Abstract
Objective
To investigate adipocytokine expression levels, platelet-to-lymphocyte ratio (PLR) and transforming growth factor (TGF)-β1/Smad signaling activity in diabetic patients with pulmonary infection.
Methods
Eighty-two type 2 diabetic patients with pulmonary infection were included in the observation group and 75 patients with simple type 2 diabetes were recruited into the control group. The fasting blood glucose (FBG), glycated hemoglobin (HbA1c), and PLR in the two groups were compared. Complement-C1q/tumor necrosis factor related protein 3 (CTRP-3), complement-C1q/tumor necrosis factor related protein 9 (CTRP-9), leptin, adiponectin, and TGF-β1/Smad signaling pathway activity in peripheral blood mononuclear cells (PBMCs) was detected.
Results
Compared with patients in the control group, patients in the observation group presented with increased levels of FGB, HbA1c, and leptin, an increase in the PLR, and decreased serum CTRP-3, CTRP-9, and adiponectin levels. TGF-β1, p-Smad2, and p-Smad3 protein expression levels were up-regulated in PBMCs from patients in the observation group compared with the control group.
Conclusions
These results show that in type 2 diabetic patients with pulmonary infection, adipocytokine expression is altered, PLR is disturbed, and the TGF-β1/Smad signaling pathways in PBMCs are significantly activated.
Keywords
Introduction
Diabetes is a multifactorial chronic disease with a metabolic disorder that may lead to disturbances in carbohydrate, lipid, and protein metabolism. 1 Diabetes compromises the immune function of patients, and the patients’ condition can be further complicated by a variety of underlying diseases. Compared with the normal population, diabetics are at higher risk of developing pulmonary infection, which will further exacerbate diabetic conditions and result in a poor prognosis.2–4 Treating the infection at an early stage is the main therapeutic strategy for diabetic patients with pulmonary infection, but it often shows unsatisfactory outcomes. 5
Adipocytokines represent a class of bioactive cytokines that were secreted by adipose tissue, including leptin and adiponectin, which are involved in regulating inflammation, oxidative stress, vascular endothelial function, and other physiological functions. Metabolic disturbances in adipocytokines are a main pathological characteristic of diabetes. 6 Previous studies have shown that diabetic patients often present with a disturbed platelet-to-lymphocyte (PLR) ratio and abnormal TGF-β1/Smad signaling pathway activation, which may play important pathological roles in diabetes and relative complications.7,8 In addition, whether pneumonia aggravates the above pathological manifestations is unclear.
Therefore, this article aims to investigate adipocytokines, the PLR, and protein expression in the TGF-β1/Smad signaling pathway in diabetic patients with pulmonary infection.
Materials and methods
General data
Type 2 diabetic patients with pulmonary infection and patients with simple type 2 diabetes who were admitted to The People’s Hospital of Jianyang City from May 2016 to October 2018 were enrolled into the observation group and the control group, respectively. General data on the two groups of patients were collected and compared, including age, gender, body mass index, disease course, alanine transaminase, aspartate transaminase, hypertension, hyperlipidemia, treatment (hypoglycemic drugs, insulin), and complications (microangiopathy, macroangiopathy, neuropathy). All patients provided written informed consent and this study was approved by the Ethics Committee at The People’s Hospital of Jianyang City (NO. JY17100012).
Inclusion and exclusion criteria
The inclusion criteria were as follows: 1) Patients with type 2 diabetes who met the criteria of the Standards of Medical Care in Diabetes guidelines that were issued by American Diabetes Association in 2012 9 , including the following: fasting blood glucose (FBG) level ≥7.0 mmol/L, 2-hour ≥11.1 mmol/L on the oral glucose tolerance test (75 g), or patients with typical hyperglycemic symptoms and a random blood sugar level ≥11.1 mmol/L; 2) patients with pulmonary infection that was diagnosed based on the criteria established by Chinese Thoracic Society of the Chinese Medical Association 10 , as follows: patients with typical symptoms or signs such as cough, expectoration, and fever, infiltrative shadows on chest X-ray, and pathogenic bacteria that is isolated from sputum culture; and 3) patients who did not receive anti-infection treatment within 2 weeks before the study.
The exclusion criteria were as follows: 1) Patients with infections in the abdominal cavity, urinary tract, skin, and other parts of the body; 2) patients with diseases such as liver and renal dysfunction, malignant tumor, pulmonary tuberculosis, myocardial infarction, blood disease, or diseases with changes in platelet or lymphocyte levels.
Identification of pathogenic bacteria
After admission, all patients underwent culture procedures to identify pathogenic bacteria. Briefly, after waking in the morning, the patients gargled with water twice, then strongly coughed twice. The first coughed-out sputum was spit out and discarded, and the second sputum was collected for culture and identification of pathogenic bacteria using the VITEK®-60 Fully Automated Microbiology System (Biomerieux, Inc., Marcy l’Etoile, France).
Determination of glucose and lipid metabolism
After admission and before initiating treatment, serum FBG, glycated hemoglobin (HbA1c), triglyceride (TG), total cholesterol, high density lipoprotein (HDL), and low-density lipoprotein (LDL) levels in the patients were measured using a HITACHI 7180 biochemistry automatic analyzer (Hitachi, Tokyo, Japan).
Determination of adipocytokine levels
After admission and before initiating treatment, 2 mL of fasting venous blood was taken from the patients and incubated at room temperature for 20 minutes. Samples were then centrifuged at 8,000 × g for 10 minutes. Serum in the upper layer was retained for analysis. Serum levels of complement C1q/tumor necrosis factor related protein 3 (CTRP-3), complement C1q/tumor necrosis factor related protein 9 (CTRP-9), leptin, and adiponectin were determined using an enzyme-linked immunosorbent assay (ELISA) kit (CUSABIO Biotech Co., Ltd., Wuhan, China).
Determination of the platelet-to-lymphocyte ratio
After admission and before initiating treatment, 2 mL of fasting venous blood was collected from the patient in a vacuum tube with heparin as an anticoagulant for routine blood examination using a DxH800 hematology analyzer (Beckman Coulter Inc., Miami, FL, USA.).
Separation of peripheral blood mononuclear cells (PBMCs)
After admission and before initiating treatment, 3 mL of fasting venous blood was collected from the patient in a vacuum tube with heparin, and 3 mL of PBS was added to dilute the blood sample. Then, 6 mL of human whole blood mononuclear cell separation medium (Beijing Solarbio Science & Technology Co., Ltd., Beijing, China) was added into a new and empty centrifuge tube, and the diluted whole blood was slowly added above the surface of the separation medium. After centrifugation at 500 × g for 30 minutes, the solution was stratified into the following four discernible layers: the top layer was plasma, followed by a PBMC layer with albuginea-like appearance, then a transparent layer of separation medium, and erythrocytes and granulocytes were deposited in the bottom layer. The PBMCs were then carefully extracted and transferred into a new centrifuge tube, washed with PBS, and centrifuged at 250 × g for 10 minutes. Finally, the PBMCs were obtained.
Western blotting
PBMCs were first treated with 100 μL of RIPA lysate (Beyotime Biotechnology, Jiangsu, China) and centrifuged at 8,000 × g for 5 minutes. Then, the protein was extracted from the supernatant and quantified using the BCA method. The concentration of the total protein sample was adjusted using PBS and then 40 μg of protein sample was mixed with loading buffer and boiled in water for 5 minutes to achieve complete denaturation. Subsequently, the sample was separated using polyacrylamide gel electrophoresis and transferred on to PVDF membranes. The PVDF membranes were then blocked with 5% bovine serum albumin at room temperature for 2 hours and primary antibodies including anti-TGF-β1 antibody (Abcam Plc, Cambridge, UK; 1:1,000), anti-Smad2 antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA, 1:500), anti-p-Smad3 antibody (Santa Cruz Biotechnology; 1:500), anti-Smad3 antibody (Santa Cruz Biotechnology; 1:500), and anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) antibody (Abcam Plc; 1:3,000) were added onto the membrane and incubated overnight at 4°C. After washing the membrane three times with TBST, the secondary antibody (Boster Biological Technology Co., Ltd, Wuhan, China; 1:5,000) was added onto the membrane and incubated at room temperature for 1 hour. The membranes were then washed three times and ECL was added as a developing solution (Beyotime Biotechnology). Images were acquired using a Gel Doc XR + gel imaging system (Bio-Rad, Hercules, CA, USA), and the gray value of each band was measured using Image Pro Plus 6.0 (Media Cybernetics, Inc., Rockville, MD, USA). TGF-β1/GAPDH, p-Smad2/Smad2, and p-Smad3/Smad3 in the control group were set as equal to 1.
Statistical analysis
SPSS Statistics for Windows, version 20.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. All measurement data had a normal distribution and were expressed as the mean ± standard deviation. Independent sample t-tests were used to compare the measurement data between the two groups. All enumeration data were expressed as the number of patients, and the χ2 test was used to compare the enumeration data between the two groups. P < 0.05 was considered to be a statistically significant difference.
Ethics
This study was approved by the Ethics Committee of The People’s Hospital of Jianyang City (No. JY17100012).
Results
Comparison of patients’ general data between the two groups
Eighty-two type 2 diabetic patients with pulmonary infection and 75 patients with simple type 2 diabetes were enrolled into the observation group and the control group, respectively. There were no statistically significant differences in age, gender, body mass index, alanine transaminase, aspartate transaminase, hypertension, and hyperlipidemia, and in the percentage of neuropathy occurrence between the two groups of patients. However, compared with patients in the control group, patients in the observation group presented with a longer disease course, a higher percentage of insulin use, microangiopathy occurrence, and macrovascular disease occurrence, and a lower percentage of hypoglycemic drug use (P < 0.05, Table 1).
General data.
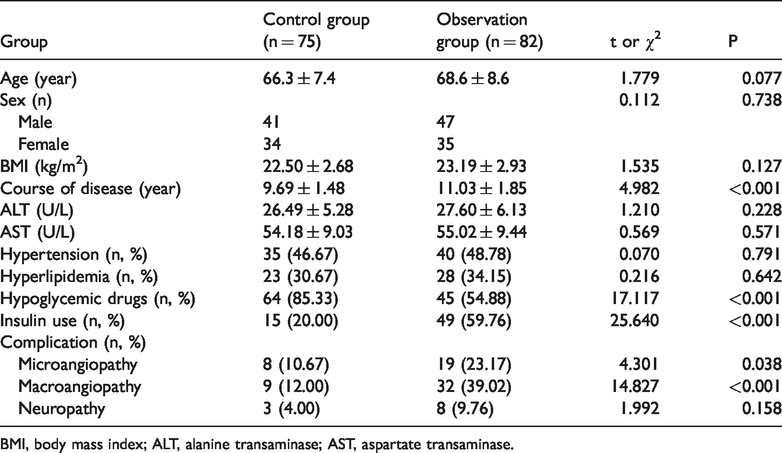
BMI, body mass index; ALT, alanine transaminase; AST, aspartate transaminase.
Identification of pathogenic bacteria
No pathogenic bacteria were isolated from 75 patients in the control group, while 91 strains of pathogenic bacteria were isolated from the 82 patients in the observation group. Among these patients, 73 had a single pathogen infection and nine had a mixed infection of two pathogens. Among the pathogenic bacteria, there were 57 strains of Gram-negative bacteria that accounted for 62.64% of the bacteria. These bacteria mainly included 19 strains of Klebsiella pneumoniae, 15 strains of Pseudomonas aeruginosa, and 12 strains of Acinetobacter baumannii, which accounted for 20.88%, 16.48%, and 13.19%, respectively. There were 27 strains of Gram-positive bacteria that accounted for 29.67%, which mainly included 13 strains of Staphylococcus aureus and eight strains of Staphylococcus epidermidis, accounting for 14.29% and 8.79%, respectively. There were seven strains of fungi that accounted for 7.69%. This mainly included five strains of Candida albicans, which accounted for 5.49%.
Comparison of glucose and lipid metabolism parameters between the two groups
Compared with the control group, the patients in the observation group presented with increased levels of FBG and HbA1c (P < 0.05). There were no statistically significant differences in TG, TC, HDL, and LDL levels between the two groups (Table 2).
Glucose and lipid metabolism.

FBG, fasting blood-glucose; HbA1c, glycated hemoglobin; TG, triglyceride; TC, total cholesterol; HDL, high density lipoprotein; LDL, low-density lipoprotein.
Comparison of serum adipocytokine levels between the two groups
The patients in the observation group had decreased serum CTRP-3, CTRP-9, and adiponectin levels and increased leptin levels compared with the control group (P < 0.05, Table 3).
Serum adipocytokine levels.
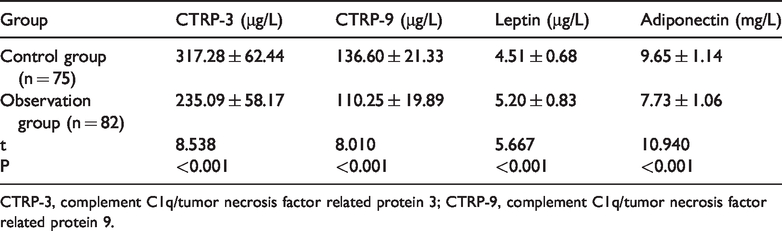
CTRP-3, complement C1q/tumor necrosis factor related protein 3; CTRP-9, complement C1q/tumor necrosis factor related protein 9.
Comparison of PLR between the two groups of patients
The PLRs of patients in the observation group were higher compared with patients in the control group (P < 0.05, Table 4).
PLRs in two groups of patients.

PLR, platelet-to-lymphocyte ratio.
Comparison of TGF-β/Smad signaling pathway protein expression in PBMCs between the two groups
Compared with patients in the control group, TGF-β, p-Smad2, and p-Smad3 protein expression levels in PBMCs from patients in the observation group were up-regulated (P < 0.05, Figure 1 and Table 5).

Expression of TGF-β1/Smad signaling pathway proteins in PBMCs.
Expression levels of TGF-β/Smad signaling pathway proteins in PBMCs.

TGF, transforming growth factor; PBMCs, peripheral blood mononuclear cells; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Discussion
Pulmonary infection is a main cause of increased medical expenses and decreased quality of life in type 2 diabetic patients. This can also greatly increase the incidence of metabolic complications such as ketoacidosis and hypoglycemia, which results in a poor prognosis. 11 Pulmonary infections and therapeutic antibiotics may also lead to insulin resistance, which would increase the complexity of blood glucose control. Moreover, the sensitivity of diabetic patients to antibiotics is poor, which may be caused by reduced antibiotic levels at the infection site as a consequence of diabetic vasculopathy.12–14 Therefore, further exploration of pathological changes in diabetic patients with pulmonary infection to reveal key mechanisms and discover therapeutic targets has significant value in facilitating better treatment implementation.
Risk factors for pulmonary infection in patients with type 2 diabetes include aging, a long disease course, high fasting blood glucose (FBG) levels, invasive procedures, and complications such as organ failure, chronic bronchitis, pulmonary tuberculosis, and emphysema.15–17 In this article, comparison of general data between the two groups revealed that the higher blood sugar level in diabetic patients with pulmonary infection may be a main cause of pulmonary infection as well as a manifestation of concomitant disease exacerbation. We also found that diabetic patients with pulmonary infection were characterized by a long disease course, more underlying diseases and complications, and the dominant treatment strategy was insulin therapy. However, this article reported no statistically significant differences in age or other relative parameters between the two groups. The inconsistency may be associated with insufficient inclusion of cases, subject grouping, and the inclusion criteria.
Our study noted changes in serum levels for a variety of adipocytokines in diabetic patients, and these adipocytokines are involved in the mechanism of insulin resistance in diabetes. 18 CTRP-3 and CTRP-9 are members of the Clq/TNF-related protein family, which plays a role in blood sugar level reduction, vascular protection, and inflammation regulation, and their serum levels in patients with type 2 diabetes were significantly decreased. 19 We also found a decrease in CTRP-3 and CTRP-9 levels in diabetic patients with pulmonary infection. This may be used to judge the severity and prognosis of diabetes mellitus in the future, and to develop targeted drugs to promote the synthesis and secretion of CTRP-3 and CTRP-9 in adipose tissue, which has potential application prospects.
Leptin and adiponectin are hormone-like substances that are mainly secreted by adipose tissue, and they play an important regulatory role in appetite control and anti-atherosclerosis intervention. 6 In this article, patients from the observation group presented with higher leptin levels and lower adiponectin levels compared with controls.
PLR reveals the inflammatory state of diabetic patients, and it is associated with many complications such as retinopathy and diabetic nephropathy. 8 In this article, our results showed that the PLR of patients in the observation group was significantly higher compared with the control group. Increased PLRs have been reported in patients with infections at different sites and infections with different kinds of pathogens such as pulmonary infections that are caused by Mycoplasma pneumoniae, Mycobacterium tuberculosis, and other pathogens. 20 Therefore, we hypothesizes that high PLR in patents with pulmonary infection is an universal phenomenon that is not limited to diabetic patients, but it may be associated with the universal hyper-inflammatory state in pulmonary infection. PLR can be simply detected by blood routine examination, which promotes its potential use in monitoring the condition of diabetic patients with pulmonary infection in the future.
The TGF-β1/Smad signaling pathway plays a critical role in diabetes-associated pathological processes in the peripheral nerves, kidney, heart, skin, and other parts of the body, and it is involved in regulation of various pathological processes, such as fibrosis proliferation, ulcer formation, inflammatory factor secretion, and response of oxidative stress.7,21 In this article, we used PBMCs from patients to compare the expression levels of TGF-β1/Smad signaling pathway proteins between the two groups. The results indicate that TGF-β1 expression levels in the observation group were significantly higher compared with the control group. Moreover, phosphorylation levels of its downstream effector molecules, Smad2 and Smad3, were significantly increased. This indicates significant activation of the TGF-β1/Smad signaling pathway in diabetic patients with pulmonary infection, which may accelerate the development of pathological processes such as renal fibrosis.
There are some limitations in this article. First, in this paper, the types and proportions of pathogenic bacteria in pulmonary infection were assessed using general statistics and descriptions, which can provide some epidemiological basis for clinical treatment. However, this paper does not discuss the differences in the pathophysiological effects of different kinds of pathogenic bacteria on diabetes. The effects of different pathogenic bacteria on diabetes may be very different. In the future, studies in this area need to be further refined. Second, although PBMCs are used frequently because of their adequate numbers and because they are easy to obtain, they cannot provide a complete picture of pathological characteristics in diabetic patients with pulmonary infection. Therefore, the conclusion needs to be verified by analyzing lung tissue and other samples in future experiments, and we will further explore differences in presence of complications, survival rate, and other long-term prognostic indicators in diabetic patients with pulmonary infection.
In conclusion, in type 2 diabetic patients with pulmonary infection, adipocytokine levels and the PLR are increased and TGF-β1/Smad signaling pathways in PBMCs are significantly activated compared with controls.
