Abstract
Dysregulated neutrophil extracellular traps (NETs) formation contributes to the pathogenesis of anti-neutrophil cytoplasmic Ab (ANCA)-associated vasculitis (AAV). Increasing evidence indicates that autophagy is involved in the process of NETs formation. In this study, we aimed to investigate whether ANCA could induce autophagy in the process of NETs formation. Autophagy was detected using live cell imaging, microtubule-associated protein light chain 3B (LC3B) accumulation and Western blotting. The results showed that autophagy vacuolization was detected in neutrophils treated with ANCA-positive IgG by live cell imaging. This effect was enhanced by rapamycin, the autophagy inducer, and weakened by 3-methyladenine (3-MA), the autophagy inhibitor. In line with these results, the autophagy marker, LC3B, showed a punctate distribution pattern in the neutrophils stimulated with ANCA-positive IgG. In the presence of rapamycin, LC3B accumulation was further increased; however, this effect was attenuated by 3-MA. Moreover, incubated with ANCA-positive IgG, the NETosis rate significantly increased compared with the unstimulated group. And, the rate significantly increased or decreased in the neutrophils pretreated with rapamycin or 3-MA, respectively, as compared with the cells incubated with ANCA-positive IgG. Overall, this study demonstrates that autophagy is induced by ANCA and promotes ANCA-induced NETs formation.
Introduction
Antineutrophil cytoplasmic Ab (ANCA)-associated vasculitis (AAV) includes microscopic polyangiitis (MPA), eosinophilic granulomatosis with polyangiitis (EGPA) and granulomatosis with polyangiitis (GPA). 1 ANCAs, the serological markers of AAV, are specific for the cytoplasmic constituents of neutrophils, in particular, proteinase 3 (PR3) and myeloperoxidase (MPO).
Neutrophil extracellular traps (NETs) were first reported in 2004 2 that were released by activated neutrophils in response to pathogenic bacteria, fungi,2,3 and also in response to pharmacological agents such as phorbol myristate acetate (PMA). 4 NETs consist of extracellular chromatin modified with various cytoplasmic and granule-derived proteins, such as MPO and PR3. The well-known function of NETs is pathogen trapping through limiting the diffusion of pathogens. 2 However, excessive NETs accumulation has been demonstrated to be associated with autoimmune disorders. 5 Recent studies showed that ANCA-induced NETs are involved in the pathogenesis of AAV, and a vicious cycle including ANCA and the regulation of NETs might be critically involved in the disease development.6–8
Autophagy is a key mechanism for cell homeostasis and survival, including damaged organelles or cellular proteins clearance and the essential nutrient supply by recycling organelles and cytosolic macromolecules. 9 Also, the autophagy pathway is involved in the pathogenesis of immunity and inflammation via pathogen clearance mechanisms that are mediated by immune cells, including neutrophils and macrophages. 10 Recently, several studies have shown that autophagy plays an important role in NETs formation. For instance, autophagy promotes NETs formation induced by the bacteria-derived peptide formyl-Met-Leu-Phe (fMLP), 11 the same as NETs formation stimulated with PMA or IL-1β. 12 In contrast, McInturff et al. found that autophagy negatively regulates the NETs release induced by LPS. 13 These reports indicate that the role autophagy plays in NETs formation varies depending on different stimulants for neutrophils. Therefore, in the current study, we investigated whether ANCA could induce autophagy and the role of autophagy played in the process of ANCA-induced NETs formation.
Material and methods
Reagents
TNF-α, 3-methyladenine (3-MA), PMA and rapamycin were purchased from Sigma. Anti-microtubule-associated protein light chain 3B (LC3B) Ab (no. 3868) was from Cell Signaling Technology and anti-LC3B Ab produced in rabbit (L7543) was purchased from Sigma. Cyto-ID Autophagy Detection kit was purchased from Enzo Life Science and 4′,6-diamidino-2-phenylindole (DAPI) was purchased from Zhongshan Golden Bridge Biotechnology (Beijing, China). The cell-permeable DNA dye Hoechst 33342, the cell-impermeable DNA dye Sytox green and goat anti-rabbit Alexa Fluor 488 Ab were from Invitrogen (Carlsbad, CA, USA).
Preparation of IgG
IgG was prepared according to the methods described previously.14,15 Healthy control IgG and ANCA-IgG were purified from plasma of healthy human volunteers and patients with PR3- or MPO-ANCA-positive primary small vessel vasculitis, respectively. Plasma was filtered through a 0.22 mm syringe filter (Gelman Sciences, Ann Arbor, MI, USA) and applied to a HiTrap Protein G column on an AKTA-FPLC system (GE Biosciences, South San Francisco, CA, USA).
Neutrophils isolation
Blood samples were collected from healthy volunteers and the neutrophils were isolated as previously described. 11 Briefly, human venous blood was drawn from healthy donors into EDTA, and then layered with Polymorphprep and Lymphoprep in the centrifuge tube, followed by centrifugation at 500 g for 30 min at room temperature (22℃). The lower layer containing neutrophils was collected and washed with PBS by centrifugation at 300 g for 5 min. To remove contaminating red blood cells, the pellet was re-suspended in red cell lysis buffer for 10 min on ice. After centrifugation at 200 g for 5 min, the neutrophils were collected and washed with PBS by centrifugation at 200 g for 5 min. Then the neutrophils were re-suspended in Roswell Park Memorial Institute (RPMI)1640 medium (phenol red-free) supplemented with 4% FBS. This research was approved by the ethics committee of the Peking University First Hospital and was in compliance with the Declaration of Helsinki.
Immunofluorescence microscopy
Isolated human neutrophils were analyzed by confocal microscopy as described previously. 16 In brief, human neutrophils on uncoated glass coverslips were placed in 24-well plates, pretreated with or without rapamycin (100 nM) or 3-MA (5 mM) for 30 min at 37℃. Then the cells were primed with TNF-α for 15 min and incubated with purified MPO-ANCA-positive IgG or PR3-ANCA-positive IgG (300 µg/ml) for up to 60 min at 37℃ in 5% CO2. Moreover, we used PMA (100 nM) in an incubator containing 5% CO2 at 37℃ for 1h as the positive control. Subsequently, cells were fixed with 4% paraformaldehyde at room temperature. They were permeabilized with methanol at −20℃ for 10 min. After three washing steps with PAB (PBS supplemented with 5 mg/ml BSA), cells were incubated with blocking buffer (PBS supplemented with 5 mg/ml BSA + 10% FBS) at 4℃ for 16 h. They were incubated with primary anti-LC3B Ab (1:200) (Cell Signaling Technology) for 1h at room temperature. After 10 washing steps with PAB (PBS supplemented with 5 mg/ml BSA), cells were incubated with 1:500 secondary goat anti-rabbit Ab coupled to Alexa 488 for one hour at room temperature, then after 10 washing steps with PAB, DAPI was added for another 10 min. Confocal images were captured with a Carl Zeiss confocal microscope (Zeiss, Oberkochen, Germany).
Live-cell imaging
The method of live-cell imaging detection was performed as previously reported. 17 Briefly, neutrophils were diluted to (5–8) × 105 cells/ml in a glass-bottom culture dish for live-cell observation (Thermo Scientific, Waltham, MA, USA) and incubated for 30 min at 37℃ in the presence of 4 µg/ml Hoechst 33342, and Cyto-ID. The cells were pretreated with or without rapamycin (100 nM) or 3-MA (5 mM) for 30 min. Then, TNF-α was used for priming. Subsequently, we incubated the neutrophils with MPO-ANCA-positive IgG or PR3-ANCA-positive IgG (300 µg/ml). The fluorescent signals were detected and imaged using a Carl Zeiss Workstation.
Western blot analysis
Western blot analysis was performed on cells treated for 60 min. At this time point, the most prominent autophagy marker LC3BII was observed after treatment with ANCA-positive-IgG. Purified human neutrophils were pelleted and re-suspended in lysis buffer supplemented with protease inhibitor, and cell debris was removed via high-speed centrifugation after incubating the lysate for 40 min on ice. Then, 25 µg of protein was separated on 12% SDS-PAGE gel. Subsequently, incubation of polyvinylidene difluoride (PVDF) membranes at 4℃ in an anti-LC3B Ab (1:1000) (Sigma) was performed for 12 h, followed by probing with a HRP-conjugated secondary Ab (1:5000) for 1h at room temperature.
Quantification of NETs formation
In this experiment, the percentage of neutrophil-releasing NETs was quantified through evaluating neutrophils displaying a nucleus that can be indicated by the cell-impermeable DNA dye Sytox Green (50 nM). In brief, the neutrophils were pretreated with or without 3-MA (5 mM) or rapamycin (100 nM) for 30 min, then cells were incubated with MPO-ANCA-positive IgG or PR3-ANCA-positive IgG (300 µg/ml) after being primed with TNF-α for 15 min. PMA, TNF-α, ANCA-positive IgG or control IgG was added to the other groups, respectively. Subsequently, the cells were fixed and seeded in black flat-bottomed 96-well plates. Then fluorescence signals of Sytox Green were measured respectively in a microplate fluorescence reader after 60 min and 180 min (TriStar Multimode Microplate Reader LB941, Berthold Technologies, Bad Wildbad, Germany).
Statistical analysis
Data are presented as means ± SD. Differences of quantitative parameters between groups were assessed using one-way analysis of variance (ANOVA). Statistical analyses were performed with GraphPad Prism 5 software and SPSS statistical software package (version 13.0, Chicago, IL, USA). Differences were considered significant if the P value was < 0.05.
Results
ANCA-positive IgG induces the typical features of autophagy
In the best-known form of autophagy, parts of the cytoplasm, intracellular organelles and long-lived proteins were sequestered within double-membrane vesicles called autophagic vacuoles or autophagosomes. These vacuoles were finally delivered to lysosomes for degradation.
18
Therefore, we tested whether ANCA-positive IgG induced the autophagy in the process of NETs formation through detecting the autophagic vacuoles using the specific dye Cyto-ID, along with the cell-permeable DNA dye Hoechst 33342 by direct live-cell imaging. Within 60 min of TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG stimulation, we observed autophagic vacuolization structures. To further confirm whether this was the structure of autophagosomes, we pretreated neutrophils with rapamycin, an inducer of autophagy through inhibition of the mammalian target of rapamycin (mTOR),
19
or the autophagy inhibitor 3-MA
20
before ANCA-positive IgG was added. We found that rapamycin-treated neutrophils exhibited more vacuolization structures compared with neutrophils treated with TNF-α plus ANCA-positive IgG. Pre-stimulation with the 3-MA diminished the observed structures (Figure 1(a) and (b)).
Autophagy was involved in ANCA-positive-IgG-induced NETs formation. Neutrophils were treated with MPO-ANCA-positive IgG (a) or PR3-ANCA-positive IgG (b). Rapamycin was used as an autophagy inducer and 3-MA was used as an autophagy inhibitor. Autophagy was detected by staining for DNA using the cell-permeable DNA marker Hoechst 33342 (blue) and for autophagic vacuolization using Cyto-ID (green).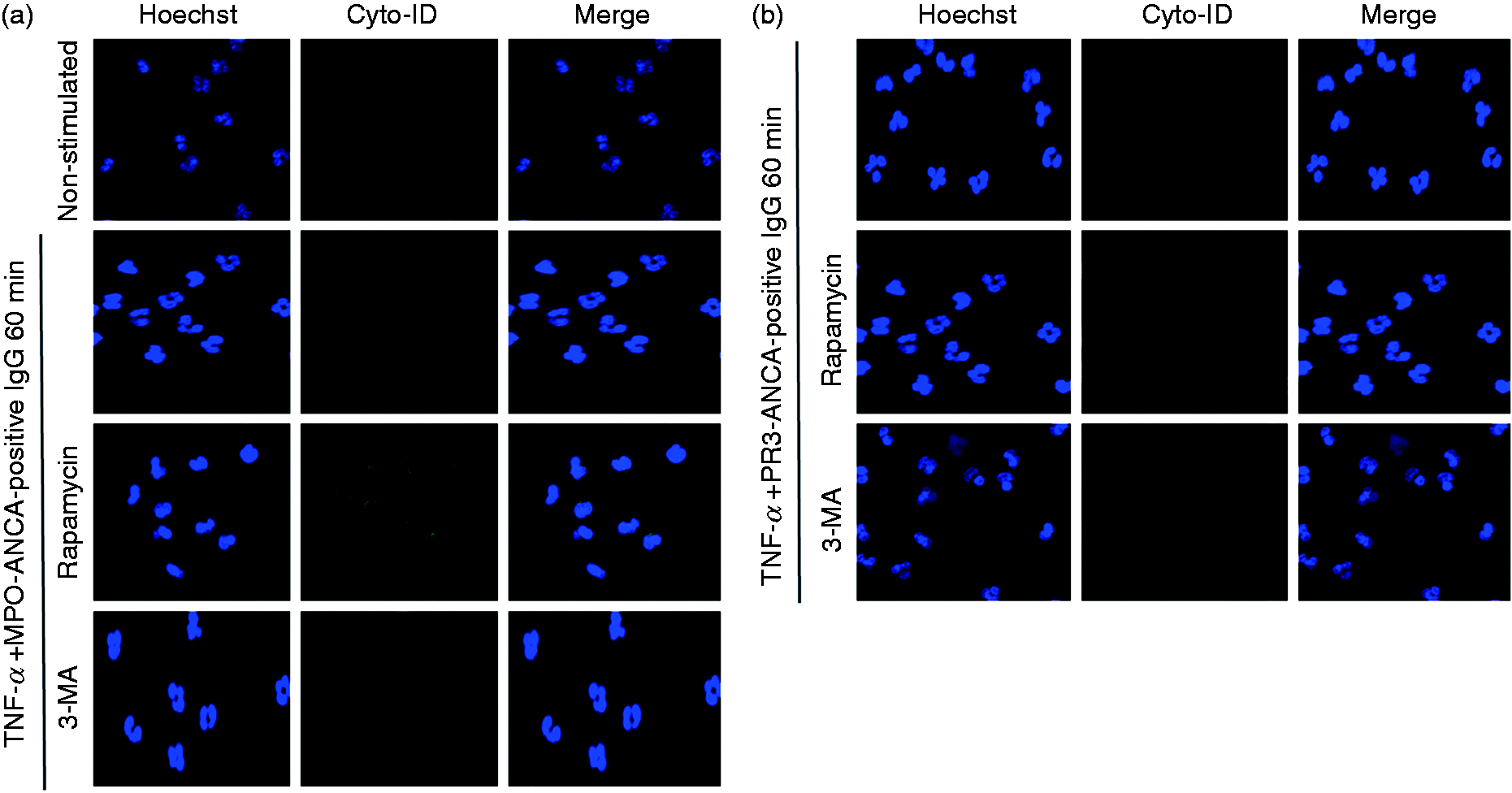
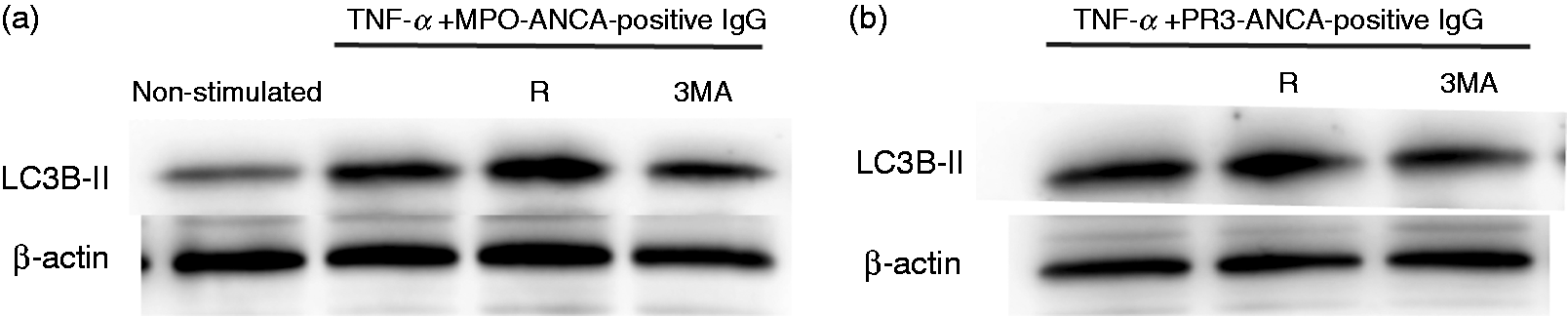
Moreover, the mechanism of autophagy in neutrophils stimulated with ANCA-positive IgG was monitored based on the localization of LC3B immunofluorescence confocal microscopy. Numerous studies have shown that LC3B can be regarded as a marker of the autophagosome, since it undergoes post-translational modification and associates with autophagic structures upon the initiation of autophagy.10,21,22 Autophagic vacuolization structures were observed in 60 min by live-cell imaging. Therefore, we also detected the localization of LC3B in 60 min. Our results showed that the immunofluorescence of punctate LC3B structures could be observed after being stimulated with TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG, which are typical for autophagy, suggesting autophagosome formation. Furthermore, we found that in the presence of rapamycin, accumulation of LC3B was further increased at 60 min post-addition of TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG. The blockade of PI3K by 3-MA led to a decrease in the fluorescent intensity of LC3B in stimulated neutrophils (Figure 2(a) and (b)). The ability of ANCA-positive IgG to accelerate autophagy was further detected via immunoblotting for LC3B. Consistently, LC3B levels, as compared with β-actin loading controls, increased with TNF-α plus ANCA-positive IgG stimulation. Moreover, this level could be increased by adding rapamycin and reduced by adding 3-MA (Figure 3(a) and (b)). Taken together, these results indicated that ANCA-positive IgG induced the formation of autophagy.
Autophagy was detected in the process of ANCA-positive-IgG-activated neutrophils by immunofluorescence staining. Immunofluorescence staining for LC3B in neutrophils treated with MPO-ANCA-positive IgG (a) or PR3-ANCA-positive IgG (b) for 60 min. Rapamycin was used as autophagy inducer and 3-MA was used as autophagy inhibitor. Autophagy was assessed based on staining for microtubule-associated protein LC3B (green) and DNA (blue). Autophagy was detected in the process of ANCA-positive-IgG-activated neutrophils by Western blotting. Immunoblotting for LC3B analysis in human neutrophils that were treated with or without 3-MA (5 mM) or rapamycin (100 nM) for 30 min, followed by stimulation with MPO-ANCA-positive IgG (a) or PR3-ANCA-positive IgG (b) after being primed with TNF-α. R: rapamycin.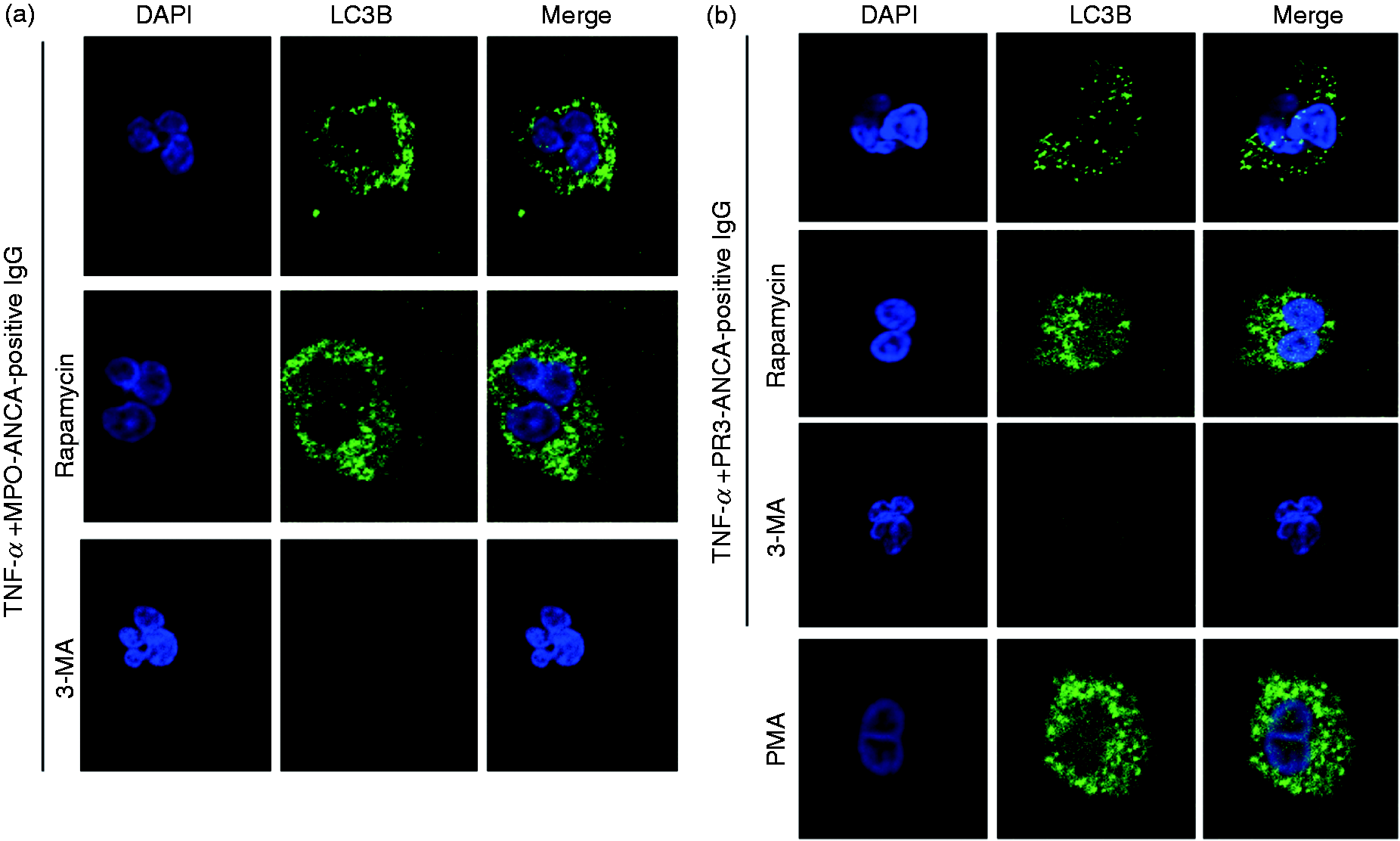
Inhibition of autophagy activity prevents ANCA-positive-IgG-induced NETosis
According to previous studies,
11
extracellular DNA release was caused by NETosis as opposed to apoptotic cell death. Then we detected the level of NETosis using the cell-impermeable DNA dye Sytox green by the microplate fluorescence reader to monitor the extracellular release of DNA from treated neutrophils. ANCA-positive IgG were prepared from four patients with active MPO-ANCA-positive vasculitis, two patients with active PR3-ANCA-positive vasculitis, and normal IgG was prepared from three healthy controls. After 60 min, in the presence of TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG, the percentage of NETosis was significantly higher than those treated with TNF-α plus IgG from healthy controls (5.80 ± 1.18% vs. 1.73 ± 0.67%, P = 0.003; 5.07 ± 1.22% vs. 1.74 ± 0.89%, P = 0.003, respectively). Pre-treatment with autophagy inhibitor 3-MA led to a significant reduction in the NETosis percentage compared with cells stimulated with TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG (2.70 ± 0.93 % vs. 5.80 ± 1.18%, P = 0.022; 2.25 ± 0.79% vs. 5.07 ± 1.22%, P = 0.001, respectively) (Figure 4(a) and (b)). After 180 min, in the presence of TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG, the percentage of NETosis was significantly higher than those treated with TNF-α plus IgG from healthy controls (12.51 ± 0.7% vs. 4.03 ± 0.96%, P < 0.001; 12.13 ± 1.95% vs. 5.51 ± 1.42%, P = 0.012, respectively) or those non-stimulated (12.51 ± 0.7% vs. 1.85 ± 1.23%, P < 0.001; 12.13 ± 1.95% vs. 2.94 ± 2.6%, P = 0.001, respectively), or those incubated with TNF-α (12.51 ± 0.7% vs. 3.85 ± 0.65%, P = 0.001; 12.13 ± 1.95% vs. 5.12 ± 3.28%, P = 0.008, respectively) or ANCA-positive IgG alone (12.51 ± 0.7% vs. 4.29 ± 1.01%, P = 0.001; 12.13 ± 1.95% vs. 4.38 ± 0.52%, P = 0.003, respectively). Pre-stimulation of neutrophils with rapamycin before the addition of TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG resulted in a significant increase of the NETosis percentage, compared with those treated with TNF-α plus ANCA-positive IgG (24.06 ± 0.76% vs. 12.51 ± 0.7%, P < 0.001; 28.77 ± 7.04% vs. 12.13 ± 1.95 %, P < 0.001, respectively), while pre-incubation with autophagy inhibitor 3-MA led to a significant reduction in the NETosis percentage compared with cells stimulated with TNF-α plus MPO-ANCA-positive IgG or PR3-ANCA-positive IgG (5.96 ± 1.64% vs. 12.51 ± 0.7%, P = 0.008; 5.23 ± 1.58% vs. 12.13 ± 1.95%, P = 0.009, respectively) (Figure 4(c) and (d)).
Increased autophagic activity facilitated NETs release. Human neutrophils were treated with or without 3-MA (5 mM) or rapamycin (100 nM) for 30 min, followed by stimulation with MPO-ANCA-positive IgG or PR3-ANCA-positive IgG after primed with TNF-α. Extracellular DNA (Sytox green) was obtained at 60 min ((a), (b)) and 180 min ((c), (d)) using the microplate fluorescence reader. Results of two time-points were obtained respectively from 12 independent experiments and presented as the mean percentage of Sytox-positive cells ± SD. *P < 0.05.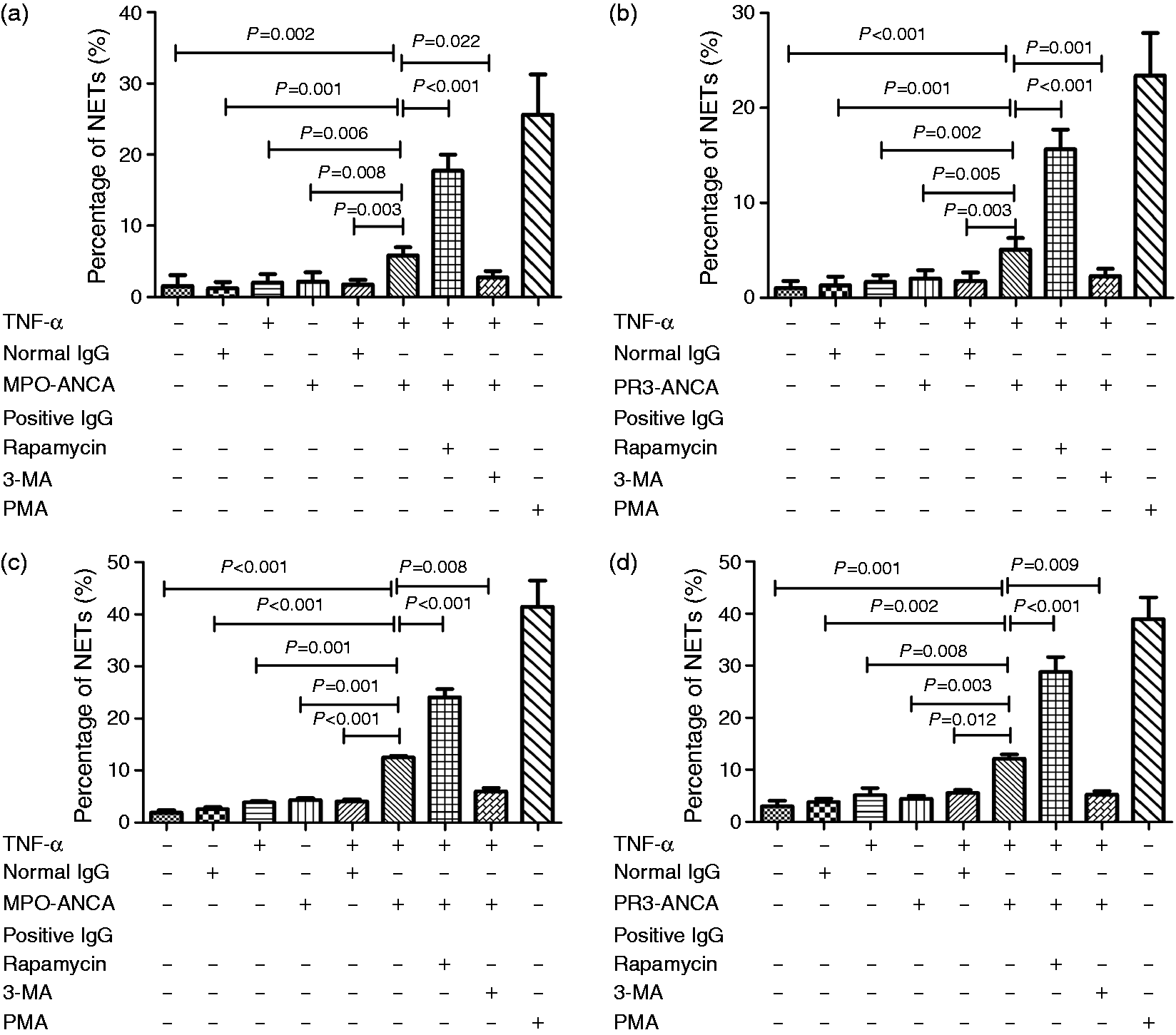
Overall, these data indicated that inhibition of autophagy activity prevents ANCA-positive IgG-induced NETosis.
Discussion
NETs are released by neutrophils in response to a wide range of stimuli, undergoing a process called NETosis. Recent studies demonstrated that ANCA-induced NETs formation triggers vasculitis and facilitates the autoimmune response against neutrophil constituents in AAV patients. 8 Autophagy is a key mechanism for cell homeostasis and survival, including damaged organelles or cellular proteins clearance and the essential nutrient supply by recycling organelles and cytosolic macromolecules. 9 Remijsen et al. found that autophagy was involved in the mechanism of NETs formation treated with PMA and promoted this process. 16 Consistently, some other studies found that autophagy played a positive role in the process of fMLP-induced or IL-1β-induced NETs formation.11,12 However, another recent study by McInturff et al. indicated that autophagy negatively regulated the NETs formation induced by LPS. 13 Therefore, it seems that whether autophagy could be induced and the role of autophagy played in NETs formation varies, depending on the stimulants for neutrophils.
MPO and PR3 were always identified as the major antigenic targets of ANCA.23–26 Subsequently, Kessenbrock et al. 8 reported that ANCA-IgG, which was specific for MPO and PR3, could induce primed human neutrophils to form NETs in vitro. In the current study, we provided evidence for induction of autophagy by MPO- and PR3-ANCA-positive IgG. NETs formation is inhibited when autophagy is blocked, as previously reported. 27 Tang et al. found that autophagy-related signaling participated in the mechanism of anti-lysosomal membrane protein-2 (LAMP-2)-Ab-induced NETs release. 17 Therefore, the current study confirmed and further extended Tang’s results, since the significance of anti-LAMP-2 Ab in AAV still remained controversial.28,29
To detect the occurrence of autophagy in the process of ANCA-positive-IgG-induced NETs formation, live-cell imaging for autophagy vacuolization was performed. Moreover, in this study, rapamycin was used as an autophagy inducer, whereas 3-MA was used as an autophagy inhibitor. We found that autophagy vacuolization was detected in neutrophils treated with MPO-ANCA-positive IgG or PR3-ANCA-positive IgG. This effect was enhanced by rapamycin, and weakened by 3-MA. We then used another two assays, the autophagy marker LC3B detection by immunofluorescence staining and Western blotting, for further study. Consistent with the results of live-cell imaging mentioned above, the typical feature of autophagy could be validated. Therefore, we suggested that autophagy could be induced by ANCA-positive IgG. To explore the potential role of autophagy in ANCA-positive-IgG-induced NETs formation, we tested NETs release via monitoring extracellular release of DNA from treated neutrophils at 60 min, which is the time-point that we detected autophagy, and 180 min, respectively. We found that in the presence of ANCA-positive IgG, the percentage of NETosis was significantly higher than those treated with IgG from healthy controls or those non-stimulated. Moreover, the ANCA-positive-IgG-induced NETs formation could be significantly enhanced by rapamycin, the autophagy inducer, and significantly suppressed by 3-MA, the autophagy inhibitor. Finally, we proposed that autophagy could facilitate the release of ANCA-positive-IgG-induced NETs.
In this study, we detected the involvement of autophagy in ANCA-positive-IgG-induced NETs formation via live cell imaging, immunofluorescence staining, Western blot and microplate fluorescence reader. However, since all the assays were performed in vitro, these results should be further confirmed by in vivo studies.
Conclusions
The results from our current studies indicate that autophagy is induced by ANCA and promotes NETs formation.
Footnotes
Acknowledgments
We are grateful to all the healthy volunteers and patients who participated in this study. We thank our associate Ms Ping Hou for providing assistance in the live cell imaging and confocal microscopic.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by three grants from the National Natural Science Fund (grant numbers 81425008, 81321064 and 81300599) and the grant from National key research and development program (No. 2016YFC0906102).
