Abstract
Carbamylation of lysine residues and protein
Introduction
Post-translational modifications (PTMs) are pivotal steps in protein maturation. PTMs increase the functional diversity of the proteome and play a key role in multiple cellular processes, including protein–protein interactions, cell signaling, differentiation and regulation of gene expression.
1
Carbamylation is a ubiquitous, non-enzymatic PTM, in which cyanate (OCN−) reacts with primary amino groups (R-NH2) at the
The abundance of MPO at inflammatory foci has sparked significant interest in the role of carbamylation in the context of chronic inflammatory diseases and atherogenesis.3,6,7 Apart from directly affecting protein function and turnover, homocitrulline residues act as neo-epitopes that can trigger primary immune responses, thereby inducing chemotaxis and proliferation of CD4+ T cells and the subsequent production of IFN-γ, IL-10 and IL-17. In addition, Abs against carbamylated residues have clinical value in that they predict a more erosive progression of rheumatoid arthritis.8,9
Cationic antimicrobial peptides (CAMPs) are essential components of human innate immunity and are produced by a variety of cells, including epithelial cells, 10 keratinocytes 11 and neutrophils. 12 In humans, CAMPs are represented by α- and β-defensins, and the cathelicidin-derived LL-37 peptide. The active form of LL-37 is characterized by a high abundance of arginine and lysine residues, which generate a net positive charge of +6 at a neutral pH. Apart from killing a wide spectrum of pathogenic bacteria,13,14 LL-37 neutralizes LPS, functions as a chemoattractant for immune cells (including T cells, monocytes, neutrophils and mast cells),15,16 profoundly affects the course of dendritic cell maturation, 17 stimulates production of cytokines, chemokines and their receptors,18,19 and triggers mast cell degranulation. 16 These effects are mediated, at least in part, by activation of at least four different receptors: formyl peptide receptor-like 1 (FPRL1), epidermal growth factor receptor, P2X7 and CXCR2. Finally, recent studies show that LL-37 has a direct effect on the cells and might be hemolytic or cytotoxic to peripheral blood mononuclear cells (PBMCs) at high concentrations.20,21 At low concentrations (< 5 μM), LL-37 induces rapid secondary necrosis of apoptotic human neutrophils. 22 To prevent collateral tissue damage due to exacerbation of inflammation, the activity of LL-37 is strictly controlled by serum proteins, 23 most likely apolipoprotein-A1 (ApoA-1). 24 Taken together, all available data indicate that the antibacterial activity of LL-37 is secondary to its immunomodulatory functions. In a twist of the paradigm, it is now generally accepted that the major role of CAMPs is not direct killing of invading microbes, but rather acting as signaling molecules for innate and acquired immunity.
Within the inflammatory milieu, the concomitant release of LL-37 and MPO by activated neutrophils may create conditions required for LL-37 carbamylation. Additionally, lysine residues within CAMPs are crucial for peptide structure and activity, and conversion of these positively charged residues to neutral homocitrulline residues would be expected to abrogate the biological activity of LL-37.
Here, we performed mass spectrometry (MS)-based sequence analysis and found that LL-37 undergoes rapid carbamylation in the presence of cyanate in a time- and concentration-dependent manner. Unexpectedly, we found that the free amino group of the N-terminal leucine was most susceptible to carbamylation under conditions that reflected the cyanate concentration in the inflammatory milieu. Prolonged incubation resulted in the generation of a mixture of variably carbamylated LL-37 molecules with impaired antimicrobial activity against both Gram-negative and Gram-positive species.
Taken together, these results suggest that carbamylation of LL-37 within an inflammatory environment might actually exacerbate inflammation and be detrimental to the host.
Materials and methods
In vitro carbamylation of LL-37 and MS analysis
LL-37 (ProImmune, Oxford, UK) was carbamylated by incubation with 10, 50 and 100 mM KCNO (Sigma-Aldrich, Oslo, Norway) in 100 mM HEPES (pH 7.8) for 3 h at 37℃. The reaction was quenched by addition of formic acid to a final concentration of 5% and samples were immediately purified using StageTips (ThermoScientific, Waltham, MA, USA). The samples were lyophilized and either analyzed directly by LC-MS/MS or subjected to proteolytic digestion with
Peptide synthesis
Native and carbamylated LL-37 were synthesized by Proimmune (Oxford, UK) by using Fmoc solid-phase peptide synthesis, diluted in 0.01% v/v acetic acid and stored at −70℃ until use.
Circular dichroism spectroscopy
The secondary structure of the LL-37 analogues was investigated by circular dichroism (CD) spectroscopy. The experiments were performed using a Jasco J-810 spectropolarimeter (Jasco, Essex, UK). Far-UV spectra were acquired at 37℃ in the 195–260 nm range at a scan rate of 50 nm/min and a band width of 1 nm. Three scans were accumulated for each sample and appropriate blanks were subtracted from each spectrum by using the software provided by the instrument manufacturers. The peptides (10 µM) were analyzed upon dilution in a 10 mM sodium phosphate buffer containing 50% v/v trifluorethanol (TFE) or a physiological salt solution resembling plasma (113 mM NaCl, 24 mM NaHCO3, 0.6 mM MgCl2, 1.3 mM CaCl2, 3.9 mM KCl) in 1.0-mm quartz cuvettes (Hellma-Analytics, Oslo, Norway). The mean ellipticity was calculated using the formula [θ] = θ/(10·c·l), where θ is the ellipticity (mdeg), 10 is a scaling factor, c is the protein concentration (M) and l is the path length of the cuvette (cm). The helical content (percentage of helix) was estimated by using the CDNN program from the molar ellipticity θ [deg.cm2 dmol–1].
Broth microdilution assay
Frozen samples of
Culture of human monocyte-derived macrophages
Human blood samples were collected from healthy volunteers by using heparin-coated tubes (BD, Franklin Lakes, NJ, USA) and diluted 1:1 in PBS without calcium and magnesium. PBMCs were isolated by density gradient separation on Lymphoprep (Axis-Shield Poc AS, Oslo, Norway) and diluted in macrophage medium (RPMI 1640 containing UltraGlutamin supplemented with 10% v/v autologous human serum and penicillin streptavidin; Sigma-Aldrich). Subsequently, 3 × 106 cells/well were plated in 24-well plates (Sarstedt, Nümbrecht, Germany) and incubated for 24 h at 37℃ in 5% CO2. Non-adherent PBMCs were removed by washing with PBS. Adherent cells were cultured in regularly changed macrophage medium for a minimum of 10 d.
Cytokine production of hMDMs
Following washing (three times) with PBS mature hMDMs were incubated with RPMI medium containing 0.5 µM native or carbamylated LL-37 together with 100 ng/ml
Hemolysis of erythrocytes
Peripheral blood was collected in heparin-coated tubes (BD) from healthy volunteers and centrifuged at 800
Chemotaxis of neutrophils
The Insall chamber was used to visualize chemotaxis. 25 For each sample, discontinuous Percoll gradient-isolated neutrophils (400 μl in RPMI, final density 1 × 106/ml) were added to acid washed (0.2 M HCl), dried and blocked (7.5%, BSA 400 μl; Sigma) coverslips (22 mm; VWR International, Radnor, PA, USA), which were then incubated at room temperature (approximately 23℃) for 30 min, to allow the cells to adhere. The coverslip was then inverted and placed at the top of the chemotaxis chamber ensuring that the chemoattractant loading bays were exposed. The desired chemoattractant (80 µl), fMLP (10 nM) or LL-37 and its modified forms (used at 20 µM after assessing a range of concentrations) or control (RPMI media) was injected into the chemoattractant channels. The cell movement was analyzed using a Zeiss Primovert microscope (Carl Zeiss Imaging, Thornwood, NY, USA) and images captured every 30 s for up to 40 frames per condition using a Q Imaging Retiga 2000R camera (Qimaging, Surry, Canada).
Binding of LL-37 and ApoA1 by surface plasmon resonance
The surface plasmon resonance experiments were performed using a BIACORE 3000 instrument (GE Healthcare, Little Chalfont, UK). The N-HisTag containing ApoA1 (NH-ApoA1) was immobilized on the surface of a NTA sensor chip (GE Healthcare), according to manufacturer’s protocol. Shortly, for direct capture of NH-ApoA1, the surface of NTA chip was prepared by 1 min injection of 0.5 mM NiCl2, then NH-ApoA1 at concentration of 2.5 µg/ml diluted in running buffer (10 mM HEPES, 150 mM NaCl, and 0.005% surfactant (v/v), pH 7.4) was injected with a flow rate of 5 μl/min to achieve capture level between 800 and 1000 resonance units (RU). The surface-stabilized ApoA1was primed with running buffer before subsequent assay steps. The binding of all LL-37 analogs were each tested in triplicate in a concentration range up to 1000 nM in running buffer. All samples were injected at a flow rate of 5 μl/min. Between experiments, the surfaces were strictly regenerated with two pulses (30 s) of 1 M NaCl at a flow rate of 20 μl/min, followed by an extensive wash procedure with running buffer. All measurements were performed at a constant temperature of 25℃. Sensorgrams corrected for the reference were aligned, and a blank run was subtracted.
Results
LL-37 undergoes rapid carbamylation, which affects the peptide structure
Multiple studies based on circular dichroism,
23
Fourier transform infrared
26
and NMR spectroscopy
27
reveal that LL-37 exists as a linear cationic, amphipathic α-helical structure within vesicles, both within the lipid bilayer and in solutions with an ionic composition resembling that of intracellular fluid or plasma. We used MS to show that LL-37 undergoes rapid modification in the presence of cyanate, with the number of carbamylated residues increasing in a concentration- and time-dependent manner (Figure 1A–D). In the presence of 10 mM KCNO (equivalent to cyanate levels found in the inflammatory milieu), multiple forms of carbamylated LL-37 were detected in the reaction mixture after only 10 min. Closer analysis of the elution pattern of modified peptides from a RP-HPLC column revealed two distinct forms of LL-37 bearing a single homocitrulline, which were the most commonly detected modifications. Although almost 70% of the modifications were represented by a single site substitution, a variety of peptides carrying multiple (two, three or four) carbamylated residues were also detected (Figure 1B). Increasing either the cyanate concentration (to 50 mM and 100 mM) or incubation time shifted the modification pattern. In 50 mM KCNO, previously predominant peptides bearing a single modification were replaced by peptides bearing multiple carbamylations on different Lys residues, resulting in a highly heterogeneous mixture. Peptides of the same molecular mass bearing one or more carbamylated amino groups resolved into several peaks (same color in Figure 1C), which depended on the particular combination of the amino groups modified. This suggests that all six lysine side chains in LL-37 were equally susceptible to carbamylation. This was confirmed by analysis of LL-37 in the presence of 100 mM cyanate, which showed the presence a of a heterogenous population of LL-37 with both five and six carbamylations (several peaks with same color Figure 1C).
The number of carbamylated amino acid residues increases with KCNO concentration. LL-37 was incubated for 3 h with increasing amounts of KCNO. (A) 0 mM, (B) 10 mM and (C) 50 mM. The samples were then analyzed by LC-MS/MS and the 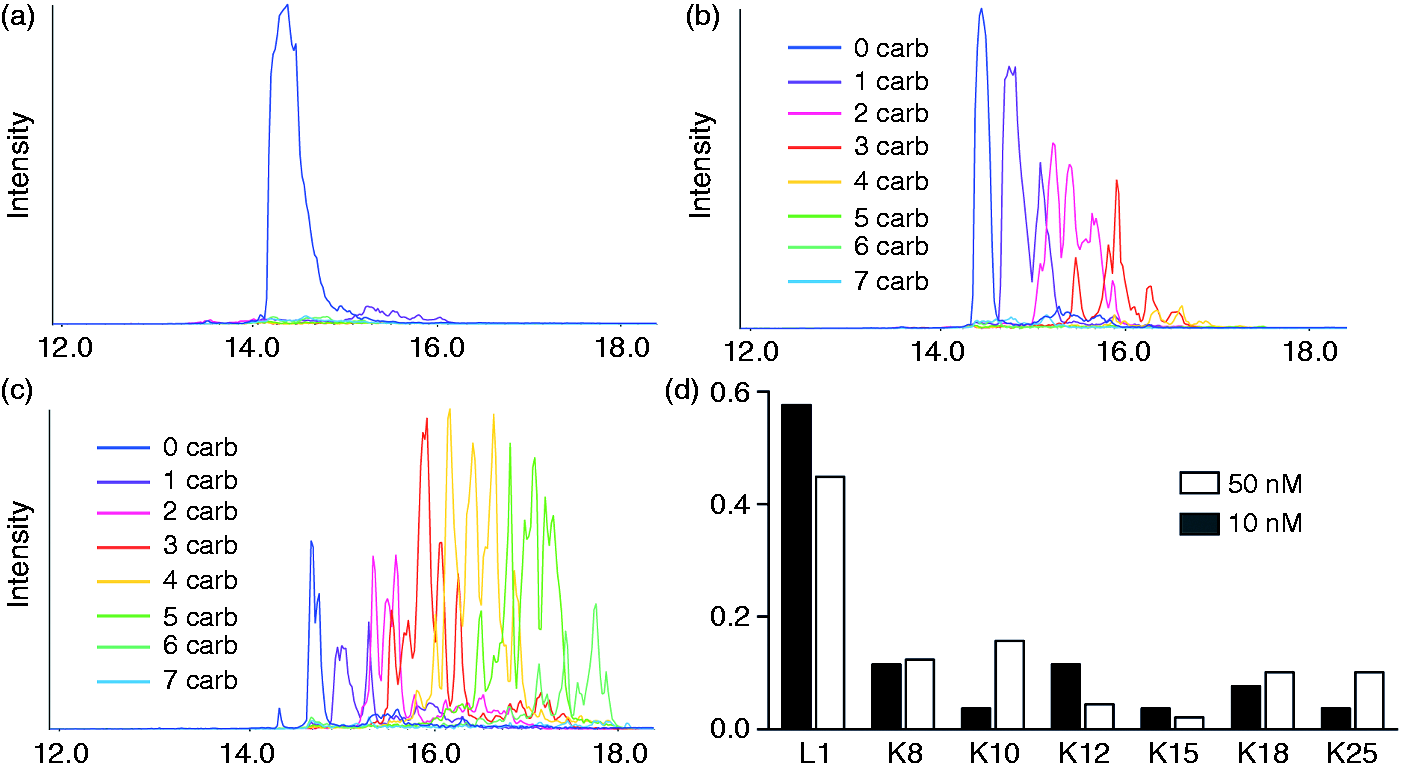
To address the functional significance of LL-37 carbamylation, we focused our attention on modifications identified after incubating the peptide in an environment mimicking that of the inflammatory milieu. To this end, we first identified the most common carbamylated variants of LL-37 under these conditions. KCNO-treated samples were digested with
LL-37 peptides synthesized and examined in this study.
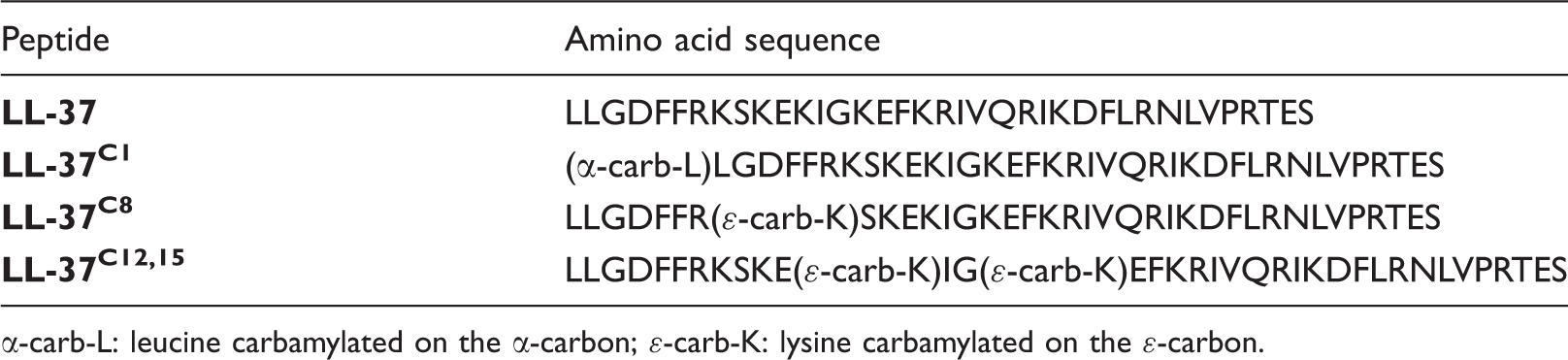
α-carb-L: leucine carbamylated on the α-carbon; ɛ-carb-K: lysine carbamylated on the ɛ-carbon.
In aqueous solution, LL-37 forms a random coil structure but adopts an α-helical conformation under physiological conditions; the α-helical structure is also adopted in TFE or lipid bilayer vesicles.
26
In the latter case, NMR studies show that LL-37 comprises three basic parts: an CD spectra of native and carbamylated LL-37. The peptide concentration was 10 μM. The TFE buffer comprised 50% TFE in 10 mM sodium phosphate buffer. The plasma buffer contained 113 mM NaCl, 24 mM NaHCO3, 0.6 mM MgCl2, 1.3 mM CaCl2 and 3.9 mM KCl. (A) Spectra represent the mean of two (50% TFE buffer) or three (plasma buffer) independent experiments. (B) Differences in the predicted α-helical content of the peptide analogues in 50% TFE buffer at 190–260 nm. (C) Difference in the predicted α-helical content of the peptide analogues in plasma buffer at 190–260 nm. (B, C) Data are expressed as the mean ± SD. Statistical significance was evaluated by one-way ANOVA, followed by Tukey’s multiple comparison test. *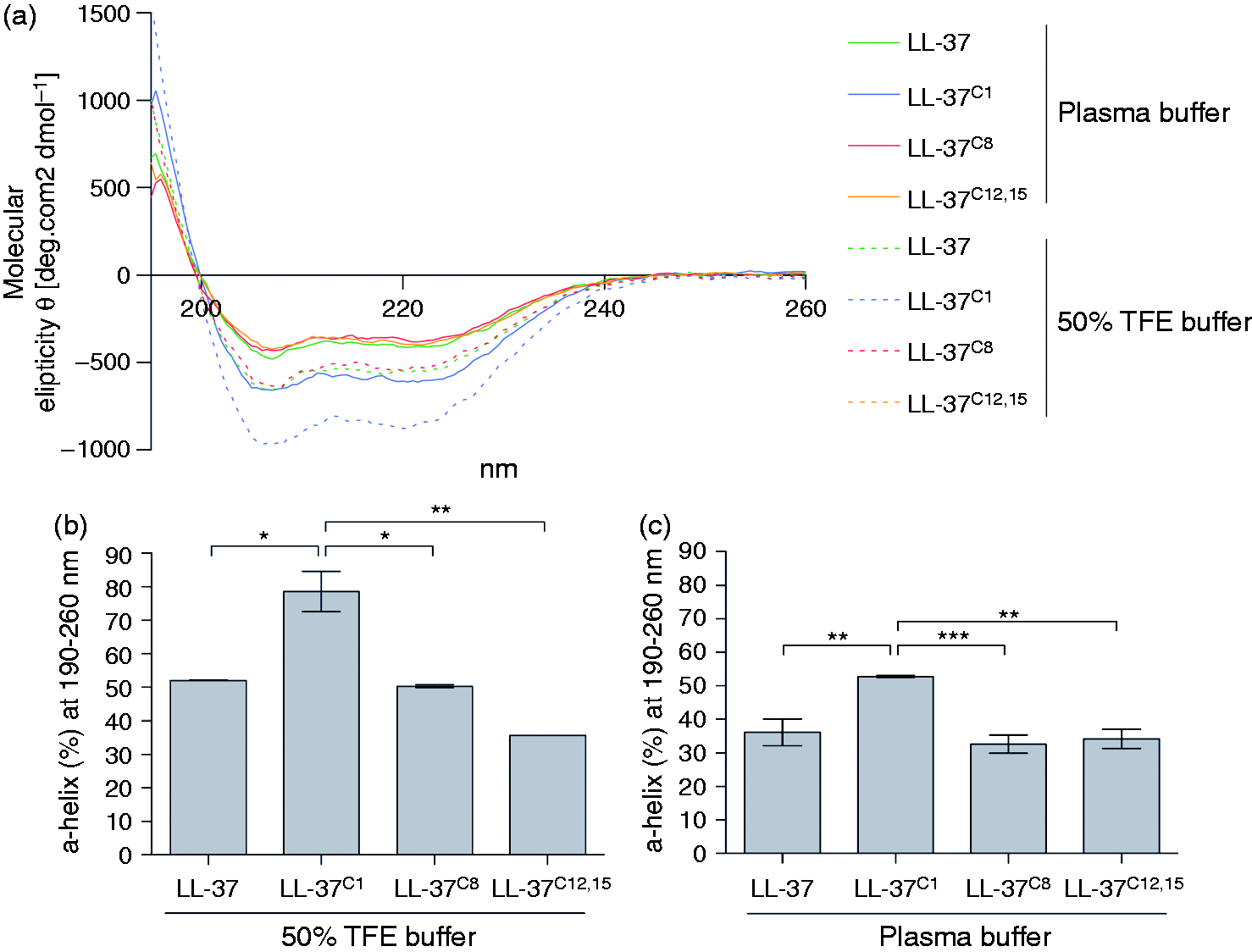
As expected, incubation with TFE increased the ellipticity and the helical content of the peptides beyond that observed in plasma buffer (Figure 2B). Under these conditions, CD analysis of the far-UV spectra predicted an α-helical content of approximately 80% for LL-37C1 but approximately 50% for the native peptide (Figure 2B). However, the difference between the peptides in TFE was similar to that observed in plasma buffer, that is, the α-helical composition of LL-37C1 was about 20-30% greater than that of the other peptides (Figure 2C).
Carbamylation abrogates the bactericidal capacity of LL-37
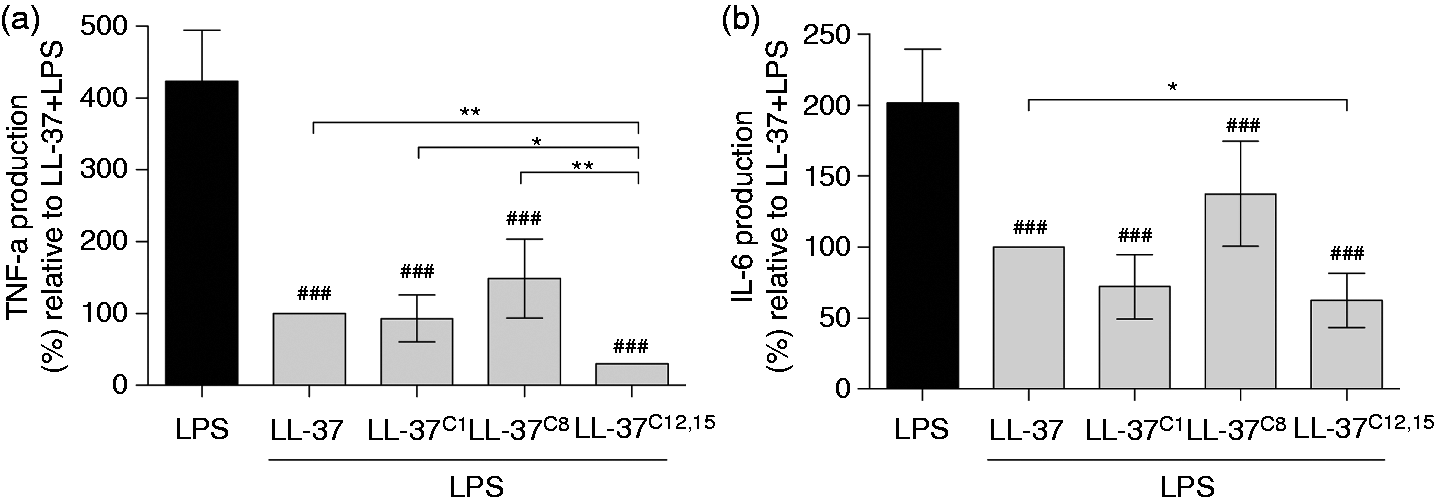
LL-37 interacts with bacteria via electrostatic and hydrophobic interactions resulting in membrane permeabilization and disruption. A broth microdilution assay revealed that the bactericidal activity of LL-37 was profoundly affected by carbamylation. At a peptide concentration of 1 µg/ml (i.e. 0.2 μM), LL-37C8 and LL-37C12,15 demonstrated impaired ability to inhibit bacterial growth of Carbamylation abrogates the antimicrobial activity of LL-37. (A–C) The number of colony-forming units formed after incubation with (A) LPS-induced cell activation remain supressed in the presence of carbamylated LL-37. Human monocyte-derived macrophages were incubated with 100 ng/ml LPS in the absence or presence of modified peptides. The levels of (A) TNF-α and (B) IL-6 in the supernatants were then measured. LL-37 + LPS was set at 100%. Data are expressed as the mean ± SD. Statistical significance was evaluated by one-way ANOVA, followed by Tukey’s multiple comparisons post-test. *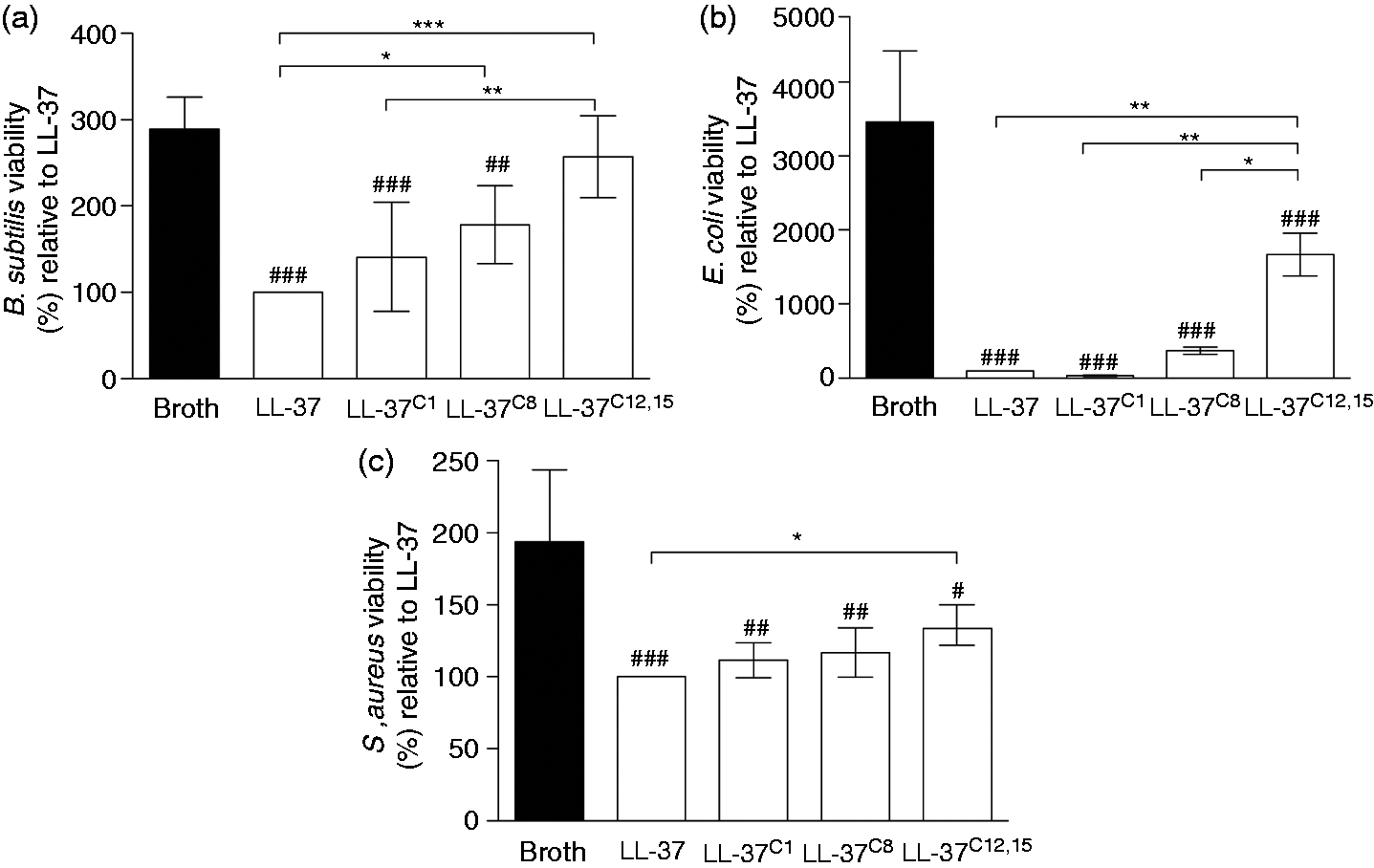
Carbamylation of LL-37 affects its affinity for ApoA1
Carbamylation of LL-37 affects its affinity for ApoA1. Surface plasmon resonance curves showing the interaction between ApoA1 and carbamylated and native LL-37 at a concentration of 1 μM.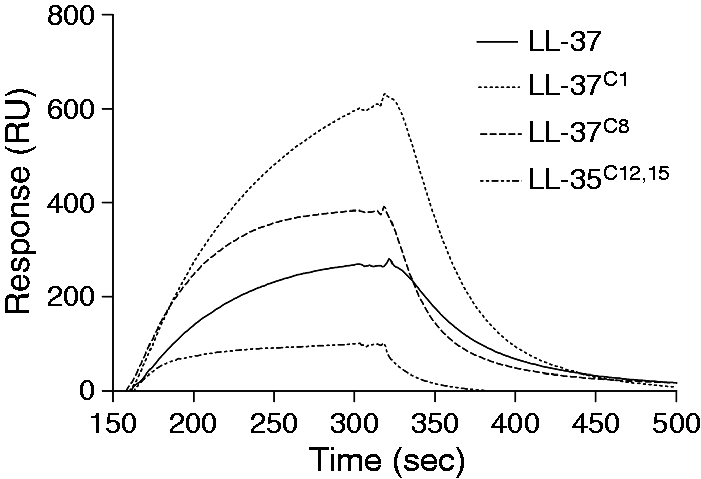
Carbamylation affects the chemotactic capacity of LL-37
The ability of neutrophils to efficiently reach sites of inflammation is pivotal for efficient elimination of pathogens and restriction of potential tissue damage. LL-37 is a strong chemoattractant for neutrophils, monocytes and T cells, via the FPRL1 receptor.
15
To investigate how carbamylation affects the chemotactic activity of LL-37, we used a ‘state-of-the-art’ approach that allowed us to observe cell migration in real time. The Insall chambers used in the study provide a gradient for the cells to migrate against, rather than simply exposing cells to the chemoattractant alone. This, in turn, allows observation of cell migration in more detail as information about speed (average speed of the cell over time in any direction), velocity (average speed of the cell in the direction of the gradient over time), and the directional accuracy of chemotaxis (expressed as the chemotactic index) can be obtained. We found that a peptide concentration of 20 μM was optimal in our assay (tested concentrations, 10–40 μM; data not shown). Both native LL-37 and the carbamylated forms induced neutrophil migration without any apparent toxicity (Figure 6A–C). However, neutrophils were significantly less responsive to the carbamylated versions of LL-37. Both the speed and velocity of migrating neutrophils were significantly lower when LL-37C1, LL-37C8 and LL-37C12,15 were used as chemoattractants. Interestingly, LL-37C1 was not only a significantly weaker chemoattractant than the other carbamylated peptides, but it also showed lower directional accuracy. Taken together, these observations suggest that, although all of the homocitrullinated peptides were able to trigger cell migration, carbamylation of the Carbamylation reduces the chemotactic potential of LL-37. Analysis of chemotaxis at a peptide concentration of 20 μM. Neutrophils from four healthy volunteers were treated with either native or modified peptide. Values for speed, velocity, chemotactic index, and the resultant vector length were analyzed. The midline of each box represents the median value. Statistical significance was evaluated by one-way ANOVA, followed by Tukey’s multiple comparisons post-test: **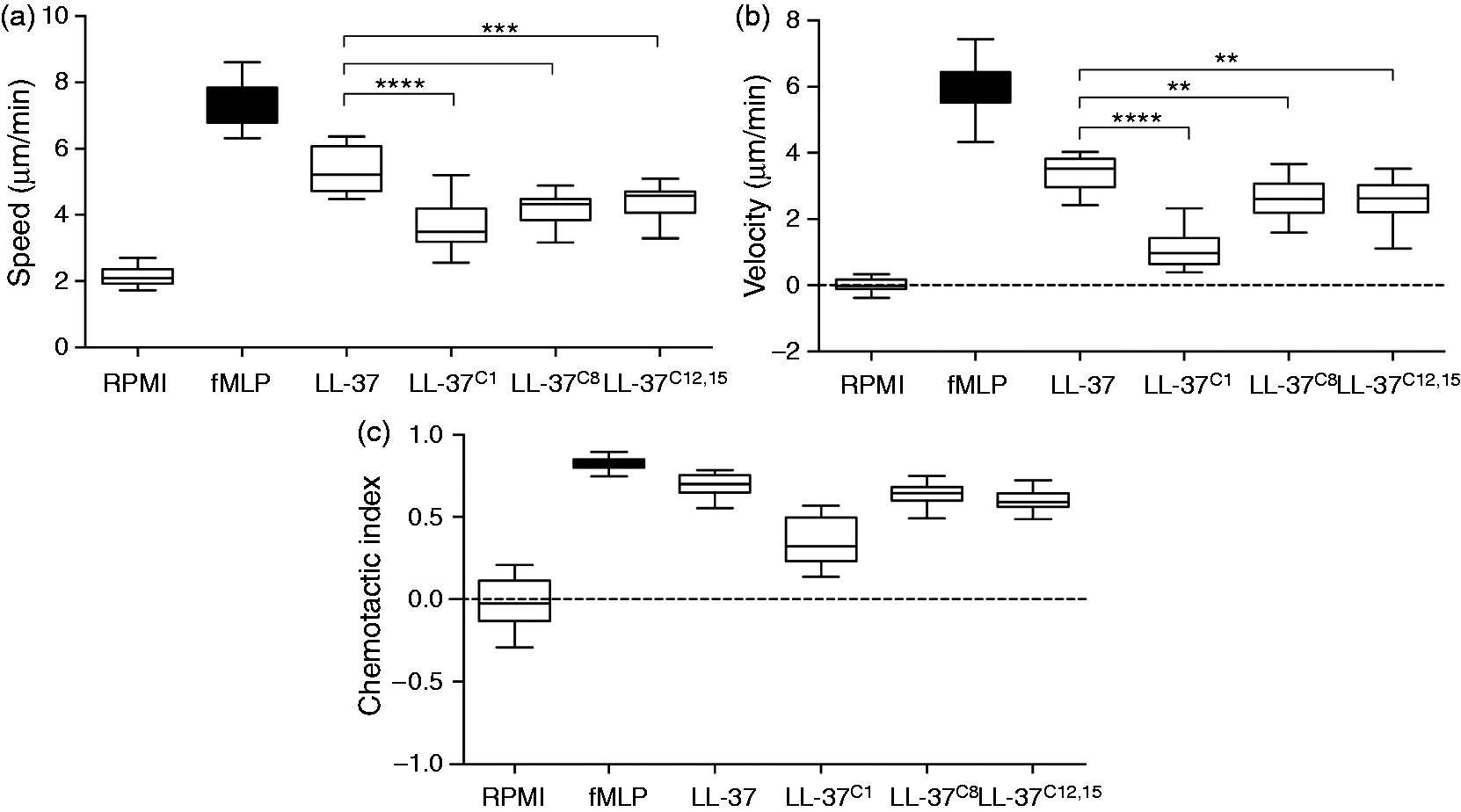
Carbamylation affects the cytotoxicity of LL-37
LL-37 can be cytotoxic to host cells. Therefore, we used a highly sensitive hemolytic assay to examine the impact of carbamylation on the cell-permeabilizing effects of LL-37. Human red blood cells (hRBC) were incubated with the peptide for 2 h at 37℃. The results showed that LL-37C1 exerted a very strong and concentration-dependent lytic effect. The peptide caused a significant increase in hemoglobin release when used at a concentration of 2 μM compared with PBS ( Carbamylation significantly affects the hemolytic capacity of LL-37. Erythrocytes were incubated for 2 h with each LL-37 analogue at concentrations ranging from 1 to 20 μM. The hemolytic activity of the peptides was evaluated by recording the release of hemoglobin from human erythrocytes at 405 nm. Data are representative of four individual experiments and are expressed as the mean ± SD. Statistical significance was evaluated by one-way ANOVA, followed by Dunnett’s multiple comparisons post-test: **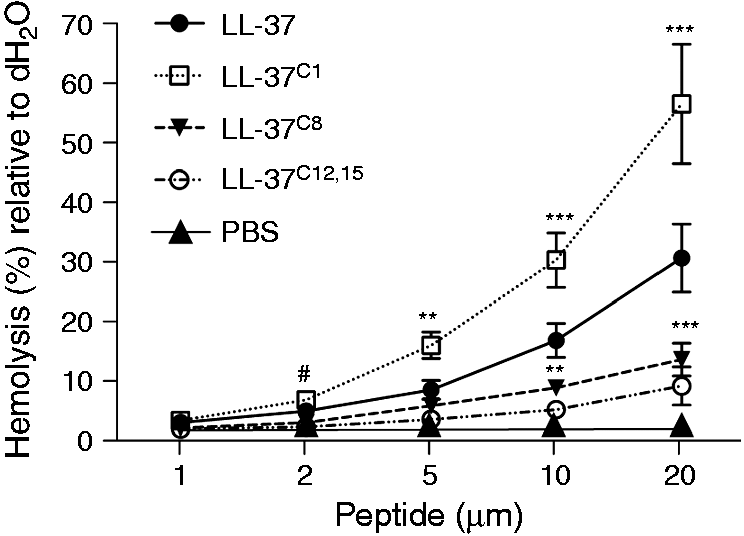
Discussion
Host antimicrobial peptides, in particular LL-37, are potential novel therapeutics, mainly owing to their wound healing, antiseptic and antimicrobial properties.14,30,31 In the era of rapidly increasing drug resistance among pathogens, novel treatments based on LL-37-derived peptides are very much needed. In addition, the immunostimulatory and cytotoxic properties of LL-37 may be useful tools for cancer treatment, 32 or even as a vaginal contraceptive. 33 It is, however, important to keep in mind that the effector functions of LL-37 are most often executed at local sites of inflammation, and mainly in the context of neutrophil activation and subsequent release of MPO and H2O2. 34 Thus, LL-37 is mainly present in microenvironments that foster carbamylation. Therefore, it is important to examine the impact of carbamylation on the immunomodulatory and cytotoxic functions of LL-37 prior to the clinical administration of cathelicidin-derived peptides. Here, we explored the effects of carbamylation on the biological functions of LL-37.
We found that in the presence of cyanate, LL-37 undergoes rapid modification to generate a pool of peptides, each with a diverse carbamylation pattern. MS analyses revealed that the
The antimicrobial and cytotoxic activities of LL-37 are effectively inhibited in human plasma. This latency is due to the interaction between LL-37 and its carrier protein, apoA-1;
24
this interaction is dependent on the hydrophobicity of the
Upon carbamylation, the bactericidal activity of LL-37 is compromised. Again, this is most likely related to the loss of two positive charges when Lys residues are converted to homocitrulline. It is likely that the reduced electrostatic attraction between LL-37C12,15 and bacterial membranes results in impaired antimicrobial activity against
Interestingly, LL-37C12,15 was the most effective LPS-detoxifying agent among all tested forms of LL-37, that is, this carbamylated peptide efficiently attenuated the LPS-induced production of TNF-α, an important mediator of endotoxic shock. 44 In addition to directly binding LPS via electrostatic and hydrophobic interactions, LL-37 also interacts with the LPS receptor, CD14, to block LPS-induced macrophage activation. 45 We hypothesize that carbamylation of Lys-12 and Lys-15 increases the binding of LL-37 to this receptor. From a clinical point of view, it is tempting to speculate that LL-37C12,15 provides therapeutic protection against septic shock without being cytotoxic. Nevertheless, it must be kept in mind that further carbamylation may alter the biological activity of the peptide.
In summary, carbamylation has a profound impact on the bactericidal, cytotoxic and pro-inflammatory activity of LL-37, which may have detrimental consequences for the host. The pattern of carbamylation is dependent on the OCN− concentration and time of exposure, suggesting that the subsequent effects of this modification will be difficult to foresee and therefore control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grants from the EC Marie Curie ITN (RAPID no. 290246) and (FP7-HEALTH-F3-2012-306029 ‘TRIGGER’). PM and AH were supported by a grant from the National Science Center (2014/14/E/NZ6/00162, Poland), and PM was supported by the Broegelmann Foundation. JP acknowledges support from the National Science Center (2012/04/A/NZ1/00051), National Institutes of Health, NIDCR (DE 09761 and DE 022597) and Polish Ministry of Science and Higher Education (2975/7.PR/13/2014/2).
