Abstract
The study examined the effect of chromium supplementation on the response of steers to an LPS challenge. Steers received a premix that added 0 (control; n = 10) or 0.2 mg/kg of chromium (n = 10) to the total diet on a dry matter basis for 56 d. Steers were fitted with jugular catheters and rectal temperature (RT) recording devices on d 52. Blood samples were collected and sickness behavior scores assigned to each steer relative to an LPS challenge (0.5 µg/kg) on d 55. Pre-LPS RT were greater in chromium-supplemented than in control steers. Post-LPS RT increased in both treatments, with control steers producing a greater change in RT than chromium-supplemented steers. Sickness behavior scores were greater in control than in chromium-supplemented steers post-LPS (P = 0.03). Cortisol concentrations did not differ between treatments pre-LPS. Post-LPS cortisol concentrations increased but did not differ due to treatment. Concentrations of IL-4 increased post-LPS but were not affected by treatment pre- or post-LPS. Treatment did not affect pre-LPS TNF-α or IFN-γ. Post-LPS TNF-α and IFN-γ increased in both treatments, with chromium-supplemented steers producing greater TNF-α (P = 0.005) and IFN-γ (P = 0.004) than control steers. Pre-LPS IL-6 was greater (P = 0.027) in chromium-supplemented steers than in control steers. Post-LPS IL-6 increased in both treatments and was greater (P < 0.001) in chromium-supplemented than in control steers. These data suggest that chromium supplementation enhances the acute phase response of steers to an LPS challenge, which may expedite recovery.
Introduction
The receiving period at the feedlot is a very stressful time for cattle. Increased handling and commingling of calves with unfamiliar cattle increases stress hormone concentrations. 1 Stress hormones, such as cortisol, are known to negatively affect immune function. Additionally, commingling between unfamiliar cattle increases the risk of pathogen transfer between cattle.1,2 Therefore, the high rate of morbidity, mainly related to bovine respiratory disease, found in receiving cattle is not unexpected. 2 High incidences of morbidity lead to increased cost to the producer due to a decrease in feed intake, an increase in the number of days on feed, and the addition of the cost of antibiotics used to treat sick cattle. 2 This has led to an increase in the need for methodologies that enhance the immune system of receiving cattle.
The potential use of immunomodulators, or substances that can be administered to enhance immune function and overall growth, is a growing area of research. One immunomodulator that has received attention as a potential supplement to enhance immune function is chromium. While originally thought to be part of the glucose tolerance factor, which is required for normal metabolism and maintenance of normal sugar concentrations in the blood,3,4 chromium is now described to be a component of chromodulin. 5 Chromodulin is a low molecular mass oligopeptide that binds chromium and is believed to modulate the actions of insulin through binding to and activating signal transduction, specifically tyrosine kinase activity, when insulin is bound to the insulin receptor.5,6 Additionally, chromium is an essential nutrient in cattle diets and may be deficient in stressed cattle. 3 During stress, glucose metabolism increases, thus increasing chromium utilization and leading to chromium deficiency.3,7 Indeed, stress has been demonstrated to increase the urinary secretion of chromium in humans8,9 and Burton et al. 10 suggested that chromium may reduce immunosuppression in stressed dairy cows. Regarding immune function, Chang et al. 11 demonstrated chromium supplementation increased the in vitro blastogenic response of leukocytes isolated from Charolais-cross calves in the presence or absence of a mitogen. Faldyna et al. 12 also observed a greater antibody (IgG2) response to tetanus toxoid vaccination in late-pregnant dairy cows that were supplemented with chromium. Although there is literature that details the effect of chromium on the adaptive immune response, literature on the effect of chromium on the innate and pro-inflammatory response is insufficient. Therefore, the objective of this study was to examine the effect of chromium, supplemented as chromium propionate, on the acute phase response of receiving steers to an LPS challenge.
Materials and methods
All experimental procedures were in compliance with the Guide for the Care and Use of Agricultural Animals in Research and Teaching and approved by the Institutional Animal Care and Use Committee at Texas Tech University (Approval number 10045-08).
Experimental design
Diet composition.
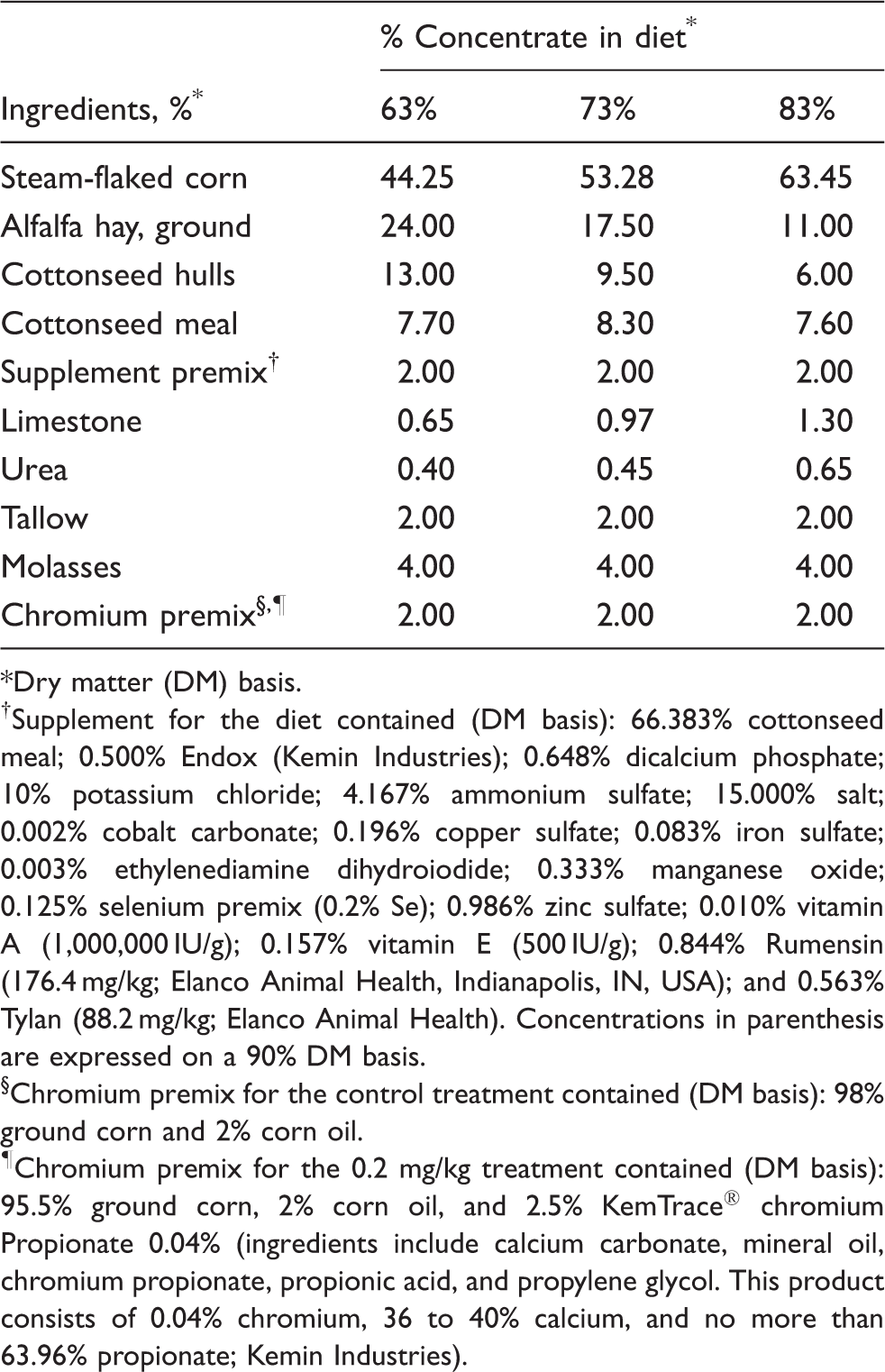
Dry matter (DM) basis.
Supplement for the diet contained (DM basis): 66.383% cottonseed meal; 0.500% Endox (Kemin Industries); 0.648% dicalcium phosphate; 10% potassium chloride; 4.167% ammonium sulfate; 15.000% salt; 0.002% cobalt carbonate; 0.196% copper sulfate; 0.083% iron sulfate; 0.003% ethylenediamine dihydroiodide; 0.333% manganese oxide; 0.125% selenium premix (0.2% Se); 0.986% zinc sulfate; 0.010% vitamin A (1,000,000 IU/g); 0.157% vitamin E (500 IU/g); 0.844% Rumensin (176.4 mg/kg; Elanco Animal Health, Indianapolis, IN, USA); and 0.563% Tylan (88.2 mg/kg; Elanco Animal Health). Concentrations in parenthesis are expressed on a 90% DM basis.
Chromium premix for the control treatment contained (DM basis): 98% ground corn and 2% corn oil.
Chromium premix for the 0.2 mg/kg treatment contained (DM basis): 95.5% ground corn, 2% corn oil, and 2.5% KemTrace® chromium Propionate 0.04% (ingredients include calcium carbonate, mineral oil, chromium propionate, propionic acid, and propylene glycol. This product consists of 0.04% chromium, 36 to 40% calcium, and no more than 63.96% propionate; Kemin Industries).
Sickness behavior
Sickness score definitions of visual signs of sickness.

Assays for cortisol and cytokines
All serum samples were analyzed in duplicate. Serum cortisol concentrations were determined using a commercially-available enzyme immunoassay according to the manufacturer’s directions (Arbor Assays, Ann Arbor, MI, USA) by comparison of unknowns to standard curves generated with known concentrations of cortisol. The minimum detectable cortisol concentration was 45.4 pg/ml and the intra- and inter-assay coefficients of variation were 3% and 6%, respectively. Data are presented as ng/ml.
Serum cytokine concentrations (IL-4, TNF-α, IFN-γ, and IL-6) were determined by a custom bovine 4-plex sandwich-based chemiluminescence ELISA kit (Searchlight-Aushon BioSystems, Inc., Billerica, MA, USA) as previously described.15,16 The minimum detectable concentrations were 2.6, 0.5, 0.1 and 3.3 pg/ml for IL-4, TNF-α, IFN-γ, and IL-6, respectively. All intra-assay coefficients of variation were less than 9% and all inter-assay coefficients of variation were less than 21% for all assays.
Statistical analysis
Prior to analysis, RT data were averaged into 30 min intervals. Data for RT, sickness behavior scores, cortisol, and cytokines were analyzed using the MIXED procedure of SAS (SAS, Inc., Cary, NC, USA) specific for repeated measures with treatment, time, and time × treatment interaction included as fixed effects. Specific treatment comparisons were made using Fisher’s Protected LSD with P < 0.05 considered significant. Data are presented as the least squares means ± the standard error of the mean.
Results
Rectal temperature
Prior to administration of LPS, RT were greater in chromium-supplemented (38.91 ± 0.02°C) steers than control steers (38.77 ± 0.02°C; P = 0.002; Figure 1). Following administration of LPS, RT increased in both treatments (P < 0.001), with peak RT occurring between 120 and 240 min (Figure 1). The change in RT from baseline values was greater in control steers (0.398 ± 0.009°C; P < 0.001) than steers supplemented with chromium (0.0255 ± 0.009°C). Steer RT returned to temperatures not different than baseline by 470 min for chromium-supplemented steers (P = 0.074) and by 790 min for control steers (P = 0.062). Additionally, RT decreased below baseline values (P = 0.042) for chromium-supplemented steers from 1250 min until the end of the study, a response that was not observed in control steers.
Rectal temperature response to an endotoxin (LPS; 0.5 µg/kg body weight) challenge (control, n = 10; chromium, n = 10). Rectal temperature data are presented as least squares means (LSM; SEM ± 0.09 for both control and chromium treatments). Baseline rectal temperature data presented as average of LSM for pre-LPS rectal temperatures (−120 to 0 min; SEM ± 0.08 for both control and chromium treatments).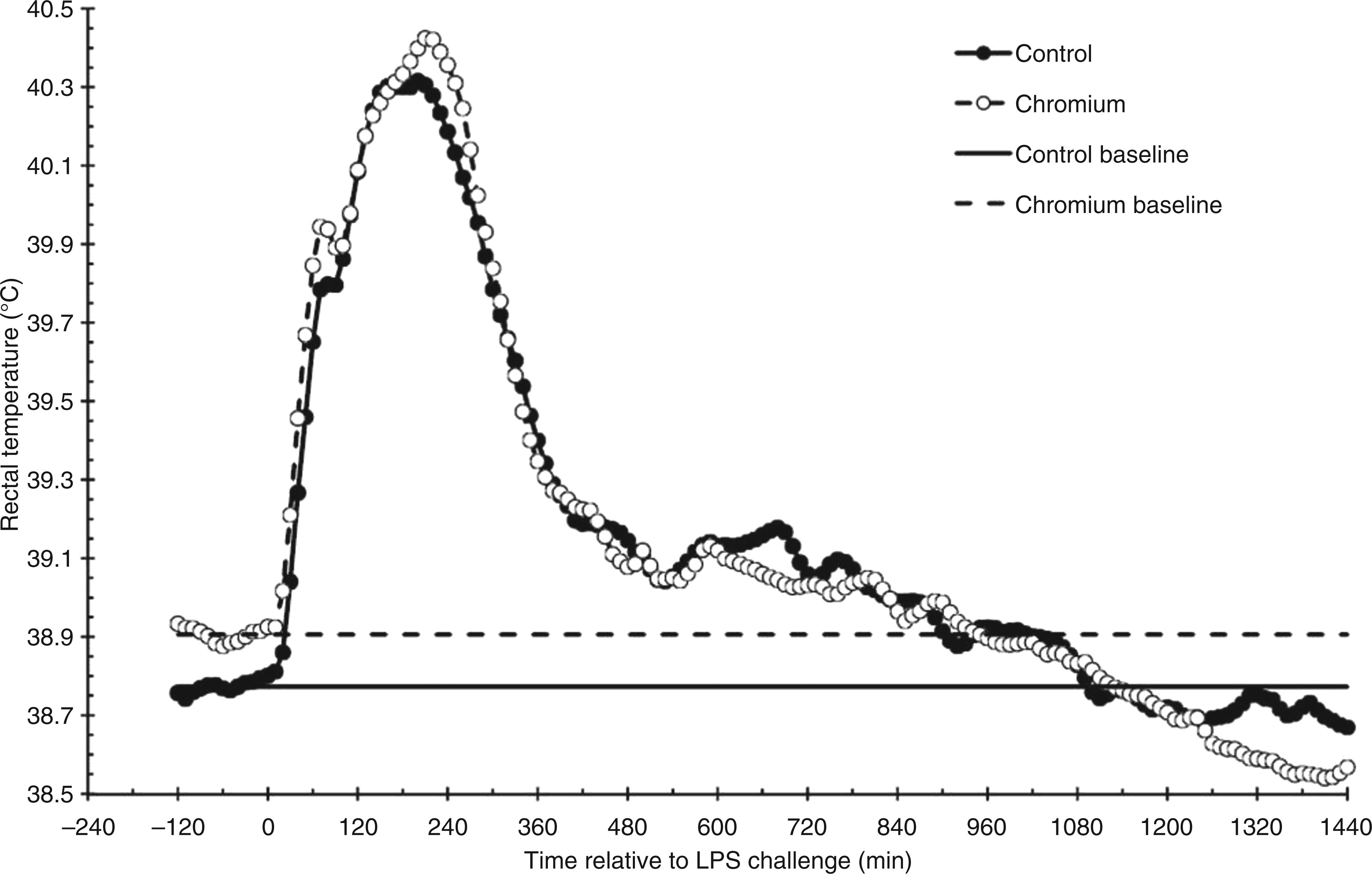
Sickness behavior
Steer sickness behavior did not differ prior to administration of LPS (P = 1.00; all steers had a score of 1 prior to the LPS challenge; Figure 2). Post-LPS sickness behavior scores increased in both treatments (P < 0.001), with control steers displaying greater behavioral signs of sickness (1.20 ± 0.01) compared with chromium-supplemented steers (1.15 ± 0.01; P = 0.030). Peak sickness behavior scores were attained at 0.5 h post-LPS. Sickness behavior scores returned to values not different than baseline values 3 h post-LPS administration in chromium-supplemented steers (P = 0.571) and 3.5 h post-LPS administration in control steers (P = 0.258).
Sickness behavior response to an endotoxin (LPS; 0.5 µg/kg body weight) challenge (control, n = 10; chromium, n = 10). Data presented as least squares means ± SEM. For sickness behavior a score of 1 represents a steer that is active or agitated and a score of 5 represents a steer that is lying on its side with labored breathing.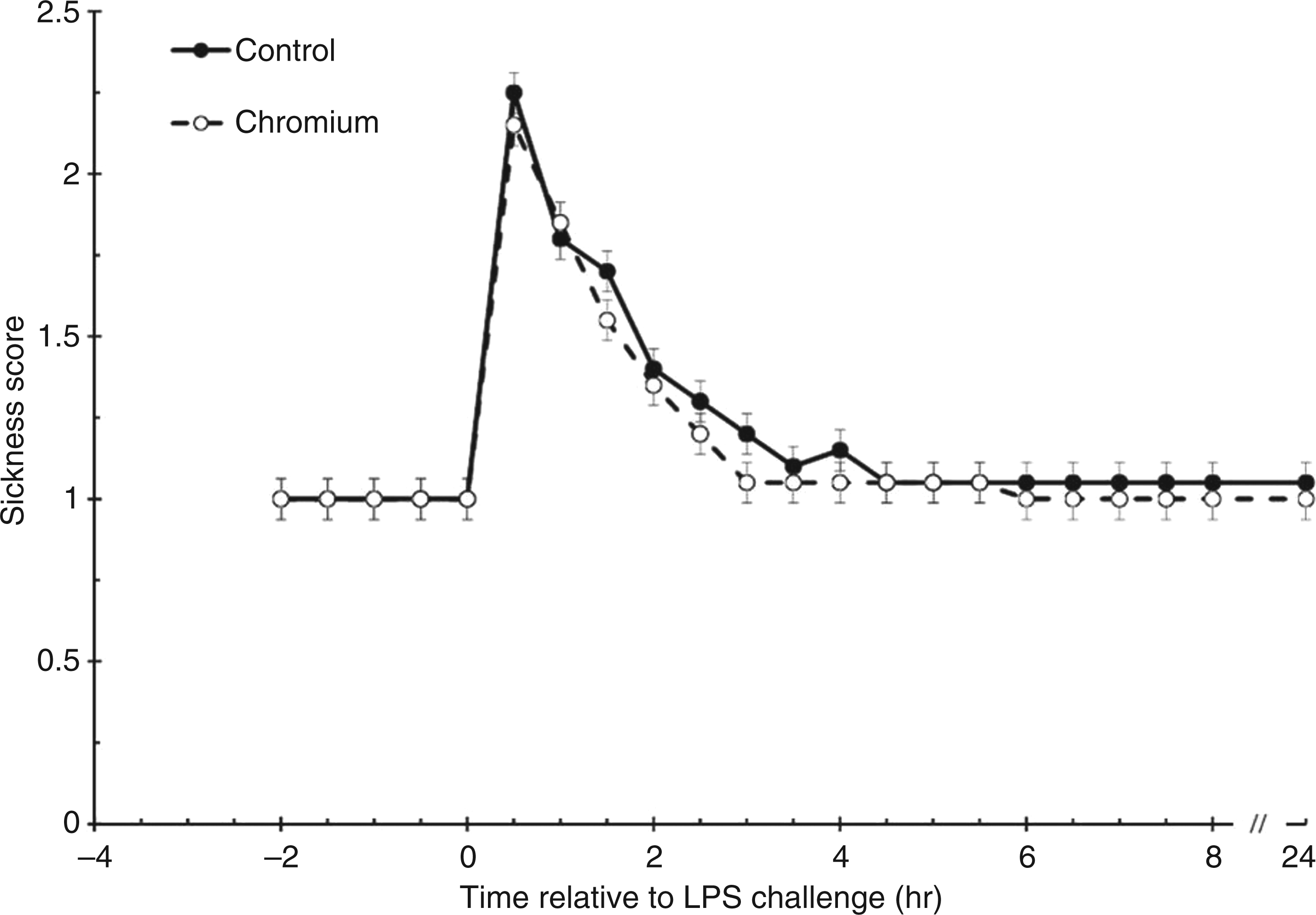
Cortisol concentrations
Prior to LPS administration, serum cortisol concentrations did not differ between treatments (P = 0.678; Figure 3). However, from −2 to 0 h there was an overall time effect (P < 0.001) such that serum cortisol concentrations increased in both groups (5.1 ± 1.5 and 17.0 ± 1.5 ng/ml at −2 and 0 h, respectively). Serum concentrations of cortisol increased in response to LPS administration at 0 h (P < 0.001), and peaked at 1 h post-LPS. Serum cortisol concentrations following LPS administration did not differ between treatments (P = 0.264). Serum concentrations of cortisol returned to baseline at 3.5 h (P = 0.075) for chromium-supplemented steers and 4 h (P = 0.147) for control steers.
Cortisol response to an endotoxin (LPS; 0.5 µg/kg body weight) challenge (control, n = 10; chromium, n = 10). Data presented as LSM ± SEM.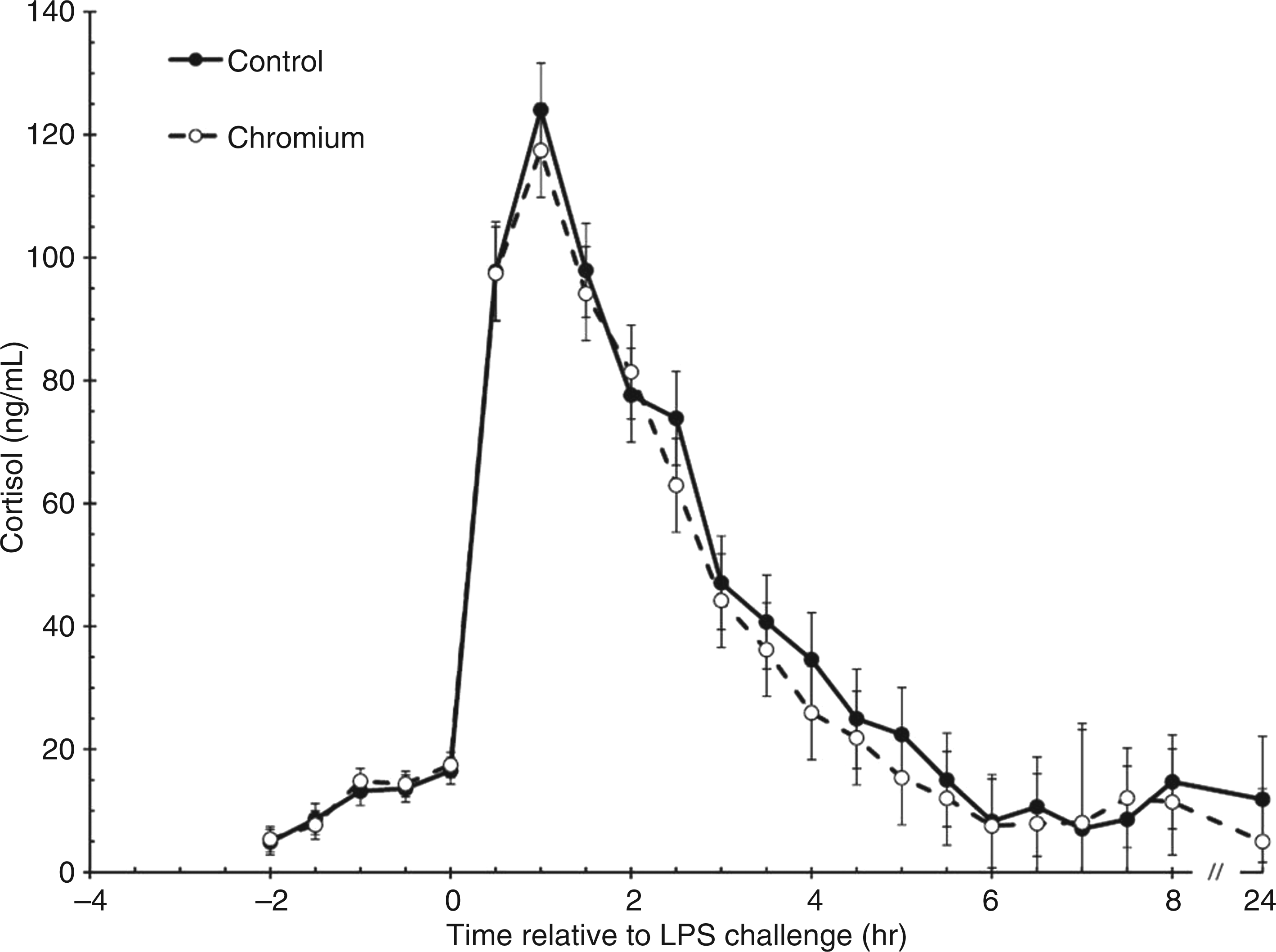
Cytokine concentrations
Serum concentrations of IL-4 were not affected by treatment pre- or post-LPS administration (P = 0.699 and 0.320, respectively; Figure 4A). Post-LPS administration, serum IL-4 concentrations increased (P < 0.001) and peaked at 3 h (16.6 ± 1.3 pg/ml) before declining. Serum IL-4 concentrations returned to baseline at 4 h for both control and chromium-supplemented steers (P = 0.239 and 0.446, respectively).
Cytokine response to an endotoxin (LPS; 0.5 µg/kg body weight) challenge (control, n = 10; chromium, n = 10). (A) IL-4; (B) TNF-α; (C) IFN-γ; (D) IL-6. Inset of (D): treatment averages of pre-LPS serum IL-6 concentrations. Data presented as least squares means ± SEM. Treatment means differ: *P < 0.05; †P < 0.08.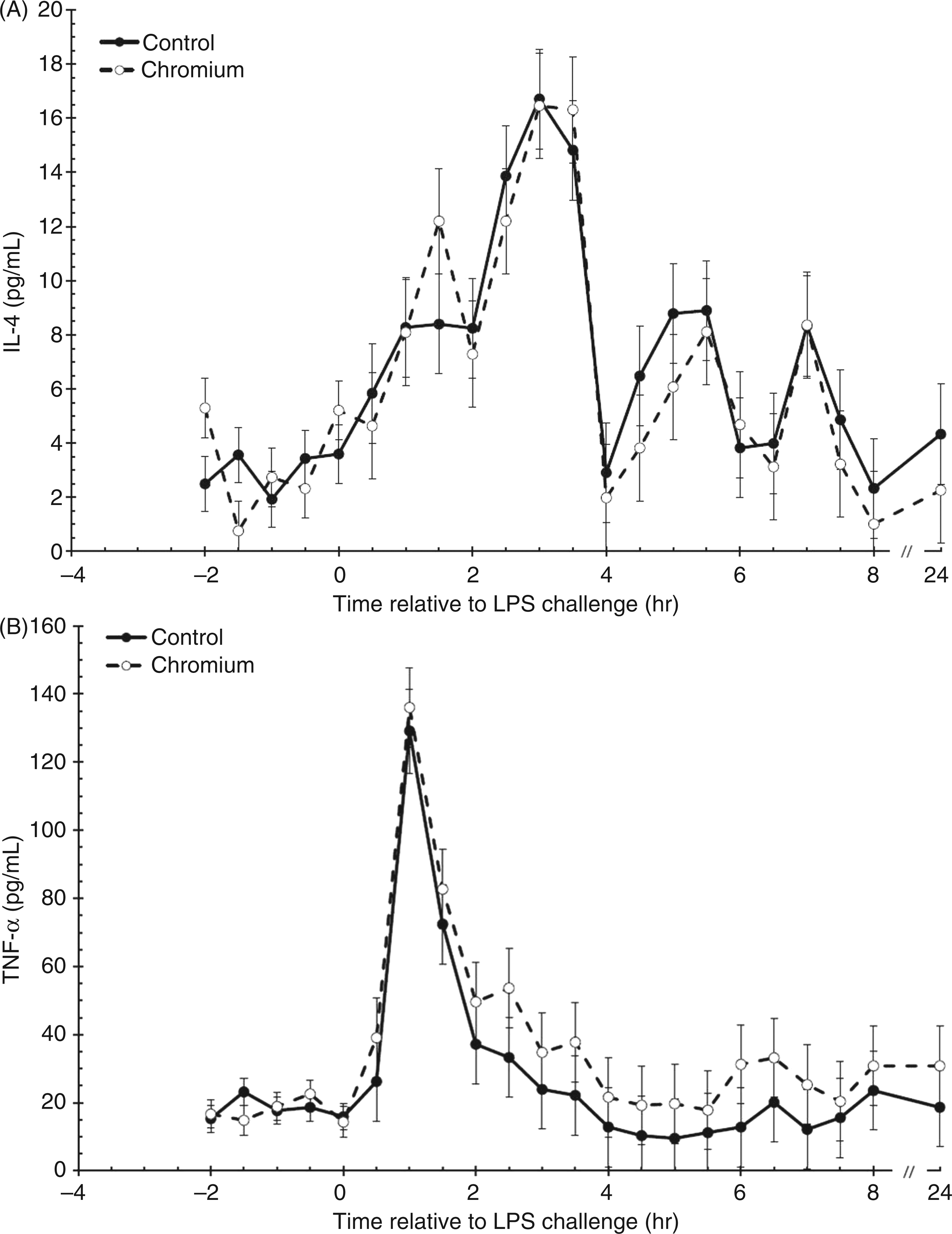
Serum concentrations of TNF-α (Figure 4B) and IFN-γ (Figure 4C) did not differ between treatments prior to LPS administration (P = 0.793 and 0.177, respectively). Following administration of LPS, serum TNF-α concentration increased (P < 0.001) and peaked within 1 h post-LPS for both treatment groups. Serum concentrations of TNF-α were greater in chromium-supplemented (40.2 ± 2.8 pg/ml) steers than control (28.9 ± 2.8 pg/ml) steers post-LPS (P = 0.005). Serum concentrations of TNF-α returned to baseline by 2 h in chromium-supplemented (P = 0.163) and control steers (P = 0.190). Serum concentrations of IFN-γ also increased in response to LPS administration (P < 0.001), with peak concentrations occurring between 2.5–3.5 h post-LPS. Similar to TNF-α, serum concentrations of IFN-γ were greater post-LPS in chromium-supplemented (19.2 ± 0.7 pg/ml) steers than control steers (15.6 ± 0.7 pg/ml; P = 0.004). Serum concentrations of IFN-γ returned to baseline by 7 h in both control and chromium-supplemented steers (P = 0.280 and 0.390, respectively).
Prior to administration of LPS at time 0 h, serum concentrations of IL-6 were greater in chromium-supplemented steers (10.7 ± 1.8 pg/ml) than control steers (4.9 ± 1.9 pg/ml; P = 0.027; Figure 4D). Following administration of LPS, serum IL-6 concentrations increased (P < 0.001), with a more robust increase in IL-6 produced in chromium-supplemented than control steers (time × treatment interaction; P = 0.020). This resulted in greater serum IL-6 being produced by chromium-supplemented (506.17 ± 34.91 pg/ml) steers than control steers post-LPS administration (296.21 ± 36.80 pg/ml; P < 0.001). Serum concentrations of IL-6 returned to baseline by 4 h for control and chromium-supplemented steers (P = 0.162).
Discussion
The influence of chromium supplemented as chromium propionate on the acute phase response of crossbred steers to LPS challenge was assessed. The results indicate that chromium can alter the acute phase response through modulation of RT, sickness behavior, and specific cytokine concentrations.
Prior to initiation of the challenge, steers supplemented with chromium had greater RT than control steers. This indicated a potential difference in metabolism between those supplemented with chromium and control steers. As chromium can affect the interaction between insulin and its receptor, it is possible that chromium influenced the inherent metabolic rate of the chromium-supplemented steers. Carroll and Forsberg 4 reported that in response to an immune challenge, an estimated 10–13% increase in metabolism is required to increase body temperature by 1°C. If the basal metabolic rate was increased through chromium supplementation, it would suggest that chromium increased the available energy resources for the immune system to utilize in response to the LPS challenge. Additionally, an increased basal metabolic rate may have been the driving factor associated with the greater basal RT exhibited by chromium-supplemented steers, thus resulting in a lower overall RT response (relative to baseline) in chromium-supplemented steers compared to control steers. It is important to note that an elevated body temperature in response to an invading pathogen is essential as it aids in the clearance of invading microorganisms. However, the peak RT values were similar in both chromium-supplemented and control steers, yet chromium-supplemented steers, relative to baseline, had a lesser increase in RT than control steers. Additionally, increases in RT are stimulated by pro-inflammatory cytokines, including TNF-α, and IL-6,17,18 which were both greater in chromium-supplemented steers than control steers post-LPS administration. However, although greater cytokine concentrations were observed in chromium-supplemented steers, this did not result in a greater increase in RT in these steers. While cytokines have a role in modulating body temperature in response to infection, other systems, including the central nervous system (e.g. catecholamines),19,20 are also involved with thermoregulation and therefore may play a more important role in regulating body temperature than cytokines in the current study.
In pigs, there was no effect of supplemental chromium as Cr(III) picolinate on RT measured by a hand thermometer. 21 Arthington et al. 22 did not report differences in RT in Holstein bull calves supplemented with a high-chromium yeast in which RT was measured daily in response to bovine herpesvirus-1 infection. Additionally, Kegley et al. 23 found no effect of chromium-nicotinic acid complex supplementation on RT measured daily following infectious bovine rhinotracheitis virus (IBRV) inoculation. In contrast, Moonsie-Shageer and Mowat 24 found a decrease in RT of Charolais-cross steers fed chromium from high-chromium yeast. However, the current study used indwelling RT recording devices that measured RT over time at 1-min intervals, while the previous studies only measured RT with a hand-held thermometer. Therefore, utilization of indwelling RT devices that measure continuously may have been able to clarify a response not seen in the previous studies due to the nature in which RT data was collected. Additionally, all previously mentioned studies utilized different chromium products.
As stated earlier, the immune response is an energy-costly process. In response to infection, cattle behavior is changed in order to reduce energy expenditure and allow energy to be redistributed to the immune system. 4 Therefore, there is an increase in the amount of time spent lying down and a decrease in the amount of time spent foraging and standing. In the current study, steers supplemented with chromium displayed less signs of sickness than control steers in response to the LPS challenge. While not a direct comparison to LPS-induced sickness behavior, Moonsie-Shageer and Mowat 24 reported decreases in morbidity in Charolais-cross steers fed chromium from a chromium yeast product for 30 days. Wright et al. 25 found no effect of chelated-chromium supplementation on morbidity in Charolais cross-bred steers, but did report a decrease in sickness relapse in supplemented steers. A change in metabolic rate, as suggested in the previous paragraph, would provide greater energy towards the immune system without the resultant need to conserve energy, thus resulting in the decreased sickness behavior observed in chromium-supplemented steers.
Cortisol concentrations were not affected by treatment prior to, or following, administration of LPS. There is conflicting evidence on the role of chromium on affecting changes in cortisol concentrations. Chromium has been reported to decrease cortisol concentrations in stressed Charolais-cross steers.24,26 In pigs supplemented with chromium as Cr(III) picolinate, Kim et al. 21 found no effect of chromium supplementation on cortisol concentrations in response to LPS. However, the pigs in the study by Kim et al. 21 were physically handled in order to collect each blood sample, potentially causing additional stress to the pig and negating any treatment effect. In cattle, Arthington et al. 22 found no effect of high-chromium yeast supplementation on plasma concentrations of cortisol or adrenocorticotropic hormone (ACTH) in Holstein bull calves that were inoculated with bovine herpesvirus-1. Kegley et al. 23 also reported no difference in cortisol concentrations between control and chromium-nicotinic acid complex-supplemented Angus-cross steers. Mowat et al. 27 suggested that early feeding of chromium during a period of stress was able to decrease cortisol concentrations. However, in the current study steers were supplemented for 56 d and cortisol concentrations pre- and post-LPS challenge were unaffected by supplementation. Spears 28 listed four reasons for discrepancies between studies discussing changes in immune function: the chromium status of cattle prior to the initiation of the study; the chromium content in the control diet; the type of chromium used for supplementation; and the stressor used during the study. An additional factor could be the concentration of chromium in the supplemental product and the rate at which it is supplemented. All of these factors could influence the varying results observed in cortisol concentrations when chromium is supplemented in the diet.
The acute phase response results in the secretion of pro-inflammatory cytokines within 1–4 h, as demonstrated in the current study. Chromium supplementation enhanced the secretion of TNF-α, IFN-γ, and IL-6 in response to LPS. Additionally, IL-6 concentrations were greater in chromium-supplemented steers prior to administration of LPS. The greater increase in IFN-γ in chromium-supplemented steers in response to LPS support the finding by Faldyna et al. 12 who found a greater increase in the IgG2 subtype in response to tetanus toxoid vaccination, as IFN-γ is known to stimulate the production of this IgG subtype.
Coincident with the proposed role of chromium to enhance the Th2 response, 7 IL-6 concentrations were not only greater in chromium-supplemented steers prior to the challenge, but those steers also produced a more robust IL-6 response to challenge with LPS. Similar to the current study, the supplementation of chromium picolinate in Landrace × Poland China gilts resulted in greater concentrations of IL-6 than in control gilts. 29 Borgs and Mallard 7 suggested that modulation of the acute phase response by chromium supplementation was not a primary goal, as there was limited effects of chromium supplementation on serum haptoglobin, total hemolytic complement activity, 30 and neutrophil phagocytosis. 31 This can be argued based on the current study’s results. The current study measured several other parameters associated with the acute phase response than the previous studies referenced by Borgs and Mallard. However, IL-6 can also be considered a Th2 cytokine because of its effects on the stimulation of the humoral immune response. Therefore, the enhancement of antibody responses by chromium supplementation may be mediated through the regulation of cytokine production. 7 Limited research has been performed on the effects of chromium supplementation on the acute phase response and, based on the current data, further research is necessary in order to elucidate specific effects. One potential option is that supplementation of chromium may negate the normal chromium deficiency that arises in stressed and morbid animals, thus enhancing immune function. 11
Several factors may have influenced the production of cytokines in response to LPS. While cortisol is known to negatively-regulate immune function during stress, cortisol concentrations were not different between control and chromium-supplemented steers in the current study. Immuno-enhancing effects of insulin have also been described. 3 Through the actions chromodulin, chromium may alter concentrations of insulin. 11 Determination of concentrations of glucose and insulin from the collected blood may answer some of the above questions. Additionally, propionate, the chromium carrier in the current study, has been demonstrated to increase concentrations of glucose and insulin in early lactating dairy cows. 32 However, the amount of propionate added to the diet was very minute. The chromium premix only contained 2.5% of the chromium propionate product, which contained 0.04% chromium and used calcium carbonate as the primary mixture carrier. The premix was fed at a level of 2% of the diet (Table 1). Additionally, the chromium propionate product was added to the premix as a one-to-one substitute for ground corn. As a result, the diet not containing chromium received more ground corn than the diet that contained chromium. Propionate is the major volatile fatty acid produced from ground corn (46.9%) in the rumen. 33 Therefore, propionate is not believed to have had a significant role in modulating glucose or insulin concentrations.
Differences between some of the variables measured, such as sickness behavior and serum concentrations of TNF-α, and IFN-γ, were very small between control and chromium-treated steers. The differences in sickness behavior alone may not justify the addition of chromium to the diet. However, greater differences were observed in regard to RT, and serum concentrations and IL-6. Enhancement of the immune system is one area in which the cattle industry can see the most improvement and, therefore, chromium may be a useful immunomodulator to improve the health of cattle entering the feedlot. Finding ways to enhance immune function prior to arrival at a feedlot and during the receiving period has the potential to reduce the costs associated with morbidity and mortality during this very stressful time period.
Conclusions
In summary, our data indicated that chromium propionate supplementation enhanced the acute phase response of steers to an LPS challenge, which may expedite recovery of stressed, receiving cattle following acute infection. Therefore, chromium may be used as an immunomodulator to alleviate potential chromium deficiency during times of stress and sickness, resulting in enhancement of immune responses in stressed feedlot cattle. Due to the known effects of chromium on insulin and glucose metabolism, it will be necessary to determine the effects of chromium on insulin and glucose concentrations in response to LPS challenge. To our knowledge, this is one of the first studies to report the effect of chromium supplemented as chromium propionate on the acute phase response of cattle to endotoxin challenge.
Footnotes
Funding
This study was supported in part by Kemin Agrifoods, North America, Inc., Des Moines, IA, USA, and the Gordon W. Davis Regents Chair Endowment at Texas Tech University.
Acknowledgements
Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture (USDA). The USDA prohibits discrimination in all its programs and activities on the basis of race, color, national origin, age, disability, and where applicable, sex, marital status, familial status, parental status, religion, sexual orientation, genetic information, political beliefs, reprisal, or because all or part of an individual's income is derived from any public assistance program. (Not all prohibited bases apply to all programs.) Persons with disabilities who require alternative means for communication of program information (Braille, large print, audiotape, etc.) should contact USDA's TARGET Center at (202) 720-2600 (voice and TDD). To file a complaint of discrimination, write to USDA, Director, Office of Civil Rights, 1400 Independence Avenue, S.W., Washington, D.C. 20250-9410, or call (800) 795-3272 (voice) or (202) 720-6382 (TDD). USDA is an equal opportunity provider and employer. The authors acknowledge the technical assistance of J.R. Carroll, J.W. Dailey, A.N. Loyd, C. Wu, D.N. Fink, M.A. Jennings, and T.R. Young.
