Abstract
The aim of this study was to investigate the protective effects of TGF-β1 on intestinal epithelial barrier, as well as canonical Smad and MAPK signal pathways involved in these protection processes by a IPEC-J2 model stimulated with TNF-α. IPEC-J2 monolayers were treated without or with TNF-α in the absence or presence of TGF-β1. The results showed that TGF-β1 pretreatment ameliorated TNF-α-induced intestinal epithelial barrier disturbances as indicated by decrease of transepithelial electrical resistance (TER) and increase of paracellular permeability. TGF-β1 also dramatically alleviated TNF-α-induced alteration of TJ proteins ZO-1 and occludin. Moreover, TGF-β1 pretreatment increased TβRII protein expression in IPEC-J2 monolayers challenged with TNF-α. In addition, a significant increase of Smad4 and Smad7 mRNA was also observed in the TGF-β1 pretreatment after TNF-α challenge compared with the control group. Furthermore, TGF-β1 pretreatment enhanced smad2 protein activation. These results indicated that the canonical Smad signaling pathway was activated by TGF-β1 pretreatment. Finally, TGF-β1 pretreatment decreased the ratios of the phosphorylated to total JNK and p38 (p-JNK/JNK and p-p38/p38) and increased the ratio of ERK (p-ERK/ERK). Anti-TGF-β1 Abs reduced these TGF-β1 effects. These results indicated that TGF-β1 protects intestinal integrity and influences Smad and MAPK signal pathways in IPEC-J2 after TNF-α challenge.
Introduction
The intestinal barrier is important as the body's first line of defense against noxious Ags and pathogens within the intestinal lumen. 1 However, numerous factors such as early weaning and infection lead to intestinal barrier damage and dysfunction.1–3 Impairment of the intestinal barrier may facilitate attack and diffusion of any deleterious factors present within the intestinal lumen and perpetuate the inflammatory process, which is the major pathogenesis of many intestinal diseases, including infectious diarrhea or inflammatory bowel diseases.1,2 Fortunately, the intestinal epithelium reveals enormous regenerative capabilities that allow rapid healing after damage. 1 It would be of interest to investigate the nutritional regulation of the epithelial barrier restoration.
TGF-β1 is a multifunctional polypeptide growth factor, which has a central role in modulating gut mucosal cell growth, differentiation, migration and epithelial restitution after injury and immune regulation.4–7 TGF-β1 is the most abundant isoform in the mucosa of the gut and may play an important role in postnatal adaptation of the gastrointestinal tract in suckling animals.8,9 TGF-β1 also has been demonstrated to be an important modulator of the intestinal development and function in post-weaning adaptation processes in the intestine of pigs.7,9 It has been reported that TGF-β found upon repair of the epithelium after injury was related to enhancing epithelial cell migration, expression and turnover of extracellular matrix proteins and cell surface integrin.10–12 However, the mechanisms have not yet been elucidated.
So far, different signaling pathways have been reported to be involved in TGF-β action, including Smad-dependent and Smad-independent pathways.13,14 The canonical TGF-β signaling pathway is mediated by Smad-family proteins. 14 Besides the canonical Smad pathways, a number of non-Smad signaling pathways such as MAPK pathways [Erk, c-jun N-terminal kinase (JNK) and p38 MAPK pathways], phosphatidylinositol-3 kinase (PI3K)/Akt pathway or Rho/Rho-associated Kinase (ROCK) pathways also have been reported to be involved in TGF-β1 actions.13–15 We hypothesized that TGF-β1 might be involved in these barrier protection processes and lead to changes in the Smad and MAPK signaling pathways. In the present study, TNF-α, a potent pro-inflammatory cytokine, was used to build on our model of intestinal barrier injury. We employed an in vitro IPEC-J2 model to investigate the protective effects of TGF-β1 on intestinal epithelial barrier function and whether the canonical Smad and MAPK signal pathways were involved in the process.
Materials and methods
Cell culture
Porcine small intestinal epithelial cells (IPEC-J2) were used for all experiments. The IPEC-J2 cell line, a non-transformed porcine intestinal epithelial cell line, originally isolated from the jejunal epithelia of a neonatal unsuckled piglet, 16 was kindly supplied by Dr Yin Yulong's laboratory (Institute of Subtropical Agriculture, Chinese Academy of Sciences, ChangSha, China). Cells were grown in DMEM-F12 (Gibco, Gaithersburg, MD, USA), supplemented with 5% FBS (Sigma, St. Louis, MO, USA), insulin (5 µg/ml), selenium (5 ng/ml), transferrin (5 µg/ml) and epidermal growth factor (5 ng/ml). The cells were grown at 37℃ in a humidified atmosphere of 5% CO2. The culture medium was changed three times a week, according to standard culture protocols. 17
Cell preparation and treatment
IPEC-J2 cells were seeded in a collagen-coated permeable polycarbonate membrane transwell chamber (0.4 µm pore size; Corning, Somerville, MA, USA) at a density of 1.5
Measurement of epithelial barrier
In the present experiment, transepithelial electrical resistance (TER) as an indicator of tight junction (TJ) permeability to ionic solutes were measured using a Millicell-ERS epithelial voltohmmeter (Millipore Corp., Billerica, MA, USA) as described by Watari et al. 19 Each TER measurement was calculated by subtracting the resistance value of the filters and fluids. In order to facilitate comparisons between conditions, TER was normalized to the initial value. Flux of FITC-conjugated dextran (FITC-dextran, 4 kDa; Sigma) across monolayers was used to stand for the paracellular permeability of intestinal epithelial barrier to uncharged macromolecules. Briefly, monolayers were gently washed with Hank's balanced salt solution (HBSS). In the apical chamber, it was replaced by 500 µl HBSS (Invitrogen, Merelbeke, Belgium) of 1 mg/ml FD4 (FITC-dextran, 4 kDa; Sigma). Then, monolayers were incubated at 37℃ for 2 h. A 100-µl sample was taken from basal chamber and measured by a fluorescence microplate reader (FLx800; Bio-Tek Instruments, Inc., Winooski, VT, USA). FITC-dextran flux was normalized to control.
Quantitative real-time PCR
Primer sequences used for real-time PCR.

Western blot
The Western blot analysis was performed according to the procedures outlined by Watari et al. 19 Briefly, after electrophoresis the proteins were transferred to polyvinylidene difluoride membrane (Millipore, Bedford, MA, USA). The membranes were incubated at 4℃ for 12 h with primary Ab and then with the secondary Ab for 120 min at 20℃. The following primary Abs were obtained from Santa Cruz (Santa Cruz Biotechnology, Santa Cruz, CA, USA): occludin, claudin-1, ZO-1, ERK, P38, JNK, TGF-β receptorI, TGF-β receptorII, Smad2/3, β-actin, rabbit IgG. Anti-phospho-ERK, phospho-JNK 1/2, phospho-P38, phospho-Smad2 were purchased from Cell Signaling Technology (Danvers, MA, USA). The secondary Ab was HRP-conjugated anti-rabbit IgG (Santa Cruz Biotechnology). An enhanced chemiluminescence detection kit (Amersham, Arlington Heights, IL, USA) was used to detect the positive bands. β-Actin was used as an internal control, which exhibited no difference among each group. The protein expression of all samples was expressed as fold changes, calculated relative to the values from the control group.
Statistical analysis
Data were analyzed using the SAS statistical package (SAS Institute, Cary, NC, USA), with all data are representative of at least three independent experiments. Results were statistically analyzed by one-way ANOVA. Differences between the means were tested using Duncan's multiple range tests. Differences were considered significant at P < 0.05.
Results
Intestinal epithelial barrier function
Intestinal epithelial barrier function.
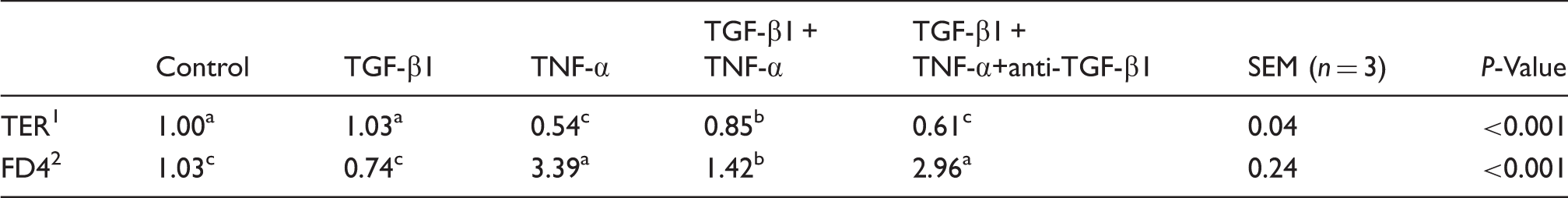
a,b,cMeans within a row with different letters differ significantly (P < 0.05).
TER: transepithelial electrical resistance (the values were normalized to control); FD4: FITC dextran (4 kDa) (the values were normalized to control).
TJ proteins expression
Figure 1 shows the effects of TGF-β1 on the expression of TJ proteins. As compared with the control, TGF-β1 alone had no effects on expression of TJ proteins ZO-1, occludin and claudin-1 in IPEC-J2 monolayers. TNF-α treatment significantly reduced (P < 0.05) the TJ proteins occludin and ZO-1 of IPEC-J2 monolayer compared with the control monolayers. However, TGF-β1 treatment significantly (P < 0.05) prevented the TJ proteins occludin and ZO-1 drop induced by TNF-α. Moreover, anti-TGF-β1 Abs blocked (P < 0.05) the beneficial protection of TGF-β1 on TJ protein ZO-1.
Effects of TGF-β1 on TJ protein levels of occludin, claudin-1 and zonula occludens-1 (ZO-1). (a) shows representative blots of occludin, claudin, ZO-1 and β-actin by different treatment, respectively. (b) Relative TJ protein expression. Values are means and SDs represented by vertical bars (n = 3). a,bMeans with different letters differ significantly (P < 0.05). The control sample was used as the reference sample. The protein expression of all samples was expressed as fold changes, calculated relative to the control group. 
 , Control;
, Control;  , TGF-β1;
, TGF-β1;  , TNF-α;
, TNF-α;  , TGF-β1 + TNF-α;
, TGF-β1 + TNF-α;  , TGF-β1 + TNF-α+siRNA.
, TGF-β1 + TNF-α+siRNA.
Canonical Smad signal pathways mRNA expression
mRNA expressions of smads signals in IPEC-J2.*
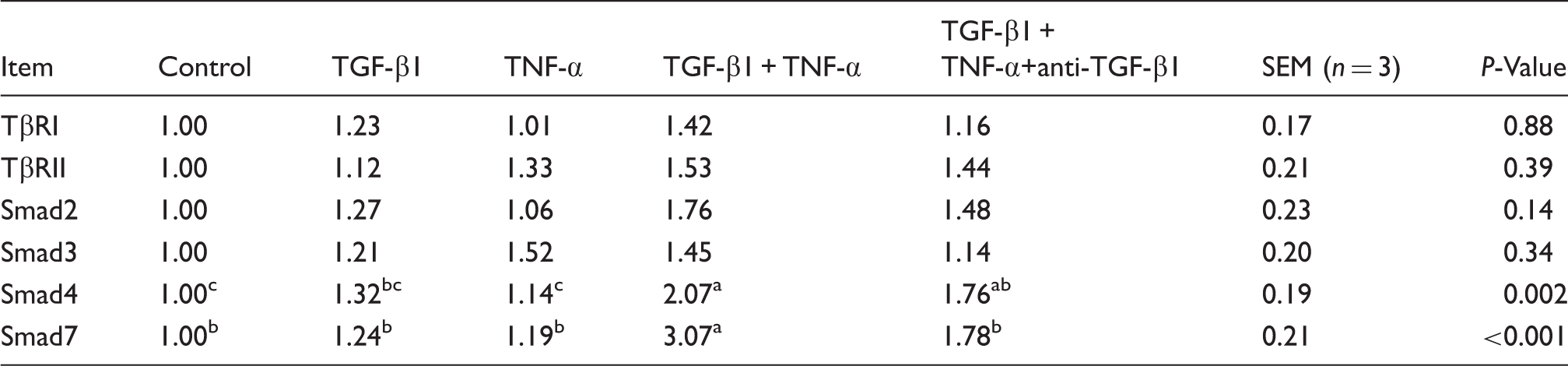
a,b,cMeans within a row with different letters differ significantly (P < 0.05). *The 2−ΔΔCt method was used to analyze the relative expression (fold changes), calculated relative to the values in samples from the control group.
Canonical Smad signal pathways protein expression
Figure 2 shows the effects of TGF-β1 on protein expression levels of TβRI and TβRII. TGF-β1 pretreatment significantly increased (P < 0.05) the protein expression levels of TβRII in IPEC-J2 monolayers after TNF-α treatment. While TGF-β1 alone and TNF-α did not have effects on protein expression levels of TβRI and TβRII. In addition, TGF-β1 pretreatment with IPEC-J2 before TNF-α challenge significantly increased (P < 0.05) the levels of phosphorylated Smad 2 compared with the control group (Figure 3). Anti-TGF-β1 Abs decreased the effects of TGF-β1 on protein expression levels of TβRII and Smad2 compared with the TGF-β1 pretreatment before TNF-α challenge group. There was no difference in the total protein levels for Smad2 among groups (P > 0.05)
Effects of TGF-β1 on protein levels of TβRI and TβRII in IPEC-J2. (a) Representative blots of TβRI and TβRII protein expression and β-actin by different treatment. (b) Relative TβRI and TβRII protein expression. a,bMean values where different letters indicate significant difference (P < 0.05). Values are means and SDs, represented by vertical bars (n = 3). The value of protein expression was the ratio of the densitometry units of TβRI, TβRII protein and β-actin. The control sample was used as the reference sample. The protein expression of all samples was expressed as fold change, calculated relative to the control group. Effects of TGF-β1 on Smad2 activation in IPEC-J2. (a) Representative blots of Smad2 by different treatments. (b) The values were calculated as the ratio of the phosphorylation level (p-Smad2) to the total level of Smad2. a,bMean values with different letters indicate statistical significance (P < 0.05). Values are means and SDs, represented by vertical bars (n = 3). The control sample was used as the reference sample. The protein expression of all samples was expressed as fold change, calculated relative to the control group. Data are means ± SD (n = 3). 
 , Control;
, Control;  ,TGF-β1;
,TGF-β1;  , TNF-α;
, TNF-α;  , TGF-β1 + TNF-α;
, TGF-β1 + TNF-α;  , TGF-β1 + TNF-α+anti-TGF-β1.
, TGF-β1 + TNF-α+anti-TGF-β1.
 , Control;
, Control;  , TGF-β1;
, TGF-β1;  , TNF-α;
, TNF-α;  , TGF-β1 + TNF-α;
, TGF-β1 + TNF-α;  , TGF-β1 + TNF-α+anti-TGF-β1.
, TGF-β1 + TNF-α+anti-TGF-β1.
MAPK signal pathways
Figure 4 shows effects of TGF-β1 on the three MAPK signaling pathways (JNK, p38, ERK) and phosphorylated forms of MAPKs (p-JNK, p-p38, p-ERK). There was no difference in the total protein levels for the JNK, p38 and ERK pathways among the groups (P > 0.05). However, compared with the control, TGF-β1 pre-treatment before TNF-α stimulation significantly increased (P < 0.05) the ratio of the phosphorylated forms and the total levels of ERK (p-ERK/ERK), and significantly decreased (P < 0.05) the ratio of phosphorylated forms and the total levels of JNK and p38 (p-JNK/JNK and p-p38/p38). TNF-α treatment significantly increased (P < 0.05) the ratio of phosphorylated forms and the total levels of JNK, p38 and ERK (p-JNK/JNK, p-p38/p38 and p-ERK/ERK). TGF-β1 alone did not have effects on phosphorylated forms of MAPKs (p-JNK, p-p38, p-ERK) compared with the control. Anti-TGF-β1 Abs attenuated (P < 0.05) the effects of TGF-β1 on promoting phosphorylated p-ERK expression, and inhibited phosphorylated JNK and p38 expression.
Effects of TGF-β1 on activation of MAPK signaling pathways in IPEC-J2. The three MAPKs are JNK, p38 MAPK and extracellular regulated kinases (ERK1/2). (a) Representative blots of MAPKs and phosphorylated MAPK protein expressions by different treatment. (b) The values were calculated as the ratios of their phosphorylation levels (p-JNK, p-p38, p-ERK) to the total levels of MAPK. The protein expression of all samples was expressed as fold change, calculated relative to the values from control group. The control sample was used as the reference sample. Data are means ± SD (n = 3). a,bMean values with different letters indicate statistical significance (P < 0.05). 
 , Control;
, Control;  , TGF-β1;
, TGF-β1;  , TNF-α;
, TNF-α;  , TGF-β1+TNF-α;
, TGF-β1+TNF-α;  , TGF-β1 + TNF-α+anti-TGF-β1.
, TGF-β1 + TNF-α+anti-TGF-β1.
Discussion
Intestinal epithelia serve multiple physiological roles, including the provision of a defensive barrier to protect against potentially harmful luminal contents. Substantial evidence has shown that the pro-inflammatory cytokine TNF-α is implicated in intestinal barrier dysfunction in various cell types, including intestinal epithelial cells.21–23 In agreement with earlier reports, the present study demonstrated that TNF-α induced alteration of intestinal barrier function in IPEC-J2, as indicated by decreased TER and increased paracellular permeability. Intestinal epithelium integrity is provided by the presence of TJ located in the apical region of intestinal epithelial cells. Previous studies showed that TNF-α induced redistribution of several TJ such as adherens junction proteins, including zonula occludens-1, occludins, or claudins.23,24 In the current study, we found that TNF-α induced reductions in occludin and ZO-1. Thus, we took advantage of the model for inducing intestinal epithelial barrier disruption by TNF-α, to investigate whether TGF-β1 could protect the barrier function.
TGF-β1 is a multifunctional cytokine with immunosuppressive properties produced by many cell types, including intestinal epithelia. TGF-β1 has been reported to play an important regulatory role in the restoration of intestinal epithelial barrier in vivo.9,11,25,26 During injury or disease, it can stimulate cell proliferation and extracellular matrix production and thereby heal or repair the injured tissue.6,25 In this study, we found that TGF-β1 prevented epithelial barrier dysfunction caused by TNF-α in IPEC-J2, as evidenced by TGF-β1 alleviating both TER decrease and paracellular permeability increase. Moreover, consistent with the improved intestinal barrier function, we observed that pretreatment with TGF-β1 prevented the TNF-α-induced reductions in occludin and ZO-1, which are thought of as the major integral membrane proteins in the barrier function. Reduced levels of claudin-1, ZO-1 and occludin accompanied the loss of barrier function. In the current experiment, TGF-β1 prevented the alteration of TJ proteins ZO-1 and occludin in IPEC-J2 intestinal epithelial monolayers exposed to TNF-α. These results indicated that TGF-β1 could ameliorate the disruption of intestinal barrier function induced by the pro-inflammatory cytokine TNF-α in IPEC-J2. Similarly, Al-Sadi et al. reported that TGF-β1 possesses barrier-protective effects on the intestinal epithelial barrier function as by prevented TER decrease and paracellular permeability increase in Caco-2 cells. 27 Howe et al. also reported that TGF-β enhanced barrier function by regulation of epithelial TJ proteins in T84 cells. 11
To demonstrate the specific role of TGF-β1 in the barrier restoration, anti-TGF-β1 Abs were used in the present study. We found that anti-TGF-β1 Abs could attenuate the actions of TGF-β1. Anti-TGF-β1 Abs blocked the protective effects of TGF-β1 on the barrier functions as indicated by TER, paracellular permeability and TJ proteins, supporting the specific role of TGF-β1 here, because the effect of TGF-β1 on barrier properties has been connected to its anti-inflammatory properties. 28 At least in our IPEC-J2 cell model, the blocking effect of TGF-β1 Abs underlines the predominant role of TGF-β1 on barrier function. Earlier findings have shown that TGF-β enhances the epithelial barrier. For example, Howe et al. demonstrated that TGF-β1 preserved epithelial barrier function after enterohemorrhagic Escherichia coli infection. 11 Hering et al. reported that TGF-β1 present in whey protein concentrate showed protective effects on IFN-γ-induced barrier disturbance in HT-29/B6 cells by attenuating the decrease in TER and increase in permeability of the paracellular marker fluorescein. 29 TGF-β also showed a striking ability to reduce the capacity of IFN-γ and Cryptosporidium parvum to diminish epithelial function.30,31 In the current study, TGF-β1 exerted beneficial effects on prevention of intestinal epithelial barrier dysfunction induced by TNF-α in IPEC-J2.
To elucidate the molecular mechanisms of TGF-β1 exerting beneficial effects on IPEC-J2 after TNF-α challenge, we examined the TGF-β1 signaling pathways. The canonical TGF-β signaling pathway is mediated by Smad family proteins. When TGF-β reaches the membrane of target cells, they bind directly to TGF-β type II receptors (TβRII), which leads to the recruitment of TGF-β type I receptors (TβRI). TβRII then trans-phosphorylates TβRI, enabling the TβRI kinase domain to act on cytoplasmic proteins and thereby propels downstream signaling actions. Following stimulation by TGF-β, Smad2 and Smad3 become phosphorylated Smad2/3, which can complex with Smad4, and then translocate to the nucleus and regulate gene expression. 32 The key factors for regulating signaling component stability and balancing the incoming TGF-β signal are the inhibitory (I-) Smads (Smad7). 33 In the present study, we evaluated the canonical downstream substrate of the TGF-β signal. We observed that TGF-β1 promoted the protein expression of TβRII in IPEC-J2 monolayers exposed to TNF-α, compared with the control group. Consistent with elevated TβRII, there was an increase of mRNA expression of Smad4 and Smad7 during TGF-β1 treatment. We also found that phosphorylation of smad2 was increased during TGF-β1 pretreatment. Consistent with the increased TβRII protein expression, this may suggest that the canonical Smad pathways were activated by TGF-β1 treatment after TNF-α challenge. In line with our study, a recent study has revealed that oral administration of TGF-β1 protects the immature gut from injury via Smad protein-dependent signaling pathways. 34 It was also reported that TGF-β enhancement of the epithelial barrier function was a Smad2/3-dependent event. 11 Thus, we speculated that TGF-β1 prevented intestinal epithelial barrier disruption by TNF-α in IPEC-J2, partly through TGF-β1 canonical Smad pathways, directly or indirectly.
Apart from canonical Smad pathways, a number of non-canonical signaling pathways, including MAPK pathways, are involved in TGF-β1 actions. 35 The MAPK signaling pathways transduce signals from a diverse array of extracellular stimuli and form a highly integrated network to regulate cellular functions such as differentiation, proliferation and cell death.36,37 In the present study we demonstrated, for the first time, that TGF-β1 pretreatment significantly increased phosphorylated ERK 1/2, while decreasing the phosphorylated p38 and JNK, suggesting that MAPK signaling pathways were involved in intestinal barrier protection when exposed to TNF-α. Anti-TGF-β Abs reduced the effects of TGF-β1 on phosphorylated ERK 1/2 and phosphorylated p38 and JNK. TNF-α has been reported to increase the relative protein levels for phosphorylated p38, JNK and ERK 1/2 in many cell types.38–41 Using a porcine model of intestinal ischemia, Shifflett et al. demonstrated that inhibition of JNK resulted in enhanced recovery of barrier function, whereas inhibition of ERK1/2 and p38 MAPK had a deleterious effect on mucosal recovery from ischemia. 37 The JNK and p38 cascades are activated by inflammatory cytokines and a wide variety of cellular stresses. 42 The inhibition of TGF-β1 on p38 and JNK signaling pathways might be caused by the anti-inflammatory properties against TNF-α by counterbalancing its barrier disruption effects. The ERK 1/2 cascade is activated by growth factors, and preferentially regulates cell growth and differentiation. 42 In fact, there is evidence that the activation of the MAPK signaling is linked to the TGF-β1 induced modulation of TJ permeability and wound closure.43,44 Agreement with our study, TGF-β1 increased wound closures via ERK MAPK activation in cultured rat intestinal epithelial cells (IEC-6). 45 TGF-β1 also stimulated re-epithelialization in human corneal epithelial cells through the Ras/MEK/ERK signaling pathway. 46 Additionally, TGF-β1 has been shown to stimulate HepG2 cell proliferation and invasion by phosphorylated MAPKs (pJNK1/2, pp38 and pERK1/2). 47 Also, TGF-β-induced activation of Ras/Erk MAPK signaling can induce TGF-β1 expression, thereby amplifying the TGF-β response and inducing secondary TGF-β responses. It is possible that the barrier protection properties against TNF-α of TGF-β1 in IPEC-J2 are related to activation of MAPKs. It has been reported that the MAPK pathways cross-talk with Smad signaling by regulating Smad phosphorylation at the linker region to increase nuclear translocation of p-Smad2/3 and also to enhance canonical TGF-β signaling. 48 However, whether these pathways regulate epithelial barrier independently or via intracellular cross-talk in this study remains to be determined in future.
In summary, the present study indicates that TGF-β1 protects intestinal integrity and influences Smad and MAPK signal pathways in IPEC-J2 after TNF-α challenge. This identifies a mechanism of the curative effect of TGF-β1 that will enable us to understand the intestinal integrity and to develop a new strategy to treat intestinal mucosal injuries.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was jointly supported by National Natural Science Foundation of China (31472103), the Special Fund for Agroscientific Research in the Public Interest (201403047), Science Technology Department of Zhejiang Province (Project 2015C02022).
