Abstract
LL-37 is a cationic peptide belonging to the cathelicidin family that has antimicrobial and immune-modulatory properties. Here we show that the mammalian mono-ADP-ribosyltransferase-1 (ART1), which selectively transfers the ADP-ribose moiety from NAD to arginine residues, ADP-ribosylates LL-37
Introduction
Cationic host defense peptides, like defensins and cathelicidins, are evolutionarily conserved key components of the innate immune system that act as natural antibiotics. They are widely distributed throughout all the kingdoms of life, and are among the most ancient host defense factors.
1
Human defensins are grouped into α- and β-defensins. Human neutrophil peptides (HNPs) 1–4 are the major components of the azurophilic granules of neutrophils, and, together with HD-5 and HD-6, two enteric human defensins, belong to α-defensins. HD-5 and HD-6 have been isolated from the granules of Paneth cells in the small intestine,
2
while β-defensins are mainly expressed in epithelial cells of different organs.3–5 The only cathelicidin produced in humans is the human cationic antimicrobial protein (hCAP)-18 from which the active peptide LL-37 is released by proteolytic cleavage.
6
The hCAP-18 protein is produced by epithelial cells of lungs, skin and gut,7–9 while preformed LL-37 is stored in peroxidase-negative granules of neutrophils.
10
LL-37 contributes in many ways to the host immune response having not only bactericidal,
11
but also chemotactic and angiogenetic activity.12,13 LL-37 can also activate epithelial cells to repair wounds and to secrete chemokines.14,15 LL-37 is a 4.5 kDa α-helical amphiphilic peptide comprised of hydrophobic and hydrophilic residues aligned on opposite sides of the helix, facilitating penetration through cell membranes. The presence of positively charged amino acids, five arginine (R) and six lysine (K) residues (Figure 1A), allows binding to bacterial membranes, as well as to polyanionic macromolecules, like nucleic acids, F-actin or glycosaminoglycans. In the airways of patients affected by cystic fibrosis, the antibacterial properties of LL-37 are strongly reduced as a consequence of direct interaction of LL-37 with DNA and F-actin.
16
Further, LL-37 complexed with self-DNA or self-RNA was found in skin lesions of patients with psoriasis,17,18 while self-DNA/LL-37 complexes were found in neutrophil extracellular traps released by neutrophils of patients with lupus erythematosus.19,20 Nucleic acid/LL-37 complexes are powerful trigger of type I IFN production by plasmacytoid dendritic cells or monocytes,17,21 and thus not only contribute to inflammation and to the aggravation of auto-immune diseases, but also to protection against viral infections.
(A) Amino acid sequence of LL-37. The arginine (R) residues that could be the possible target of ART1 are in bold. (B) Schematic representation of the ART1-catalyzed reaction. ART reactions proceed through a nucleophilic substitution-2 (Sn2)-like mechanism in which the nucleophilic attack by the preferred substrates arginine and arginine analogs such as agmatine accelerate the rate of the NAD hydrolysis linked to the ADP–ribose transfer.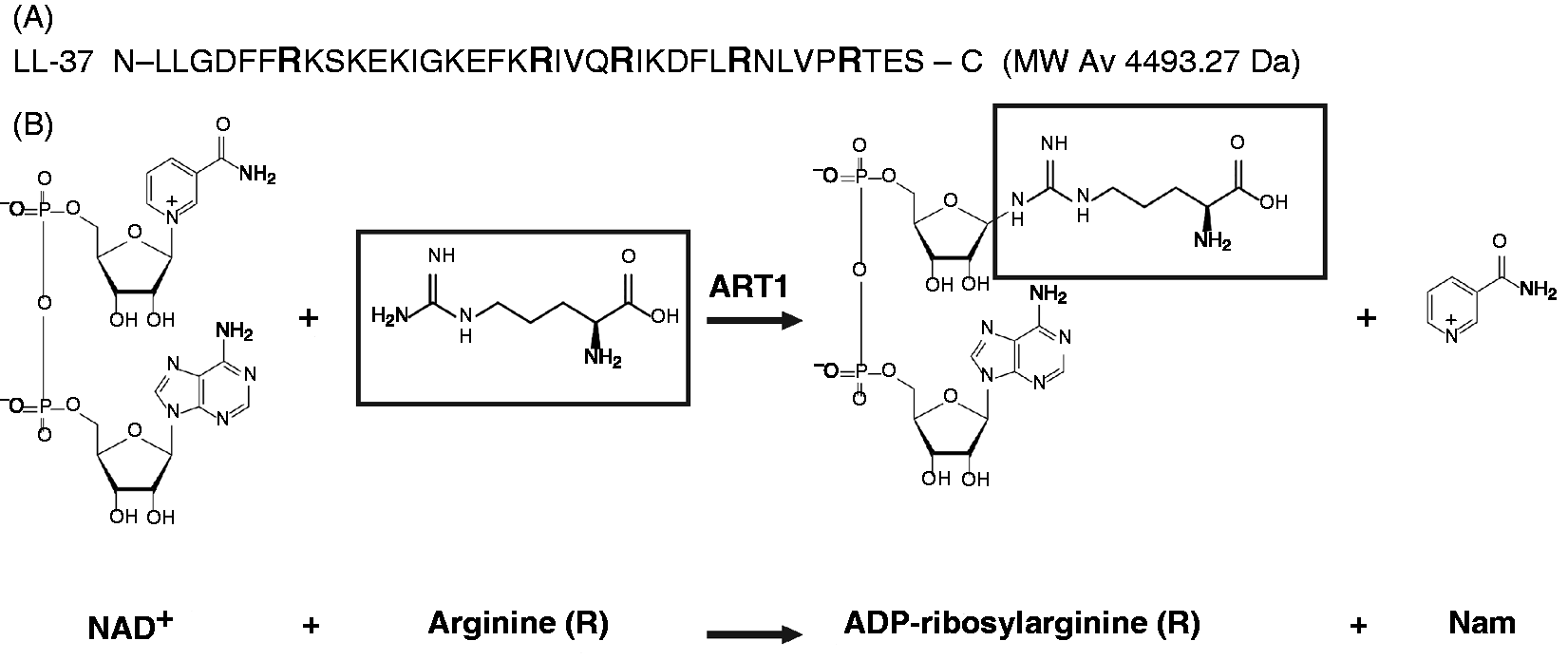
ADP-ribosyltransferases (ARTs) are a family of prokaryotic and eukaryotic enzymes that split the
In agreement with the amino acid and substrate selectivity of ARTs, HNP-1 is modified at R14 only by ART1 and by some bacterial ADP-ribosylating toxins.28,29
The presence of five arginine residues in LL-37 prompted us to evaluate whether this peptide could be recognized as substrate by ART1.
In this study, we show that LL-37 is mono-ADP-ribosylated at arginine residues in a NAD concentration-dependent fashion by human ART1
Material and methods
Reagents
The synthetic peptide LL-37 and mutants were synthesized at the Protein and Peptide Chemistry Facility of the University of Lausanne, Switzerland. [
Preparation of artificial sputum medium
The artificial sputum medium (ASM) was prepared by adding 4 g DNA from fish sperm to 250 ml sterile water very slowly over a period of at least 6 h.
The mucin from porcine stomach (type II, 5 g) was slowly added to 250 ml sterile water. To dissolve completely the mucin, the solution was stirred at 4℃ for 18 h.
We then dissolved 0.25 g of each essential and nonessential
We adjusted the pH to 6.9 with 1 M Tris (pH 8.5) and brought the volume to 1 l with sterile water. The ASM was sterilized by filtration using a Millipore Steritop filter unit with a pore and neck size of 0.22 µm. Filtered ASM is stored at 4℃ in the dark for a maximum of 1 mo.
Preparation of ART1
Human ART1 was synthesized as a GPI-linked protein on the surface of V79 cells after induction for 48 h with 2 µg/ml final concentration of doxycicline. The soluble form of ART1 was collected following treatment of intact V79 cells with 1 U/ml phosphatidylinositol-specific phospholipase C for 1 h at 37℃.
30
ADP-ribosyltransferase activity of ART1 was tested by monitoring the enzymatic transfer of ADP–ribose to poly-arginine with a filter plate-based assay.
31
Briefly, assays were performed in a total volume of 0.3 ml with 0.6 mg poly-arginine and 0.1 mM [
Detection of biotin ADP-ribosylated LL-37 by immunoblot analysis
ADP-ribosylation of LL-37 was obtained by incubating LL-37 (3 µg) with ART1 (5.5 nmol/h) in 50 mM potassium phosphate buffer (pH 7.4) in the presence of biotinylated NAD (biotin-NAD; 10 µM) in a final volume of 20 µl at 30℃ for 1 h.
To confirm that the reaction was enzymatic, either 2 mM NAD or 2 mM ADP–ribose were added to the assay mixture. As negative controls, ART1 was either omitted from the reaction mixture or heat-inactivated. The ADP-ribosylated LL-37 was resolved by SDS-PAGE in a 10% NuPAGE gel using 2-(N-morpholino)ethanesulfonic acid (MES) as running buffer and transferred to nitrocellulose using a dry system (I-Blot; Invitrogen). The membrane was blocked for 1 h with 5% BSA in PBS containing 0.05% Tween-20 (PBS-T), extensively washed and then incubated in the same buffer containing streptavidin–HRP conjugate (1:10,000 dilution) and mixed for 1 h on an orbital shaker at 22–25℃ (room temperature). After several washings with PBS-T, bound streptavidin was detected using the ECL immunoblotting detection system (Bio-Rad) according to the manufacturer’s instructions. Molecular masses were estimated from the calibration standard included in each gel.
Detection of ADP-ribosylated LL-37 by Coomassie blue staining
ADP-ribosylated LL-37 was also detected using NAD instead of biotin-NAD in the conditions described above, except that the reactions were incubated for 18 h. An aliquot of each assay was run on a 16% tricine gel using tricine running buffer, followed by staining of the gel with Coomassie blue. Molecular masses were estimated referring to the calibration standard included in each gel.
NAD–glycohydrolase assay
NAD–glycohydrolase (NADase) activity was monitored using a radioactive assay based on the release of nicotinamide (NAM) as previously described.
32
Briefly, the assay was carried out by incubating 0.1 mM [
MS analysis of ADP-ribosylated LL-37
Peptide molecular masses were determined using a MALDI-TOF/TOF mass spectrometer UltraFlex (Bruker Daltonics, Bremen, Germany). Ions generated by laser desorption at 337 nm (N2 laser) were recorded at an acceleration voltage of 20 kV in linear mode. In general, about 200 single spectra were accumulated to improve the signal/noise ratio and analyzed by FlexAnalysis version 2.4 (Bruker Daltonics). Briefly, 1 µl of reaction solution (20–60 pmol) was added to 1 µl of a saturated solution of sinapinic acid (3,5-dimethoxy-4-hydroxy-trans-cinnamic acid) in 30% (vol/vol) acetonitrile, 0.1% (vol/vol) trifluoroacetic acid. Then, 2 µl of peptide/matrix mixture was spotted on a stainless steel sample target and air-dried at room temperature. Peptide mass spectra were calibrated using external peptide calibration standard (Bruker Daltonics).
Protein assay
The protein content was determined with the Bradford protein assay kit (Bio-Rad) using BSA for standardization.
Results
LL-37 is ADP-ribosylated by ART1
The mature form of LL-37 contains five arginine residues, which are potential acceptors of ADP–ribose (Figure 1A). Therefore, we wondered whether LL-37 could be recognized as a substrate by ART1. As shown in Figure 2, ADP-ribosylated LL-37 appeared upon incubation with ART1 but not when ART1 was omitted or heat-inactivated. The antibiotic novobiocin, which is known to inhibit ART-catalyzed reactions,
33
strongly decreased LL-37 ADP-ribosylation. As shown in Figure 1A, LL-37 contains six lysine residues (K) that could non-enzymatically react with free ADP-ribose.34,35 However, the addition of a 200-fold excess of ADP-ribose to the reaction had only a minor effect on the incorporation of biotin-NAD, while the addition of a 200-fold excess of NAD completely abolished the incorporation of biotin-NAD (Figure 2), strengthening the conclusion that the observed modification of LL-37 was catalyzed by ART1. To mimick more closely the environment of the airways during inflammation, we performed the ADP-ribosylation reaction in ASM. As shown in Figure 2B, ART1 ADP-ribosylated LL-37 in ASM.
Enrico Balducci – 05/lug/14 09:48. LL-37 is ADP-ribosylated by ART1 (A). The activity of ART1, released by V79 transfected cells as described in Material and Methods, was tested by measuring the transfer of ADP-ribose to poly-arginine using a standard assay as described.31 LL-37 (3 µg) was incubated with ART1 (5.5 nmol/h) in the presence of 10 µM biotin-NAD in 50 mM potassium phosphate buffer, pH 7.5, at 30℃ for 1 h (ART1). The same reaction was performed in the presence of 2 mM NAD (ART1 + NAD), or 2 mM ADP-ribose (ART1 + ADP- ribose). Control reactions were carried out in the conditions above described with heat-inactivated enzyme (HI-ART1), in the absence of ART1 (-ART1), or in the presence of 2 mM Novobiocin (Novobiocin). (B). The ADP-ribosylation of LL-37 (3 µg) was also performed with ART1 (11.5 nmol/h) in the presence of 10 µM biotin-NAD with ASM medium replacing potassium phosphate buffer, in the identical conditions above described. A control reaction with heat-inactivated ART1 was also run (HI-ART1). The ADP-ribosylated peptides were resolved by SDS-PAGE in a 10% NuPAGE gel, using MES as running buffer and transferred onto a nitrocellulose membrane. After blocking with 5% BSA in PBS-T for 1 h, the blot was incubated with streptavidin-HRP conjugated (1:10000 dilution) for 1 h at RT in the same buffer. The biotin-ADP-ribose labeled bands were visualized by chemiluminescence. The blot was subsequently stripped and incubated with mouse anti-LL-37 antibody to check for equal loading of peptide. Open arrows indicate LL-37 (theoretical MW 4493.27 Da) while close arrows indicate ADP-ribosylated LL-37. Results are representative of three independent experiments.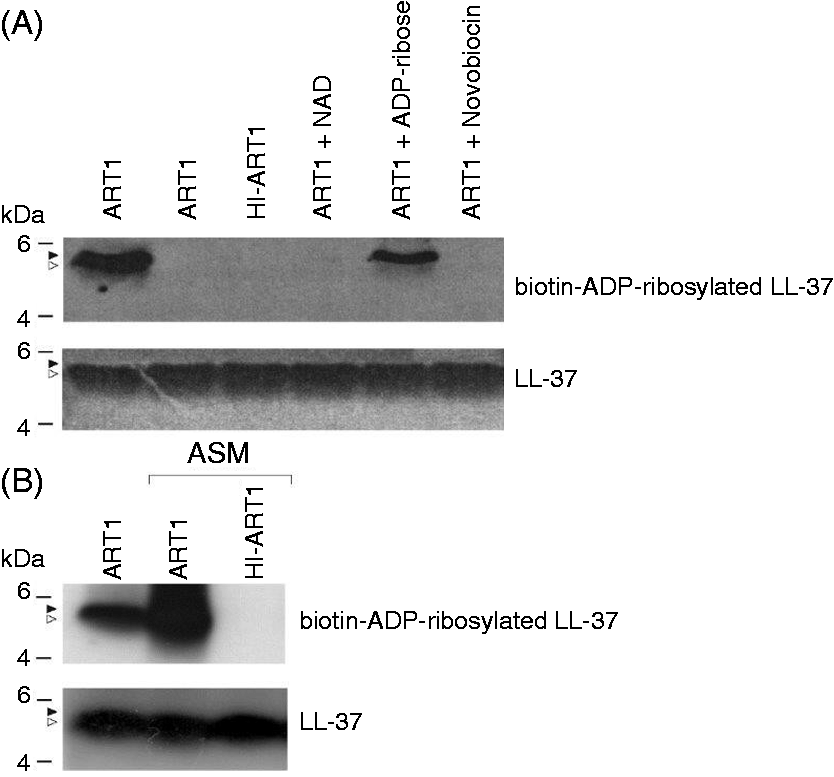
LL-37 is ADP-ribosylated at multiple R residues
As LL-37 contains five arginine residues as potential target for ADP-ribosylation, it could be modified at multiple sites. To verify whether one or more modifications occur, we incubated LL-37 with ART1 in the presence of increasing concentrations of NAD (Figure 3A). A band migrating slower than LL-37, likely corresponding to ADP-ribosylated form(s) of LL-37, appeared in the tricine gel, which improves the resolution of the bands, at NAD concentrations ranging from 1 to 25 mM but not at 0.1 mM. The apparent discrepancy with the experiment shown in Figure 2, in which we detected ADP-ribosylated LL-37 using 0.01 mM biotin-NAD, depends on the higher sensitivity of chemiluminescence versus Coomassie blue staining.
(A) NAD-dependent ADP-ribosylated of LL-37 by ART1. LL-37 (15 µg) was incubated with or without (last lane) ART1 (5.5 nmol/h) and NAD at different concentrations in 50 mM potassium phosphate buffer pH 7.4 at 30℃ overnight. An aliquot of each reaction was run on a 16% Tricine gel and stained with Coomassie blue. The appearance of a band with a molecular mass slightly higher than that of LL-37 at higher concentrations of NAD corresponded to the ADP-ribosylated forms of LL-37. (B) MS analysis shows that LL-37 is ADP-ribosylated at multiple sites by ART1. MS analysis of the above reaction run in the presence of ART1 and 5 mM NAD confirmed the mono-ADP-ribosylation at multiple arginine sites by ART1 (lower panel). A mass control reaction, i.e. LL-37 incubated at 30℃ for 18 h without ART1, is shown in the upper panel. Asterisks correspond to Na+ adduct (+22 Da), while triangles correspond to K+ adduct (+38 Da).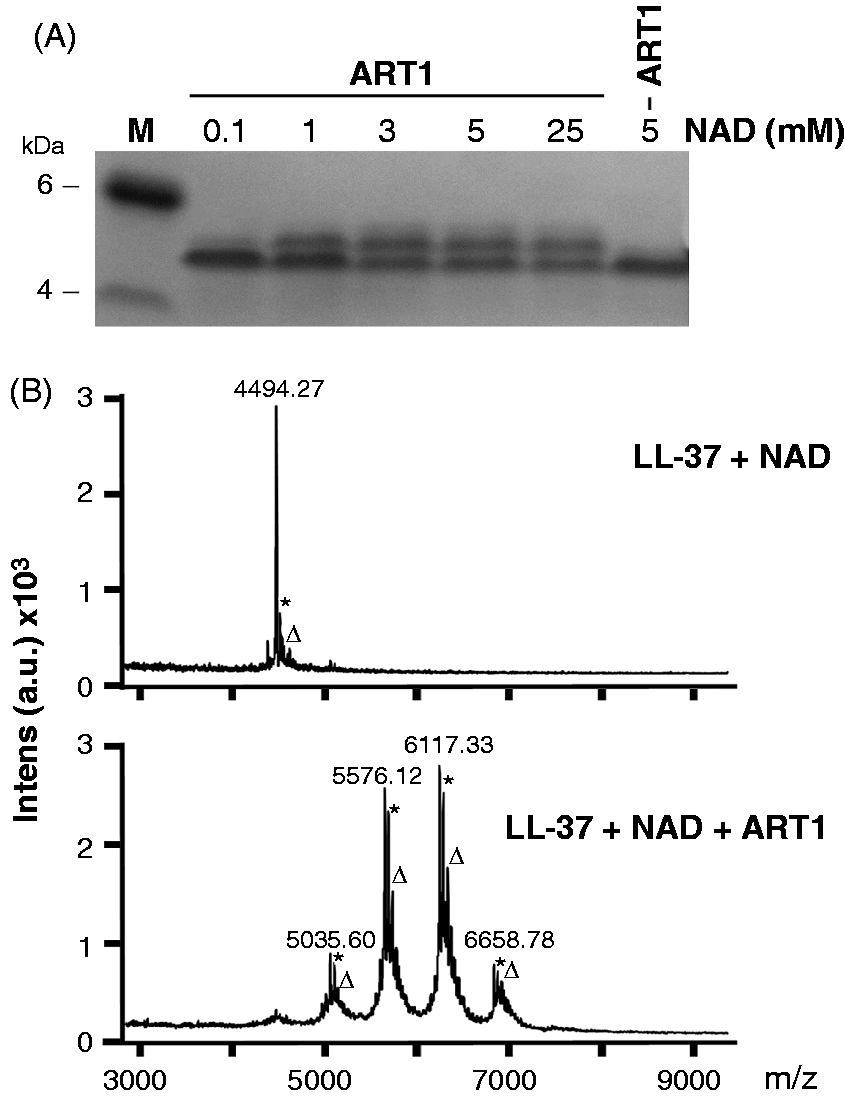
MS analysis of the reaction run with 5 mM NAD without ART1 showed a molecular mass of 4494.27 Da, which is in agreement with that of unmodified LL-37 (4493.27 Da), while analysis of the reaction run with 5 mM NAD with ART1 showed four additional peaks (Figure 3B). These peaks were in agreement with modification of LL-37 owing to the addition of 1, 2, 3 or 4 ADP–ribose moieties (+541 Da each). MS analysis of the reaction with 25 mM NAD showed the same peaks (data not shown).
Identification of the R residues ADP-ribosylated by ART1
MS analysis suggested that only four out of the five arginine residues of the LL-37 molecule were ADP-ribosylated at the same time. To identify which of the arginine residues was not ADP ribosylated by ART1, we used a panel of LL-37 mutants in which four R were conservatively replaced by K so that only one R was left in each mutant (Figure 4B). As shown in Figure 4C, each mutated form of LL-37 was ADP ribosylated upon incubation with ART1 and biotin-NAD with the exception of the R19 mutant that contains only the arginine at position 19, which was not modified. The analysis of the 3D model of LL-37 indicated that R19, differently from the other arginine residues, is poorly exposed to the solvent on the surface of LL-37 (Figure 4A). This is probably the reason why R19 is not accessible to ART1.
(A) LL-37 is not ADP-ribosylated at R19. Solvent accessibility of the arginine residues of LL-37. (B) Sequences of the LL-37 mutant peptides in which four R out of the five Rs present in LL-37, R7, R19, R23, R124 and R34, were conservatively replaced by K. (C) R19 is the residue that is not ADP-ribosylated by ART1. LL-37 mutant peptides (0.5 µg) were incubated at 30℃ for 1 and 3 h in 50 mM potassium phosphate buffer (pH 7.5) with 10 μM biotin-NAD and in the presence of ART1 (4.8 nmol/h, upper panel). Control blot incubated with α-LL-37 polyclonal serum (1:10,000 dilution) is also shown (lower panel).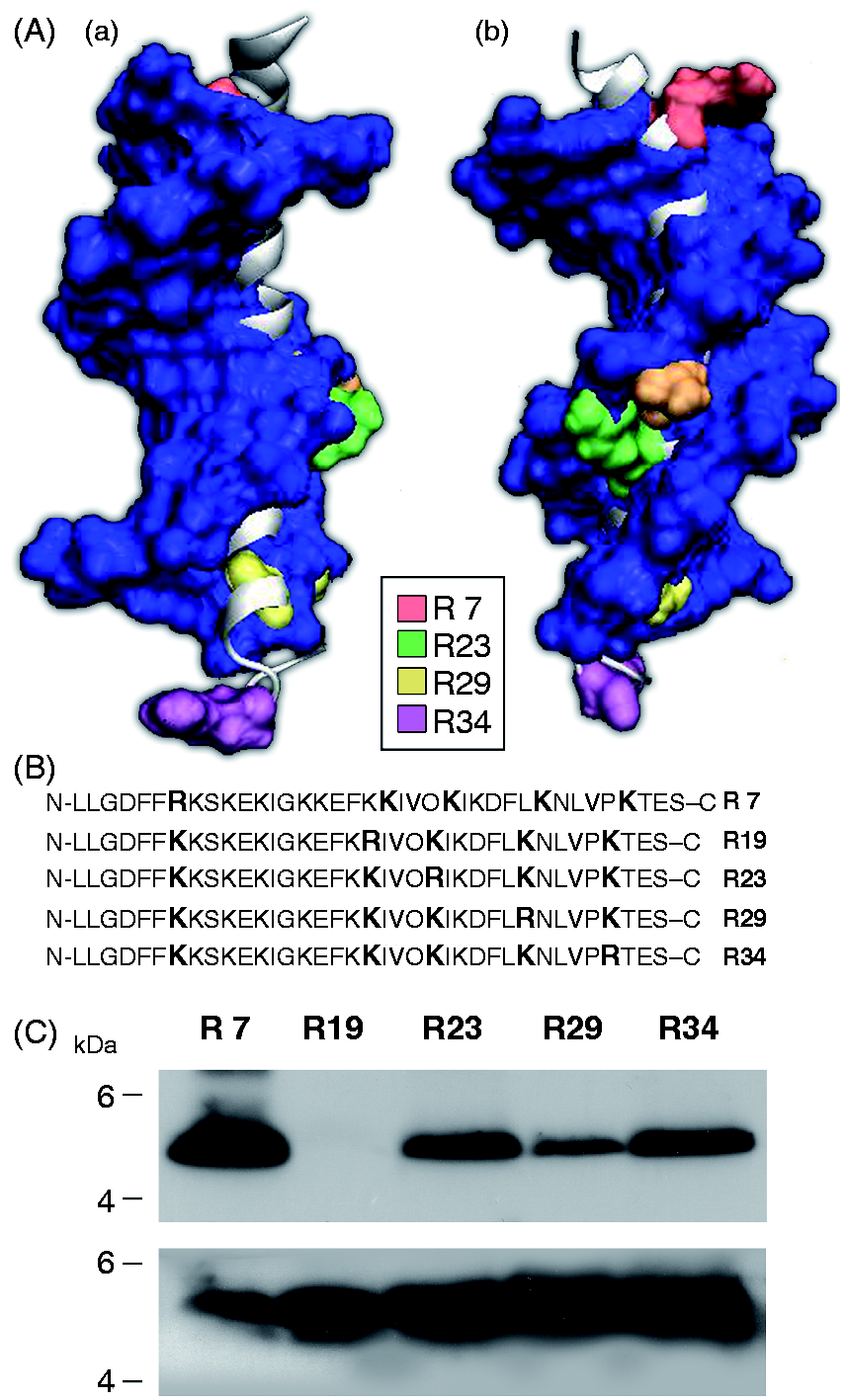
LL-37 does not modulate ART1 transferase activity
HNP-1 not only is a substrate of ART1, but also modulates ART1 enzymatic activity.
28
To assess whether LL-37 could also have such a regulatory role on ART1, we used a NAM release-based assay. In the NAD-glycohydrolase catalyzed reaction, water serves as a nucleophilic ADP–ribose acceptor, and nucleophilic substrates, including guanidine compounds, accelerate NAM release coupled to the transfer of ADP–ribose. ART1 cannot hydrolyze NAD; thus, with this assay we measure only transferase activity. As shown in Figure 5, LL-37 did not affect the release of NAM.
NAM release from NAD catalyzed by ART1 is enhanced by the presence of LL-37. ART1 (5.5 nmol/h) was incubated with 0.1 mM [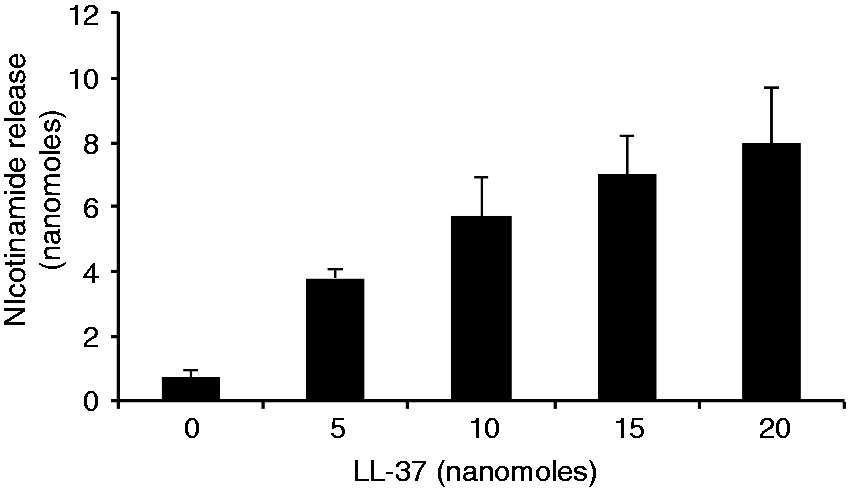
Discussion
ART1 is a surface-exposed enzyme expressed on the apical surface of human airway epithelial cells, 26 which catalyzes the transfer of the ADP–ribose moiety from NAD to arginine residue(s) on target proteins. LL-37 is a host defense peptide, usually released at a high concentration in the airway milieu during inflammatory diseases,36,37 which, owing to its high content of arginine residues, could be a substrate for ART1.
In this report, we present evidence that LL-37 can, indeed, be ADP-ribosylated by ART1 at multiple arginine residues
LL-37 can assume a random coil monomeric conformation at a low concentration, 40 while at a high concentration, LL-37 assumes an oligomeric structure. 40 Oligomeric conformation can expose selected arginines on the surface, which can be ADP-ribosylated by ART1. Although discrepancies exist among the studies that define the parts responsible for the biological activities of LL-37, it is clear that the subtle interplay of structural and physicochemical parameters such as cationicity, hydrophobicity and amphipaticity contributes to the antibacterial and cytotoxic activities of LL-37.41,42 The attachment of one or more ADP–ribose groups (approximately 540 Da), which is five times bigger than an amino-acid (110 Da), causes a notable change in the size of LL-37, the total molecular mass of which is only 4493.27 Da. This modification in size could change the structure of LL-37 and thus interfere with biological activities that imply interaction with macromolecules or receptors. Moreover, the overall cationic nature of LL-37 and the presence of particular amino acids constitute the chemical basis of the ability of LL-37 to interact with polyanionic molecules. 43 Therefore, the attachment of one or more negatively charged ADP–ribose moieties on arginine residues would result in a reduced cationicity. Consequently, ADP-ribosylated-LL-37 has a diminished ability to interact with biological membranes or polyanionic molecules such as DNA, RNA, actin or glycosaminoglycans.16–18
ADP-ribosylation of LL-37 could represent a novel mechanism that can regulate the formation of the complexes between LL-37 and negatively charged molecules, and thus affect the LL-37 immunomodulatory activities. Recent discoveries have provided accessible and easy methods to synthesize peptides containing ADP-ribsoylated amino acids.44–46 Therefore, it is conceivable that in diseases such as cystic fibrosis and psoriasis, selected ADP-ribosylated fragments of LL-37 could be used to develop new therapeutic compounds.
Footnotes
Funding
We thank Novartis Vaccines and Diagnostics for financially supporting this research project, and the Province of Siena and the Monte dei Paschi di Siena Foundation, which granted the fellowship of Marta Castagnini.
Acknowledgements
Chinese hamster fibroblast V79 cells transfected with pTet-ON-ART1 cDNA were kindly provided by Dr. J Mac Dermot. We are grateful to Giorgio Corsi for his precious artwork, to Simone Pasquini for preparing the artificial sputum medium, to Eleonora Talluri for helpful discussion valuable advice and assistance, to Alessio Bonucci for the modeling of LL-37 and to Nathalie Norais for the MS analysis.
