Abstract
The T-cell adjuvanticity of mycobacterial cord factor trehalose 6,6'-dimycolate (TDM) is well established. The identification of the C-type lectin Mincle on innate immune cells as the receptor for TDM and its synthetic analogue trehalose 6,6'-dibehenate (TDB) has raised interest in development of synthetic Mincle ligands as novel adjuvants. Trehalose mono- (TMXs) and diesters (TDXs) with symmetrically shortened acyl chains [denoted by X: arachidate (A), stearate (S), palmitate (P), and myristate (M)] were tested. Upon stimulation of murine macrophages, G-CSF secretion and NO production were strongly augmented by all TDXs tested, in a wide concentration range. In contrast, the TMXs triggered macrophage activation only at high concentrations. Macrophage activation by all TDXs required Mincle, but was independent of MyD88. The superior capacity of TDXs for activating macrophages was paralleled by direct binding of TDXs, but not of TMXs, to a Mincle-Fc fusion protein. Insertion of a short polyethylene glycol between the sugar and acyl chain in TDS reduced Mincle-binding and macrophage activation. Immunization of mice with cationic liposomes containing the analogues demonstrated the superior adjuvant activity of trehalose diesters. Overall, immune activation
Introduction
Activation of innate immune cells with structurally well-defined synthetic ligands for PRRs is an attractive strategy for development of novel adjuvants for immunization purposes. The C-type lectin receptor Mincle is the receptor for the mycobacterial cord factor trehalose-6,6'-dimycolate (TDM),1,2 can also be activated by the synthetic glycolipid trehalose 6,6'-dibehenate (TDB), a strong Th1/Th17-inducing adjuvant.2,3 Mincle signalling is initiated by the associated immunoreceptor tyrosine-based activation motif-containing adapter protein Fc receptor gamma chain (FcRγ) that recruits the kinase Syk leading to activation of the Card9–Bcl10–Malt1 complex, which, in turn, promotes expression of target genes encoding chemokines, growth factors and cytokines.
Liposomes based on TDB and the cationic surfactant dimethyldioctadecylammonium (DDA) bromide [also referred to as cationic adjuvant formulation 1 (CAF01); Statens Serum Institut, Denmark] have been shown to be a promising adjuvant in several preclinical animal vaccination models using recombinant protein antigens originating from
Currently, there is intense interest in further development of synthetic Mincle ligands for use as adjuvants. Of particular interest are compounds with increased solubility as compared to TDB, and which are readily synthesized. Recent publications describing synthetic trehalose-based glycolipids include fatty acid esters and synthetic corynomycolates.8–11 Besides introducing new vaccine adjuvant candidates, such studies also generate valuable mechanistic knowledge that defines the minimal requirements for Mincle-dependent activation of macrophages in terms of sugar headgroup and acyl chain length.
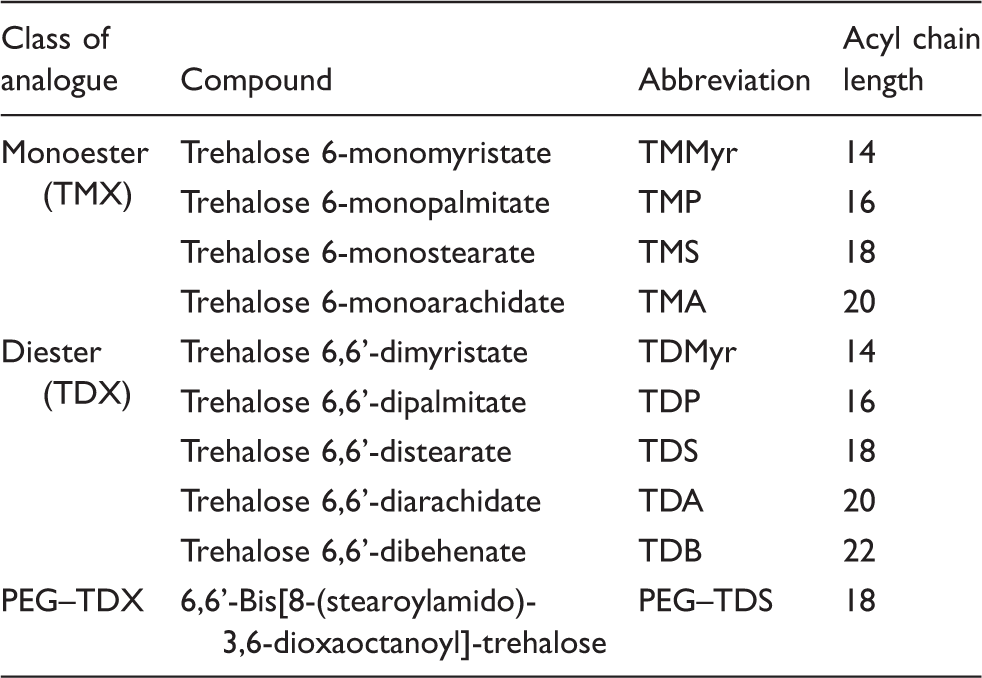
Here, we tested an array of synthetic TDB analogues (Figure 1A) to elucidate the influence of: (i) the chain length, (ii) the degree of acylation (mono- vs. diesters) and (iii) the headgroup display by insertion of a short, hydrophilic polyethylene glycol (PEG) spacer between the headgroup and the acyl chain. The analogues were tested for their capacity to bind to murine Mincle Study overview and structure of TDB analogues. (A) Graphical overview of the study. (B) Structures of mono- (TMX) and diester (TDX) analogues of TDB with symmetrically shortened acyl chain, and an analogue (PEG–TDS) with a hydrophilic spacer inserted between the trehalose headgroup and each of the acyl chains.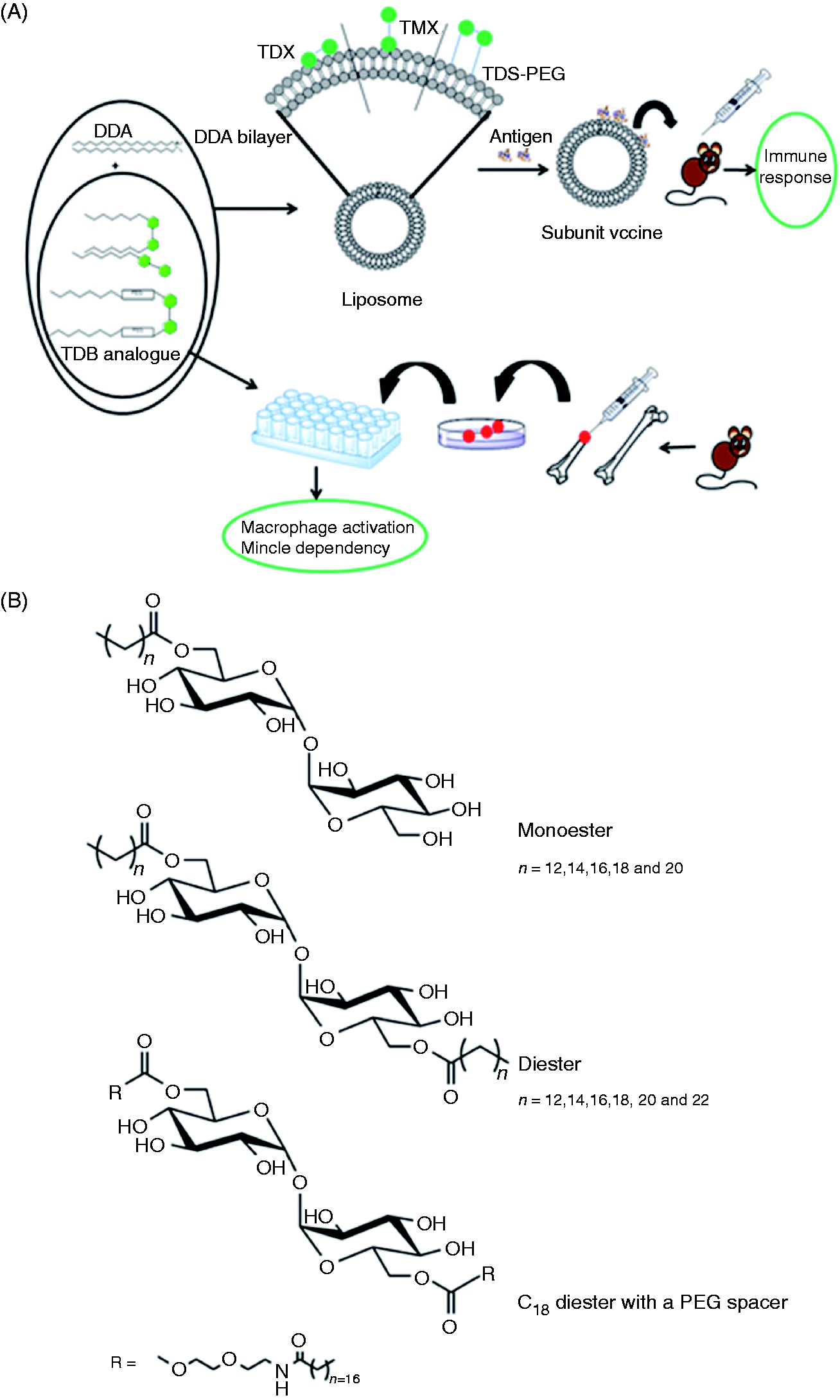
Materials and methods
Materials
DDA was obtained from Avanti Polar Lipids (Alabaster, AL, USA). Two reference batches of TDB were obtained from Avanti Polar Lipids (referred to as TDB-1) and synthesized by Clausen-Kaas A/S (Farum, Denmark; referred to as TDB-2). LPS was from Sigma (catalogue number L2880; St Louis, MO, USA), recombinant murine IFN-γ was from Peprotech (Hamburg, Germany), B-type CpG ODN 1826 was from TIB MOLBIOL (Berlin, Germany) and TDM prepared form
Synthesis of trehalose esters
Array of TDB analogues.
Differentiation of macrophages and murine BMDC
C57BL/6 mice were originally obtained from Charles River (Sulzfeld, Germany). Mincle-deficient (Clec4e–/–) mice have been described and were originally generated by the Consortium of Functional Glycomics on a C57BL/6 background.2,12 MyD88–/– mice were obtained from Dr Shizuo Akira and maintained on a C57BL/6 background. C57BL6, Mincle-deficient and MyD88–/– mice were bred at the animal facility of the Medical Faculty of the Friedrich-Alexander University Erlangen-Nürnberg (Germany). The mice were allowed free access to water and food. Sex-matched mice were euthanized at 8–12 wk of age. The bone marrow from the tibias and femurs was flushed with cold PBS; after erythrocyte lysis, bone marrow cells were set into 10-cm Petri dishes (Sarstedt, Kleinstadt, Germany) for overnight (between 16 and 24 h) adherence in cDMEM supplemented with 10% FCS (Biochrom, Berlin, Germany), 1% penicillin/streptomycin (Sigma Aldrich) and 50 µM β-mercaptoethanol, with 10% (v/v) L cell-conditioned medium (LCCM) as a source of M-CSF. On the following day, non-adherent cells were set into 10-cm Petri dishes at 5 to 6 × 106 cells per 10-cm petri dish (medium as before). On day 3, 5 ml cDMEM + 10% (v/v) LCCM were added, and differentiated bone marrow macrophages (BMM) were harvested on day 6 or 7. For generation of DCs, bone marrow cells were cultured in cDMEM + 10% (v/v) conditioned medium of X63 cells producing GM-CSF for 7 d with additional supplementation with GM-CSF-containing medium on d 3 and d 5 of culture. Non-adherent differentiated bone marrow dendritic cells (BMDCs) were harvested by gentle pipetting on day 7, washed and counted.
Stimulation of macrophages and DC with glycolipids coated on dishes
TDB and the TDB analogues were dissolved in sterile isopropanol and stored at −20℃. For coating, the solutions were heated to 60℃ until the solutions were completely clear. Dilutions were prepared in sterile isopropanol and administered to 96-well cell culture plates. After evaporation of isopropanol, the plates were used for stimulation of BMM or BMDC. Soluble control stimuli (LPS, IFN-γ) were diluted in cDMEM. BMM were harvested with Accutase (Life Technologies, Carlsbad, CA, USA) and administered to 96-well plates, coated with the stimuli, at a density of 0.2 × 106 cells per well. Stimulations were performed in triplicate (BMM) or duplicate (BMDC). For each condition, single stimulation and co-stimulation with IFN-γ (50 ng/ml) was performed. After 48 h, the morphology of the cells was observed and the supernatants were harvested.
Measurement of G-CSF by ELISA
G-CSF production of single stimulations (i.e. no IFN-γ) in the supernatants was assessed by a sandwich ELISA (R&D, Minneapolis, MN, USA), performed according to the manufacturer‘s protocol.
Determination of nitrite levels using the Griess assay
The NO production in IFN-γ co-treated BMM was assessed by measuring the nitrite levels with the Griess assay. Nitrite levels are displayed as Δnitrite = nitrite [stimulus] – nitrite [isopropanol].
Binding of Mincle–Fc fusion protein to plate-coated glycolipids
Production of the mouse Mincle–Fc fusion protein has been previously described. 13 Briefly, cDNA encoding the extracellular part of murine Mincle was ligated into the pFuse–hIgG1–Fc expression vector (InvivoGen, San Diego, CA, USA). The Mincle–Fc-encoding vector was transiently transfected into CHO cells using the FreeStyle Max CHO-S Expression System (Life Technologies, Darmstadt, Germany). The cell supernatant containing Mincle–Fc was collected, and Mincle–Fc was purified using a HiTrap Protein G HP column (GE Healthcare, Piscataway, NJ, USA). SDS-PAGE with subsequent Coomassie stain, as well as Western Blot, were performed to confirm the identity and the purity of Mincle–Fc. The Mincle–Fc concentration was determined using the Micro BCA Protein Assay Kit (Thermo Scientific, Rockford, IL, USA). The plates were coated with the glycolipids as described above. If not otherwise indicated, 2 µg of each trehalose ester per well were used. The plates were blocked with 3% (w/v) BSA in HBSS, and the purified Mincle–Fc fusion protein was applied. After extensive washing with HBSS, bound fusion protein was detected with an HRP-coupled anti-Fc Ab and addition of 3,3',5,5'-tetramethylbenzidine substrate.
Preparation of liposomes
Liposomes consisting of binary mixtures of DDA and glycolipid (TDB-2, TDS, TMS or PEG–TDS) at a molar ratio of 89:11 were prepared by using the thin film method essentially as previously described, combined with probe sonication to ensure a narrow size distribution of the resulting liposomes.8,14 Briefly, weighed amounts of DDA and glycolipid were dissolved in CHCl3-MeOH (9:1, v/v) in a round-bottomed flask. The organic solvents were evaporated under vacuum resulting in the formation of a thin lipid film. The film was stripped twice with EtOH and dried overnight to remove trace amounts of organic solvents. The lipid film was rehydrated by adding Tris buffer (10 mM, pH 7.4), and sonicated for 5 min using a Sonifier® cell disruptor (Branson, Danburry, CT, USA), followed by heating to 60℃ for 1 h with 2 min of vigorous vortex mixing every 10 min. After 20 min of rehydration, the liposomes were tip-sonicated for 20 s with a 150 W Branson tip-sonicator (50% of the duty cycle) to reduce the size of the liposomes. The final concentration of DDA and TDB-2/TDS/TMS/PEG–TDS were 2.5 mg/ml and 0.5, 0.44, 0.31 or 0.59 mg/ml, respectively, corresponding to a molar ratio of 89:11. The liposomes were stored at 4℃ until further use. For each immunization, fresh batches of liposomes were prepared immediately prior to immunization. The quality of each liposome batch was confirmed by measuring the particle size distribution, the polydispersity index and the zeta-potential, as previously described (results not shown). 8
Immunization of mice
Female C57BL/6 mice were obtained from Harlan Laboratories (Boxmeer, The Netherlands). The mice were allowed free access to water and food. The immunization experiments were approved by the Danish Council for Animal Experiments and done in accordance with EU directive 2010/63EU for animal experiments. The vaccines were prepared by mixing the liposomal dispersions with 5 µg
Detection of IFN-γ and IL-17a from activated T cells by ELISA
Isolated splenocytes were cultured in duplicate in round-bottomed 96-well microtiter plates at a density of 2 × 105 cells/well in a volume of 200 µl RPMI supplemented as described above. The cells were re-stimulated with 5 µg/ml MOMP and allowed to incubate for 3 d at 37℃. Wells containing medium alone or 3 µg/ml concanavalin A (Sigma-Aldrich) were included as negative and positive controls, respectively. After incubation, supernatants were collected, and the IFN-γ and IL-17a production was quantified by a standard ELISA protocol. Briefly, purified rat α-mouse IFN-γ or IL-17a (BD Biosciences, San Jose, CA, USA) were used as capture Abs, and biotin-conjugated rat α-mouse IFN-γ or IL-17a (BD Biosciences) were used as detection Abs, respectively, followed by HRP-conjugated streptavidin (BD Biosciences) and 3,3’,5,5’-tetramethylbenzidine Plus Ready-to-use substrate (Kem-En-Tec, Taastrup, Denmark). The reaction was stopped at the optimal colour development with 0.2 M H2SO4, and the absorbance was read at 450 nm with wavelength correction at 570 nm.
Intracellular cytokine staining analysis
Isolated splenocytes (1 × 106 cells/well) were stimulated for 1 h with 5 µg/ml MOMP in the presence of 1 µg/ml anti-CD28 and anti-CD49d mAbs (both BD Pharmingen, San Diego, CA, USA) in a total volume of 200 µl supplemented with RPMI 1640. The cells were subsequently incubated for 5 h at 37℃ after addition of 10 µg/ml brefeldin A (Sigma Aldrich) and 0.7 µl/ml monensin/Golgi-stop (BD Pharmingen). Following overnight storage at 4℃, the cells were washed in FACS buffer and stained with anti-CD4 (APC) and anti-CD44 (FITC) mAbs (BD Biosciences) in FACS buffer for 30 min at 4℃. Cells were washed with FACS buffer, permeabilized by using the Cyto-fix/Cyto-perm kit (BD Pharmingen) according to the manufacturer’s instructions, and stained intracellularly for 30 min at 4℃ using anti-IFN-γ (PE–Cy7) and anti-IL-17a (PerCP–Cy5.5) mAbs (eBiosciences, San Diego, CA, USA) in Perm wash buffer. After washing, the cells were re-suspended in FACS buffer and analysed by flow cytometry using a six-colour BD FACS Canto flow cytometer (BD Biosciences). Responses were analysed with the FlowJo software (Tree Star Inc., Ashland, OR, USA).
Statistical analysis
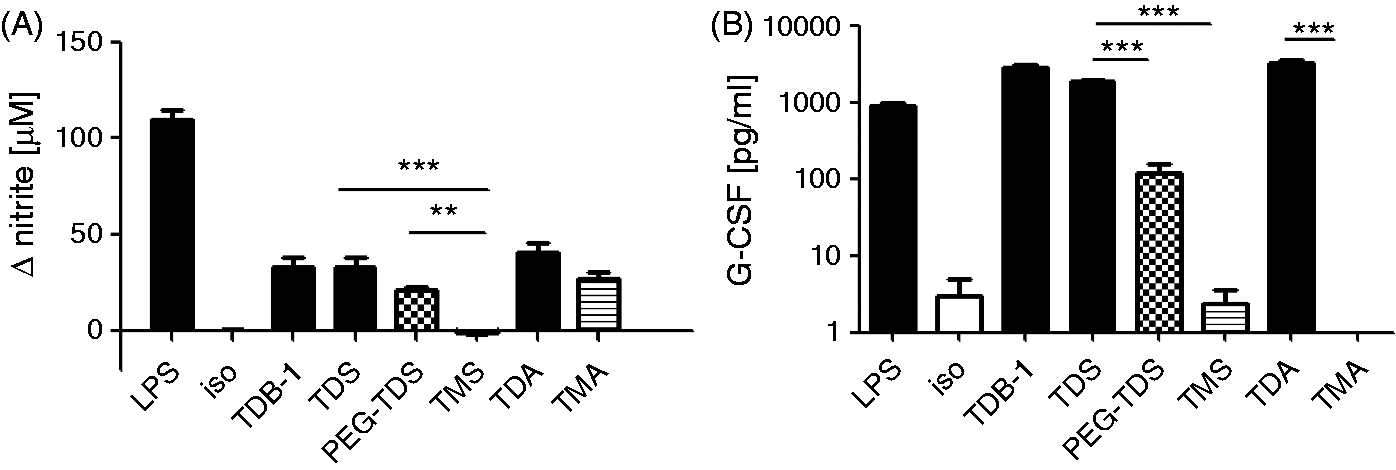
Graphical display and statistical analysis of the Comparison of macrophage activation by trehalose mono- and diesters. Murine BMM were stimulated for 48 h with 5 µg/ml of the indicated compound. For monitoring NO production, 50 ng/ml IFN-γ was added. CpG (0.5 µM), LPS (100 ng/ml) and TDM (5 µg/ml) served as positive controls, respectively, while isopropanol served as negative control. (A) NO levels are displayed as ΔNO, i.e. (with stimulus) – (isopropanol control). (B) The G-CSF values are displayed as absolute concentrations. Data represent at least nine replicates per condition pooled from four independent experiments, except for TDM (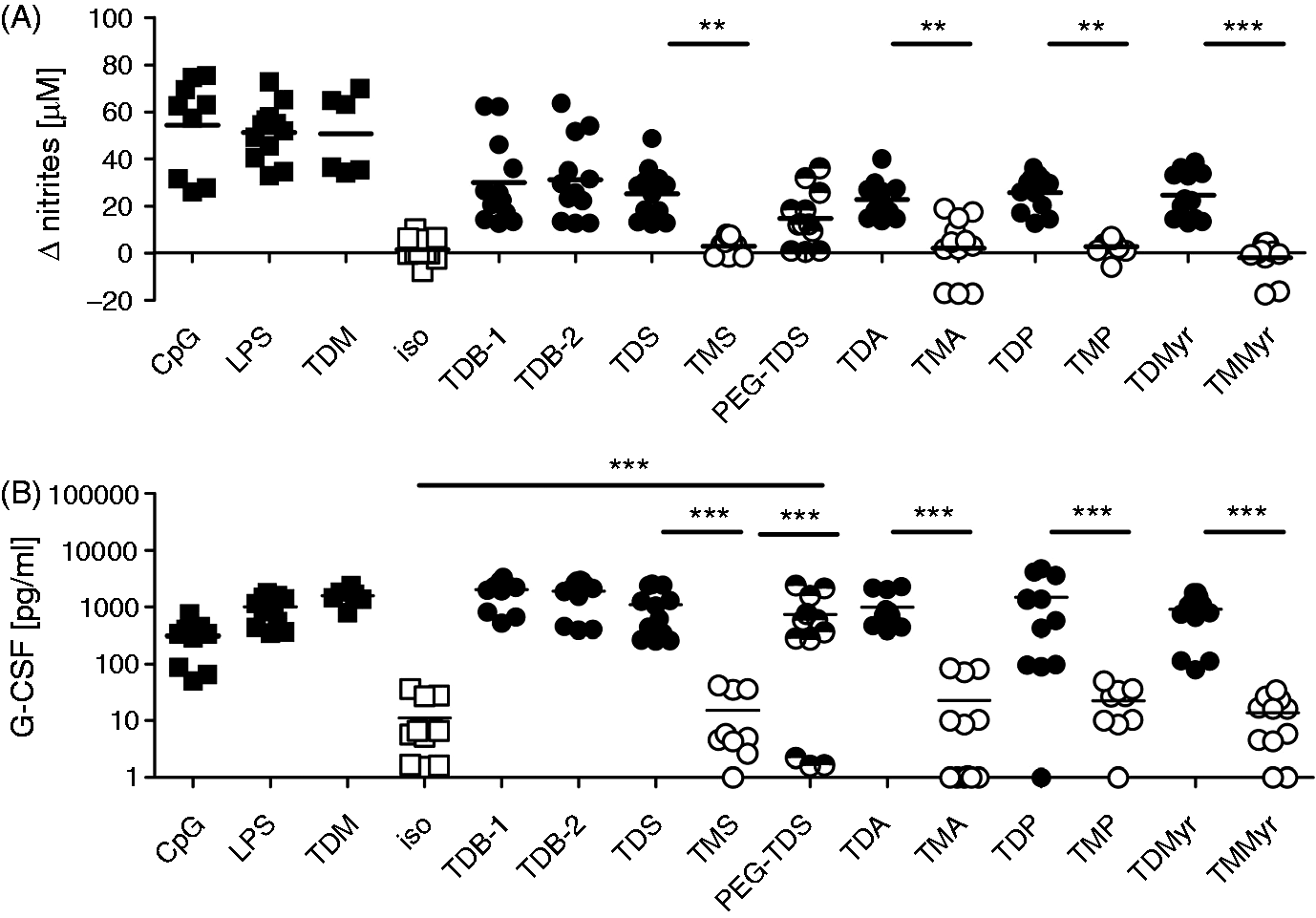
Results
Superior activation of macrophages by trehalose diesters as compared to the monoesters
To investigate the stimulatory activity of monoester and diester analogues of TDB, displaying different acyl chain length, on macrophages (Figure 1B, Table 1), mouse BMM differentiated in the presence of M-CSF were seeded into wells coated with synthetic glycolipids. The production of G-CSF by macrophages after 48 h of incubation was determined in the cell culture supernatants by ELISA. The release of NO by macrophages, co-stimulated with IFN-γ in addition to the glycolipids, was measured after 48 h by using the Griess assay. First, we compared the stimulatory capacity of the compounds at a fixed concentration of 5 µg/ml (corresponding to 1 µg coated glycolipid per well) (Figure 2). The TLR ligands LPS and B-type CpG ODN 1826, as well as the natural cord factor TDM, served as stimulation controls. Reference TDB samples obtained from two different commercial suppliers (TDB-1 and TDB-2) were included as positive controls, and both gave rise to comparable levels of G-CSF and NO production (Figure 2). While all TDXs tested induced robust production of G-CSF and NO, the TMX did not induce significant release of these mediators by the macrophages (Figure 2). However, the acyl chain length of the diester analogues did not strongly influence the output of NO and G-CSF by the BMM (Figure 2). The PEG–TDS analogue containing the hydrophilic spacer between the trehalose headgroup and the stearoyl chains did also induce significantly higher G-CSF production than the TMS analogue (Figure 2).
Since the lack of macrophage stimulation observed for the TMXs might indicate a quantitative, albeit not qualitative, difference, the analogues were titrated over a wide concentration range. The results for TDS and TMS are shown as representative examples in Figure 3. TDS stimulated the production of G-CSF in single-treated macrophages and of nitrite in IFN-γ-co-stimulated macrophages starting at a concentration of 0.2 µg/ml, with increasing responses up to 5 µg/ml (TDS). In contrast, the corresponding monoester TMS did not trigger measurable NO or G-CSF release at concentrations in the range of 0.2–5.0 µg/ml. At the higher concentration of 25 µg/ml robust NO release and very low levels of G-CSF were detectable (Figure 3A, B). Increasing the glycolipid concentration to 125 µg/ml again abrogated the production of both G-CSF and NO for TMS (Figure 3A, B), which was due to TMS-induced toxicity (data not shown). Dose–response curves were also determined for the other TMX/TDX pairs (data not shown), confirming the results obtained with the stearate analogues and indicating the requirement for an approximately 100-fold higher concentration of the monoester analogue to elicit significant macrophage activation. PEG–TDS showed intermediate activity and reached similar levels of NO and G-CSF at approximately fivefold higher concentrations, as compared to TDS (Figure 3).
Dose–response curves of mono- and diester activity in a titration experiment. The trehalose mono- and diesters with stearic acid were used to stimulate BMM at a range of test concentrations. TDS was tested from 0.2 to 5.0 µg/ml, resulting in a clearly detectable response. TMS was tested from 0.2 µg/ml to a concentration of 25 µg/ml, while PEG–TDS was tested from 1 to 25 µg/ml. (A) ΔNO levels in BMM after 48 h in the presence of 50 ng/ml IFN-γ and (B) G-CSF from BMM stimulated for 48 h without IFN-γ. Data represent mean and SD of at least six replicates per condition pooled from two to four independent experiments. The error bars are sometimes smaller than the symbols. TDX are more potent stimulators of BMDC. The indicated glycolipids were coated in 96-well plates at a concentration of 5 µg/ml. LPS was used at 100 ng/ml in solution. BMDC were added to the plates. IFN-γ was (A) added or (B) not, and supernatants were harvested after 48 h. (A) NO was determined by Griess assay; (B) G-CSF by ELISA. Shown are mean and SD of six biological replicates per condition pooled from three independent experiments. Statistical analysis: one-way ANOVA with Bonferroni correction, **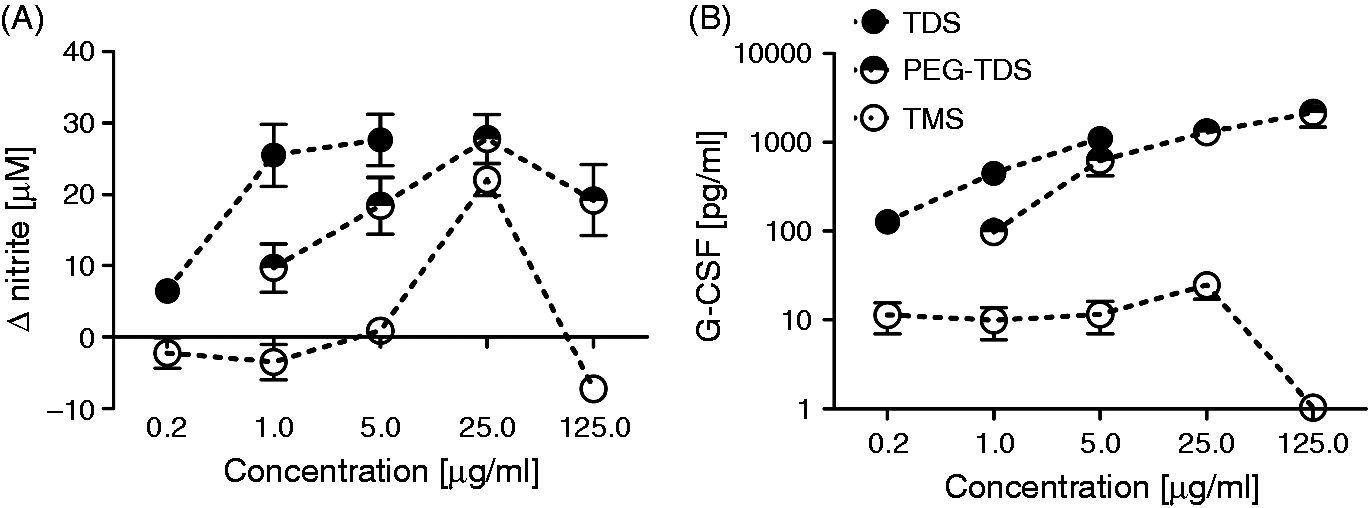
Subsequently we investigated whether the higher stimulatory capacity of TDX would be conserved between different antigen-presenting cell types. Therefore, we stimulated BMDC differentiated in the presence of GM-CSF with the trehalose esters of 18:0 or 20:0 fatty acids. For the stearic acid esters, the monoester TMS failed to increase production of NO and release of G-CSF, whereas TDS triggered strong production and PEG–TDS was moderately active (Figure 4A, B). The 20:0 arachidate monoester TMA induced NO release from BMDC but to a lesser degree as compared to the diester TDA (Figure 4A). TMA failed to trigger G-CSF production from BMDC (Figure 4B). Overall, the stronger activation of antigen-presenting cells by the TDXs as compared to TMXs was conserved between macrophages and DC.
Secretion of G-CSF and NO is abrogated in Mincle-deficient macrophages but intact in the absence of MyD88
TDB activates macrophages by direct binding to the C-type lectin receptor Mincle and via activation of the Syk-Card9 signalling pathway through the adapter protein FcRγ. Therefore, it was next tested whether Mincle is also required for the stimulatory capacity of the other TDXs investigated. Indeed, the release of G-CSF and NO in response to all TDXs were completely abolished in Mincle-knockout macrophages that responded normally to stimulation with the TLR4 ligand LPS and the TLR9 ligand CpG ODN 1826 (Figure 5). In contrast, MyD88-deficient macrophages showed a reduced response to LPS and were unresponsive to CpG stimulation but produced unaltered amounts of G-CSF or NO when stimulated with the glycolipids. Thus, TDX glycolipids with symmetric acyl chain lengths of 14–22 carbon atoms activate macrophages via Mincle signalling independently of MyD88. Notably, the G-CSF secretion triggered by PEG–TDS was dependent on both Mincle and MyD88 (Figure 5B), whereas the quite low levels of NO induced by this compound were not MyD88-dependent (Figure 5A).
TDX stimulate macrophages via Mincle independently of MyD88. The glycolipids were immobilized at a fixed concentration of 5 µg/ml in 96-well plates; isopropanol was used as a negative control. BMM of the indicated genotypes were added in (A) the presence or (B) the absence of IFN-γ. (A) NO and (B) G-CSF were measured in the supernatants after 48 h. Mean and SD of 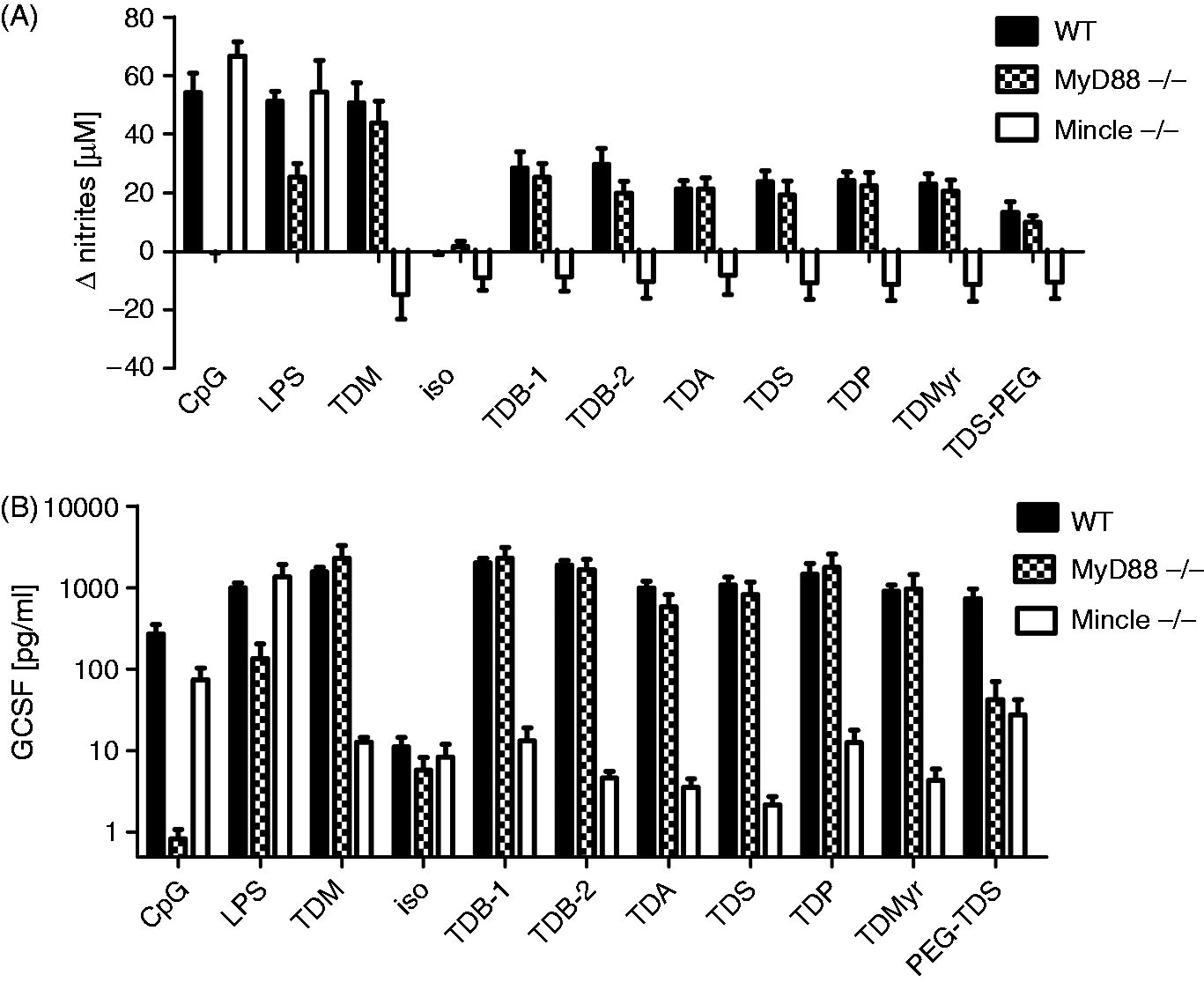
Binding of trehalose esters to Mincle-Fc protein correlates with macrophage activation
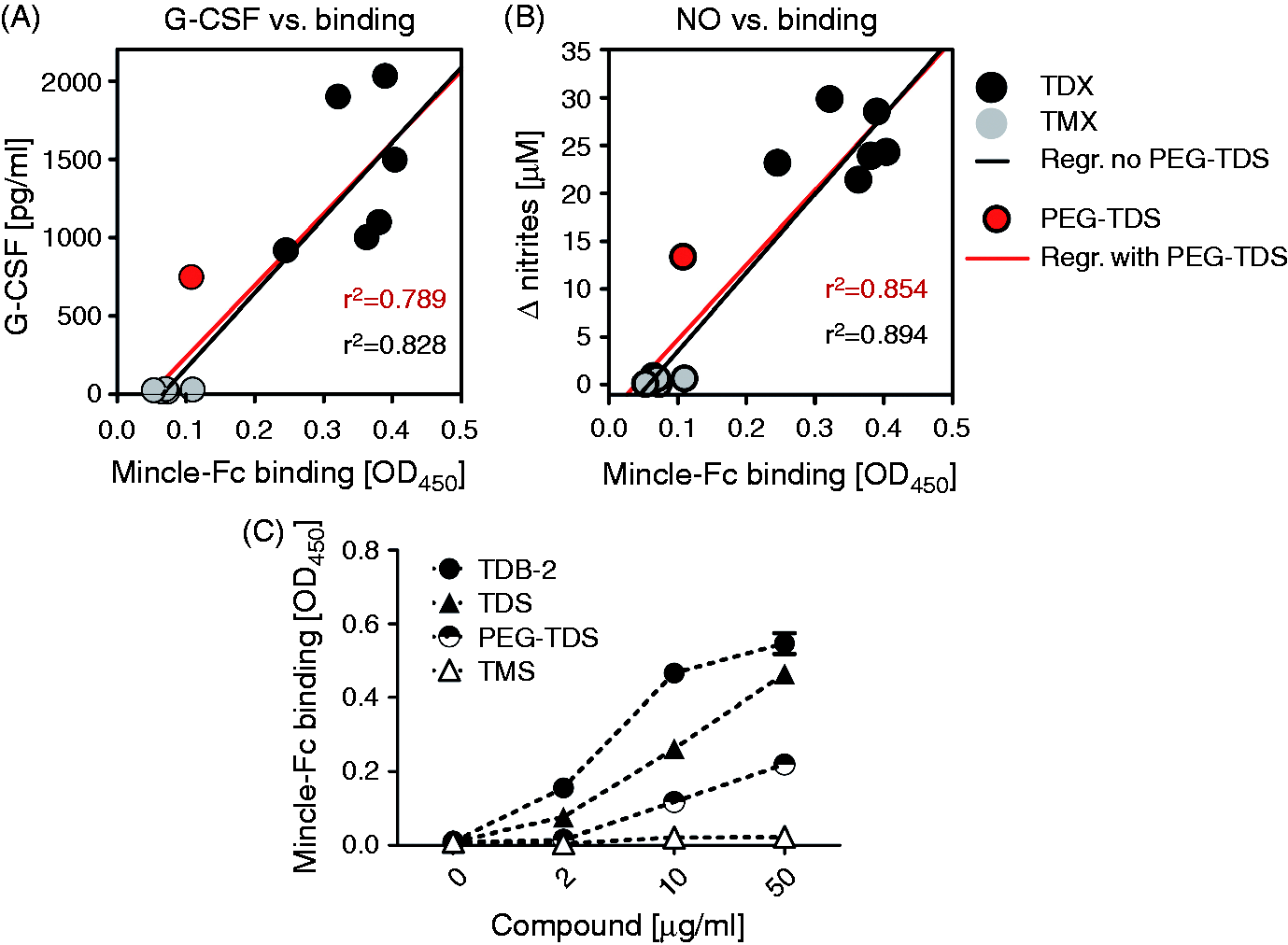
The strongly reduced macrophage activation by the TMXs, as opposed to the TDXs, may be due to a lack of binding to the Mincle receptor or to impaired activation of Syk-Card9, despite conserved binding to Mincle. To differentiate between these possibilities, we used a recombinant fusion protein of mouse Mincle and human Fc to test binding to the coated glycolipids (Figure 6).17,18 Using a fixed concentration of 10 µg/ml glycolipid, we observed comparably strong binding of this Mincle-Fc for all TDXs tested (i.e. TDB, TDA, TDS, TDP, TDMyr), whereas no detectable signal above the isopropanol background control was found for the monoester compounds (i.e. TMA, TMS, TMP, TMMyr) and PEG–TDS. To correlate the binding of glycolipids with their capacity to stimulate macrophages, we plotted the OD450 values from the Mincle–Fc assay against the mean values of G-CSF and NO production (Figure 7A, B). Overall, the ability to bind directly to Mincle correlated closely with the capacity of the glycolipids to trigger G-CSF and NO release from macrophages, except for PEG–TDS, which induced some macrophage activation without significant binding (Figure 7A, B). To address this discrepancy, the binding assay for the stearate analogues was extended to include a range of test concentrations for TDS, TMS and PEG–TDS. PEG–TDS showed binding to Mincle–Fc when coated at higher concentrations, whereas TMS did not bind, even at 50 µg/ml (Figure 7C).
Correlation between binding to Mincle and NO/G-CSF production. The data shown in Figures 2 and 5 were used to correlate the binding of the compounds to Mincle–Fc with: (A) G-CSF and (B) NO production. Linear regression and r2 coefficients of correlation are given in each graph, differentiating between inclusion (red) and exclusion (black) of PEG–TDS in the analysis. (C) Mincle–Fc binding assay using increasing amounts of glycolipids. Mean values of quadruplicate samples from one experiment.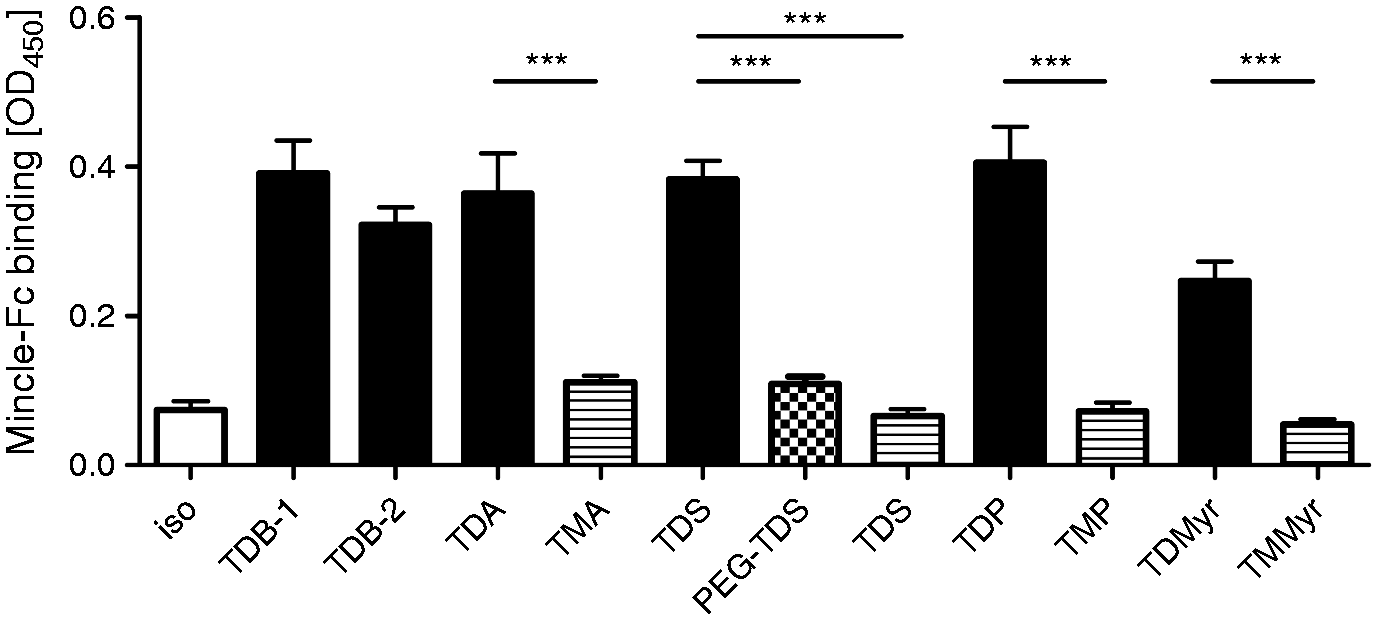
TDS incorporated into DDA liposomes induces more robust and superior Th1/Th17 responses than TMS and PEG–TDS in mice
Based on the data obtained in the
Co-administration of all liposomal dispersions, with the MOMP Ag surface-adsorbed to the liposomes, resulted in a statistically significant increase in the IFN-γ and IL-17a production as compared to naïve control mice and mice immunized with Ag alone (Figure 8A). Comparable IFN-γ levels were observed when co-administering MOMP with DDA/TDB-2 and DDA/TDS (Figure 8A, left). In contrast, the IFN-γ levels induced by the DDA/TMS and the DDA/PEG–TDS formulations were significantly lower (approximately 25% and 30% decreases, respectively) than the IFN-γ levels induced by the DDA/TDS liposomes (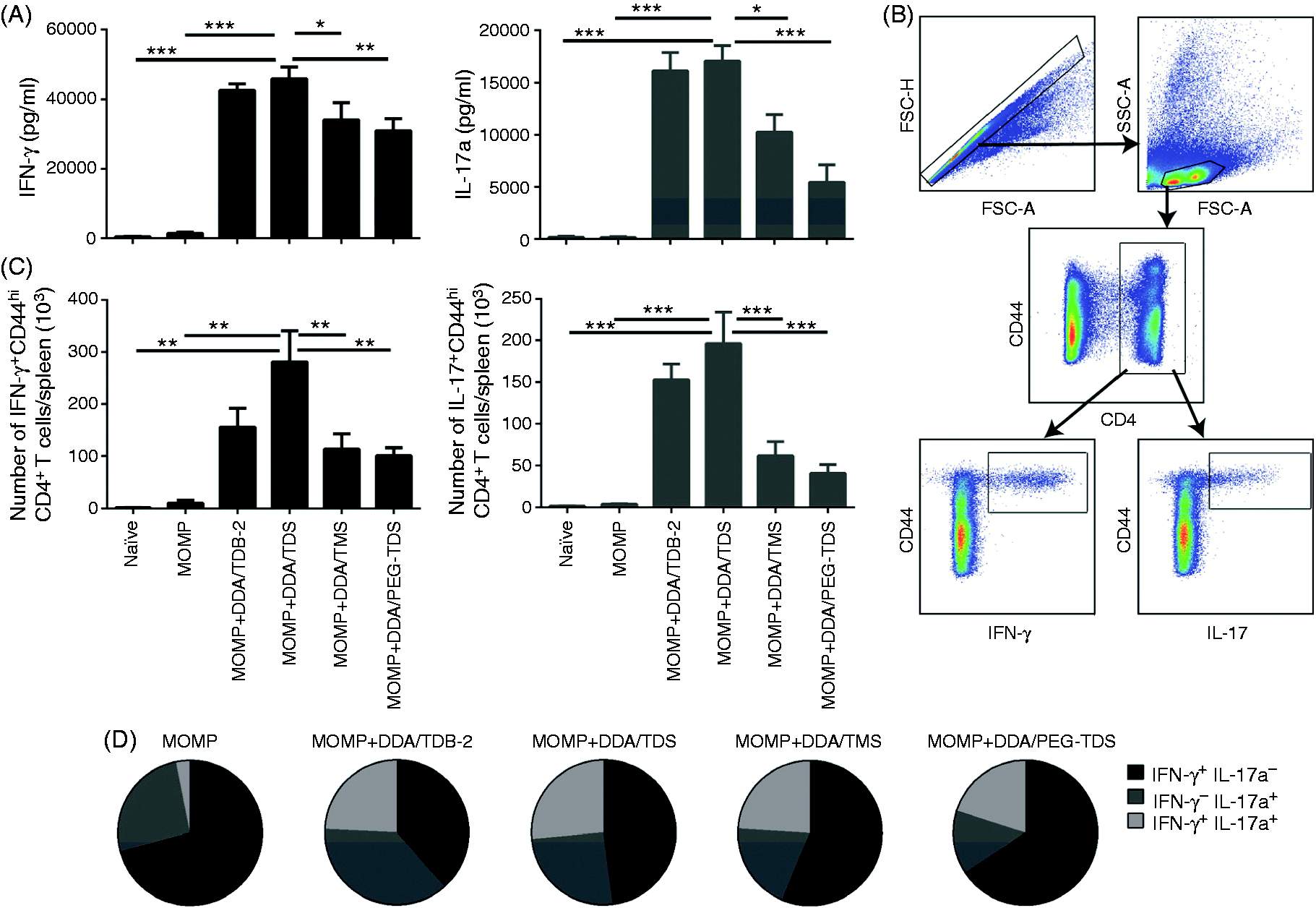
The Th1/Th17 responses were further evaluated at a single-cell level by intracellular cytokine staining (the gating strategy is illustrated in Figure 8B). The numbers of IFN-γ+CD44+CD4+ triple-positive T cells were significantly decreased for the DDA/TMS and DDA/PEG–TDS groups, as compared to the group immunized with DDA/TDS [approximately a 50% decrease (
The polyfunctionality of the T cells was evaluated by combinatorial Boolean gating analysis in order to compare the cytokine expression profiles (Figure 8D). Upon immunization with MOMP–DDA/TDB-2, a mixed Th1/Th17 response with equal percentages of IFN-γ+ and IL-17a+ T cells was observed. Furthermore, in comparison to the group vaccinated with MOMP alone, a larger percentage of the T cells were double-positive (IFN-γ+/IL-17a+), although this observation is only based on few cells and should be confirmed. A correlation between the number of IFN-γ+/IL-17a+ double-positive cells and protection after immunization of mice with DDA/TDB and a chlamydia antigen (PmpG) has previously been reported. 20 Vaccination with the formulations adjuvanted with DDA/TDS, DDA/TMS or DDA/PEG–TDS, respectively, did also drive the detected immune response in a Th1/Th17 double-positive direction compared with the response for the antigen alone. However, there was a decrease in the fraction of single-positive IL-17a+ T cells in the order DDA/TDS > DDA/TMS > DDA/PEG–TDS. For all adjuvanted groups, the fraction of double-positive T cells (IFN-γ+/IL-17a+) was almost constant.
The findings from the ELISA analyses and the intracellular cytokine staining thus collectively suggest that the diester TDS leads to a more robust immune activation than found for both the monoester TMS and the PEG–TDS construct.
Discussion
In this study, we tested an array of analogues of the synthetic glycolipid immunostimulator TDB with systematically shortened chain length, varied degree of acylation (mono- vs. diesters), and headgroup display mode. We analysed their potential as: (i) activators of murine macrophages and DC, (ii) ligands of the cord factor receptor Mincle
Recently, low-resolution crystal structures for both human and bovine Mincle were reported independently by two laboratories,21,22 demonstrating the presence of two sugar binding sites and a hydrophobic groove, of which the latter most likely accommodates the acyl chains attached to the trehalose headgroup.21,22 However, because of the low solubility of the hydrophobic natural TDM ligand for Mincle, the crystals were obtained in the presence of trehalose and citrate, respectively. Nevertheless, the influence of the length of the acyl chains on the binding to Mincle has been addressed by using binding assays with the carbohydrate recognition domain only. Short soluble diester derivatives of trehalose have been shown to compete with 2.5–7-fold higher efficiency than the corresponding monoesters for binding to the mouse Mincle carbohydrate recognition domain, which is consistent with our finding that trehalose 6,6'-diesters display superior binding and activation as compared with the monoesters. 23
Previously, activation of mouse macrophages by trehalose esters has been investigated,9,10 and our results corroborate the finding that a strong activation may be achieved with 20:0, 22:0 and 26:0 trehalose diesters. 9 While the 18:0 (stearate) diester was only minimally active in the study by Khan et al., 9 we observed strong activation also by TDS, as well as by TDMyr (14:0) and TDP (16:0). The discrepancy between our present results and those of Stocker et al. regarding the potential of TMXs to activate macrophages appears even more striking. 10 Thus, we observed a strongly reduced capacity of the monoester analogues to induce NO and G-CSF production, whereas Stocker et al. found comparable macrophage activation by the corresponding monoester derivatives with similar fatty acid length. 10 Differences in the experimental conditions may account for these discrepancies. We differentiated macrophages from bone marrow cells in the presence of M-CSF, whereas the Stocker group used GM-CSF to differentiate macrophages from bone marrow cells, which is, in fact, a commonly used protocol to generate mouse BMDC, although recent work has revealed that GM-CSF mouse bone marrow cultures comprise a mix of monocyte-derived macrophages and conventional DC.24,25 To address this potential role of the exact cell type used for stimulation, we compared BMM generated with M-CSF and BMDC differentiated with GM-CSF, but observed that the stronger activity of the diester compounds was observed in both cell types (Figure 4). Kinetics may also play a role, as we measured the accumulation of G-CSF and NO after a stimulation period of 48 h, while the Stocker group harvested the supernatants after 96 h. Finally, the physical presentation of the glycolipids [either adsorbed to the bottom of the well (as in our study) or solubilized by addition of DMSO] to the cells may affect the interaction of the glycolipids with Mincle, and thereby influence the cellular responsiveness owing to differences in their supramolecular structure.
A recent study has described the synthesis of corynomycolic acids and their esters with the disaccharide trehalose and the monosaccharide glucose. 11 Testing of Mincle activation by these compounds in dose–response assays with a reporter cell line showed that the trehalose mono-corynomycolates were as potent in activating murine and human Mincle reporter cells as the trehalose di-corynomycolates. 11 Strikingly, even glucose Glc mono-corynomycolates activated Mincle signalling, albeit requiring 10-fold higher concentrations, suggesting that the requirement for the trehalose headgroup initially reported by Ishikawa et al. is not absolute. 1 However, when the glucose headgroup was linked not to the branched corynomycolate but to a simple fatty acid of similar length (i.e. behenic acid), Mincle activation was completely abrogated. 11 Together with our present findings, it appears that TDX of simple fatty acids (of a certain length) possess affinity to Mincle. By contrast, in order for the glucose derivatives to elicit activation, the acyl chain has to be more complex (i.e. a corynomycolate instead of a simple fatty acid). Mcl, a closely related C-type lectin receptor, has been described as an additional receptor for the cord factor. 26 Mcl was recently shown to increase Mincle surface expression through complex formation.27,28 With regard to direct interaction with the cord factor, Mcl appears to bind with much lower affinity as compared with Mincle. Thus, it is at present not entirely clear to which extent the requirement for Mcl reflects its function as a receptor for the glycolipids or as a chaperone for Mincle.
The poor solubility of long-chain trehalose diesters potentially decreases their attractiveness as adjuvant candidates, and it may explain some of the variance observed in studies using similar compounds under different assay conditions in different laboratories, as discussed above. Therefore, the recent report on the first soluble ligand for Mincle, namely brartemicin, is of considerable interest for functional studies.
29
Brartemicin, isolated from bacteria of the genus
For the
The reduction in adjuvanticity (when comparing TDS with TMS) was not as pronounced in the
The design of adjuvants with a mixed Th1/Th17 response profile has been suggested to be important for vaccination-induced control of lung infectious diseases, such as tuberculosis.31,32 IL-17 has been shown to be essential for the formation of iBALT, which has resulted in a special interest in Th17-inducing mucosal vaccines.33–35 However, T cells with IFN-γ+/IL-17a+ polyfunctionality are not well described in the literature and their significance is thus largely unknown. The induction of IFN-γ+/IL-17a+ co-expressing T cells has been correlated with vaccine-induced protection upon vaccination with DDA/TDB liposomes and MOMP.
20
In the
The results from the
Footnotes
Acknowledgements
The authors wish to thank Rune Fedelius Jensen (Statens Serum Institut), Mikkel Lohmann Schiøth and Sarah Justesen (both University of Copenhagen), Barbara Bodendorfer (University Hospital Erlangen) and the Department for Biological Services (Statens Serum Institut) for excellent technical assistance. The support given by Katrin Jozefowski and Manfred Kirsch in mouse genotyping and husbandry is appreciated. The support by Stephanie Zimmermann in Mincle-Fc generation and production is also gratefully acknowledged.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KSK and DC are employed by Statens Serum Institut, a non-profit government research facility, which holds a patent on the use of TDB in adjuvant formulations and of which the CAF adjuvants are proprietary products.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by The Danish Council for Independent Research/Medical Sciences (grant number 09-067412) and the Drug Research Academy, Faculty of Health and Medical Sciences, University of Copenhagen, Denmark (R.S.K.). Work in the laboratory of C.F. was further supported by Innovation Fund Denmark [previously The Danish National Advanced Technology Foundation (grant numbers 007-2007-1 and 069-2011-1) and the Danish Strategic Research Council (Centre for Nano-Vaccine, grant number 09-067052)]. Work at SSI was supported by the EU’s Seventh Framework Programs NEWTBVAC (grant agreement no. FP7-HEALTH-F3-2009-241745) and ADITEC (grant agreement no. FP7-HEALTH-F2010-280873). The NMR equipment used in this work was purchased via grant number 10-085264 from The Danish Council for Independent Research/Nature and Universe. Work in the laboratory of R.L. was supported by grants from Deutsche Forschungsgemeinschaft (SFB 796, TP B6) and by EU FP7 NEWTBVAC. Work in the laboratory of B.L. was supported by grants from the Deutsche Forschungsgemeinschaft (SFB 765) and the German Federal Ministry of Education and Research (BMBF).
