Abstract
Otitis media (OM), one of the most prevalent diseases in young children, is clinically important owing to its high incidence in children and its potential impact on language development and motor coordination. OM is the most common reason for the prescription of antibiotics (accounting for 25% of prescriptions) due to its extremely high incidence. A recent increase in antibiotic resistance among OM pathogens is emerging as a major public health concern globally, which led us to consider non-antibiotic approaches for the management of OM. In this study, we evaluated gene transfer of an antimicrobial peptide, human β-defensin 2 (DEFB4), using an adenoviral vector (Ad5 with deletions of E1/E3/E4) as a potential therapeutic approach. We demonstrated that the transduction of human β-defensin 2 induces the production of human β-defensin 2 and suppresses non-typeable Haemophilus influenzae (NTHi) adhesion to human middle ear epithelial cells. Moreover, intratympanic inoculation of Ad-DEFB4 was found to attenuate NTHi-induced middle ear effusions without eliciting a significant immune response. Most importantly, intratympanic inoculation of Ad-DEFB4 appeared to significantly augment clearance of NTHi from middle ear cavity. Collectively, our results suggest that intratympanic gene delivery of antimicrobial molecules may serve as an alternative/adjuvant approach for the management of OM.
Introduction
A majority of young children is known to experience at least one episode of otitis media (OM) by their third birthday. 1 Considering that over 20 million visits annually to physicians are due to OM, 2 OM in children is a tremendous burden to families owing to OM-related medical and socioeconomical costs. 3 Moreover, children with recurring episodes of OM are at higher risk for developing hearing loss and balance impairment,4–6 which can lead to detrimental effects on the development of language and motor coordination.
‘Watchful waiting’ is an acceptable alternative to immediate antibiotics for the management of nonsevere acute OM 7 because it is frequently cleared without drug intervention. However, antibiotics are still most commonly used for the treatment of OM, 8 and their over use is emerging as a global concern due to appearance of superbacteria resistant to multiple antibiotics. Therefore, a non-antibiotic therapeutic approach is of potential value against infectious diseases such as OM. The first line of tubotympanal defense against OM pathogens is mucociliary systems and secretory antimicrobial molecules, such as defensins. 9 Antimicrobial molecules are ideal candidates for the non-antibiotic therapeutic agents to treat bacterial diseases as antimicrobial molecule-deficient animals are susceptible to bacterial infections.10–12 Moreover, we have demonstrated that defensins effectively inactivate common OM pathogens, 13 which led us to further determine the therapeutic potential of defensins for the management of OM.
Defensins are a large family of small cationic peptides that are able to form membranous pores, leading to bacterial lysis.14,15 Among the defensins, the β-defensins are highly expressed in the tubotympanal epithelium. 16 In particular, human β-defensin 2 (also known as DEFB4 or DEFB4A) is inducible by a common OM pathogen, non-typeable Haemophilus influenzae (NTHi), in human middle ear epithelial cells,17,18 and has a potent antimicrobial activity against NTHi, 13 indicating its critical involvement in the pathogenesis of OM caused by NTHi.
NTHi, a Gram-negative rod-shaped bacterium, exists as a commensal organism in the human nasopharynx. 19 NTHi is considered to be strictly pathogenic in humans, causing OM and sinusitis in children, and exacerbating chronic lung disease in adults.20,21 In this study, NTHi was used as a model OM pathogen as intratympanic injection of NTHi induces middle ear inflammation and mucosal hyperplasia in mice. 22
Although recombinant antimicrobial molecules and their mimetics are partly protective for bacterial infections,23,24 there are significant challenges to application of a recombinant peptide/protein drug due to proteolytic inactivation and high production cost.25–27 Therefore, we aimed to deliver the human β-defensin 2 gene to induce its over-expression using a viral vector. We selected adenoviral vectors because they are able to infect diverse types of cells, 28 a target gene is transiently induced without chromosomal incorporation and a new generation of adenoviral vectors is rarely immunogenic.29–31
Here, we present the finding that local gene delivery of human β-defensin 2 is capable of significantly reducing experimental OM and enhancing bacterial clearance from the middle ear cavity. We also demonstrate that the adenoviral vector used in this study delivers the human β-defensin 2 gene to the middle ear epithelium without significant inflammatory reactions. As a proof-of-principle, this study shows the feasibility of adenoviral vector-mediated local over-expression of human β-defensin 2 for the management of OM.
Materials and methods
Bacterial culture
NTHi 12 (provided by Dr. Xin-Xing Gu, NIH), originally a clinical isolate from the middle ear fluid of a child with acute OM, 32 and GFP-conjugated NTHi 86-028NP (provided by Dr. Lauren Bakaletz, Ohio State University) 33 were used in this study. The bacteria were streaked onto a fresh chocolate agar plate and incubated overnight in a 37℃-incubator with 5% CO2. A single colony was grown in 3 ml of brain–heart infusion broth (BHI; Becton Dickinson, Cockeysville, MD, USA), supplemented with hemin (10 µg/ml) and NAD (10 µg/ml), and placed in a shaking incubator overnight (16 h). After the addition of fresh BHI broth, bacteria were further grown for 4 h to a mid-log phase (A600 = 0.4–0.6).
Cell culture
The human middle ear epithelial cell line (HMEEC) 34 was maintained in a 1:1 mixture of DMEM and bronchial epithelial basal medium (Lonza, Walkersville, MD, USA) supplemented with bovine pituitary extract (50 µg/ml), hydrocortisone (0.5 µg/ml), human epidermal growth factor (0.5 ng/ml), epinephrine (0.5 µg/ml), transferrin (10 µg/ml), insulin (5 µg/ml), triiodothyronine (6.5 ng/ml), retinoic acid (0.1 ng/ml), gentamicin (50 µg/ml) and amphotericin-B (50 ng/ml). A549 cells (CCL-185) were purchased from the ATCC (Manassas, VA, USA) and maintained in DMEM supplemented with 10% FBS. All cells were cultured at 37℃ in a humidified atmosphere with 5% CO2.
Adenoviral vector construction and animal experiments
Ad-null, Ad-DEFB4, Ad-EGFP and Ad-β-galactosidase were generated in collaboration with Dr. Douglas E Brough (GenVec, Gaithersburg, MD, USA), which are Ad5-based vectors with deletions in the E1, E3 and E4 genes, 35 and is driven by the human CMV constitutive promoter expression cassette. All aspects of animal handling were performed according to the approved Institutional Animal Care and Use Committee guidelines. For the analysis of bacterial clearance, 11 105 CFU of live NTHi was transtympanically inoculated into the middle ear cavity of C57BL/6 mice with or without recombinant DEFB4 (0.5–2.5 µg/ear), or adenoviral vectors [5 × 107 particle unit (p.u.)/ear] using a 30-G needle and syringe under a surgical microscope. Animals were euthanized 36 h after inoculation. After dissection of the temporal bones, middle ear effusions and lavages were collected and cultured in chocolate agar plates for bacterial colony counting. Then, temporal bones were fixed with 4% paraformaldehyde for hematoxylin and eosin (H&E) staining as described. 36 For tissue X-gal staining, 37 the mouse bullae were partially opened with a sharp chisel and washed with PBS. X-Gal solution (Life Technologies, Grand Island, NY, USA) 1 mg/ml was applied onto the middle ear epithelium. After incubation for 4 h at 37℃, the middle ear epithelium was observed under the dissecting microscope. X-Gal staining was quantitatively analyzed using Image J software (NIH, Bethesda, MD, USA).
Real-time quantitative PCR
After purification of total RNAs using the RNeasy kit (Qiagen, Valencia, CA, USA), cDNA was synthesized using the TaqMan reverse transcription kit (Applied Biosystems, Foster City, CA, USA). Multiplex PCR was performed using the ABI7500 Real Time PCR System (Applied Biosystems) with gene-specific primers, a FAM-conjugated probe for DEFB4 (Hs00175474_m1; Applied Biosystems) and a VIC-conjugated probe for cyclophilin (4326316E; Applied Biosystems). The cycle threshold (CT) values were determined according to the manufacturer’s instructions. The relative quantity of mRNAs was determined using the 2−(ΔΔCT) method. 38 CT values were normalized to the internal control (cyclophilin), and the results were expressed as fold-induction of mRNA, with the mRNA levels in the nontreated group set as 1.
ELISA
Culture supernatants and cell lysates were collected from the HMEEC cells infected with Ad-null or Ad-DEFB4. The total protein level of the samples was quantified using a BCA Protein Assay kit (Pierce Biotechnologies, Rockford, IL, USA). To measure adenovirus-mediated production of human β-defensin 2 in the HMEEC cells, ELISAs were conducted according to the manufacturer’s instructions (Phoenix Pharmaceuticals, Burlingame, CA, USA) as described. 18 Sample concentrations were determined by extrapolation to the standard curve, and data were expressed as pg/50 µl for the supernatants and pg/100 µg total proteins for the cell lysate.
NTHi adhesion assay
HMEEC cells (2 × 104) were infected with 1 × 106 p.u. of Ad-null or Ad-DEFB4 (50 p.u./cell) overnight, and were exposed to 2 × 106 CFU of live GFP-expressing NTHi (100 CFU/cell) as described. 39 Nonadherent bacteria were removed by PBS washing 2 h after infection, and samples were fixed with 4% paraformaldehyde. Mounted samples were then viewed and photographed using a confocal microscope equipped with an image analyzer (CTR6500; LEICA, Wetzlar, Germany). For quantitative analysis of NTHi attachment, GFP-positive NTHi was directly counted, and the fluorescence intensity was measured using Image J software (NIH).
Statistical analysis
All experiments were carried out in triplicate and repeated twice. Results were expressed as means ± SD. Statistical analysis was performed with a Student’s t-test and ANOVA, followed by Tukey’s post hoc test using R2.14.0 software (The R Foundation for Statistical Computing, Vienna, Austria). For the analysis of middle ear effusions and bacterial clearance, Pearson’s χ2 test was conducted. A P-value < 0.05 was considered significant.
Results
Recombinant human β-defensin 2 improves NTHi clearance from the middle ear cavity
As we have demonstrated that recombinant human β-defensin 2 effectively kills a common OM pathogen, NTHi,
13
we sought to determine if intratympanic application of recombinant β-defensin 2 provides protection against OM. Intratympanic application of recombinant human β-defensin 2 appeared to enhance NTHi clearance in a dose-dependent manner. In particular, 2.5 µg of human β-defensin 2 resulted in > 70% enhancement of NTHi clearance from middle ear cavity (Figure 1). Our findings suggest that recombinant human β-defensin 2 is protective for experimental OM in vivo, and is consistent with our prior study.
13
However, potential problems associated with the application of a peptide/protein drug led us to consider adenoviral vector-mediated delivery of the β-defensin 2 gene.
Intratympanic injection of recombinant human β-defensin 2 improves NTHi clearance from the mouse middle ear cavity. Bacterial colony counting shows that the number of live NTHi cultured from the middle ear lavage is reduced in the mice with intratympanic injection of recombinant human β-defensin 2 (>1 µg). The experiments were performed in triplicate and repeated twice. Values are given as the mean ± SD (n = 3). *P < 0.05.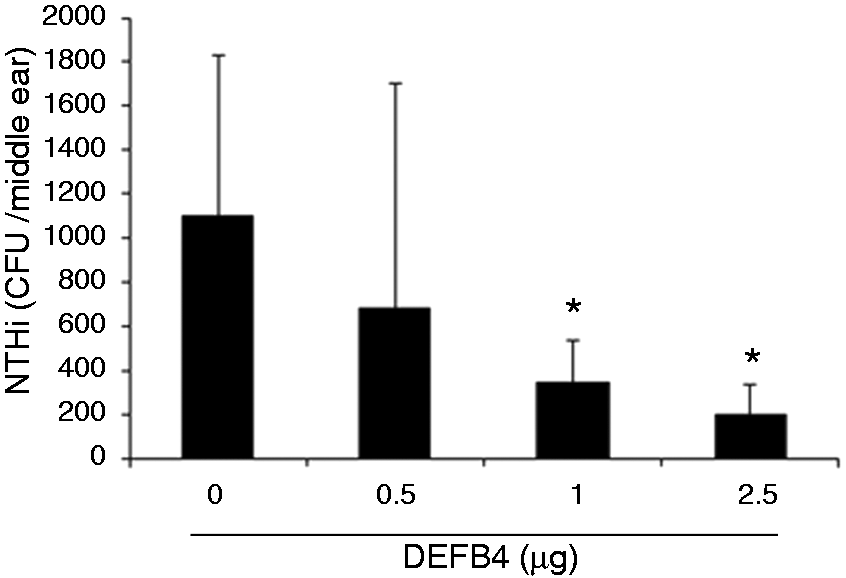
Adenoviral vector expressing human β-defensin 2 (Ad-DEFB4) up-regulates β-defensin 2 in human middle ear cells
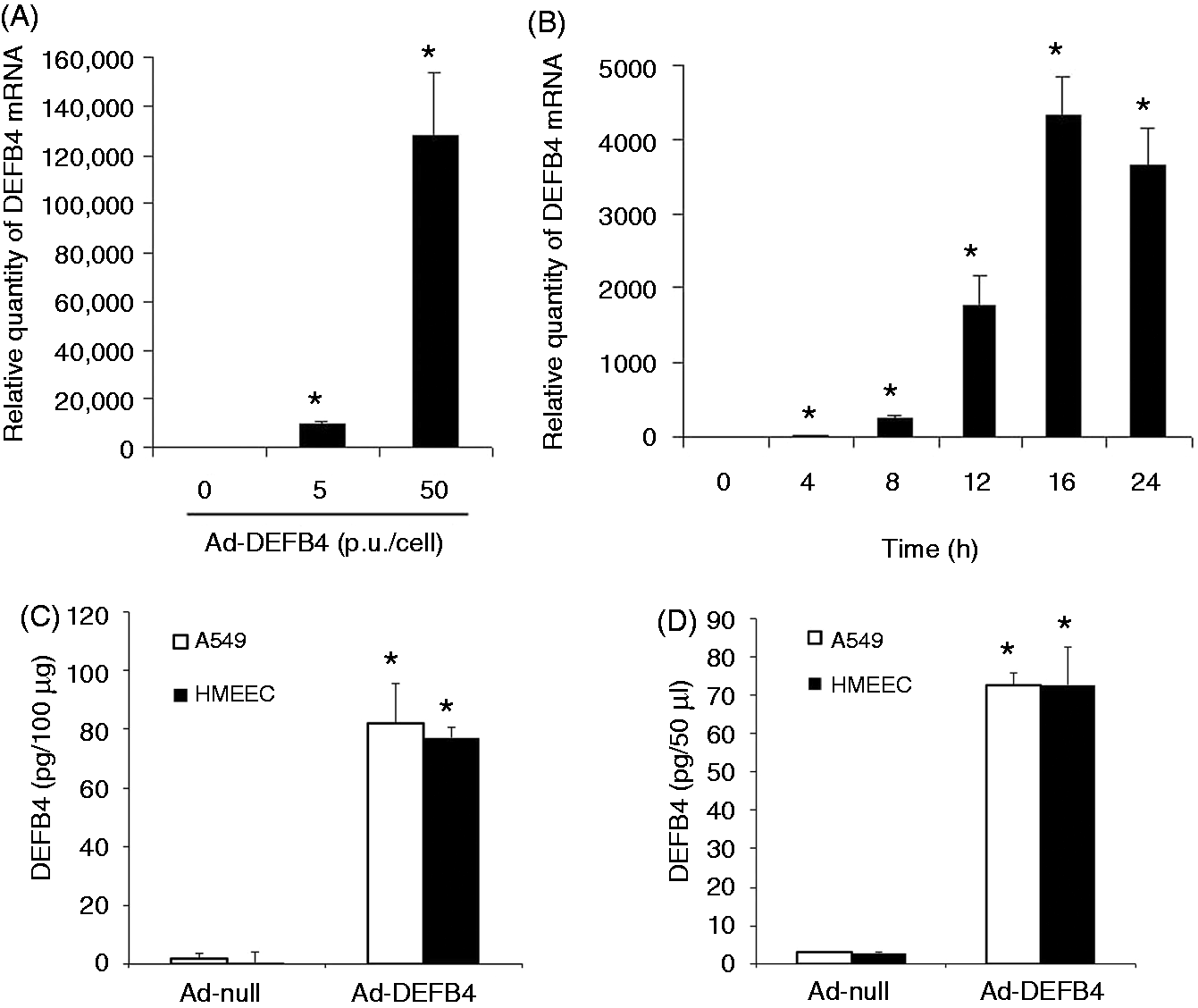
Gene delivery is a promising modality for the treatment of various incurable diseases.40–42 Among viral and nonviral vehicles studied for gene delivery, adenoviral vectors have been reported as a useful tool for gene delivery to the inner ear tissue.43,44 Hence, we sought to determine adenoviral vector-mediated in vitro gene delivery of the human β-defensin 2 gene to the middle ear epithelial cells in this study. To test the feasibility of adenoviral vector-mediated gene delivery to the middle ear epithelial cells, HMEEC cells were infected with an adenoviral vector expressing either enhanced GFP (EGFP; Ad-EGFP) or β-galactosidase (Ad-βgal). The adenoviral vector induced expression of EGFP and β-galactosidase in the HMEEC cells in a dose-dependent manner (Figure 2). To determine if an adenoviral vector can effectively induce expression of the human β-defensin 2 gene, the HMEEC cells were infected with Ad-DEFB4. Real-time quantitative RT-PCR analysis showed that the HMEEC cells with infection of Ad-DEFB4 highly up-regulate the expression of human β-defensin 2 gene in a dose- and a time-dependent manner (Figure 3A,B). Moreover, ELISA analysis showed that Ad-DEFB4 infection up-regulates the production and release of human β-defensin 2 in A549 and HMEEC cells compared with an empty vector (Ad-null; Figure 3C,D). Taken together, our results indicate that adenoviral vector-mediated gene delivery is an effective approach to over-express human β-defensin 2 in the human epithelial cells.
The adenoviral vector efficiently delivers genes to the middle ear epithelial cells. (A) Fluorescent microscopy (Fluorescence) and X-gal staining (X-gal) show adenoviral vector-mediated delivery of the EGFP and β-galactosidase genes to HMEEC cells in a dose-dependent manner. Original magnification×100. Ad-DEFB4 up-regulates human β-defensin 2 expression in the human middle ear cells. Quantitative RT-PCR analysis shows that infection of the HMEEC cells with Ad-DEFB4 up-regulates human β-defensin 2 expression in a (A) dose- and (B) time-dependent manner. ELISA analysis shows that the HMEEC and A549 cells infected with Ad-DEFB4 increase (C) production and (D) release of human β-defensin 2 compared with the cells infected with a control adenovirus (Ad-null). The experiments were performed in triplicate and repeated twice. Values are given as the mean ± SD (n = 3). *P < 0.05.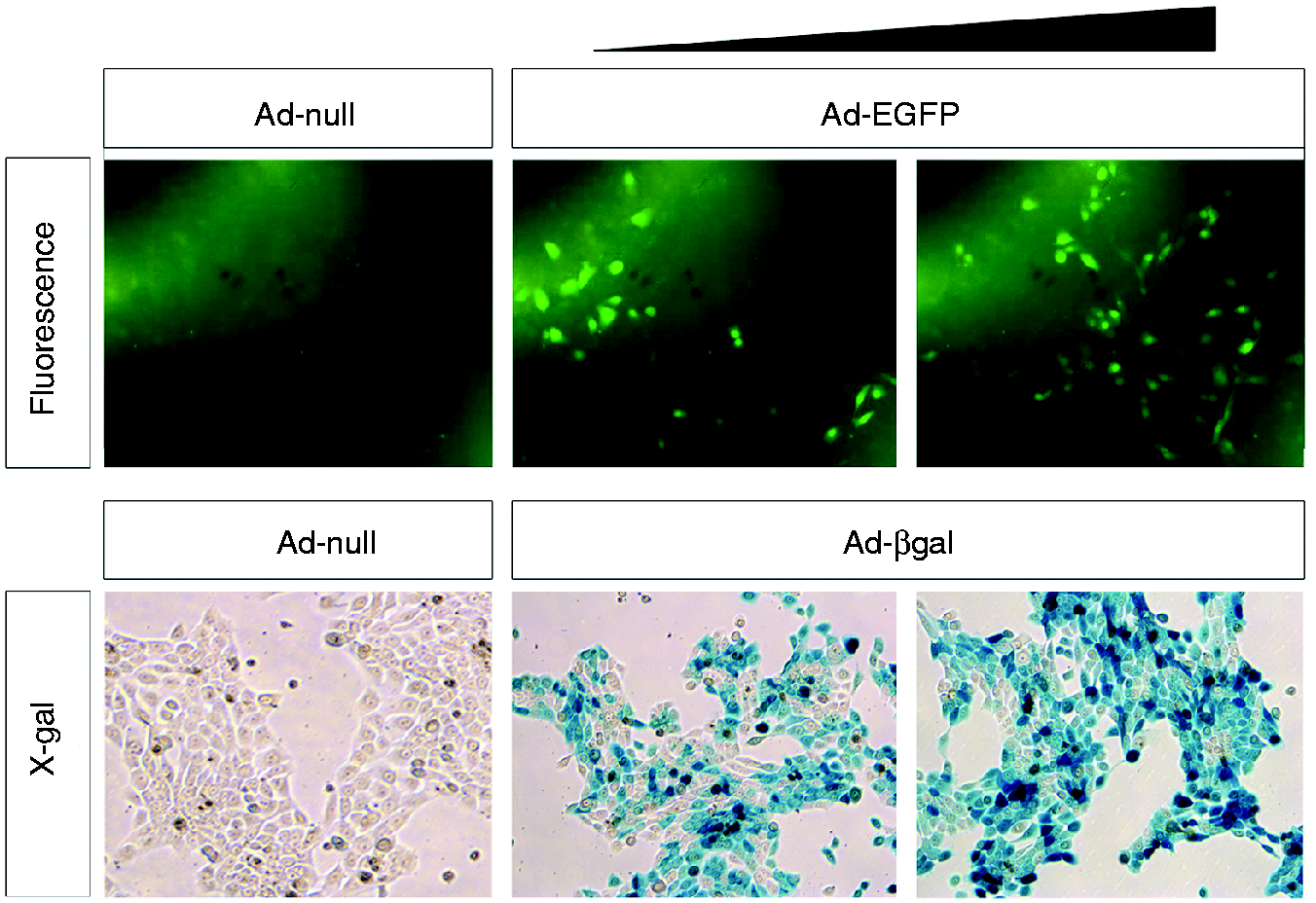
Ad-DEFB4 inhibits NTHi adhesion to human middle ear epithelial cells
For the development of middle ear infection, OM pathogens first need to adhere to the nasopharyngeal epithelium, which enables pathogens to enter the middle ear through the Eustachian tube.
9
Hence, we sought to determine if adenovirus-mediated over-expression of human β-defensin 2 affects NTHi adhesion to epithelial cells. For bacterial adhesion assays, the HMEEC cells were infected with Ad-null or Ad-DEFB4, and were exposed to live, GFP-expressing NTHi, and nonadherent bacteria were removed by PBS washing. As shown in Figure 4, Ad-DEFB4 inhibited NTHi adhesion to the HMEEC cells compared with Ad-null, indicating the functional activity of human β-defensin 2 induced by Ad-DEFB4 infection.
Ad-DEFB4 inhibits NTHi adhesion to the human middle ear epithelial cells. Confocal microscopic images show that NTHi (green) attaches more to the HMEEC cells infected with (A) Ad-null compared with (B) Ad-DEFB4. Blue: nucleus. Red: actin. Original magnification × 630. Attached NTHi was quantitatively analyzed by (C) direct counting of GFP-positive NTHi and (D) measuring of fluorescence intensity. *P < 0.05.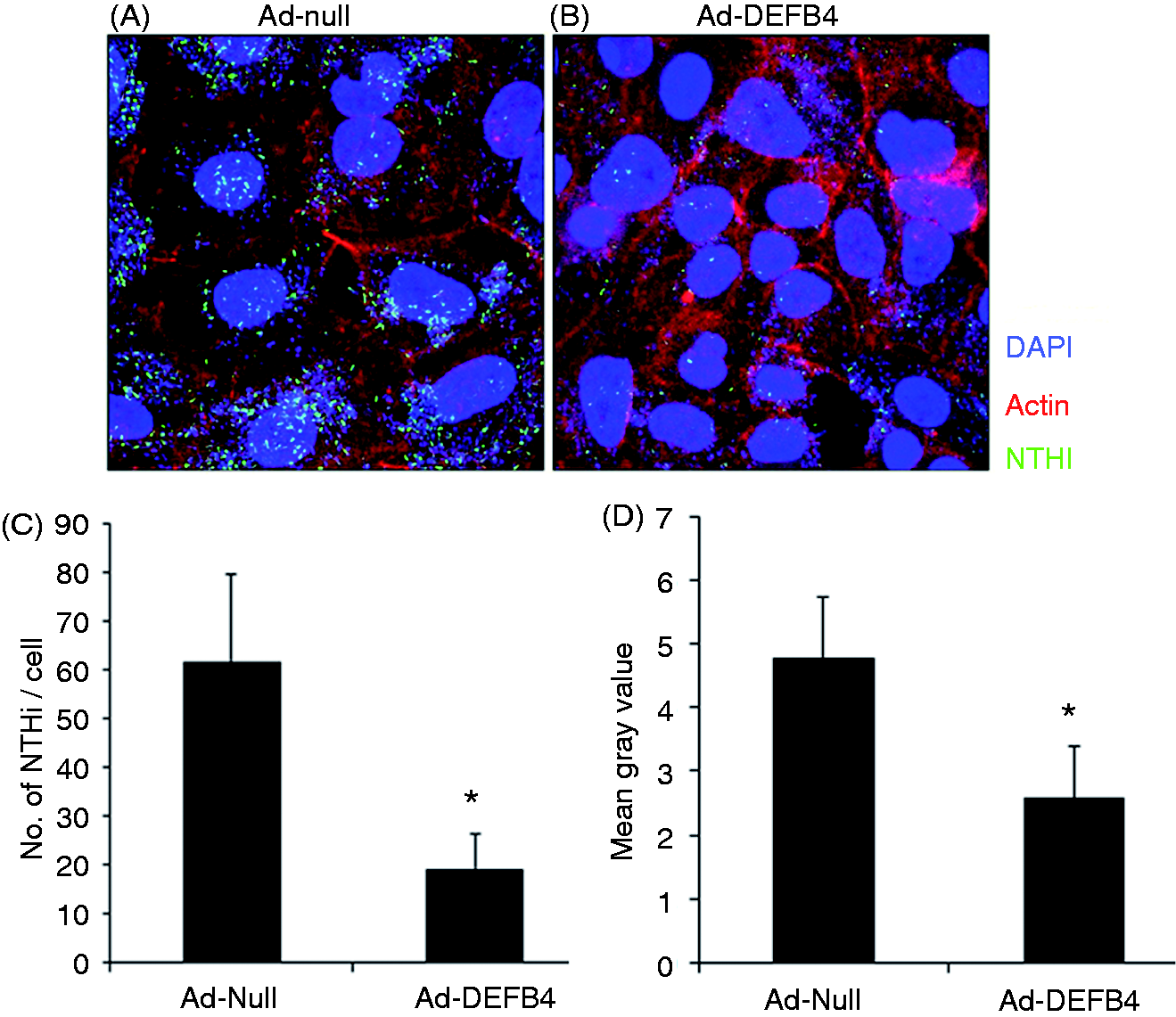
Adenovirus is able to deliver a gene to the middle ear epithelium without inflammatory reactions
As viral vectors are known to frequently elicit immune responses in the host,
45
we sought to determine if an adenoviral vector induces middle ear inflammation in vivo. H&E staining showed that middle ear inflammation is not induced by intratympanic inoculation of either Ad-null or Ad-DEFB4 (Figure 5A). To determine adenoviral vector-mediated gene delivery to the middle ear epithelium in vivo, 5 × 107 p.u. of Ad-βgal or Ad-null were intratympanically inoculated to the mouse middle ear cavity. The middle ear epithelial cells infected with Ad-βgal showed higher β-galactosidase activity compared with the endogenous β-galactosidase activity of the Ad-null-infected cells (Figure 5B,C). Taken together, we demonstrated the feasibility and utility of an adenoviral vector-mediated intratympanic gene delivery model.
Adenoviral vectors are able to deliver a gene to the middle ear epithelium without inflammatory reactions. (A) H&E staining shows that intratympanic injection of adenoviral vectors (Ad-null and Ad-DEFB4) does not induce inflammation in the mouse middle ear cavity. Saline: a negative control. M: middle ear cavity. Original magnification ×50. (B) X-Gal staining of the middle ear mucosa and (C) densitometric analysis show that β-galactosidase activity is up-regulated in the middle ear epithelium of the mice by intratympanic injection of the adenoviral vector expressing β-galactosidase (Ad-βgal) 48 h after injection compared with the endogenous activity of β-galactosidase in the Ad-null-infected mouse. *P < 0.05. #Bony capsule of the cochlea.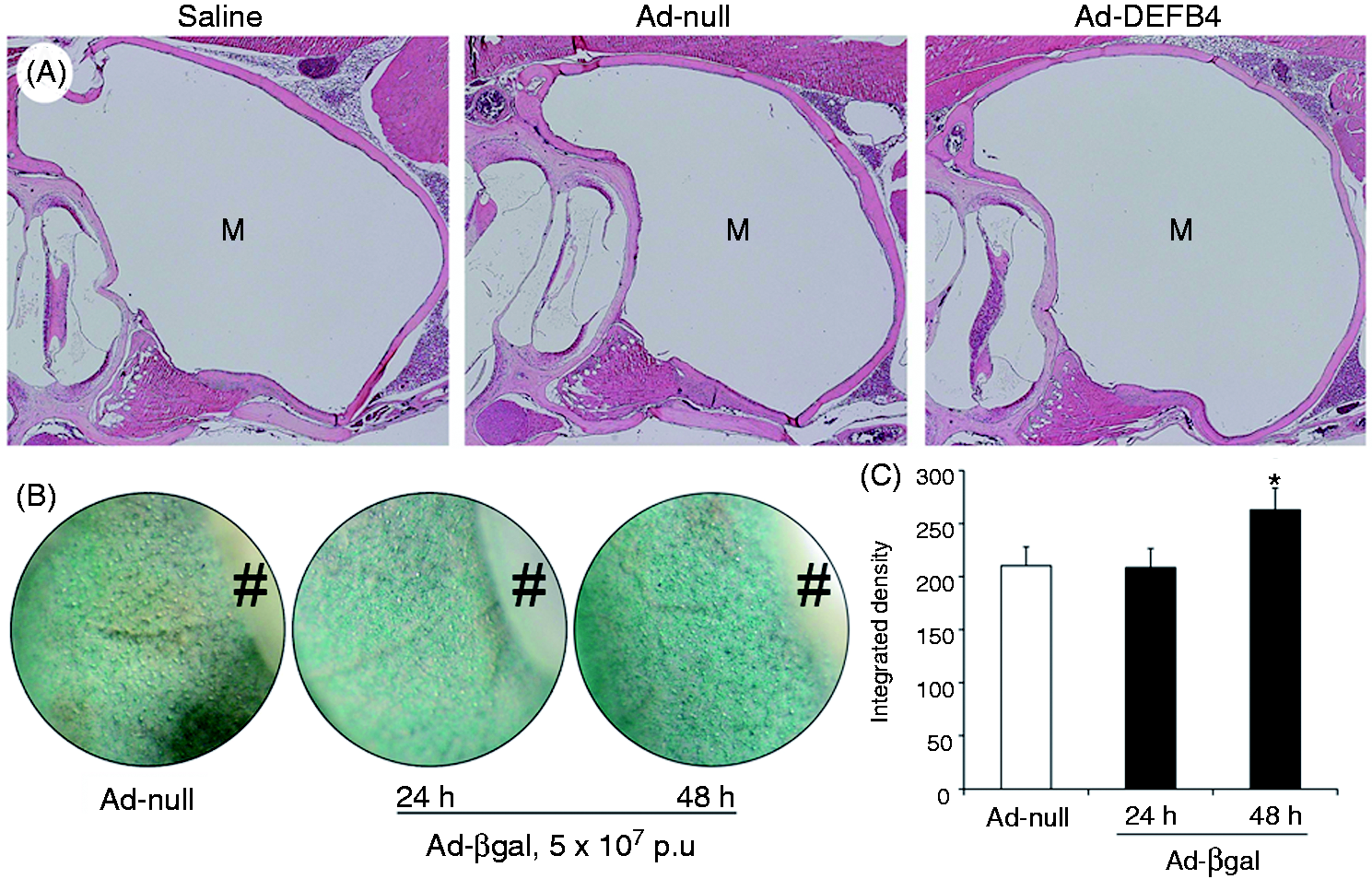
Intratympanic injection of Ad-DEFB4 ameliorates experimental OM
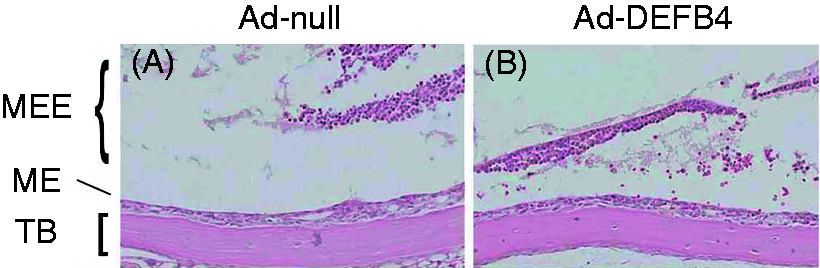
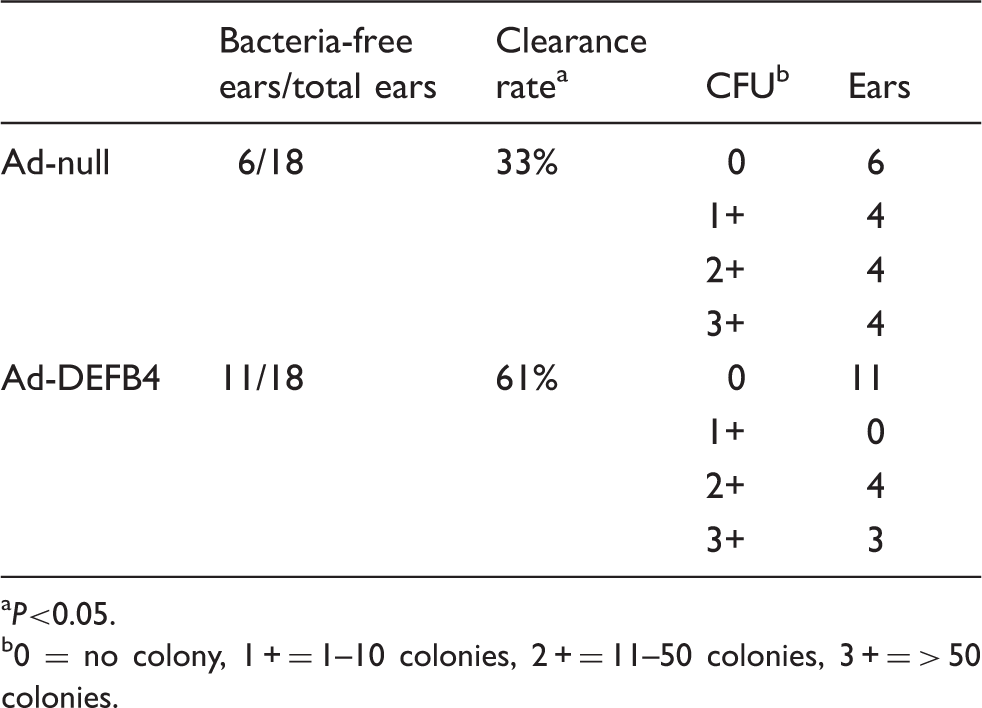
Clinical manifestations of OM include fever, otalgia and middle ear effusions. In experimental OM in a mouse model, intratympanic injection of live NTHi led to a range of middle ear effusions, which were graded as mild, moderate and severe effusions (Figure 6). To determine whether over-expression of human β-defensin 2 by Ad-DEFB4 can reduce the incidence and severity of middle ear effusions, Ad-DEFB4 or Ad-null were intratympanically inoculated with live NTHi into the mouse middle ear cavity. As shown in Table 1, 16/24 (67%) ears in the mice inoculated with Ad-null vector exhibited high-grade effusion (moderate and severe effusions). In contrast, 10/24 (44%) ears in the mice inoculated with the Ad-DEFB4 vector showed high-grade no effusions. Two of 24 (8%) ears had no effusion in the Ad-null-injected mice, while 4/24 (17%) ears had no effusions in the Ad-DEFB4-injected mice. Furthermore, severe effusions developed in six (25%) ears of the Ad-null-injected mice, but only in three (13%) ears of the Ad-DEFB4-injected mice. There was no statistical significance between Ad-DEFB4 and Ad-null when effusions were grouped into four subgroups. However, Ad-DEFB4 appeared to significantly attenuate middle ear effusions when effusions were divided into two groups. In addition, we performed histological analysis of the middle ear mucosa to determine an effect of Ad-DEFB4 on mucosal hyperplasia. As shown in Figure 7, Ad-DEFB4 seemed to inhibit NTHi-induced mucosal hyperplasia, but we failed to see a statistical significance. Together, these findings indicate that adenovirus-mediated β-defensin 2 over-expression attenuates both incidence and severity of NTHi-induced middle ear effusions.
NTHi-induced middle ear effusions. Intratympanic injection of live NTHi leads to a range of middle ear effusions: (A) mild effusion with a minimal amount of effusion; (B) moderate effusion with an air-fluid level (arrow) near a midline between the bony E-tube (*) and the posterior margin of the bulla; and (C) severe effusion with two air-fluid levels or an air bubble. #Tympanic membrane and external auditory canal. Original magnification ×50. Ad-DEFB4 insignificantly affects mucosal hyperplasia of the middle ear. (A, B) H&E staining shows NTHi-induced mucosal hyperplasia is not attenuated by infection of Ad-DEFB4 compared with Ad-null. TB: temporal bone; MEE: middle ear effusion. Original magnification ×100. Effects of intratympanic injection of Ad-DEFB4 on NTHi-induced middle ear effusions.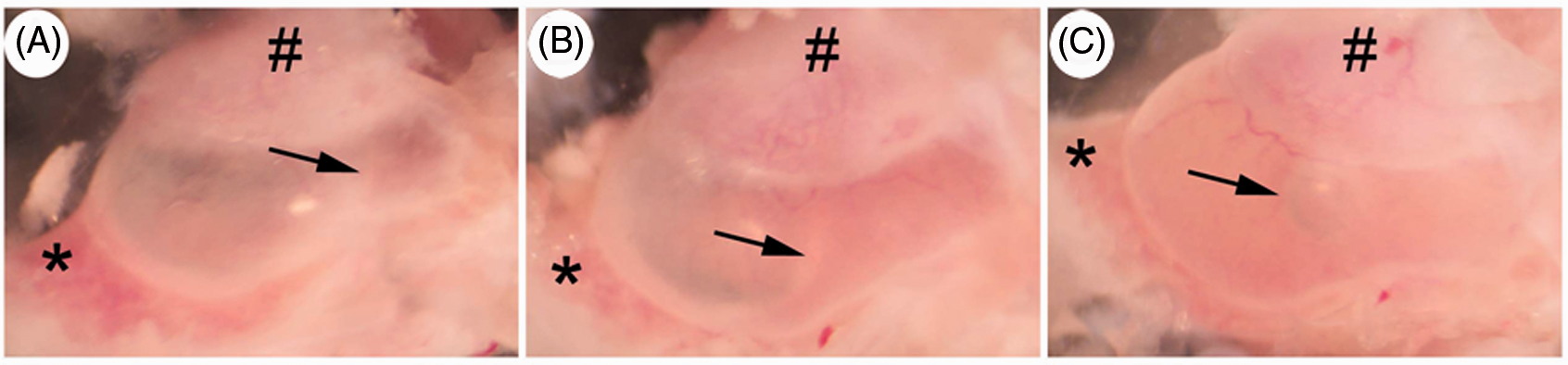

Effects of intratympanic injection of Ad-DEFB4 on NTHi clearance from the middle ear cavity.
P<0.05.
0 = no colony, 1 + = 1–10 colonies, 2 + = 11–50 colonies, 3 + = > 50 colonies.
Discussion
In this study, we have demonstrated that adenoviral vector-mediated gene delivery is effective for local over-expression of human β-defensin 2 in the middle ear cavity, resulting in significant amelioration of experimental OM. Human β-defensin 2 was up-regulated in HMEEC infected with Ad-DEFB4, which suppressed adhesion of a common OM pathogen, NTHi. Ad-DEFB4 treatment also reduced the severity and incidence of experimental OM with effusions. Most importantly, we found that Ad-DEFB4 treatment enhances NTHi clearance from the middle ear cavity without eliciting an immune response to the adenoviral vector. Hence, we have shown that adenoviral vector-mediated over-expression of antimicrobial peptides may serve as a non-antibiotic approach to manage OM.
Although Ad-DEFB4 appeared to ameliorate middle ear effusions and enhance NTHi clearance, we failed to show statistical significance in Ad-DEFB4-mediated attenuation of middle ear mucosal thickening. In a mouse model, intratympanic injection of NTHi is known to induce middle ear mucosal thickening, which is prolonged in mice lacking MyD88, a common adaptor of TLRs. 22 As our previous study showed that inhibition of MyD88 suppresses NTHi-induced DEFB4 up-regulation, 18 we anticipated that Ad-DEFB4 would attenuate mucosal thickening. To determine an effect of Ad-DEFB4 on mucosal thickening, further studies are necessary, with consideration of various factors such as duration of infection, dosage of Ad-DEFB4 and CFU of NTHi.
There exists a large amount of diverse antimicrobial molecules in the tubotymapanum, 9 which may be associated with self-resolution of acute OM without antibiotic treatment. Among antimicrobial molecules, defensins, small cationic antimicrobial peptides, target anionic lipids of bacterial membrane and disrupt lipid bilayer structure. 46 Mammalian cells are relatively resistant to defensins because the mammalian cell membrane is mainly composed of electrically neutral phospholipids, such as phosphatidyl choline. 46 Previously, we demonstrated antimicrobial activities of defensins against common OM pathogens, 13 but there is evidence demonstrating bacterial countermeasures to defensins. 47 In the case of NTHi, acylation of lipooligosaccharides decreases susceptibility of NTHi to β-defensin 2, 48 and the products of the ‘sap’ (sensitivity to antimicrobial peptides) operon of NTHi play an important role in resistance to defensins. 49 However, it is unlikely that bacteria have generally developed effective resistance to defensins as the essential structures for survival and virulence of pathogens are usually targeted, which explains why defensins have a long history in the animal kingdom as a highly effective antimicrobial molecule.
Although recombinant human β-defensin 2 appeared to significantly improve NTHi clearance from the middle ear cavity, there are many challenges to a therapeutic application of peptide/protein drugs, such as difficulties in the entry of hydrophilic proteins into cellular compartments, 25 and the negative impacts of protein degradation and clearance on half-life and effectiveness. 26 In addition, it takes time and effort to produce a peptide/protein that essentially requires post-translational modifications such as glycosylation, phosphorylation and proteolytic cleavage. 27 Furthermore, our results indicate that a high dose of human β-defensin 2 is required for effective enhancement of NTHi clearance, but the potential cost of production should be considered. For example, the cost of producing a sufficient amount of recombinant β-glucocerebrosidase to treat Gaucher’s disease is estimated to be >$100,000 per patient annually in USA. 50 Besides the high cost, high concentrations β-defensin 2 may synergistically act on host cytotoxicity of β-defensin 2 through an increase in the local oncotic pressure of the middle ear mucosa. Furthermore, ionic concentrations in a working solution of recombinant defensins should carefully be adjusted because an antimicrobial activity of defensins is highly sensitive to a salt environment. 51
In contrast, gene therapy enables bioactive peptides/proteins to be stably produced for a relatively long period with minimal cost; however, there are many limitations to be overcome for successful clinical application. In particular, the difficulty in delivery of negatively charged large DNAs into the cells without degradation remains challenging. 52 Ideal vehicles for gene delivery are able to remain in the bloodstream or tissue fluids without being removed by the phagocytic system, and efficiently transport DNAs into the cytoplasm without lysosomal degradation. 53 Viral vectors are useful for gene delivery, but their clinical applications remain limited owing to the potential for incoporation into host chromosomes 54 and triggering of immunological reactions.55,56
Among viral vectors, adenoviral vectors are capable of infecting a wide variety of cell types. 28 Although early generations of the adenoviral vector were highly immunogenic, 55 the current generations are able to deliver genes without significant inflammatory reactions.29–31 In this study, we have shown that intratympanic injection of an Ad5-based vector with deletions in the E1, E3 and E4 genes 35 rarely induces middle ear inflammation. Moreover, as adenoviral vectors are rapidly cleared from the host without chromosomal incorporation, they are not adequate for chronic or genetic disorders, 28 but are useful for acute diseases, such as acute OM.
In conclusion, we suggest that local gene delivery of human β-defensin 2 is able to significantly ameliorate experimental OM and augment bacterial clearance from the middle ear. Combined with technological advances in the safety and effectiveness of gene delivery, defensins are excellent potential therapeutic agents that can serve as an alternative or adjuvant therapy to conventional antibiotics to fight against infectious diseases in the future, particularly considering a global increase in drug-resistant pathogens.
Footnotes
Funding
This work was supported, in part, by National Institutes of Health grants DC005025 and DC011862.
Conflict of interest
Jeong-Im Woo, Sung-Hee Kil, Yoo Jin Lee, David J Lim, and Sung K Moon have no potential conflicts of interest to declare. Dr. Douglas E Brough works for GenVec Inc., Rockville, MD, USA. He constructed and provided Ad-null, Ad-EGFP, Ad-βgal and Ad-DEFB4, but was not involved in other experiments conducted as part of this study.
Acknowledgements
We would like to thank Drs. Xin-Xing Gu (NIH) and Lauren Bakaletz (Ohio State University) for providing the bacterial strains used in this study.
