Abstract
Burn induces an immunopathological response involving multiple immune cell types that includes γδ T-cells. Nonetheless, the role of γδ T-cells at the wound site after burn is not clearly defined. Wild type and γδ T-cell receptor deficient (δ TCR-/-) mice were subjected to a major burn or sham procedure. At 1–7 d thereafter, skin samples were collected and T-cell populations analyzed. The majority of T-cells in the skin of sham mice were γδ T-cells. After burn, however, an increase in the total T-cells was observed at the wound site and these cells were predominantly αβ T-cells. Their influx was γδ T-cell dependent, as it was markedly reduced in injured δ TCR-/- mice. Burn wound γδ T-cells were activated with increased expression of TLRs and CD69. In contrast, the infiltrating αβ T-cells TLR and CD69 expressions were attenuated after burn. Thus, burn is associated with of γδ T-cell activation at the injury site, which initiates a massive infiltration of the wound with αβ T-cells that likely facilitate the transition from the inflammatory to the proliferative phase of healing.
Introduction
Burn- and trauma-related morbidity and mortality can, in part, be attributed to immunoinflammatory derangements that contribute to the subsequent development of systemic inflammatory response (SIRS) and multiple organ failure (MOF).1–3 Nonetheless, inflammation has a beneficial role at times and plays a major role in the complex process of wound repair. The regulation and propagation of inflammatory responses involves multiple immune cell types (i.e. T-cells, macrophages and neutrophils) working in concert in a highly regulated way. T-cells constitute the most abundant leukocyte subset in human skin wounds4,5 and, in general, migrate into wounds. Their numbers peak during the late proliferative and early remodeling phases of wound healing. 5 A recent study in a murine model lacking both T- and B-cells has demonstrated that scar formation is attenuated in the absence of these lymphocytes. 6
Burn and other forms of trauma induce a generalized loss of T-cell reactivity that is associated with Th-1/Th-2 shift1,7–9; however, most burn studies have focused on αβ T-cells from the circulation or lymphoid tissues.8,10 Recent evidence suggests that γδ T-cells are central in the response to burn injury. 11 T-cells expressing the γδ T-cell receptor (TCR) normally represent a small percentage of cells in lymphoid tissues, but are abundant in the skin and other epithelial tissue beds. 12 Importantly, γδ T-cells differ from αβ T-cells with regard to tissue localization and Ag recognition, where γδ T-cells recognize Ags directly, without requiring any processing or presentation.13,14 γδ T-cells are functionally specialized and are involved in different disease processes, suggesting a role in both innate and acquired immunity.15–18 With regard to trauma, recent studies have shown the presence of activated γδ T-cells in the circulation of patients with severe SIRS, demonstrating the important role of these cells in the early response to severe injury. 19
Earlier studies from our laboratory using a mouse injury model have established an important role of γδ T-cells in burn-induced immunopathology influencing macrophage function, distal organ injury and wound repair.10,11 Burn is also associated with an increased level of circulating activated γδ T-cells with increased TLR expression. 20
The role of γδ T-cells in the recruitment of inflammatory cells to the injury site after burn has been described, as γδ T-cell deficient mice displayed a significant reduction in the cellular infiltrate, which was associated delayed wound healing. 21 Plasma and tissue chemokine levels were attenuated in γδ T-cells deficient mice, indicating that the γδ T-cells are an important source of chemokines. 22 In this regard, Boismenu et al. 23 have also shown that γδ T-cells produce a number of chemokines, such as lymphotactin, macrophage inflammatory protein (MIP)-1α, MIP-1β and RANTES for recruitment of other immune cells, such as neutrophils, monocytes, and so on. 23 The current study was undertaken to better characterize the role of wound γδ T-cells in the regulation of the wound inflammatory response.
Materials and methods
Mice
C57BL/6 wild type (WT) and mice lacking γδ T-cells (δ TCR-/-; C57BL/6 J-Tcrdtm1Mom) (male, 18–25 g; Jackson Laboratory, Bar Harbor, ME, USA) were used for all the experiments. Mice were allowed to acclimatize for at least 1 wk prior to experimentation and maintained in ventilated cages under specific pathogen-free conditions. Animals were randomly assigned to either sham or burn group. All animal protocols were approved by the Institutional Animal Care and Use Committee of the University of Texas Health Science Center at San Antonio. This study was conducted in compliance with the Animal Welfare Act, the implementing Animal Welfare Regulations, and the principles of the Guide for the Care and Use of Laboratory Animals.
Burn procedure
Mice received a scald burn as described previously. 24 Briefly, the mice were anesthetized by i.p. injection of ketamine/xylazine, and the dorsal surface was shaved. The anesthetized mouse was placed in a custom-built, insulated mold exposing 12.5% of their total body surface area (TBSA) along the right dorsum. The mold was immersed in 70℃ water for 10 s to produce a third degree burn. The burn procedure was repeated on the left dorsal side, yielding a total burn size of 25% TBSA. Previous studies have verified this injury to be a full-thickness burn, as defined by observed damage to the epidermal, dermal and sub-dermal layers.24,25 The mice were then resuscitated with 1 ml of Ringer's lactate solution administered by i.p. injection and returned to their cages. The cages were placed on a heating pad until the mice were fully awake, at which time they were returned to the animal facility. Sham treatment consisted of anesthesia and resuscitation only.
Skin tissue collection, digestion and cell isolation
At 1, 3 or 7 d after burn or sham procedure, skin samples were collected and wet mass was measured. Normal, non-injured skin was collected from sham, and injured skin from burn site was collected from burn mice. Skin samples from the burn site included injured skin and the wound margin. The burn-injured skin was excised, down to the level of the musculofascia, including the submucosal layer, by sharp dissection.
Collected skin tissues were washed in PBS with 50 U/ml penicillin and 50 µg/ml streptomycin (Gibco/Life Technologies, Grand Island NY, USA) in a 60-mm petri dish (Corning Life Sciences, Tewksbury, MA, USA) and the skin was minced with scissors into small pieces of approximately 2–3 mm in size and placed into dispase II medium (0.05%; Roche Applied Science, Indianapolis, IN, USA) for overnight digestion at 4℃ on an orbital rocker. The following day the skin samples were further minced into smaller pieces and then digested by agitating in trypsin-GNK (0.3%, Glc/dextrose, NaCl and KCl buffer; Sigma-Aldrich, St Louis, MO, USA) for 30 min at 37℃ in a water bath shaker. Heat-inactivated FBS (Gibco/Life Technologies) was added (10% total volume) to stop the digestion reaction and the dissociated cells were sieved through a 100-µm mesh. The cell suspension was centrifuged at 400 g for 10 min at 4℃ and re-suspended in RPMI culture medium [RPMI with 10% FBS, 50 µM of 2-mercaptoethanol (Sigma-Aldrich), 2 mM
Cell phenotyping by flow cytometry
The cells were washed in staining buffer (PBS with 0.2 % BSA and 0.09% NaN3) and treated with Fc-blocking Ab (anti-CD16/CD32; BD Biosciences) for 15 min. The cells were stained with the following directly conjugated Abs: anti-CD3 [phycoerythin (PE) or APC-Cy7] in combination with anti-β TCR (PerCPCy5.5), anti-δ TCR (FITC), anti-CD4 (PECy7), anti-CD8 (APC-Cy7), anti-CD69 (PE-Cy7), anti-TLR2 (PE) anti-TLR4 (APC) and anti-TLR9 (PB). After 30 min of incubation on ice, the cells were washed and re-suspended in staining buffer. Appropriate isotype controls were used for all staining. All data were acquired using a LSRII (BD Biosciences) and analyzed using FlowJo (Tree Star) software. A minimum of 50,000 events was collected and live cells were gated according to forward- and side-scatter properties. Absolute cell count was determined as % cells and total cells per gram of wet mass of skin tissue.
Statistical analyses
Data are expressed as mean ± SEM. Comparisons between groups were analyzed using ANOVA and SigmaPlot 11.0 software (Systat Software, San Jose, CA, USA). Further post-hoc analysis employed the Dunnett’s method for multiple comparisons versus the control group. A P-value < 0.05 was considered to be statistically significant for all analyses.
Results
Infiltration of the burn wound with T-cells
Skin T-cells, isolated from full-thickness skin samples were studied after 1, 3 and 7 d after sham or burn procedure. A profound influx of total CD3+ T-cells was observed at the wound site at all time points after burn (Figure 1). The percentage and absolute numbers of T-cells were significantly elevated as early as 1 d post-burn (fourfold) and remained elevated up to 7 d (threefold) in comparison with the T-cells percentages and numbers in skin from sham mice (Figure 1). In uninjured skin from burn mice the percentage and number of T-cells were comparable to that of sham mice and were not different at d 1 or d 7 (data not shown). However, at 3 d after burn, approximately a 2.5-fold increase in the percentage of T-cells was observed in the uninjured skin of burn mice (10.7% ± 1.8 versus 4.3% ± 0.6% for uninjured burn and sham, respectively; P < 0.05). A similar 2.5-fold increase in the number of viable cells was also observed in the uninjured skin of burn mice (data not shown). Analysis of the αβ T-cell and γδ T-cell subsets revealed that the mean fluorescence intensity (MFI) for CD3 was significantly higher on the γδ T-cells than the αβ T-cells and that with injury CD3 expression (i.e. MFI) significantly decreased compared with cells from sham skin (Table 1.)
Analysis of skin T-cells. At 1, 3 or 7 d after sham or burn procedure, skin cells from WT mice were prepared and studied for T-cell phenotype by flow cytometry as described in the ‘Materials and methods’. (A) Representative data for the isotype Ab and CD3 for cells from sham and burn mice at 3 d after injury. (B) Data for CD3+ T-cell percentages. (C) Data for the number of T-cells/g wet mass of tissue. Data are mean ± SEM for 5–11 mice/group. *P < 0.05 versus respective sham, †P < 0.05 versus d 1 burn. CD3 expression (MFI) on skin T-cells. Data are expressed as mean ± SEM of the percentage (n = 4–5 mice/group). P < 0.05 versus respective sham. P < 0.05 versus respective CD3+ β TCR+.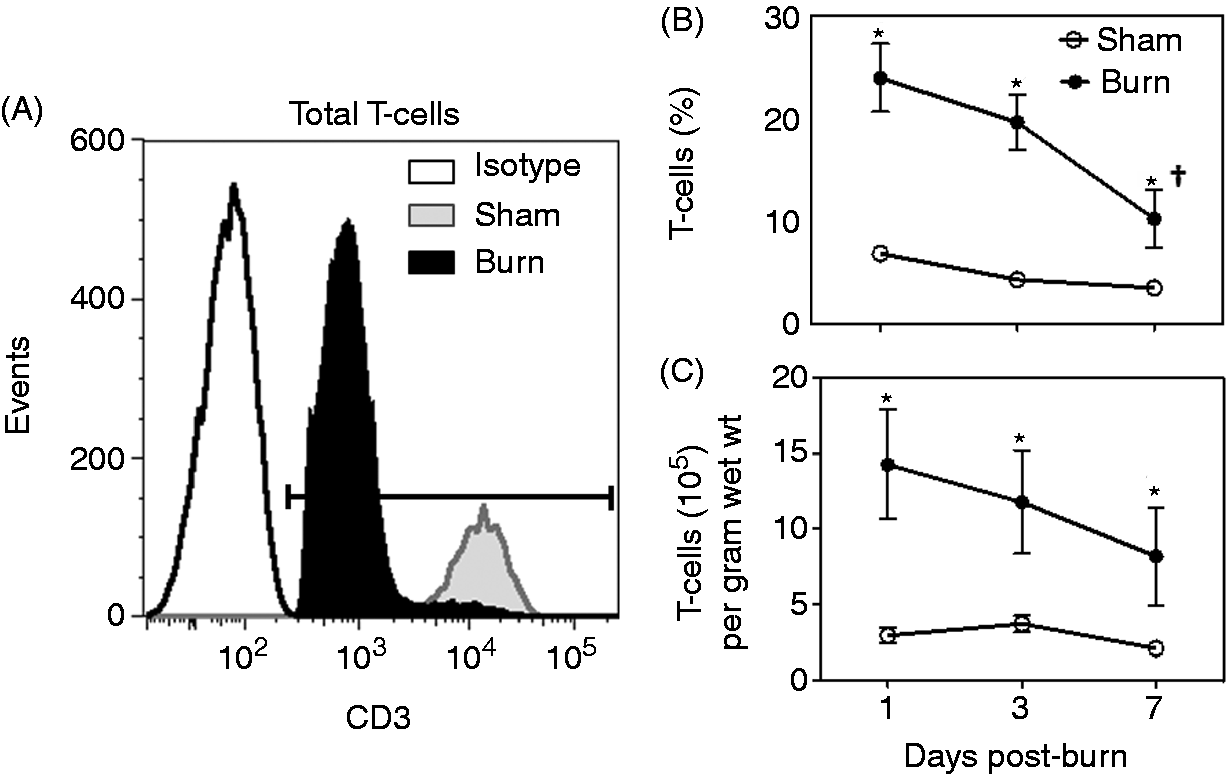

Isolated skin cells were stained with anti-CD3 in combination with δ TCR and β TCR Abs to characterize γδ T-cells (Figure 2) and αβ T-cells (Figure 3), respectively. The majority of T-cells in the uninjured skin of sham mice were of the γδ T-cell receptor lineage. γδ T-cells contributed from 80–90% of the total T-cells in the uninjured skin of sham mice (Figure 2A, B) and burn mice (data not shown). The overall percentage of γδ T-cells was reduced by 70–95 % at the wound site, as compared with T-cells from uninjured skin of sham mice (Figure 2A, B). Figure 2A shows representative data at d 3 after burn. While the percentage of the γδ T-cells was reduced after burn, the absolute number of the cells remained comparable to sham skin at 1 and 3 d post-burn. At d 7 post-burn γδ T-cell numbers were significantly reduced compared with sham skin.
Analysis of skin γδ T-cells. At 1, 3 or 7 d after sham or burn procedure, skin cells from WT mice were prepared and studied for T-cell phenotype by flow cytometry as described in the ‘Materials and methods’. (A) representative data for CD3 and δTCR based on the CD3+ gate for cells from sham and burn mice at d 3 after injury. (B) Data for CD3+ δTCR+ T-cell percentages. (C) Data for the number of γδ T-cells/g wet mass of tissue. Data are mean ± SEM for 3–11 mice/group. *P < 0.05 versus respective sham, †P < 0.05 versus d 1 burn. Analysis of skin αβ T-cells. At 1, 3 or 7 d after sham or burn procedure, skin cells from WT mice were prepared and studied for T-cell phenotype by flow cytometry as described in the ‘Materials and methods’. (A) Representative data for CD3 and βTCR based on the CD3+ gate; the numbers indicate the percentage of αβ T-cells gated from total T-cells. (B) Data for CD3+ βTCR+ T-cell percentages. (C) Data for the number of αβ T-cells/wet mass of tissue. Data are mean ± SEM for 3–11 mice/group. *P < 0.05 versu respective sham.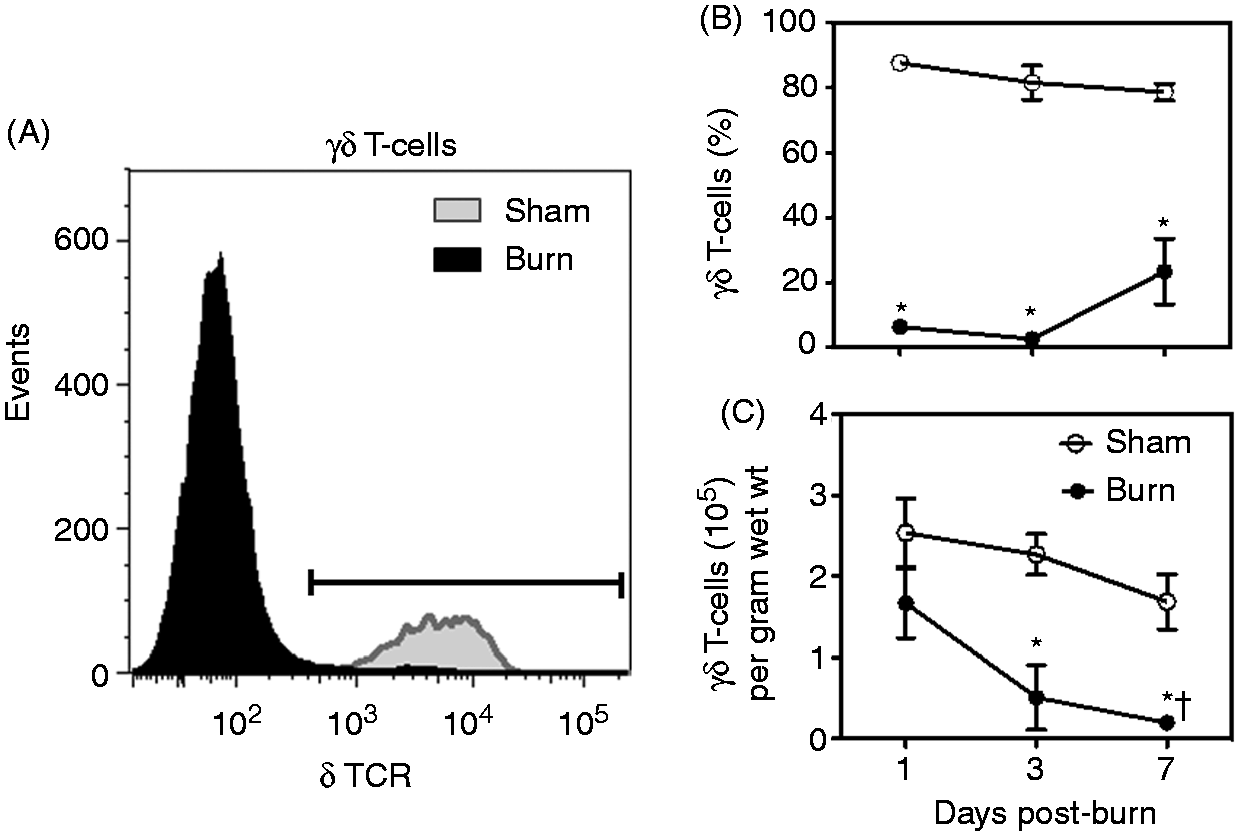
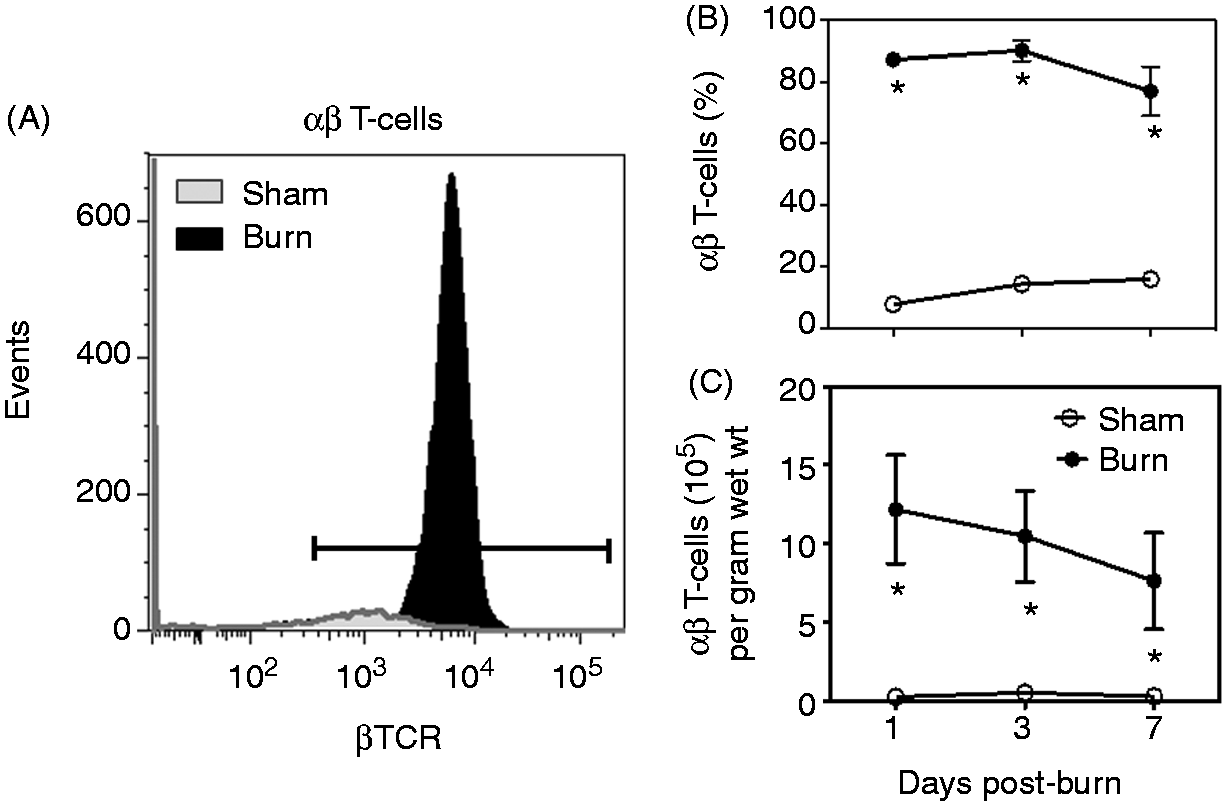
Burn was also associated with a profound increase in both the percentage and absolute numbers of αβ T-cells at the wound site, contributing to approximately 95% of the total T-cells present at the wound site (Figure 3). αβ T-cell numbers and percentage increased as early as d 1 post-burn with approximately a 50-fold, increase and remained elevated by approximately 20-fold at 7 d after injury.
Burn increases γδ T-cell TLR expression
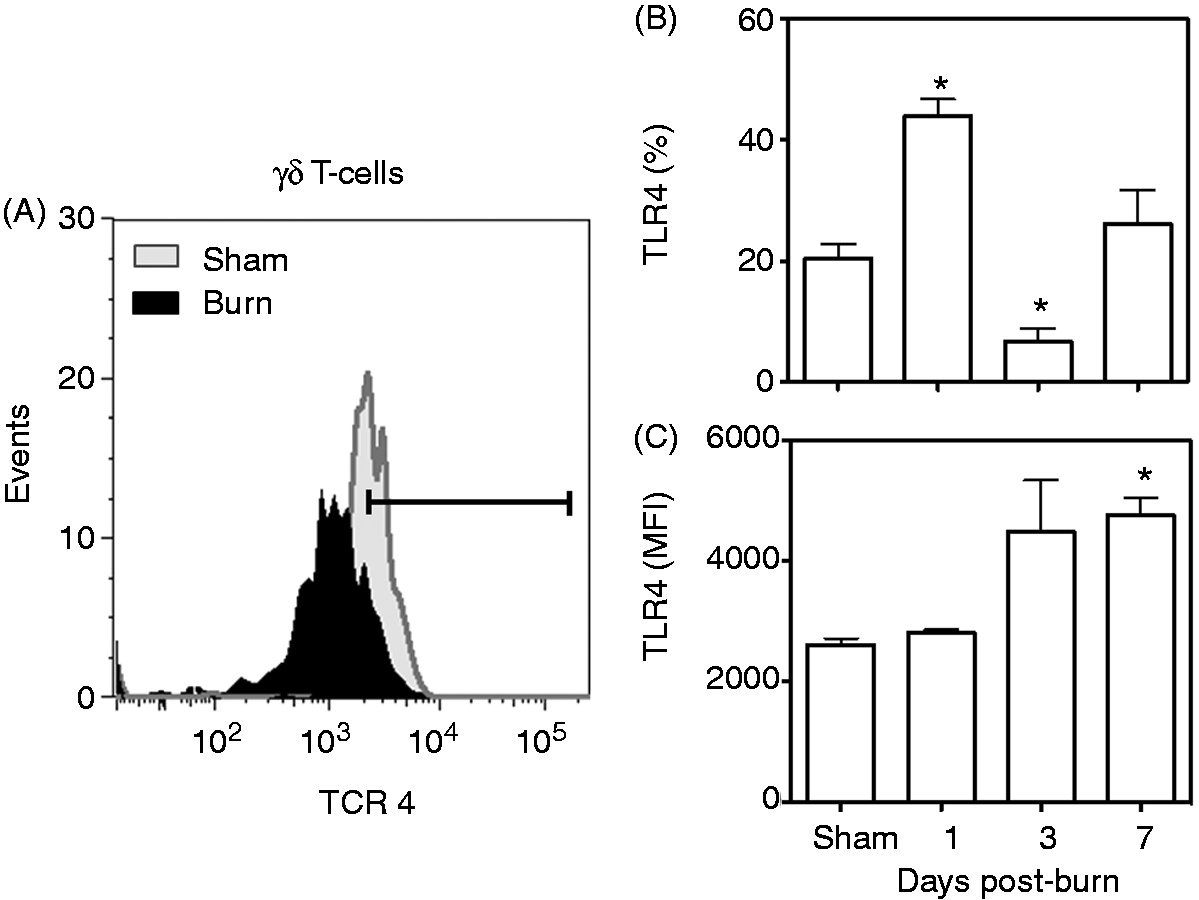
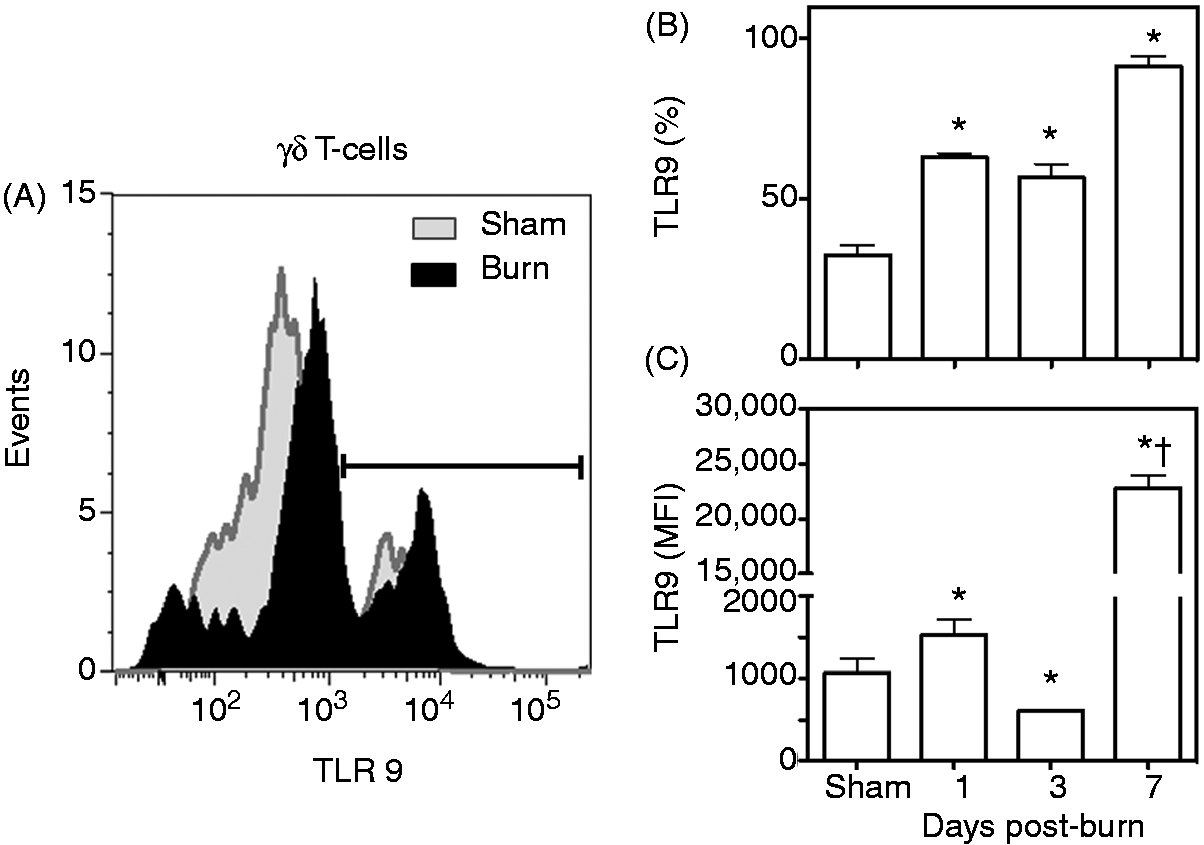
The percentage of the γδ T-cells expressing TLR2 increased significantly after the injury compared with sham mice (Figure 4). At d 3 after burn, but not earlier, a significantly greater proportion of the γδ T-cell population at the wound site was TLR2+-positive compared with uninjured skin from the sham mice (Figure 4A, B). In addition, the MFI for TLR2 (an indicator of the relative number receptors per cell), was also significantly increased at d 3 after burn (Figure 4C). By 7 d post-injury γδ T-cell TLR2 expression was not significantly different between the sham and burn groups. In contrast to TLR2, TLR4 expression on γδ T-cells after burn was biphasic. The expression of TLR4 doubled at d 1 after burn, was suppressed at d 3 after burn and returned to sham levels at 7 d post-injury (Figure 5). A twofold increase in the MFI for TLR4 was observed at 7 d after burn, but not earlier (Figure 5C). The percentage of the γδ T-cells expressing TLR9 increased significantly as early as 1 d after injury (Figure 6). By 7 d post-injury ∼ 90% of the γδ T-cells were positive for TLR9 (Figure 6B). This change at 7 d after burn was paralleled by a profound 22-fold increase in the MFI for TLR9 compared with cells from sham mice (Figure 6C).
TLR2 expression on γδ T-cells. At 1, 3 or 7 d after sham or burn procedure, skin cells from WT mice were prepared and studied for γδ T-cells and TLR2 phenotype by flow cytometry as described in the ‘Materials and methods’. (A) Representative data for δTCR and TLR2 based on the CD3+ δTCR+ cells from the lymphocyte gate at d 3 after injury. (B) Data for CD3+ δTCR+ TLR2+ T-cell percentages. (C) Data for CD3+ δTCR+ TLR2+ T-cell MFI. Data are mean ± SEM for 4–9 mice/group; *P < 0.05 versus sham. TLR4 expression on γδ T-cells. At 1, 3 or 7 d after sham or burn procedure, skin cells from WT mice were prepared and studied for γδ T-cells and TLR4 phenotype by flow cytometry as described in the ‘Materials and methods’. (A) Representative data for δTCR and TLR4 based on the CD3+ δTCR+ cells from the lymphocyte gate at d 3 after injury. (B) Data for CD3+ δTCR+ TLR4+ T-cell percentages. (C) Data for CD3+ δTCR+ TLR4+ T-cell MFI. Data are mean ± SEM for 4–9 mice/group; *P < 0.05 versus sham. TLR9 expression on γδ T-cells. At 1, 3 or 7 d after sham or burn procedure, skin cells from WT mice were prepared and studied for γδ T-cells and TLR9 phenotype by flow cytometry as described in the ‘Materials and methods’. (A) Representative data for δTCR and TLR9 based on the CD3+ δTCR+ cells from the lymphocyte gate at d 3 after injury. (B) Data for CD3+ δTCR+ TLR9+ T-cell percentages. (C) Data for CD3+ δTCR+ TLR9+ T-cell MFI. Data are mean ± SEM for 4–9 mice/group; *P < 0.05 versus sham. †P < 0.05 versus d 1 and d 3 post-burn.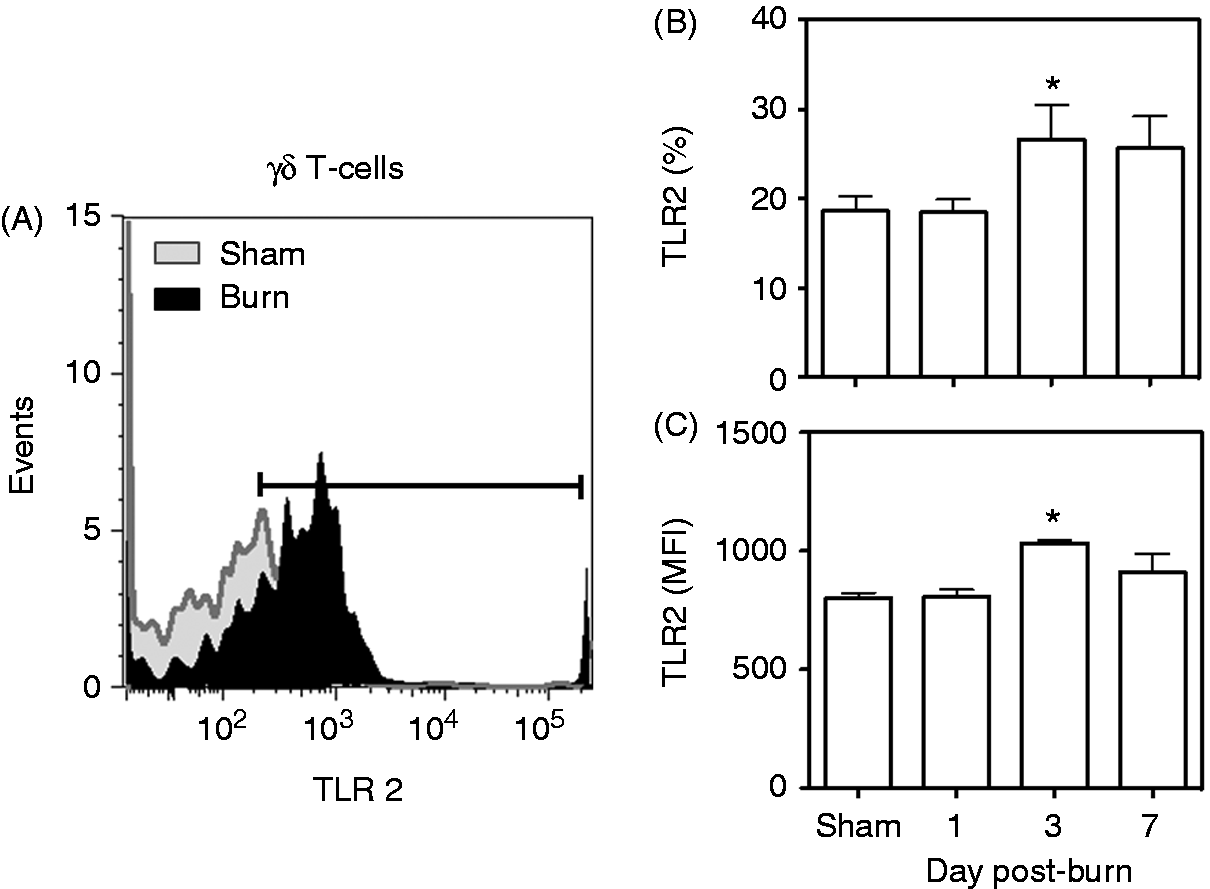
TLR expression on αβ T-cells.
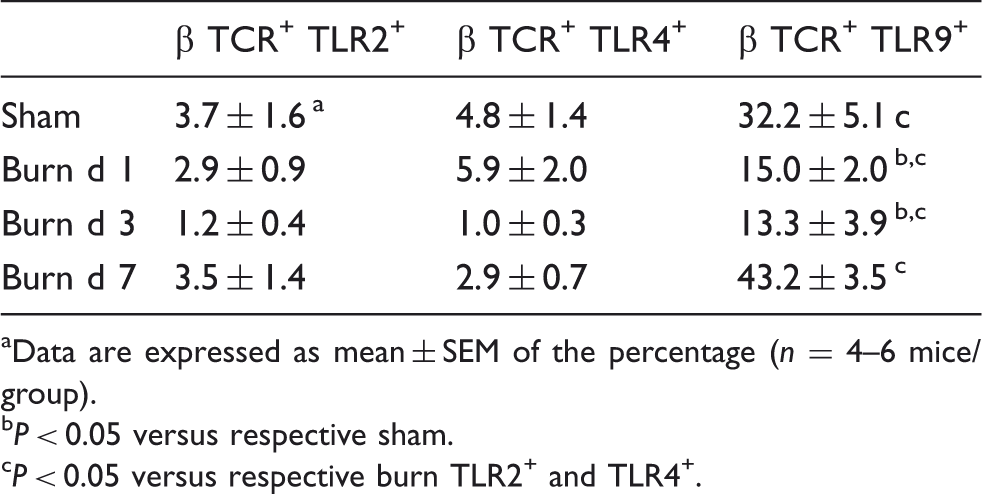
Data are expressed as mean ± SEM of the percentage (n = 4–6 mice/group).
P < 0.05 versus respective sham.
P < 0.05 versus respective burn TLR2+ and TLR4+.
Burn increases γδ T-cell CD69 expression, but suppresses αβ T-cell CD69 expression
Further analysis of γδ T-cells for their activation status revealed that the γδ T-cells had increased expression of CD69 (Figure 7A). Similar to TLR4, a biphasic response was observed after burn. A small percentage of γδ T-cells from normal skin of sham mice expressed CD69 (∼ 5%). At d 1 after burn CD69 expression doubled; at d 3 after burn expression was not elevated compared with sham; and at d 7 after burn CD69 expression was again elevated ∼ twofold. A greater percentage of αβ T-cells from normal skin of sham mice expressed CD69 (∼ 10%) than γδ T-cells (Figure 7C). Unlike γδ T-cells, the CD69 expression on αβ T-cells was significantly reduced at all time points after burn. While percentages of CD69+ cells changed after burn compared with sham, the MFI for CD69 on either T-cell population did not change significantly (Figure 7B, D). Figure 8 shows representative data for CD69 expression on γδ and αβ T-cells at 3 d post-injury.
CD69 expression on γδ and αβ T-cells. At 1, 3 or 7 d after sham or burn procedure, skin cells from WT mice were prepared and studied for CD69 expression on γδ and αβ T-cells by flow cytometry as described in the ‘Materials and methods’. (A) percentages and (B) MFI of the aggregated data for the CD3+ δTCR+ CD69+ γδ T-cells from the CD3+ δTCR+ population from the lymphocyte gate. (C) Percentages and (D) MFI of the aggregated data for the CD3+ βTCR+ CD69+ αβ T-cells that are gated from the CD3+ βTCR+ population from the lymphocyte gate. Data are mean ± SEM for 3–9 mice/group; *P < 0.05 versus respective sham. CD69 expression on γδ and αβ T-cells at 3 d post-injury. Skin cells from WT mice were prepared and studied for CD69 expression on γδ and αβ T-cells by flow cytometry as described in the ‘Materials and methods’. (A–D) Cells within the lymphocyte gate (based on forward and side scatter) that were positive for δTCR and βTCR in sham and burn mice. The numbers represent the percentage of lymphocyes positive for δTCR and βTCR. (E–H) γδ T-cells and αβ T-cells positive for CD69 expression. The numbers represent the percentage of γδ or αβ T-cells positive for CD69. Data are from representative mice of 3–9/group. FSC: forward scatter.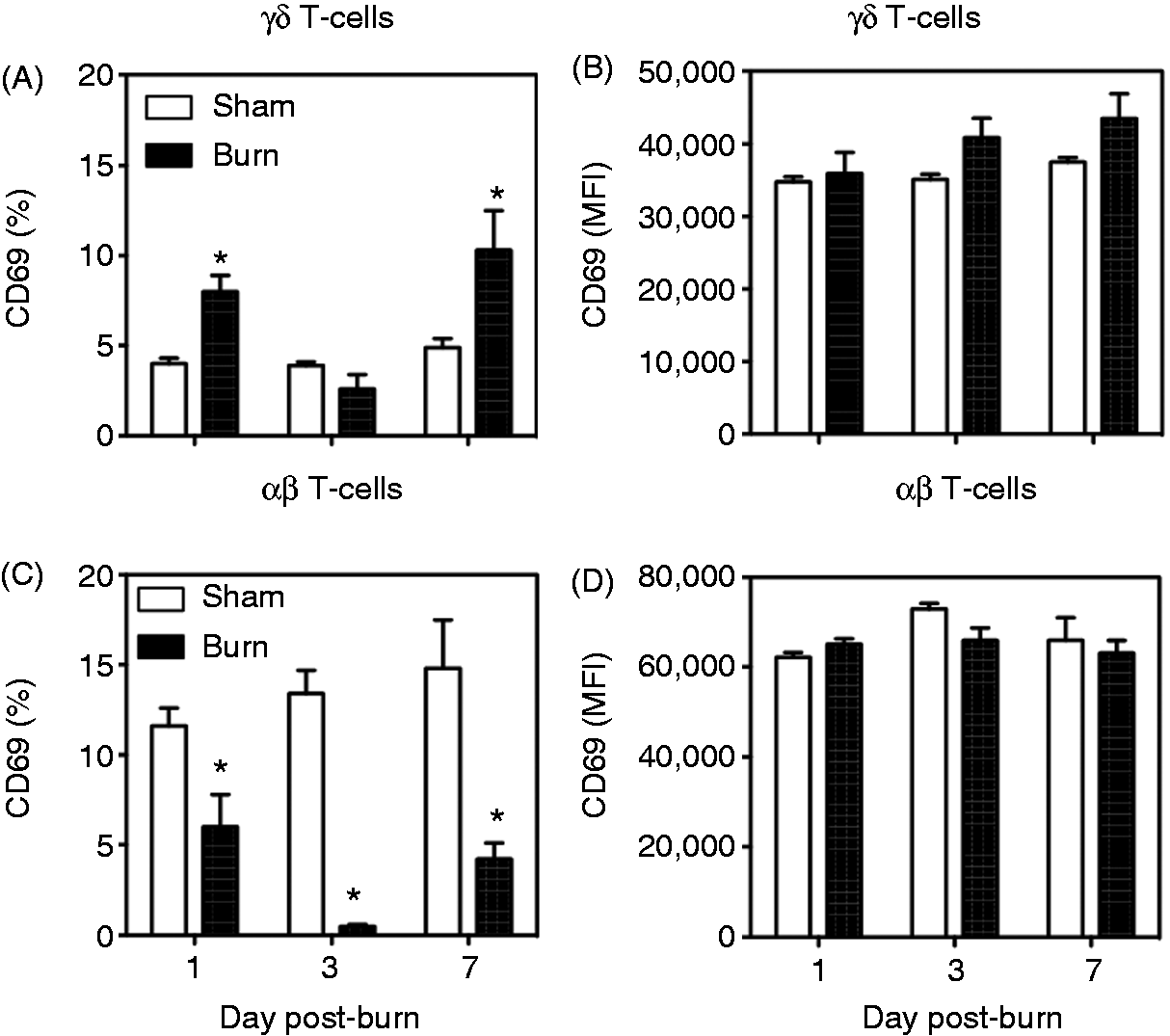
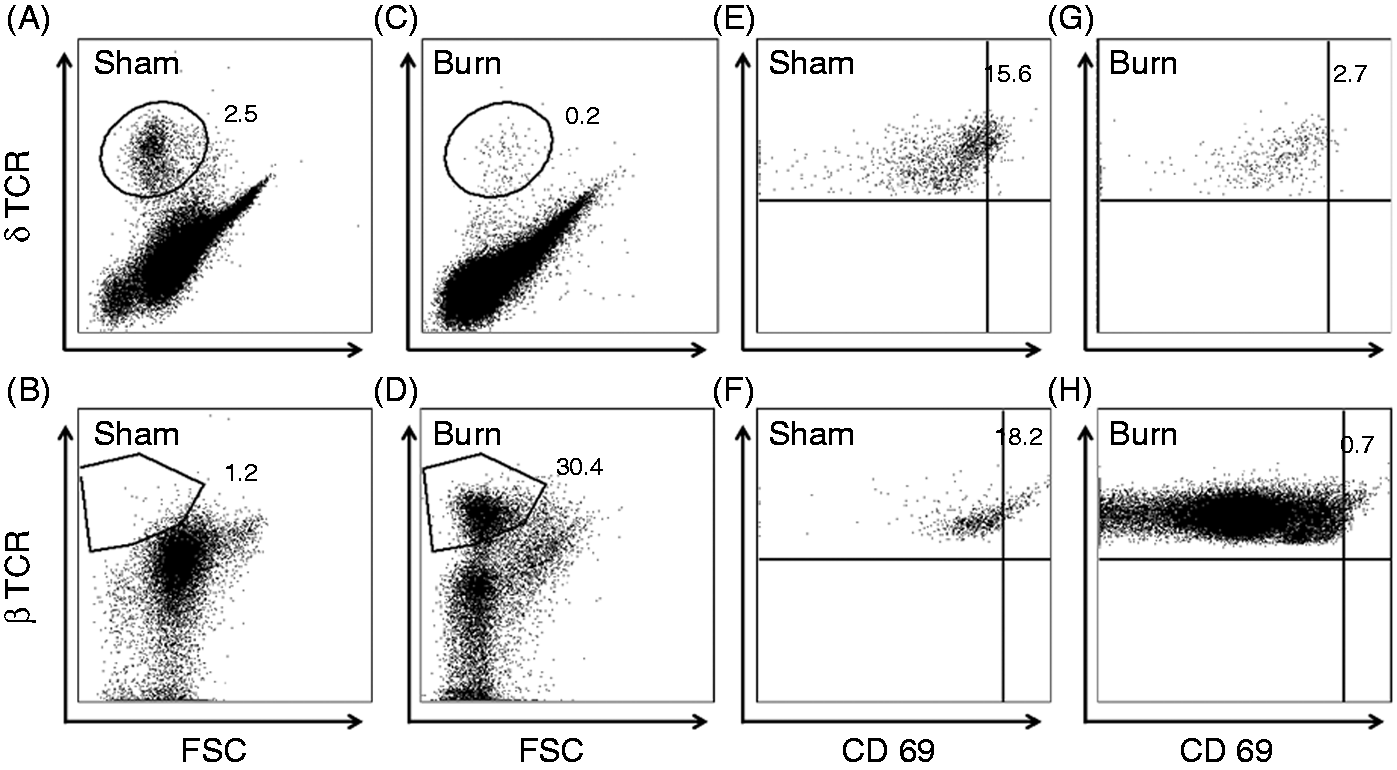
γδ T-cells are required for T-cell infiltration of the burn wound
To investigate whether γδ T-cells affect the wound infiltration by αβ T-cells, skin cells were isolated at 3 d after sham or burn procedure from WT or δ TCR-/- mice. The 3-d time point was chosen based on the observation as the time of maximal αβ T-cell infiltration (Figure 3). A profound fourfold attenuation in the burn-induced infiltration of αβ T-cells was observed at the burn wound site in δ TCR-/- mice in comparison with that observed in WT mice (Figure 9). T-cell percentages in the wounds of δ TCR-/- mice were not different of that from WT sham mice or δ TCR-/- sham mice (Figure 9C, D).
Impact of γδ T-cells on wound T-cell infiltration. Three d after sham or burn procedure, skin cells from WT or δ TCR-/- mice were prepared and analyzed for T-cell phenotype by flow cytometry as described in the ‘Materials and methods’. (A) Representative data for all CD3+ cells. (B) βTCR+ cells from the CD3+ gate. (C) Cumulative data (%) for T-cells; (D) cumulative data for αβ T-cells. Data are mean ± SEM for 3–7 mice/group. *P < 0.05 versus sham, †P < 0.05 versus WT.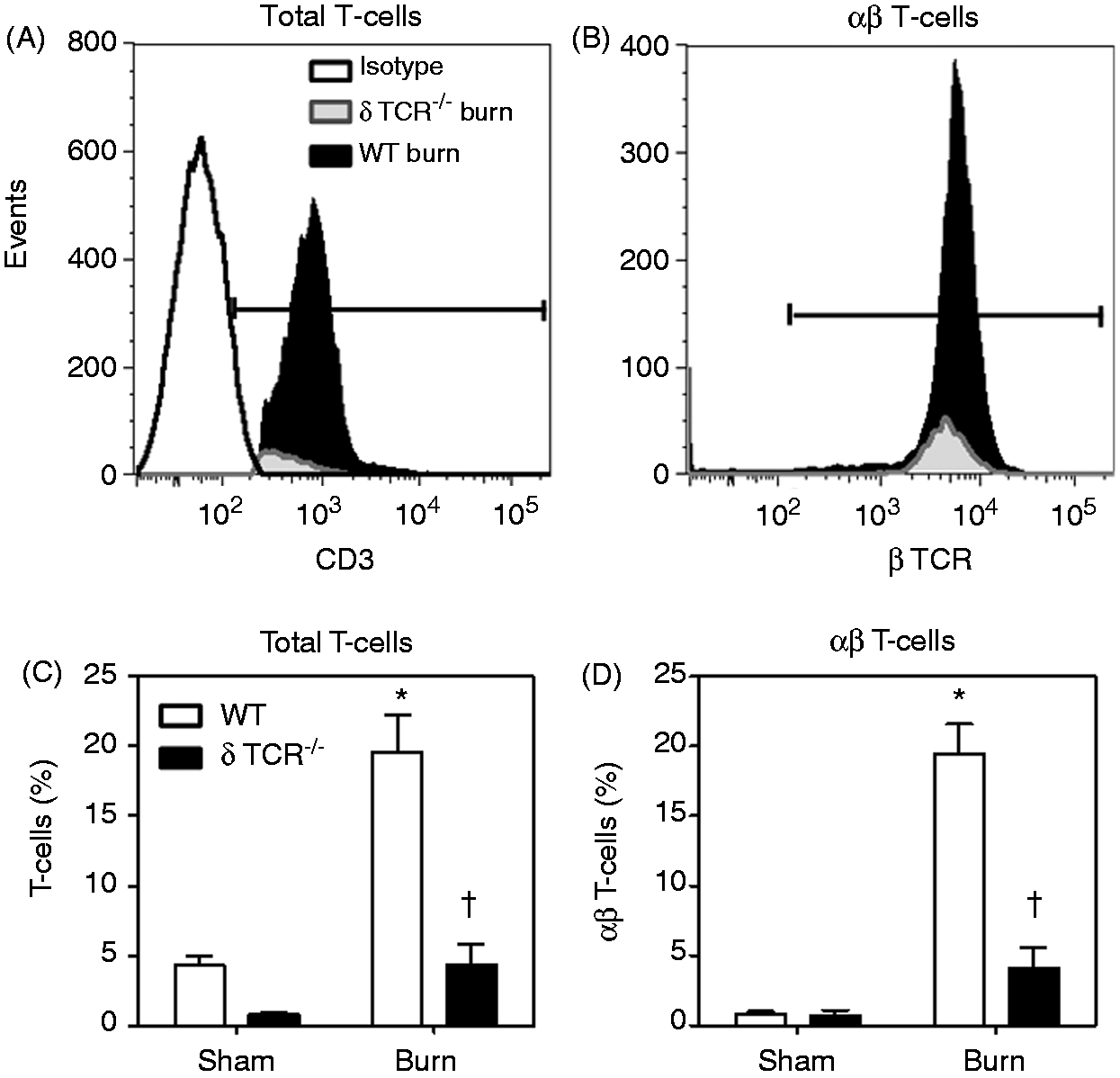
Discussion
The current study demonstrates that the resident skin γδ T-cells are activated after burn injury and essential to the massive influx of the αβ T-cells to the injury site, as this infiltration of the wound site by αβ T-cells was not evident in mice deficient in γδ T-cells. Moreover, the infiltrating αβ T-cells are relatively anergic and unresponsive to activation, which may be a contributing factor to wound-healing complications after burn.
It has been well established that most T-cells found in the mouse epidermis under naïve conditions are of the γδ TCR lineage,26,27 which our findings confirm. After burn, the percentage of γδ T-cells in the overall T-cell population dramatically decreased at the wound site; however, their absolute number remained relatively comparable to that found in uninjured skin, suggesting that the γδ T-cells in the wound were resident skin cells, rather than infiltrating cells. The wound γδ T-cells were activated, as evidenced by up-regulation of CD69, TLR2 and TLR4 expression on their surface. There is a growing body of literature describing this phenomenon of γδ T-cell activation after inflammation, injury and infection. A clinical study by Matsushima et al. 19 has demonstrated activated γδ T-cells in the circulation of SIRS patients. Earlier studies from our laboratory have shown the activation of circulatory γδ T-cells by TLR2 and TLR4 surface markers after 24 h of burn. 20 In another study, activation of the circulating γδ T-cells was observed early post-burn (i.e. 3 h) and was associated with increased CD120 expression. 20 A study by Cairns et al. 28 has also shown the increased TLR4 expression by splenic T-cells at later time post-burn injury, that is, 14 d.
Our current findings showing an elevation in TLR2, TLR4, and TLR9 expression in γδ T-cells suggests that damage associated molecular patterns (DAMPs) may play an important role in their activation after injury. The Danger Theory, proposed by Matzinger,29,30 suggests that the function of the immune system is to recognize and prevent attack from harm in the context of ‘Danger’ signals or DAMPs. The theory states that the mechanism by which a cell dies governs whether an immune response is initiated in that DAMPs are generated by injured or dying cells, which activate the system. Potent immune activation can be mediated by DAMPs via pattern recognition receptors, such as TLRs, which represents an important link between tissue injury, infection and inflammation. Zhang et al. 31 have recently demonstrated that mitochondrial DAMPs released by cellular disruption after trauma are present in the circulation and capable of activating neutrophils and inducing SIRS. γδ T-cells may also be activated after injury by DAMPs via TLRs in order to alert the host for its nominally sterile injury. 32 For example, cellular activation via TLRs can induce the expression of genes that regulate cell migration. 33 Thus, the post-burn changes in γδ T-cell TLR expression may be related to the early DAMP-mediated inflammatory response at the injury site. Our recent finding have demonstrated that γδ T-cells can be activated by mitochondrial DAMPs. 34
During the tissue remodeling phase of wound healing, T-cells constitute the most abundant leukocyte subset in human skin wounds.4,5 Several studies suggest that delayed T-cell infiltration and concentration in the wound site is associated with impaired wound-healing, while others have reported that CD4+ T-cells have a positive role in wound-healing, and that CD8+ T-cells play an inhibitory role in wound healing. 35
We observed a profound infiltration of the wound with αβ T-cells in terms of both the percentage and absolute number of cells. αβ T-cells are the predominant T-cell population in the draining lymph nodes. In this regard, Purcell et al. 36 have shown that burn causes CD4 T-cell proliferation in the lymph nodes draining from the injury site, but not in other lymphoid (i.e. spleen) tissues. 36 It is conceivable that this is related to our observation that αβ T-cells infiltrate the burn wound. The influx of αβ T-cells is γδ T-cell dependent, as it was virtually absent in mice lacking γδ T-cells. It is well established that γδ T-cells secrete a variety of chemokines that direct the migration and recruitment of specialized cells to the site of inflammation or damage.23,27,37 Jameson et al. 37 showed that the migration of macrophages and αβ T-cells to the epidermis is delayed in mice lacking γδ T-cells. Boismenu et al. 23 have also shown that γδ T-cells produce a number of chemokines for recruitment of various inflammatory cells and that the migration of CD8+ αβ T-cells was induced by activated γδ T-cells. While we did not measure chemokines in the current study, our earlier finding suggests that γδ T-cells are also an important source of chemokines early post-burn as plasma and tissue chemokine levels were reduced in δ TCR-/- mice. 22 More recently, we have shown that murine γδ T-cells can be activated to secrete RANTES, an important T-cell, by mitochondrial DAMPs. 34 DAMPs are likely to be in high concentrations at the burn wound site owing to the massive tissue damage present. Although the current study provides evidence that γδ T-cells regulate T-cell infiltration 3 d post-burn, regulation by γδ T-cells at other times cannot be excluded, as activation of γδ T-cells was observed earlier (d 1) and later (d 7). We chose to focus on 3 d post-injury because of the previously observed differences in growth factors at the burn site in δ TCR-/- mice at that time.21,38
Our findings also showed at profound decrease in CD3 expression by both αβ and γδ T-cells after burn. Traditionally, decreased CD3 expression, after exposure to Ags, is considered to be evidence of activation. 39 It can be speculated that T-cells at the injury site are activated by a number of exposed Ags as a result of the massive tissue injury associated with burn. The observed decrease in CD3 expression can also be explained by the shift from a predominantly γδ TCR+ population in sham skin to a αβ TCR+-predominant population in burn skin. An important characteristic of γδ T-cells is that they express much higher levels of the CD3/TCR complex than αβ T-cells.40,41 Venet et al. 41 have suggested that reduced CD3 expression by T-cells may contribute to suppressed Ag presentation after septic shock and be responsible, in part, for development of secondary infections. A similar scenario is possible after burn, which is also associated with increased susceptibility to infection. 1 The relationship between loss of CD3 expression by T-cells after burn and susceptibility infection remains to be elucidated.
While αβ T-cells were the predominant T-cell population at the wound site, these cells appear to not be activated. TLR2 and TLR4 expression by αβ T-cells was negligible under any conditions, consistent with our previous observations 20 and the infiltrating αβ T-cells showed a suppressed phenotype, as the expression of TLR9 was suppressed at 1–3 d after burn and the classic activation marker CD69 was also markedly attenuated. It has been shown by various research laboratories that inducible nitric oxide synthase (iNOS)-derived NO is produced in large amounts after burn.42–44 We speculate that αβ T-cells may be suppressed by iNOS-derived NO, as a direct role of iNOS in the inhibition of T-cell function is well established.45,46 Our recent findings show that γδ T-cells are important in the expression of iNOS at the burn wound site. 42
Conclusions
Our findings establish the regulatory role of γδ T-cells in the immunoinflammatory response to burn. They act as the initiators of the response and are essential in the recruitment of αβ T-cells at the wound site, which are likely to be important in the transition from the inflammatory to tissue remodeling stages of wound-healing.
Footnotes
Acknowledgements
These finding were presented in part at the 43 rd annual meeting of the American Burn Association in Chicago, IL, USA. We thank Ms. Teresa Craig for her excellent technical support and assistance in the preparation of this manuscript. MR was responsible for the animal experiments, cell isolation, FACs, data analysis and drafting of the manuscript. QZ were responsible for the animal experiments, cell isolation and FACs. MRS and APC were responsible for scientific design and interpretation. MGS was responsible for scientific conception, design and interpretation and assisted in the final drafting of the manuscript. All authors read and approved the final version of the manuscript. The opinions or assertions contained herein are the private views of the author and are not to be construed as official or as reflecting the views of the Department of the Army or the Department of Defense. The authors declare that they have no competing interests.
Funding
This work was supported by grant funding from the National Institutes of Health (GM079122).
