Abstract
Amphotericin B (AmB) is a commonly used antifungal drug, with well-documented effects on cellular immune responses. We determined that AmB-stimulated γδ T-cell activation and proliferation in vitro at very low concentrations. AmB also enhanced IFN-γ production by NK cells in combination with IL-18. AmB had a greater effect on IFN-γ production in cells isolated from very young animals. Although innate immunostimulatory aspects of AmB have been defined, AmB has not been extensively applied in non-fungal infection settings. Given that γδ T cells are increased and activated in Salmonella infection in cattle, we assessed the effects of AmB in protection from Salmonella enterocolitis in calves. One injection of AmB, at approximately one-tenth of the concentration used in human patients to counter fungal infection, or saline control, was delivered intravenously to calves prior to infection with Salmonella. This single injection caused no adverse effects, reduced disease symptoms from Salmonella enterocolitis and significantly reduced Salmonella bacteria shed in feces of infected animals. Our findings suggest that AmB may be an inexpensive and readily available prophylactic approach for the prevention of bacterial infection in calves.
Introduction
Amphotericin B (AmB) is a polyene antimicrobial commonly used to treat systemic fungal infections. Early studies indicated that AmB was not simply affecting the fungal cells, but also had immunomodulatory effects.1–3 AmB is delivered at fairly high i.v. doses, and is associated with adverse reactions, such as fever and chills.4–8 AmB-induced changes in expression of TNF-α, IL-6 and IL-1 receptor agonist have been proposed to be the reason for these adverse reactions.6,9,10 The drug also augments macrophage phagocytosis and antibacterial capabilities.11–13 For example, the drug primes macrophages for the release of higher amounts of TNF-α, reactive oxygen metabolites and NO.13–17 In murine macrophages AmB increased nitrite production, especially in the presence of IFN-γ. 17 AmB enhances the ability of human alveolar macrophages to kill Aspergillus fumigatus in vitro, 18 and augments TNF-α production following priming with IFN-γ. 14 TLR2, TLR4, CD14 and MyD88 have each been implicated in AmB recognition and signaling, but their use may be concentration-, formulation- and tissue-dependent.19,20 It seems clear that AmB is not only an effective antifungal, but it also modulates innate macrophage responses.
In addition to macrophages, AmB also affects lymphocytes and was shown to enhance the proliferation of a specific subset of lymphocytes. 21 AmB and other polyene antibiotics can act as mitogens for B lymphocytes and can induce polyclonal Ab production from these cells.21–24 AmB has also been described as stimulatory to T cells.2,23,25 However, there is also some evidence that AmB suppresses T- and B-cell activity, at high doses in particular.2,26–30 Though both the immunomodulatory and fungicidal effects of AmB have been widely documented, AmB has not been extensively tested in in vivo models of bacterial infection.
Here, we have further defined the immunomodulatory properties of AmB and determined that AmB had stimulatory effects on γδ T cells. γδ T cells are found in high numbers in circulation in young animals, including humans. In bovine calves they can comprise up to 50% of circulating lymphocytes, and are activated during Salmonella enterica serovar Typhimurium (ST) infection. 31 γδ T cells are a major source of innate IFN-γ and have critical protective roles in a number of infectious disease contexts. As such, we sought to identify specific agonists that stimulate this critical protective subset. We determined that AmB has a potent activation effect on γδ T cells at low concentrations. At low nanogram concentrations, AmB induced increased expression of activation markers and proliferation of γδ T cells from cattle, humans and mice in vitro. In addition to activating γδ T cells, AmB also augmented expression of IFN-γ by NK cells in the presence of IL-18. Interestingly, AmB had a greater effect on cells derived from very young animals. These novel effects on innate lymphocytes have not been described for AmB, and could contribute to the documented immune alterations induced by AmB.
Considering the responses of γδ T cells to ST infection, 31 the importance of IFN-γ and the high incidence of this disease in calves, we applied low-dose AmB in vivo to protect calves against ST-induced disease and bacterial shedding. One dose of AmB was delivered intravenously at approximately one-tenth of the dose used for antifungal effects in human patients, and calves had no measurable adverse effects. After 24 h, calves were infected orally with ST. Compared with calves that received saline-only injections, calves injected with AmB exhibited considerably fewer disease symptoms, including less fever and significantly fewer bacteria shed in feces. This inexpensive and already approved drug could provide substantial protection to calves at risk for ST and other early life infections. In addition to protecting the individual animal from disease symptoms, entire farm or ranch operations are likely to benefit from decreased bacterial shedding resulting from AmB treatment. Furthermore, enhanced innate immunity can non-specifically protect against a broad spectrum of infectious insults. As young cattle are prone to infection from various agents, treatment with low-dose AmB may confer protection from infection with other agents, as well as Salmonella.
Materials and methods
Preparation of PBMCs
Whole blood was drawn from newborn Holstein bull calves (<12 h old), older Holstein bull calves (1–3 mo old) and adult (>2 yr old) Holstein cows into sodium heparin tubes (Becton Dickinson, Franklin Lakes, NJ, USA). Histopaque 1077 (Sigma-Aldrich, St. Louis, MO, USA) was used to separate the leukocytes from whole blood, as per the manufacturer's instructions, as previously described. 32 Cell preparations were maintained in X-VIVO 15 medium (Cambrex; East Rutherford, NJ, USA) at 1 × 106 cells/ml at 37℃, 10% CO2 in the presence of known or test agonists, or medium only as controls for 24 h, unless otherwise indicated. Samples from a minimum of three calves were obtained and analyzed per experiment. Cells from human subjects were derived from peripheral blood and similarly purified using Histopaque 1077. Mouse spleens were collected from TCR-α–/– mice, under aseptic conditions, and spleens were homogenized. This strain has increased numbers of γδ T cells, facilitating their analyses. 33 Red blood cells were water lysed before culturing cells in X-VIVO media. All experiments were performed in accordance with and approved by the institutional animal care and use committee of Montana State University, and the use of human subjects was approved by the institutional review board of Montana State University.
Preparation of AmB formulations
Commercially available sources of AmB were screened for their ability to stimulate γδ T cells. A water-soluble deoxycholate source for use in cell culture (Cat# A2942; Sigma-Aldrich) performed better than other commercial sources and was thus chosen for these studies.
Endotoxin screening
To analyze the concentration of endotoxin that may have been present in the preparations of AmB, a kinetic turbidimetric Limulus amebocyte lysate (LAL) (Associates of Cape Cod; East Falmouth, MA, USA) assay was performed as per the manufacturer’s instructions. AmB was diluted to concentrations ranging from 56.25 to 7.03 µg/ml in LAL reagent water and analyzed for endotoxin using LAL reagent re-suspended in Glucashield buffer. A ThermoMax Microplate Reader (Molecular Devices, Sunnyvale, CA, USA) was utilized to measure LAL agglutination, and each of the samples was analyzed in triplicate. All samples were found to be negative for detectable endotoxin.
FACS-based analyses
A BD FACSCalibur equipped with a high-throughput screening (HTS) loader (Becton Dickinson) or a BT FACSCanto (Becton Dickinson) was used to analyze cells by flow cytometry. Bovine cells were stained with mAbs against IL-2Rα (LCTB2A) (VMRD) and/or γδTCR (clone GD3.8). The mAbs were directly labeled (FITC, PE, PE–Cy5, PE–Cy7, APC or APC–Cy7) or indirectly labeled with goat-antimouse PE, FITC or APC (Jackson ImmunoResearch, West Grove, PA, USA). Human cells were stained for γδ TCR (11F2; BD Biosciences) and anti-human CD69 (BioLegend, San Diego, CA, USA). Mouse γδ T cells were stained with Ab specific for γδ TCR (clone GL3; BD Biosciences). In order to determine the level of background staining in the FACS analyses, second-stage reagents were used alone.
IL-2Rα expression on gated γδ T cells was determined by multicolor FACS after cells were cultured in the presence or absence of varying concentrations of AmB for 48 h. In order to determine cell proliferation after incubation with AmB, PBMCs or mouse splenocytes were labeled with 2.5 μM carboxyfluorescein diacetate succinimidyl ester (CFSE) for 5 min in Hank’s Balanced Salt Solution (HBSS; Mediatech, Manassas, VA, USA), washed and then cultured at 37℃, 10% CO2, in X-VIVO medium for 5 d in the presence or absence of AmB. AmB-stimulated bovine cells proliferated in the absence of additional IL-2, while IL-15 or IL-2 (each at 1 ng/ml) was added to human or mouse CFSE cultures, respectively. Quantification of cell division was done with multicolor FACS analysis to determine the percentage of γδ T cells that had divided at least once, based on a 50% or greater reduction in the intensity of CFSE. Con A (1 µg/ml; Sigma-Aldrich) was used as a positive control. Appropriate negative controls were also employed, including buffers and assay medium only.
IL-18 activation assays
To test for priming effects by AmB, bovine PBMCs were isolated and incubated in X-VIVO 15 medium at 37℃ and 10% CO2 in the presence of AmB (125 ng/ml) or medium only for approximately 24 h. Cells were then washed with X-VIVO and re-suspended in X-VIVO 15 medium in the presence or absence of 1 ng/ml (ELISA) or 10 ng/ml (flow cytometry) recombinant human (rhu) IL-18 (R&D Systems, Minneapolis, MN, USA). To measure secreted IFN-γ, cells were subsequently incubated approximately 20 h and the supernatant fluids were collected for IFN-γ quantification by a bovine IFN-γ ELISA kit (MABTECH, Cincinnati, OH, USA).
To determine the cellular source for IFN-γ production by bovine cells by flow cytometry, intracellular staining was performed. Following cell stimulation as described above, brefeldin A (eBioscience, San Diego, CA, USA) was added to the culture for 4–6 h. Cells were then stained with a directly labeled mAb against γδ TCR (GD3.8) or Alexa Fluor 488-conjugated CD335 (AKS1, specific for bovine NK cells; AbD Serotec, Raleigh, NC, USA). Cells were then fixed with 2% paraformaldehyde for at least 10 min, washed once with PBS + 2% horse serum and then washed once with 0.2% saponin (Sigma-Aldrich) in PBS + 2% horse serum. Intracellular bovine IFN-γ was detected using a PE-conjugated mouse IgG1 mAb against IFN-γ (MCA1783E; ABD Serotec). For isotype controls, cells were stained with a PE-conjugated mouse IgG1 Ab (BioLegend). IFN-γ Abs and isotype controls were re-suspended in 0.2% saponin. Cells were stained for 25 min at room temperature. After staining, cells were washed, then analyzed using a FACSCalibur with HTS.
Infection model
Calves were housed in ABSL2 specific pathogen-free conditions from 1–6 wk of age to ensure equivalent health status prior to ST infection studies. Calves were tested pre-infection for common pathogens, and all were found negative for ST infection. Calves did not naturally acquire any infections in our facilities. At 6 wk, calves were orally infected with 6 × 106, 1 × 107 or 6 × 107 CFU of a natural calf isolate of ST (Type O, Group B). This isolate has been utilized in prior studies, 31 and causes a predictable and severe infection in calves. Prior to infection, 1 μl of ST stock was added to 4 ml of LB media and shaken for 18 h at 37℃. Bacteria were then subcultured 1:40 then shaken again for 2 h at 37℃. CFU counts were verified by plating serial dilutions of bacterial suspensions on LB agar plates. Plasma and fecal samples were collected at infection and at 24 h intervals after infection. Calves were monitored twice daily, and hourly when disease symptoms became apparent. Enterocolitis was defined as fever (>40.6℃) and diarrhea between 24 and 48 h after infection. Fecal samples were collected daily and utilized for bacterial DNA extraction, and also submitted to the Montana State Veterinary Diagnostic Service. Those from experimentally infected calves tested positive for the input strain of ST and negative for other infectious agents. All animal protocols were reviewed and approved by the MSU Institutional Animal Care and Use Committee.
Bacterial DNA extraction and q-PCR
Fecal samples were collected daily from infected calves, and small aliquots were weighed and stored at –80℃ until DNA was extracted. Bacterial DNA was extracted using the QIAamp DNA Stool Mini Kit (QIAGEN, Frederick, MD, USA) according to manufacturer's instructions and stored at –80℃ until further processing. This approach effectively removes PCR inhibitors found in fecal samples. Real-time qPCR was run using SYBR Green Mastermix (Bio-Rad, Berkely, CA, USA) and read on the MYiQ PCR Detection System (Bio-Rad), both according to the manufacturer’s instructions. All primers were ordered from IDT (Coralville, IA, USA), and standard curves were generated for each primer pair. Values for Salmonella species were detected using primers specific to invA, a gene involved in invasion of the intestinal epithelium. 34 These values were normalized to total bacteria levels using primers that are specific to a conserved region among many bacterial species. These values were then normalized to the mass of the fecal aliquot.
Statistical analyses
Data presented were analyzed in GraphPad (GraphPad, La Jolla, CA, USA) and statistical significance was assessed using the unpaired Student’s t-test(two-tailed) or two-way ANOVA as indicated. Data were considered significant when P-values were <0.05.
Results
AmB induces cell division and activation of γδ T cells
HTS assays identified both AmB and a related antifungal drug, Nystatin (data not shown), as potent inducers of IL-2Rα expression on bovine γδ T cells. Non-γδ T cells, including αβ T cells had minimal responses to these drugs. AmB had higher specific activity and was utilized in all further studies concerning the effects of polyenes on γδ T cells. We first measured cell activation induced by AmB by evaluating the percentage of γδ T cells expressing IL-2Rα and the change in mean fluorescence of this receptor. AmB induced increased expression of IL-2Rα on γδ T cells, similar to the response induced by another T-cell agonist, ConA (Figure 1). Figure 1 demonstrates that AmB affected γδ T cells at very low (ng/ml) concentrations. The cells responded strongly after 48 h of incubation with the antifungal drug, but 24 h was sufficient to induce the increased IL-2Rα expression (data not shown). The effect of AmB on IL-2 receptor expression was specific to the γδ T cells within the PBMC population (Figure 1A), but not all γδ T cells were activated even after exposure to the highest (non-cytotoxic) concentrations. This suggests that the drug may only activate specific subsets of γδ T cells. Furthermore, it was observed that removal of monocytes from the PBMC preparation caused a decrease in the AmB effect on γδ T cells (data not shown), indicating a contributory role for monocytes in the response of γδ T cells to AmB. A similar assay was performed using human PBMCs, measuring the increased expression of CD69 as an indicator of activation for human γδ T cells. Similar to bovine cells, human γδ T cells increased expression of CD69 after stimulation with AmB (Figure 1D, E). These data indicate that AmB stimulates γδ T cells from both cattle and humans at low doses.
AmB activates γδ T cells in vitro. Freshly collected PBMCs were cultured with several concentrations of AmB or medium alone. The cells were then analyzed by flow cytometry in two-color FACS assays to determine the percentage of γδ T cells (from at least three animals or donors) expressing activation markers, and the fluorescence intensity of the stain was measured. (A) FACS plots depicting increased expression of IL-2Rα on bovine γδ T cells after treatment with X-VIVO medium, ConA (1 µg/ml, positive control) or AmB (112.5 ng/ml). (B) The percentage of bovine γδ T cells expressing IL-2Rα after treatment with AmB compared with X-VIVO medium alone. (C) The mean fluorescence intensity (MFI) of staining for IL-2Rα on bovine γδ T cells after treatment with AmB or X-VIVO medium. (D) The percentage of human γδ T cells expressing CD69 after treatment with AmB compared with X-VIVO medium alone. (E) The MFI of staining for CD69 on human γδ T cells after treatment with AmB or X-VIVO medium. Error bars represent the SE of mean expression from cells from three donors. Representative of results from at least three independent experiments.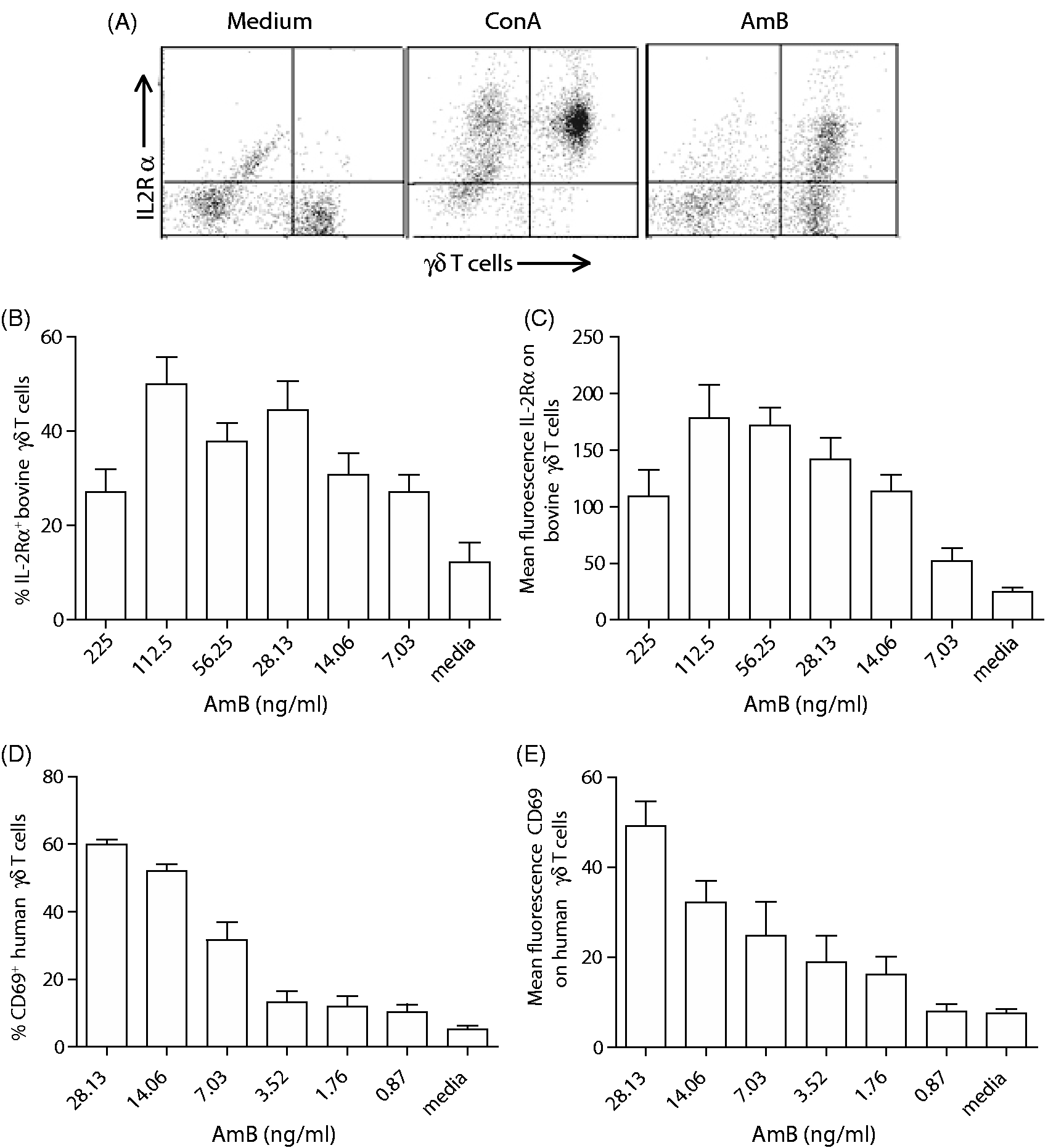
In addition to enhanced IL-2Rα expression, AmB induced proliferation of γδ T cells (Figure 2A, B), detected in a 5-d CFSE assay, and calculated based upon a decrease in CFSE staining at each cell division. The AmB-induced proliferation of γδ T cells was dose-dependent, as lower doses caused few cell divisions above medium-treated cells, while doses of AmB above 112.5 ng/ml were slightly toxic to the cells and caused less proliferation (Figure 2B). These results were consistent with the dose curve related to AmB-induced IL-2Rα expression on γδ T cells, as doses higher than 112.5 ng/ml showed decreased activation, likely owing to cytotoxicity at higher concentrations. Proliferation was also specific to cells staining positive for the γδ TCR, and had little effect on γδ TCR negative cells in the lymphocyte gate (data not shown). A similar proliferation assay was performed using human PBMCs and mouse splenocytes. Figure 2C demonstrates that subsets of human and mouse γδ T cells and γδ TCR-negative cells also proliferated in response to AmB in combination with IL-15 (human) or IL-2 (mouse). These data indicate that the γδ T-cell stimulatory effect is conserved amongst these three species.
AmB causes γδ T cell proliferation in vitro. PBMCs were labeled with CFSE and γδ TCR mAbs, and proliferation was assessed in response to AmB. (A) Representative CFSE FACS plots demonstrating reduction in CFSE staining (indicating cell proliferation) on γδ T cells after treatment with medium alone, 1 µg/ml ConA or 112.5 ng/ml AmB. (B) The percentage of γδ T cells that proliferated at least once after treatment with different concentrations of AmB. Representative results from three independent experiments, performed with three donor calves per experiment. (C) AmB similarly primes human and murine γδ T cells for augmented expansion in vitro. CFSE-labeled human PBMCs or murine TCR-α–/– splenocytes were cultured with AmB diluted in X-VIVO-15 medium for 48 h. Medium was replaced with X-VIVO-15 containing 1 ng/ml IL-15 (human) or IL-2 (murine), and cells were cultured for an additional 72 h. Cells were stained with anti-human or anti-mouse γδ TCR Abs conjugated to PE and analyzed via FACS for γδ T-cell proliferation. Percent total cell proliferation over cytokine alone is given for each scatter plot.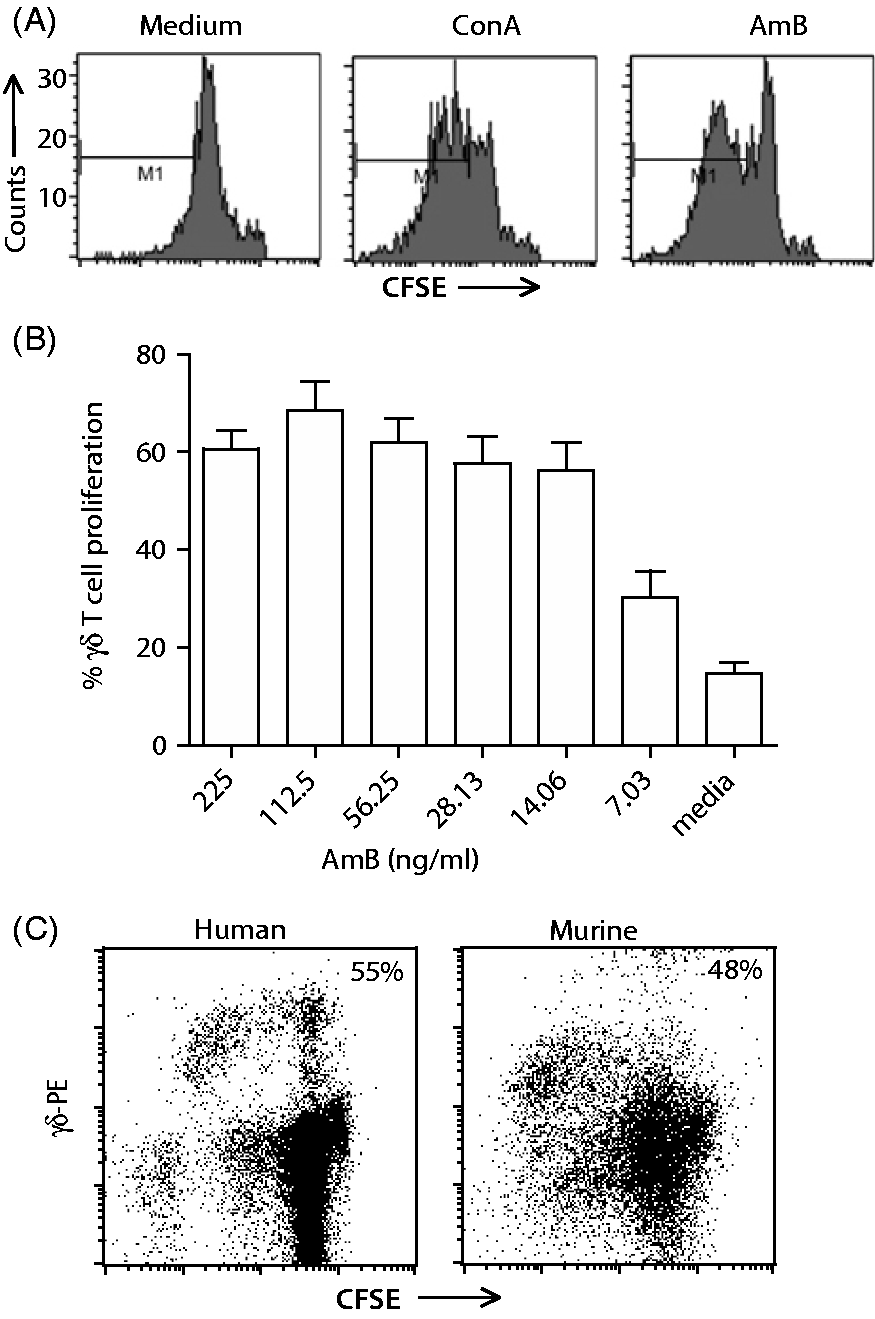
AmB enhances IFN-γ production by bovine lymphocytes
As γδ T cells are a major source of IFN-γ, we assessed the capacity of AmB to stimulate the production of this cytokine in combination with IL-18. IL-18 is produced by stimulated macrophages and, together with IL-12, stimulates the production of IFN-γ. First, we assessed expression of the activation marker IL-2Rα in response to AmB in combination with IL-18. We determined that when combined with IL-18, AmB greatly augmented γδ T-cell activation compared with IL-18 or AmB alone (Figure 3A, B). In addition to IL-2Rα, we determined that the combination of AmB and IL-18 consistently enhanced IFN-γ production by PBMCs (from multiple animals) compared with IL-18 alone (Figure 3C). These results indicate that AmB in low doses acts as an agonist that primes cells for activation by other stimulants in vitro. As IL-18 is expressed in vivo during infection, these data suggest that AmB could enhance the production of this important cytokine during infection. We also sought to determine the cellular source of IFN-γ in AmB- and IL-18-stimulated PBMCs using intracellular flow cytometry. When cells were stained for intracellular IFN-γ and γδ TCR, it was clear that the IFN-γ was emanating from a non-γδ T-cell source (data not shown). This was followed up by staining with an Ab specific for CD335, which is a marker for bovine NK cells.
35
The plots in Figure 3D suggest that the primary source of IFN-γ in AmB- and IL-18 stimulated PBMCs were NK cells. Thus, AmB primes γδ T cells for IL-2-induced proliferation and NK cells for enhanced production of IFN-γ in the presence of IL-18.
AmB primes bovine PBMCs for enhanced stimulation after exposure to IL-18. (A) The percentage of γδ T cells expressing IL-2Rα after PBMCs were cultured with AmB alone (14.06 ng/ml), rhu IL-18 alone (10 ng/ml), both or X-VIVO medium only for 24 h at 37℃ in 10% CO2 and analyzed by flow cytometry in IL-2Rα two-color FACS assays. (B) The mean fluorescence intensity of staining for IL-2Rα on γδ T cells after treatment. Error bars represent the SE from a minimum of three animals. Representative results from three independent experiments. (C) PBMCs from six calves were incubated with 28.13 ng/ml AmB, 10 ng/ml rhu IL-18, both or X-VIVO medium alone for 24 h. Supernatant fluids were collected and concentration of IFN-γ was measured by ELISA. Treatments were compared by two-way ANOVA. P-values for differences in means are *P < 0.05; **P < 0.01; ***P < 0.001; and ****P < 0.0001. (D) Cells were treated with Brefeldin A and stained for intracellular IFN-γ and CD335, a marker for NK cells. Right panel indicates NK cells as the major source for AmB-induced IFN-γ.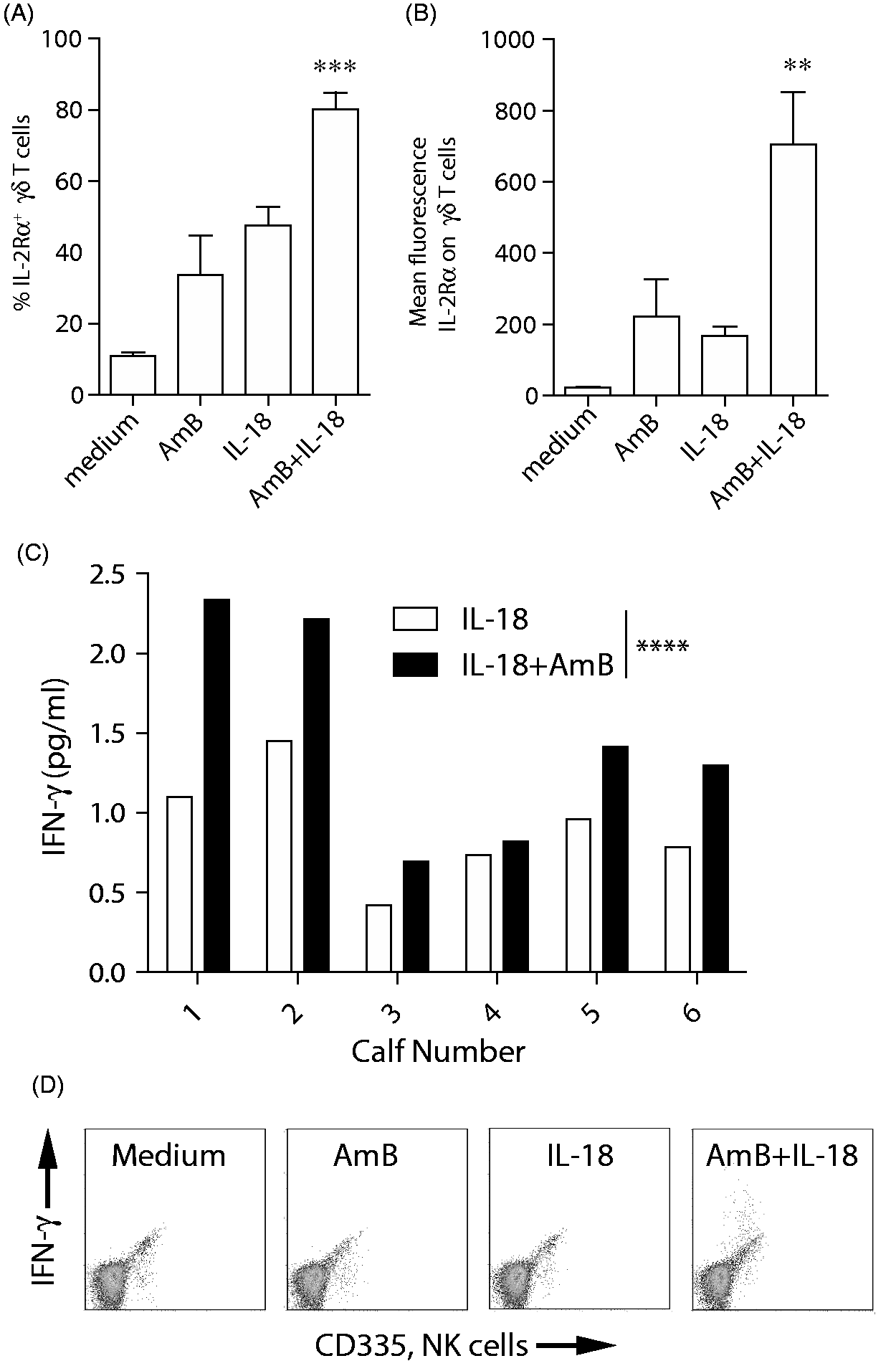
Next we assessed the effects of age differences on responses to AmB in calves. The in vitro production of IFN-γ was assessed in PBMCs derived from newborn calves (<12 h old), 6-12-wk-old calves and adult (>2 yr old) cattle. When 6–12-wk-old calves were compared with Holstein cows, PBMCs derived from calves produced significantly more IFN-γ in response to AmB alone and AmB combined with IL-18 than those derived from mature cow/heifers (Figure 4A). In addition, when newborn calves were compared to 6–12-wk-old calves, newborn calf PBMCs produced significantly more IFN-γ upon stimulation than PBMCs from the older calves (Figure 4B). Together, these data suggest that the ability for AmB to enhance IFN-γ production by NK cells may decrease with increasing age of the animal. Therefore, AmB may be optimally applied in younger populations.
AmB priming of bovine PBMCs for enhanced IFN-γ production after exposure to IL-18 decreases with age. (A) PBMCs derived from bull calves (n = 4) and cows (n = 5) were treated with AmB or medium only, washed, then treated with rhu IL-18 or medium only. IFN-γ levels in the supernatant fluids were then measured by ELISA. (B) Cells from newborn bull calves (n = 4) and older bull calves (n = 4) were similarly compared as for (A). All samples were measured in duplicate. Graphs represent pooled results and error bars represent SEM. Significance was determined by two-way ANOVA. *P < 0.05, **P < 0.01, ***P < 0.001.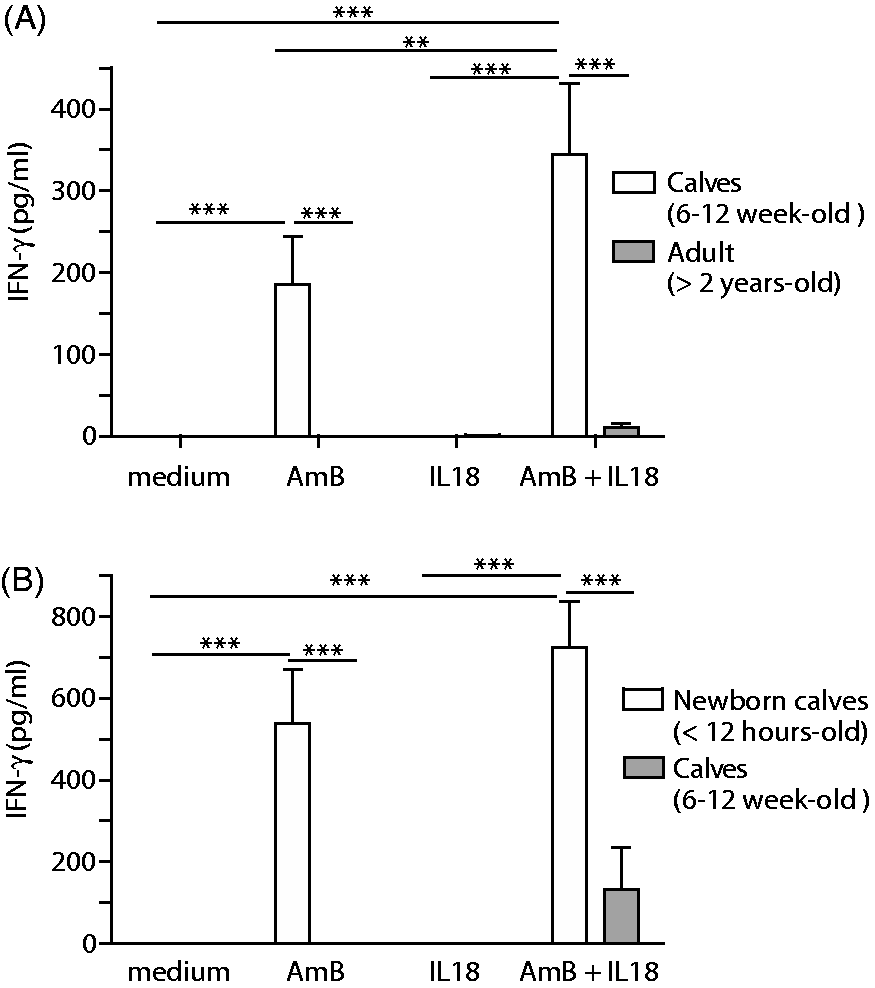
AmB enhances protection against Salmonella infection in calves
Because of its profound effects on multiple responses of the innate immune system, we applied AmB in a non-fungal infectious challenge model. Calves are highly prone to infection in early life and Salmonella is a major problem for ranchers and farmers. We reasoned that, as γδ T cells and NK cells respond to low doses of AmB in vitro, a low dose delivered in vivo would not induce reported toxic side effects, yet would stimulate these important cell subsets to contribute to protection from infection. Six-wk-old calves (average mass 54–59 kg) were injected once i.v. with approximately 0.029–0.031 mg/kg AmB (1.7 mg in 10 ml of saline), or saline only. The dose of AmB is approximately 10-fold less than the recommended 0.25–0.30 mg/kg dose delivered daily to human patients for treatment of fungal infections. Injection of AmB at this low dose did not induce fever or other apparent detrimental effects in the animals (Figure 5; time 0 is 24 h post-AmB injection). After 24 h the calves were infected with ST by the oral route at a range of 6 × 106–6 × 107 CFU. Calves were initially monitored twice daily, and hourly after 48 h of infection. Animals were scored on a 1–4 scale for attitude, appetite, fever and fecals. Figure 5 shows that animals (n = 4 per group) infected with 1 × 107 CFU began to develop fevers at about 24 h after infection, and fevers were most severe by 52 h after infection. The calves that received AmB developed significantly lower fevers than did saline injected calves (Figure 5A). In addition, the calves that received AmB injections had less severe disease based on other parameters. When the four assessments were combined and averaged, the calves that received AmB had lower scores at every time point than the saline-injected calves (Figure 5B). Thus, one injection of low-dose AmB significantly reduced ST-induced disease symptoms.
Treatment with low-dose AmB protects from ST-induced symptoms. Calves (n = 4 per group, pooled from two independent experiments) were injected with either AmB or saline only and infected 24 h later with 1 × 107 CFU ST. Calves were monitored twice daily. (A) AmB treatment caused no change in temperature as measured 24 h post-injection (time 0 post-infection) and significantly reduced fever responses in response to ST infection. At 72 h post-infection, the saline-treated calves had an average temperature of 41 ± 0.17℃; the error bar is not visible. (B) Calves were assessed for attitude, appetite, fever and fecals on a twice-daily basis, and these scores were totaled and averaged within treatment groups. Calves that received AmB also had reduced assessment scores compared with saline-treated calves. Error bars represent SD. Groups were compared using two-way ANOVA, **P < 0.01, ***P < 0.001.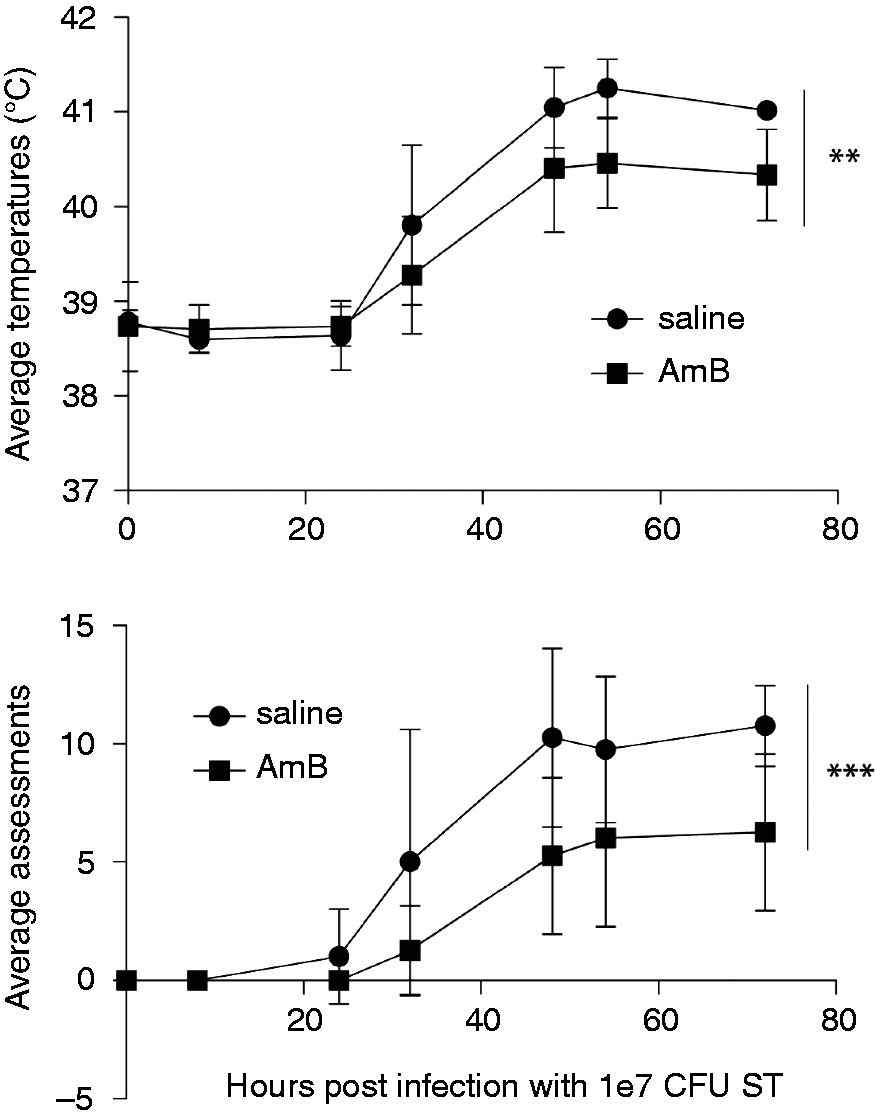
In one experiment, multiple injections of AmB prior to infection were also assessed. In this case, the calves that received four daily injections prior to infection developed a more severe disease than did saline-injected calves (data not shown). One potential reason for this outcome may be excessive TLR4 agonist signaling leading to immune suppression (tolerance), rather than stimulation. 36 However, our data suggest that one low-dose injection of AmB was sufficient to provide enhanced protection from ST-induced enterocolitis.
To determine if γδ T cells were activated or altered by injection or ST infection, cell populations in peripheral blood were assessed. In each experiment, we measured activation markers on lymphocytes isolated directly from blood, and purified cultured cells re-stimulated with LPS (TLR4 agonist), Pam3cys (TLR2 agonist) and ConA/IL-2 in an attempt to assess AmB-induced priming responses ex vivo. However, no consistent changes in γδ T cells or NK cells could be determined in these studies (data not shown). This is consistent with an earlier assessment of γδ T cells during ST infection in which changes were primarily detected in lymphatic cells trafficking from the mucosal tissue. 31 Following i.v. administration, AmB rapidly localizes to and is retained in tissues. 37 Thus, its activation effect may not be reflected in the blood. Furthermore, the primary γδ T-cell subset in circulation localizes to sites of inflammation. 38 It is possible that activated γδ T cells and NK cells similarly localized to the ST-infected tissues and were therefore not detected in circulation in this model. In addition, we measured IL-6 expression in serum collected daily. There were no differences in serum concentrations of IL-6 in calves injected with AmB compared with saline-only injected calves (data not shown). Thus, this low dose of AmB did not appear to cause inflammatory effects similar to those reported for human patients receiving this drug at the therapeutic dose for fungal infections.
To assess bacterial shedding, fecal samples were collected daily from ST-infected calves and assessed for relative amounts of ST. Calves were injected with one dose of AmB or saline only and infected either with either 1 × 107 or 6 × 107 CFU ST. All fecal samples were tested in the Montana State Diagnostic Laboratory for ST using routine culture methods before and after ST infection, and post-infection samples were positive for ST only, as expected. We also developed a quantitative assay using qPCR to detect ST DNA. Bacterial DNA was extracted from daily fecal samples. Primers specific for a conserved region of the 16 S rRNA were used to measure total bacteria for normalization, and ST-specific primers were used to quantify ST in samples.
34
Samples collected at 0 and 24 h did not contain ST, as evidenced by non-specific qPCR products and low normalized CT values. Thus, only values collected at 48 and 72 h after infection were used for statistical comparisons. For both of these infection doses, prior injection with AmB clearly reduced the amount of ST shed in the feces detectible at 48 and/or 72 h after infection (Figure 6). This assay is highly quantitative, as calves infected with the higher dose of ST shed about 10-fold more ST than calves infected with the lower dose. These data suggest that calves that were injected with AmB were not only protected from disease symptoms and severity, but they also subsequently shed less bacteria into the environment than did saline-treated calves. These data indicate that AmB can ameliorate symptoms induced by bacterial infection and counter bacterial replication in calves.
AmB treatment reduced the amount of ST shed in feces. Fecal samples were collected daily from infected calves. Bacterial DNA was extracted from feces and ST was measured by qPCR for an ST-specific gene. Total bacteria (conserved 16 S sequence) was also measured, and ST quantities were normalized to total bacteria and to feces mass. (A) Infection with 1 × 107 CFU. (B) Infection with 6 × 107 CFU. Data from 48 and 72 h (contained in boxes) were used to calculate significance by two-way ANOVA.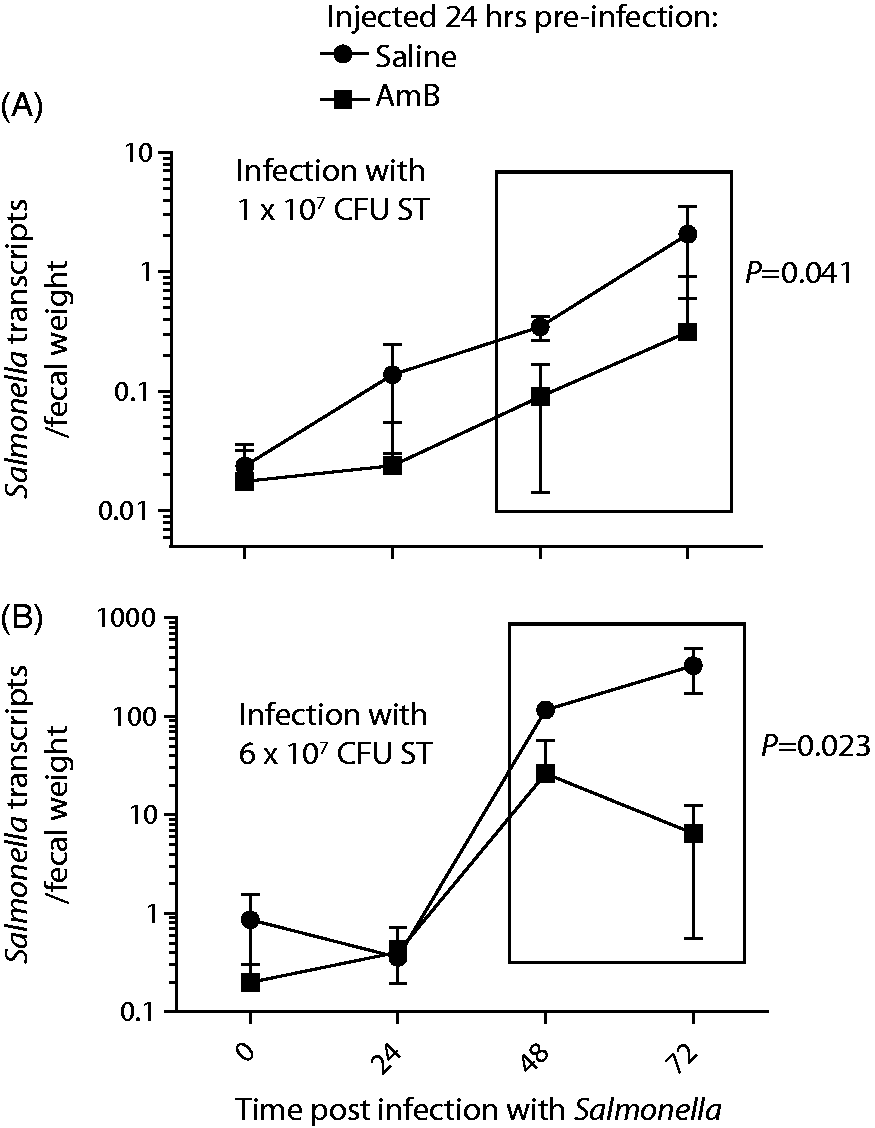
Discussion
AmB is a safe, well-characterized and readily available drug that may be repurposed for enhancing innate protection from disease. As a therapy for invasive fungal infections, AmB is administered i.v. and is given slowly over several hours. The length of treatment with AmB varies depending upon the type of infection being treated, but it is usually administered at dosages between 0.25 and 1.0 mg/kg body mass per day. In these studies, we utilized one dose of 1.7 mg/calf at an average mass of 54–59 kg, which amounts to 0.029–0.031 mg/kg, or approximately one-tenth of the antifungal dose. At this dose, no fever, IL-6 induction or other adverse side effects were noted. The in vitro effects on γδ T cells and NK cells could not be measured in vivo, but this minimal dose protected from Samonella-induced disease. Thus, low-dose AmB may have a subtle priming effect on innate cells in vivo. Upon infection these primed innate cells would have the capacity to respond more readily to infection, and this response likely occurs primarily in the tissue microenvironment. Considering that AmB has been used extensively in human patients and is well characterized in terms of pharmacokinetics and toxicology, its use to enhance innate immune protection at very low doses is warranted.
Human patients that receive large daily doses of AmB can experience severe adverse inflammatory effects. Our data suggest that a very minimal dose can enhance innate protection from infection, without severe side effects. When we utilized multiple daily doses of AmB, ST infection was enhanced in AmB treated calves. This finding is consistent with the results of repeated treatment with TLR agonists, such as LPS, leading to tolerance and reduced TLR signaling. 36 These results underscore the findings that AmB signals similarly to PAMPs through TLRs with similar outcomes.
The utility of these findings may be extended when the pulmonary delivery of AmB is considered. AmB can be delivered directly to the lung and may also be effective for pulmonary infections. 39 Direct delivery of AmB to the lung has been applied human transplant patients to protect from fungal infection. 40 We have observed that AmB induces TLR-dependent macrophage activation, cytokine release and enhanced innate resistance to bacterial challenge in the lungs of mice (unpublished observations). Respiratory infections are very common and problematic in cattle. 41 Thus, it is possible that a simple nasal delivery of low-dose AmB might be a useful prophylactic prior to shipping or other stressful and infection prone situations.
One potential problem of using an antifungal in the absence of any fungal infection is the development of resistant strains. The literature suggests that this is highly unlikely. Species resistant to AmB are rarely seen clinically, and the resistance to AmB has not been profound enough to affect clinical treatment of a wide variety of fungal infections with the drug. Furthermore, it has been repeatedly demonstrated that resistance in fungal species does not emerge during therapy with AmB.42–44 More recently, it was determined that resistance to AmB evolves at considerable costs to fitness, unlike resistance to other antifungal drugs, and this limits the emergence of such variants in the clinic. 45
Infections that result in ‘scours’ in young cattle are a significant problem for the dairy and beef industries, and Salmonella is a major cause of disease in young cattle. Our data suggests that low-dose AmB may enhance innate immune responses to Salmonella infection resulting in protection from disease symptoms and reduced bacterial shedding. An important advantage of enhanced innate responses is broad-spectrum protection from infection. Thus, such treatment is not specific to one pathogen, and the cause of the infection or outbreak need not be specifically identified. In this era of increasing resistance to antibiotics, it is critical to discover and apply novel preventative and treatment measures. AmB could be used as a complementary approach to reduce the necessity or dose of antibiotics. We have also determined that AmB appears to be most effective in very young cattle, especially newborns. Providing this benefit may be particularly important in cases where calves are immune suppressed owing to insufficient colostrum. This young population is most susceptible to a variety of infectious insults. We propose that AmB may be applied as a one- or limited-time treatment for protection of young cattle from infection.
Footnotes
Funding
This project is supported by the Agriculture and Food Research Initiative competitive grant no. 2014-67016-21552 of the USDA National Institute of Food and Agriculture, with partial funding through NIH-NCCAM (AT0004986-01), NIH grant GM103500, M.J. Murdock Charitable Trust and The Montana State University Agricultural Experimental Station.
Conflicts of interest
The authors have no potential conflicts of interest to declare.
