Abstract
Innate γδ T cells expressing Vγ6 produce IL-17A at an early stage following infection with
Introduction
Conventional Th17 cells are exported from the thymus as naive cells, and then acquire effector functions, and produce IL-17A upon Ag encounter in the periphery. However, some γδ T cells are functionally committed to be IL-17A-producing effector cells within the thymus. Therefore, these γδ T cells are also called ‘innate γδ T cells’.1–5 Innate γδ T cells are disproportionately distributed in mucosal tissues such as the uterus, lung and peritoneal cavity,3–5 and spontaneously produce IL-17A at an early stage following infection with
Various exogenous signals, via the common gamma (γc) chain receptor (R), play an important role in regulating γδ T-cell IL-17A or IFN-γ production. The IL-15/CD125 (IL-2Rβ) axis is important for γδ T-cell IFN-γ production, 3 while the IL-7/CD127 (IL-7Rα) axis is critical to IL-17 A production.14,15 IL-21 is a pleiotropic γc cytokine secreted by activated CD4+ T and NKT cells. The IL-21 R is widely expressed on various cell types within the immune system, including macrophages, dendritic cells, NKT, CD4+ and CD8+ T cells, and B cells.16–22 The widespread lymphoid distribution of IL-21 R leads to pleiotropic actions of IL-21 in the innate and adaptive immune responses, including host defense against infection, antitumor activity, and pathogenesis of inflammatory diseases such as autoimmunity and allergic diseases.20–22
Do et al. reported that IL-21 was dispensable for γδ T-cell IL-17 A expression in lymph nodes (LN),
23
while Moser et al. reported that IL-21R signaling suppressed the IL-17A-producing γδ T-cell response in lung after influenza A virus infection.
24
Thus, the role of the IL-21/IL-21R axis in innate γδ T-cell response remains obscure. In this study, we examined the role of IL-21 in the regulation of IL-17A- producing innate γδ T-cell response following BCG infection, using IL-21R knockout (KO) mice and IL-21-producing recombinant BCG mice (rBCG-Ag85B-IL-21), which we constructed. We found that IL-17A-producing Vγ1−Vγ4− γδ T cells in rBCG-Ag85B-IL-21-inoculated mice significantly decreased, whereas they increased in BCG inoculated
Materials and methods
Mice
Age- and sex-matched C57BL/6 (B6) male mice were purchased from Japan KBT Inc. (Tosu, Japan).
Microorganisms
Lyophilized
Detection of IL21-Ag85B fusion protein
rBCG-Ag85B-IL-21, rBCG-Ag85B or BCG were lysed in lysis buffer for 30 min or cultured in 7H9 medium for 1 wk. After centrifugation, supernatants were harvested. Cell lysate or supernatant protein quantification was performed using Ultrospec 3300 pro (Amersham Biosciences, GE Healthcare Biosciences, Piscataway, NJ, USA). Samples were diluted to the same amount of protein, separated by SDS-PAGE, and proteins blotted and detected with anti-IL-21 mAbs, (Biolegend, San Diego, CA, USA). Culture supernatant IL-21 was measured using a DuoSet ELISA (R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s instructions.
Flow cytometry
Single cells were isolated from PECs as previously described. 3 Briefly, PECs were obtained by lavage with 5 ml HBSS. After washing, PECs were pre-incubated with an FcRγ-blocking mAb (CD16/32, clone 2.4G2; BD Biosciences, San Jose, CA, USA) for 20 min at 4℃ to prevent Ab binding to the FcR. Cells were then stained with various combinations of mAbs. We purchased the following mAbs: anti-CD11b-fluorescein isothiocyanate (FITC) (clone M1/70), anti-NK1.1-phycoerythrin (PE) (clone PK136), anti-MHC class II (clone M5/114.15.2), anti-CD3e-APC-Cy7 (clone 145-2C11), anti-CD4-peridinin chlorophyll protein complex (PerCP) and allophycocyanin (APC) (clone RM4-5), anti-CD8α-PerCP and FITC (clone RM53-6.7),), anti-Gr1-PE (clone RB6-8C5) (all BD Biosciences), anti-CD45R-APC (clone RA3-6B2), anti-F4/80-APC (clone BM8), anti-MHC class II-PerCP-eFluor 710 (clone M5/114.15.2), anti-TCRβ-PE (clone H57-597) (all eBioscience, San Diego, CA, USA), anti-CD3e-PE (clone 145-2C11), anti-TCRγδ-APC (clone GL3), anti-TCRγδ-BrilliantViolet421 (clone GL3), anti-TCRγδ-PE/Cy7 (clone GL3), anti-TCR Vγ1-PE and FITC (clone 2.11), anti-TCR Vγ4-PE and FITC (clone UC3-10A6), and anti-IL-21R-PE (clone 4A9) (all Biolegend). Stained cells were analyzed on a FACSCalibur and a FACSVerse flow cytometer (BD Biosciences). The data were analyzed using CellQuest (BD Biosciences) and FlowJo software (Tree Star, Ashland, OR, USA). γδ T-cell apoptosis was determined by staining with Annexin V–APC conjugate according to the manufacturer’s instructions (eBioscience).
Intracellular cytokine staining
Freshly isolated PECs were re-suspended in RPMI 1640 containing 10% fetal bovine serum (FBS), 100 U/ml penicillin, 100 µl/ml streptomycin and 50 µm 2-ME. PECs were cultured for 24 h in 96-well plates in the presence or absence of rIL-21 (25 or 50 ng/ml; R&D Systems) and further incubated without any stimulation for indicated time at 37℃ and 5% CO2. Brefeldin A (BFA) (Sigma-Aldrich) (10 µg/ml) was added for the last 4 h of culture and the cells washed with FACS buffer and surface stained with various combinations of mAbs for 30 min at 4℃. Intracellular staining was then performed according to the manufacturer’s instructions (BD Biosciences). Briefly, 100 µl Cytofix/Cytoperm solution (BD Biosciences) was added to the cell suspension, mixed and the mixture placed on ice for 20 min at 4℃. Fixed cells were washed twice with 500 µl of Perm/Wash buffer (BD Biosciences) and stained intracellularly with anti-IL-17 A-PE (clone TC11-18H10), anti-IFN-γ-FITC (clone XMG1.2) (both BD Biosciences), anti-IL-17 A-PerCP-Cy5.5 (clone eBio17B7; eBioScience) and anti-Bim-PE (clone C34C5; Cell Signaling, Danvers, MA, USA) for 30 min at 4℃.
Statistics
Statistical significance was evaluated by Student’s
Results
Kinetics of IL-17 A-producing γδ T cells in the peritoneal cavity of il21r−/− mice after BCG inoculation
We examined the kinetics of γδ T cells producing IL-17 A in Kinetics of peritoneal γδ T cells in 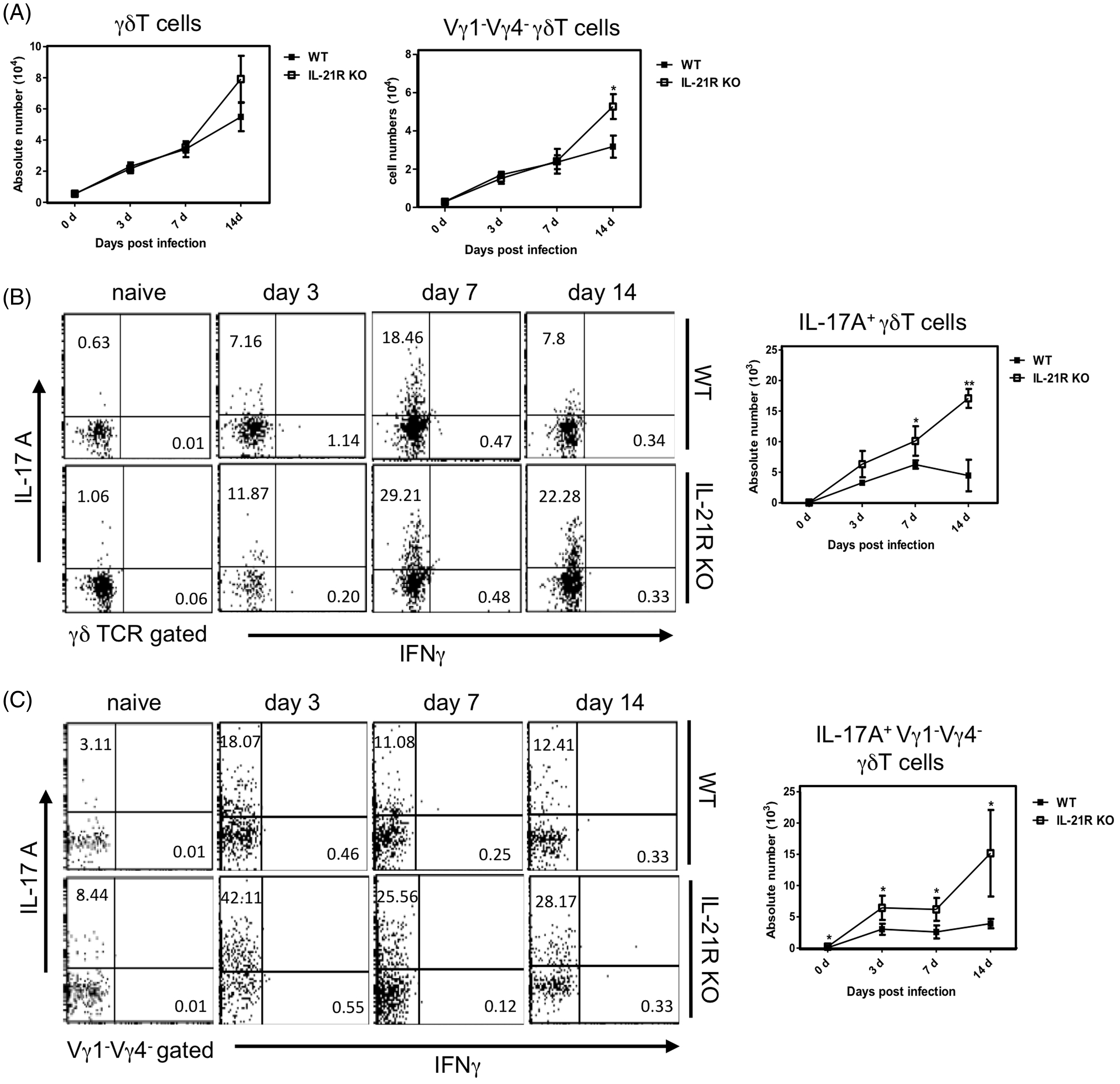
Detection of Ag85B-IL21 fusion protein from rBCG-Ag85B-IL-21
We constructed an rBCG expressing Ag85B-IL-21 fusion protein using the pNN2-mIL-21-Ag85B vector (Figure 2A). Lysates or culture supernatants of rBCG-Ag85B-IL-21, rBCG-Ag85B or BCG were blotted and reacted with anti-IL-21 mAbs to detect Ag85B-IL-21. Protein was detected at ∼47 kDa in the cell lysates and culture supernatant of the rBCG-Ag85B-IL-21 lane but not in the rBCG-Ag85B or the BCG lanes (Figure 2B). Thus, Ag85B-IL-21 was confirmed in rBCG-Ag85B-IL-21. rBCG-Ag85B-IL-21 and rBCG-Ag85B were cultured in 7H9 medium for 6 wk. Notably, IL-21 production peaked during the second wk, as determined from the rBCG-Ag85B-IL-21 culture supernatant (Figure 2C). Together, these data suggest rBCG-Ag85B-IL-21 was successfully constructed.
Construction of the mIL-21 expression plasmid. (A) Construction of pNN2-Ag85B-mIL-21. (B) Bacteria were cultured in 7H9 medium for 1 wk and supernatants harvested after centrifugation. After supernatant and lysate protein quantitation, samples were separated by SDS-PAGE, then blotted and reacted with anti-IL-21 mAb. Arrows indicate Ag85B-IL-21 fusion protein at ∼47 kDa. (C) The concentration of IL-21 in culture supernatants at the indicated days after 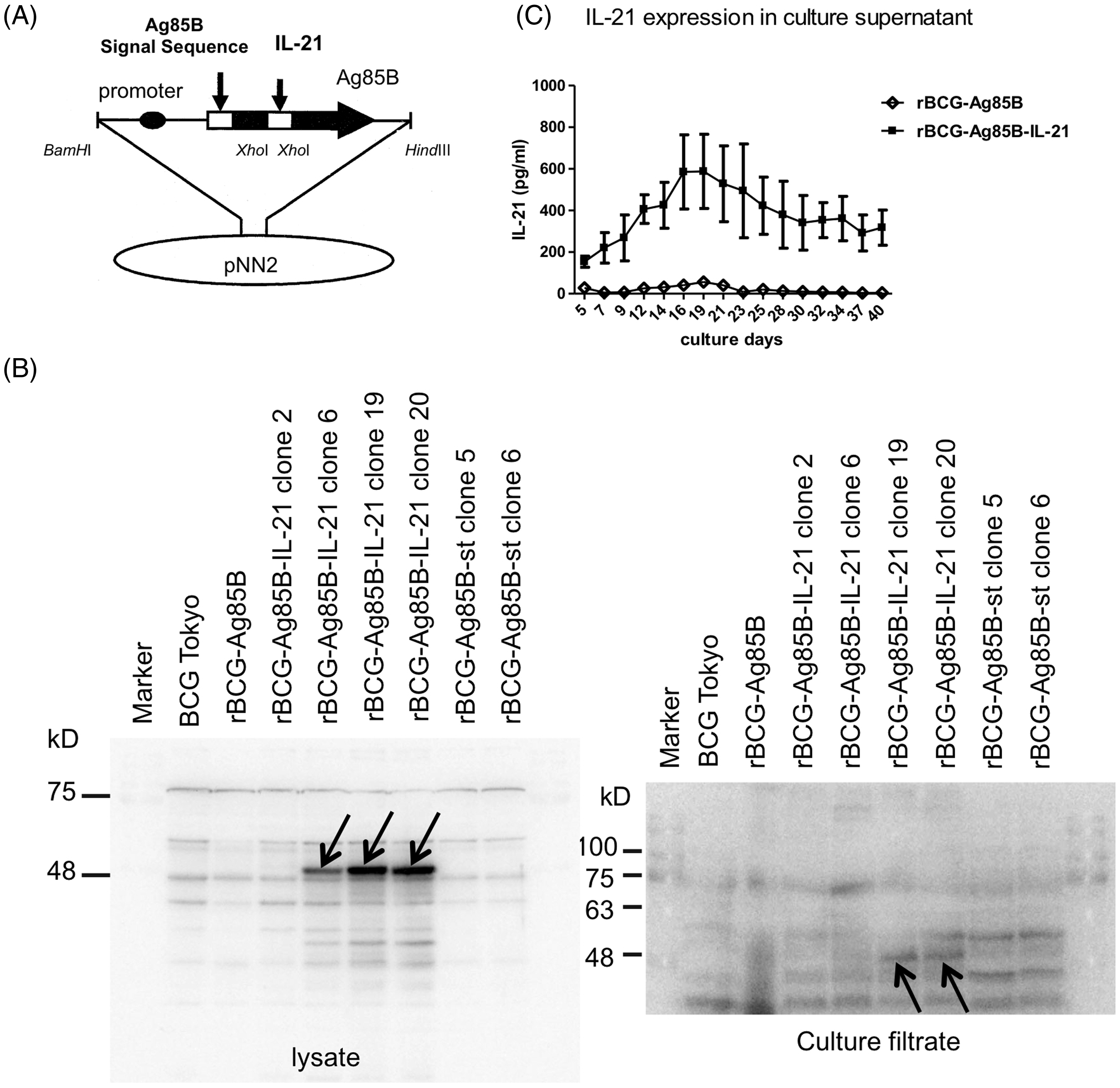
Kinetics of PEC IL-17A-producing γδ T cells in rBCG-Ag85B-IL-21-inoculated mice
We examined the kinetics of bacterial growth and PECs in the peritoneal cavity of WT mice at the early stage after i.p. inoculation with 2 × 106 rBCG-Ag85B-IL21 or rBCG-Ag85B. No obvious differences were observed in the number of bacteria in the peritoneal cavity between rBCG-Ag85B-IL-21- and rBCG-Ag85B-inoculated mice (Figure 3A). However, neutrophil number was significantly higher on d 7 and 14 after rBCG-Ag85B inoculation than after rBCG-Ag85B-IL-21 inoculation (Figure 3B, C; Figure S1). There was no difference in the number of macrophages (CD11b+ Gr-1low F4/80high) between rBCG-Ag85B-IL-21- and rBCG-Ag85B-inoculated mice (Figure 3C).
Kinetics of bacterial growth and PECs in the peritoneal cavity of WT mice after rBCG-Ag85B-IL-21 inoculation. WT mice were inoculated i.p. with 2 × 106 CFU of rBCG-Ag85B-IL-21 or rBCG-Ag85B. (A) The number of bacteria recovered from the peritoneal cavity of inoculated mice was determined on the indicated days. (B) Flow cytometry analysis of peritoneal neutrophil (CD11b+ Gr-1high F4/80low) kinetics and total numbers from inoculated mice. PECs were harvested on the indicated days after rBCG-Ag85B-IL21 or rBCG-Ag85B inoculation and surface stained with various mAbs. (C) Kinetics of peritoneal neutrophils shortly after rBCG-Ag85B-IL21 or rBCG-Ag85B inoculation. Neutrophil (CD11b+ Gr-1high F4/80low) number was calculated by multiplying the total PEC number by the percentages of each PEC subset. Data are represented as the mean ± SD ( Kinetics of peritoneal γδ T cells at an early stage of rBCG-Ag85B-IL21 or rBCG-Ag85B inoculation. PECs were harvested on the indicated days and surface stained with mAbs. Kinetics of peritoneal γδ T cells (A) or peritoneal Vγ1−Vγ4− γδ T cells (C) at the early stage rBCG-Ag85B-IL21 or rBCG-Ag85B inoculation. The absolute number was calculated by multiplying the total PEC number by the percentages of each PEC subset. (B) Data are represented as the mean ± SD (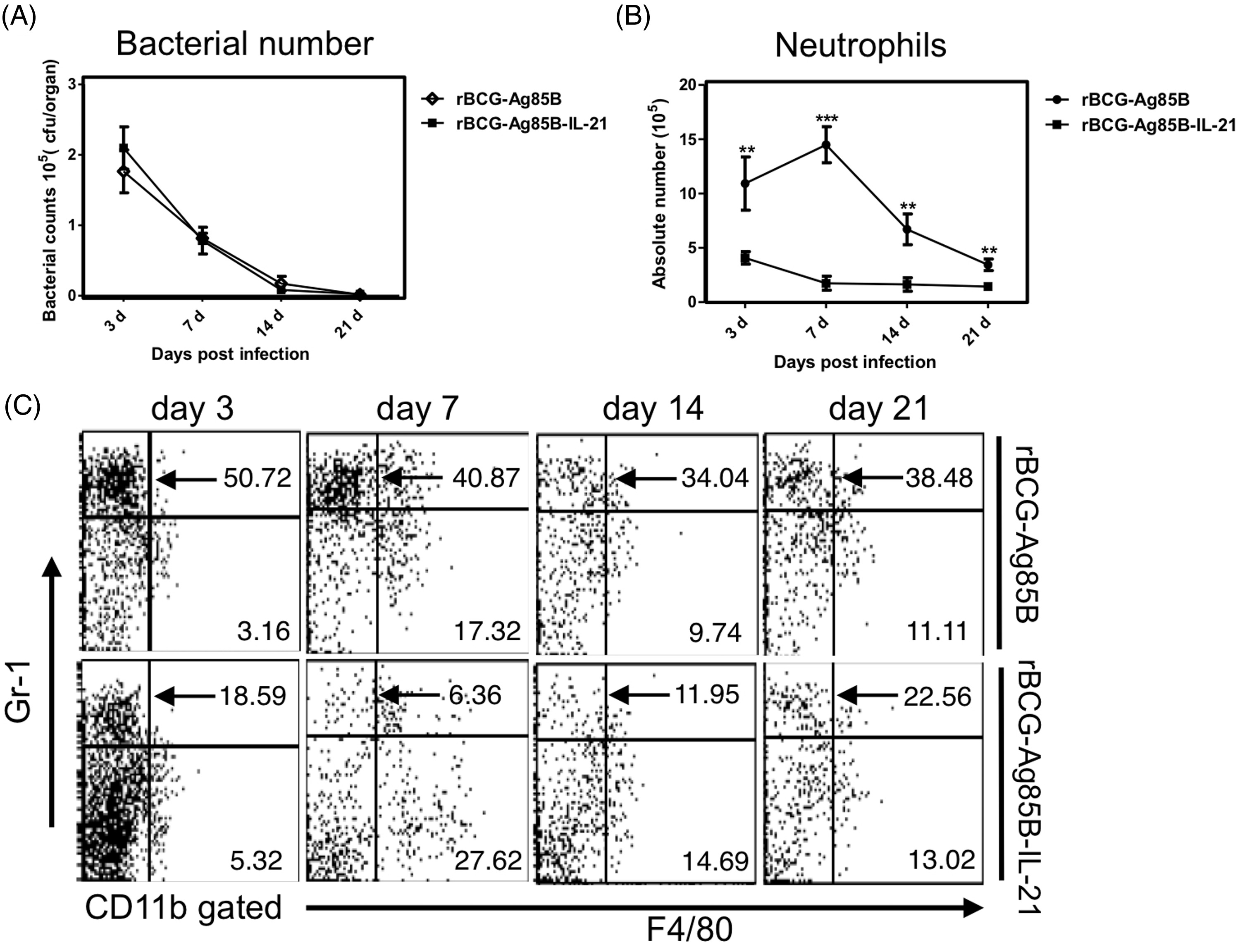
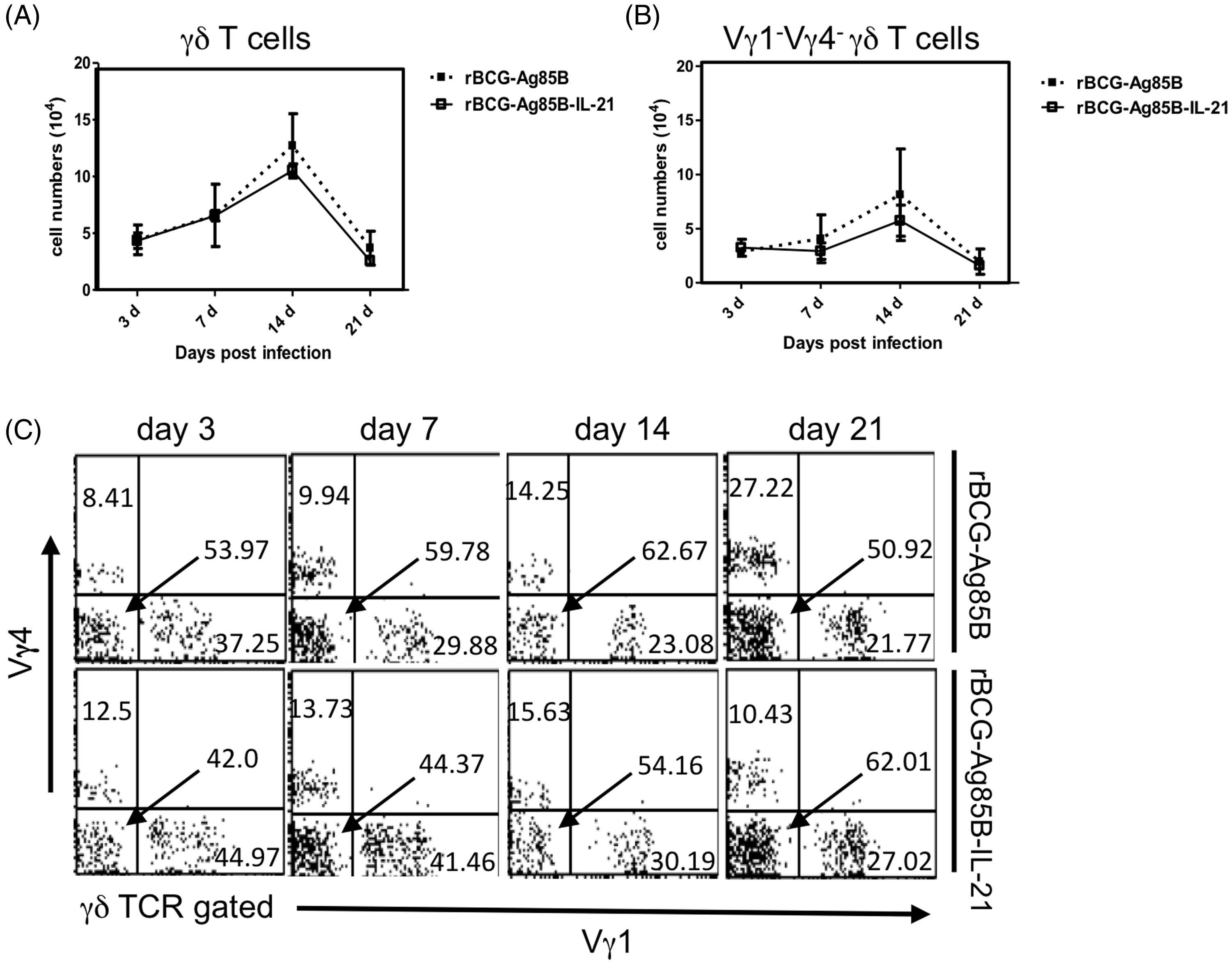
We next examined the kinetics of innate IL-17A-producing γδ T cells after rBCG-Ag85B inoculation, by intracellular cytokine flow cytometory analysis without stimulation. There were also no significant differences in the absolute number of γδ T cells and Vγ1−Vγ4− γδ T cells between rBCG-Ag85B-IL-21- and rBCG-Ag85B-inoculated mice on d 3, 7 and 14 (Figure 4A–C; Figure S2). Innate γδ T cells preferentially appear at an early stage after BCG inoculation, detectable by intracellular cytokine flow cytometry analysis without Kinetics of IL-17A+ γδ T cells shortly after rBCG-Ag85B-IL-21 or rBCG-Ag85B inoculation. PECs were harvested on the indicated days, cultured with BFA for the last 4 h, stained intracellularly for IL-17A or IFN-γ and analyzed by flow cytometry. Kinetics of IL-17A+ γδ T cells (A) and IL-17A+ Vγ1− Vγ4− γδ T cells (B) in PECs during early stage rBCG-Ag85B-IL-21 or rBCG-Ag85B inoculation. Data are represented as the mean ± SD (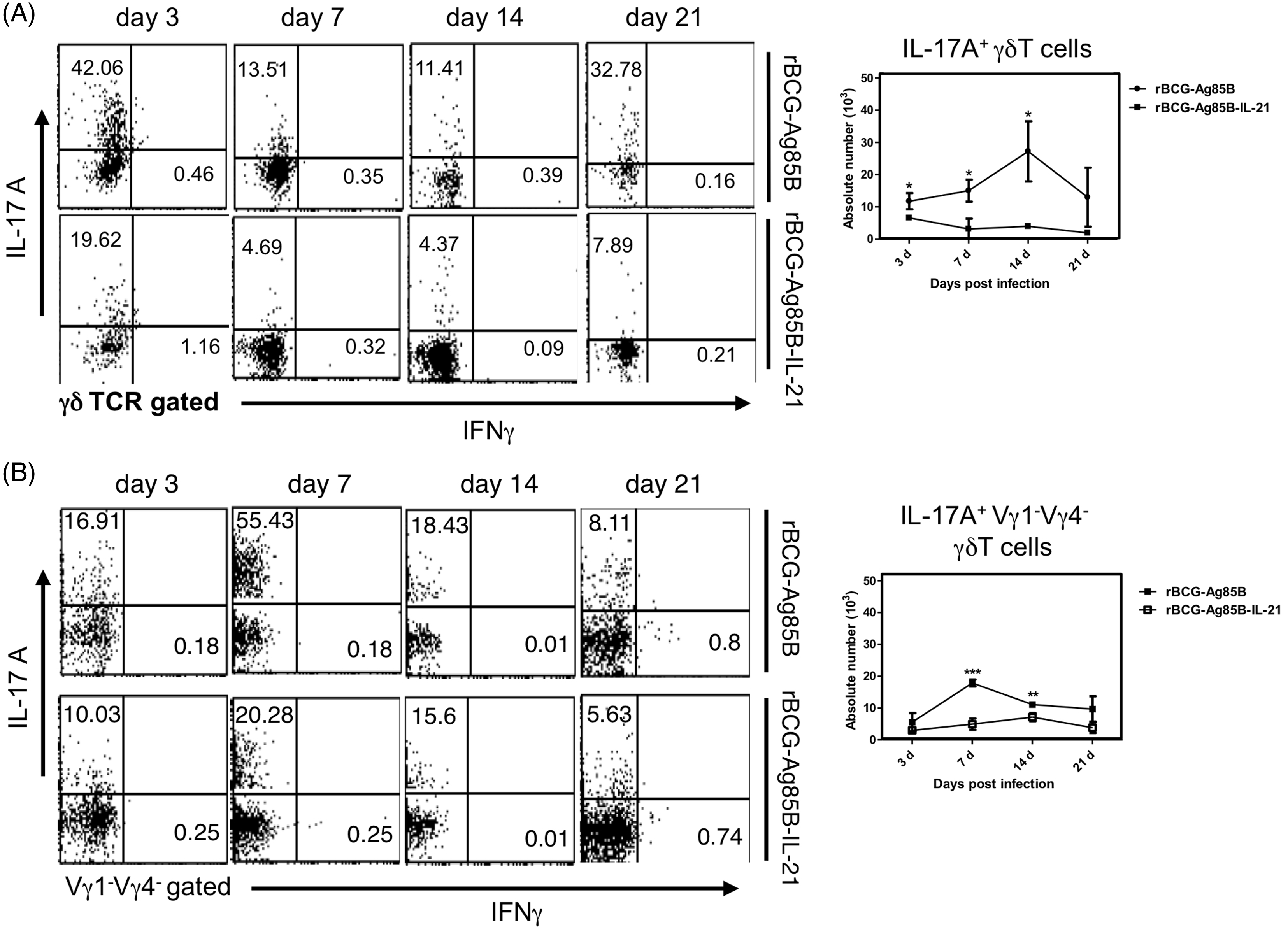
Effect of IL-21 on apoptosis of IL-17A producing γδ T cells in vitro
To further examine how IL-21 suppresses the response of IL-17A-producing γδ T cells, we examined the effect of rIL-21 on IL-17A-producing γδ T cells Effect of exogenous IL-21 on IL-17A+ γδ T cells 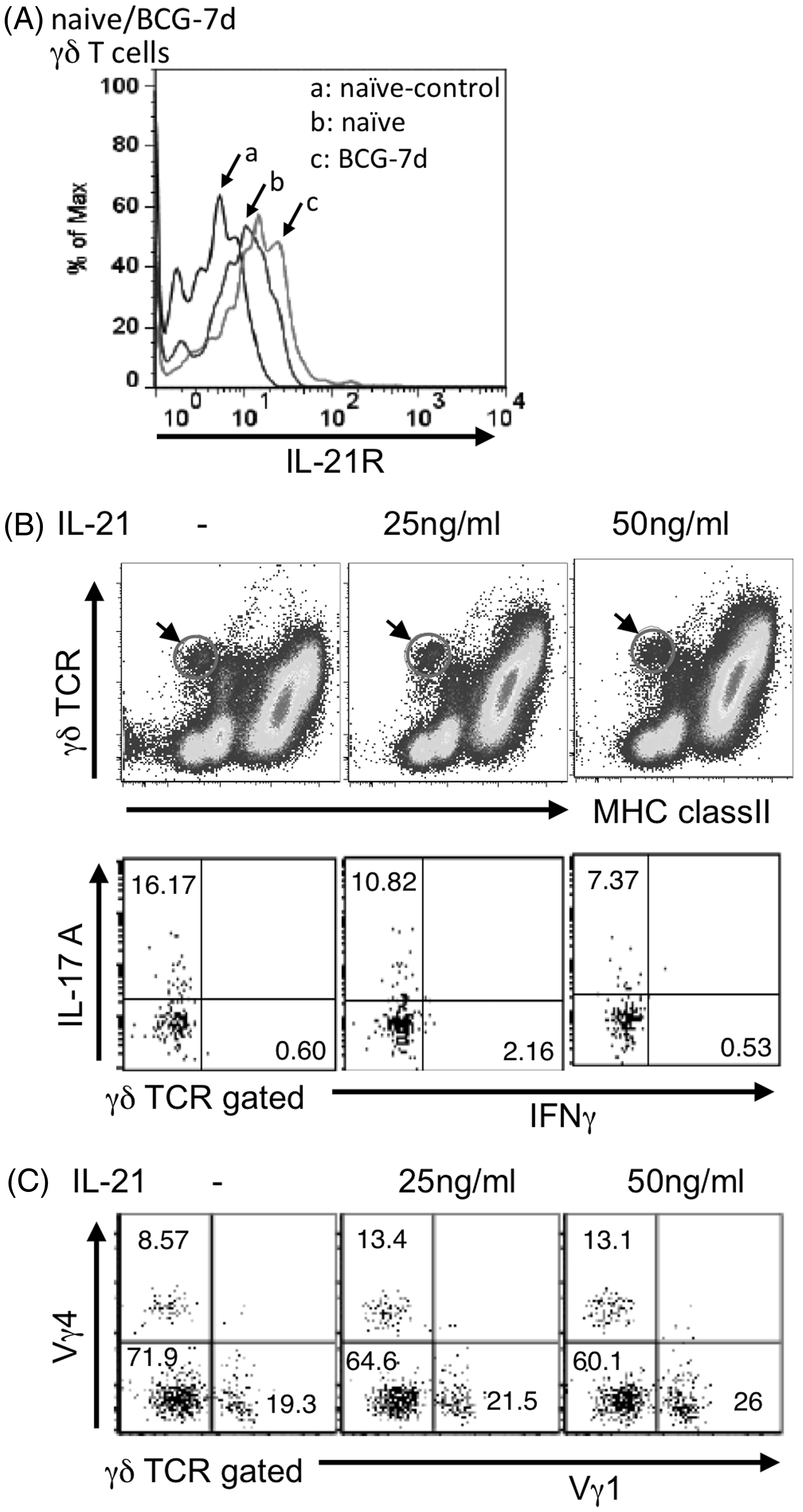
IL-21 induces apoptosis of some lymphoid and myeloid cells.
28
To further examine whether IL-17A-producing cells underwent apoptosis upon IL-21 stimulation, we used IL-17A-reporter ( Effect of exogenous IL-21 on apoptosis of IL-17A+ γδ T cells 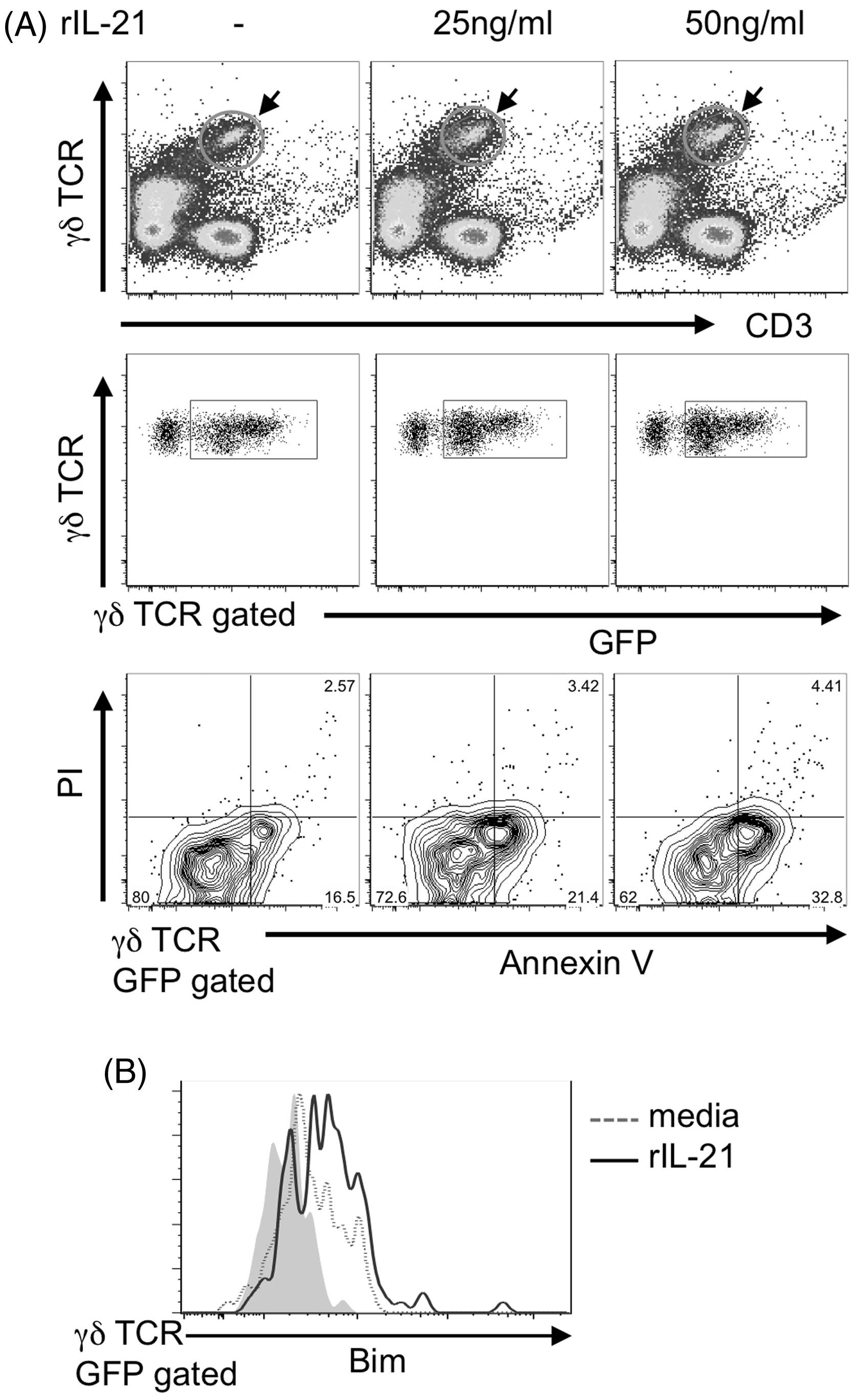
Discussion
We have shown that the IL-21/IL-21R plays an important role in regulating innate IL-17A-producing γδ T cells after BCG infection. The role of IL-21 in regulating IL-17A-producing T cells is controversial. Several lines of evidence suggest IL-21 plays a critical role in the expansion of IL-17A-producing αβ T (Th17) cells, although it is not required for the differentiation of Th17 cells from naive CD4+ T cells.29,30 In contrast, Ertelt et al. reported that IL-21 suppressed IL-17A production by
The mechanism whereby IL-21R signaling induces apoptosis of IL-17A-producing γδ T cells remains to be determined. IL-21R is composed of two subunits, IL-21R and Cγ.
19
IL-21R activates Jak1 and Jak2, causing phosphorylation, dimerization and nuclear translocation of STAT3, and, to a lesser extent, STAT1, STAT5a and STAT5b.19–22 IL-21 induces B-cell apoptosis through a Bim- and caspase-dependent pathway, although it stimulates B cells in the context of a co-stimulatory T-cell signal.17,32,33 Similarly, IL-21 induces NK cell apoptosis, although it co-stimulates cytokine-activated NK cells.34,35 Recently, it has been reported that IL-21 induces apoptosis of conventional dendritic cells via a STAT3 and Bim-dependent mechanism.
36
Consistent with these reports, our present study suggests that IL-21 reduces IL-17A-producing γδ T cells by apoptosis via Bim expression. An important finding in the present study is that only Vγ1−Vγ4− γδ T cells were affected by IL-21. We previously reported that the Vγ1−Vγ4− subset expressing Vγ6/Vδ1 mRNA in the peritoneal cavity produced IL-17A after BCG infection without
γδ T cells are innate sources of IL-17A, closely linked to the recruitment of neutrophils after bacterial infection, including
Conclusion
We have demonstrated that the number of IL-17A-producing γδ T cells, particularly Vγ1−Vγ4−γδ T cells, significantly decreased after inoculation with rBCG-Ag85B-IL-21, while IL-17A-producing Vγ1−Vγ4−γδ T cells increased in
Footnotes
Acknowledgements
We thank Akiko Yano, Miki Kijima and Yumiko Kitada for their secretarial assistance and all the members of our laboratory for helpful discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid (25670213; 26293098) from the Japan Society for the Promotion of Science, Yakult Bioscience Foundation (Y.Y.), and the Takeda Science Foundation (Y.Y.).
