Abstract
Aldose reductase (AR; gene
Introduction
Atherosclerosis is an inflammatory disease of the arterial wall. 1 During the course of atherogenesis, blood monocytes adhere to the endothelium, transmigrate into the subendothelial space and differentiate toward macrophages. 1 This differentiation process is modulated by cytokines, chemokines and lipoproteins within the subendothelial space. Depending on the local micromilieu, it may result in differentially polarized macrophages.2,3 Accordingly, the cells may assume different polarization phenotypes: the pro-inflammatory M1 phenotype induced by IFN-γ and LPS, and the rather anti-inflammatory M2 phenotype induced by Th2 cytokines such as IL-4 represent the extremes of this polarization spectrum. 4 Intermediate or additional polarization types may also occur within atherosclerotic lesions.2,3
Diabetes is one of the most important risk factors promoting atherogenesis and plaque destabilization.5,6 The underlying mechanisms are manifold.6,7 One mechanism associated with hyperglycemia seems to be the induction of aldose reductase (AR), the rate-limiting enzyme of the polyol pathway. Under normoglycemia, AR is involved in detoxyfication of aldehydes.8,9 Based on its Km of 0.66 µmol/l for
We have previously demonstrated AR expression in human atherosclerotic plaque macrophages that is synergistically increased by both high levels of oxidized LDL and Glc.
15
Interestingly, AR expression was not consistently found in all plaque macrophages. We therefore hypothesized that AR expression may be restricted to a certain macrophage polarization type. Here, we investigated AR expression and activity in differentially polarized monocyte-derived macrophages
Material and methods
Monocyte-derived human macrophages
The study was approved by the institutional review committee (S-045/2011). All participants gave informed consent. Monocyte-derived macrophages were generated in serum-free media with M-CSF as described previously.16–18 M1 and M2 polarization of macrophages was induced as described previously. 19 Recombinant human M-CSF (Cat. No. 300-25), IFN-γ (Cat. No. 300-02,) and IL-4 (Cat. No. AF-200-04) were purchased from Peprotech (Wiesbaden, Germany). LPS (Cat. No. L4516-1 mg) was purchased from Sigma (Taufkirchen, Germany). To study the effects of hypo- or hyperglycemia, cells were kept in media containing 0, 5 or 30 mmol/l Glc for the last 18 h of cell culture. To study the role of the Glc transporter GLUT-1, STF-32, was used as a specific GLUT-1 inhibitor. 20
Quantitative RT-PCR
Quantitative PCR was performed as described previously.
18
Primer sequences were obtained from Primer Bank:
21
Flow cytometry
Intracellular flow cytometry was performed as described previously. 18 AR was stained using a goat anti-human polyclonal IgG Ab (Cat. No. SC-17732, clone N-20; Santa Cruz, Heidelberg, Germany), a Cy3-conjugated mouse anti-goat Anti-Goat IgG (Jackson ImmunoResearch, Suffolk, UK) was used as secondary Ab. For control stainings, the first Ab was left out.
AR activity assay
AR activity was measured as described previously.8,15,23 Briefly, activity was measured spectrophotometrically by determination of the decrease in absorbance at 340 nm in the absence and presence of glyceraldehyde as substrate reflecting the consumption of NADPH (Prod. No. N5130; Sigma).8,23,24 AR activity was normalized to protein content and expressed in mU/µg (nmol NADPH oxidized per min per µg protein).
Immunofluorescence
Human post-mortem coronary arteries were obtained from the University of Virginia Department of Pathology/Tissue bank. Immunofluorescence staining for AR and CD68 was performed as described previously. 15 Depending on their staining pattern, macrophages were classified CD68+AR−, CD68+AR+ and CD68+AR++. AR+ and AR++ were determined through semiquantitative analysis by a reviewer blinded in regards to the patients’ diagnoses.
Statistics
Quantitative PCR and flow cytometry data were analyzed by paired non-parametric Mann–Whitney testing. Multiple comparisons were done using the Kruskall–Wallis test with Dunn’s multiple comparison testing. Proportions were compared using χ2testing. All statistical analyses were performed using Prism (GraphPad Inc., La Jolla, CA, USA).
Results
AR is differentially expressed in human atherosclerotic plaque macrophages
Macrophages have been demonstrated to be a key source of AR expression within atherosclerotic lesions.
15
We have previously described differential AR expression in macrophages located within the intima, the adventitia, or perivascular fat.
15
To better understand which conditions may regulate macrophage AR expression, we performed immunofluorescence staining of human post-mortem coronary arteries against CD68 and AR in different post-mortem donors. As shown in Figure 1, CD68+AR−, CD68+AR+ and CD68+AR++ macrophages could be identified in most lesions. CD68+ cells were mostly AR+ or AR++, with a rather small proportion of CD68+AR− macrophages. The proportion of CD68+AR+ and CD68+AR++ macrophages differed substantially between patients (2–69% for CD68+AR+ and 26–56% for CD68+AR++; Figure 1b). Therefore, further experiments were conducted to elucidate the underlying reasons for differential AR expression in plaque macrophages.
AR is differentially expressed in human atherosclerotic plaque macrophages. (a) Immunofluorescence staining of a post-mortem coronary artery against CD68 (green) and AR (red). DAPI (blue) as nuclear stain. Yellow arrow indicates a CD68+AR++, orange arrow a CD68+AR+ and green arrow a CD68+AR− macrophage. Scale bars indicate 100 µm. (b) Relative proportion of CD68+AR− (green), CD68+AR+ (orange) and CD68+AR++ (yellow) macrophages of all CD68+ plaque macrophages in post-mortem coronary arteries derived from eight different individuals. ‘n’ indicates the number of CD68+ macrophages analyzed.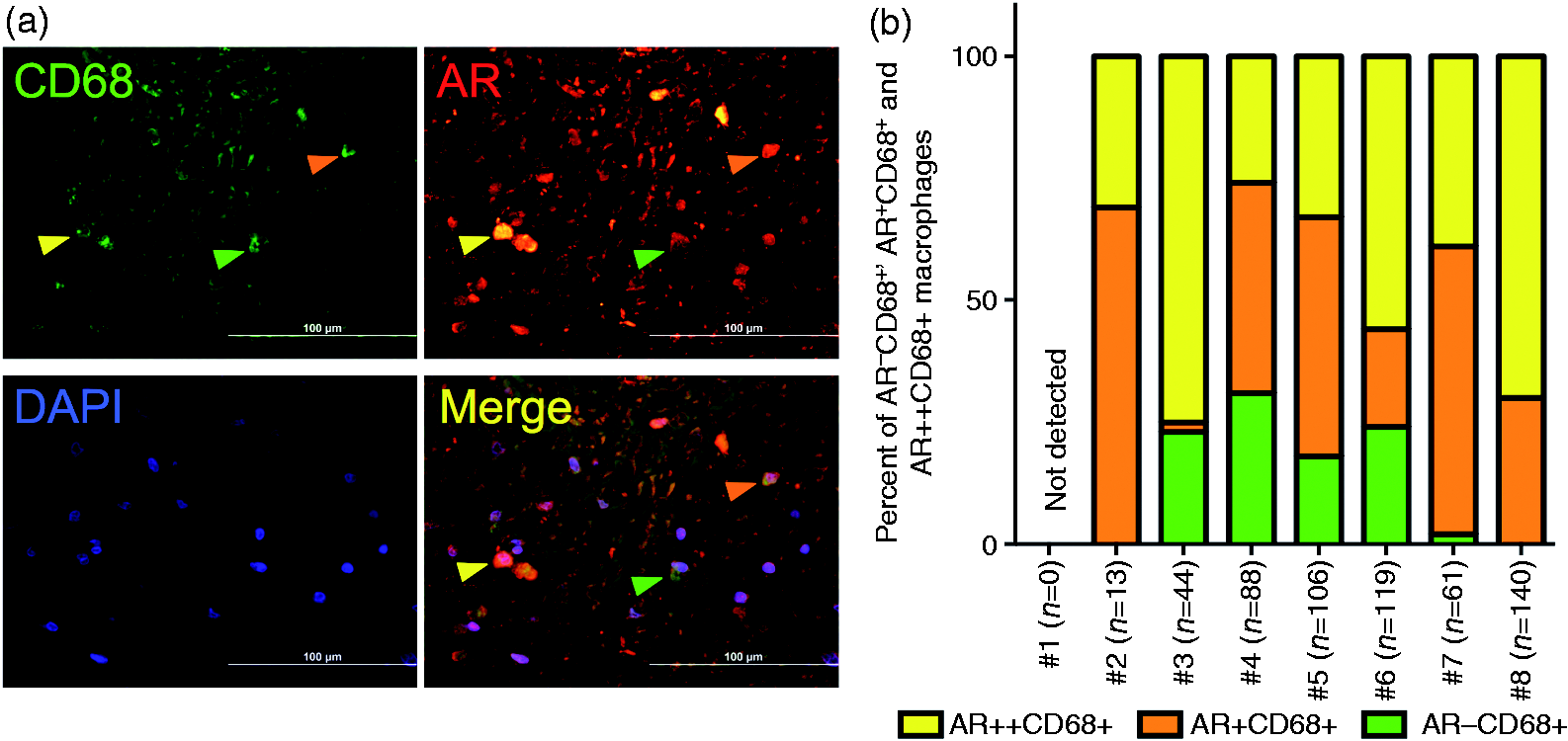
AR is preferentially expressed by M1-polarized macrophages
AR is preferentially expressed by M1 polarized macrophages. Gene expression of (a) M1 and (b) M2 markers in M1 or M2 polarized human monocyte-derived macrophages as determined by quantitative PCR. 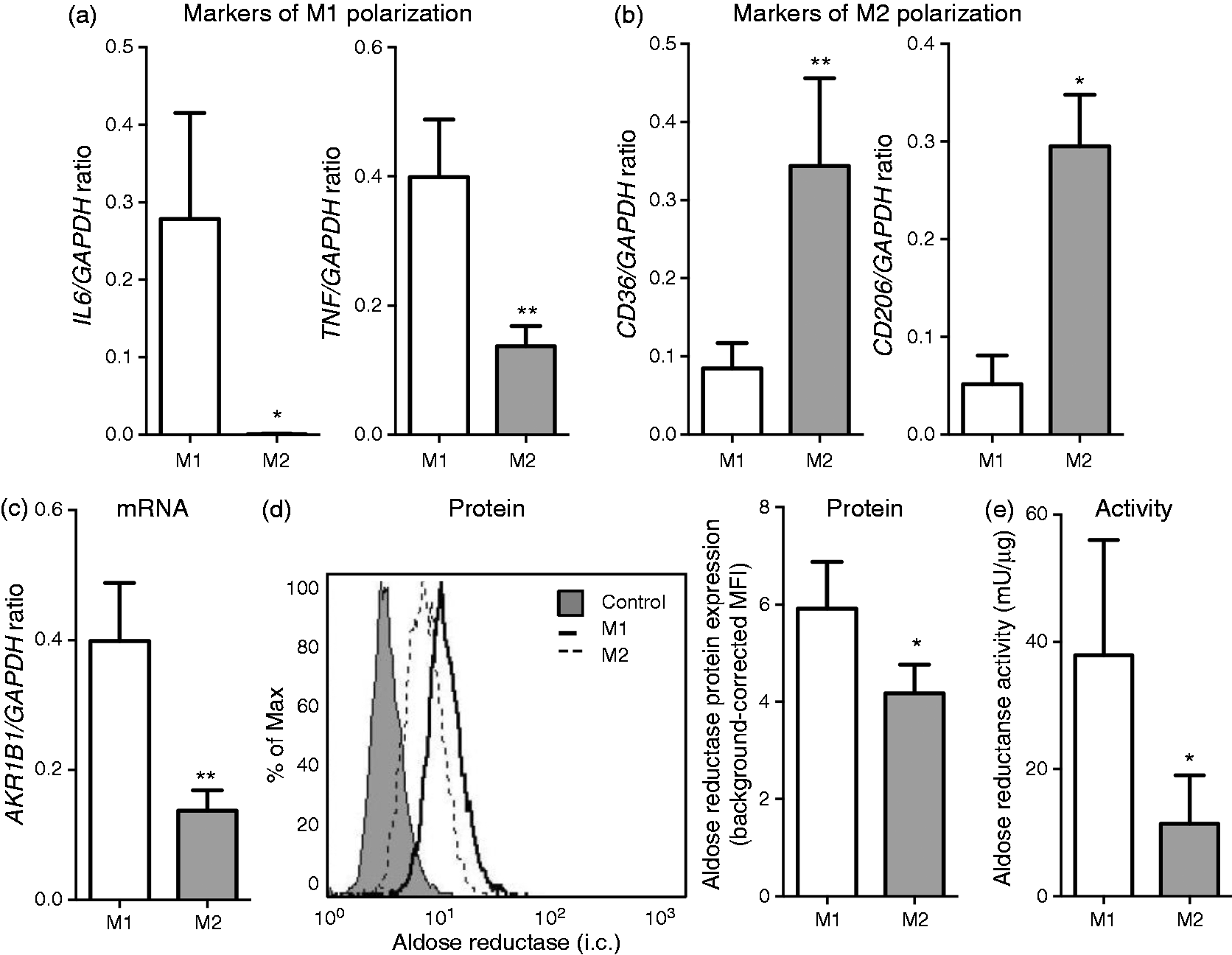
The notion that AR expression is associated with M1 polarization was furthermore confirmed when correlating the expression levels of
Differential AR expression between M1 and M2 macrophages depends on hyperglycemia
As many other Overexpression of AR in M1 macrophages depends on hyperglycemia. (a) Relative 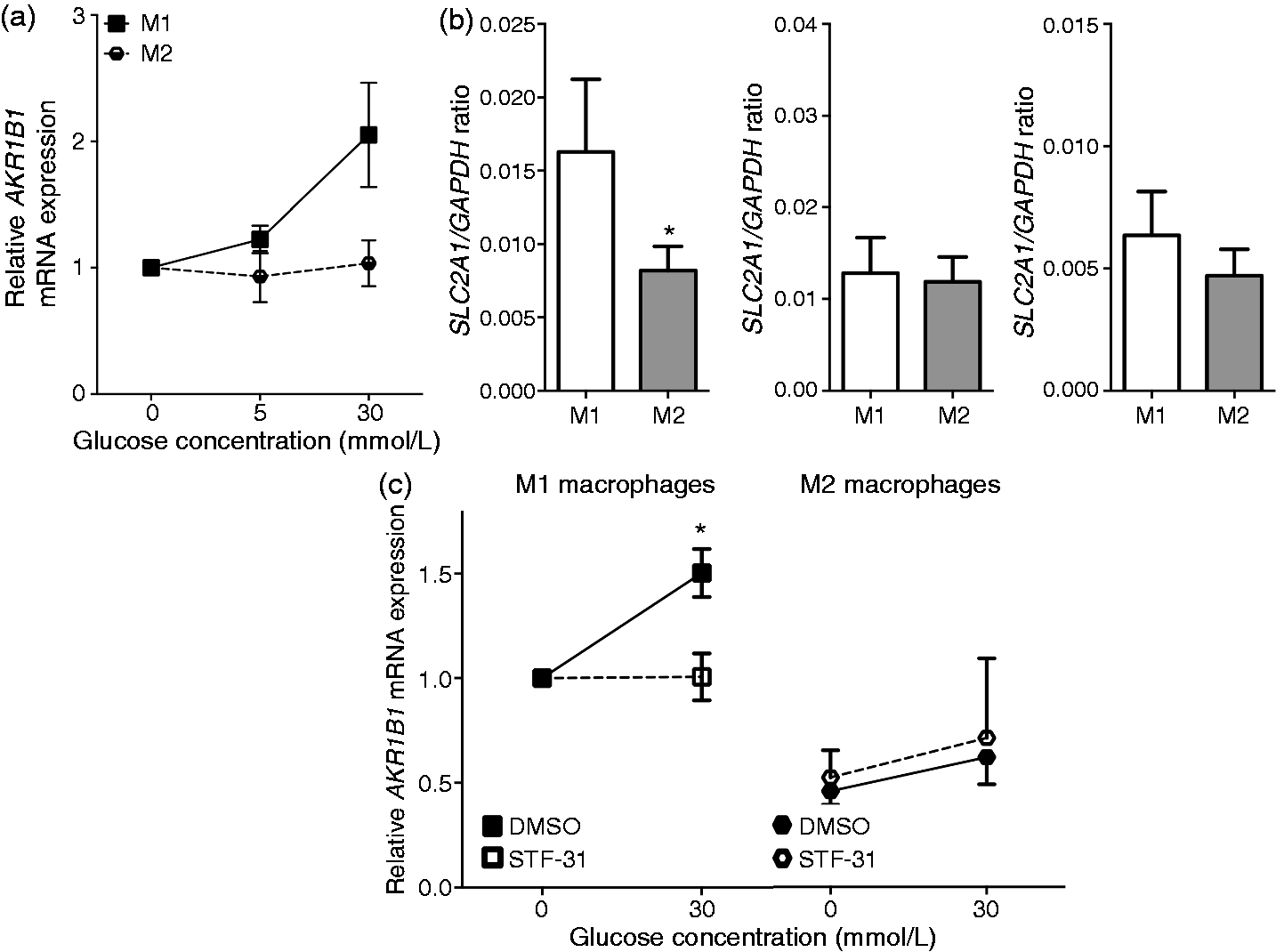
As
To test whether increase of GLUT-1 expression was causally involved in increased AR expression in M1 macrophages, STF-32 was used as a specific pharmacological inhibitor. While treatment macrophages with STF-32 completely inhibited Glc-induced AR up-regulation, it did not affect AR expression in M2 macrophages (Figure 4c). These data suggest that differential AR expression in plaque macrophages may at least partially depend on increased Glc levels.
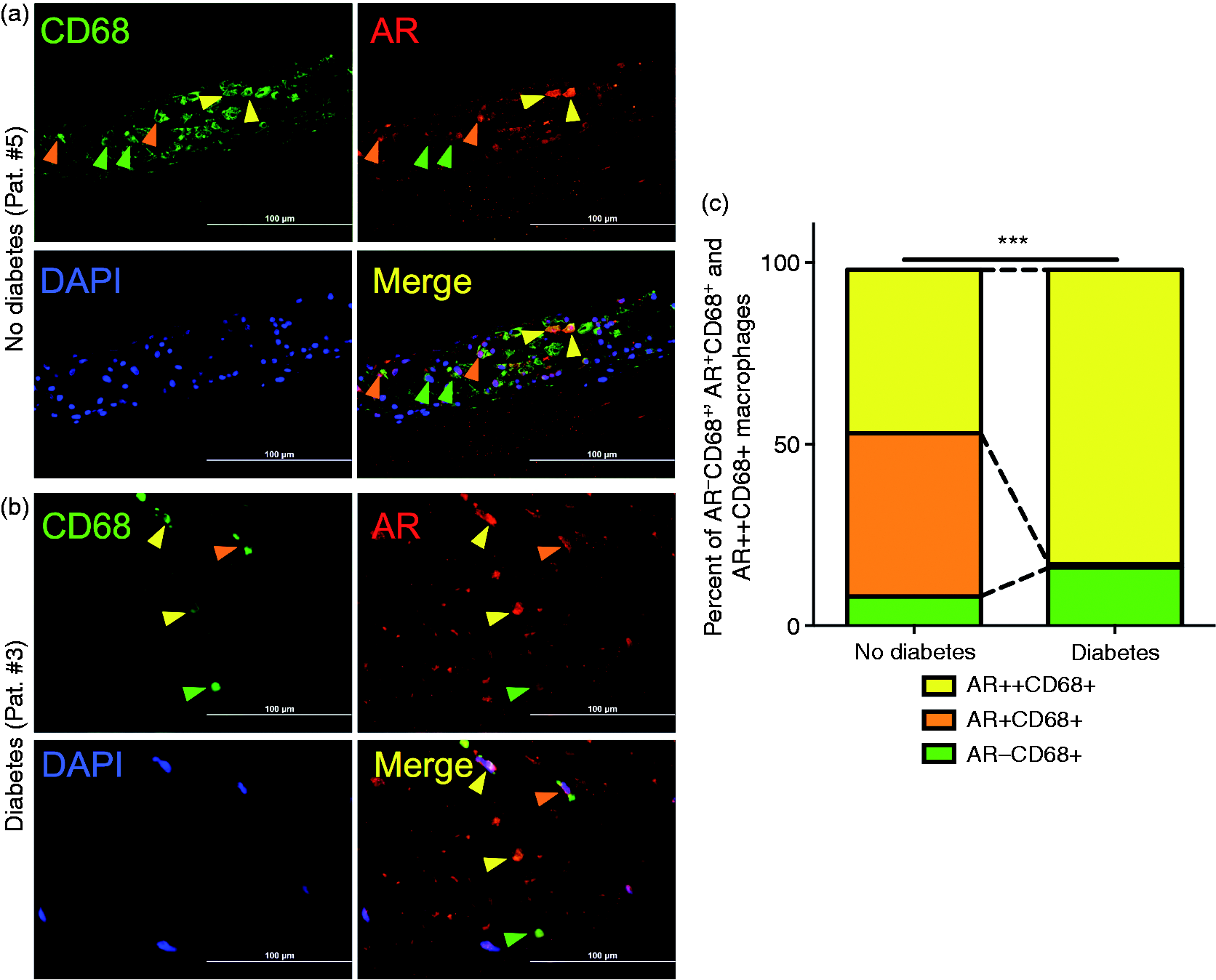
Diabetic patients display a greater proportion of CD68+AR++ macrophages than non-diabetic patients
Our
While the percentage of CD68+AR- macrophages was moderately higher in diabetic than in non-diabetic patients (increased by eight percentage points), the proportion of CD68+AR++ was significantly higher (increased by 36 percentage points, Correlation between Coronary artery plaques from diabetic patients display a greater degree of macrophage heterogeneity regarding AR expression. Representative immunofluorescence stainings of post-mortem coronary arteries against CD68 (green) and AR(red) from (a) a non-diabetic and (b) a diabetic patient. DAPI (blue) as nuclear stain. Yellow arrows indicate CD68+AR++, orange arrows CD68+AR+ and green arrows CD68+AR− macrophages. Scale bars indicate 100 µm. (c) Relative proportions of a CD68+AR++ (yellow), CD68+AR+ (orange) and CD68+AR− macrophages in non-diabetic and diabetic individuals (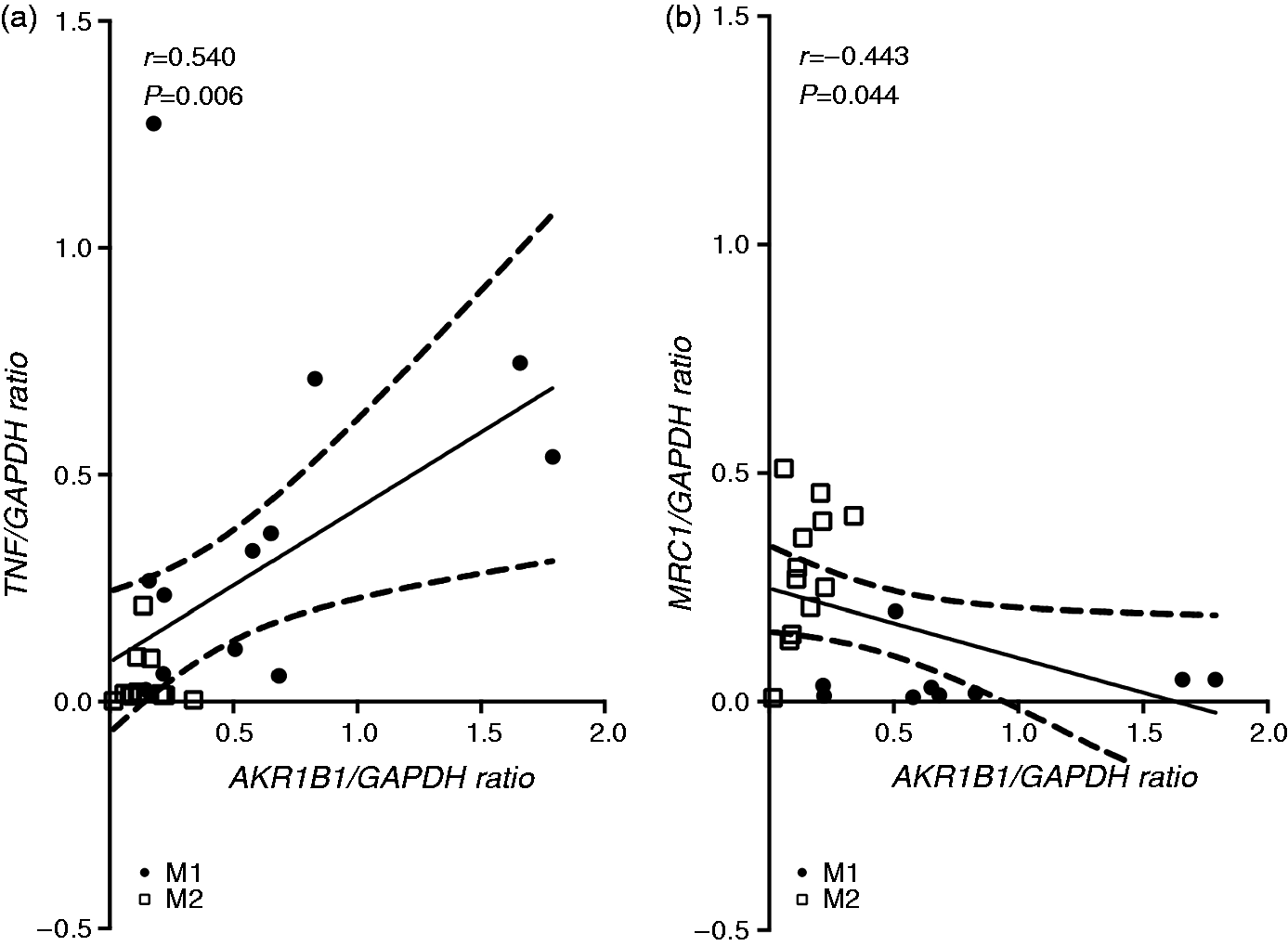
Discussion
Diabetes may promote atherogenesis and destabilization of atherosclerotic plaques by various mechanisms.6,7 One of the mechanisms associated with hyperglycemia is the induction of AR, the rate-limiting enzyme of the polyol pathway. We have previously shown that a subset of human plaque macrophages expresses AR and that its expression is synergistically induced by hyperlipidemia and hyperglycemia.
15
Here, we expand these findings by showing that AR expression is mainly found in M1 macrophages, while expression in M2 macrophages is rather low. Also, we find that differential AR expression depends on hyperglycemia, that is, with increasing Glc levels AR expression increases in M1 but not M2 macrophages. As underlying mechanism, we propose increased expression of the Glc transporter GLUT-1 in M1 macrophages facilitating Glc uptake and presumably inducing increased AR (
The pathophysiological relevance of macrophage heterogeneity in human atherosclerosis has clearly been recognized.2,3 In 2006, Bouhlel et al.27 demonstrated expression of both M1 and M2 markers within atherosclerotic lesions. Subsequent studies by Waldo et al., 28 Bolye et al., 29 our own group30,31 and others have shown that other macrophage types induced by GM-CSF, hemorrhage or CXCL4 can be identified within atherosclerotic lesions. It is conceivable that plaque destabilization and rupture are promoted by a pro-inflammatory macrophage subset within the lesion. Thus, identification of pro-atherogenic macrophage phenotypes may be of diagnostic and therapeutic value when developing strategies to prevent plaque rupture and subsequent myocardial infarction.
Our data demonstrate that differential macrophage polarization may, in some cases, be affected by cardiovascular risk factors such as hyperglycemia. Thus, differential AR expression between M1 and M2 polarized macrophages clearly depends on hyperglycemic conditions. One explanation may be given by increased expression of GLUT-1 in M1 macrophages: GLUT-1 is one of the three major Glc transporters expressed by human macrophages.26,32 Pharmacological inhibition of GLUT-1 completely abrogated Glc-dependent AR up-regulation; thus, increased GLUT-1 expression in M1 macrophages may lead to increased Glc uptake, which may, in turn, stimulate an osmotic response element that induces AR expression and activity. 25
This
AR has been shown to promote atherosclerosis in hyperglycemic, diabetic
In conclusion, we demonstrate that polarization of plaque macrophages may be affected by cardiovascular risk factors such as hyperglycemia. Our data therefore illustrate the complex interplay between systemic and local factors in the pathogenesis of atherosclerotic disease.
Footnotes
Acknowledgments
We thank Dr Klaus Ley (La Jolla Institute for Allergy & Immunology, La Jolla, CA, USA) for providing post-mortem coronary artery specimens; we also acknowledge John Sanders and Nadine Wambsganss for their excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Cardiology Career Program (University of Heidelberg) to G.R., by the DZHK (German Centre for Cardiovascular Research) and by the BMBF (German Ministry of Education and Research) to H.A.K
