Abstract
Gram-negative bacterial LPS induce murine B-cell activation and innate (polyclonal) Ab production. Mouse B cells express the LPS signaling receptor (TLR4), yet how LPS activates B-cell responses in vivo is not known. Can LPS directly stimulate B cells to induce innate Ab production? Is activation of non-B cells also required? To address these questions, we transfused LPS-responsive (Tlr4+/+) or non-responsive (Tlr4−/−) B cells into LPS-responsive or non-responsive mice. Increased expression of the early activation markers CD69 and CD86 could be induced on transfused Tlr4−/− B cells by injecting LPS subcutaneously into Tlr4+/+ mice, demonstrating indirect activation of B cells by TLR4-responsive non-B cells in vivo, but the Tlr4−/− B cells did not increase serum IgM levels. In contrast, when Tlr4−/− recipients were transfused with Tlr4+/+ B cells, LPS induced large amounts of serum IgM and LPS could also enhance specific Ab production to a protein that was co-injected with it (adjuvant response). Thus, LPS-exposed non-B cells mediated increased surface expression of early B-cell activation markers, but this response did not predict innate Ab responses or LPS adjuvanticity in vivo. Direct stimulation of B cells by LPS via TLR4 was necessary and sufficient to induce B cells to produce Ab in vivo.
Introduction
LPS is the classical murine B lymphocyte mitogen, capable of stimulating polyclonal B cells to proliferate, differentiate into plasma cells and secret innate Abs in vitro. 1 Whether accessory cells, such as macrophages and dendritic cells, are required for LPS-induced B-cell activation and Ab production has been debated for many years. Some investigators have argued that direct LPS–B-cell contact is required to trigger Ab production (‘one nonspecific signal’2–4) and offered evidence that macrophages suppress direct B-cell activation by LPS, 5 while others found that accessory cells and/or their products are required for LPS to induce murine B-cell proliferation and/or Ab production.6–9 These studies were performed in vitro using isolated cells; the induction of Ab-secreting cells was often the experimental end point. Now we know that the ‘one nonspecific signal’ comes from the major LPS receptor on murine B cells, TLR4,3,10–12 and that the signal may be enhanced by another LPS receptor, RP105, 13 yet whether LPS-induced B-cell intrinsic signaling is necessary and sufficient for B-cell activation and innate Ab production in vivo is not established.
We have previously found that the host’s endogenous LPS-degrading enzyme, acyloxyacyl hydrolase (AOAH), 14 limits LPS-induced polyclonal B-cell responses and Ab production in vivo.15,16 Mice that lacked AOAH had exaggerated polyclonal Ab production in response to LPS injected subcutaneously. We further showed that this effect was probably caused by the persistence of a reservoir of bioactive LPS at the site of injection for more than a month in AOAH-deficient mice. 17 Evidence that subcutaneously injected LPS can directly contact B cells in draining lymph nodes and then stimulate B cells to produce Abs has been lacking.
LPS is also an adjuvant, capable of enhancing Ab production to a protein Ag that is mixed and co-injected with it. Pasare and Medzhitov found that activation of TLR4 on B cells is required for LPS to be an adjuvant, 18 but whether TLR4 expression on B cells is sufficient for this outcome is not clear.
In this study, we distinguished indirect from direct LPS–B-cell interactions by infusing Tlr4+/+ or Tlr4−/− B cells into recipient mice of the same or opposite TLR4 genotype. After administering LPS subcutaneously, cell-surface expression of CD69 and CD86 was used to monitor early activation of lymph node B cells, and serum IgM titers were measured to quantitate polyclonal Ab production. To differentiate innate Abs produced by donor from recipient B cells, we used Tlr4+/+ or Tlr4−/− MD4 mice, which differ in their IgM allotypes but share the C57BL/6 background. 19 To assess adjuvant responses, LPS was also injected with hen egg lysozyme (HEL), which can be recognized by the MD4 mouse B cell receptor, and anti-HEL Ab levels were measured. By using immunofluorescent microscopy and flow cytometry, we studied the direct contact of LPS with B cells in draining lymph nodes. The results provide strong evidence that direct stimulation of B cells by LPS is both necessary and sufficient for LPS to induce polyclonal Ab responses and for LPS to be an adjuvant in vivo.
Materials and methods
Mice
C57BL/6-Tg(IghelMD4)4Ccg/J (MD4), B6.B10ScN-Tlr4lps-del/JthJ (Tlr4−/−), C57BL/6 J (CD45.2) and B6.SJL-Ptprc a Pepc b /BoyJ (CD45.1) mice were obtained from Jackson Laboratories (Bar Harbor, ME, USA). MD4 and Tlr4−/− mice were crossed to produce Tlr4−/− MD4 mice. Mice were housed in a specific pathogen-free facility and studied using protocols approved by the Institutional Animal Care and Use Committee, UT-Southwestern Medical Center, or by the Animal Resources Center of the National Institute of Allergy and Infectious Diseases (ASP LCID11E). All the experiments were performed in accordance with the approved guidelines.
Reagents
Escherichia coli O111 LPS was from Sigma-Aldrich (L2630, purified by phenol extraction; St. Louis, MO, USA). Neisseria meningitidis LPS, purified from a group B (L3,7,9) strain, was a generous gift from M. Apicella (University of Iowa Medical Center, Iowa City, IA, USA).20–22 E. coli O14 was obtained from the Statens Serum Institut (Copenhagen, Denmark) and its LPS (Ra chemotype) 23 was prepared by the phenol–chloroform–light petroleum method. 24 The ability of each of the LPS preparations to induce TNF-α production was at least 20-fold less in TLR4−/− macrophages than in TLR4+/+ macrophages. E. coli O14 LPS–FITC was made as described.25,26 OVA-Alexa 594 was from Life Technologies (Carlsbad, CA, USA). Abs used for flow cytometry: anti-CD69 (Clone H1.2F3), anti-CD86 (Clone GLI), B220 (Clone RA3-6B2), CD45.1 (Clone A20) and CD45.2 (Clone 104) were from BD Biosciences (Franklin Lakes, NJ, USA).
In vivo and in vitro stimulation
Single-cell suspensions were made from spleens and lymph nodes of MD4 and Tlr4−/− MD4 mice (IgMa B cells). Red blood cells were lysed by using red blood cell lysing buffer (Sigma-Aldrich). Cells were blocked with 0.5 mg/ml normal mouse Ig (Invitrogen, Carlsbad, CA, USA), 0.5% BSA, 2 mM EDTA on ice for 30 min. B cells were purified by using anti-B220 magnetic beads (MACS; Miltenyi Biotec, Bergisch Gladbach, Germany). In total, 2 × 107 Tlr4+/+ or Tlr4−/− B cells (≥95% pure) were transfused into Tlr4+/+ and Tlr4−/− C57BL/6J mice, which express IgMb on their B cells, via the lateral tail vein. Twenty-four h later, 5 µg N. meningitidis LPS was injected into one footpad. After 16 h, the draining and non-draining popliteal nodes were dissected and analyzed for CD69 and CD86 expression on B220+ B cells by flow cytometry. Donor and recipient B cells were identified by their surface IgM allotype. We found that 1.58 ± 0.72% of the B cells in the non-draining nodes were donor B cells; there was no significant difference in donor B-cell frequency among the groups.
To assay CD69 up-regulation on B cells in vitro, B cells were purified from spleens and lymph nodes, first by MACS and then by FACS. More than 99% of the purified cells were B220+ (Figure 1a). One million B cells per well were seeded in 24-well plates in the absence or presence of 5 × 105 negatively selected Tlr4+/+ cells (B220 negative cells) and treated with various doses of N. meningitidis LPS for 16 h at 37℃. Cells were analyzed for CD69 expression by flow cytometry.
LPS induces CD69 surface expression on B cells in vitro when TLR4 is expressed either on B cells or on splenic non-B cells. Tlr4+/+ or Tlr4−/− B cells were purified to over 99% purity and treated with N. meningitidis LPS in (a) the absence or (b) presence of Tlr4+/+ non-B cells for 16 h at 37℃. CD69 expression on B220+ B cells was measured by flow cytometry. (c) Untreated B cells; (d) 100 ng/ml LPS treated Tlr4+/+ B cells. (e) The up-regulation of CD69 expression was determined after treatment with increasing doses of LPS. Each replicate is shown (n = 2). This experiment was repeated with similar results.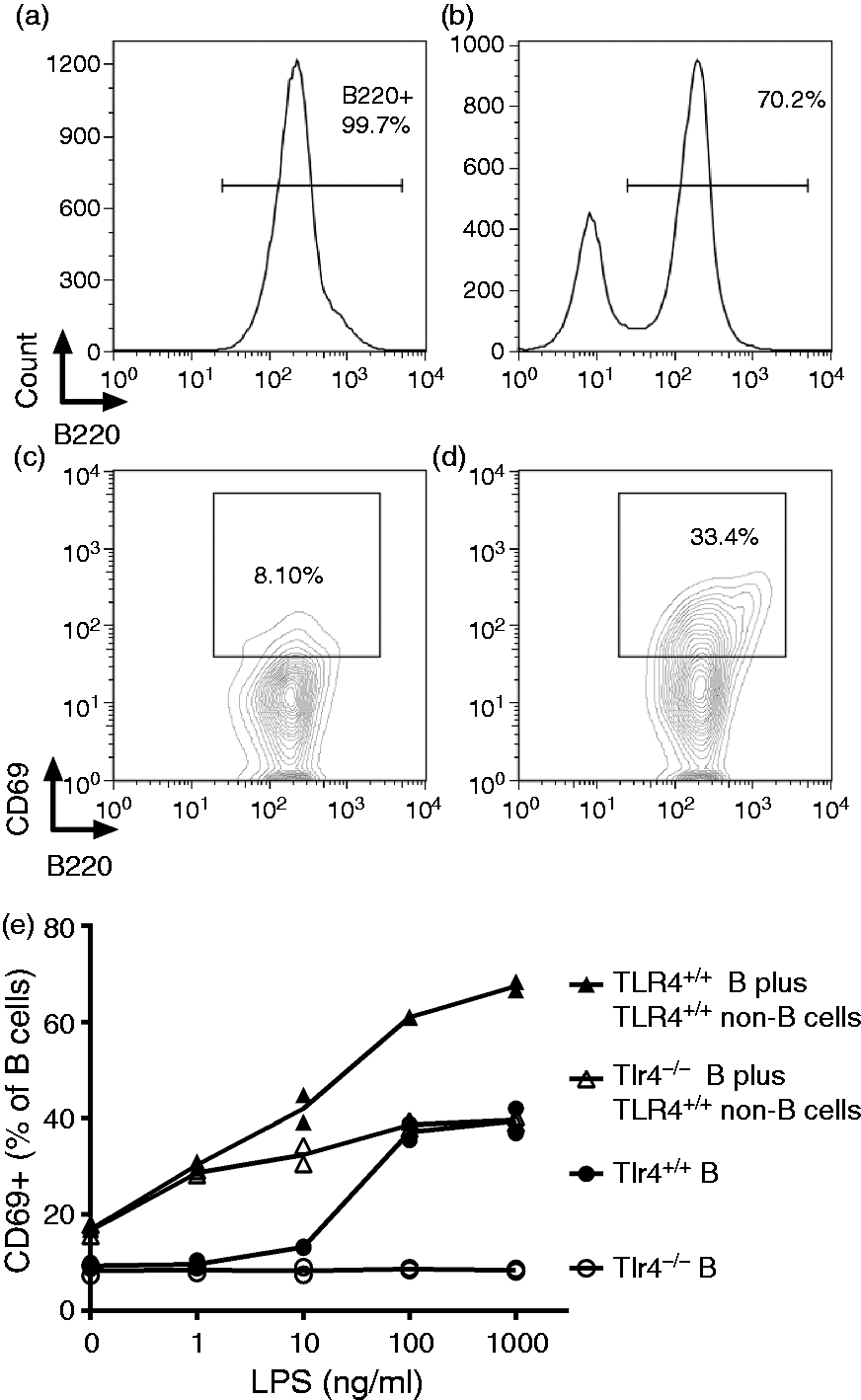
IgMa B-cell transfer and Ab production experiment
B cells were purified from MD4 and Tlr4−/−, MD4 mice as described above. In total, 1 × 107 purified TLlr4+/+ and Tlr4−/− IgMa B cells were transfused into Tlr4+/+ and Tlr4−/− C57BL/6J mice (IgMb). Twenty-four h later, 10 µg E. coli O111 LPS was injected into the footpads of recipient mice; 2.5 µg was given on d 4 and 10 µg LPS on d 12 and 18. Blood samples were drawn on d 7 and 21 and IgMa concentrations were determined by ELISA. To test LPS adjuvanticity, 50 µg HEL, 5 µg LPS O14 or a mixture of both was injected into the mouse right footpad. Serum samples were collected on d 6 and anti-HEL IgMa levels was measured by ELISA.
ELISA methods
Microtiter wells were coated with goat anti-mouse IgM or HEL (Sigma-Aldrich). The detection Abs were HRP-conjugated goat anti-mouse IgM (Sigma) and anti-mouse IgMa-Biotin (BD, San Jose, CA, USA) plus streptavidin–HRP. HRP substrate (BD) was used. Plates were read on a MRX Microplate Reader (Dynex Technologies Incorporation, Chantilly, VA, USA). IgM standards were kindly provided by E. Vitetta (UT-Southwestern Medical Center, Dallas, TX, USA). In all cases, differences between post- and pre-immunization are reported.
Immunofluorescence assay
Mice were injected with 5 µg LPS-FITC in their footpads. The draining lymph nodes (popliteal nodes) were harvested 3 min after injection and immediately fixed in buffered 4% formaldehyde for 24 h. The lymph nodes were washed with PBS and embedded in OCT compound (Sakura Finetek, Alphen aan den Rijn, the Netherlands) and snap frozen in isopentane chilled in liquid nitrogen. The frozen nodes were cut to 8-µm sections and stained with anti-B220 Ab as described. 26 Images were taken using an Olympus BX61 motorized confocal microscope.
In vivo LPS binding assay
C57BL/6 J mice (CD45.2) were injected with 5 µg LPS–FITC in the right footpad; after 0.5, 1, 2, 5 and 24 h, the right and left popliteal nodes (draining and non-draining lymph nodes respectively) were harvested and processed with lymph nodes from CD45.1 mice by using the frosted ends of glass slides. Single-cell suspensions were made and stained with B220, CD45.1 and CD45.2 Abs (BD). The percentage of FITC+ B cells was measured by FACS. The percentage of FITC+ CD45.1 B cells is considered to indicate non-specific binding of LPS-FITC during the processing of lymph nodes.
Statistic analysis
Unpaired Student’s t-test (two-tailed) was used for comparisons between groups.
Results
TLR4 expression on B cells is not required for early activation
To evaluate the contribution of macrophages and other non-B cells to LPS-induced B-cell activation, we treated co-cultures of purified B cells (>99% B220+) and non-B cells (B220–) with LPS in vitro (Figure 1a, purified B cells only; Figure 1b, purified B cells mixed with non-B cells) and measured the surface expression of an early activation marker, CD69 (Figure 1c, untreated B cells; Figure 1d, purified TLR4+/+ B cells treated with 100 ng/ml LPS). CD69 was up-regulated on purified Tlr4+/+ cells, as well as on Tlr4−/− B cells in the presence of Tlr4+/+ non-B (accessory) cells (Figure 1e). LPS can thus increase CD69 expression on B cells in vitro either directly (Tlr4+/+ B cells) or indirectly (via Tlr4+/+ accessory cells). With lower concentrations (1 and 10 ng/ml) of LPS, the indirect effects were greater than the direct effects, while at higher concentrations (100 and 1000 ng/ml), similar levels of B-cell activation were observed through B and non-B cell TLR4 signaling. The response of the Tlr4+/+ B cells co-cultured with the non-B cells from Tlr4+/+ mice was approximately the sum of the direct and indirect responses.
We transfused Tlr4+/+ or Tlr4−/− IgMa+ B cells (from MD4, Tlr4+/+ and MD4, Tlr4−/− mice, respectively)
19
into Tlr4+/+ or Tlr4−/− recipient C57BL/6 mice, which express IgMb on their B cells (Figure 2a). The frequency of transferred B cells in the recipient lymph nodes was similar among the four groups. Surface expression of CD69 and CD86, measured 16 h after LPS injection into a footpad, increased on draining lymph node Tlr4−/− B cells when the recipient mice expressed TLR4; in contrast, CD69 and CD86 expression increased much less on adoptively transferred Tlr4+/+ B cells following LPS administration to Tlr4−/− recipient mice (Figure 2b, c). At these early time points after LPS injection, accessory cell stimulation was sufficient to activate B cells in vivo.
LPS–B cell contact is required for polyclonal Ab production. Tlr4+/+ or TLR4−/− MD4 (IgMa) B cells were transfused into Tlr4+/+ and Tlr4−/− IgMb recipient mice 24 h before they received a footpad injection of 5 µg LPS. Sixteen h after LPS injection, the popliteal nodes were collected and CD86 and CD69 expression on endogenous (IgMb) and transfused (IgMa) B cells was measured using flow cytometry. (a) Gating strategy to determine CD86 and CD69 expression on donor and recipient B cells. B220+ B cells from lymph nodes were gated first. The donor and recipient B cells were differentiated by IgMa expression. CD86 or CD69 expression was then determined on B cells. Top right histogram: blue dotted line, Tlr4−/− donor B cells (in a Tlr4−/− recipient); blue solid line, Tlr4+/+ donor B cells (in a Tlr4−/− recipient). Right bottom histogram: black dotted line, Tlr4−/− recipient B cells (the host mice received Tlr4−/− B cells); black solid line, Tlr4−/− recipient B cells (the host mice received Tlr4+/+ B cells). (b, c) LPS induces early B-cell activation when either the host or the infused B cells express TLR4. The data shown are combined from two independent experiments, each with three or four mice per group. The x-axis labels indicate infused (donor) or endogenous (recipient) B cells. (d–g) LPS-induced serum Ab production requires TLR4 expression on B cells. Purified Tlr4+/+ (closed boxes) or Tlr4-/- (open boxes) splenic and lymph node IgMa B cells were transfused into Tlr4+/+ and Tlr4−/− IgMb recipient mice. LPS (10 µg) was given into one footpad 24 h later and boosted with doses of 2.5, 10 and 10 µg on d 4, 12 and 18, respectively. Serum IgMa (d, e) and total IgM (f, g) levels were measured 7 and 21 d after LPS injection. Tlr4−/− mice that received Tlr4+/+ IgMa B cells had the strongest IgMa response (d, closed boxes). Ab responses are expressed as the change (Δ) above baseline levels. n = 4 animals/group. Similar results were obtained in two other experiments, each with 3 or 4 mice/group. (b–g) *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.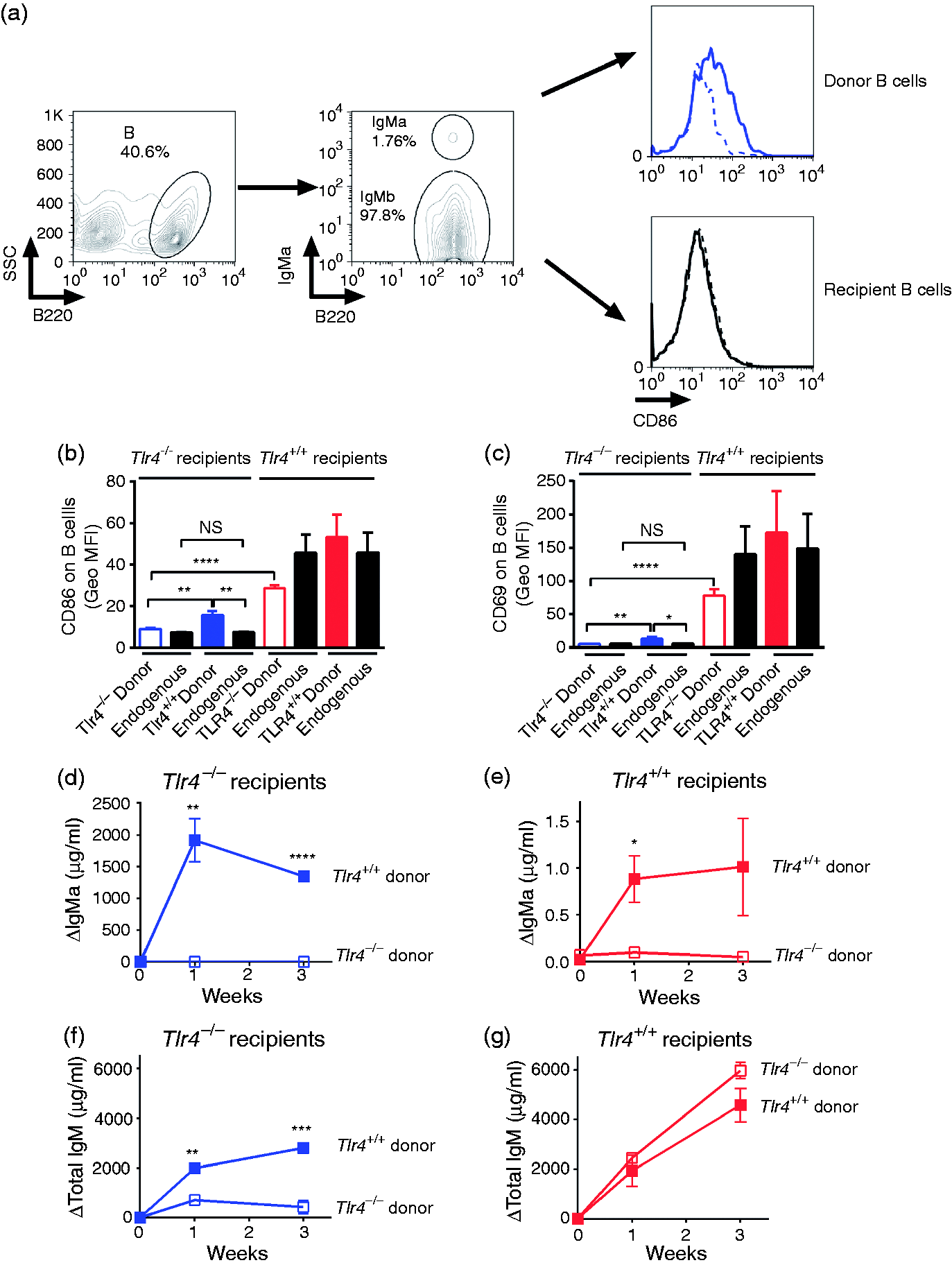
TLR4 expression on B cells is necessary and sufficient for LPS-induced Ab responses
We next asked whether TLR4 expression on B cells or accessory cells is required to enable induction of innate Ab responses by LPS. Tlr4+/+ or Tlr4−/− MD4 IgMa+ B cells were transfused into Tlr4+/+ or Tlr4−/− C57Bl/6 mice (IgMb+ B cells). LPS was then injected s.c. and serum IgMa and total IgM titers were measured 1 and 3 wk later. The Tlr4−/− mice that received Tlr4+/+ IgMa B cells had the strongest LPS-induced IgMa response (Figure 2d), whereas Tlr4+/+ recipients infused with Tlr4−/− IgMa+ B cells did not increase serum IgMa (Figure 2e). TLR4 expression on B cells was thus necessary and sufficient for LPS-induced polyclonal Ab production, strongly suggesting that direct contact between LPS and Tlr4+/+ B cells occurred in vivo. This experiment also showed that accessory cell stimulation, which could activate Tlr4−/− B cells in vitro (Figure 1) and in vivo (Figure 2b, c), was unable to induce serum Ab production by Tlr4−/− B cells in vivo (Figure 2e, open boxes). Unexpectedly, Tlr4+/+ mice that received Tlr4+/+ IgMa B cells (Figure 2e, closed boxes) had much lower IgMa responses than did Tlr4−/− recipients (Figure 2d). As donor B cells accounted for <2% of the lymph node B cells, the injected LPS may have activated large numbers of endogenous B cells in Tlr4+/+ recipients and their IgMb production greatly exceeded the IgMa produced by the donor Tlr4+/+ cells. It is also possible that Tlr4+/+ non-B cells negatively regulate Ab production. Tlr4−/− IgMa B cells did not produce IgMa when they were present in Tlr4+/+ recipient mice, yet the endogenous (total IgM) response was robust (Figure 2f, g), confirming the recipients’ responsiveness to LPS.
Following subcutaneous injection, B cells in draining lymph nodes have direct contact with LPS
To test whether B cells in draining lymph node can be exposed to LPS injected s.c., mice were injected with a mixture of LPS–FITC and OVA-Alexa594 in one footpad. Three min after injection, their draining lymph nodes (popliteal nodes) were dissected for study using immunofluorescence (Figure 3a). OVA-Alexa594 highlighted the conduits in the lymph nodes. LPS–FITC was found co-localizing with OVA in the sub-capsular sinus (SCS) and in some conduits beneath the SCS, suggesting that some LPS–FITC was delivered via the conduit system to B cells. To seek direct evidence that B cells in draining lymph node contact LPS, C57BL/6J (CD45.2) mice were injected with LPS–FITC in one footpad. The draining lymph nodes (popliteal nodes) were then harvested at various time points. To exclude ex vivo capture of LPS–FITC during the processing of lymph nodes, the draining lymph nodes were processed together with lymph nodes from congenic B6.SJL (CD45.1) mice. From 1 h to 24 h after injection, there was significantly greater association of LPS with B cells in draining lymph nodes than there was non-specific binding (Figure 3b, c). The amount of specifically-bound LPS increased over time, suggesting that LPS drains to lymph nodes following s.c. injection and directly stimulates B cells.
Draining lymph node B cells contact LPS after s.c. LPS injection. LPS–FITC and OVA-Alexa Fluor 594 were mixed and injected into one footpad. Three min later, the draining popliteal nodes were harvested and fixed with paraformaldehyde before imbedding in OCT. OVA-Alexa Fluor 594 highlighted the conduit system in the lymph node and the B cells were stained with anti-B220 Ab-Alexa Fluor 350 respectively (a). C57BL/6J mice (CD45.2, dots) were injected with 5 µg LPS–FITC in one footpad. 0, 0.5, 1, 2, 5 and 24 h after injection, their draining lymph nodes (DLN) and non-draining lymph nodes (NDLN, NLN) were dissected (filled symbols) and processed along with lymph nodes (LN) from CD45.1 mice (open symbols). B cells that were associated with LPS were measured by FACS. Data are combined from two experiments. n = 6 animals/group (b, c). *P < 0.05; **P < 0.01; ***P < 0.001.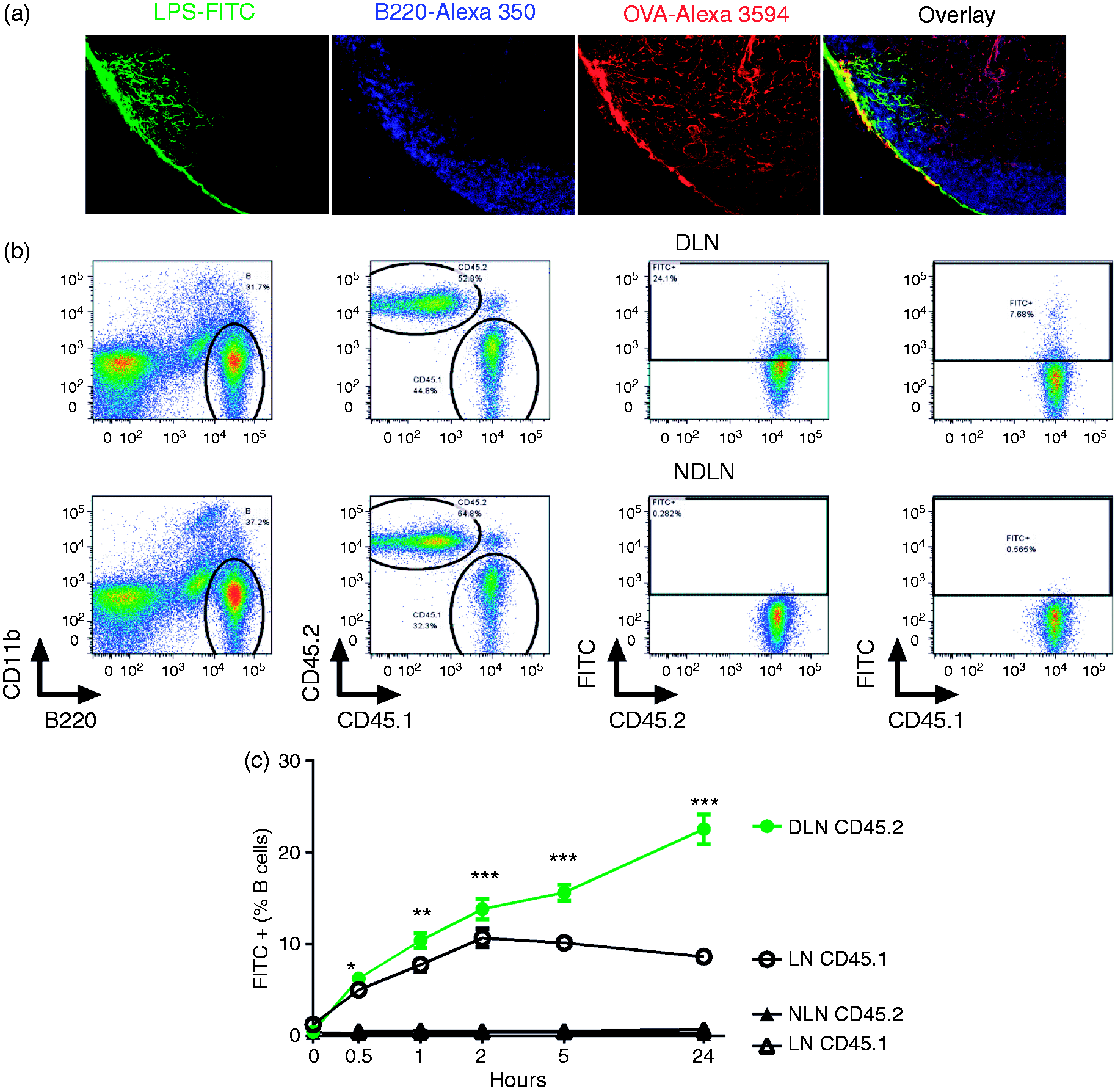
TLR4 expression on B cells is necessary and sufficient for LPS adjuvanticity
Pasare and Medzhitov have shown that TLR4 is required for LPS adjuvanticity in vivo.
18
When TLR4 is solely expressed on B cells, can LPS still be an adjuvant? Tlr4+/+ or Tlr4−/− IgMa+ B cells were transfused into Tlr4+/+ or Tlr4−/− C57Bl/6 mice (IgMb + B cells) and the mice were immunized with a mixture of LPS and HEL (IgMa B cells from MD4 mice recognize an epitope of HEL
19
). Anti-HEL IgMa levels were measured in serum samples collected 6 d after immunization. In Tlr4−/− C57Bl/6 mice transfused with Tlr4+/+ IgMa+ B cells, the amount of anti-HEL IgMa was significantly higher in mice that had received LPS and HEL than in mice that had been injected with HEL or LPS, demonstrating that LPS can be an adjuvant when TLR4 is exclusively expressed on B cells (Figure 4). In agreement with the results of Pasare and Medzhitov, in Tlr4+/+ C57Bl/6 mice transfused with Tlr4−/− IgMa+ B cells, immunization with HEL and LPS did not elicit an anti-HEL IgMa response. Tlr4+/+ C57Bl/6 mice transfused with Tlr4+/+ IgMa+ B cells had a significant but very low anti-HEL response, similar in magnitude to the polyclonal Ab responses to LPS in these mice (Figure 2d).
LPS–B-cell contact is necessary and sufficient for adjuvanticity of LPS. Tlr4+/+ or Tlr4–/– MD4 (IgMa) B cells were transfused into Tlr4+/+ and Tlr4–/– IgMb recipient mice 24 h before they received a footpad injection of 5 µg LPS, 50 µg HEL or both. Six d after injection, serum samples were collected and anti-HEL IgMa levels were measured by ELISA. Data shown are combined from two experiments. n = 7–8. ***P < 0.001, ****P < 0.0001.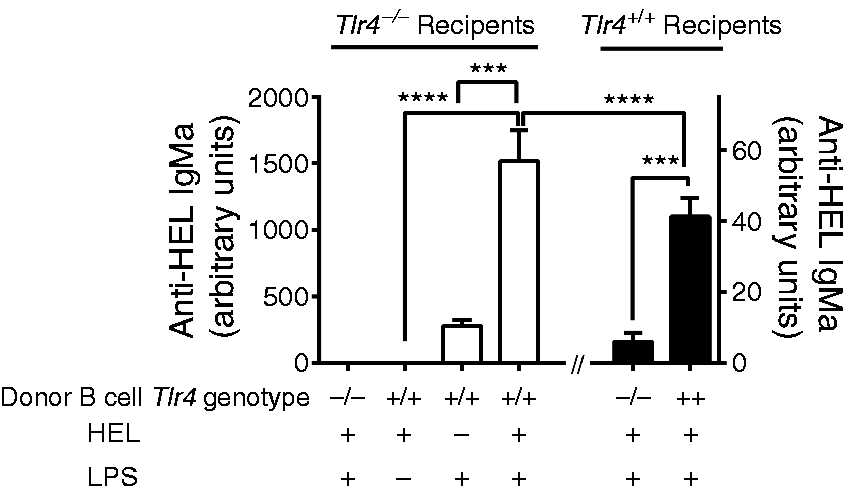
Discussion
Gram-negative bacterial LPS stimulates polyclonal B cells to differentiate and to produce Abs, regardless of B-cell receptor (BCR) ligation or cognate T-cell help. 27 How accessory cells such as macrophages and dendritic cells contribute to the LPS–B cell interaction has been debated for decades. As murine B cells are now known to express LPS signaling receptor TLR4 and a cooperative receptor, RP105, we designed in vivo experiments to test whether a B-cell intrinsic TLR4 signal is sufficient to induce Ab production. Instead of using B-less μMT mice as recipients (they have abnormal lymph node architecture), we transfused purified Tlr4+/+ or Tlr4−/− B cells into Tlr4+/+ or Tlr4−/− recipient mice, in which the lymph node structure is intact. Our results demonstrate that TLR4 expression on B cells (provided that RP105 abundance/activity is not affected by TLR4 deficiency) is both necessary and sufficient for LPS-induced innate Ab responses in vivo; contributions from accessory cells are dispensable. The conclusion is consistent with the ‘one nonspecific signal’ theory. 2 Our findings also confirm that, in mice that lack the LPS-degrading enzyme AOAH, bioactive LPS that drains persistently from an injection site to regional lymph nodes can directly stimulate B cells to produce large amounts of Ab.15,17
When B cells are activated, they up-regulate their cell surface expression of CD69 and CD86 molecules, then they proliferate and differentiate to Ab-secreting plasma cells. Induced cell-surface CD69 expression leads to down-regulation of S1P1, promoting lymphocyte retention in lymphoid tissue; 28 CD86 is a co-stimulatory molecule for Ag presentation to T cells. We found that B-cell extrinsic TLR signals (accessory cell-derived) could cause CD69 and CD86 up-regulation, both in vitro and in vivo, indicating that LPS induced accessory cell activation can promote early B-cell activation. Whether direct cell–cell interaction or soluble mediators (such as type I interferon18,19) produced by accessory cells leads to B-cell activation requires further investigation. Our in vitro and in vivo results also showed that B-cell extrinsic TLR signaling leads to even greater CD69 and CD86 surface expression than does B-cell intrinsic TLR signaling (Figure 2b, c), yet these signals were not sufficient for Ab production. Thus, early B-cell activation did not lead to Ab secretion in vivo, suggesting that B-cell retention in lymphoid organs and Ag presentation are regulated differently from B-cell differentiation and Ab production. Whether the nature or the duration of stimulation determines B cell fate needs to be studied further. Indeed, direct TLR stimulation increases B-cell expression of transcription factors (Blimp-1 and XBP-1 s) that are pivotal for plasma cell differentiation.29,30 Early activation did not presage IgM production in B cells that lacked TLR4, casting doubt on the utility of CD69 and CD86 as predictors of productive B-cell stimulation in vivo. 31
How lymph-borne LPS is delivered to B cells in lymph nodes has rarely been studied. Small protein Ags (<70 kDa Ags or with small molecular radius) enter a system of conduits that extend to B-cell follicles, 32 or diffuse via gaps in the SCS floor to penetrate the B-cell compartment. 33 Gretz et al. showed that, after s.c. injection, LPS–FITC was mainly detected in the SCS and medulla of draining lymph nodes. 34 The smooth LPS molecules used in their study (from E. coli O55), are heterogeneous in size, with molecular masses from 3.4 to approximately 100 kD. The reason that LPS was excluded from conduits was thought to be the large size of the LPS aggregates. We used LPS from E. coli O14, which produces an LPS that lacks an O-Ag (Ra chemotype). Rough LPS aggregates may become monomers in vivo when albumin, LBP and soluble CD14 are present, 35 enabling them to enter the conduits. However, in a previous study we found LPS–FITC in the sub-capsular and medullary sinuses, co-localizing with CD169+ SCS macrophages and medullary LYVE+ lymphatic endothelium; no LPS–FITC was found in the B-cell area. 17 In that study, we immediately embedded the lymph nodes in OCT after excision and fixed the tissue sections with methanol, so that cell-free LPS, such as LPS in conduits or in tissue interstitial spaces, could have washed away. In the current study, we fixed draining lymph nodes using paraformaldehyde before we embedded the nodes in OCT. With this fixation protocol, LPS was detectable in SCS and some was found in conduits surrounded by B cells. Our data do not exclude the possibilities that LPS diffuses through gaps in the SCS floor to reach B-cell follicles or that SCS macrophages facilitate the delivery of LPS to B cells.33,36 When measuring LPS–FITC binding to B cells using a more sensitive flow cytometry method, we found that B cells bound LPS over time and that binding was maintained during processing and performance of flow cytometry, confirming that lymph node B cells contact LPS. In addition, we did not observe differences in LPS movement from the injection site to the draining lymph nodes between Tlr4+/+ and Tlr4−/− mice (data not shown). Thus, our imaging, FACS and functional analysis all provide evidence that B cells in lymph nodes contact lymph-borne LPS directly, become activated and secrete Abs.
For thymus-dependent Ags, Ab responses require BCR ligation and cognate T-cell help through CD40. Ag-specific B cells are essential Ag-presenting cells for priming CD4+ T cells in vivo.37–40 Whether B-cell intrinsic TLR signals can enhance adaptive Ab responses has been controversial.18,41 Pasare and Medzhitov showed that TLR4 expression on B cells was necessary for LPS to be an adjuvant. 18 In our study, we investigated whether B-cell intrinsic TLR4 signaling is sufficient for LPS to enhance the Ab responses to a protein Ag. In our transfer system, when TLR4 was exclusively expressed on B cells, HEL did not induce much anti-HEL IgM, while HEL mixed with LPS elicited strong responses. Importantly, HEL + LPS induced more anti-HEL IgM than LPS did, excluding the possibility that only non-specific (innate) Ab responses were induced by LPS. Thus, B-cell expression of TLR4 is sufficient for LPS adjuvanticity. Whether the enhanced anti-HEL response is due to cognate T-cell help or simply reflects the synergistic effects of TLR and BCR signaling in B cells requires further investigation. 42 Interestingly, Tlr4−/− recipients transfused with Tlr4+/+ B cells produced much more anti-HEL IgMa in response to HEL and LPS immunization than did Tlr4+/+ recipient mice that received Tlr4+/+ B cells. The results are similar to the innate IgM Ab responses shown in Figure 2 (d, e). Although the explanation for this finding is not clear, the transfused B cells were only ∼2% of the total B-cell population and it is possible that LPS-induced endogenous polyclonal B-cell activation interfered with HEL-specific exogenous B-cell differentiation in the Tlr4+/+ animals that received Tlr4+/+ B cells.
We conclude that the signal that leads to polyclonal and Ag-specific Ab production in vivo is delivered by LPS to B cells directly through TLR4 and does not require stimulation of other cell types. B-cell expression of the LPS transmembrane receptor molecule, TLR4, was necessary and sufficient for both LPS-induced polyclonal Ab production and LPS adjuvanticity in vivo.
Footnotes
Acknowledgements
We thank Sandip Datta, Stefanie Vogel, Patricia Valdez and Jiyang O-Wang for critiquing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant AI18188 from NIAID to the University of Texas Southwestern Medical Center, Dallas, TX(R.M.), by the Division of Intramural Research, NIAID, NIH(R.M.), by grant 14ZR1401700 from Shanghai Committee of Science and Technology (M.L.) and by grant 31570910 from National Natural Science Foundation of China (M.L.).
