Abstract
The family of kallikrein-related peptidases (KLKs) has been identified in a variety of immunolabeled human tissue sections, but no previous study has experimentally confirmed their presence in the human neutrophil. We have investigated the expression and bioregulation of particular KLKs in the human neutrophil and, in addition, examined whether stimulation by a kinin B1 receptor (B1R) agonist or fMet-Leu-Phe (fMLP) induces their secretion. Western blot analysis of neutrophil homogenates indicated that the MM of the KLKs ranged from 27 to 50 kDa. RT-PCR showed that blood neutrophils expressed only KLK1, KLK4, KLK10, KLK13, KLK14 and KLK15 mRNAs, whereas the non-differentiated HL-60 cells expressed most of them, with exception of KLK3 and KLK7. Nevertheless, mRNAs for KLK2, KLK5, KLK6 and KLK9 that were previously undetectable appeared after challenging with a mixture of cytokines. Both kinin B1R agonist and fMLP induced secretion of KLK1, KLK6, KLK10, KLK13 and KLK14 into the culture medium in similar amounts, whereas the B1R agonist caused the release of lower amounts of KLK2, KLK4 and KLK5. When secreted, the differing proteolytic activity of KLKs provides the human neutrophil with a multifunctional enzymatic capacity supporting a new dimension for its role in human disorders of diverse etiology.
Introduction
Neutrophils function not only as eminent phagocytes, but also contribute to orchestrating the immune/inflammatory response. 1 We postulated that proteases formed and stored in the neutrophil may play a seminal role in tissues to remove cellular debris and in infection acting to kill microorganisms, apart from regulating immune defense. The question we considered was whether the innate presence of the multigene group of serine proteases known as the human tissue kallikrein (KLK) family, could be established in the human neutrophil, as they may serve to control infections and immune-related human disorders. Experiments were therefore designed to determine whether members of the tissue KLK family are expressed in the neutrophil and whether their secretion could be stimulated by cytokines and bioactive agonists. The human tissue KLK family comprises a closely related multigene group of 15 serine endopeptidases, designated as KLK1–KLK15, located in tandem in a tightly clustered locus of >320 kb on chromosome 19 q13.3–13.4. The corresponding proteins include the classical (true) tissue kallikrein (KLK1) and the kallikrein-related peptidases (KLK2–KLK15), a group of proteases with tryptic or chymotryptic activity.2–6 Because KLK1 has the functional capacity to form pro-inflammatory kinin peptides from endogenous protein substrates called H- and L-kininogens, it is also classified as a kininogenase. The kinin peptides have a major role in the acute inflammatory response as they can mimic the cardinal signs of inflammation, namely erythema, increase in vascular permeability (edema) and pain.7,8 Furthermore, the local acidic conditions that prevail during tissue injury favor kinin accumulation. 7 In addition, the human neutrophil, a pivotal player of acute inflammation, has all of the components of the kinin cascade, including membrane-bound kininogens,9–11 KLK112 and the kinin B1 receptor (B1R), 13 a G protein-coupled receptor characterized by seven-transmembrane spanning helices. Although in most cells the B1R seems to be functionally latent under normal conditions,8,14,15 in the human neutrophil it is constitutively expressed in such a manner that its stimulation activates chemotaxis, 13 release of neutrophil proteases and up-regulation of CD11b/CD18 integrins. 16
Experiments with recombinant KLK-related peptidases appeared to indicate that they may function in a tissue-specific manner in many cellular and molecular events. For example, KLK3 (prostate-specific Ag) is an enzyme secreted by the prostate, responsible for the resolution of the seminal clot, whereas in the skin, KLK5 and KLK7 participate in desquamation of the superficial epidermal layers and activate antimicrobial peptides like the cathelicidins. 17 Despite these tissue-specific physiological functions, most research on KLK-related peptidases has been orientated to investigate their role in carcinogenesis, as several of its members have been proposed as biomarkers for prostate, ovarian and breast cancer among others. Nevertheless, KLK-related peptidases may be important players during inflammation and host defense as KLK6 and KLK13 have shown an inherent ability to degrade some components of the extracellular matrix, KLK5 catalyzes formation of functional antimicrobial peptides and KLK13 activates IL-1β from protein precursors. 18
Previous studies have established the expression of KLK1 in the human neutrophil, based on the use of inhibitors, kinin formation and immunological recognition by specific Abs in radioimmunoassay and immunocytochemistry.12,19–21 Furthermore, the presence of KLK1 in the human neutrophil has been confirmed by RT-PCR, 22 in situ hybridization and by Abs raised against the recombinant protein and against an N-terminal 11-amino acid peptide present in the pro-enzyme but not in the active protease. 23 By comparison, the presence of KLK-related peptidases has been detected only by immunoreactivity in polymorphonuclear leukocytes embedded in a variety of human tissue sections (summarized in Petraki et al. 24 ). Furthermore, previous to the current study, the identity of the neutrophil family of KLK-related peptidases had not been tested experimentally using molecular biology techniques. In view of the functional importance of these proteases in inflammation and cancer biology, we designed experiments to precisely identify each member of the neutrophil family of KLKs by using a number of molecular and immunolabeling techniques. The next question we addressed was whether members of this family would be secreted into the extracellular microenvironment by leukocytes stimulated with bioactive mediators. Evidence for such an effect was examined through stimulation of human neutrophils by the chemotactic bacterial peptide formyl-Met-Leu-Phe (fMLP), a cytokine mixture, LPS and an agonist of the kinin B1R.
Materials and methods
Ethical permission
Blood was collected by venipuncture from 15 volunteers following the guidelines stipulated by the medical ethical committee of Universidad Austral de Chile and the Declaration of Helsinki principles. Thirteen of the volunteers were clinically healthy and were not receiving any medication, nor was any volunteer suffering from a cold or had an allergic or any other inflammatory disorder. The two remaining volunteers had a localized bacterial skin infection. Blood from these two volunteers was collected on the first day of symptoms, before medication, and 3 wk later when they were symptom free. The age of the volunteers ranged from 20 to 45 yr old and they had a body mass index < 27 kg/m2.
Isolation of human neutrophils
Cells were isolated from anti-coagulated whole blood obtained from each volunteer. All procedures were carried out under sterile conditions using siliconized material as previously described. 16 Briefly, blood (35 ml) collected in 50-ml sterile tubes containing 3.8% trisodium citrate was mixed with 35 ml of dextran (6% MM 500,000; Sigma Aldrich, St. Louis MO, USA) and 105 ml PBS. After 20 min at room temperature (20–22℃) the upper leukocyte-enriched plasma was centrifuged and the cell pellet re-suspended in 1.5 ml of 50% Percoll (Sigma Aldrich) solution that was placed onto a Percoll gradient to separate neutrophils. Cell viability, assessed by trypan blue exclusion, was 99%.
Human HL-60 cells
The HL-60 cell line derived from an acute promyelocytic leukemia patient (ATCC, Manassas, VA, USA) was used as a source of neutrophil precursors. Cells were cultured at 37℃ and 5% CO2 in Iscove's Modified Dulbecco's medium (Life Technologies, Carlsbad, CA, USA) supplemented with 20% FBS, 4 mM
Effect of pro-inflammatory mediators on neutrophil KLKs
Neutrophils isolated from healthy human participants were re-suspended in 200 µl Hank’s balanced salt solution (HBSS) containing a mixture of the following cytokines: 1500 U/ml GM-CSF, 38 ng/ml TNF-α, 20 ng/ml IL-8, 1.5 ng/ml IL-1β and 150 U/ml IFN-γ. These are pro-inflammatory cytokines that are usually elevated in individuals during acute inflammation and that may also have effects on enzyme releases and changes in neutrophil gene expression, among others.25,26 In addition, exacerbation of inflammation has been observed following administration of GM-CSF, whereas gene targeting or blockade by an anti-GM-CSF Ab in animal models ameliorates inflammation. 27 Another group of neutrophils was directly stimulated with 1 µg/ml LPS. In both cases, cells were stimulated for 1, 3 or 6 h at 37℃ and 5% CO2. After stimulation, incubation media was stored at –80℃ until used, whereas cells were utilized to prepare total mRNAs or to visualize KLKs proteins by Western blotting.
Neutrophil stimulation by fMLP and by a kinin B1R agonist
Neutrophils (107 cells/500 µl HBSS) were incubated for variable periods of time at 37℃ in an atmosphere of 5% CO2–95% air with 100 nM fMLP or 100 nM of the kinin B1R agonist, Lys-des[Arg 9 ]bradykinin (LDBK), as previously described.13,16,28 Negative controls were performed by incubating the neutrophils under the same conditions, but in the absence of stimulus. fMLP was suspended in DMSO, whereas LDBK was directly dissolved in HBSS. Incubation of neutrophils with DMSO diluted with HBSS at the same concentration used in the experiments described above (0.01%) did not induce the release of neutrophil content (not shown). Cell viability, assessed by trypan blue exclusion, before and after experiments, was 98%. Proteins present in each incubation medium were precipitated using a ProteoExtract Protein Precipitation Kit (Calbiochem, La Jolla, CA, USA), re-suspended in electrophoresis sample buffer and proteins separated by 12.5% SDS-PAGE.
RT-PCR
Primers sequences used for PCR amplification.

Immunocytochemistry
Isolated neutrophils were smeared with aid of a cytocentrifuge on 22 × 22-mm coverslips. After washing with Dulbecco's PBS (DPBS) cells were fixed with methanol at −20℃ for 45 min, completely air dried and stored at −80℃ until used. For immunolabeling the fixed cells were incubated at room temperature for 30 min with DPBS containing human serum (1:20) to block Fc receptors and 3% BSA to prevent non-specific labeling. Next, cover slips were incubated overnight (12 h) at 4℃ with each of the anti-KLK Abs (Abcam, Cambridge, UK) according to manufacturer instructions in DPBS–1% BSA. After washing, cells were incubated with an anti-mouse or anti-rabbit F(ab’)2 fragments conjugated to Alexa 488 (1:1000; Molecular probes, Eugene, OR, USA). Nuclei were detected with 0.5 µg/ml of 4',6-diamidino-2-phenylindole (DAPI) in DPBS. Immunolabeling was evaluated by epifluorescence or in an Olympus Fluoview FV1000 confocal microscope (Olympus, Tokyo, Japan) using the 488-nm Argon-Krypton and 405 nm of diode lasers to the excitation of Alexa 488 and DAPI, respectively. Optical sections taken every 0.4 µm were digitalized and images processed using Image J (NIH, Bethesda, MD, USA). Negative controls included omission of primary Ab or its replacement by matching non-immune immunoglobulins at the same dilution.
Western blotting
Neutrophils were homogenized by the addition of ice-cold radioimmunoprecipitation assay buffer (50 mM Tris-HCl, pH 7.4, containing 150 mM NaCl, 0.25% deoxycholic acid, 1% Nonidet P40, 1 mM EDTA, 1 mM Na3VO4, 250 µg/ml p-nitrophenyl phosphate, 1 mM phenylmethane-sulphonyl fluoride, 1 mM NaF, 1 µg/ml leupeptin, 1 µg/ml pepstatin and 1 µg/ml aprotinin). Cells were sonicated and the amount of protein measured. Equal amount of protein (50 µg), dissolved in sample buffer containing 2.5% β-mercaptoethanol were subjected to SDS-PAGE and then transferred onto immobilon P. Proteins precipitated from incubation media of stimulated and non-stimulated neutrophils were processed simultaneously and in an identical manner. Blots were incubated for 2 h with each of the anti-KLK Abs (Abcam) in PBS containing 0.1% Tween 20 (Sigma Aldrich) and 5% BSA. Bound Abs were detected using the Super Signal West Dura Extended Duration chemiluminescence kit (Pierce, Rockford, IL, USA).
Quantitative image analysis and statistics
The intensity of immunoreactive protein bands, visualized after immunoblotting, was quantified using an automated image digitizing system (Un-Scan-It, Silk Scientific Inc., Orem, UT, USA) as described previously.13,16,28 At least three independent experiments were performed for each measurement and representative images from different experiments are depicted in the figures. Student’s t-test was used to analyze differences between stimulated and non-stimulated neutrophils (GraphPad InStat, San Diego, CA, USA). Values are expressed as mean ± SEM, and significance was considered acceptable at the 5% level (P < 0.05).
Results
Immunoreactivity to members of the neutrophil family of KLKs
When used on cell smears, all the Abs reacted strongly with their corresponding KLK family member (Figure 1A). Immunoreactivity to all KLKs was homogeneously distributed in the neutrophil cytoplasm with absence of membrane-associated labeling. In fact, confocal microscopy images of neutrophils immunostained for each KLK showed a granular pattern of variable size that seemed to be diversely distributed between the different kallikreins (Figure 1A). Immunoreactivity was only observed when neutrophils were incubated with the corresponding anti-KLK Ab, but not when it was omitted or replaced by non-immune immunoglobulins at the same dilution (Figure 1A).
Immunodetection of the neutrophil KLK family by immunocytochemistry and Western blotting. (A) Neutrophils isolated from volunteer peripheral blood were attached to round coverslips with the aid of a cytocentrifuge. After fixation with methanol they were incubated with each anti-KLK Ab and then with the corresponding Alexa-labeled secondary Ab (green); nuclei were stained with DAPI (blue). KLK1, true kallikrein; KLK2–15, KLK-related peptidases. Control corresponds to neutrophils that were only incubated with the Alexa-labeled secondary Ab. (B) Neutrophils were homogenized and proteins separated by SDS-PAGE and transferred onto Immobilon P and then immunoprinted for each kallikrein (KLK1–15). Representative Western blots and images of three independent experiments (n = 3) are shown.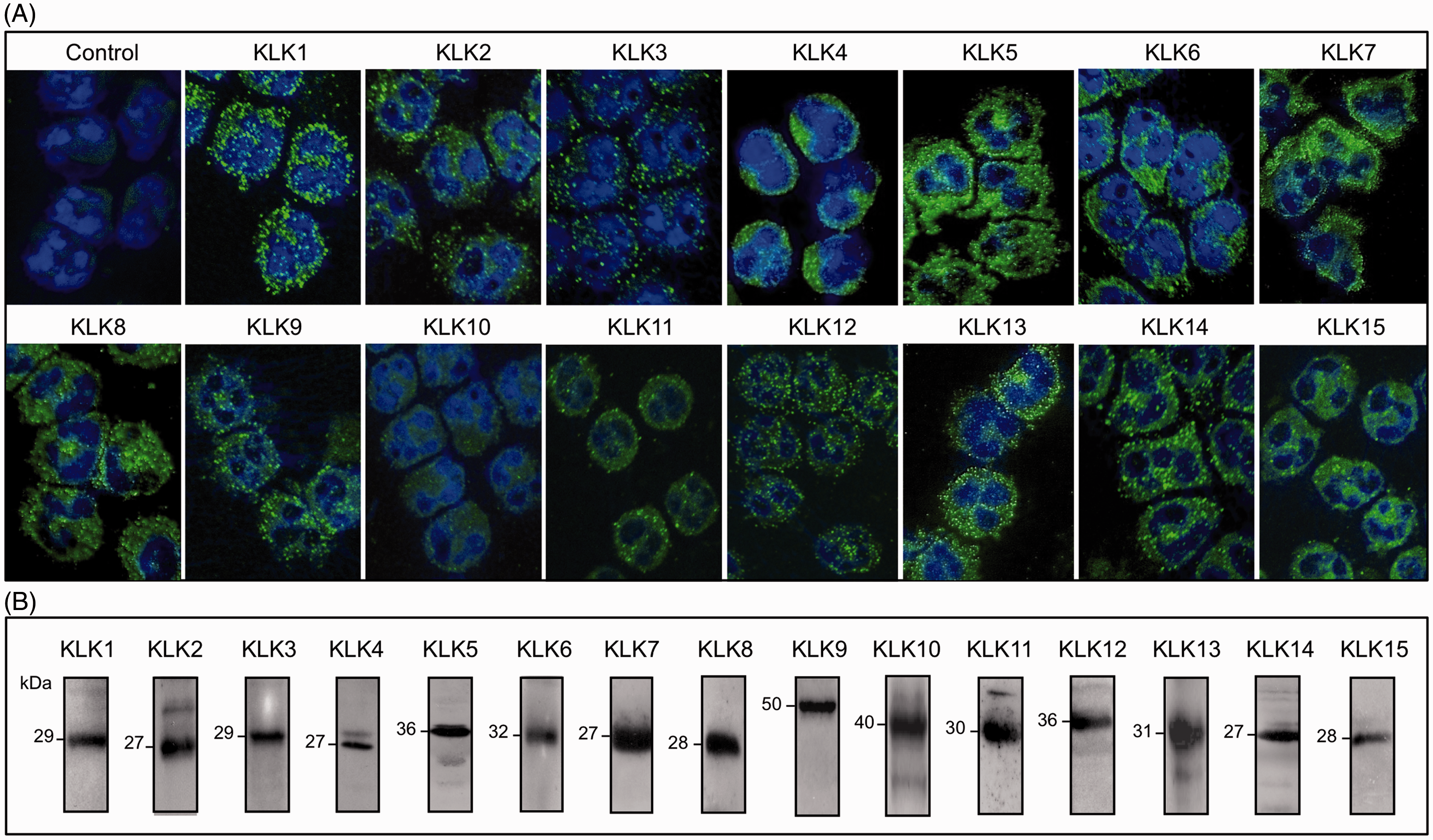
Cell homogenates prepared from isolated human neutrophils and subjected to SDS-PAGE and Western blotting with Abs directed to each KLK family member displayed the expected immunoreactive bands for each protease with relative molecular masses ranging between 27 and 50 kDa (Figure 1B). Positive controls performed with human MCF-7 breast cancer cells, HaCaT keratinocytes and human prostate showed immunoreactive bands of similar size (not shown).
Expression of the neutrophil family of KLKs is diverse
In contrast to that observed for KLKs protein expression when we probed for the expression of KLKs mRNAs using primers, which had been used previously to successfully identify these proteases in various tissues,29,30 we only obtained amplified transcripts for KLK1, KLK4, KLK10, KLK13, KLK14 and KLK15 (Figure 2). As most of circulating human neutrophils are well-differentiated cells, we performed the same protocol on the promyelocytic HL-60 cell line. This approach identified expression of additional KLK mRNAs those of KLK2, KLK5, KLK6, KLK8, KLK9, KLK11 and KLK12. However, expression of KLK3 and KLK7 was not observed in these cells. As expected, the same primers amplified the corresponding KLK in its matching positive control (Figure 2). Controls in which reverse transcriptase was omitted did not amplify the corresponding cDNA (Figure 2).
Identification of the neutrophil KLK family by RT-PCR. KLK mRNA transcripts were identified in neutrophils or in HL-60 cells. Positive controls correspond to mRNAs isolated from HaCaT keratinocytes (KLK6, KLK7, KLK8, KLK9, KLK10, KLK11, KLK12, KLK13 and KLK15), MCF-7 breast cancer cells (KLK1, KLK4, KLK5 and KLK14) and human prostate (KLK2, KLK3 and KLK4). Representative images of four independent experiments (n = 4) are shown.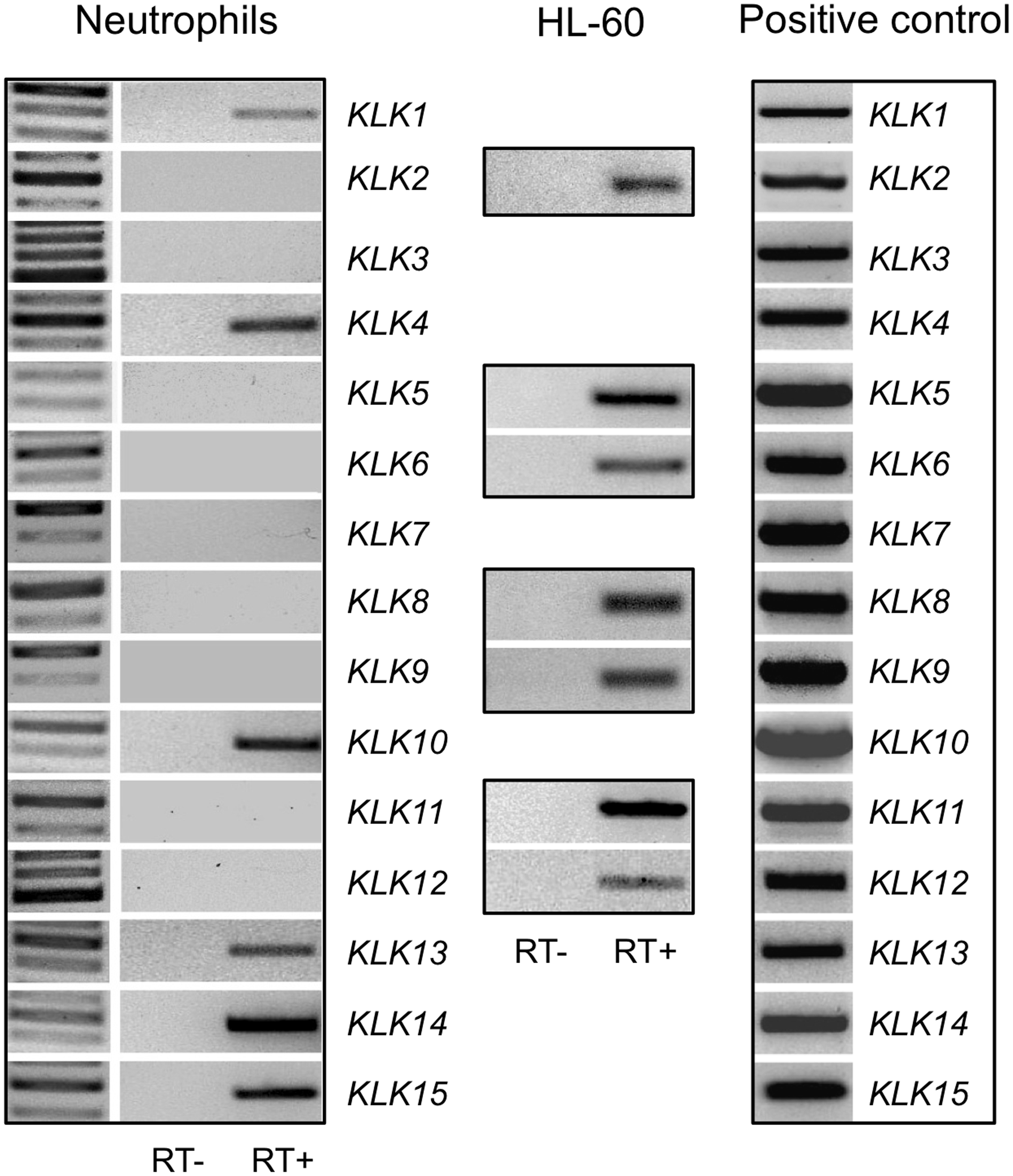
KLK mRNAs are expressed in blood neutrophils challenged with pro-inflammatory agents
Because some KLK mRNAs (i.e. KLK2, KLK5, KLK6, KLK8 and KLK9) were not expressed in the circulating neutrophil we asked the question as to whether they could be expressed under an appropriate mediator challenge. For this purpose, freshly isolated neutrophils were stimulated either with a mix of cytokines or LPS. Surprisingly, KLK mRNAs that were previously undetectable became expressed following the challenge indicating that their expression was dependent on cytokine stimulation (Figure 3A); in fact, unchallenged neutrophils obtained from the same individuals did not express the KLK mRNA (not shown). However, only KLK mRNAs corresponding to KLKs 2, 5, 6 and 9 were expressed, whereas KLK8 was not (Figure 3A). In contrast, neutrophils isolated from the two volunteers who were suffering an acute inflammatory process expressed at least four of these KLK mRNAs, namely KLKs 2, 5, 8 and 9, but not KLK6, that were previously undetected in leukocytes isolated from healthy individuals (Figures 2 and 3A).
Effect of a mixture of cytokines or tissue infection on the expression of specific members of the neutrophil KLK family. (A) Expression of KLK mRNAs transcripts in neutrophils incubated with a mixture of cytokines for 3 h or directly isolated from individuals with acute inflammation due to skin infection. Positive controls correspond to mRNAs isolated from HaCaT keratinocytes (KLK6, KLK8 and KLK9) or MCF-7 breast cancer cells (KLK2 and KLK5). Representative agarose gel images of two independent experiments (n = 2) are shown. (B) Protein levels of KLK2, KLK5 KLK6 and KLK9 in neutrophils treated with a mixture of cytokines or LPS for 6 h. (C) Protein levels of the same KLKs in the incubation media after treatment of the neutrophils with the same stimulus. Representative Western blots are shown and data represent the mean ± sem of three independent experiments (n = 3). *P < 0.05, **P < 0.01 between untreated and stimulated cells.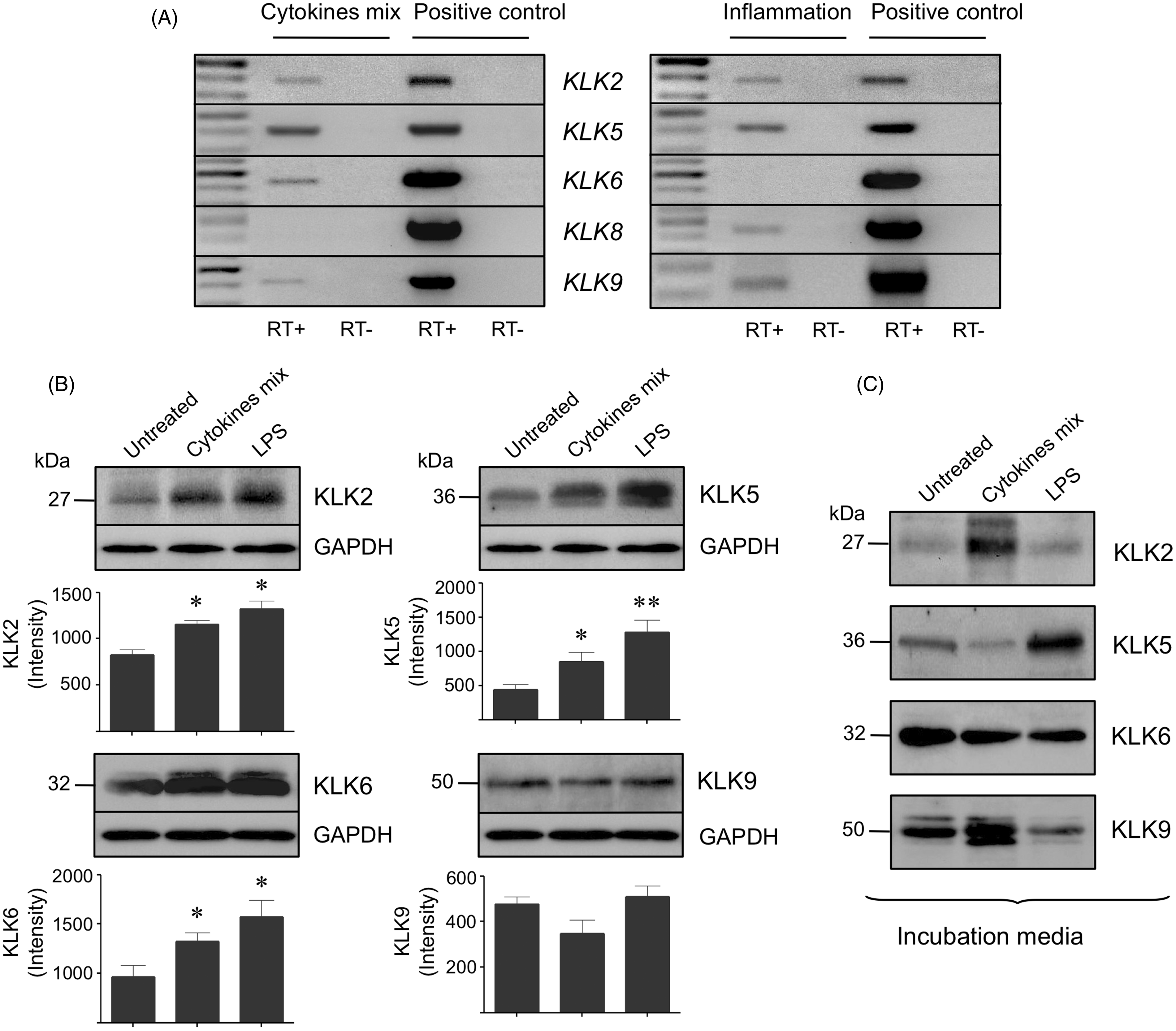
Additionally, we evaluated the protein levels of KLK2, KLK5, KLK6 and KLK9 in both unchallenged and challenged neutrophils, and in the incubation media at the end of incubation period to determine whether they are secreted after stimulation. We observed that challenging of freshly isolated neutrophils with a cytokine mixture or LPS for 6 hr increased levels of KLK2, KLK5 and KLK6 proteins when compared with unchallenged neutrophils from the same individuals (Figure 3B). Interestingly, the levels of KLK2 and KLK9 in the incubation media increased only after challenging with a cytokine mix, but not with LPS (Figure 3C). The opposite result was observed for KLK5 as levels of this protease in the medium increased only after treatment of the cells with LPS (Figure 3C). However, KLK6 levels released into the incubation medium did not change significantly during stimulation with LPS or the cytokine mixture (Figure 3C).
Members of the neutrophil family of KLKs are secreted after stimulation with fMLP or a kinin B1R agonist
Because the immunolabeled KLKs showed a granular appearance, we asked the question whether activation of human neutrophils by fMLP or by a kinin B1R agonist such as LDBK would cause the secretion of KLK6 and KLK13 to the incubation media. We chose these two KLKs because they have the ability to hydrolyze several components of the extracellular matrix, an event that may be important for neutrophil migration after they leave the circulation to enter into the inflamed microenvironment. When neutrophils were incubated with 100 nM fMLP, a clear, time-dependent release of both KLK6 and KLK13 was observed (Figure 4A, C). Secretion of KLK6 was maximal when the cells were stimulated for 15 min (Figure 4C), whereas that of KLK13 peaked largely between 15 and 30 min after stimulation (Figure 4A). By comparison, a similar response was observed when neutrophils were stimulated with 100 nM LDBK under same conditions. Although the time-dependent secretion of KLK6 was similar to that observed after stimulation of neutrophils with fMLP, maximal release of KLK13 occurred 5 min after stimulation with the B1R agonist (Figures 4B, D). Release of KLKs from stimulated and non-stimulated neutrophils was confirmed when the level of the corresponding KLK was evaluated in homogenates of these neutrophils (not shown).
Release of KLK6 and KLK13 from human neutrophils stimulated with fMLP or LDBK. Neutrophils were stimulated with 100 nM fMLP or LDBK for various periods of time and then the proteins present in the cell-free supernatants were precipitated and separated by SDS-PAGE, transferred onto Immobilon P and immunoprinted for KLK13 and KLK6. Specificity of the kinin B1R agonist LDBK was evaluated by stimulating the cells for 5 (KLK13) or 15 min (KLK6) with 100 nM LDBK after pre-incubation for 30 min with 1 μM of the B1R antagonist (B1R Ant) desArg
9
[Leu8]-bradykinin. Representative Western blots and images of three independent experiments (n = 3) are shown. Data represent the mean ± SEM (n = 3). *P < 0.05, **P < 0.01 between unstimulated and stimulated cells. #P < 0.01 between neutrophils stimulated with LDBK and those pre-incubated with the B1R antagonist and then stimulated with LDBK.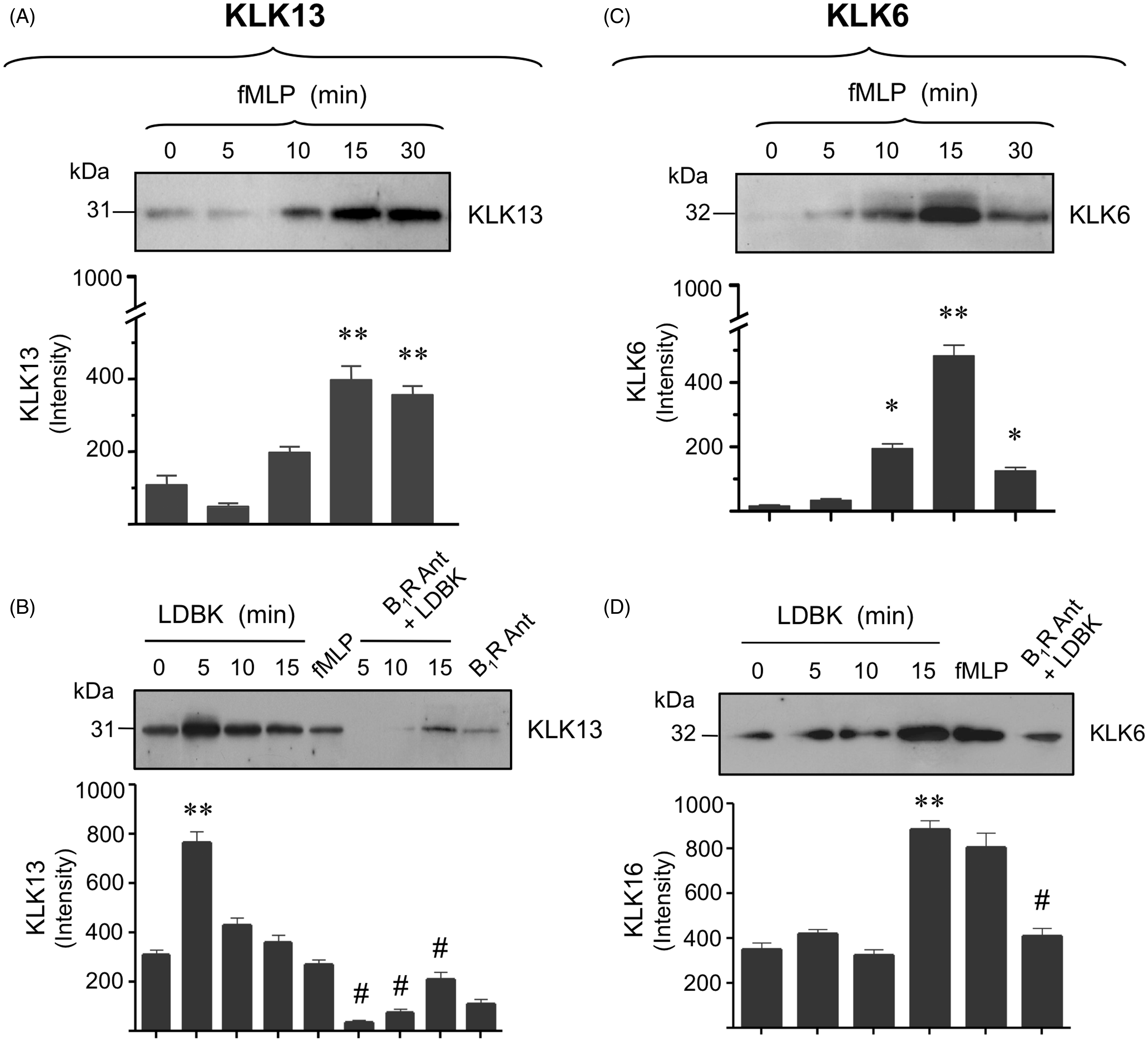
Once conditions to assess the secretion of KLK6 and KLK13 into the incubation media following stimulation with fMLP or LDBK had been established, we proceeded to examine the secretion of KLK1 (true kallikrein, kininogenase) and, in addition, KLKs 2, 4, 5, 10 and 14 (Figure 5). Our experiments using fMLP as a stimulus indicated that KLK1 and KLKs 2, 4, 5, 10 and 14 were secreted into the media in similar amounts to those described for KLK6 and KLK13. By contrast, KLKs 2, 4 and 5 were secreted in minimal amounts in response to stimulation with LDBK (Figure 5). Pretreatment of human neutrophils with the kinin B1R antagonists, des[Arg9]-Leu8-bradykinin or Lys-des[Arg9]-Leu8-bradykinin entirely blocked the secretion of KLKs induced by LDBK, thereby confirming that secretion of these neutrophil KLKs was, in fact, mediated through activation of the B1R (Figure 4B, D and Figure 5).
Release of various members of the neutrophil KLK family following stimulation with fMLP or LDBK. Proteins present in the cell-free supernatants of cells stimulated with 100 nM fMLP or LDBK for various periods of time were precipitated and then separated by SDS-PAGE, transferred onto Immobilon P and immunoprinted for the corresponding KLK. Specificity of the effect produced by LDBK was evaluated by stimulating the cells for 10 min with 100 nM LDBK (KLK1 and KLK10) after pre-incubation for 30 min with 1 μM of the B1R antagonist (B1R Ant) desArg9[Leu8]-bradykinin. Data represent the mean ± SEM (n = 3). *P < 0.05; **P < 0.01.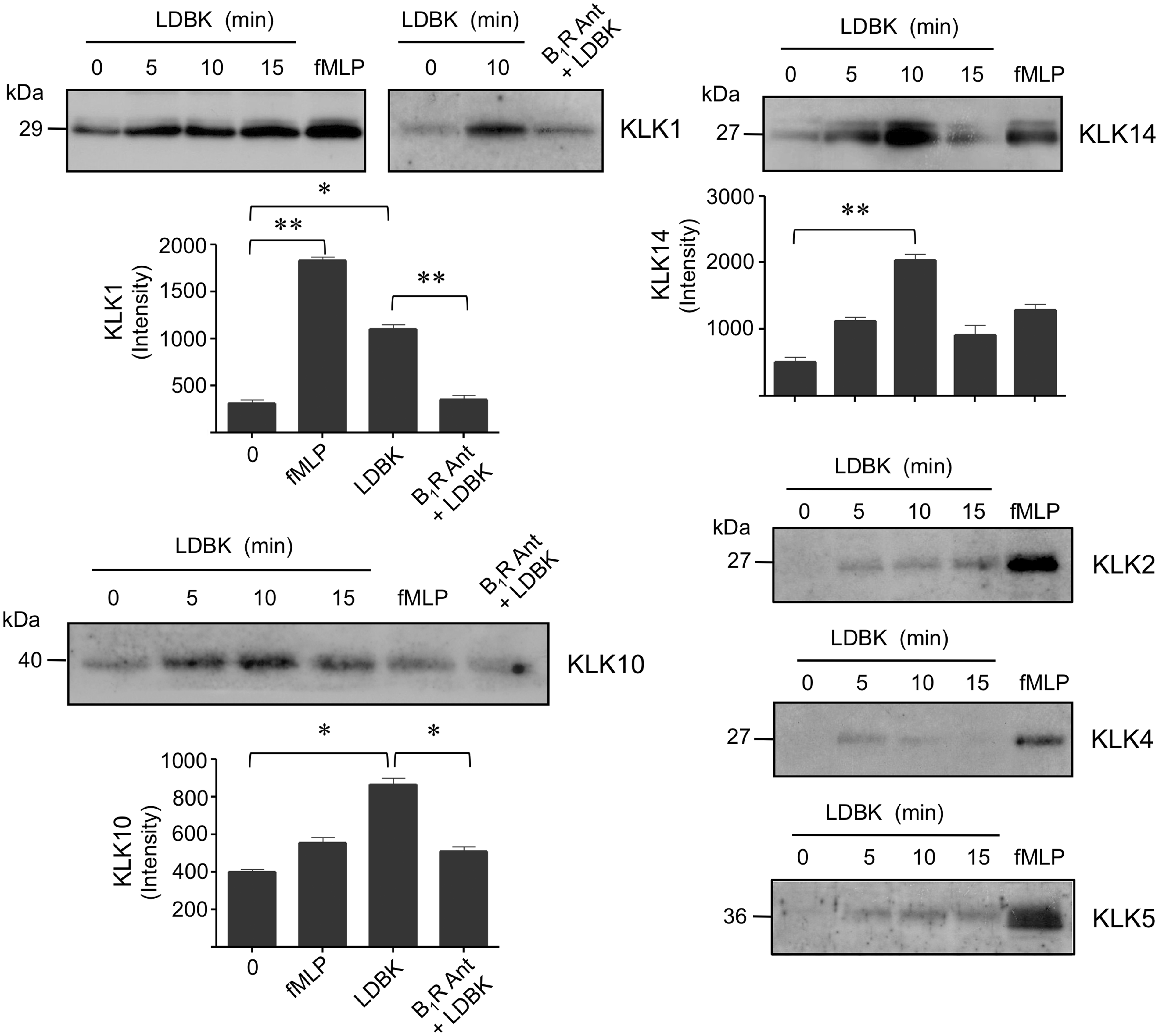
Additional controls performed by incubating neutrophils with the solvents used to suspend the B1R agonist and antagonists (0.9% NaCl) as well as fMLP (DMSO), and then diluted with HBSS at the same concentrations as those used in the experiments described above, did not induce release of KLKs (not shown).
Discussion
In this study, we have clearly established the presence of a neutrophil family of KLK-related peptidases using molecular biology and immunolabeling techniques, and we have also examined the concept that various members of this family are secreted into the extracellular microenvironment after stimulation by particular bioactive mediators.
KLK1 (true tissue kallikrein, kininogenase or kallikrein 1) was the first member of the family identified in human neutrophils by specific enzyme activity, inhibitors profile, formation of bioactive kinin peptides and immunological recognition by specific Abs both in radioimmunoassay and on cell smears prepared from either whole peripheral blood or bone marrow.12,31 Subsequently, its presence in the human neutrophil was confirmed by different techniques that included mRNA and pro-enzyme determination.22,23 Later reports indicated that thrombin-stimulated neutrophils release immunoreactive KLK1, suggesting a possible linkage between blood coagulation and inflammation. 19 We have previously reported that KLK1 is also secreted by neutrophils after stimulation with fMLP, thrombin or immune complexes and that it can produce biologically active kinin peptides in the presence of purified kininogens. 21 Here, we show that the kinin B1R agonist, LDBK induces the release of KLK1 from human neutrophils, raising the possibility that more kinin peptides could be generated by the enzymatic activity of KLK1 on plasma kininogens. In addition to KLK1, purified KLK2 has kininogenase activity as it cleaves H-kininogen at specific Arg residues to yield bradykinin and several other peptides, but this property has been demonstrated only in in vitro experiments. 32 Kinins generated by KLK1 during acute inflammation are considered to increase pain and vascular permeability, induce neutrophil chemotaxis, degranulation and up-regulation of CD11b/CD18 via activation of kinin B1 receptors.13,16,28
KLK-related peptidases are a family of serine proteases with functions, which have not yet been comprehensively investigated in the neutrophil. In the current study, we provide experimental evidence for the presence of a neutrophil family of KLKs. Although all of the 15 KLK proteins were detected, equivalent mRNAs were identified only for KLKs 1, 4, 10, 13, 14 and 15 in the circulating human neutrophils suggesting that immunoreactive protein levels do not necessarily correspond with the spectrum of mRNAs. By comparison, mRNA levels for KLKs 2, 5, 6, 8, 9, 11 and 12, which appeared to be absent in circulating neutrophils, were easily detected in HL-60 cells. As most of the KLK mRNAs were detected in HL60 cells with the exception of the KLK3 and KLK7, one possible explanation would be that very low levels of mRNA may not be detectable in these undifferentiated cells. Lack of expression of most KLK mRNAs in circulating neutrophils with respect to HL-60 cells suggests that synthesis of these proteins occurs largely during cell differentiation and, therefore, in the circulating neutrophil the mRNAs might be already degraded, or silenced.
As variable levels of KLK mRNAs were present in the human neutrophil, we investigated the possibility that individual KLK mRNAs and their corresponding proteins may vary after contact of neutrophils with pro-inflammatory cytokines or in neutrophils obtained from individuals suffering acute inflammatory episodes. Surprisingly, those KLK mRNAs that were undetectable in circulating neutrophils isolated from healthy individuals became expressed after exposure to a mixture of different pro-inflammatory cytokines thereby providing evidence of their latent presence in the human neutrophil. Furthermore, this finding was supported by the detection of mRNAs for KLKs 2, 5, 8 and 9 in neutrophils isolated from the two volunteers who had an inflammatory process. Additionally, in both cells and incubation media increased levels of the equivalent proteins accompanied the augmented levels for KLK2, KLK5, KLK6 and KLK9 mRNAs in the stimulated neutrophils.
A role for KLK5 in inflammation and antibacterial protection is supported by our finding that this peptidase is secreted by neutrophils stimulated with LPS and also by evidence indicating that its activity is not inhibited at acidic pH, a characteristic of the inflammatory milieu. Under these circumstances, KLK5 secreted into the inflamed site may, by autoactivation, initiate the known KLK cascade, resulting in activation of other KLKs such as pro-KLK14, which, in turn, increases activation of pro-KLK5 and also simultaneously activates pro-KLK1, the kininogenase responsible for kinin formation.33,34 Interestingly, KLK14 is considered to be the physiologic activator of pro-KLK1 and thereby to bioregulate kinin formation. 35 Furthermore, as KLK5 regulates activation of the cathelicidin precursor and formation of LL-37 on the skin surface and processes defensin-1α in vitro,36,37 it is likely that neutrophil KLK5 may enhance the generation of LL-37 traditionally assigned to proteinase-3. 38 Active LL-37 is also a potent chemoattractant for neutrophils, monocytes and T lymphocytes a feature that may improve recruitment of inflammatory cells at the site of injury.39,40 In addition, KLK5 may also process LL-37 to smaller peptides, which, in vitro, display higher antimicrobial activity against various types of bacteria such as Escherichia coli and Staphylococcus aureus, and the yeast Candida albicans than intact LL-37.36,41 In addition to cathelicidin and defensin-1α, the precursor of IL-1β in saliva is mostly activated by KLK13 by cleaving its internal Leu113–Leu114 bond. 42 Interestingly, in the epidermis, LL-37 acts synergistically with IL-1β to increase immune response. 43 Thus, activation of antimicrobial peptides by KLK5 and IL-1β by KLK13 strongly supports a role for these members of the neutrophil kallikrein family in innate immunity.
Additionally, we have shown that bacterial peptides like fMLP or the bioactive mediator LDBK stimulate secretion of various members of the human neutrophil family of KLKs. We have evaluated the secretion of KLKs 1, 2, 4, 5, 6, 10, 13 and 14 from neutrophils and observed that the release of KLKs 1, 6, 10, 13 and 14 following stimulation by the B1R agonist was similar to that produced by fMLP. By comparison, KLKs 2, 4 and 5 were secreted at higher levels when stimulated with fMLP than with the B1R agonist. These experiments suggest that the type of stimulus (i.e. an inflammatory mediator vs. a bacterial product) may influence the differential secretion of particular members of the neutrophil KLK family.
In contrast to KLK5, the hydrolytic activity of KLKs 2, 6 and 13 seems to be pH independent, 44 a characteristic that makes these members highly relevant in the inflamed microenvironment where local pH is reduced to values <6. In fact, KLK6 activity generally increases in acidic conditions such as those that occur during inflammation, hypoxia and tissue damage. 44 Once activated, several KLKs have the ability to degrade different components of the extracellular matrix such as collagen, fibronectin, laminin and vitronectin (KLKs 2, 5, 6, 8, 13 and 14), and activate pro-metalloproteases 2 and 9, and procollagenase (KLK1).7,18,45 These proteolytic activities may be highly relevant to neutrophil function during acute inflammation, especially if they become released during its mobilization across the extracellular matrix.
Evidence suggests that members of the neutrophil KLK family may function as proteases in inflammation, allergy, autoimmune diseases and cutaneous chronic disorders. For instance, an anomalous overproduction of antimicrobial peptides generated by deregulation of members of the KLK family has been shown to have negative consequences in rosacea. 40 KLKs are also highly up-regulated in psoriasis producing over-expression of LL-37, which, in turn, enhances chronic inflammation favoring the arrival of inflammatory cells. 46 KLK6 is secreted by T cells; further, its levels are increased in macrophages during spinal cord injury, 47 and also in CD4+ and CD8+ T cells and in macrophages at sites of demyelination in experimental models of multiple sclerosis. 48
In summary, our experiments have clearly shown the expression of a neutrophil family of KLK-related peptidases, some of which may be up-regulated and secreted after stimulation with a mixture of pro-inflammatory cytokines and also secreted by stimulation with a bacterial peptide or a kinin B1R agonist that may be generated during inflammation by the enzymatic action of KLK1 on extravasated plasma kininogens. Thus, the family of KLK-related peptidases and KLK1 provide the human neutrophil with an expanded proteolytic capacity that may be of crucial relevance in a wide range of human disorders of varying etiology.
Footnotes
Funding
This work was supported by the Fondo Nacional de Investigacion Cientifica y Tecnologica (FONDECYT, Chile) (grant number 1110464).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
