Abstract
Medicinal mushrooms have been used for centuries in Asian countries owing to their beneficial effects on health and longevity. Previous studies have reported that a single medicinal mushroom may produce both stimulatory and inhibitory effects on immune cells, depending on conditions, but the factors responsible for this apparent dichotomy remain obscure. We show here that water and ethanol extracts of cultured mycelium from various species (
Introduction
Traditional Chinese medicine (TCM) represents an ancient form of medical treatment that was established several thousand years ago. While evidence supporting this practice is mostly based on tradition, folklore and anecdotal accounts, considerable interest has been devoted recently to assess the efficacy and safety of TCM.1,2 Several bioactive compounds derived from herbal remedies commonly used in TCM have been identified and approved for human use, including the anti-malaria drug artemisinin (for which the Nobel Prize in Physiology or Medicine was awarded in 2015), the amphetamine-like compound ephedrine and the immunosuppressive molecule fingolimod.1,3 We recently showed that high molecular mass polysaccharides isolated from
A vast group of medicinal mushrooms that includes
Among the biological effects described, the immunomodulatory properties of medicinal mushrooms have been the subject of numerous studies (reviewed recently 7 ). Surprisingly, a single mushroom has been shown to produce both stimulatory and inhibitory effects on immune cells, 16 but the reason for this apparent dichotomy remains unclear. In the present study, we examined the possibility that water and ethanol extracts of medicinal mushrooms may produce opposite effects on innate immune cells.
Materials and methods
Chemical reagents
Αlpha-minimum essential medium (α-MEM) without ribonucleosides and deoxyribonucleosides, RPMI 1640 medium, horse serum and FBS were purchased from Invitrogen (Carlsbad, CA, USA). Folic acid, glutamine, inositol, JNK inhibitor [SP600125 (SP)], ERK1/2 inhibitor [PD98059 (PD)], p38 MAPK inhibitor [SB203580 (SB)] and 2-mercaptoethanol were purchased from Sigma (St. Louis, MO, USA). Inhibitors were dissolved in dimethyl sulfoxide and stored at −20℃ before use.
Preparation of mycelium extracts
Mycelium strains (ABM, AC, GL and HSM) were initially isolated from fresh mushroom fruiting bodies and characterized by Chang Gung Biotechnology. Species identification was performed by sequencing of rDNA and internal transcribed spacer genes (ITS-1 and ITS-2) and comparison with sequence database (≥ 99% homology was observed with strains AJ133376, AJ496398, AF506371, and AJ245559; Bioresource Collection Research Center, Hsinchu, Taiwan). DNA sequence search and alignment was performed using BlastN 2.2.29+ (NCBI, Bethesda, MD, USA).
To prepare the mycelium extracts, 400 g dried mycelium was mixed with 10 l of either double-distilled water or 95% ethanol (w/v) in a bioreactor (Bioflo 4500 20L; New Brunswick Scientific, Enfield, CT, USA). After incubation at 122℃ for 30 min with gentle agitation, the solution was centrifuged at 5900
Cell culture
The human NK cell line NK92 was obtained from the ATCC (CRL-2407; Manassas, VA, USA). The cell lines were initially derived from a case of non-Hodgkin's lymphoma, as described previously.17 NK92 cells were cultured in α-MEM containing 2 mM L-glutamine, 1.5 g/l sodium bicarbonate, 10% FBS, 12.5% horse serum and 100 IU/ml IL-2 (Peprotech, Rocky Hill, CT, USA). The human cell line K562, initially derived from a case of chronic myelogenous leukemia,18 was obtained from the ATCC (CRL-243) and cultured in RPMI 1640 containing 10% FBS.
Abs
Mouse anti-human Abs raised against granulysin, NKG2D, NKp30, NKp44, NKp46 or perforin were used (R&D Systems, Minneapolis, MN, USA). Rabbit polyclonal Abs that recognize ERK, JNK, p38, phosphorylated-ERK (Thr202/Tyr204), phosphorylated-JNK (Thr183/Tyr185) or phosphorylated-p38 (Thr180/Tyr182) were obtained from Cell Signaling Technology (Danvers, MA, USA). Mouse monoclonal anti-actin Ab was purchased from Chemicon International (Temecula, CA, USA). Abs were used according to the manufacturer’s instructions.
Cell viability assay
NK92 cells were grown at 1 × 105 cells/well in 96-well plates (Corning, Corning, NY, USA). Cells were incubated in culture medium (90 μl) containing the indicated concentration of water or ethanol mycelium extract for 24 h. Ten μl MTT reagent (5 mg/ml, MTT In Vitro Toxicology Assay kit; Sigma) was added to the wells and the plates were incubated for 3 h at 37℃. Solubilization solution (100 μl) was added followed by incubation for 18 h. Cell viability was determined based on instructions from the supplier. Experiments were done in triplicate.
NK cell killing assay
Cell killing was monitored using a commercial assay (CytoTox 96 Non-Radioactive Cytotoxicity Assay; Promega, Madison, WI, USA). The assay measures colorimetric changes representing the presence of lactate dehydrogenase, a cytosolic enzyme released from cells following lysis. Briefly, NK effector cells cultured at densities of 5 × 104, 2.5 × 104 and 5 × 103 cells/well in 96-well plates were treated with water or ethanol mycelium extract for 24 h at the dose indicated. Cells were pretreated with MAPK inhibitors for 30 min followed by treatment with 0.1–5% mycelium extract for 4 h. Alternatively, cells were pretreated with MAPK siRNA for 18 h. Washed K562 target cells (5 × 103/well) cultured in complete cell culture medium were added at the effector-target ratios of 1:1, 5:1 or 10:1. Microplates were centrifuged for 4 min at 250
Flow cytometry analysis
The following fluorophore-coupled Abs were used for cell sorting: anti-NKG2D-allophycocyanin (APC), anti-NKp46-fluorescein-isothiocyanate (FITC), anti-NKp30-phycoerythrin (PE; R&D Systems), and anti-NKp44-phycoerythrin (Becton Dickinson, Franklin Lakes, NJ, USA). Blocking of non-specific Ags on NK92 cells (5 × 105) was performed using cold 1% BSA–PBS at 4℃ for 30 min. Cells were treated with Abs in the dark at 4℃ for 30 min prior to washing twice in cold PBS and re-suspension in 1 ml cold PBS. Flow cytometry was done using a FACSCalibur™ flow cytometer (Becton Dickinson).
RNA isolation and PCR analysis
NK92 cells were treated with mycelium extracts for 24 h. mRNA were isolated using the commercial Qiagen RNeasy kit (Qiagen, Valencia, CA, USA). Total RNA was converted to cDNA using a reverse transcriptase kit (Taqman; Applied Biosystems, Foster City, CA, USA). Quantitative PCR was done by using 1/50 of the cDNA preparation in an Mx3000P (Stratagene, La Jolla, CA, USA), using a final volume of 25 μl with Brilliant QPCR Master Mix (Stratagene). The PCR protocol consisted of initial denaturation at 95℃ for 10 min, followed by 50 cycles of 95℃ for 30 s, 55℃ for 1 min and 72℃ for 1 min.
Western blot analysis
NK92 cells (106/well in six-well plates) were cultured with water or ethanol extract for 24 h at the dose indicated. Washed cells were lysed using the Mammalian Protein Extraction Reagent (Pierce Chemicals, Rockford, IL, USA). Proteins (40 µg/lane) were separated on a 10% SDS polyacrylamide gel and electro-blotted onto polyvinylidenedifluoride membranes (Immobilon-P; Millipore, Billerica, MA, USA). Membranes were blocked with 5% non-fat milk for 2 h at 25℃ in Tris-buffered saline (Tris 10 mM, NaCl 150 mM, pH 7.6) containing 0.1% Tween 20. Membranes were probed for 18 h at 4℃ with primary Ab (1:1,000 for anti-ERK, anti-granulysin, anti-JNK, anti-NKG2D, anti-p38, anti-perforin, anti-phosphorylated-ERK, anti-phosphorylated-JNK and anti-phosphorylated-p38; 1:10,000 for anti-actin). Membranes were incubated with HRP-conjugated secondary Ab (1:10,000). Protein signals were developed using enhanced chemiluminescence (Amersham Pharmacia Biotech, Piscataway, NJ, USA).
Gene silencing
siRNA against human p38 (sc-29433), JNK-1 (sc-29380), ERK-2 (sc-35335), NKG2D (sc-42948), NKp44 (sc-72170), NKp46 (sc-63344), NKp30 (sc-42950) and control siRNA (sc-37007) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). siRNA were transfected into NK92 cells by electroporation using a commercial kit (Amaxa, Gaithersburg, MD, USA).
Statistical analysis
Statistical analysis was performed using ANOVA tests (SPSS 12.0 software; IBM, Armonk, NY, USA) with correction for multiple comparisons. Results giving a
Results
Effects of water and ethanol mycelium extracts on NK cell viability and cytotoxic activity
Medicinal mushrooms such as ABM, AC, GL and HSM are non-comestible owing to their rough, wood-like texture and, for this reason, have usually been prepared in water and served as soups or decoction. Alcohols, mainly ethanol, have also been used as extraction solvents in order to obtain tincture containing active compounds of hydrophobic nature. We therefore prepared water and ethanol extracts of ABM, AC, GL and HSM mycelium and monitored the effects of these extracts on the viability of NK92 cells. While treatment with 0.1% water extract did not affect NK cell viability compared with control water, treatment with 2%, or in some cases 5%, enhanced cell viability (Figure 1a). In comparison, the ethanol extracts (0.5–2%) did not affect cell viability compared with control ethanol (Figure 1b).
Effects of water and ethanol mycelium extracts on NK cell viability. NK92 cells were treated with (a) water or (b) ethanol extracts of the indicated mycelium for 24 h and cell viability was assessed using the MTT assay. In (b), NK92 cells were treated with 1% or 2% of a 95% ethanol solution as controls. (a) At 5%, water extract enhanced NK92 cell viability. The experiments shown in this study were performed in triplicate (#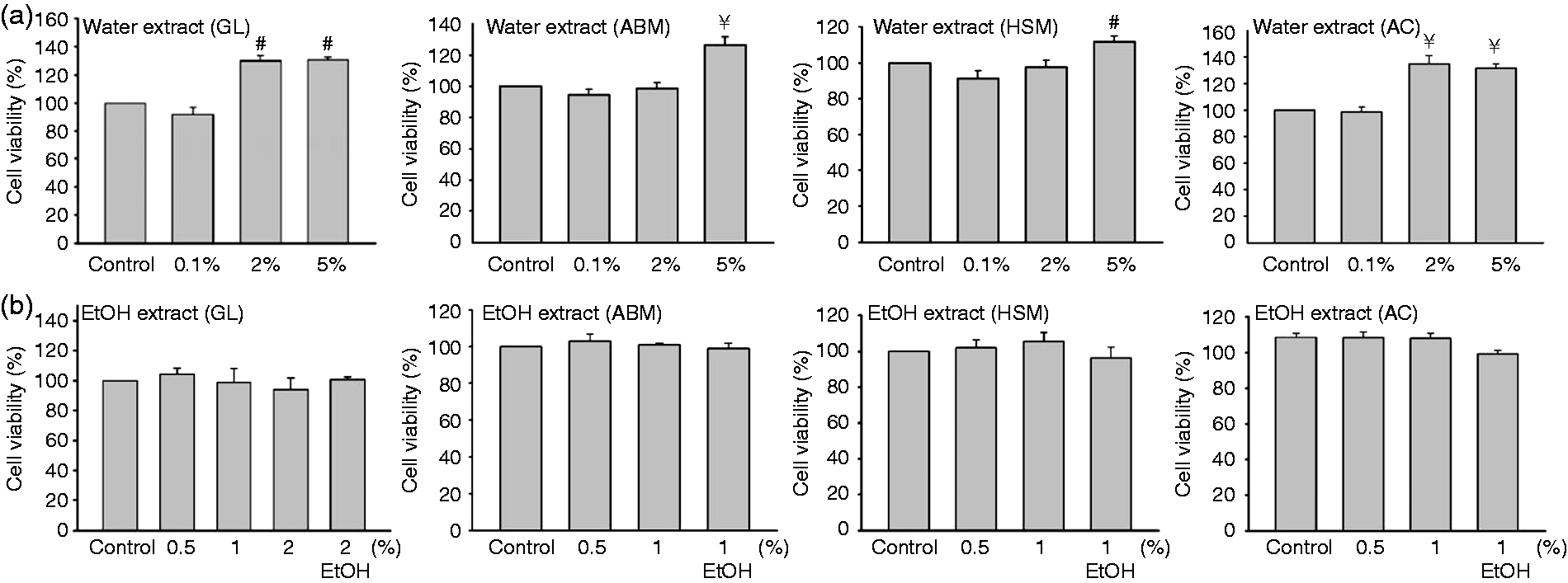
We monitored the effects of the mycelium extracts on NK cytotoxic activity against K562 leukemia cancer cells. For these experiments, NK cells used as effectors were cultured with target cancer cells at different effector-target ratios (1:1, 5:1 and 10:1), which were determined experimentally based on previous work.
19
As expected, NK cell cytotoxic activity increased as a function of the effector–target ratio (Figure 2a,b, ‘Control’ curves). Notably, we observed that the mycelium water extracts enhanced the cytotoxic activity in a dose-dependent manner (Figure 2a), whereas the ethanol extracts reduced cytotoxicity (Figure 2b). These results suggest that the water and ethanol extracts of the same mycelium may produce opposite effects on NK cells.
Water and ethanol mycelium extracts differentially modulate the cytotoxicity of NK cells. Effector NK92 cells were cultured with (a) water or (b) ethanol extracts of the indicated mycelium for 24 h prior to incubation with target K562 myelogenous leukemia cells at various effector-target ratios (1:1, 5:1, and 10:1). Cell killing was measured based on the release of lactate dehydrogenase. Data are expressed as cell death percentages (%).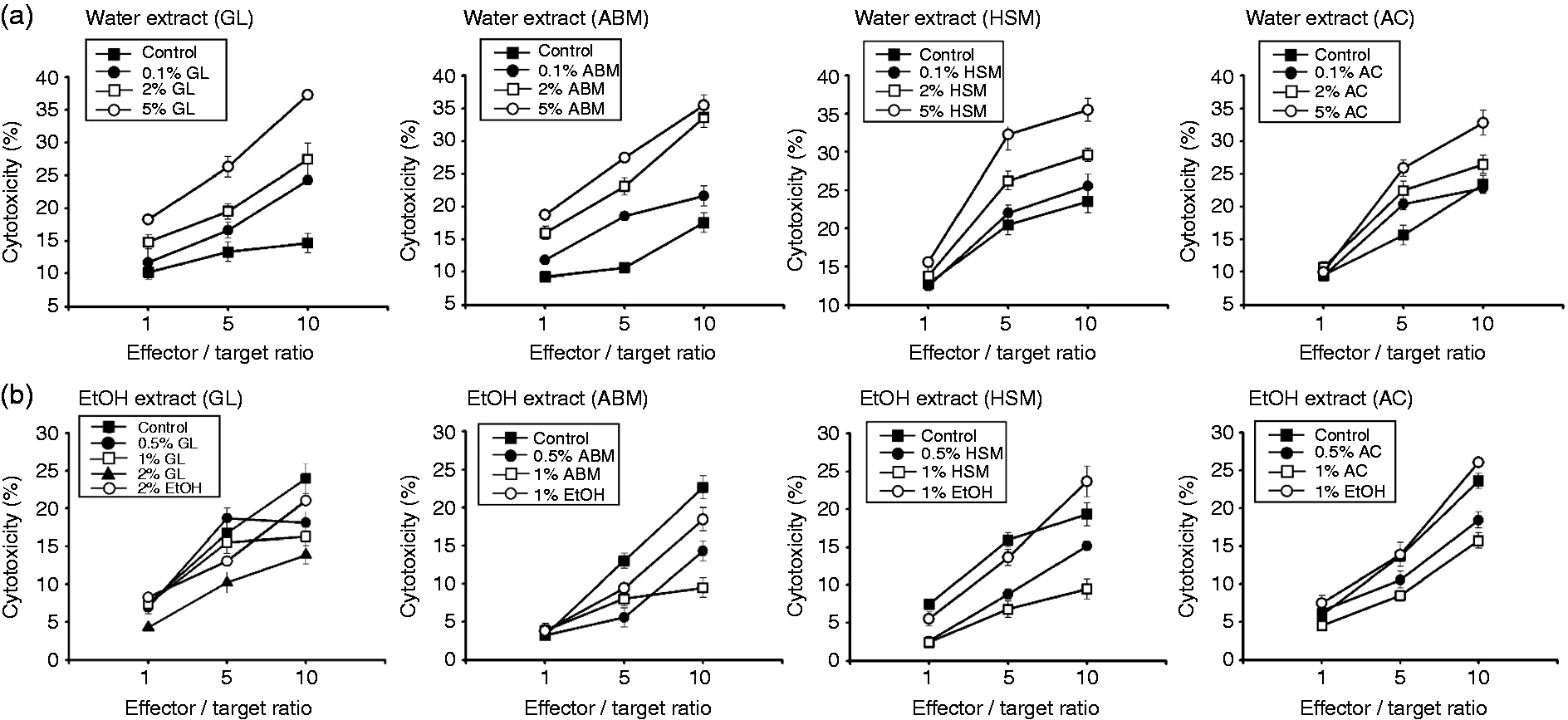
Modulation of perforin and granulysin production by water and ethanol mycelium extracts
We have shown earlier that perforin is a pore-forming protein that mediates the delivery of pro-apoptotic proteins such as granulysin and granzyme into the cytosol of target cells, thereby inducing apoptosis.20–23 We examined the possibility that water and ethanol mycelium extracts may regulate the expression and production of perforin and granulysin by NK cells. Indeed, treatment of NK cells with the water mycelium extracts for 24 h enhanced perforin and granulysin mRNA expression in a dose-dependent manner (Figure 3a). In contrast, treatment with the ethanol extracts reduced perforin and granulysin mRNA expression (Figure 3b).
Water and ethanol extracts of mycelium differentially affect the expression and secretion of perforin and granulysin. NK cells were cultured with (a, c) water or (b, d) ethanol extracts of mycelium for 24 h, followed by measurement of (a, b) mRNA and (c, d) protein levels of perforin and granulysin. RT-PCR was used to quantify mRNA transcriptional level, while Western blot analysis was used for protein quantification. The data represent means of three independent experiments performed in triplicate (*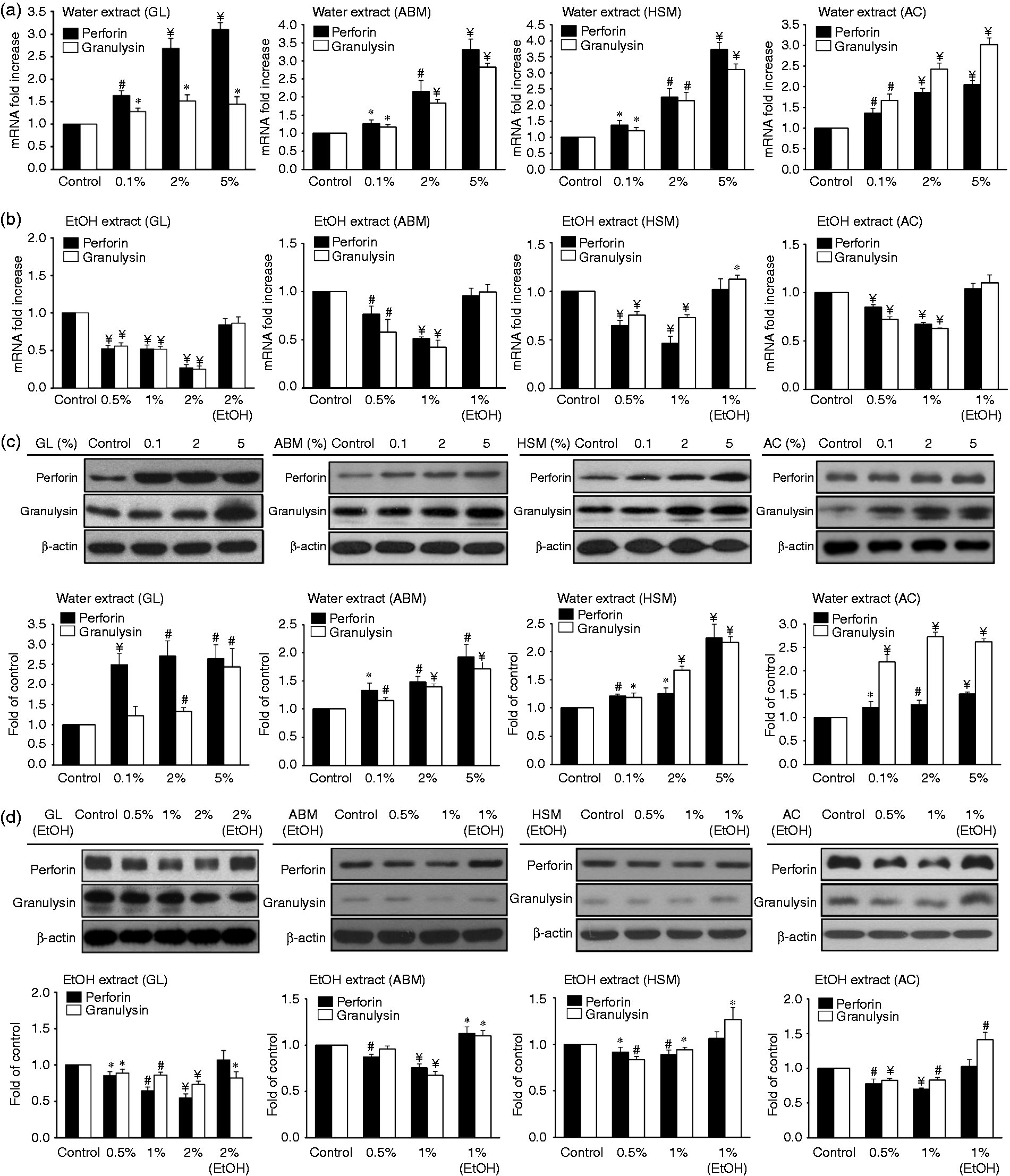
Western blot analysis was performed to determine whether the mycelium extracts modulate perforin and granulysin protein levels. As shown in Figure 3c, water mycelium extracts enhanced the production of perforin and granulysin proteins in a dose-dependent manner. In comparison, ethanol extracts inhibited the production of cytolytic proteins (Figure 3d). The modulatory effects of water and ethanol extracts on cytolytic proteins are consistent with the effects produced by the extracts on NK cell cytotoxic activity as described above (Figure 2a,b), further supporting the concept that water and ethanol extracts produce opposite effects on NK cell function.
NK cell receptor expression is differentially affected by water and ethanol mycelium extracts
Expression of the cytolytic proteins perforin and granulysin in NK cells is induced by activation of signaling pathways acting downstream of NKG2D, NKp30, NKp44 and NKp46 cell surface receptors.19,24 Moreover, activation of NK cell cytotoxicity may induce mRNA and protein expression of NK cell surface receptors,
19
a process that possibly enhances NK cell activation. To examine whether the expression level of these NK cell receptors is affected by mycelium extracts, we measured their mRNA levels using real-time qRT-PCR, 24 h after treatment with the mycelium extracts. mRNA levels of the receptors increased in a dose-dependent manner following treatment of NK cells with water extracts (Figure 4a). However, treatment with ethanol extracts inhibited receptor mRNA expression (Figure 4b).
Effects of water and ethanol mycelium extracts on NKG2D and NCR receptors in NK cells. NK92 cells were treated with (a, c) water or (b, d) ethanol extracts of mycelium for 24 h and total cellular mRNA and proteins were prepared for quantification of NKG2D and NCR. mRNA levels were quantified using (a, b) real-time RT-PCR, while receptor protein expression was monitored using (c, d) Western blotting. The results correspond to means of three independent experiments performed in triplicate (*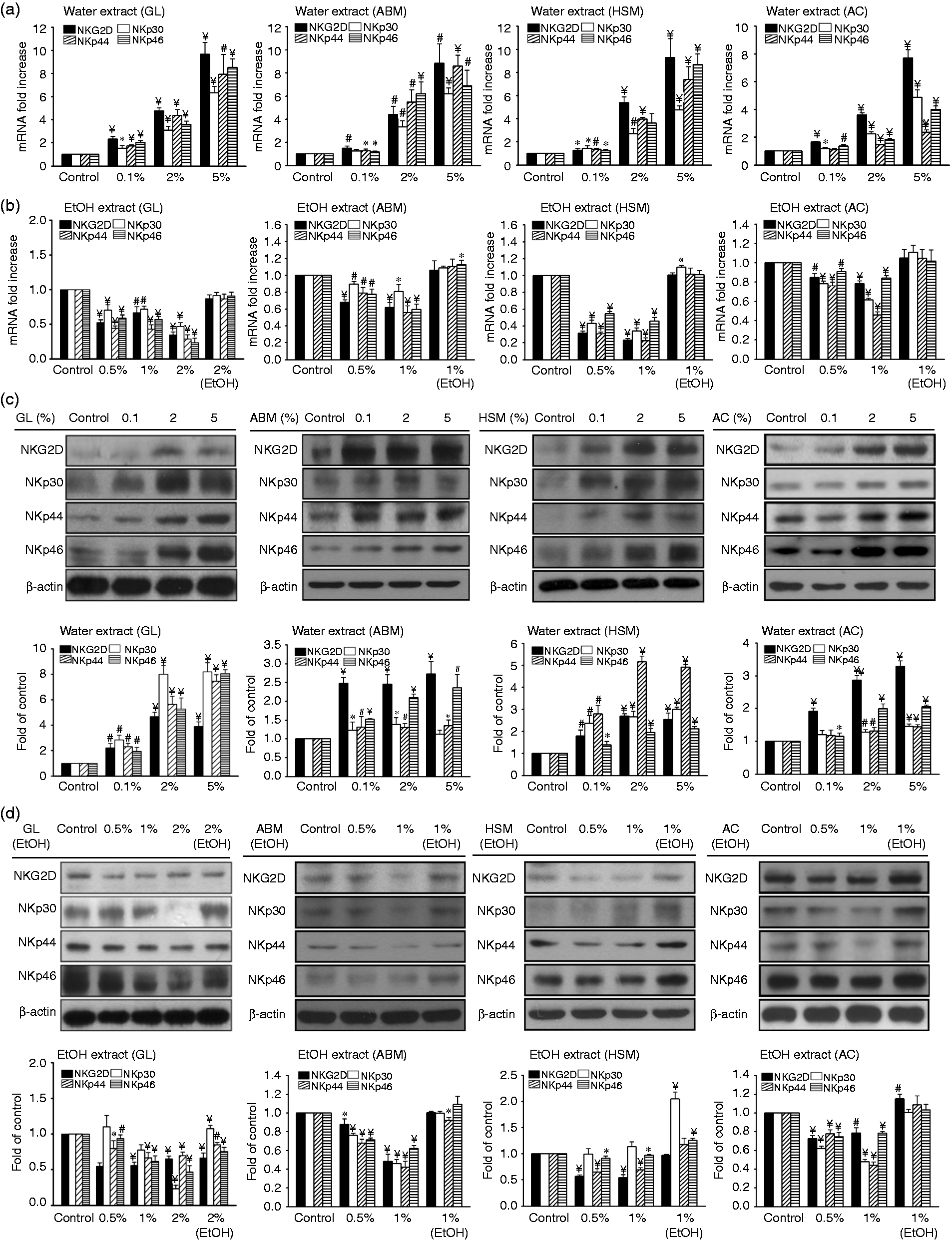
Similar to the results obtained for mRNA expression, the level of NK cell receptor proteins in cell lysate was also modulated differentially by the mycelium extracts. Treatment of NK cells with water mycelium extracts increased production of the receptor proteins in a dose-dependent manner (Figure 4c), while ethanol extracts slightly reduced protein levels of the receptors (Figure 4d).
The levels of NK cell surface receptors were also monitored using flow cytometry. As with the results of total cellular protein levels (Figure 4c), treatment of NK cells with water extracts increased production of the cell surface receptors in a dose-dependent manner (Figure 5a). However, cell surface receptor levels remained at basal levels in cells treated with ethanol extracts (Figure 5b). In these experiments, non-specific binding was ruled out using a matched isotype control Ab (Figure 5, ‘Isotype’ columns). Taken together, these observations indicate that water and ethanol mycelium extracts produce opposite effects on the production of cytolytic proteins as well as cell surface receptors responsible for activation of cytotoxicity.
Quantification of NK92 cell receptors as monitored by flow cytometry. Effects of (a) water and (b) ethanol extract on cell receptor levels. Data are representative of four independent experiments performed as described in ‘Materials and methods’.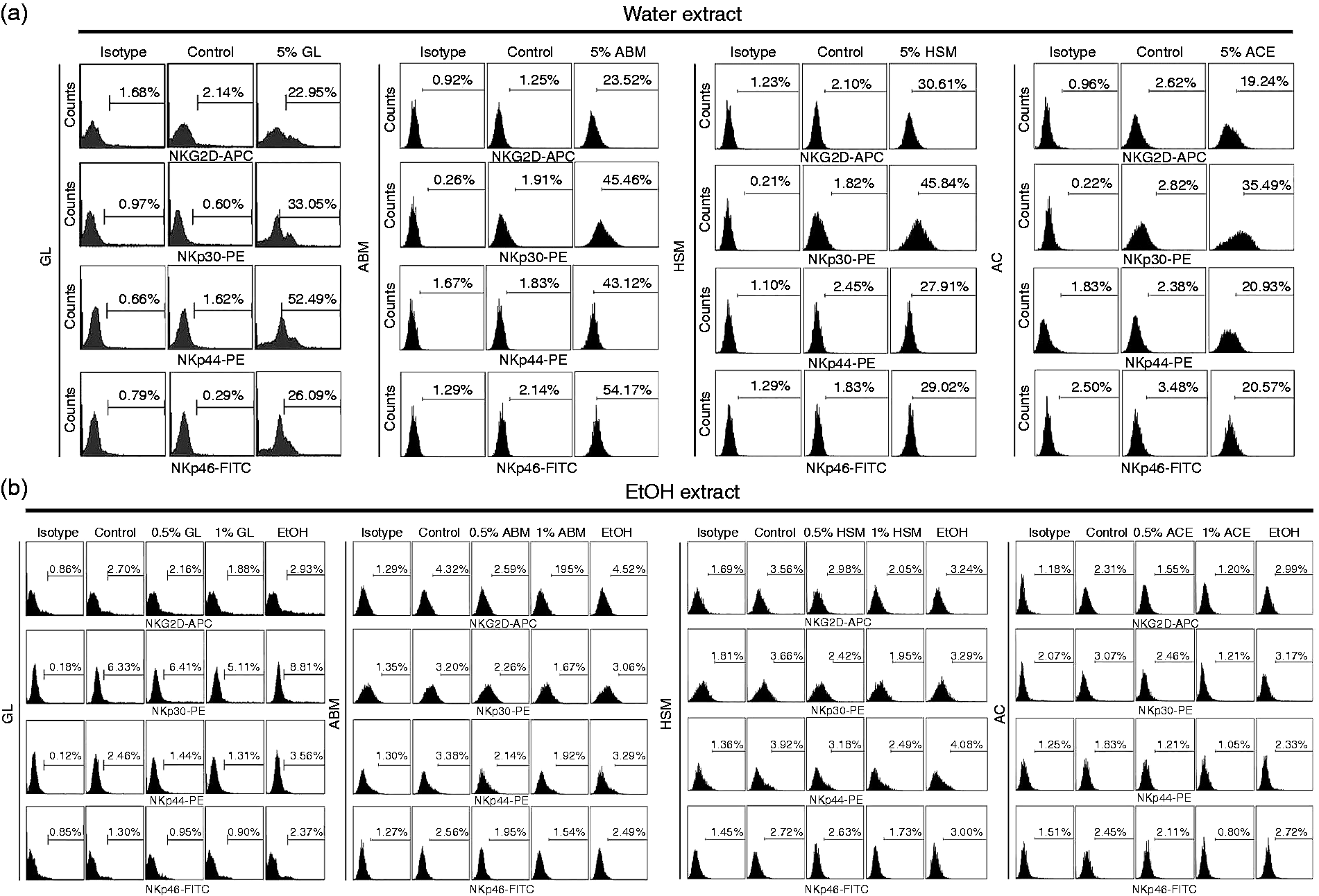
Water mycelium extracts enhance MAP kinase activity in NK cells
MAP kinases act downstream of NK cell receptors and induce the expression of cytolytic proteins.25–27 Accordingly, inhibition of JNK and ERK-1/2 kinases abrogates NKG2D-mediated NK cytotoxic activity.
28
Therefore, we examined whether the water mycelium extracts modulate MAP kinase activity by monitoring the level of phosphorylated (active) kinases using Western blot analysis. As shown in Figure 6a, phosphorylated kinase levels (p-JNK, p-ERK, and p-p38) were enhanced in a dose-dependent manner in NK cells treated with the water extracts.
Effects of water mycelium extract on MAP kinase activity in NK cells. (a) NK92 cells were treated with water extract for 30 min before measuring phosphorylation (P) and total (T) protein levels of ERK, JNK and p38 using Western blot. (b–d) Cells were pretreated for 30 min with MAPK inhibitors against JNK (SP, SP600125, 10 mM), (c) ERK1/2 (PD, PD98059, 10 mM) or (d) p38 (SB, SB203580, 10 mM), prior to treatment with 5% water mycelium extract for 4 h. The cytotoxic activity of treated NK cells against target K562 myelogenous leukemia cells was evaluated at the indicated effector–target ratios. The data represent means of three independent experiments performed in triplicate.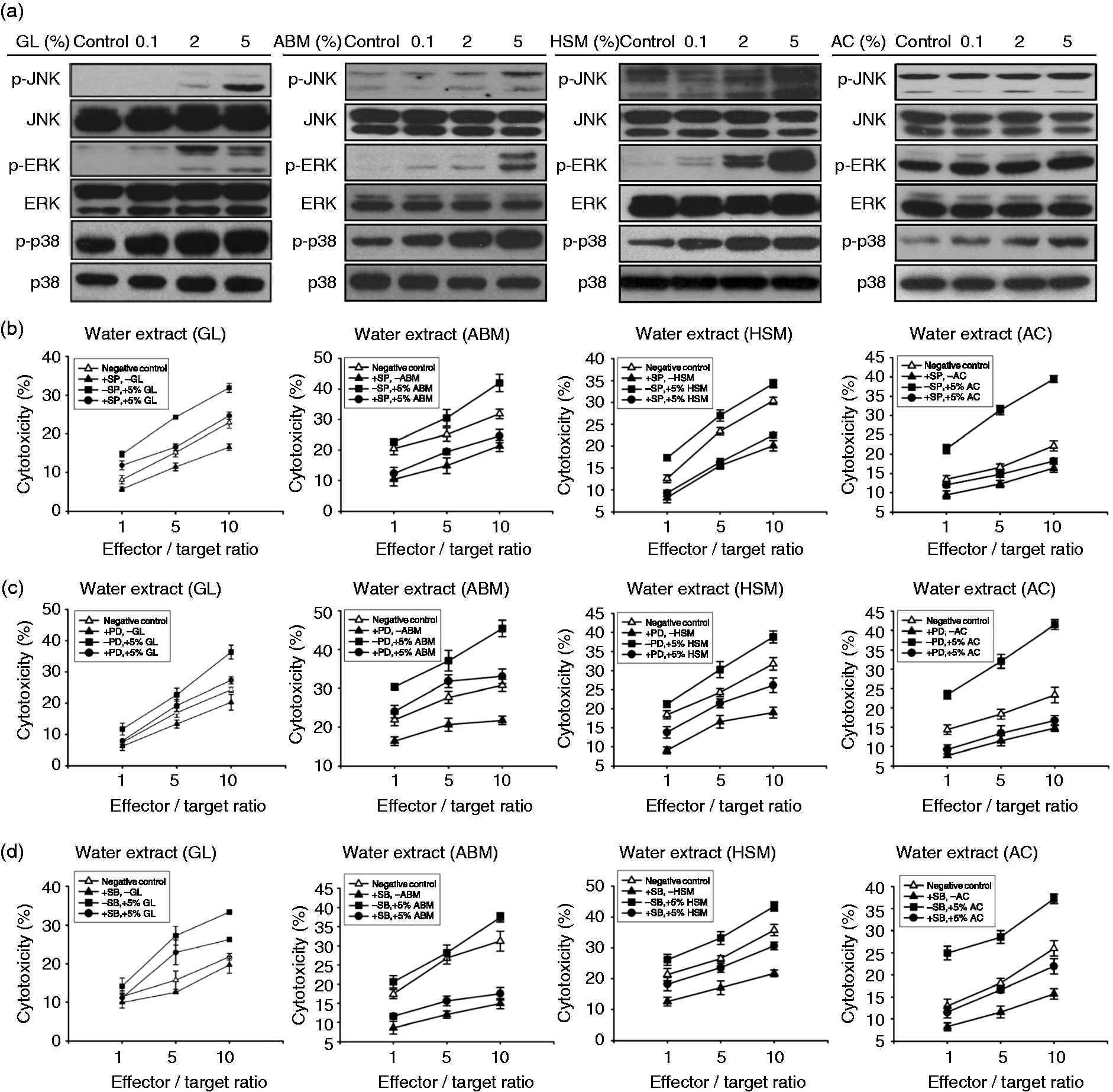
To confirm the involvement of MAP kinases in modulating the effects produced by mycelium extracts, we used chemical inhibitors that specifically target each kinase. Blocking of JNK kinase using the SP inhibitor reduced NK cytotoxic activity induced by treatment with the water extracts (Figure 6b). Similarly, blocking ERK1/2 or p38 reduced NK cytotoxic activity induced by the water mycelium extracts (Figure 6c,d; inhibitors labeled as PD and SB, respectively). These results indicate that MAP kinases are involved in mediating the effects of water extracts on NK cell function.
We also performed knockdown assays using small interfering RNA (siRNA) to silence MAP kinase expression and confirmed the effects of these treatments on NK cell cytotoxicity. Expression of siRNA effectively reduced kinase protein expression, whereas the electroporation treatment, expression of the empty vector or a scramble control had no effect (Figure 7a). While treatment with water mycelium extracts increased perforin and granulysin protein levels, both proteins were down-regulated following silencing of JNK kinase (Figure 7a). Moreover, silencing of JNK reduced NK cytotoxicity induced by the water extracts (Figure 7b). Using the same knockdown strategy, we observed that knockdown of ERK and p38 reduced perforin and granulysin protein levels (Figure 7c,e) and inhibited cytotoxicity in NK cells treated with water mycelium extracts (Figure 7d,f). We conclude that the mycelium extracts modulate NK cell killing activity at least partially by affecting MAP kinase activity.
Mycelium extracts regulate perforin and granulysin secretion by modulating kinase activity. NK92 cells were transfected with MAPK siRNA against (a, b) JNK, (c, d) ERK and (e, f) p38 by electroporation, followed by treatment with 5% water mycelium extracts. Scramble siRNA was used as negative control. Kinase protein levels and cytotoxic activity was analyzed using Western blot and the MTT assay, respectively. The results are representative of three independent experiments performed in triplicate (*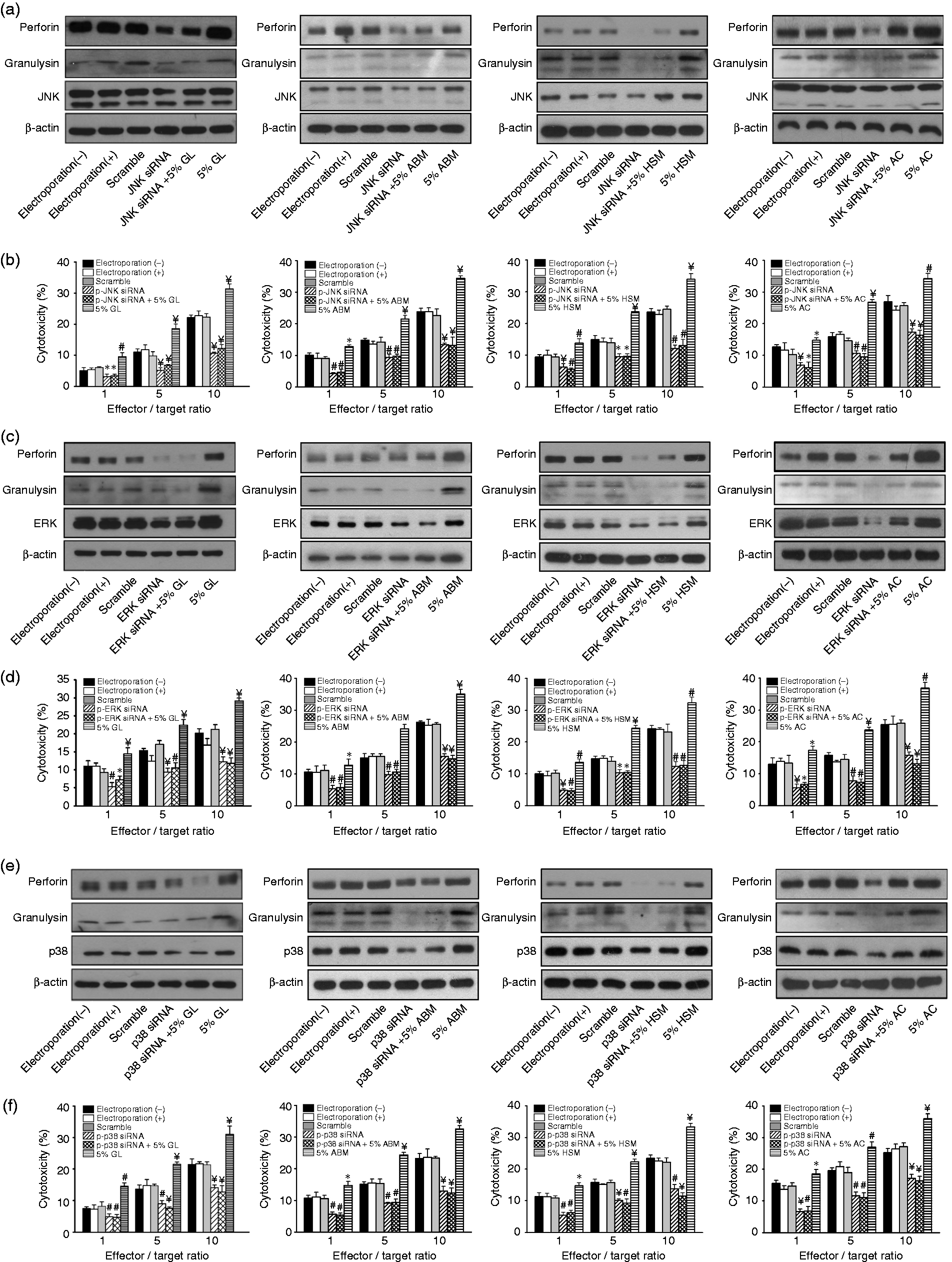
Discussion
Previous studies have shown that a single medicinal mushroom may produce both immunostimulatory and immunosuppressive effects on cultured cells and animals.7,29 The reason for this apparent contradiction has not been thoroughly addressed in the literature. In the present study, we observed that water extracts of medicinal mushrooms activate NK cell cytotoxicity, whereas ethanol extracts reduce cytotoxicity. Our results thus indicate that the extraction protocol may determine the effects of medicinal mushrooms on immune cells, presumably owing to the differential solubility of the bioactive compounds in the extracts. Given that medicinal mushrooms are currently being evaluated for the prevention and treatment of various human diseases,7,9 our results provide a convenient experimental platform for determining the optimal extraction method required to produce specific immunological effects in humans.
Consistent with the hypothesis proposed here, a large majority (83%) of studies that examined the effects of mushroom and mycelium extracts on cultured cells and animal models show that water extracts of the four medicinal mushrooms studied here activate immune responses, while ethanol (or methanol) extracts inhibit immune cells (see Supplementary Table 1; 83 studies out of 100 selected at random support the hypothesis). Of note, the immunomodulatory effects described in the previous studies listed in Supplementary Table 1 cover a wide range of immune cells (B cells, dendritic cells, or DCs, eosinophils, macrophages, neutrophils, NK cells, T cells), primary cells and immortalized cell lines (ANA-1, GG2EE, HeNC2, J774A.1, NK92, NR8383, R309, RAW264.7, THP-1, U937), species (humans, mice, rats), disease models (allergy, cancer, cerebral ischemic injury, colitis, graft rejection, hypoxia, infection, inflammation, insulin resistance, lupus, obesity, skin wound), and immune reactions (Ab production; cell differentiation, proliferation and maturation; cytokine expression and secretion; phagocytosis; targeted cell killing), suggesting that this phenomenon is not limited to NK cells or the experiments presented here.
However, some studies do not support the hypothesis presented here (Supplementary Table 1; 17 studies highlighted with an asterisk out of 100), including a study in which a single OS water extract produced stimulatory effects on immature DCs but inhibitory effects on mature DCs. 30 This study illustrates the importance of the physiological state of the tested organism. Based on these observations, it appears that, in addition to the solvent used for extraction, additional factors—such as the source of fungal species, the amount and potency of immunomodulatory compounds found in each species or specimens, the sample preparation used, or the physiological condition of the cell or organism on which samples are tested—may play a role in determining the immunological activity of mushroom and mycelium extracts.
Another factor possibly affecting the activity of mycelium extracts
Further studies are needed to identify the active compounds responsible for the immunological effects described in the present study. Most immunosuppressive compounds currently in use in humans can be classified as hydrophobic compounds (e.g. azathioprine, cortisol, dexamethasone, laquinimod, opioids, prednisone, sirolimus, tacrolimus; with some compounds such as ciclosporin and fingolimod being derived from fungi and mushrooms), whereas several known immunostimulatory compounds consist of hydrophilic molecules (e.g. cytokines, β-glucan, histamine, polysaccharides). However, notable exceptions have been noted: for instance, vitamin D and estrogens possess hydrophobic properties but are usually considered as immunostimulants. These observations indicate that, while a trend can be observed between the water–ethanol solubility of immunomodulatory compounds and their effects on the immune system, exceptions also exist.
The present study was performed to examine why a medicinal mushroom may produce dual effects on immune cells, a phenomenon that has possible implications for disease treatment in humans. For instance, in a phase I/II clinical trial, Deng et al.
34
showed that the medicinal mushroom maitake (
In addition to providing information regarding the preparation of mushroom extracts for the prevention and treatment of human diseases, our results should also prove useful for the identification of bioactive compounds found in medicinal mushrooms, a field of research that offers promise for the development of novel disease treatments.
Supplemental Material
Supplemental material for Immunomodulatory properties of medicinal mushrooms: differential effects of water and ethanol extracts on NK cell-mediated cytotoxicity
Supplemental Material for Immunomodulatory properties of medicinal mushrooms: differential effects of water and ethanol extracts on NK cell-mediated cytotoxicity by Chia-Chen Lu, Ya-Jing Hsu, Chih-Jung Chang, Chuan-Sheng Lin, Jan Martel, David M Ojcius, Yun-Fei Ko, Hsin-Chih Lai and John D Young in Innate Immunity
Footnotes
Acknowledgements
We thank the personnel of Chang Gung Biotechnology for the isolation and preparation of the mycelium extracts used in this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Y.-F.K. is President of Chang Gung Biotechnology. J.D.Y. is Chairman of the Board of Chang Gung Biotechnology. The authors (with the exception of Y.-J.H.) have applied for patents related to the preparation and use of medicinal mushrooms and their active compounds.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Primordia Institute of New Sciences and Medicine, grants MOST103-2320-B-182-027-MY3 and MOST-104-2320-B-030-009 from the Ministry of Science and Technology of Taiwan, and grants CMRPDm1E0072, CMRPD1F0121 and QZRPD120 from Chang Gung Memorial Hospital at Linkou.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
