Abstract
Infection and inflammation can result in the rapid loss of muscle mass and myofibrillar proteins (muscle atrophy). In addition, aspartate (Asp) is necessary for protein synthesis in mammalian cells. We hypothesized that Asp could attenuate LPS-induced muscle atrophy in a piglet model. Twenty-four weaning piglets were allotted to four treatments, including non-challenged control, LPS challenged control, LPS+0.5% Asp and LPS+1.0% Asp. On d 21, the piglets were injected with i.p. injection of LPS (100 ug/kg BM) or saline. At 4 h post-injection, blood, gastrocnemius and longissimus dorsi muscles samples were collected for biochemical and molecular analyses. Asp decreased the concentrations of cortisol and glucagon in plasma. In addition, Asp increased protein and RNA contents in muscles, and decreased mRNA expression of muscle atrophy F-box (MAFbx) and muscle RING finger 1 (MuRF1). Moreover, Asp decreased phosphorylation of AMPKα but increased phosphorylation of Akt and Forkhead Box O (FOXO) 1 in the muscles. Our results indicate that Asp suppresses LPS-induced MAFbx and MuRF1 expression via activation of Akt signaling, and inhibition of AMPKα and FOXO1 signaling.
Introduction
Skeletal muscle, the most abundant tissue in the body, plays an important role in supporting an animal’s physical and biochemical activities. 1 The rapid loss of skeletal muscle mass and myofibrillar proteins can arise as a secondary symptom related to sepsis and inflammation, and consequently results in muscle atrophy.1,2 The stimulation of proteolysis observed during atrophy takes place, at least in part, owing to an activation of the ubiquitin-proteasome pathway. 3 In the ubiquitin-proteasome pathway, the muscle-specific ubiquitin ligases muscle atrophy F-box (MAFbx) and muscle RING finger 1 (MuRF1) are considered to be the key regulators of muscle protein degradation. 4 In the case of sepsis-induced atrophy, MAFbx and MuRF1 are up-regulated several fold.3,5 They can be regarded as the early markers of the atrophy process, facilitating the differential diagnosis of muscle diseases.3,5
Several lines of evidence indicate that the signaling pathways, mediated by the nuclear transcription factors termed Forkhead Box O (FOXO), regulate the expression of the muscle-specific ubiquitin ligases (i.e. MAFbx and MuRF1).3,5 In skeletal muscle, the activation of FOXO has been identified in conditions of muscle atrophy. 6 Various protein kinases, including Akt and AMPK, have been reported to regulate FOXO activation.1,3,5,7,8 Akt, a serine-threonine protein kinase, suppresses protein degradation through inhibiting the FOXO proteins from entering the nucleus and preventing transcription of FOXO target genes, i.e., MAFbx and MuRF1. 6 AMPK, which is composed of one catalytic (α) and two non-catalytic subunits (β and γ), also controls protein degradation. 8 Interestingly, unlike Akt, AMPK mediates muscle loss and catabolism through increasing the activity of FOXO transcription factors. 8
Aspartate (Asp), which can be synthesized de novo, is categorized as a non-essential amino acid in mammals. 9 Importantly, Asp has been shown to exert various functions in the physiological and biological processes. Firstly, Asp is used in the biosynthesis of nucleotides and proteins. 10 Beyond its role as an amino acid in proteins, Asp provides carbon backbone for de novo pyrimidine biosynthesis and is necessary for the conversion of IMP to AMP in de novo purine biosynthesis. 10 In addition, Asp can be metabolized by skeletal muscle, and its combinations with other substances play an important role on muscle metabolism.11,12 For example, Lancha et al. 11 reported that Asp and asparagine supplementation increased glycogen concentration and modulated the glucose uptake in rat muscle. Lancha et al. 12 also reported that Asp, asparagine, and carnitine supplementation increased the capacity of muscle to utilize free fatty acids and spare glycogen. However, there is little research on how Asp modulates muscle atrophy.
Accordingly, we hypothesized that Asp supplementation would protect against muscle atrophy through controlling the expression of muscle ubiquitin ligases. In this study, Escherichia coli LPS was administrated to mimic endotoxaemia. 13 In addition, a piglet model was used to establish the model for studying nutrition and physiology of human, specifically child and adolescent with muscles undergoing rapid growth.14,15 Our objective was to evaluate whether Asp could inhibit LPS-induced muscle atrophy, and to elaborate its molecular mechanism(s).
Materials and methods
Animals and experimental design
All procedures performed on the animals were approved by the Animal Care and Use Committee of Wuhan Polytechnic University. In all, 24 weaned castrated barrows (Duroc × Large White × Landrace, weaned at 21 ± 1 d of age), weighing 8.9 ± 0.2 kg, were acquired. The piglet was individually housed in 1.80 × 1.10 m2 pen at a temperature of 25–27℃, in a humid and light-controlled room with free access to food and water. The basal diet (Supplementary Table 1) was prepared on the basis of the nutrient requirements of National Research Council. 16
The 24 piglets were randomly assigned to the following four treatments (six replicate pens for per treatment): (1) non-challenged control (CONTR; piglets fed a control diet and injected with 0.9% NaCl solution); (2) LPS-challenged control [LPS; piglets fed the same control diet and injected with E. coli LPS (E. coli serotype 055: B5; Sigma Chemical Inc., St. Louis, MO, USA)]; (3) LPS + 0.5% Asp treatment (piglets fed a 0.5% Asp diet and injected with LPS); and (4) LPS + 1.0% Asp treatment (piglets fed a 1.0% Asp diet and injected with LPS). The Asp doses (purity > 99%; Amino Acid Bio-Chemical Co., Wuhan, China) were selected according to our previous studies.17,18 Our previous studies showed that, prior to LPS challenge, dietary 0.5% and 1.0% Asp supplementation had no effect on growth performance, total and differential leukocyte counts, and serum biochemical parameters of weaning piglets, 17 suggesting that the Asp level in basal diet was enough to meet the requirements of weanling piglets’ growth and physiological function in normal physiological condition. However, our previous studies also showed that, after LPS challenge, both 0.5% and 1.0% Asp attenuated the alteration of these variables induced by LPS, 17 suggesting that exogenous Asp supply exerted beneficial effects under pathological conditions. Thus, in this experiment, we focused on investigating the effect of dietary 0.5% and 1.0% Asp supplementation on muscle variables in LPS-challenged pigs, and did not investigate the effect of Asp in pigs without LPS challenge. We added 0.67%, 0.34% and 0% alanine (purity > 99%; Amino Acid Bio-Chemical Co.) to the control, 0.5% Asp and 1.0% Asp diets, respectively, to get isonitrogenous diets. After 19 d of feeding with the control, 0.5% Asp and 1.0% Asp diets, the challenged groups were treated with i.p. injection of LPS at 100 µg/kg BM, and the non-challenged group was treated with the same volume of 0.9% NaCl solution. The experiment was conducted for 19 d, according to our previous study. 19 In addition, the LPS dose was chosen in accordance with our previous experiments, which demonstrated that this dose of LPS caused tissue damage in weaning piglets.20,21
Blood and muscle sample collections
At 4 h after administration with saline or LPS, blood samples were collected and centrifuged to separate plasma. Plasma was kept at −80℃ for further measurement of cortisol and glucagon concentrations. Following blood collection, the piglets were humanely euthanized by i.m. injection of pentobarbital (80 mg/kg BM; Zao Zhuang Ke Shun Chemical Co., Ltd., Shandong, China). The gastrocnemius and longissimus dorsi (LD) muscles were collected under the semitendinosus and biceps flexor cruris (left hindlimb) and from the tenth rib (right side of the carcass), respectively. The muscle samples were frozen immediately in liquid nitrogen, and then stored at −80℃ for further measurement. In several studies, the gastrocnemius and LD muscles were selected to study muscle atrophy.22,23 High levels of MAFbx were found in the gastrocnemius and LD muscles in piglets with muscle fiber atrophy. 23 Therefore, we chose these two muscles to investigate muscle atrophy. In addition, the time point of 4 h following LPS or saline injection was selected according to the previous experiments.21,24,25
Plasma cortisol and glucagon concentrations
Plasma cortisol and glucagon concentrations were measured with 125I RIA assay kits (Beijing North Institute of Biological Technology, Beijing, China). All experimental procedures were performed according to the manufacturer's instructions.
Muscle protein, DNA and RNA contents
The methods to analyze muscle protein, DNA and RNA contents were the same as previously described. 20
Real-time PCR
RNA isolation, quantification, cDNA synthesis and real-time PCR were performed as previously described. 21 The primer pairs used are presented in Supplementary Table 2. The 2−ΔΔCT method 26 was used to determine changes in gene expression between treatment groups with the GAPDH CT as the correction factor. We analyzed the stability of several housekeeping genes using the method of Vandesompele et al. 27 Our results demonstrated that the expression of GAPDH is more stable than that of other housekeeping gene, and GAPDH did not exhibit any difference among four treatments or two muscles. Relative mRNA abundance of each target gene was normalized to the control group.
Western blot
Western blot analysis was performed according to the method of Liu et al. 21 Briefly, the muscle samples were homogenized and centrifuged, and the supernatants were collected to measure protein concentrations using the BCA reagent (Sigma Chemical Inc.). Equal amounts of muscle proteins (65 µg) were loaded onto polyacrylamide gels, separated through SDS-PAGE, transferred to blotting membranes, and then incubated with the primary antibodies (Abs). Next, the membranes were incubated with the secondary Ab. The following primary Abs were used: total AMPKα (tAMPKα; 1:1000; #2532), phosphorylated AMPKα (pAMPKα, Thr172; 1:1000; #2535), total Akt (tAkt, 1:1000; #9272), phosphorylated Akt (pAkt, serine473; 1:1000; #9271), total FOXO 1 (tFOXO1; 1:1000; #9454) and phosphorylated FOXO 1 (pFOXO1, serine256; 1:1000; #9461) (all from Cell Signaling Technology Inc., Danvers, MA, USA). An Enhanced Chemiluminescence Western blotting kit (Amersham, Little Chalfont, UK), a Gene Genome bioimaging system (Alpha Innotech, Kasendorf, Germany), and a GeneTools software (Syngene, Cambridge, UK) were used to develop, visualize and analyze the bands, respectively. Phosphorylated forms of AMPKα, Akt and FOXO1 were normalized with the total protein content.
Eight samples from four treatments (n = 2) were run in one gel. Thus, 24 samples from four treatments (n = 6) were run in three gels at one time to minimize the variations of gel to gel. The analyses were conducted in duplicate.
Statistical analysis
Data were analyzed by the mixed procedure of SAS (SAS Institute Inc., Cary, NC, USA) appropriate for a randomized complete-block design. The comparison between CONTR and LPS (0% Asp) group was to determine the effect of LPS on the piglets. Linear and quadratic polynomial contrasts were used to determine the response to dietary Asp supplementation among LPS-challenged piglets. The data were expressed as means and pooled SEM in Tables 1–3, or as means and SEs in Figures 1–4. The statistical significance level for all analyses was set at P ≤ 0.05, and 0.05 < P ≤ 0.10 were considered as trends.
Results
Plasma cortisol and glucagon concentrations
Effects of Asp supplementation on plasma cortisol and glucagon concentrations in weaning piglets at 4 h after the administration of E. coli LPS challenge. a
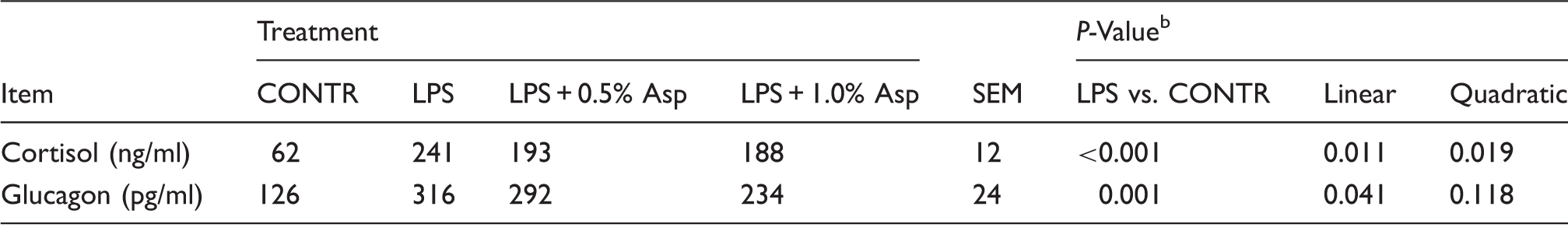
Values are means and pooled SEM, n = 6 (one pig/pen).
The LPS pigs were compared with CONTR pigs to determine the effect of LPS. Linear and quadratic polynomial contrasts were used to determine the response to Asp supplementation among LPS-challenged pigs.
Protein, DNA and RNA contents
Effects of Asp supplementation on muscle protein, DNA and RNA contents in weaning piglets at 4 h after the administration of E. coli LPS challenge. a
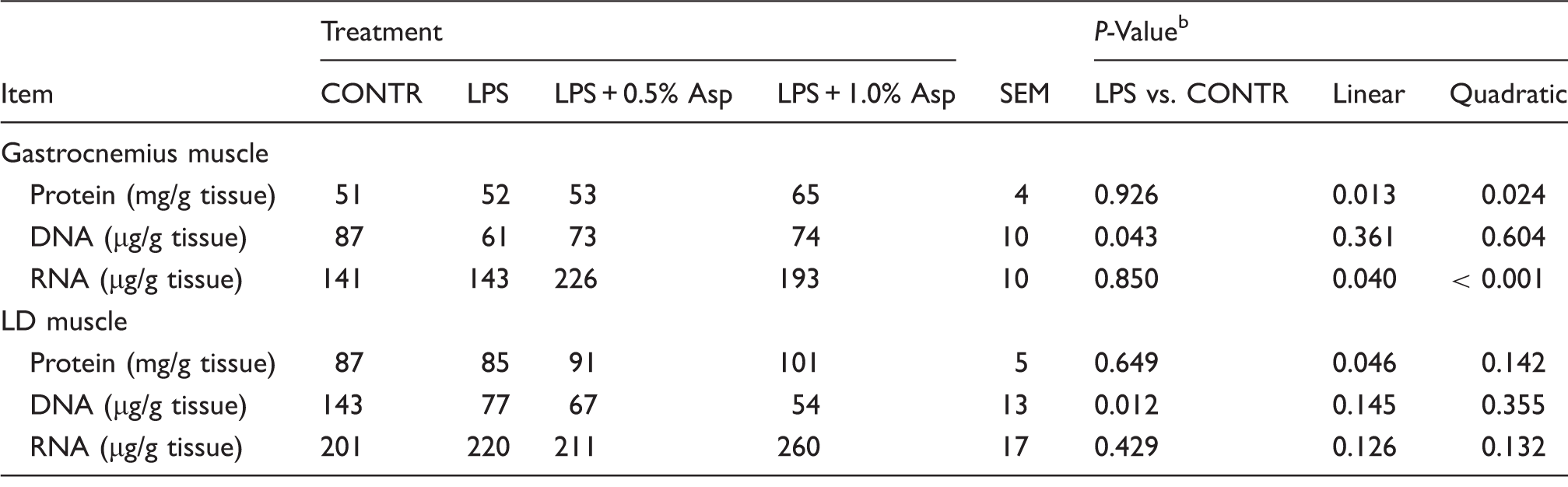
Values are means and pooled SEM, n = 6 (one pig/pen).
The LPS pigs were compared with CONTR pigs to determine the effect of LPS. Linear and quadratic polynomial contrasts were used to determine the response to Asp supplementation among LPS-challenged pigs.
mRNA expression of MAFbx and MuRF1
Relative to CONTR piglets, LPS challenge increased mRNA expression of MuRF1 in gastrocnemius and LD muscles (P < 0.05; Figure 1), and MAFbx in LD muscle (P < 0.001). Among the LPS-challenged piglets, Asp supplementation decreased mRNA expression of MAFbx in gastrocnemius (linear, P < 0.05) and LD (linear, P = 0.095; quadratic, P = 0.086) muscles, and MuRF1 in gastrocnemius (linear, P = 0.055; quadratic, P = 0.056) and LD (linear, P = 0.001; quadratic, P < 0.01) muscles.
Effects of Asp supplementation on the mRNA expression of (a) MAFbx and (b) MuRF1 in gastrocnemius and LD muscles of weaning piglets at 4 h after LPS challenge. The LPS (0% Asp) pigs were compared with CONTR pigs (LPS vs. CONTR) to determine the effect of LPS. Linear (L) and quadratic (Q) polynomial contrasts were used to determine the response to dietary Asp supplementation among LPS-challenged pigs. Values are means with SEs, n = 6 (one pig/pen).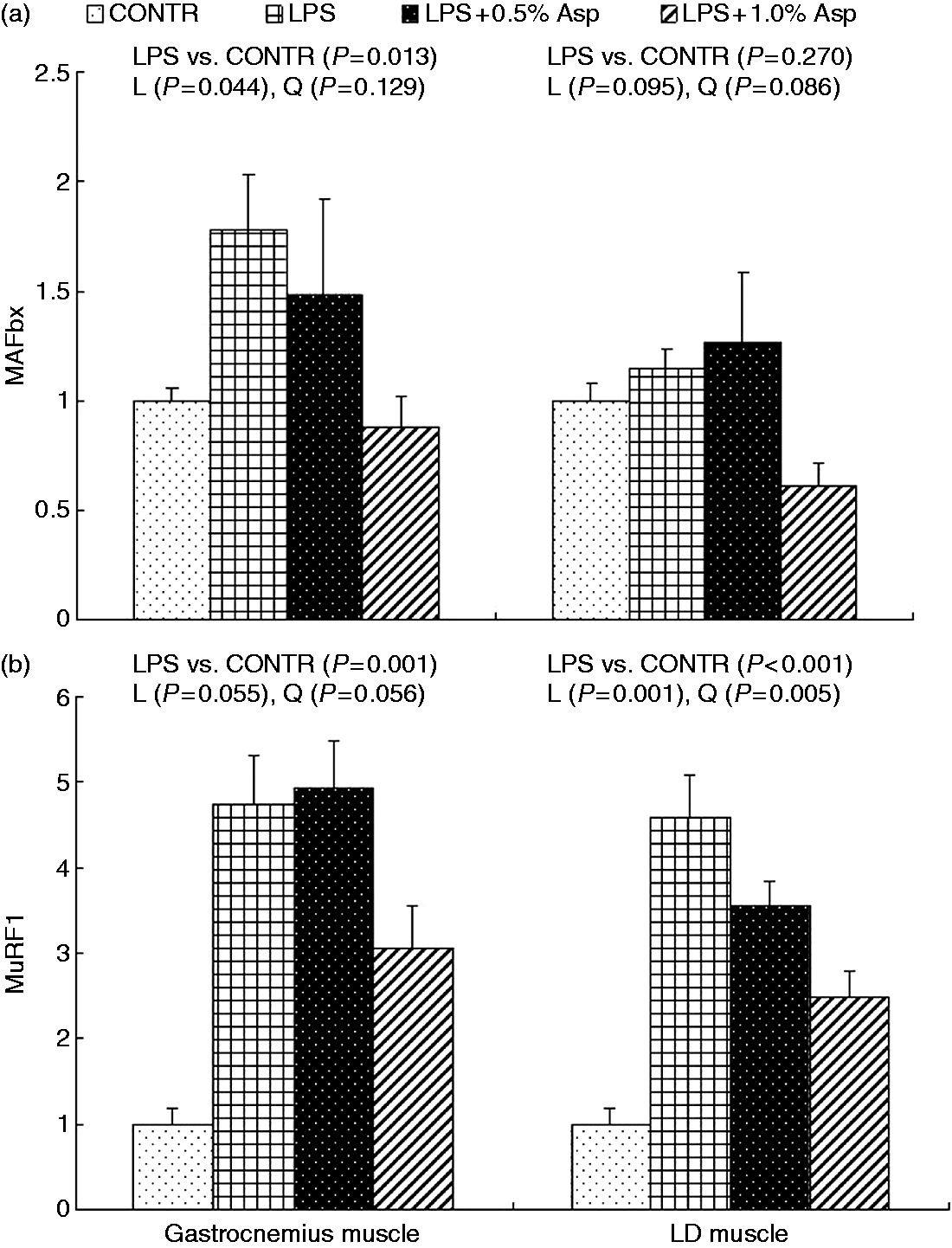
mRNA expression of AMPKα, Akt1 and FOXO1
Effects of Asp supplementation on muscle mRNA expression of AMPKα, Akt1 and FOXO1 in weaning piglets at 4 h after the administration of E. coli LPS challenge. a
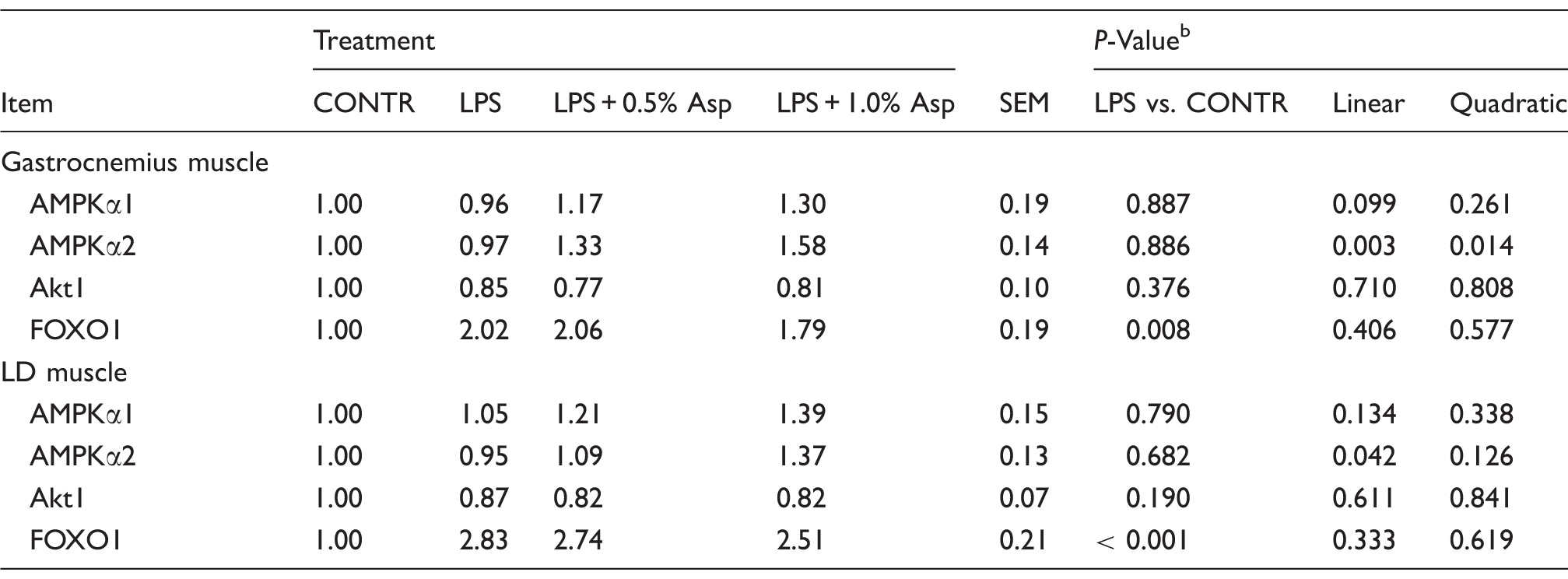
Values are means and pooled SEM, n = 6 (one pig/pen). The LPS pigs were compared with CONTR pigs to determine the effect of LPS. Linear and quadratic polynomial contrasts were used to determine the response to Asp supplementation among LPS-challenged pigs.
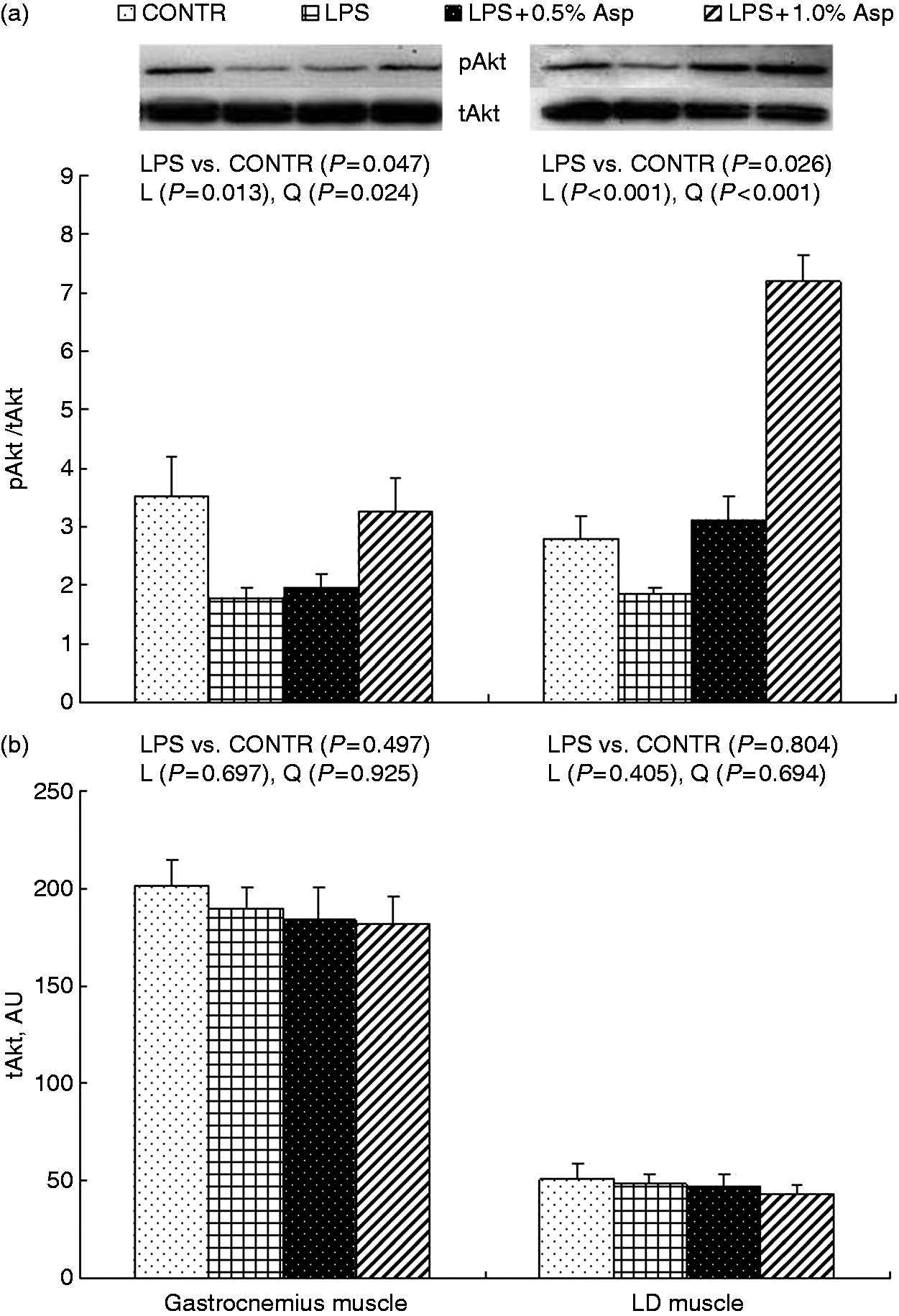
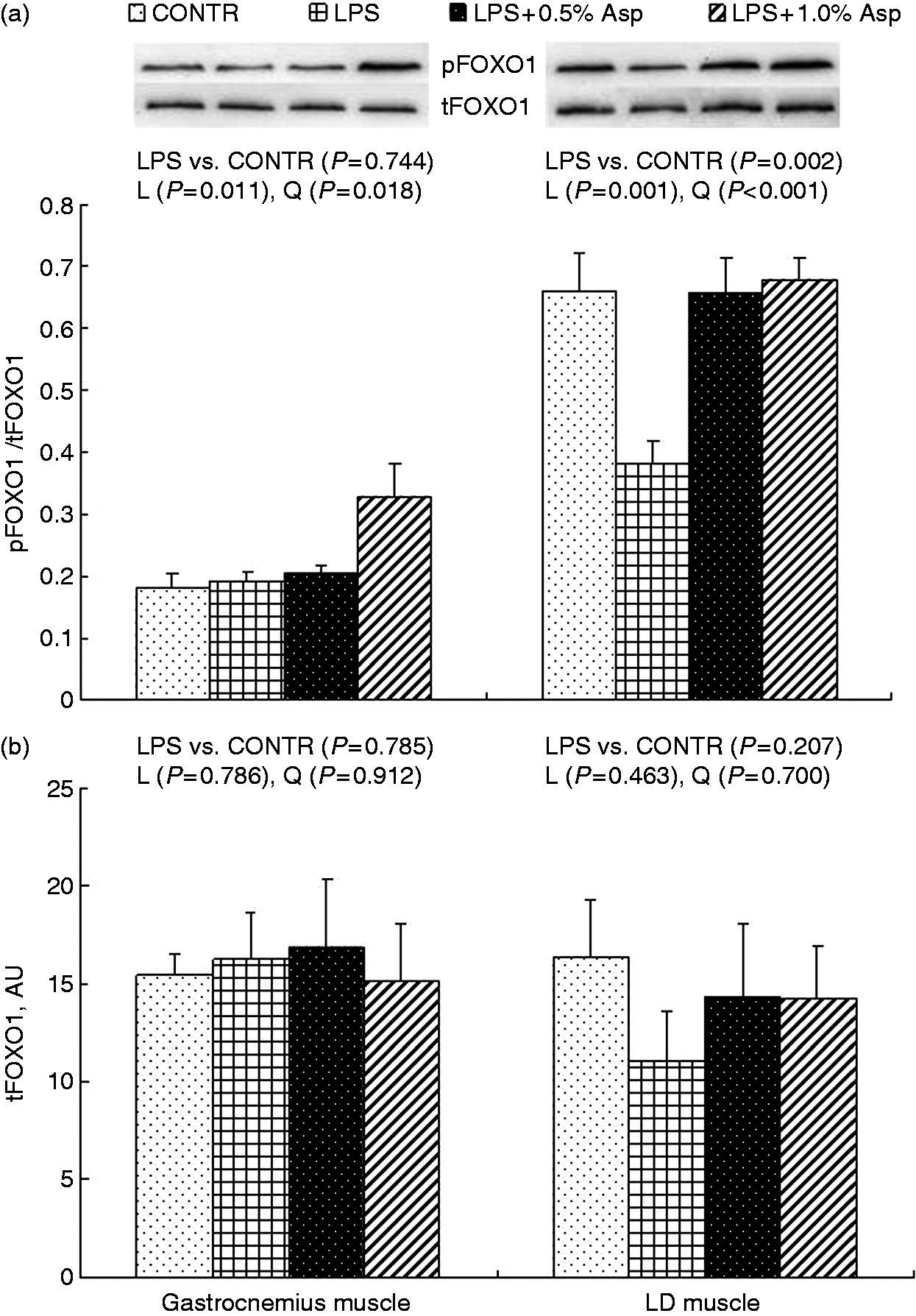
Protein phosphorylation and abundance of AMPKα, Akt and FOXO1
Relative to CONTR piglets, LPS challenge decreased the ratio of pAkt/tAkt in gastrocnemius and LD muscles, and the ratio of pFOXO1/tFOXO1 in LD muscle (P < 0.05; Figures 2–4). Among the LPS-challenged piglets, Asp supplementation decreased the ratio of pAMPKα/tAMPKα in gastrocnemius muscle, increased the abundance of tAMPKα in LD muscle, and increased the ratio of pAkt/tAkt and the ratio of pFOXO1/tFOXO1 in gastrocnemius and LD muscles (linear, P < 0.05; quadratic, P < 0.05).
Effects of Asp supplementation on the (a) pAMPKα:tAMPKα ratio and (b) protein abundance of tAMPKα in gastrocnemius and LD muscles of weaning piglets at 4 h after LPS challenge. The bands shown are the representative Western blot images of pAMPKα (62 kDa) and tAMPKα (62 kDa). The LPS (0% Asp) pigs were compared with CONTR pigs (LPS vs. CONTR) to determine the effect of LPS. Linear (L) and quadratic (Q) polynomial contrasts were used to determine the response to dietary Asp supplementation among LPS-challenged pigs. Values are means with SEs, n = 6 (one pig/pen). AU: arbitrary units. Effects of Asp supplementation on the (a) pAkt: tAkt ratio and (b) protein abundance of tAkt in gastrocnemius and LD muscles of weaning piglets at 4 h after LPS challenge. The bands shown are the representative Western blot images of pAkt (60 kDa) and tAkt (60 kDa). The LPS (0% Asp) pigs were compared with CONTR pigs (LPS vs. CONTR) to determine the effect of LPS. Linear (L) and quadratic (Q) polynomial contrasts were used to determine the response to dietary Asp supplementation among LPS-challenged pigs. Values are means with SEs, n = 6 (one pig/pen). AU: arbitrary units. Effects of Asp supplementation on the (a) pFOXO:tFOXO ratio and (b) protein abundance of tFOXO in gastrocnemius and LD muscles of weaning piglets at 4 h after LPS challenge. The bands shown are the representative Western blot images of pFOXO (82 kDa) and tFOXO (82 kDa). The LPS (0% Asp) pigs were compared with CONTR pigs (LPS vs. CONTR) to determine the effect of LPS. Linear (L) and quadratic (Q) polynomial contrasts were used to determine the response to dietary Asp supplementation among LPS-challenged pigs. Values are means with SEs, n = 6 (one pig/pen). AU: arbitrary units.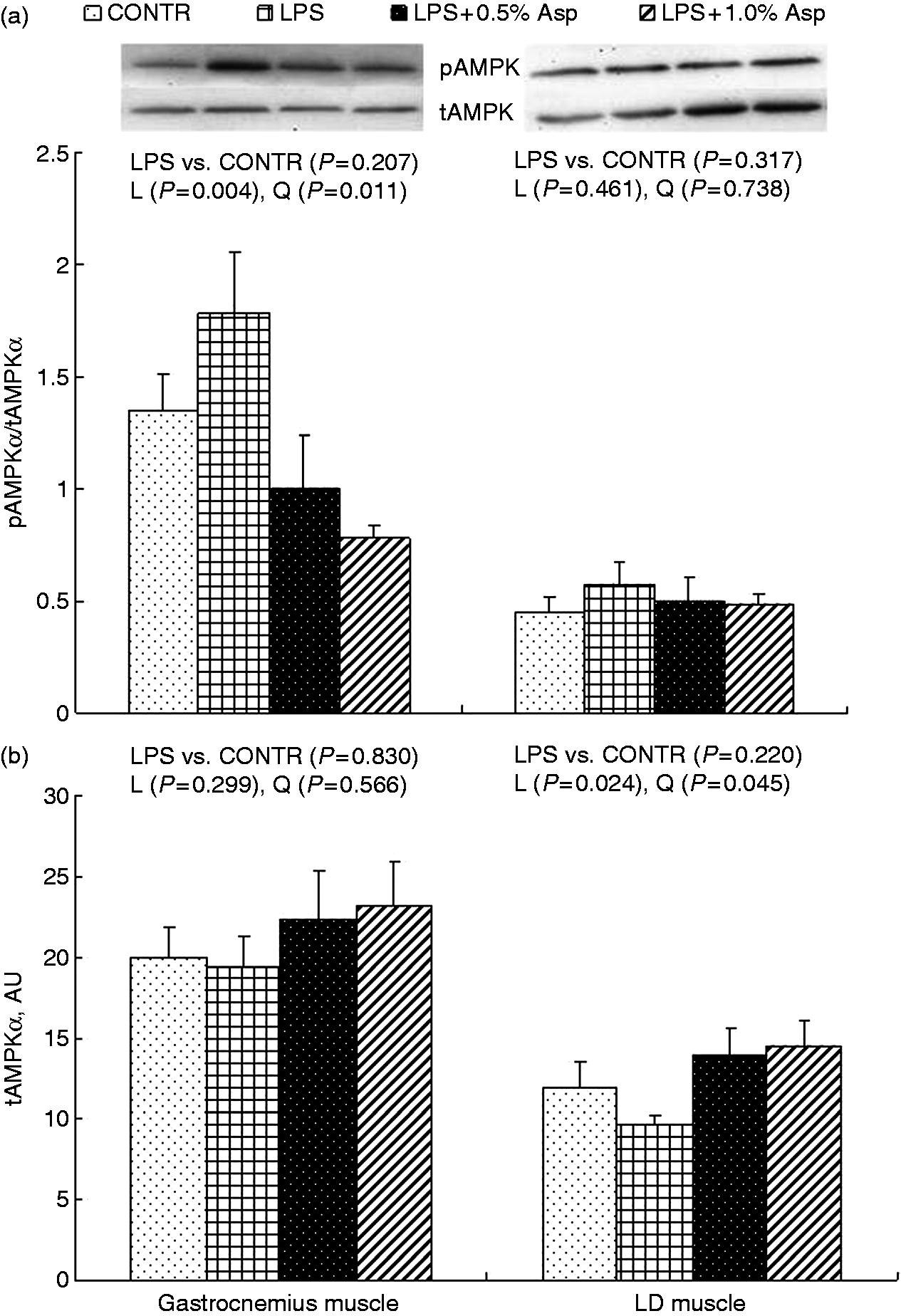
Discussion
LPS is a potent endotoxin and acute inflammation induced by i.p. LPS injection can cause serious skeletal muscle wasting.28,29 LPS challenge has been shown to decrease the diameter of myofibers and myotubes, and increase myosin heavy chain degradation and induce the loss of muscle strength and mass in mice. 30 Our previous research showed that dietary addition of 0.5% and 1.0% Asp alleviated LPS-induced intestinal injury in weaning piglets. 18 In this study, we extended our previous observations into the function of Asp in muscle tissue. Our results showed that Asp attenuated LPS-induced increase of plasma TNF-α, 18 cortisol and glucagon concentrations, suggesting that Asp attenuates LPS-induced systemic inflammatory and stress responses.
Skeletal muscle atrophy during sepsis is primarily a result of accelerated protein degradation, and is characterized by the increased expression of the muscle-specific E3 ubiquitin ligases (MAFbx and MuRF1). 31 MAFbx and MuRF1 are induced early in the atrophy process, and the increase in their mRNA expression precedes the loss of muscle mass. 32 The muscle protein, RNA and DNA are common metrics for assessing the protein synthetic capacity, translational efficiency and cell size, and they are negatively related to protein degradation. 33 In this study, we demonstrated that LPS injection increased mRNA expression of MAFbx and MuRF1, and decreased DNA content in muscles, indicating that LPS induced muscle atrophy. Our results are consistent with the findings of Schakman et al., 28 in rats, and Jamart et al., 34 in mice. Asp supplementation to the LPS-challenged piglets restored the protein and RNA contents, and decreased mRNA expression of MAFbx and MuRF1 in muscles, suggesting that Asp attenuated LPS-induced muscle atrophy. To our best knowledge, our results demonstrated for the first time that Asp can alleviate muscle atrophy.
The family of FOXO transcription factor has been implicated in proteolysis during muscle atrophy. 35 In particular, FOXO1 participates in the regulation of MAFbx and MuRF1 expression in sepsis-induced muscle wasting. 36 Akt and AMPK are considered to regulate protein degradation in muscle through FOXO and FOXO target genes (i.e. MAFbx and MuRF1).1,3,5,7,8 Phosphorylation of Akt by insulin or insulin-like growth factor-1 reduces muscle proteolysis through phosphorylating and inactivating FOXO transcription factors. 35 However, AMPK, in an active (phosphorylated) state, reduces the level of phosphorylated forms of FOXO and activates it. 37
In our study, LPS challenge decreased phosphorylation of Akt (the ratio of pAkt/tAkt) and phosphorylation of FOXO1 (the ratio of pFOXO1/tFOXO1). Similarly, Crossland et al. 13 reported that during LPS-induced endotoxemia, Akt, in a predominantly dephosphorylated/inactive state, enabled FOXO factors to enter the nucleus and up-regulate MAFbx and MuRF1 expression, promoting ubiquitin-proteasome pathway-mediated protein degradation. Consistent with decreased mRNA expression of MAFbx and MuRF1 in muscles, Asp supplementation to the LPS-challenged pigs increased phosphorylation of Akt and FOXO1, and decreased phosphorylation of AMPKα (the ratio of pAMPKα/tAMPKα). These results indicate that Asp prevented LPS-induced inhibition of Akt signaling and activation of AMPKα and FOXO signaling. So far, it is not known whether Asp directly or indirectly modulates muscle AMPKα, Akt and FOXO1 signaling. Asp may serve as substrate for protein synthesis or a signaling molecule, or both. Lancha et al. 11 reported that Asp combinations could increase glycogen concentration in rat muscle. Glycogen can allosterically inhibit AMPK activity. 38 In this way, Asp might inhibit AMPK activity through increasing muscle glycogen concentration. In addition, it is also possible that Asp can be converted to other substances such as oxaloacetate 39 and arginine, 40 to regulate these signaling pathways. Recent studies have demonstrated that both oxaloacetate and arginine increased levels of phosphorylated Akt.41,42 Williams et al. 43 reported that oxaloacetate supplementation increased lifespan via an AMPK/FOXO-dependent pathway.
In our study, the results of Asp on some muscle measurements showed difference between gastrocnemius and LD muscles. This may be attributed to different anatomy and physiology among the various segments of muscles. 44 In addition, our observations found that mRNA and protein of some measurements were differentially expressed, which is consistent with the findings of Maier et al. 45 and Koussounadis et al. 46 The distinction may be related to different levels of regulation between transcript and protein product.45,46 For the proof of concept, we focused on samples collected only at one time point (4 h) in LPS-induced muscle atrophy. In our future studies, we plan to conduct the measurements of multiple time points to further dissect the dynamic interplay and the long-term effects of Asp on muscle atrophy. Moreover, direct measurement of muscle mass and protein synthesis/degradation will be taken for the determination of the role of Asp on muscle growth.
In general, dietary Asp supplementation suppresses LPS-induced expression of muscle atrophy-associated genes and thus inhibits protein degradation. Asp may regulate muscle protein metabolism through controlling the expression of muscle ubiquitin ligases by regulating Akt, AMPK and FOXO1 signaling. However, according to our present study, it is difficult to draw the definite conclusion about how Asp affect the muscle atrophy. This is of concern and merits further investigation.
Supplemental Material
Supplemental material for Aspartate inhibits LPS-induced MAFbx and MuRF1 expression in skeletal muscle in weaned pigs by regulating Akt, AMPKα and FOXO1
Supplemental Material for Aspartate inhibits LPS-induced MAFbx and MuRF1 expression in skeletal muscle in weaned pigs by regulating Akt, AMPKα and FOXO1 by Yulan Liu, Xiuying Wang, Weibo Leng, Dingan Pi, Zhixiao Tu, Huiling Zhu, Haifeng Shi, Shuang Li, Yongqing Hou and Chien-An Andy Hu in Innate Immunity
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their gratitude to the National Natural Science Foundation of China (31422053 and 31372318) and the Hubei Provincial Department of Education (T201508) for their financial support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
