Abstract
In this study, the anti-inflammatory effects and mechanisms of baicalin on LPS-induced NLRP3 inflammatory pathway were investigated in piglet mononuclear phagocytes (control, LPS stimulation, LPS stimulation + 12.5 µg/ml baicalin, LPS stimulation + 25 µg/ml baicalin, LPS stimulation + 50 µg/ml baicalin and LPS stimulation + 100 µg/ml baicalin). The levels of reactive oxygen species (ROS), the secretion levels of IL-1β, IL-18 and TNF-α, mRNA expression levels of IL-1β, IL-18, TNF-α and NLRP3, as well as the protein levels of cleaved caspase-1 p20 were significantly increased after LPS-challenge in vitro. However, LPS stimulation did not influence apoptosis-associated speck-like protein and caspase-1 mRNA levels, which are also components of the NLRP3 inflammasome. Baicalin at 50 µg/ml and 100 µg/ml could inhibit the production of ROS, TNF-α, IL-1β and IL-18, and down-regulate mRNA expression of IL-1β, IL-18, TNF-α and NLRP3, as well as expression of cleaved caspase-1 p20. These results showed that the anti-inflammatory effects of baicalin occurred via the regulation of the release of ROS and mRNA expression of NLRP3. The anti-inflammatory activity of baicalin could be related to the suppression of NLRP3 inflammasome pathway under LPS stimulation.
Keywords
Introduction
LPS of Gram-negative bacteria is, depending on its structure, known as one of the most representative immune stress inducers that can activate the expression of inflammatory mediators/cytokines, such as IL-1β, IL-6 and TNF-α,1–4 as well as of cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), and the synthesis of prostaglandin E2 (PGE2) and NO, all of which participate in the body's inflammatory response process. 5 Persistent inflammatory reaction caused by these overwhelming productions of pro-inflammatory cytokines may lead to multiple organ injury in, e.g., lung and liver, and to shock and even death.6,7 LPS stimulation is mediated by TLR4/MD2 activating the signaling pathway of NF-κB in monocytes/macrophages. 5 In addition, cross talk between TLRs and NLRs in the secretion of mature IL-1β and IL-18 during microbial infection had been reported. 8
The inflammasome plays a critical role in the inflammatory reaction as an important innate immune pathway that exists mainly in monocyte/macrophage and epithelial cells, and is activated by a wide variety of particles, crystals, bacterial toxins, viruses, bacteria and fungi.4,9 It was reported that the caspase-1-dependent antimicrobial response was promoted by microbial infection triggering assembly of inflammasome complexes. 10 Moreover, the nucleotide-binding domain leucine-rich repeat proteins NLRP1, NLRP3 and NLRC4, and the pyrin and HIN200 domain-containing protein AIM2 have been confirmed to initiate the formation of inflammasome complexes. 11 Among them, the NLRP3 inflammasome has attracted the most interest because it is associated with a wide range of diseases, including cryopyrin-associated periodic syndromes, gout, type 2 diabetes, Crohn’s disease and atherosclerosis. 12 The NLRP3 inflammasome is activated in response to various signals, including PAMPs and danger-associated molecular patterns (DAMPs), and consists of NLRP3, apoptosis-associated speck-like protein (ASC) and pro-caspase-1, which influence the regulation of pro-inflammatory cytokine secretion, particularly the generation of IL-1β and IL-18. 13 The NLRP3 inflammasome is tightly controlled by a priming step that is dependent on NF-κB and highly inducible in response to pro-inflammatory stimuli such as LPS. 11 Mitochondrial damage and reactive oxygen species (ROS) generation represent the main means to activate it.14,15
Baicalin (BA), obtained from the Scutellaria baicalensis Georgi as a flavonoid, has significant anti-inflammatory, antibacterial and free radical scavenging properties. 16 BA can inhibit macrophage activation by LPS and protect mice from endotoxin shock, 17 reduce the release of high-mobility group box 1 from macrophages induced by LPS and improve survival in experimental mice sepsis. 18 It can also improve survival in a murine model of polymicrobial sepsis via suppression of the inflammatory response and lymphocyte apoptosis. 6 The anti-inflammatory functions of BA have been proposed as inhibition of the LPS-stimulated PGE2 production, 19 hyperpermeability, expression of cell adhesion molecules (CAMs) and adhesion/transendothelial migration of leukocytes. 20 It plays an anti-inflammatory role through reducing NF-κB and p38 phosphorylation in Staphylococcus aureus-induced mastitis, 21 and the apoptosis by regulating TLR2 and TLR2-related apoptotic factors in mouse mammary glands. 22 Currently, it is thought that the anti-inflammatory response of BA may be involved in the blocking of the TLR4/NF-κB signaling pathway.23–26
Our previous findings suggested that the TLR-NF-κB and NLRP3-caspase-1 pathway could be activated by LPS in piglet mononuclear phagocytes.3,8 However, the effects of BA on LPS-mediated activation of the NLRP3 inflammasome have not been determined. In an effort to elucidate both the effect and mechanism responsible for the anti-inflammatory action of BA, we explored the anti-inflammatory activity of BA in LPS-treated piglet mononuclear phagocytes by investigating the secretion of IL-1β, IL-18 and TNF-α, as well as on the protein level of cleaved caspase-1 p20. In addition, the production of cell ROS, as well as of the mRNA expression of IL-1β, IL-18 and NLRP3 inflammasome complex (NLRP3, ASC and caspase-1), were analyzed to evaluate the mechanism of BA suppression of NLRP3 inflammasome signaling.
Materials and methods
Isolation and culture of peripheral blood monocytes
Three crossbred healthy piglets (Duroc × Landrace × large white) obtained from Wuhan Cofco Meat Product Co., Ltd (Wuhan, China), weighing 7-10 kg and aged 5 wk were used for in vitro experiments. The piglets had not been given any drugs before. The animal use protocol for this research was approved by the Animal Care and Use Committee of Hubei Province.
The blood used for monocyte isolation was collected by the precaval vein from the piglets. Heparinized blood was layered carefully on an equal volume of PBS (pH 7.4) in a conical centrifuge tube, mixed upside down and then an equal volume of mixed blood was carefully layered on the surface of lymphocyte separation medium. The suspension was centrifuged at 358 g for 20 min at about 20℃. The cells of the lymphocyte layer were collected and washed three times with PBS by centrifugation at 201 g for 15 min at 4℃. Cells were re-suspended in 5 ml RPMI-1640 (Gibco, Waltham, MA, USA) and seeded in a six-well cell culture plate (Costar; Corning, NY, USA). Suspension (3 ml) was added to each well, and these were pre-incubated in a constant temperature incubator at 37℃ with 5% CO2 for 3 h in RPMI-1640 containing 10% FCS. Cells were washed twice with PBS and then washed with RPMI-1640 medium pre-warmed at 37℃ in order to discard the non-adherent cells. Attached cells (monocytes) were detached using a cell scraper and suspended in RPMI-1640 medium. Mononuclear cells were counted and their viability was determined by Trypan blue exclusion.
Experimental design
BA was obtained from National Institutes for Food and Drug Control (B110715-201318; Beijing, China). The drug was dissolved and diluted using RPMI-1640 medium without any other solvent.
Piglet monocytes were randomly divided into six groups: control treatment (normal cells with normal medium without adding LPS and BA); LPS (from Escherichia coli, serotype type O55:B5; Sigma, St. Louis, MO, USA) stimulation treatment (normal cells with normal medium adding LPS without drugs); LPS stimulation + 12.5 µg/ml BA treatment; LPS stimulation + 25 µg/ml BA treatment; LPS stimulation + 50 µg/ml BA treatment; LPS stimulation + 100 µg/ml BA treatment. Each treatment was repeated three times. After incubation with BA for 1 h, LPS was added (the final concentration was 1 µg/ml) for 3 h (ROS detection; mRNA analysis of IL-1β, IL-18, TNF-α, NLRP3, ASC and caspase-1; production analysis of TNF-α); 10 h (active caspase-1 p20 analysis) and 16 h, and 20 h continued incubation (production analysis of IL-1β and IL-18). Cytokines were measured by ELISA (in a 96-well plate at 5.0 × 105 cells/ml). The mRNA of IL-1β, IL-18, TNF-α, NLRP3, ASC and caspase-1 were measured by RT-PCR (in a 24-well plate at 3.0 × 106 cells/ml). Active caspase-1 was measured by Western blot analysis (in a six-well plate at 5.0 × 106 cells/ml). ROS was measured by fluorescence microscope (in a 24-well plate at 1.0 × 106 cells/ml).
Detection of ROS
ROS was observed by a fluorescence microscope. After 3 h of LPS stimulation and discarding the supernatant, the unstimulated and stimulated cells (1 ml, 1.0 × 106 cells/ml) were rinsed twice in ice-cold PBS, and 1.2 ml DCFH-DA was added (the final concentration was 10 µmol/l), and incubation at 37℃ with 5% CO2 was continued for 45 min. Then, samples were investigated by a fluorescence microscope.
Analysis of cytokines concentrations
Concentrations of IL-1β, IL-18 and TNF-α obtained from monocyte culture supernatants (in 96-well plate at 5.0 × 105 cells/ml) were measured using ELISA kits according to manufacturer’s instructions (Blue Gene, Shanghai, China). IL-1β (0–1000 pg/ml), IL-18 (0–1000 pg/ml) and TNF-α (0-1000 pg/ml) represented the standards for calibration, and the sensitivity of all assays was 1 pg/ml.
Total RNA extraction and RT-PCR
Sequence of primers used in current investigation by RT-PCR.
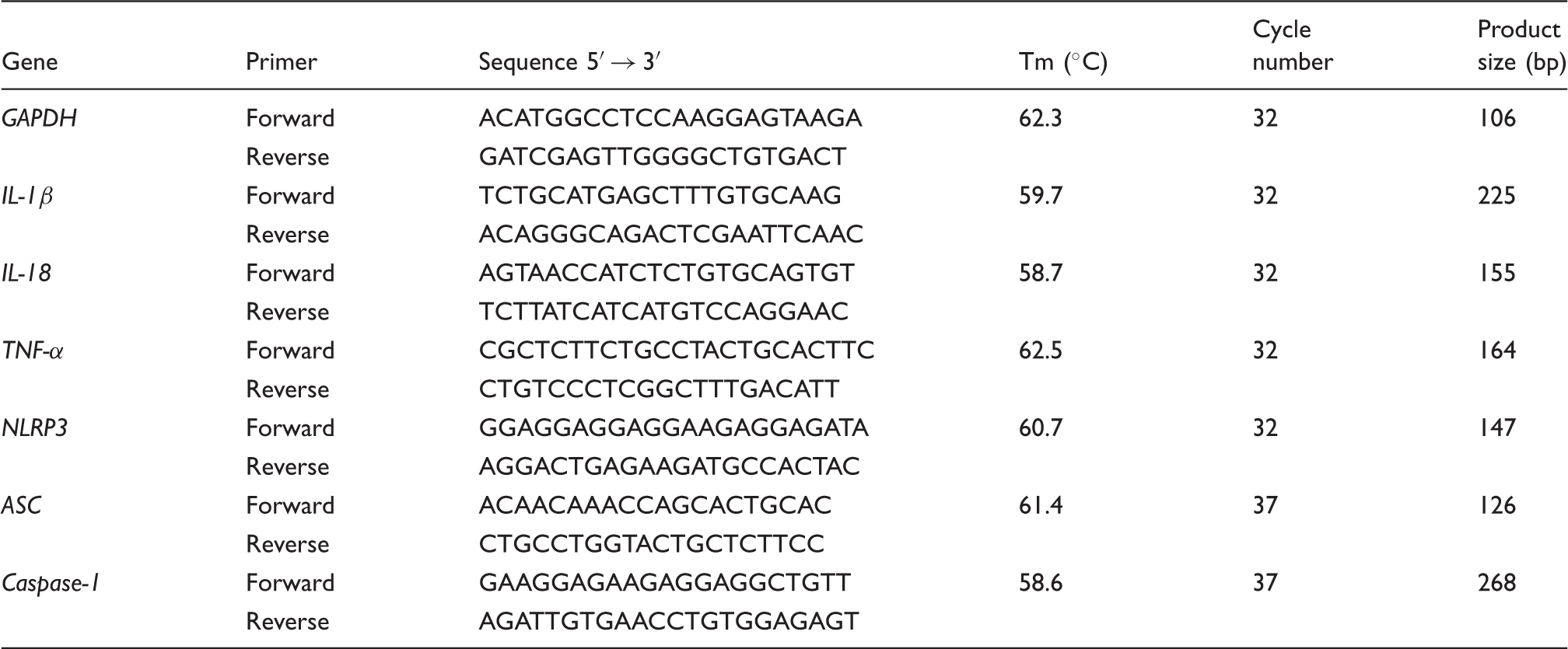
The PCR program was set as follows: 94℃ for 5 min followed by 32 cycles (37 cycles for ASC and caspase-1) of amplification at 94℃ for 30 s, melting temperature (Tm) for 32 s and 72℃ for 30 s, and then a final extension at 72℃ for 5 min. Finally, the results were analyzed by agarose gel electrophoresis and GIS image processing system (Tanon GIS 4100, Biotanon, Shanghai, China). Finally, the ratio of target gene and GAPDH Gray level was calculated.
Western blot analysis
Ten h after LPS induction, the unstimulated and stimulated cells (2 ml, 5.0 × 106 cells/ml) were rinsed twice with ice-cold PBS and the protein was extracted using total protein extraction kit following the manufacturer’s instructions (Beyotime, Shanghai, China). The protein concentration was determined with a BCA protein assay kit. The proteins were separated by 12% SDS-PAGE and transferred onto a PVDF membrane (60 mA, 1 h). The PVDF membrane was blocked with 5% skimmed milk at 20℃ for 3 h on a rotary shaker and followed by TBS-T washing, then incubated with cleaved caspases-1 p 20 Ab (sc-1218-R; Santa Cruz Biotechnology, Santa Cruz, CA, USA) or β-actin Ab (Cell Signaling Technology, Danvers, MA, USA) for 12 h at 4℃. Afterwards, the PVDF membrane was incubated with HRP-linked Ab at 20℃ for 3 h and visualized using Thermo pierce ECL (NCI5079; Thermo) solution. The level of cleaved caspase-1 p20 and β-actin was detected by ChemiDoc MP Imaging System (Bio-rad, Hercules, California, USA).
Statistical analysis
Results are expressed as mean ± SD. Data were analyzed using ANOVA followed by LSD method for independent means with SPSS Statistics 17.0 (IBM, Armonk, NY, USA) where appropriate, to determine which conditions were significantly different from each other. A probability value of P < 0.05 was considered significant.
Results
Effects of BA on ROS production in LPS-induced piglet monocytes
Three h after LPS-stimulation, the content of ROS in piglet monocytes significantly increased (P < 0.01), as observed from the pictures analyzed by Image-Pro Plus and by summed integrated optical density (Sum IOD) in the same area (Figure 1). BA in all concentration groups (12.5, 25, 50 and 100 µg/ml) could significantly (P < 0.01) inhibit the LPS stimulation and reduce production of ROS in cells.
Effects of BA on LPS-induced ROS production by piglet mononuclear phagocytes in vitro. Cells (1.0 × 106 cells/ml) were pretreated for 1 h with BA, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of ROS in the cells were measured by fluorescence microscopy. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; **P < 0.01 vs. LPS induction).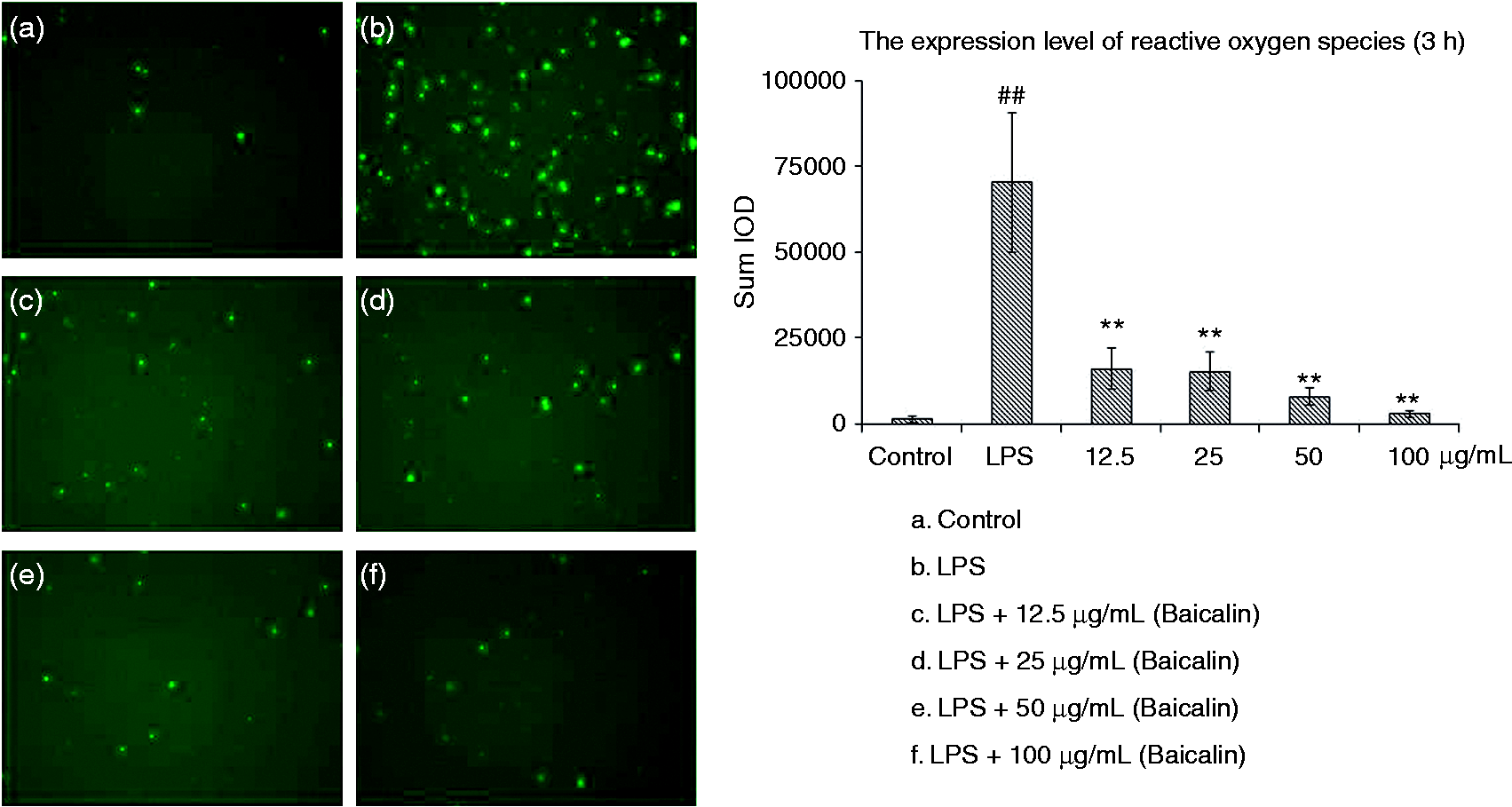
Effects of BA on IL-1β, IL-18 and TNF-α production in LPS-stimulated piglet monocytes
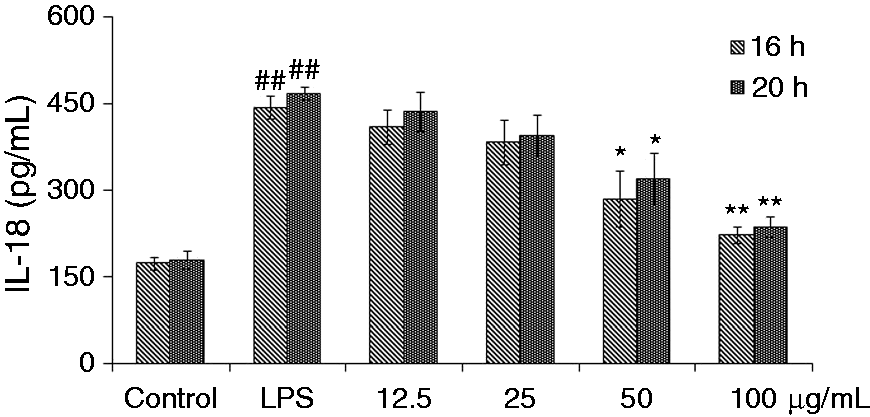
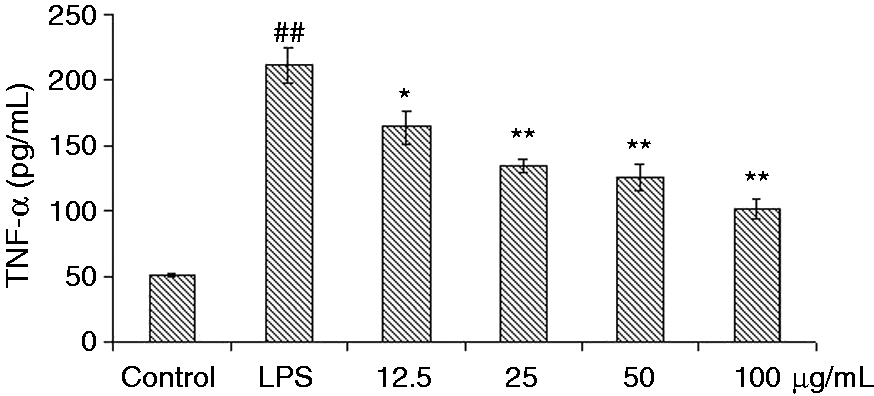
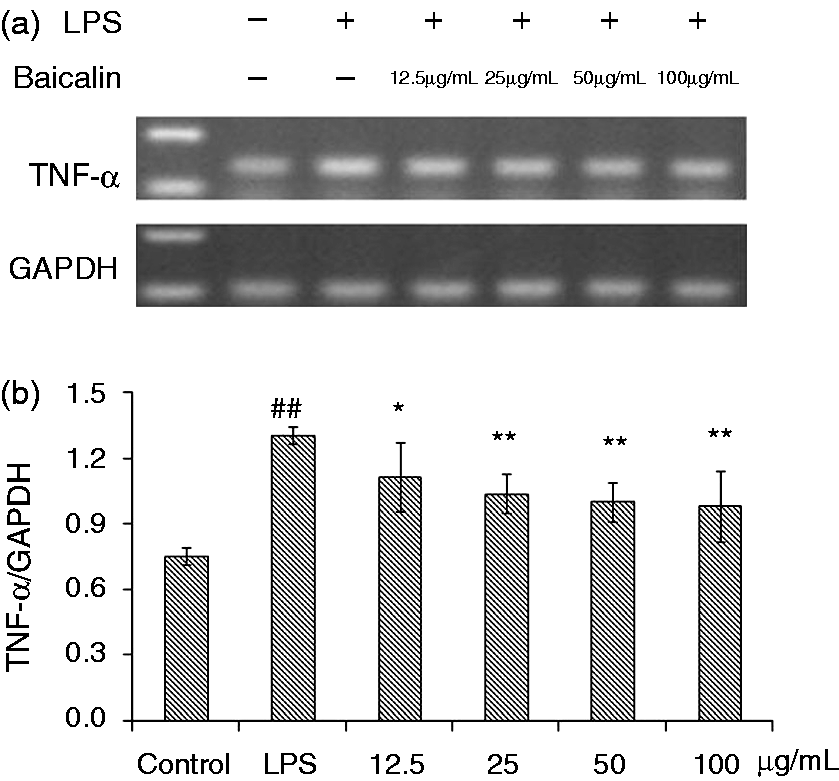
The effects of BA on IL-1β, IL-18 and TNF-α expression in LPS-simulated piglet monocytes were evaluated. LPS stimulation significantly enhanced the levels of IL-1β and IL-18 in the cells after 16 h and 20 h compared with the control. The IL-1β and IL-18 production induced by LPS could be suppressed by all concentrations of BA (Figures 2 and 3). Meanwhile, BA (50 and 100 µg/ml) could significantly (P < 0.05 and P < 0.01, respectively) inhibit the secretion of IL-1β and IL-18, while with concentrations of 12.5 and 25 µg/ml it suppressed the increase of IL-1β and IL-18 to a certain extent. Compared with the control group, LPS stimulation also significantly (P < 0.01) increased TNF-α levels in piglet monocytes. Treatment with all four concentrations of BA reduced TNF-α release in a dose-dependent manner (Figure 4). Effects of BA at 25, 50 and 100 µg/ml (P < 0.01) were stronger than that at 12.5 µg/ml (P < 0.05).
Effects of BA on LPS-induced IL-1β production by piglet mononuclear phagocytes in vitro. Cells (5.0 × 105 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 16 and 20 h. The levels of IL-1β in the supernatant were measured by ELISA. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; *P < 0.05 vs. LPS-induced; **P < 0.01 vs. LPS induction). Effects of BA on LPS-induced IL-18 production by piglet mononuclear phagocytes in vitro. Cells (5.0 × 105 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 16 and 20 h. The levels of IL-18 in the supernatant were measured by ELISA. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; *P < 0.05 vs. LPS-induced; **P < 0.01 vs. LPS induction). Effects of BA on LPS-induced TNF-α production by piglet mononuclear phagocytes in vitro. Cells (5.0 × 105 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of TNF-α in the supernatant were measured by ELISA. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; *P < 0.05 vs. LPS-induced; **P < 0.01 vs. LPS induction).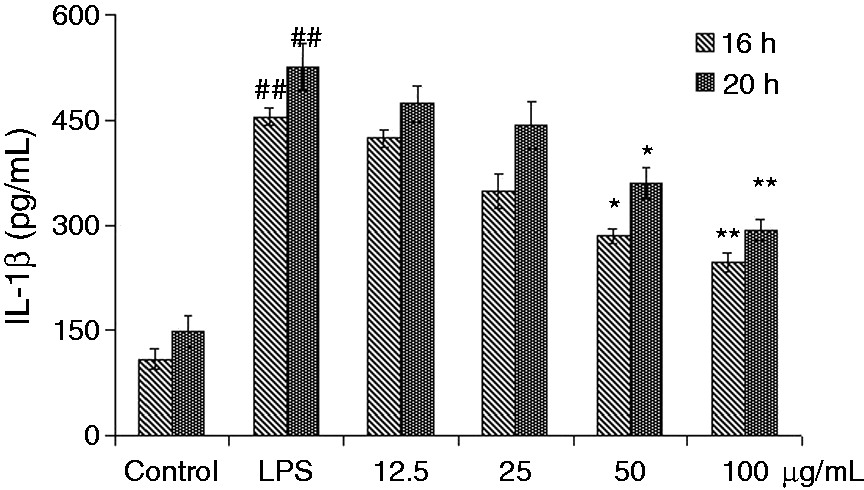
Effects of BA on the mRNA expressions of IL-1β, IL-18 and TNF-α genes in LPS-stimulated piglet monocytes
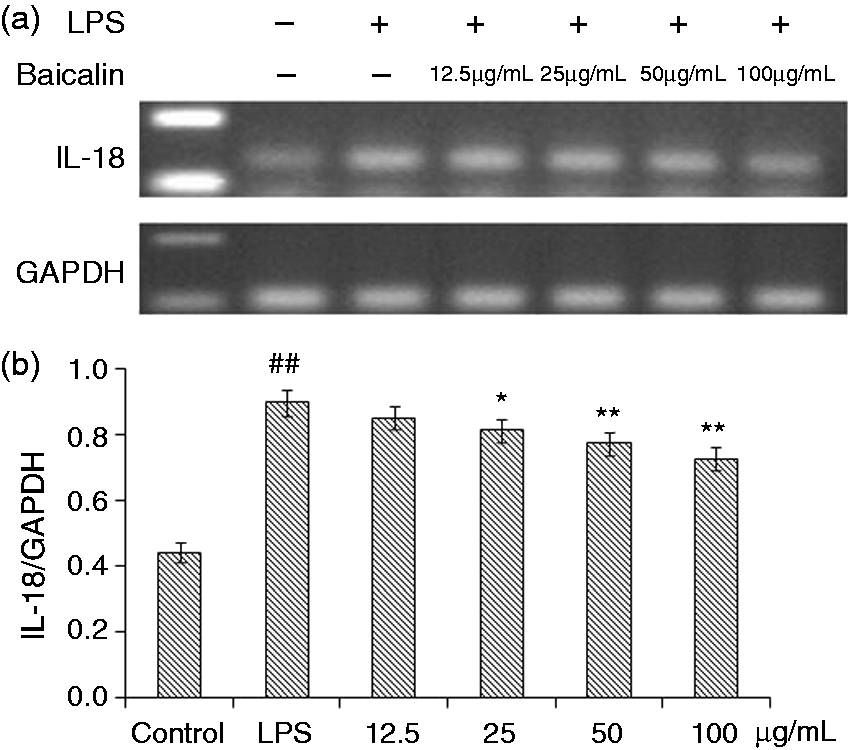
Data are shown in Figures 5–7. The mRNA expressions of IL-1β and IL-18 in the monocytes treated with LPS were significantly higher after 3 h (P < 0.01) compared with the control. The four BA treatment groups generally showed a tendency to decrease the mRNA expressions of IL-1β and IL-18; however, BA at 50 and 100 µg/ml could significantly reduce these mRNA expressions (IL-1β, P < 0.05; IL-18, P < 0.01). BA at 25 µg/ml decreased the level of IL-18 mRNA (P < 0.05). After LPS induction for 3 h, the mRNA expression of TNF-α was significantly (P < 0.01) increased (Figure 7). Compared with control group, treatment with BA (12.5, 25, 50 and 100 µg/ml) could significantly decrease this mRNA, with better effects of treatment with 25, 50 and 100 µg/m (P < 0.01) than that of 12.5 µg/ml (P < 0.05).
Effects of BA on LPS-induced mRNA expressions of IL-1β by piglet mononuclear phagocytes in vitro. (a) Cells (3 × 106 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of mRNA expression of IL-1β in the cells were measured by RT-PCR. (b) The relative level of IL-1β was measured using an image analyzer. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; *P < 0.05 vs. LPS-induction). Effects of BA on LPS-induced mRNA expression of IL-18 by piglet mononuclear phagocytes in vitro. (a) Cells (3 × 106 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of mRNA expression of IL-18 in the cells were measured by RT-PCR. (b) The relative level of IL-18 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; *P < 0.05 vs. LPS-induced; **P < 0.05 vs. LPS induction). Effects of BA on LPS-induced mRNA expression of TNF-α by piglet mononuclear phagocytes in vitro. (a) Cells (3 × 106 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of mRNA expression of TNF-α in the cells were measured by RT-PCR. (b) The relative level of TNF-α was measured using an image analyzer. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; *P < 0.05 vs. LPS-induced; **P < 0.05 vs. LPS induction).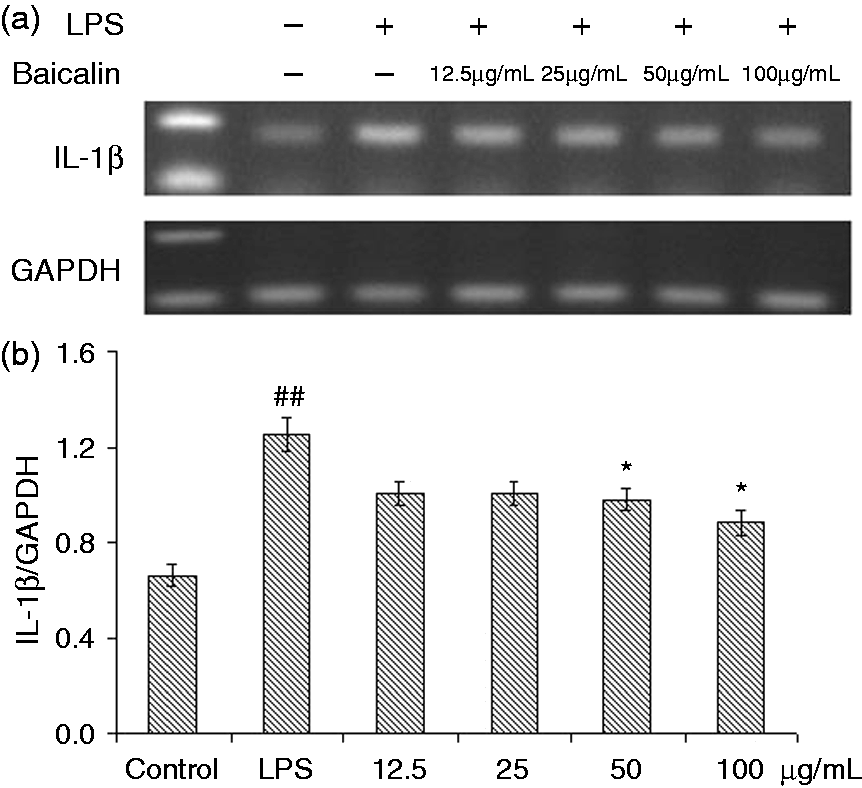
Effects of BA on the mRNA expression of NLRP3 inflammasome components in LPS-stimulated piglet monocytes
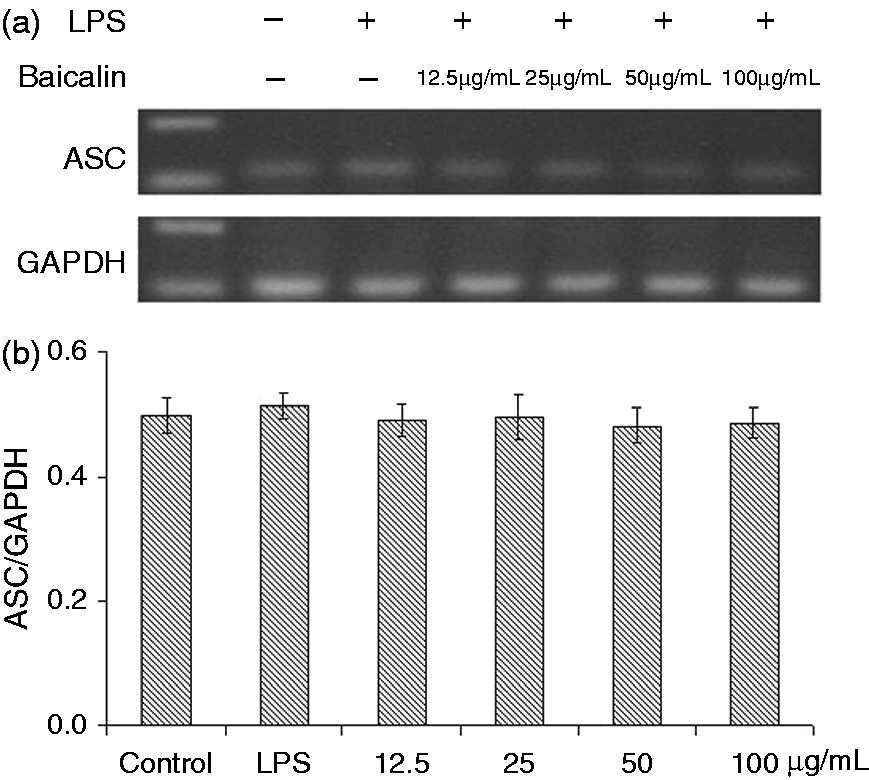
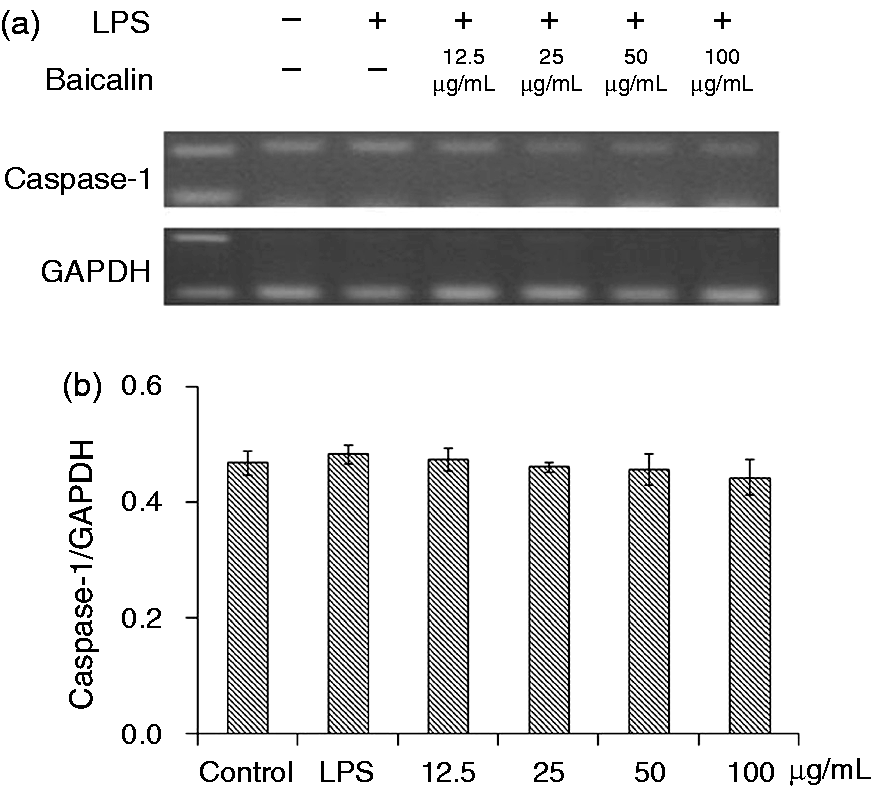
As shown in Figure 8, the mRNA expression of NLRP3 was significantly increased (P < 0.01) after LPS treatment for 3 h, while those of ASC and caspase-1 almost unified between the control and LPS groups (Figures 9 and 10). BA at 12.5, 25 and 50 µg/ml (P < 0.05) and 100 µg/ml (P < 0.01) could significantly prevent LPS induction and decreased the mRNA expression of NLRP3.
Effects of BA on LPS-induced mRNA expression of NLRP3 by piglet mononuclear phagocytes in vitro. (a) Cells (3 × 106 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of mRNA expression of NLRP3 in the cells were measured by RT-PCR. (b) The relative level of NLRP3 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; *P < 0.05 vs. LPS-induced; **P < 0.05 vs. LPS induction). Effects of BA on LPS-induced mRNA expression of ASC by piglet mononuclear phagocytes in vitro. (a) Cells (3 × 106 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of mRNA expression of ASC in the cells were measured by RT-PCR. (b) The relative level of ASC was measured using an image analyzer. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method. Effects of BA on LPS-induced mRNA expressions of caspase-1 by piglet mononuclear phagocytes in vitro. (a) Cells (3 × 106 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 3 h. The levels of mRNA expressions of caspase-1 in the cells were measured by RT-PCR. (b) The relative level of caspase-1 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method.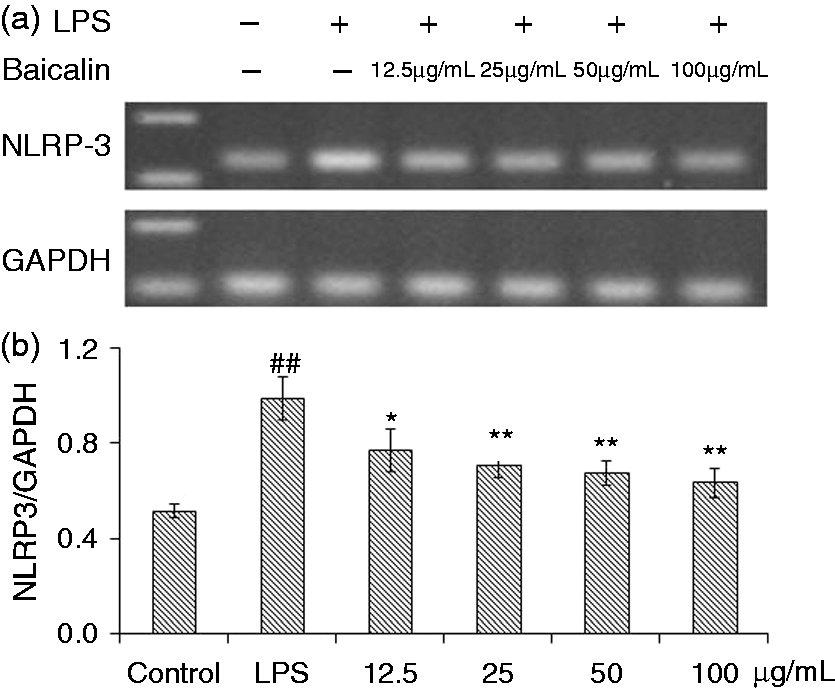
Effect of BA on cleaved caspase-1 p20 production in LPS-stimulated piglet monocytes
Protein expressions of cleaved caspase-1 p20 were significantly increased (P < 0.01) after 10 h of LPS induction (Figure 11). BA at 25, 50 and 100 µg/ml could significantly (P < 0.01) reduce the expression of cleaved caspase-1 p20.
Effects of BA on LPS-induced expression of cleaved caspase-1 p20 by piglet mononuclear phagocytes in vitro. (a) Cells (5 × 106 cells/ml) were pretreated with BA for 1 h, and then stimulated with LPS (1 µg/ml) for 10 h. The levels of cleaved caspase-1 p20 in the protein extraction supernatant were measured by Western blot analysis. (b) The relative level of active caspase-1 was measured using an image analyzer. The values presented are the means ± SD of three independent experiments and differences between mean values were assessed by LSD method (##P < 0.01 vs. control; **P < 0.05 vs. LPS induction).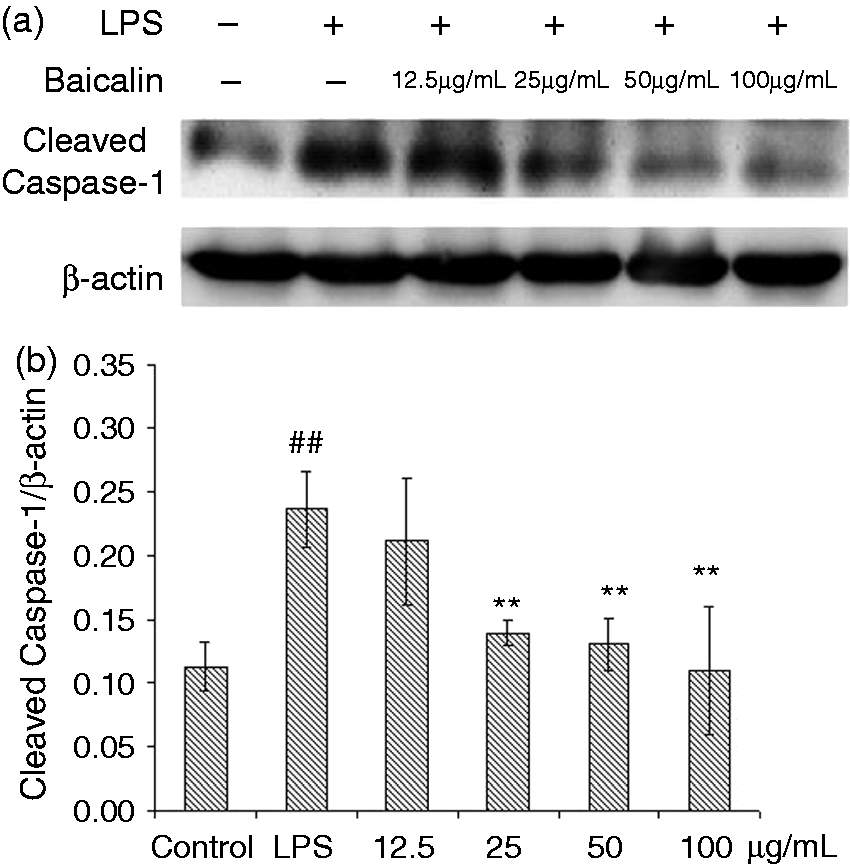
Discussion
Sepsis-related disorders are currently thought to be induced by a characteristic pathophysiology of connected overwhelming inflammatory reactions with the compensatory immune depression. TLRs and NLRs are innate immunity sensors that provide an early/effective response to pathogenic or injury conditions. 8 The inflammasomes are multiprotein complexes localized within the cytoplasm of the cell that are responsible for the maturation of pro-inflammatory cytokines. In pigs, it is not different from that in humans or mice. 27 The NLRP3 inflammasome is now recognized as a critical molecular mechanism for many degenerative diseases. 27 Several models have been developed to describe how NLRP3 inflammasomes are activated, including K+ efflux, lysosome function, ER stress, intracellular calcium, ubiquitination, microRNAs and, in particular, ROS. 15 The activation of the NLRP3 inflammasome complex can lead to the homo-type of the PYD-PYD regional interaction, then recruitment of ASC, through contact with molecules of the CARD-CARD area to mediate the maturation of inactive pro-caspase-1 into active caspase-1 (cleaved caspase-1), which is the central stimulator promoting the maturation of pro-IL-1β and pro-IL-18, respectively; finally, these cytokines cause inflammation.
LPS is the major component of most Gram-negative bacterial cell envelopes, representing, depending on its structure, the endotoxin that interacts with the membranes of macrophages, endothelial cells and neutrophils to induce the release of pro-inflammatory cytokines. 28 Flavonoids such as luteolin, glabridin and orientin have been reported to possess the ability of inhibiting LPS-triggered secretions in cells and to protect mice from septic shock.29–31 Also, BA has been studied in the treatment of sepsis-related diseases.17,18,20 It has been frequently used as an anti-inflammatory drug that can inhibit LPS-induced inflammation as an active ingredient through signaling the NF-κB pathway in epithelial, peripheral blood mononuclear and macrophage cells, as well as inhibiting hyperpermeability, expression of CAMs, and adhesion and migration of leukocytes.20,23–25 However, the anti-inflammatory effects of BA on inflammasome signaling pathway have not been fully elucidated yet. Therefore, this study provides evidence to understand the mechanisms of BA treatment through NLRP3 inflammasome pathway. According to our previous studies, the piglet mononuclear phagocyte inflammatory model could be successfully installed with 1 µg/ml LPS. 8
Mitochondrial damage and ROS generation were considered to induce NLRP3-dependent lysosomal damage and to represent the main origin of NLRP3 inflammasome activation.14,32–35 ER stress also primed cells for the expression of pro-IL-1β via NF-κB activation and promotes IL-1β secretion. Enhanced IL-1β secretion depended on the activation of the NLRP3 inflammasome through a mechanism involving ROS formation and activation of thioredoxin-interacting protein. 27 In addition, the role of ROS on NLRP3 inflammasome activation was at the level of the priming step, instead of affecting the regulation directly. 36 TNF-α can activate the downstream TLR–NF-κB signaling pathway, which played an important role in rapid response to inflammatory immune stress. 37 The activated NF-κB could induce the secretion of cytokines (e.g. TNF-α, IL-6, etc.), as well as the activation of the NLRP3 inflammasome. In this study, LPS stimulation could increase the production of ROS in piglet monocytes and the release of TNF-α in the supernatant, both rapidly and significantly, which indicated that NLRP3 activated by LPS might be related to the induction of the production of ROS and TNF-α. The increased content of TNF-α could result in mitochondrial damage, and release large numbers of ROS to induce oxidative stress. After BA treatment, the increased generation of ROS and TNF-α was dose dependent. Earlier, we found that N-acetylcysteine possessed anti-inflammatory effect via the TLR–NF-κB and NLRP3-caspase-1 pathways in LPS-challenged piglets as a scavenger of ROS. 8 Thus, the effectiveness of BA might be related to its ability as inhibitor of ROS production and inflammatory mediators through the direct free radical scavenging activity.2,16 A previous study also provided evidence that the anti-inflammatory activities of flavonoids may be related to their ability to inhibit LPS-induced increases in TNF-α levels. 20 In addition, the effects of BA treatment on ROS, TNF-α, NLRP3 inflammasome and inflammatory cytokines expression were different, and the inhibition on the production of ROS and expression of TNF-α was most effective.
In this study, it has been found that after 3 h of LPS stimulation, the mRNA expression of NLRP3 was significantly increased, while there were no significant differences for the ASC and caspase-1 genes, similar to the report by Bauernfeind et al. 38 ASC was redistributed to achieve the assembly of the NLRP3 inflammasome from the nucleus to the cytosol by the activation of PAMPs and DAMPs, but the precise mechanism of inflammasome regulation is not clear. 12 The results suggested that BA blocked NLRP3 inflammasome activation just by interfering with the priming step, which is required to induce NLRP3 expression, whereas direct NLRP3 activation is not affected. 38
Our present study confirmed that BA can work as anti-endotoxin agent (Figure 12). First, BA could inhibit the production of TNF-α to scavenge ROS, which could induce the mRNA expression of NLRP3. Second, it could reduce the production of inflammatory cytokines (IL-1β, IL-18, TNF-α, etc.) by inhibiting the activation of NF-κB pathway. Finally, BA with the ability of suppressing NLRP3 inflammasome activation could reduce the expression of caspase-1 protein, and ultimately decrease the expression of IL-1β and IL-18. Moreover, BA as the definite antioxidant drug could significantly inhibit the production of ROS induced by LPS; thus, it might be a key signal factor to inhibit the activation of NLRP3.
Proposed anti-inflammatory mechanism of BA in LPS-induced piglet mononuclear phagocytes. Solid bold lines represent confirmation and dashed lines represent hypothesis.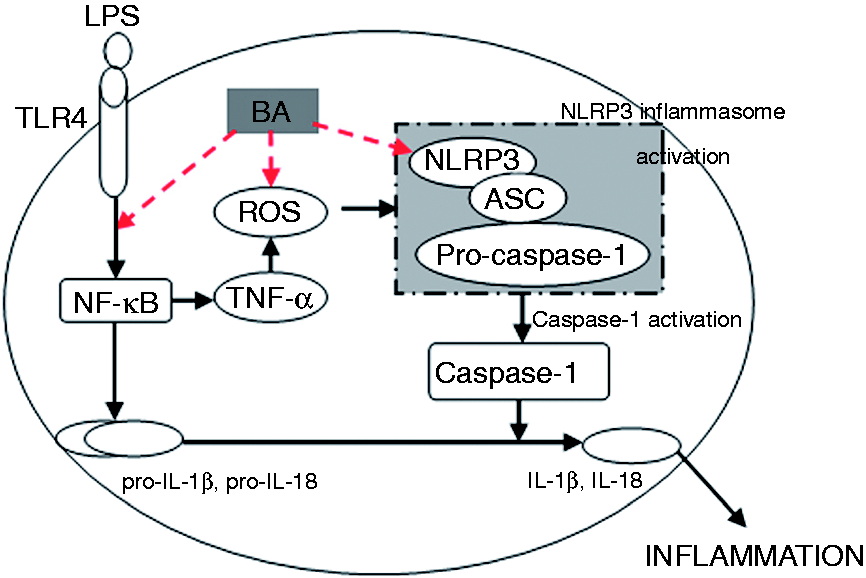
In summary, BA could significantly inhibit the production of cytokines, down-regulate the mRNA expression of NLRP3 and suppress the activation of caspase-1 in LPS-stimulated piglet mononuclear phagocytes. The anti-endotoxin effects of BA may be closely related to the inhibition of LPS-induced ROS production and mRNA expressions of NLRP3. Our results indicate that BA could significantly suppress the activation of NLRP3 inflammasome and suggeste it had an anti-inflammatory activity, which was related to the inhibition the activation of mononuclear cells through the suppression of NLRP3 inflammasome pathway. This study provides evidence to further understand the therapeutic effects of BA on inflammatory diseases in clinical practice, and it might be a reasonable veterinary drug for the prevention and treatment of piglet endotoxemia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 30972213, 31572572).
