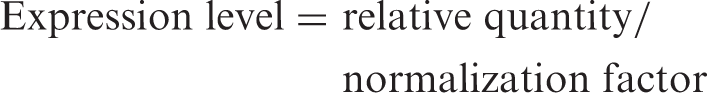Abstract
The aims of this study were to investigate OX40 ligand expression in sinus tissue from patients with nasal polyposis compared with patients with chronic rhinosinusitis without nasal polyps (NPs), and to determine if OX40 ligand expression is related to eosinophilic sinus infiltration. Twenty patients with chronic rhinosinusitis (11 with and nine without NPs) and seven controls were enrolled in the study. The mRNA expression of OX40 ligand and thymic stromal lymphopoietin and its receptor were analyzed. The immunoreactivity score for OX40 ligand and the eosinophil count were obtained. The mRNA expression and immunoreactivity score of OX40 ligand were higher in patients with nasal polyposis than in patients without NPs, as well as healthy controls. The mRNA expression of thymic stromal lymphopoietin and its receptor was significantly higher in nasal polyposis than in the control, but not significantly higher than in chronic rhinosinusitis without NPs. A correlation between the number of OX40 ligand-positive cells and the number of eosinophils in sinus biopsies was found only in patients with nasal polyposis. In conclusion, the thymic stromal lymphopoietin/OX40 ligand axis is up-regulated in nasal polyposis and is related to the intensity of eosinophilic inflammation.
Introduction
Chronic rhinosinusitis without (CRSsNP) and with nasal polyps (CRSwNP) are recognized as separate disease entities based on inflammatory and remodeling profiles. 1 In the vast majority of patients, CRSsNP is characterized by a Th1-skewed inflammatory response, whereas the inflammatory response in the CRSwNP group is rather heterogeneous, with the Th2-biased phenotype being more frequent in Caucasian patients.2,3 Recently, the concept that the epithelial barrier and altered innate immunity are fundamental to the onset of a TH2-inflammatory response has been supported by the finding that thymic stromal lymphopoietin (TSLP) represents a key molecule at the epithelial cell‐dendritic cell (DC) interface to initiate allergic inflammation. 4 Human airway epithelial cells are triggered to produce TSLP upon exposure to allergens, viral infections and pro‐inflammatory cytokines, such as TNF‐α and IL-1β. TSLP is involved in the innate immune response through the promotion of cytokine secretion by mast cells, basophils, eosinophils and macrophages. TSLP also results in the polarization of DCs, which drives the differentiation of naïve T-cells to inflammatory Th2 cells through the up-regulation of co-stimulatory molecules (CD40, CD80), MHC II molecules and OX40 ligand (OX40L).5,6 OX40L and its receptor (OX40) belong to the superfamily of TNF/TNF receptor proteins. 7 OX40L is preferentially expressed on APC, such as myeloid and plasmacytoid DCs and B-cells, and may also be expressed on vascular endothelial cells, 8 mast cells 9 and NK cells. 10 According to previous observations, the OX40/OX40L interaction is involved in Th2-associated inflammatory diseases, particularly allergic inflammation, 11 where it is crucial for T-cell activation and polarization towards a Th2 inflammatory pattern.
TSLP activates immature DCs to promote the expression of OX40L and to drive the differentiation of naïve CD4+ T-cells into Th2 inflammatory cells that produce IL-4, IL-5, IL-13 and TNF-α. 4 The expression level of TSLP in the nasal epithelial layer is higher in patients with NPs than in patients without NPs. 12 Higher expression of TSLP receptor (TSLPR) and OX40L were also detected in DCs of nasal mucosa of patients with NPs. 13 Recently, TSLPR expression has been found to be significantly higher not only in the epithelial cells, but also in the inflammatory cells isolated through sinus biopsies from patients with CRS compared with the control group, with no difference between CRSwNP and CRSsNP patients. 14 This suggests that the activation of the innate immune response, which promotes TSLP-R expression, is not specific to eosinophilic inflammation, which is only found in CRSwNP. This is not surprising as TSLPR expression may be induced by a variety of triggers (allergens, viral infection and pro-inflammatory cytokines, such as TNF-α and IL-1β), which may also be present in CRSsNP. 15 As the OX40/OX40L interaction plays a major role in promoting Th2 polarization and eosinophilic inflammation‐the hallmark of CRSwNP‐we investigated OX40L expression in human sinus tissue from patients with CRSwNP and CRSsNP. We hypothesized that OX40L expression is increased in CRSwNP and that this expression is related to eosinophilic tissue infiltration. To test our hypothesis, we evaluated OX40L and eosinophilic infiltration in sinus biopsies from patients with CRSwNP compared with CRSsNP and healthy controls.
Materials and methods
Patients
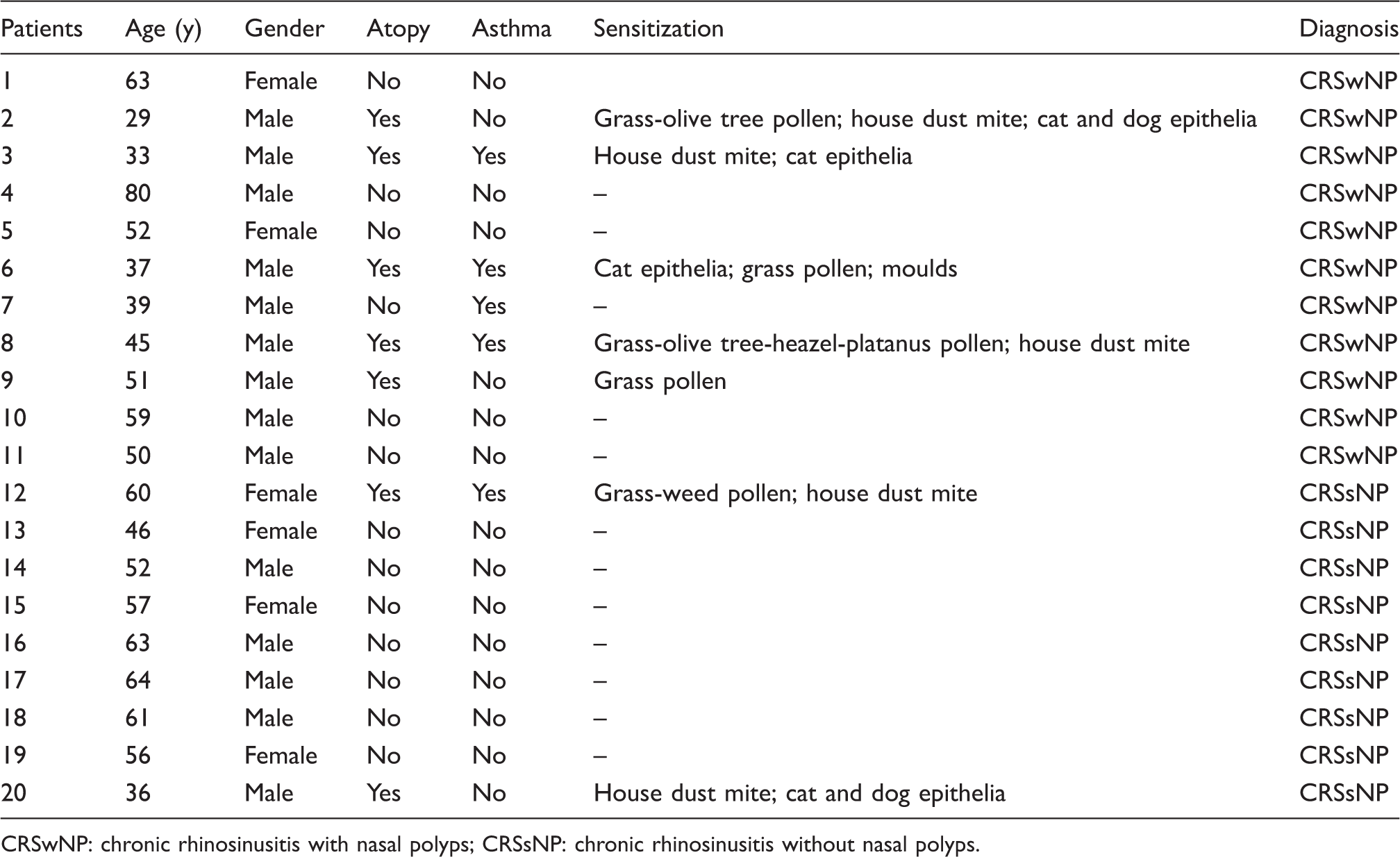
Twenty consecutive Caucasian patients (14 men, six women) with a mean age of 52.84 ± 12.63 y (range 29–80 y) recruited for functional endoscopic sinus surgery at the 1st ENT Division of the University of Turin-Italy were enrolled in this study after approval by the local ethics committee and after written informed consent was obtained. All of the patients were affected by CRSsNP (n = 11) or CRSsNP (n = 9). Diagnosis of CRSwNP and CRSsNP was based on symptoms, fiber optical examination and sinus computed tomography scan, according to the European Position Paper on Rhinosinusitis 2007 criteria. 16
Skin prick tests for a panel of 14 inhalant standard allergens (ALK Abello’, Hørsholm, Denmark) were performed in all patients. Atopy was defined as a wheal diameter of 3 mm or greater in the presence of expected results in control solutions (histamine dihydrochloride 20 mg/ml as a positive and solvent as a negative control). Measurement of specific IgE Abs against common allergens (CAP-Phadia, Uppsala, Sweden) was performed in patients if the skin prick test could not be performed.
All patients were questioned about lower respiratory symptoms, and the diagnosis of asthma was based on intermittent experience of a combination of symptoms, including wheezing, cough, shortness of breath and chest tightness, which were relieved by inhaled β2-agonists, together with documented reversible airway obstruction (FEV1 increase more than 12% of baseline after inhaled salbutamol 200 + 200 µg) and/or bronchial hyper-responsiveness (methacholine PD20FEV1 < 800 µg). None of the patients used medications of any type 2 wk prior to surgery.
Seven non-atopic patients (four men, three women) with a mean age of 48 ± 10 y (range 32–62 y), who underwent to cosmetic rhinoplasty surgery served as the control group.
Sinus biopsies
Biopsy specimens were collected from NPs in patients with CRSwNP and from the ostio-meatal complex mucosa in patients without NPs and controls. For real-time PCR, all of the specimens were immediately placed in RNA Later (Ambion, Grand Island, NY, USA) at –20℃ to preserve RNA quality. To extract RNA, samples were thawed, washed with PBS and further processed as described below. For immunohistochemistry, all specimens were immediately placed in a 10% buffered formalin solution, and then routinely processed and embedded in paraffin.
Eosinophils and cell counting
Hematoxylin and eosin staining was used to identify eosinophils in tissue sections. Healthy sinus mucosal samples were used as the control group and analyzed as described for the CRS specimens. The number of eosinophils and inflammatory cells was obtained after five randomly selected visual fields were counted at a magnification of 400 × using an objective micrometer with a diameter of 0.54 mm (area = 0.229 mm2). The average of the five fields was calculated and used as the final count for each section.
Immunohistochemistry
Slices that were 4 µm thick were deparaffinized with xylene and rehydrated in a series of graded alcohols. For the detection of OX40L, CD3, CD20 and CD68-PGM1, heat-induced epitope retrieval was performed using Target Retrieval Solution (Dako, Carpinteria, CA, USA) in an electric pressure-cooker for 20 min at 120℃. Heat-induced epitope retrieval was not performed for mast cell Tryptase staining. Endogenous peroxidase was blocked with 3% H2O2 for 25 min, and tissue sections were then incubated overnight (about 15 h) at 4℃ with primary AbS. The primary Abs used in these experiments were monoclonal mouse anti-human OX40L (clone 159403; dilution 1:27; R&D Systems, Abingdon, UK); monoclonal mouse anti-human CD3 (clone PS1; dilution 1:50; Novocastra, Leica Biosystems); monoclonal mouse anti-human CD20 (clone L26; dilution 1:200; Dako); monoclonal mouse anti-human CD68-PGM1 (dilution 1:50; Dako); and monoclonal mouse anti-human Mast Cell Tryptase (clone 10D11; dilution 1:150; Novocastra, Leica Biosystems, Milano, Italy). Sections were incubated with FBS as a negative control. The sections were subsequently incubated with biotinylated secondary Abs rabbit-anti mouse IgG (H + L), dilution 1:2000; Sigma Pharmaceutical, South Croydon, Australia) and then incubated with avidin–biotin peroxidase complex solution (Dako). Diaminobenzidine tetrahydrochloride was used as a chromogen. Slides were then counterstained with Mayer’s hematoxylin for 5 s, dehydrated and mounted in Clarion (Biomeda, Foster City, CA, USA). The evaluation of immunostaining for all of the Abs was performed in a coded manner by three independent observers (BV, JM, MB) without knowledge of the pathological parameters. To confirm reproducibility, 25% of the slides were chosen at random and scored twice. All duplicates were similarly evaluated. OX40L expression was interpreted in five selected visual fields using the objective micrometer with the diameter 0.54 mm at a magnification of 400 × (area = 0.229 mm2). After detecting the percentage of positive cells, the staining intensity for positive cells was scored as 0 (negative), 1 (weak), 2 (medium) and 3 (strong). The immunoreactivity score (IRS) was determined by multiplying the staining intensity score by the percentage of positive cells (intensity × positive cells: maximum IRS 300).
Real-time PCR analysis for mRNA expression of TSLP, OX40L and TSLPR
Total RNA was extracted after sample lysis in Trizol Reagent (Invitrogen Life Technologies, Monza, Italy). RNA samples were reverse transcribed at 42℃ with the Reverse Transcription System (Promega, Madison, WI, USA) according to the manufacturer’s instructions, and cDNA was used for real-time PCR. The quantitative real-time PCR assay was performed using primers that specifically amplify TSLP, OX40L and TSLPR. The primers and fluorogenic probes for TSLP (Hs00263639_m1), OX40L (Hs00182411_m1), TSLPR (Hs00171074_m1), G6PD (Hs00188728_m1) and β-actin (Hs99999903_m1) were purchased from Applied Biosystems Life Technologies (Italia, Monza, Italy). RT-PCR was performed using a BioRad iQ iCycler (BioRad, Hercules, CA, USA) and analyzed with the Gene Expression Analysis for iCycler iQ Real Time PCR Detection System (BioRad).
The amplification efficiency of the specific primers, β-actin and G6PD were validated in a preliminary experiment. For PCR analysis, each sample was run in duplicate in separate tubes. The results were normalized using two different housekeeping genes (β-actin and G6PD) and the mRNA expression level was reported as arbitrary units, calculated, according to Vandesompele et al.,
17
using the following equation:
Statistics
All statistical analyses were carried out using GraphPad Prism 4. For normal distribution data, one-way ANOVA was performed followed by Dunn's Multiple Comparison Test; the Kruskal‐Wallis test was performed for non-parametric distribution. All tests were two-tailed, and a probability value of P < 0.05 was considered statistically significant.
Results
Demographic and clinical characteristics of patients.
CRSwNP: chronic rhinosinusitis with nasal polyps; CRSsNP: chronic rhinosinusitis without nasal polyps.
Immunohistochemical characterization of inflammatory infiltrate
The number of eosinophils, as detected by immunohistochemical staining with hematoxylin and eosin, was significantly higher in CRSwNP compared with CRSsNP and controls [CRSwNP, 216.6 ± 49.4/high powered fields (HPF); CRSsNP 13.2 ± 4.2/HPF; controls 5.6 ± 1.06 P < 0.001] (Figure 1A). The number of inflammatory cells positive for CD3, CD68-PGM1 and tryptase were similar between the CRSsNP and CRSwNP groups (Figure 2).
Individual and mean values of eosinophils (A), OX40L IRS (B) and OX40L-positive cells in the lamina propria (C) in chronic rhinosinusitis with nasal polyps (CRSwNP), chronic rhinosinusitis without nasal polyps (CRSsNP) and controls (*P < 0.05, **P < 0.01, ***P < 0.001). Heavy eosinophilic infiltration [hematoxylin and eosin (H&E)], with no difference in lymphocytes (CD3+ ), mast cells (tryptase + ) and DCs (CD68–PGM1+) may be appreciated in sinus biopsies of a patient with chronic rhinosinusitis with nasal polyps (CRSwNP) (left panels) compared with chronic rhinosinusitis without nasal polyps (CRSsNP) (right panels). Original magnification 200× (inset 400×).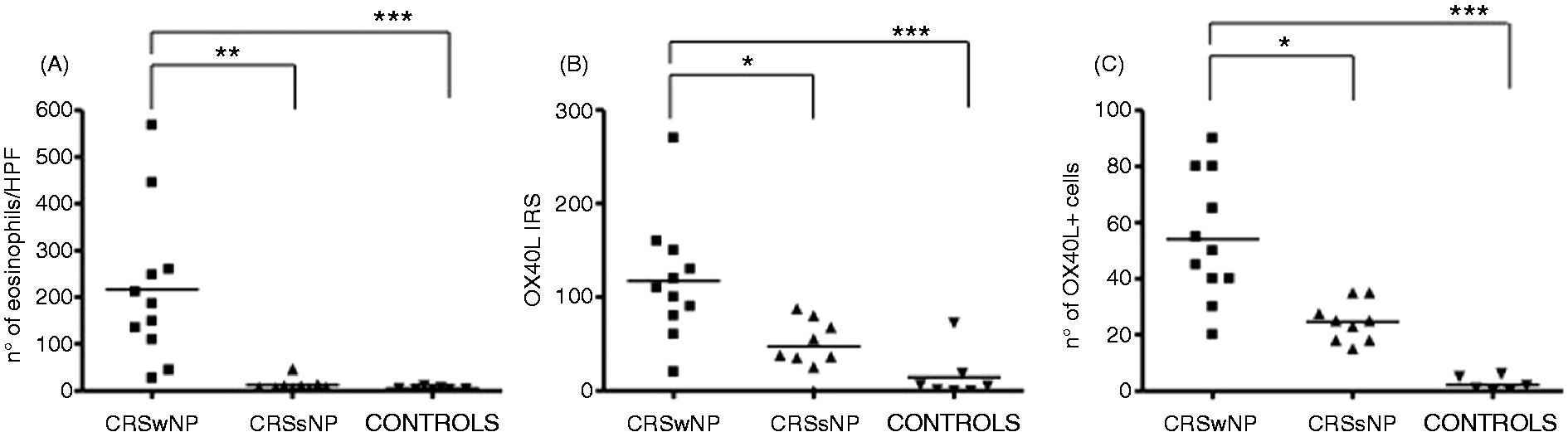
Expression of OX40L as evaluated by immunohistochemistry
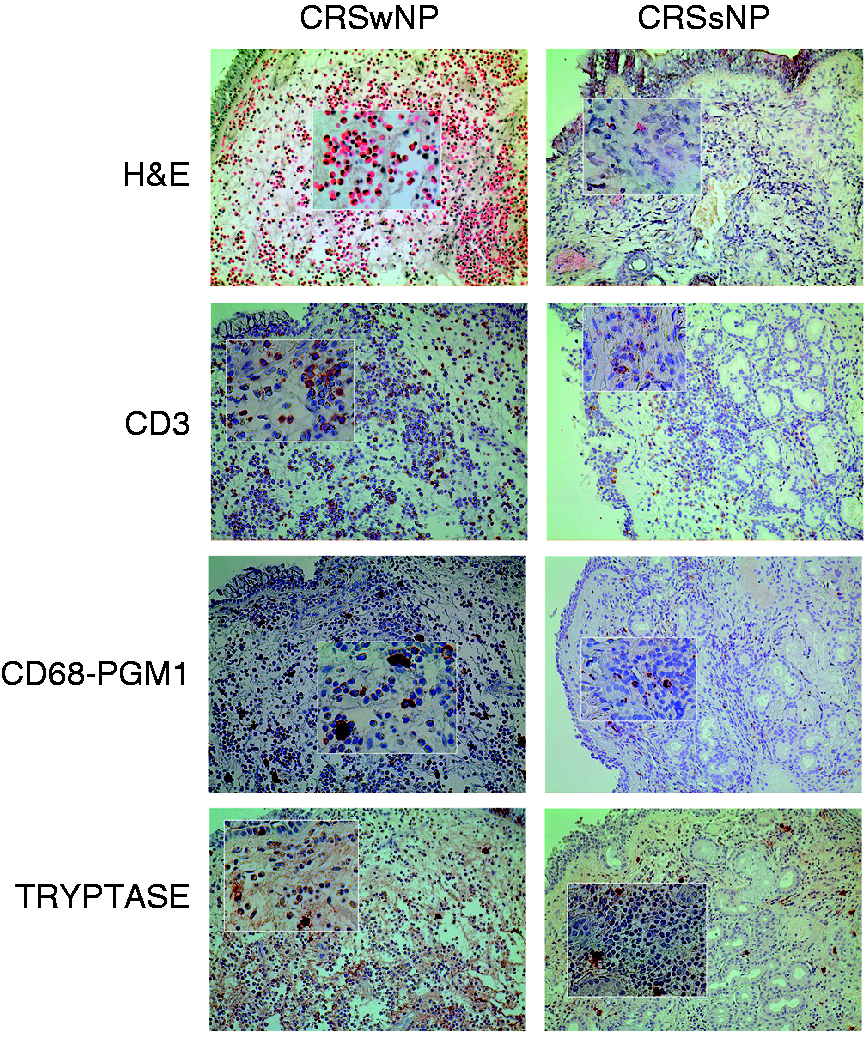
OX40L expression was observed in many inflammatory cells of the stromal compartment, which included eosinophils, macrophages and DC, CD3-positive T-cells, and mast cells. OX40L expression was detected to a lesser extent in epithelial cells (Figure 3). The immunoreactivity score for OX40L was significantly higher in CRSwNP than in CRSsNP (P < 0.05) and controls (P < 0.001) (CRSwNP 117.3 ± 19.4, CRSsNP 47 ± 9.3, controls 14.5 ± 9.8) (Figure 1B). The mean (SD) number of OX40L-positive cells in the lamina propria was significantly increased in patients with CRSwNP (54.09 ± 22.34) compared with CRSsNP (24.61 ± 7.13, P < 0.05) and controls (2.3 ± 2.58, P < 0.001) (Figure 1C). The immunoexpression of OX40L in sinus biopsies obtained from one patient with CRSsNP and one patient with CRSwNP is reported in Figure 3.
Immunohistochemical expression of OX40L in control specimen (A) in one patient with chronic rhinosinusitis with nasal polyps (CRSwNP) (patient #2) (B) and in one patient with chronic rhinosinusitis without nasal polyps (CRSsNP) (patient #13) (C) at original magnification 200× (inset 400×).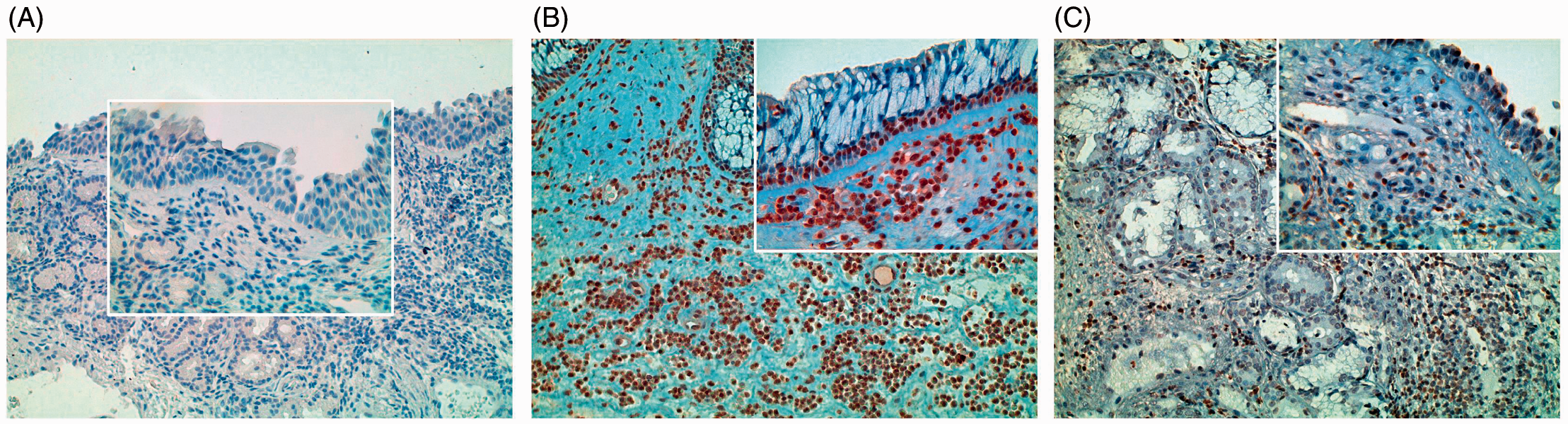
Correlation between OX40L-positive cells and eosinophils
There was a statistically significant correlation between OX40L-positive cells and the number of eosinophils in sinus biopsies of patients with CRSwNP (r = 0.605, P = 0.048), but not in patients with CRSsNP (Figure 4).
(A) Correlation between OX40L+ cells and eosinophils in chronic rhinosinusitis with nasal polyps (CRSwNP) group. (B) Relationship between OX40L+ cells and eosinophils in chronic rhinosinusitis without nasal polyps (CRSsNP) group.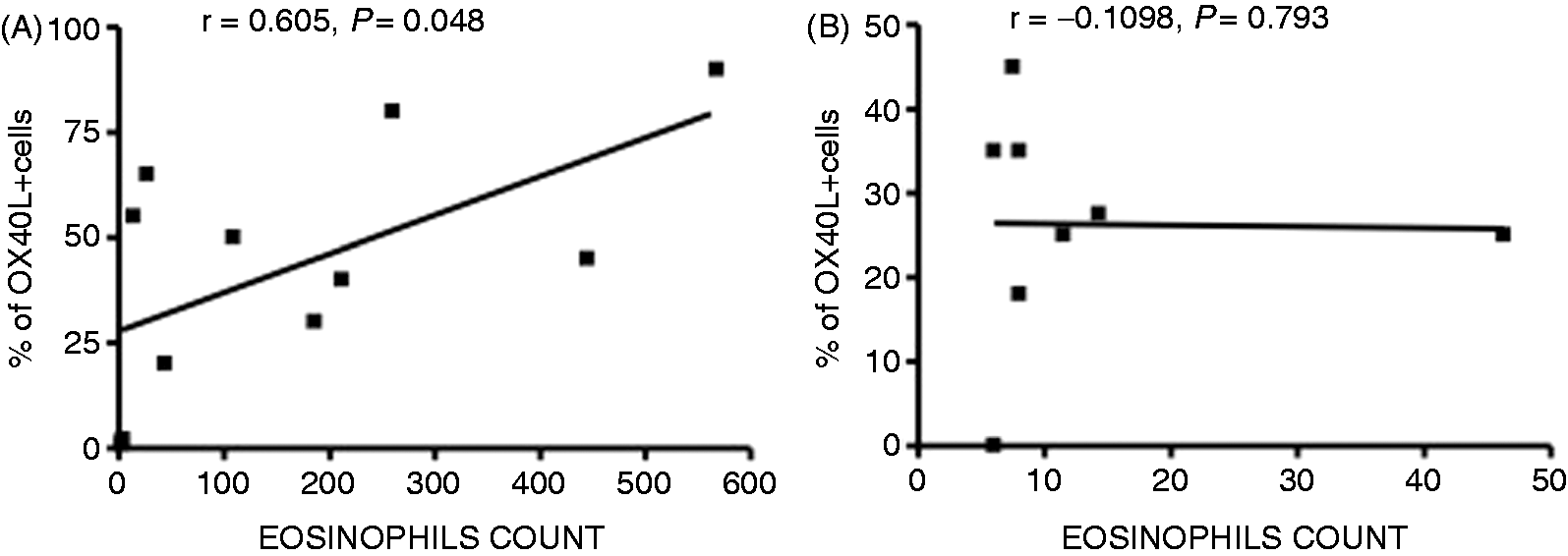
mRNA expression of OX40L, TSLP and TSLPR in patients with CRS and in controls
The mRNA expression of OX40L was higher in CRSwNP (2.4 ± 0.26) than in CRSsNP (1.38 ± 0.19, P < 0.05) and controls (0.49 ± 0.14, P < 0.001). The mRNA expression of TSLP and TSLPR was significantly higher in CRSwNP than in controls (2.5 ± 0.32 versus 0.98 ± 0.17, P < 0.01, and 1.42 ± 0.31 versus 0.25 ± 0.07, P < 0.001, respectively), but not significantly higher compared with CRSsNP (2.5 ± 0.32 versus 1.94 ± 0.25, P > 0.05, and 1.42 ± 0.31 versus 0.87 ± 0.24, P > 0.05, respectively) (Figure 5).
OX40L (A), TSLP (B) and TSLPR (C) mRNA expression in chronic rhinosinusitis with nasal polyps (CRSwNP), chronic rhinosinusitis without nasal polyps (CRSsNP) and controls (*P < 0.05, **P < 0.01, ***P < 0.001).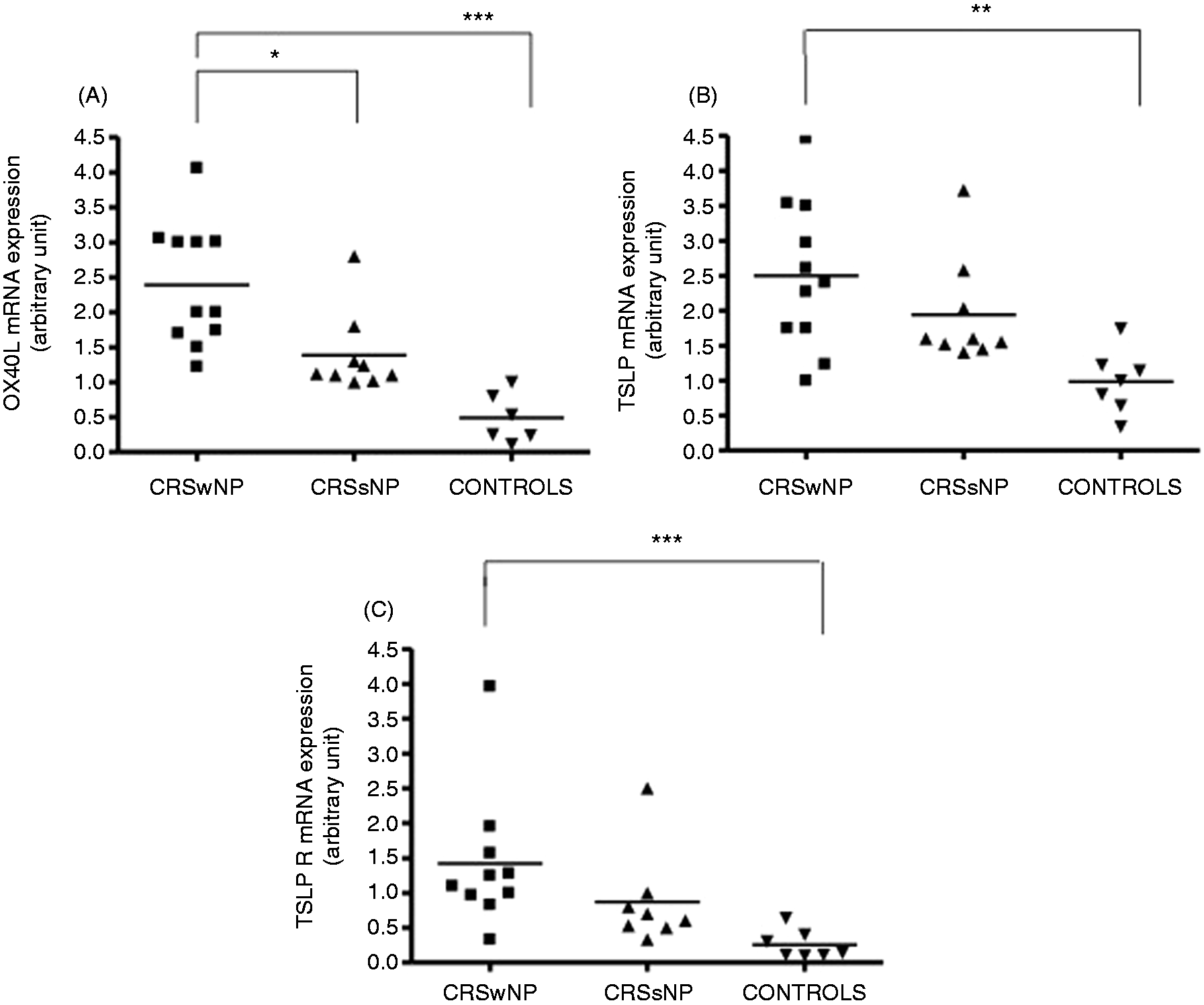
IRS and mRNA OX40L expression in atopic and non-atopic patients
Neither immunoreactivity nor mRNA expression of OX40L were significantly different between allergic patients and non-allergic patients (85 ± 14.7 versus 86 ± 20.13, P > 0.05 for IRS; 1.88 ± 0.47 versus 1.93 ± 0.42, P > 0.05 for mRNA).
Discussion
We report, for the first time, that the expression of OX40L, as determined by immunohistochemistry and real-time PCR, is increased in the lamina propria of sinus tissue of patients with CRSwNP and correlates significantly with eosinophilic infiltration. The expression of OX40L in DCs is promoted by TSLP, an IL-7-like cytokine produced by epithelial cells, and provides an important interaction between the epithelial and DCs. 18 Numerous OX40L-positive cells were identified in the stromal tissue of CRSwNP, and it is likely that other cells also express OX40L in addition to DCs. Growing evidence suggests that OX40L is also present on activated macrophages, mononuclear cell, mast cells and granulocytes,19,20 all of which were observed in patient biopsies from this study. Data indicating that TSLP and OX40L expression is increased in CRSwNP provide further evidence for a potential role of this axis in CRS. In mouse models of allergic inflammation, genetic deletion of OX40 results in the significant reduction of a Th2 response to allergens and a reduction in airway eosinophilia. 21 Critically, OX40L neutralization in murine models of allergic inflammation inhibited TSLP-induced immune responses, including Th2 inflammatory cell infiltration. 22 Consistent with these observations, our findings showed a close association between the number of eosinophils and OX40L expression in the lamina propria. The number of eosinophils in NPs has been previously reported to correlate with the number of TSLP+ cells. 12 All of these observations support the hypothesis that TSLP may have a major role in regulating eosinophilic inflammation in the NPs, driving Th2-type inflammation that subsequently leads to the expression of eosinophilic chemoattractants. The number of OX40/OX40L cells in the lamina propria of airways is associated with eosinophilic inflammation in patients with asthma, 23 suggesting common pathogenic mechanisms in CRSwNP and the eosinophilic phenotype of asthma.
We did not observe significant differences in the mRNA expression of TSLP and TSLPR between CRSwNP and CRSsNP, while the mRNA expression of OX40L was significantly increased only in CRSwNP and correlated with sinus eosinophilic infiltration. Although our findings related to TSLP/TSLPR are dependent on a small sample of patients, our results potentially suggest that the activation of an early inflammatory pathway, common to CRSwNP and CRSsNP, may originate from TSLP–TSLPR expression as a consequence of epithelial cell damage by various hits (allergens, viral infection, cytokines, etc.). Afterwards, the TSLP-OX40 axis plays a major role in promoting Th2 polarization and eosinophilic inflammation only in CRSwNP. Recent research of the biology of innate immunity, particularly that of the innate type-2 immune response has revealed an ever more complex and perplexing model of cellular interactions and effector functions. Cytokines produced by epithelial cells, such as IL-33 and IL-25, are able to activate innate cells, including eosinophils, basophils and mast cells, to regulate adaptive immunity by enhancing Th2 cytokine production and a subsequent eosinophilic inflammatory response. 21 Elucidation of early innate pathways involved in the induction of Th2- associated immune responses could help to establish novel therapeutic approaches for the treatment of Th2 inflammatory diseases in humans, 24 as the role of allergic sensitization in CRSwNP is controversial.
The prevalence of IgE-mediated allergy to environmental allergens in patients with CRS (both with and without NP) is estimated at 60% compared with 30–40% for the general population. 25 Histopathologic studies of ethmoidal tissue and NP tissue have demonstrated that allergic patients with CRS have chronic allergic inflammation with local T-cell infiltration and production of classic Th2 cytokines, including IL-4, IL-5 and IL-13.26,27
Despite these associations, the intensity of eosinophilic inflammation in patients with CRS with or without NP is independent of the presence of underlying systemic allergy.28,29 However, the number of TSLP+ cells in NPs from atopics has been reported to be significantly greater than that of non-atopics and in the allergic nasal mucosa. 12
The immunoreactivity score and mRNA expression of OX40L observed in this study were not significantly different between allergic patients compared to non-allergic patients. The TSLP–OX40–OX40L interaction is implicated in a number of inflammatory diseases in addition to allergic inflammation.30,31 Indeed, human airway epithelial cells are triggered to produce TSLP not only upon exposure to allergens, but also in response to viral infection and pro-inflammatory cytokines, such as TNF-α and IL-1β. Interestingly, OX40L expression by DCs is increased by exposure to environmental fungi 32 and endotoxins, 33 which have both been implicated in severe asthma exacerbations associated with chronic rhinosinusitis. In conclusion, we have shown that the TSLP/OX40L interaction is up-regulated in CRSwNP and that it is related to the intensity of eosinophilic inflammation. This observation confirms the Th2 bias of inflammatory response in CRSwNP. Our results suggest that TSLP/OX40L is involved in the pathogenesis of CRSwNP. Further investigation is required to determine the potential of these co-stimulatory molecules as novel therapeutic targets for the treatment of CRSwNP.
Footnotes
Funding
This research was not funded by a specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Acknowledgements
We thank Dr. Daniele Corino, 2nd service of Pathological Anatomy, Department of Molecular Biotechnology and Health Science, University of Turin, for important and professional help with immunohistochemistry techniques and interpretation.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.