Abstract
Phagocytosis commences with particle internalization and culminates with the activation of innate and adaptive immune responses. However, the role of miRNAs in phagocytosis remains largely unknown. In this study, we examined the role of miR-24, miR-30b and miR-142-3p in Ab Fc receptor (FcR)-mediated phagocytosis by macrophages (MΦ) and dendritic cells (DC). The expression of these miRNAs was reduced following phagocytosis of both IgG-opsonized beads and Escherichia coli, indicating their regulatory role in the process. Further, overexpression of these miRNAs impaired the uptake of IgG-coated latex beads, which corroborated the reduced secretion of the pro-inflammatory cytokines TNF-α and IL-8 and down-regulation of PKC-α, as well as superoxide-generating enzyme NADPH oxidase 2 expression level. Mechanistically, MΦ and DC transfected with miRNA mimics show marked reduction in expression of FcRs including FCGR2A, FcɛR1G and FCER2. We show that FcɛR1G expression is not affected at the transcription level, rather it is post-transcriptionally regulated by miR-30b. Finally, we demonstrate that siRNA-mediated knockdown of FcɛR1G leads to reduced uptake of IgG-opsonized beads, indicating its involvement on Ab-mediated phagocytosis. These results uncover miR-24, miR-30b and miR-142-3p as an essential component of FcR-mediated phagocytosis and associated innate immune responses.
Introduction
Phagocytosis is an evolutionarily conserved and complex process that can be broadly categorized into three steps: ligand binding, signal transduction and internalization. Two major categories of receptors involved in pathogen recognition are PRR and opsonic receptors.1,2 Several PRRs with their respective ligands have been identified. For instance, class A scavenger receptor binds to LPS while, CD206 and Dectin-1 recognize polysaccharides present on yeasts.3,4 Soluble molecules like Ig circulating in blood can detect and deposit on foreign particles. Phagocytes express receptors that can recognize these opsonized molecules. 5 As an example, FcγRII and FcγRIII can bind to IgG opsonized particles. 6 Pathogen recognition is generally followed by receptor clustering that leads to activation of signaling pathways. For FcγR, signaling commences with the phosphorylation of two tyrosine residues on the cytosolic immunoreceptor and tyrosine-based activation motif (ITAM) region of the Fc receptor. This is followed by a cascade of phosphorylation that triggers dynamic changes in membrane lipids and actin polymerization eventually leading to particle engulfment.2,7
MiRNAs are small (20–22 nucleotides), noncoding RNA molecules that bind to mRNA harboring complementary sequences, resulting in target transcript degradation or translation inhibition.8–10 MiRNAs, by fine-tuning the transcriptome, regulate a plethora of fundamental biological processes, including cell differentiation, signaling and pathogen response. It is increasingly clear that miRNAs exert regulatory functions in cellular immune responses.11,12 For example, by direct targeting of genes that mount pro- and anti-inflammatory signaling, miR-155 and miR-146a can regulate cellular immune responses.13–15 Similarly, miRNA expression is attuned accordingly during cell differentiation to facilitate key cellular processes signifying that expression of miRNAs in closely associated with cell differentiation and function. Aberration in miRNAs levels can lead to abnormalities in cell function. For example, higher miR-142-3p levels are reported in various malignancies, including leukemia, neoplasm, etc.16–19
Phagocytosis is the first step towards activation of innate and adaptive immune response; however, the role of miRNAs in Ab-mediated uptake is poorly studied. This is an important pathway in clearing diverse Ags, including viruses, bacteria and inert particles, and have been implicated in the pathogenesis of various immune disorders, including immune complex (IC)-mediated autoimmune diseases. We therefore examined miRNA regulation of Ab-mediated Ag uptake in MΦ and DC, key members of the mononuclear phagocytes system that express multiple Fc receptors (FcRs). In our previous study, we demonstrated that miR-24, miR-30b and miR-142-3p were differentially down-regulated during monocyte to MΦ and DC development.20,21 We, and others, have shown that ectopic expression of these miRNAs modulate inflammatory response and differentiation in myeloid cells. For instance, miR-142-3p directly binds to the 3′UTR of PKCα and N-Wasp, thereby regulating TLR activation and cell motility, respectively.21,22 miR-30b has been demonstrated to affect differentiation of myeloid and epithelial cells by regulating key signaling cascades and modulating cellular functions including immune responses and cell migration in various cell types. This indicates a broad spectrum role of this ubiquitous miRNA.20,23–25 Similarly, the role of miR-24 in differentiation, priming immune response and cell migration has been shown.20,21,26–28 These studies indicate an overlapping yet mutually exclusive regulatory role of these miRNAs.
In this study, we examined the role of miR-24, miR-30b and miR-142-3p in Ab-mediated phagocytosis. Our results show negative regulation of bead uptake and subsequent innate immune responses by miRNAs in myeloid inflammatory cells. These miRNAs interfere with bead uptake by targeting various critical components of FcR-mediated phagocytosis, including the novel miR-30b target FcɛR1G. These findings uncover a role of miR-24, miR-30b and miR-142-3p in regulating key biological processes in MΦ and DC.
Materials and methods
Primary human monocyte isolation and differentiation
Freshly prepared buffy coats were collected from healthy donors (Sylvan N Goldman Oklahoma Blood Institute, Oklahoma City, OK, USA) by density gradient centrifugation as described previously.20,21 Briefly, PMBCs were purified using Ficoll Paque-based density centrifugation (GE Healthcare, Piscataway, NJ, USA). PBMCs were incubated with magnetic-labeled CD14 beads (Miltenyi Biotech, Cologne, Germany), according to the manufacturer’s instructions. The purity of CD14+ cells was > 95%, as determined by flow cytometry. For MΦ differentiation, monocytes were plated at 2 × 106/ml in DMEM supplemented with penicillin (100 U/ml) and streptomycin (100 µg/ml). After 2 h the media was substituted with media containing 10% FBS (Life Technologies, Grand Island, NY, USA), and rhM-CSF (50 ng/ml; Peprotech, Rocky Hill, NJ, USA). For DCs, monocytes were cultured in RPMI-1640 supplemented with rhGM-CSF (1000 U/ml) and rhIL-4 (500 U/ml) (both from Peprotech). Media were replaced every 48 h. At d 7, cells were harvested and differentiation confirmed by flow cytometric analysis as described earlier.20,21
Transient miRNA/siRNA transfections
miR-24, miR-30b and miR-142-3p mimics were purchased from Qiagen (Gaithersburg, MD, USA). As a negative control, miRNA mimic with no potential human targets was used (Qiagen). The transient transfection was performed using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). MΦ and DC were transfected at a final concentration of 50 nM oligos. For FcɛR1G knockdown, four siRNAs (Flexitube siRNAs) targeting different locations of the mRNA were purchased from Qiagen. Scramble siRNA was used as control. Cells were transfected with 40 nM siRNA for 48 h and subsequently used for RNA isolation or functional assays. Red siGLO oligos (ThermoScientific, Waltham, MA, USA) were used to determine transfection efficiency.
Phagocytosis assay
Differentiated MΦ and DC were transfected with miScript miRNA mimics, inhibitors or control miRNA mimics (Qiagen). After 36 h, phagocytosis assay was performed with FITC-conjugated rabbit IgG opsonized latex beads (Cayman Chemicals, Ann Arbor, MI, USA) as per the manufacturer’s instructions. Briefly, the opsonized beads were diluted 1:10 in complete culture media (Gibco, Grand Island, NY, USA) and cells incubated for the indicated time points. As a negative control, phagocytosis was inhibited in cells treated with Cytochalasin D (5 µg/ml; Sigma-Aldrich, St. Louis, MO, USA). After assay incubation, cells were washed three times with PBS, fixed with 2% PFA and mounted in Prolong Gold Antifade with DAPI (Life Technologies). For flow cytometry analysis, cells were harvested with Accutase treatment (Cell Biolabs, San Diego, CA, USA) to dislodge adherent macrophages and also to remove fluorescent particles bound to the cell surface and permit evaluation of only internalized particles. Cells were washed twice with 1 × PBS/0.1% BSA and analyzed on BD Fortessa (BD Biosciences, San Jose, CA, USA). Cells were gated using beads and unstained cells to rule any possible contamination of beads.
Phagocytosis assays were also performed using rhodamine-labeled E. coli (Life Technologies) as described earlier. 21
Confocal imaging
Images were captured on Zeiss LSM 710 confocal microscope with 40 × /1.2 Water DIC C-Apochromat objective (ZEISS Microscopy, Oberkochen, Germany). Confocal images were processed on ZEN lite software (ZEISS Microscopy). Images from five randomly selected fields were captured for each donor (n = 4).
Total RNA isolation, cDNA synthesis and quantitative PCR
Day 7-differentiated MΦ and DC were incubated with FITC-IgG beads (Cayman Chemicals) or pHrodo™ E. coli BioParticles® (Life Technologies), as per the manufacturer’s instructions. After 4 h, total RNA was isolated using miRNeasy micro kit (Qiagen) and first strand cDNA was synthesized from 250 ng total RNA using first strand cDNA synthesis kit (Invitrogen). The expression levels of PKC-α, NAPDH oxidase (NOX2), Elf-1, GABPα, Sp1 and normalization control GAPDH were analyzed by real-time PCR using a StepOne 7500 thermocycler (Applied Biosystems, Carlsbad, CA, USA). For miR-24, miR-30b and miR-142-3p quantification, miScript primers and miScript II RT Kit were purchased from Qiagen. One hundred ng total RNA was reverse transcribed according to manufacturer’s instructions. The reactions were run using miRNA-specific primers and universal primer in the PCR mix buffer. RNU6B was used as endogenous control. The Ct values of replicates were analyzed to calculate relative fold change using the ΔΔCt method.
ELISA
Supernatants levels of cytokines TNF-α and IL-8 were measured by ELISA (Cytoset; Invitrogen). Supernatants were diluted (1:5) before performing the assay. The absorbance was measured at 450 nm on SpectraMax M2 (Molecular Devices, Sunnyvale, CA, USA).
Flow cytometry
Cells were harvested after treatments, washed with ice-cold PBS and fixed with 2% paraformaldehyde for 15 min. After washing, cells were re-suspended in 50–100 μl FcR blocking reagent (Miltenyi Biotech), followed by 15 min incubation at 25℃ (RT) to allow blocking of Fc receptors. The cell pellet was washed twice, re-suspended in 50 μl 2% BSA/TBS (w/v) and incubated with fluorochrome-conjugated Abs. The following Abs were used: human CD23 and CD32 Abs were purchased from BD Biosciences (BD Pharmingen, San Jose, CA, USA), while human FcɛR1α and FcɛR1G Abs were purchased from eBiosciences (San Jose, CA, USA). Samples were analyzed using a FACScan or BD Cyan flow cytometer using CellQuest software (BD Biosciences). Further analysis was performed using FlowJo software (Tree Star Inc., Ashland, OR, USA).
For detection of some FcRs, cells were stimulated by treating with recombinant human IFN-γ at a concentration of 100 ng/ml (Peprotech). Human IgG and IgE were used at a concentration of 1 µg/ml (Abcam, Cambridge, UK).
MiRNA target prediction, cloning of 3′UTR and luciferase reporter assays
We scanned the 3′UTR of all the FcRs for potential miR-24, miR-30b and miR-142-3p using the miRWalk search engine (http://zmf.umm.uni-heidelberg.de/apps/zmf/mirwalk/micrornapredictedtarget.html). This analysis predicted miR-30b complementary sequences on FCɛR1G 3′UTR. For 3′UTR cloning, genomic DNA was isolated from freshly prepared PBMCs using QIAamp DNA mini kit (Qiagen) according to manufacturer’s instructions. The 3′UTRs of FCɛR1G was PCR-amplified using Phusion Taq polymerase (New England Biolabs, Ipswich, MA, USA) using forward (CTCGAGCTTTAGAATAGATGCGGT) and reverse primers (GCGGCCGCGAGTCCAGTCCATGGCAGTT). The amplified products were digested with XhoI and NotI and ligated downstream to the luciferase reporter gene in psiCHECK™-2 vector (Promega, Madison, WI, USA). The colonies were screened by restriction digestion and three positive clones for each gene were verified by DNA sequencing. Dual experiments were carried out in a 48-well format. In brief, HEK293 cells were seeded at a density of 3 × 104 in DMEM supplemented with 10% FBS. All the transfections were performed in quadruplicate using 0.5 μl lipofectamine 2000, 120 ng dual luciferase reporter plasmids, and a final concentration of 1 pmol or 5 pmol of synthetic miRNA mimics (Thermo Fisher Scientific, Lafayette, CO, USA). After 36 h post-transfection, cells were lysed in passive lysis buffer (Promega) and dual luciferase assays (Promega) were performed using the Lumat (Turner BioSystems, Sunnyvale, CA, USA) luminometer. For each reporter 3′UTR construct, the Rluc/Fluc value obtained was normalized to the value obtained for psiCHECK™-2 no-insert control (EV) co-transfected with the same miRNA mimic. The values obtained were plotted as histograms, where EV is set at 1.
Statistical analysis
Data were analyzed using GraphPad Prism (GraphPad Inc., La Jolla, CA, USA). The results are represented as SD or ± SEM from three independent replicates and experiments were conducted at least thrice. P-Values were calculated using Students t-test and P < 0.05 was considered significant.
Results
Cellular levels of miR-24, miR-30b and miR-142-3p are responsive to phagocytosis
MiRNA genes are highly responsive to extracellular signals and exhibit temporal transcriptional expression.9–11 Previously, we demonstrated that expression of miR-24, miR-30b and miR-142-3p decreases during monocyte to MΦ and DC differentiation.
20
Enforced expression of these miRNAs renders myeloid cells less functional with regard to innate responses.20,21 We therefore monitored whether expression of miR-24, miR-30b and miR-142-3p were modulated during phagocytosis, an integral function of APCs. At d 7 differentiated MΦ and DC were challenged with IgG-opsonized beads or heat-killed E. coli and miRNA levels monitored after 4 h. In both MΦ and DC, expression of all the three miRNAs was down-regulated by ∼30–60% during the uptake of IgG opsonized beads (Figure 1a). For bacterial uptake mediated by scavenger receptors, we noticed marked reduction (∼25–40%) in the expression of miR-24 and miR-142-3p, suggesting that these miRNAs are responsive to phagocytosis (Figure 1b). Conversely, miR-30b levels were not significantly altered in MΦ but were significantly reduced in DC (Figure 1b). It can be noted that compared with MΦ, miRNA expression in DC is more responsive to both IgG-opsonized beads and bacterial challenge indicating cell-specific activation. These results suggest that down-regulation of miR-24, miR-30b and miR-142-3p expression occurs during activation of MΦ and DC by FcR, as well as TLR ligands.
MiR-24, miR-30b and miR-142-3p expression is modulated during phagocytosis. MΦ and DC were challenged with (a) IgG-opsonized beads for 4 h or (b) E. coli (EC) for 2 h and miRNA levels were assayed by qRT-PCR. RNU6 was used as internal control. The data presented as ± SEM of four donors and the experiments were performed three times. Student’s t-test was conducted to calculate P-values. *P < 0.05, **P < 0.01.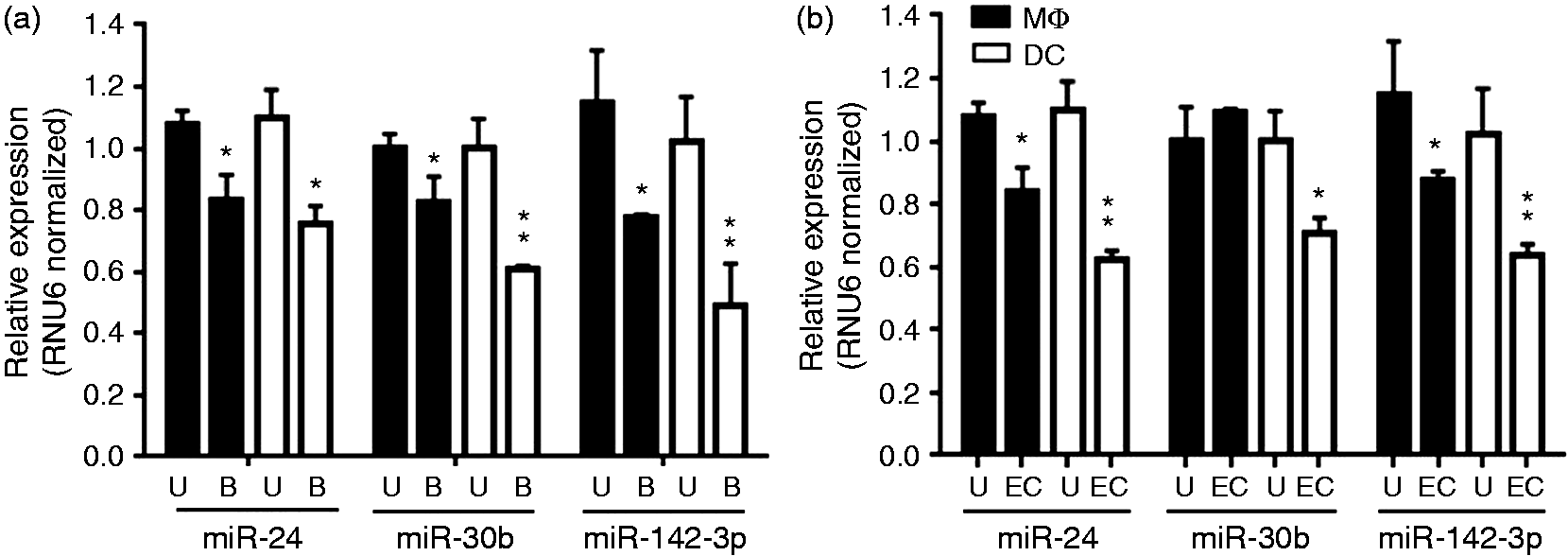
Overexpression of miR-24, miR-30b and miR-142-3p attenuate phagocytosis of Ab-opsonized beads by primary human monocyte-derived MΦ and DC
In our previous study, we demonstrated the inhibitory role of miR-24, miR-30b and miR-142-3p in the uptake of both Gram-positive and Gram-negative bacteria. 21 Besides direct recognition of PAMPs and subsequent pathogen uptake and clearance, phagocytes also internalize Ab-opsonized particles. This alternate mode of phagocytosis simultaneously allows uptake of pathogens that can evade PRR recognition, as well as inert particles that cannot be directly detected by phagocytic receptors. Mononuclear phagocytes express various FcRs that recognize a specific class of Ig. To test if these miRNAs modulate phagocytosis involving a different set of receptors, we examined their role in Ab receptor-mediated phagocytosis. Prior to performing the experiments with miRNAs, we wanted to examine the population of oligo-transfected cells undergoing phagocytosis. To mimic miRNA transfection, we used DY-547-labeled siRNA to assess the population of MΦ and DC positive for FITC-labeled IgG-coated latex beads. Flow cytometric data show that ∼82% and ∼78% of cells are double positive (siRNA and beads) in MΦ and DC, respectively (Supplementary Figure S1A, B). These results show that significant population of siRNA transfected is undergoing active phagocytosis.
Next, MΦ were transiently transfected with miRNA or control mimics and FcR-mediated phagocytosis of IgG beads assessed. Cells overexpressing miR-24, miR-30b or miR-142-3p, but not control mimics, showed reduced uptake of beads as reflected by low FITC signal under confocal microscopy (Figure 2a). The differential interference contrast (DIC) images also show fewer beads (marked with yellow arrows in lower panel) accumulated inside miRNA-transfected cells compared to control (Figure 2a). To quantify the inhibitory effect of these miRNAs, we analyzed the cells by flow cytometry. Compared with control, miR-24-, miR-30b- and miR-142-3p-transfected MΦ showed approximately 43%, 31% and 42% reductions in FITC-positive cells, respectively (Figure 2b–e).
Overexpression of miRNA mimics attenuate uptake of IgG-opsonized latex beads in MΦ. At d 7 differentiated primary monocyte-derived MΦ were transfected with miR-24, miR-30b and miR-142-3p mimics or control mimic and phagocytosis assays performed by replacing media with IgG-opsonized beads, as described in the ‘Materials and methods’. After 2 h incubation, cells were fixed and images from three different fields were captured on a confocal microscope. (a) Representative images showing internalized FITC-conjugated beads. Scale bar = 20 µm. (b–d) Histograms showing uptake of FITC beads phagocytized by miRNA or control mimics transfected MΦ. (e) Percent geometric mean florescence intensity (MFI) as assessed by flow cytometry. Untreated cells challenged with beads were used as a positive control. Data are presented as ± SEM from five independent donors. **P < 0.01, ***P < 0.001 (two-tailed Student’s t-test).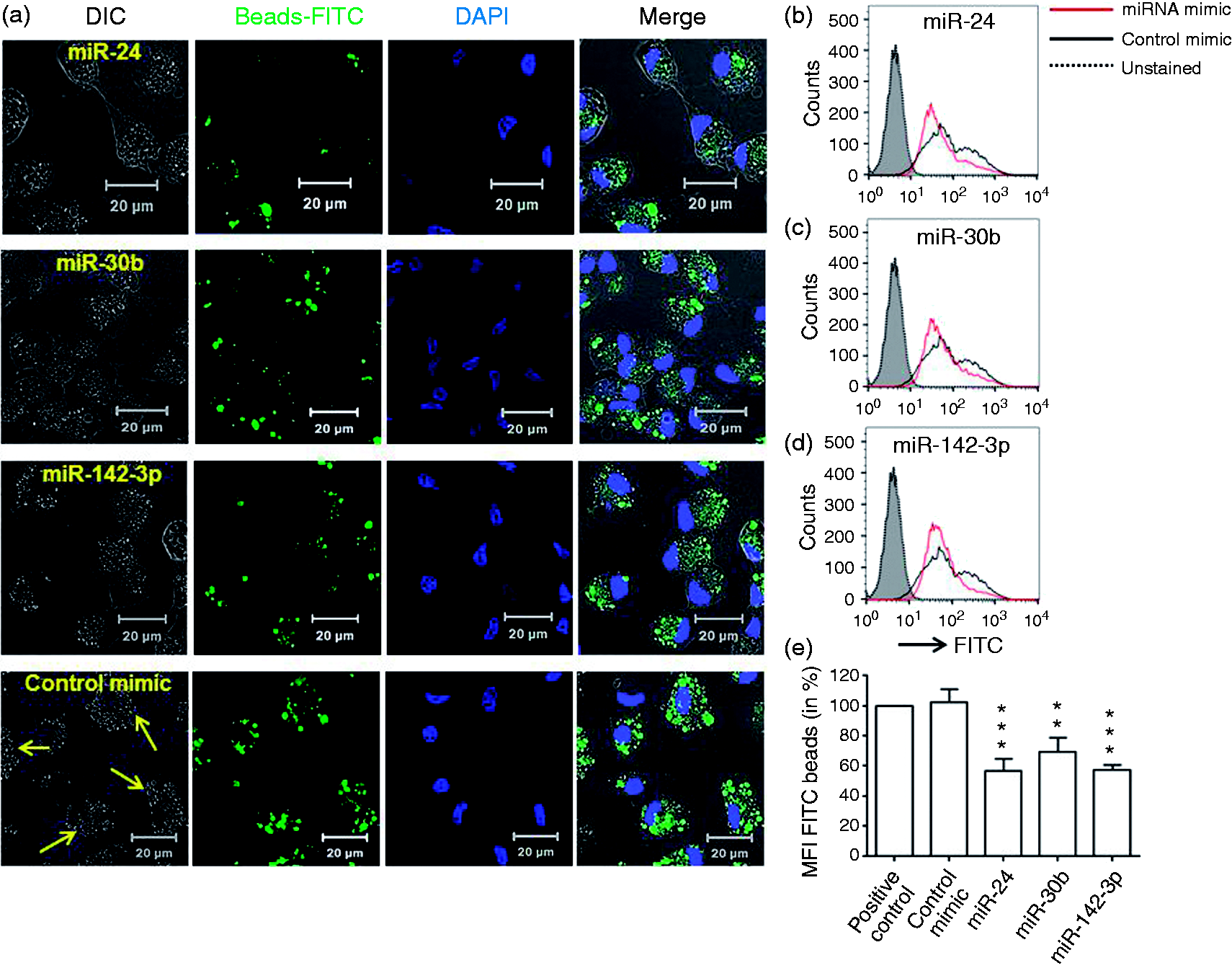
Further, we examined the impact of these miRs on the uptake of opsonized beads by monocyte-derived DC. Figure 3a shows reduced internalization of beads in DC transfected with miR-24, miR-30b and miR-142-3p mimics compared with control, which correlated with bead accumulation observed in DIC images. Similar to MΦ, significant reduction (∼30–40%) in the DC FITC-positive population was noted by flow cytometric analysis (Figure 3b–e). Taken together, these results demonstrate that miR-24, miR-30b and miR-142-3p negatively regulate Ab receptor-mediated phagocytosis in monocyte-derived MΦ and DC.
Overexpression of miRNA mimics attenuate uptake of IgG-opsonized latex beads in DC. At d 7 differentiated primary monocyte-derived DC were transfected with miR-24, miR-30b and miR-142-3p mimics or control mimic and phagocytosis assays performed by replacing media with IgG-opsonized beads. (a) Confocal images are representative of assays on four independent donors. Scale bar = 20 µm. (b–d) Flow cytometry analysis showing histograms of FITC beads phagocytized by miRNA or control mimics transfected DC. (e) Percent geometric mean florescence intensity (MFI) as assessed by FACS. Untreated cells challenged with beads were used as a positive control. Data are presented as ± SEM from five independent donors. *P < 0.05, **P < 0.01, ***P < 0.001 (two-tailed Student’s t-test).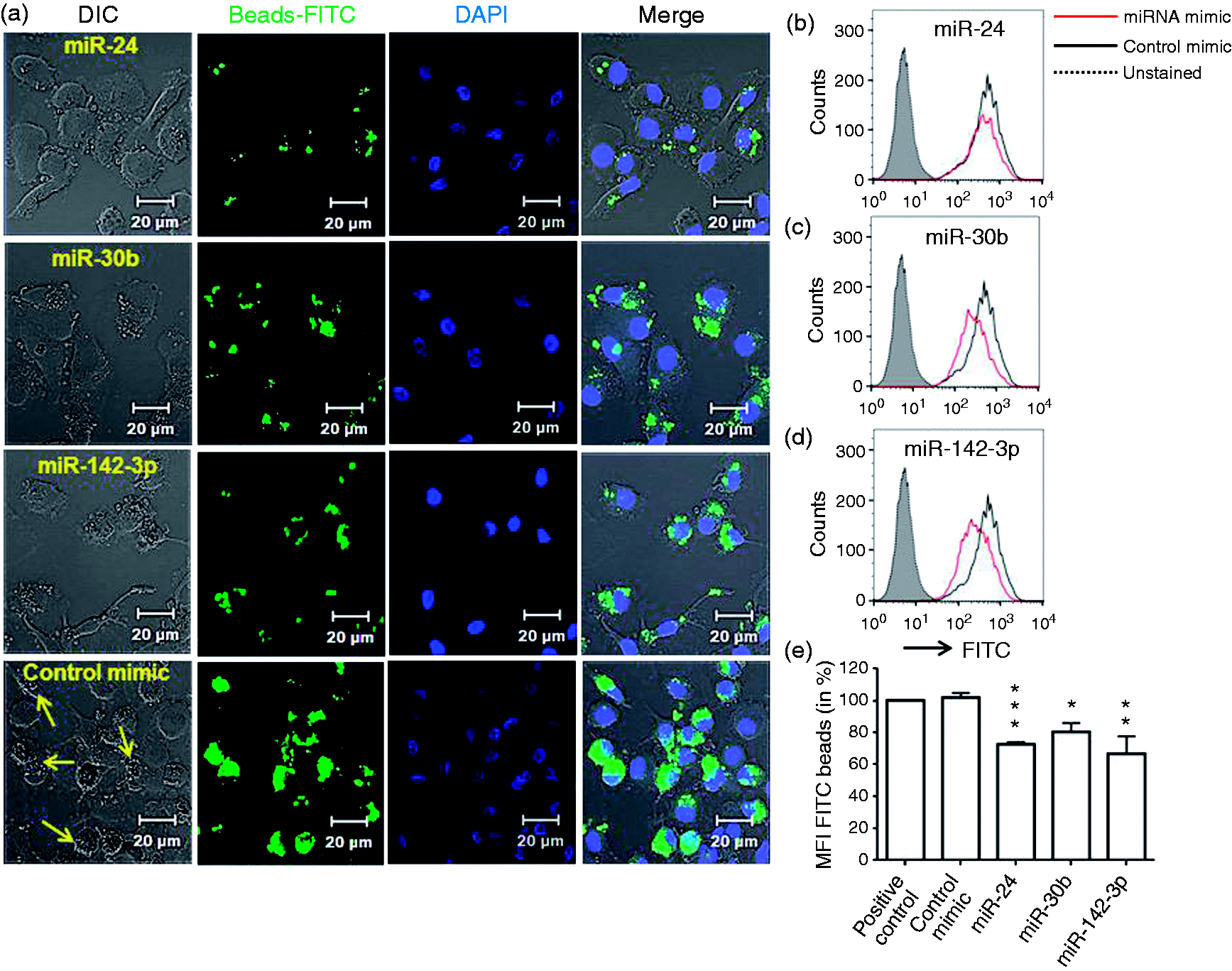
miRNA inhibition of phagocytosis is associated with reduced pro-inflammatory cytokine production by targeting PKC-α and down-regulate expression of ROS generating enzyme NOX2
Ligation of FcR triggers secretion of pro-inflammatory cytokines and is an important component of the innate response.
29
We therefore monitored supernatant levels of TNF-α and IL-8 in cells challenged with IgG beads for 4 h. MiRNA-transfected MΦ and DC show significant reduction in TNF-α levels except for DC overexpressing miR-24 mimics (Figure 4a). For IL-8, we noted significant down-regulation in MΦ and a trend for lower cytokine production in DC, albeit not significant (Figure 4b). Compared with DC, the impact of miRNAs on cytokine production by MΦ was more pronounced.
Reduced secretion of pro-inflammatory cytokines and down-regulation of PKC-α and NOX2 expression in miRNA-transfected MΦ and DCs. MiRNA or control mimic transfected MΦ and DC were incubated with IgG-opsonized beads for 4 h. Quantification of (a) TNF-α and (b) IL-8 release in the culture supernatants of MΦ and DC. Real-time PCR analysis showing mRNA expression of (c) PKC-α and (d) NOX2 in IgG beads challenged cells. GAPDH served as internal control for normalization. The data presented as ±SEM of four donors and the experiments were performed three times. Student’s t-test was conducted to calculate P-values. *P < 0.05, **P < 0.01, ***P < 0.001.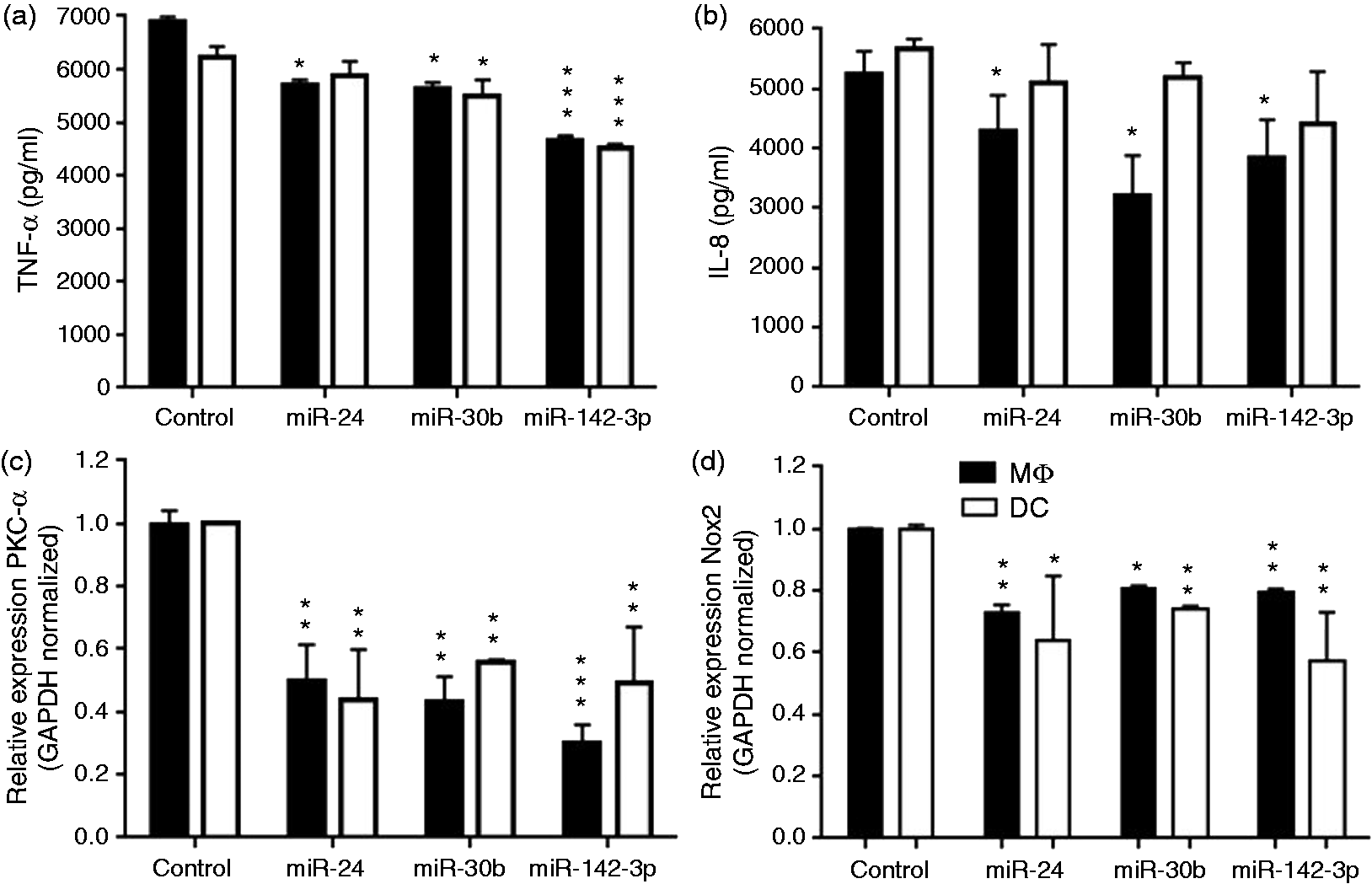
Reduced secretion of cytokines in miRNA-transfected MΦ and DC undergoing phagocytosis of IgG-opsonized beads indicated a compromised activation of downstream signaling. PKC-α plays a central role in FcR signaling that culminates with the induced pro-inflammatory cytokine secretion. Next, we monitored the expression of PKC-α expression in phagocytosis. Quantitative RT-PCR results show marked decrease (∼50–75%) in the PKC-α mRNA levels in miR-24-, miR-30b- and miR-142-3p-transfected MΦ and DC (Figure 4c). These results suggest that attenuation of phagocytosis by miRNA mimics correlates with the reduced expression of genes involved in microbial clearance, activation of signaling cascade and pro-inflammatory cytokine secretion.
The bactericidal activity of phagocytes requires activation of free radical-producing genes, including NAPDH oxidase 2 (NOX2).30,31 To evaluate the functional impact of miRNAs during phagocytosis, we examined the expression of a critical component of microbicidal activity, NOX2. MΦ and DC were transfected with miRNA mimics and 24 h later challenged with IgG-opsonized beads. Quantitative RT-PCR analysis show significantly reduced expression of NOX2 in miRNA transfected MΦ and DC compared with control (Figure 4d). This indicates that miRNA-mediated defect in phagocytosis is linked to pathways involved in pathogen clearance.
miR-24, miR-30b and miR-142-3p down-regulate expression of FcR genes and impairs downstream signaling
Previously we showed that overexpression of miR-24, miR-30b and miR-142-3p in MΦ and DC leads to down-regulation of several genes involved in phagocytosis, including FcRs.
21
Of the six FcR genes examined [FCGR1A (CD64), FCGR2A (CD32), FCGR3B (CD16b), FCER2 (CD23), FCER1A and FCER1G], we noted reduced expression of CD32, CD23, FCER1A and FCER1G.
21
However, miRNA-mediated changes were not similarly affected in MΦ and DCs. For instance, we noticed that only one gene, FCER1G, was significantly altered in MΦ at the mRNA level. To further validate these findings, we examined the surface expression of CD32, CD23 and FCER1G in miRNA-overexpressing cells. MΦ and DC were transfected on d 3 and expression of molecules associated with phagocytosis assayed on d 7. MiR-24-, miR-30b or miR-142-3p-transfected MΦ exhibited decreased (∼25–35%) CD32 expression while, miR-30b and miR-142-3p significantly down-regulated (25–35%) CD32 expression in DCs (Figure 5a, b). No significant changes were observed in CD32 levels in miR-24-transfected DC (Figure 5b).
miR-24, miR-30b and miR-142-3p down-regulate expression of Fc receptors. Cells were transfected with miRNA mimics, inhibitors or control mimics at d 3 and surface expression of FcR were monitored at d 7 by flow cytometry. FACS analysis showing reduced CD32 positive in miR-24, miR-30b and miR-142-3p transfected (a) MΦ and miR-30b and miR-142-3p transfected (b) DC. Surface expression of CD23 in (c) untreated or (d) IFN-γ stimulated MΦ. (e) Reduced CD23 expression correlates with decreased TNF-α secretion by MΦ. At d 3 miRNA or control mimic transfected cells were incubated with IgE (1 µg/ml) and supernatant levels of TNF-α were examined by ELISA. Histograms showing miRNA mediated down-regulation FcɛR1G expression assessed by flow cytometry in (f) MΦ and (g) DC. Student’s t-test was conducted to calculate P values. *P < 0.05, **P < 0.01, ***P < 0.001.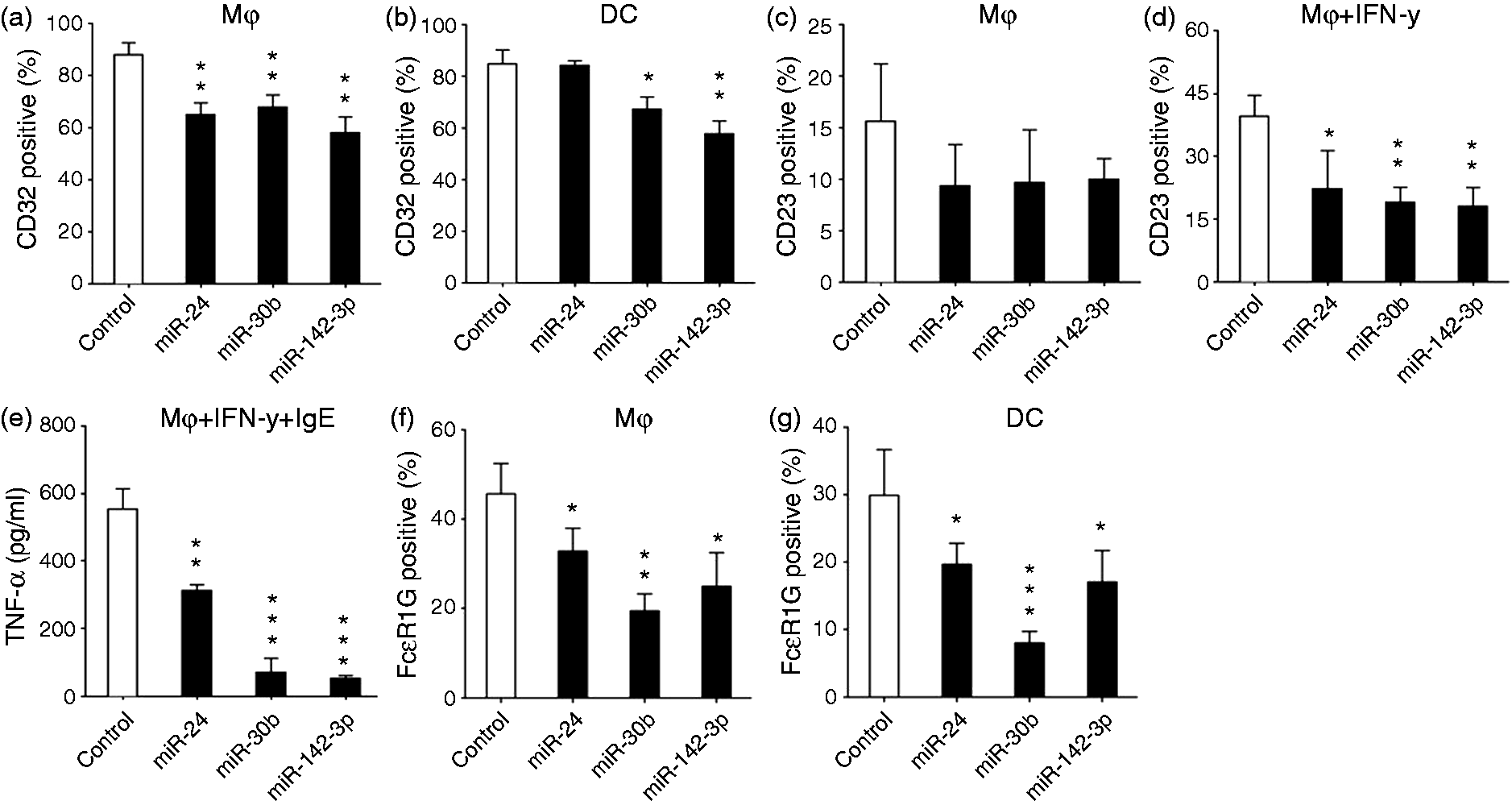
Next, we checked surface expression of CD23 and found low levels of CD23 expression on MΦ but not DC (data not shown). Subsequent experiments were therefore performed with MΦ to determine the impact of miRNA mimics on CD23 expression. Cells transfected on d 3 of differentiation with miR-24, miR-30b and miR-142-3p mimics displayed a trend for lower CD23 expression at d 7, but this was not statistically significant (Figure 5c). However, IFN-γ-induced up-regulation of CD23 expression was significantly reduced in cells overexpressing miR-24, miR-30b and miR-142-3p (Figure 5d). Ligation of FcR leads to the activation of pro-inflammatory cytokine production, including TNF-α. Because CD23 is a low-affinity IgE receptor, we therefore examined the secreted levels of TNF-α in miRNA-transfected cells stimulated with IFN-γ and IgE. MiRNA-mediated inhibition of CD23 expression translated to lower TNF-α production as a result of CD23 cross-linking via the addition of IgE (Figure 5e).
Finally, the impact of miR-24, miR-30b and miR-142-3p transfection on the expression of FcɛR1G was examined. Compared with control mimic, significantly reduced levels of FcɛR1G were observed in miR-24, miR-30b and miR-142-3p transfected MΦ and DC (Figure 5f and g). However, miR-30b mediated down-regulation was more pronounced compared with either miR-24 or miR-142-3p. Overall, these results demonstrate negative correlation of miRNA overexpression with FcR genes.
FcɛR1G expression is post-transcriptionally regulated by miR-30b
FcɛR1G is a cytoplasmic subunit of high-affinity IgE-binding FcɛR1 receptor and key determinant of maturation and surface expression levels of FcɛRI on APCs. The FcɛR1G also interacts with other Fc gamma (FCGR1 and FCGR3) and alpha (FCAR) receptors to form functional signaling complexes indicative of its association in IgG and IgA recognition and signaling. 32 We therefore focused on elucidating the mechanisms underlying miRNA-mediated FcɛRIG down-modulation. The FcɛR1G gene is transcriptionally regulated by multiple transcription factors, including Sp-1, Elf-1 and GAPBα. 33 Because we noticed miRNA-mediated changes in the FcɛRIG expression both at mRNA and protein level, we questioned if these changes are indirectly associated with reduced expression of transcription factors. To address this, the mRNA levels of FcɛRIG-associated transcription factors were examined in miR-24-, miR-30b- and miR-142-3p-transfected MΦ and DC. Quantitative PCR analysis showed that expression of all the three transcription factors, Sp-1, Elf-1 and GABPα, exhibit similar expression profile in miR-24, miR-30b and miR-142-3p or control mimic transfected cells (Supplementary Figure S2). These results suggest miRNA-mediated changes in the FcɛR1G gene is either caused by their direct interaction with FcɛR1G 3′UTR or indirectly through other regulatory factors.
To determine if the regulation of FcɛR1G genes is directly mediated by miR-24, miR-30b or miR-142-3p, we screened the 3′ UTR of FcɛR1G genes. Using the miRwalk search engine, we identified a conserved miR-30b binding site spanning 168–189 nts on the FcɛR1G 3′ UTR (Figure 6a). Besides FcɛR1G, we identified various FcR/Fc receptor-like (FcRL) genes with putative miRNA binding sites (Supplementary Table S1). However, miR-30b binding sites were most consistent predicted by five out of 10 different programs selected. FcɛR1G was one of the FcR genes down-regulated in miR-30b overexpressing MΦ and DC.
21
To confirm this in silico prediction, we cloned the complete 3′UTR downstream to renilla luciferase in psiCHECK-2 vector. Dual luciferase assays show significant reduction (∼30%) in reporter gene activity in the presence of miR-30b (1pmol) compared with control mimic transfection (P < 0.001; Figure 6b). We noticed a further reduction ( ∼ 50%) in renilla activity upon increasing the miR-30b concentration to 5 pmol (Figure 6b).
FcɛR1G is a novel target of miR-30b. (a) Sequence alignment of predicted miR-30b binding site in the 3′UTR of FcɛR1G. (b) HEK293 cells were co-transfected with FcɛR1G 3′UTR construct and with either miR-30b (1 and 5 pmol) or control mimic. Renilla activity was normalized to firefly activity, and the ratios subsequently normalized to empty vector transfected with miR-30b mimic set as 1. Data are expressed as ± SEM of three independent experiments. (c) Primary MΦ and DCs were transfected with four siRNAs targeting different regions of FcɛR1G transcript. After 48 h, expression of FcɛR1G was examined by RT-PCR to assess knockdown by each siRNA. GAPDH served as endogenous control. (d) MΦ and DC transfected with four siRNA targeting FcɛR1G. After 48 h, phagocytosis assays were performed using IgG opsonized beads. Cell were harvested after 2 h and analyzed on flow cytometry and the geo. MFI values (in %) are presented. Data are expressed as ± SEM of four independent transfections. *P < 0.05, ***P < 0.001.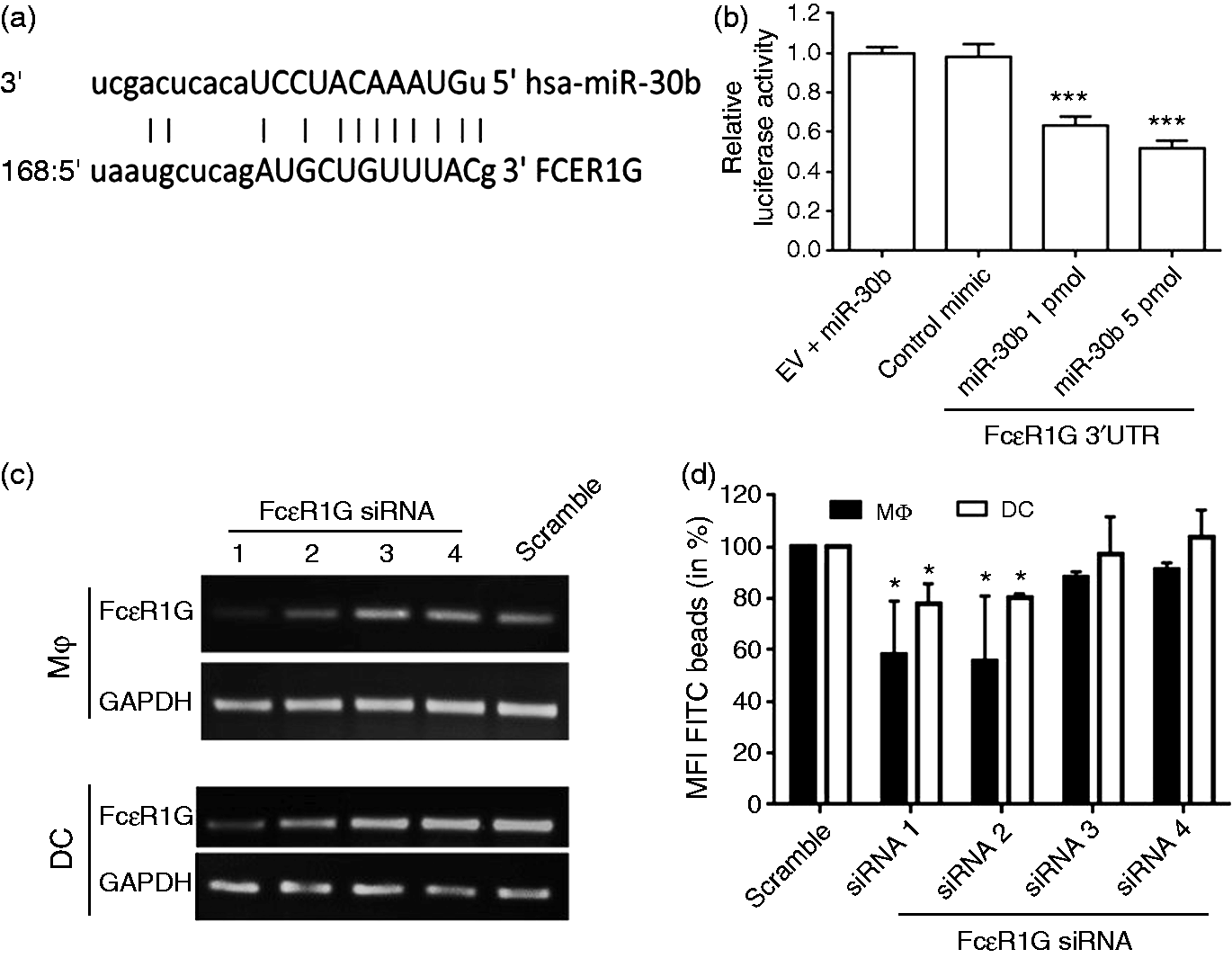
Finally, to evaluate the role of FcɛR1G in the uptake of IgG-opsonized beads, both MΦ and DC were transiently transfected with FcɛR1G-targeting siRNAs. We screened four siRNAs targeting different regions of FcɛR1G. Quantitative RT-PCR shows that efficient RNAi was achieved with siRNA 1 and 2 but not siRNA 3 or 4 (Figure 6c). We then performed phagocytosis assays in MΦ and DC transfected with FcɛR1G-targeting siRNAs. Cells transfected with siRNA 1 or 2 but not siRNA 3 or 4 show reduced uptake of IgG-opsonized beads compared with scramble control (Figure 6d). These results unequivocally show that specific silencing of FcɛR1G impairs Ab receptor-mediated phagocytosis.
Discussion
In this study we demonstrate a significant role for miR-24, miR-30b and miR-142-3p in regulating the uptake of Ab receptor-mediated phagocytosis. Recently, we reported that overexpression of these miRNAs negatively affects phagocytosis of both Gram-positive and Gram-negative bacteria. 21 These results suggest a universal inhibitory role of these miRNAs in mediating particle internalization and activation of innate immune responses. This is further supported by our data showing that the expression of these miRNAs was responsive to challenge with Ab-opsonized beads and bacteria at an early time point, suggesting transcriptional inhibition of these miRNAs upon myeloid cell activation. Consistent with this, others and our laboratory have reported notable reduction in the expression of these miRNAs during myeloid cell differentiation, indicating that their down-regulation is required to acquire APC functionality.20,21,23,34–36
Opsonized particles are recognized by FcRs present on various immune cells. In humans, eight FcR-encoding genes have been identified. These include three high-affinity receptors, FcγRIA, FcγRIB and FcγRIC, and five low-affinity receptors, FcγRIIA, FcγRIIB, FcγRIIC, FcγRIIIA and FcγRIIIB.37,38 Exposure to pro-inflammatory cytokines like TNF-α and IFN-γ or LPS induces expression of activating FcγRs, thereby promoting Ab-mediated phagocytosis. 32 In agreement, this study and previous results show that overexpression of miR-24, miR-30b and miR-142-3p suppresses secretion of TNF-α during phagocytosis of Ab-opsonized beads and bacteria. 21 Similarly, miR-142-3p-mediated down-regulation of IL-6 secretion upon TLR4 activation, indicating multiple levels of immune response regulation.21,39 This correlates with the reduced expression of various FcR transcripts, including FCGR1A, FCGR2A, FCGR3B, FCER2, FCER1A and FCER1G. 21 In this study we show down-modulation of FCGR2A and FCER2 at the protein level which correlated with reduced secretion of TNF-α by miR-24, miR-30b and miR-142-3p overexpressing cells. FcRs are also associated with Ag processing/presentation. Indeed, both activating and inhibitory FcRs control DC activation and adaptive cellular immune responses.40,41 In our Ag uptake and processing assays, we noted marked reduction in both of these concurrent processes in miRNA-overexpressing APCs (Naqvi et al., accepted). Together, these results provide strong evidence for the potential role of these miRNAs in regulating FcR-mediated innate and adaptive immune responses.
Several pathogens, especially viruses, exploit Ab-mediated uptake as a pathway to internalize and infect target cells.42,43 Delivery of pathogens to APCs and concomitant activation of immune responses through this pathway play a protective role against intracellular pathogens. Our results show that overexpression of miRNAs negatively regulates Ab-mediated phagocytosis and the expression of multiple FcRs. This can profoundly affect the ability of APCs to productively present Ag to T cells and Th cell skewing.44,45 Indeed, FcRs are important in pathogen clearance and altered expression has been reported during pathogen infection. For instance, HIV infection inhibits FcR-mediated phagocytosis by monocyte-derived MΦ, primarily by interfering with Fc gamma RI signaling through post-transcriptional down-regulation of gamma-chain mRNA.46,47 Because miRNAs are key regulators of post-translational silencing, it is highly likely that miRNA may mediate this effect. Consistent with the above observations, we show that enforced expression of miR-30b repress CD32 and CD23 in both DC and MΦ and directly regulates its novel target FcɛR1G by binding to the 3′UTR. Both CD32 and CD23 are activating receptors and changes in their surface expression will have a profound effect on FcR-mediated phagocytosis. This is further supported by the observation that miR-30b was the most significantly induced miRNA in HIV BaL (CCR5-tropic)-infected CD4+ PBMCs suggesting an important role in FcγR-mediated phagocytosis and pathogen clearance. 48 Our findings of miRNA-mediated regulation of phagocytosis may be of relevance in elucidating mechanisms underlying pathological disorders and pathogen infection.
Cross-linking of FcR by IgG or IgE stimulates downstream signaling cascades leading to the activation of NF-κB and NFAT that control transcription of pro-inflammatory cytokines, including TNF-α, IL-6, IL-8, IL-12 and others.49–52 Our results show reduced secretion of TNF-α and IL-8 (in MΦ) following challenge with IgG-opsonized beads in miRNA mimic-transfected cells. Similarly, we noted lower TNF-α secretion in IgE-stimulated MΦ overexpressing miRNA mimics. We have previously demonstrated down-regulation of NF-κB subunit p65 by miR-24, miR-30b and miR-142-3p in LPS-stimulated APCs. 20 However, compared with TLR signaling we noted comparatively less miRNA-mediated impact on cytokine secretion upon FcR activation, which likely resulted owing to differences in the PRR-specific cytokine responses. These results underscore the finding that miRNAs can directly target components involved in activation of pro-inflammatory pathways, thus highlighting their potential anti-inflammatory role. In Langerhans cells, high levels of FcɛR1 are associated with the pathophysiology of atopic dermatitis.53–55 Further, we showed that FcɛRIG, a common signaling subunit for FCGR1A and FCGR3A, is down-regulated at the protein level by miR-24, miR-30b and miR-142-3p. However, we did not notice any changes in expression of transcription factors that regulate FcɛRIG transcription, suggesting that these miRNAs do not alter transcription of FcɛRIG but regulate it post-transcriptionally. We only identified a putative binding site for miR-30b but not miR-24 or miR-142-3p on the FcɛRIG 3′UTR and confirmed this interaction using dual luciferase reporter assays. This indicates that other indirect pathways through which miR-24 or miR-142-3p could act to reduce FcɛRIG levels may be present and require further investigation. Importantly, our results demonstrate a role of FcɛRIG in the phagocytosis of IgG-opsonized beads as observed by attenuated uptake in APCs with FcɛRIG RNAi. The FcɛRI, in human APCs, is a trimeric protein made up of one FcɛRIα and two FcɛRIγ subunits. Moreover, besides acting as a signal-transducing unit of FcɛRI, the gamma subunits are postulated to be the key determinants of maturation and surface expression levels of FCGR1A, as well as FCGR3A, on APCs.56–58 Lack of functional FCGR3A is also demonstrated in FcɛRIG–/– mice. Moreover, FcɛRIG–/– mice exhibit reduced opsonophagocytosis and increased susceptibility to fungal and bacterial infections.59,60 During differentiation of monocyte-derived MΦ and DC, the expression of FcɛRI increases remarkably while miR-30b levels are down-regulated. This indicates an antagonistic expression pattern of miR-30b and its novel target FcɛRIG. These observations unequivocally demonstrate that aberrant expression of miRNAs can modulate MΦ and DCs activation and underlies their role in precisely fine tuning these responses.
In conclusion, the results of this study elucidate a new mechanism whereby miR-24, miR-30b and miR-142-3p influence uptake of IgG-opsonized beads by modulating expression of FcRs. These observations suggest that endogenous miRNAs exert an important role in regulating phagocytosis, a crucial function of APCs, and highlight their therapeutic potential for the treatment of pathological conditions associated with poor Ag uptake and clearance by mononuclear phagocytes.
Supplemental Material
Supplemental material for MicroRNA target Fc receptors to regulate Ab-dependent Ag uptake in primary macrophages and dendritic cells
Supplemental Material for MicroRNA target Fc receptors to regulate Ab-dependent Ag uptake in primary macrophages and dendritic cells by Afsar Raza Naqvi, Jezrom B Fordham and Salvador Nares in Innate Immunity
Footnotes
Acknowledgments
We thank Dr. Ganesh Balaji for his valuable help with flow cytometry.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the NIDCR/NIH [DE021052] and in part by the UIC College of Dentistry Edward C. Wach Fund.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
