Abstract
Gamma delta T-cells have been shown to be important in the early immunoinflammatory response to injury, which can be independent of infection. This sterile inflammatory response is believed to be, in part, associated with danger-associated molecular patterns (DAMPs). Mitochondrial DAMPs (MTDs) have been shown to be important in trauma-induced neutrophil activation, but it is unknown whether MTDs activate other innate immune cells, such as γδ T-cells. To study this, splenic CD3+ γδ T-cells were isolated from αβ T-cell-deficient C57BL/6 mice and mitochondria isolated from wild type mouse livers. MTDs were isolated from mitochondria by sonication and centrifugation. Gamma delta T-cells were incubated with various concentrations of MTDs (0–500 µg/ml) for 24 h. T-cells were phenotyped for TLR expression by flow cytometry and the supernatants assayed for cytokine and growth factor content. MTDs caused a dose-dependent increase in TLR2 and TLR4 expression by γδ T-cells. Both the percentage of cells positive for TLRs and the degree of expression increased. MTDs also induced the production of IL-1β, IL-6, IL-10, RANTES, fibroblast growth factor-basic and vascular endothelial growth factor by γδ T-cells. These findings support the concept that the MTDs released after tissue/cellular injury are capable of activating γδ T-cells, thus initiating sterile inflammation, as well as subsequent healing processes.
Introduction
The involvement of γδ T-cells in a wide variety of disease processes is indicative of an important role for this T-cell subset in both innate and acquired immunity.1–3 With regard to this, a critical role for γδ T-cells in inflammation and tissue repair has been shown.4–7 Studies from our laboratory suggest an important role for γδ T-cells in the immune response to major injury (i.e. burns).8–11 In these studies, increased activation of the circulating γδ T-cells was observed and the findings also indicated that γδ T-cells play a role in post-burn immune dysfunction, neutrophil-mediated tissue damage and wound healing. Normally, γδ T-cells are present only in small numbers in peripheral lymphoid tissues (i.e. spleen), but relatively abundant in the skin epithelia, intestine, uterus and tongue. 1 However, γδ T-cells have a phenotype of spontaneous activation, and under pathological conditions they can quickly expand and infiltrate lymphoid compartments and other tissues.12,13 Nonetheless, the mechanism(s) and mediator(s) responsible for mobilization and activation of this T-cell subset are under such conditions are unclear.
The ‘Danger Theory’, proposed by Matzinger,14,15 suggests that the function of the immune system is to prevent and recognize attack from harm in the context of ‘Danger’ signals or danger-associated molecular patterns (DAMPs). The theory states that the mechanism by which a cell dies governs whether an immune response is initiated, in that DAMPs are generated by injured/dying cells, which activate the system. Matzinger and Seong have also proposed that pathogen-associated molecular patterns (PAMPs) and DAMPs have similar conserved hydrophobic portions that engage the same pattern recognition receptors (PRRs) to elicit comparable responses. 16 DAMPs include mitochondrial (mt)DNA; high mobility group box 1 (HMGB1), a DNA binding protein; and S100A8, a calcium binding protein.17,18 Potent immune activation can be mediated by DAMPs via PRRs, such as TLRs, which represent a key link between tissue injury, infection and inflammation.
Hauser et al. 19 have recently demonstrated that mitochondrial DAMPs (MTDs) released by cellular disruption after trauma are present in the circulation and capable of activating neutrophils. These findings provide a key link between injury, inflammation and systemic inflammatory response syndrome (SIRS). The current study was undertaken to determine whether γδ T-cells are also capable of being activated by MTDs.
Materials and methods
Animals
C57BL/6 male mice lacking αβ T-cells (Strain: B6.129S2-Tcratm1Mom/J, 12–14-wk-old; Jackson Laboratories, Bar Harbor, ME, USA) were used for all experiments. The mice were allowed to acclimatize in the animal facility for at least 1 wk prior to experimentation. The study was approved by the Institutional Animal Care and Use Committee of the University of Texas Health Science Center at San Antonio. The study was conducted in compliance with the Animal Welfare Act, the implementing Animal Welfare Regulations, and the principles of the Guide for the Care and Use of Laboratory Animals.
Isolation of splenic γδ T-cells
The γδ T-cells used for these studies were obtained from the spleens of C57Bl/6 J mice lacking αβ T-cells (TCR αβ−/−), as described previously. 20 The mice were sacrificed by ketamine/xylazine overdose and the spleens were removed aseptically. Splenocyte suspensions were prepared in PBS. Splenic T-cells were enriched using an EasySep Mouse T-Cell Enrichment Kit according to the manufacturer’s recommendations (Stemcell Technologies, Vancouver, BC, Canada). The enriched T cells were ∼90% CD3 + γδ TCR+.
Generation of MTDs
MTDs were generated as described previously by Hauser et al. 19 In brief, mitochondria were isolated from frozen mouse liver samples using a mitochondria isolation kit (Thermo Scientific, West Palm Beach, FL, USA), according to the manufacturer’s recommendations. The mitochondria were subjected to five cycles of freeze–thawing in liquid nitrogen. The resulting disrupted mitochondrial suspensions were centrifuged at 12,000 g for 10 min at 4℃ and subsequently at 100,000 g at 4℃ for 30 min. The residual supernatants were used for the experiments. The protein concentration of the MTD solution was determined by BCA Protein Assay (Thermo Scientific, West Palm Beach, FL, USA).
In vitro activation of γδ T-cells
T-Cells (106/ml) were cultured in complete RPMI ((RPMI 1640 containing 10% heat-inactivated FBS, 5 µg/ml gentimycin and 100 µg/ml of streptomycin and penicillin; GibcoBRL, Grand Island, NY, USA) for 24 h with MTD (0–500 µg/ml). Cells were collected for flow cytometry, and supernatants for Bioplex analysis of cytokines and growth factors.
Determination of cellular phenotype
The cells were stained with a combination of Abs (γδ TCR, CD3, TLR2, TLR4) conjugated to various fluorochromes [FITC, phycoerythrin (PE), APC or PE-C7] to assess cellular phenotype. The manufacturer’s recommended methodology was employed (BD Biosciences, Mountain View, CA, USA). Appropriate isotype controls were included for all Abs. The stained cells were analyzed with a Becton-Dickinson LSR II flowcytometer (BD Biosciences) with a minimum of 30,000 events being collected. The data were analyzed with FloJo software (Tree Star, Ashland, OR, USA).
Determination of cytokine and growth factor levels
The Bioplex (Bio-Rad, Hercules, CA, USA) system was used for cytokine and growth factor level analysis in culture supernatants according to the manufacturer’s recommendations. The following factors were assessed: IL-1β, IL-6, IL-10, IL-17, TNF-α, RANTES, fibroblast growth factor (FGF)-basic and vascular endothelial growth factor (VEGF).
Statistical analysis
Data are expressed as mean ± SE. Comparisons between groups were analyzed using ANOVA and SigmaPlot 11.0 software (Systat Software, San Jose, CA, USA). Further post-hoc analysis employed the Dunnett’s method for multiple comparisons versus the control group. A P-value <0.05 was considered to be statistically significant for all analyses.
Results
MTD increase TLR expression by γδ T-cells
Under naive conditions approximately 40% of the γδ T-cell population expressed TLR2 on their cell surface. Following in vitro exposure to MTDs (500 µg/ml), almost all γδ T-cells expressed TLR2 (∼95%; Figure 1A). In addition, MTD (500 µg/ml) also enhanced the mean fluorescence intensity (MFI) for TLR2 on the γδ T-cells’ population approximately 2.5-fold, indicative of an increase in the number of receptors on each cell (Figure 1B). MTDs also increased the uniformity of TLR2 expression (i.e. decreased MFI range) by γδ T-cells, consistent with activation (Figure 1C). MTDs at lower concentrations did not significantly alter TLR2 expression.
Effect of MTD on TLR2 expression by γδ T-cells. Splenic γδ T-cells were isolated and cultured for 24 h with various concentrations of MTD, as described in the ‘Materials and methods’. The lymphocyte population was identified by forward and side scatter, and analyzed for CD3, γδ TCR and TLR2 expression. The data in (A) show the percentage of γδ T-cells positive for TLR2 and the data in (B) show the MFI for TLR2 in the CD3+ γδ TCR+ cell population. (C) Representative data of the effect of MTD (500 µg/ml) on TLR2 expression by the CD3+ γδ TCR+ cells. Data are mean ± SEM for n = 5/group. *P < 0.05 vs control (0 µg/ml MTD).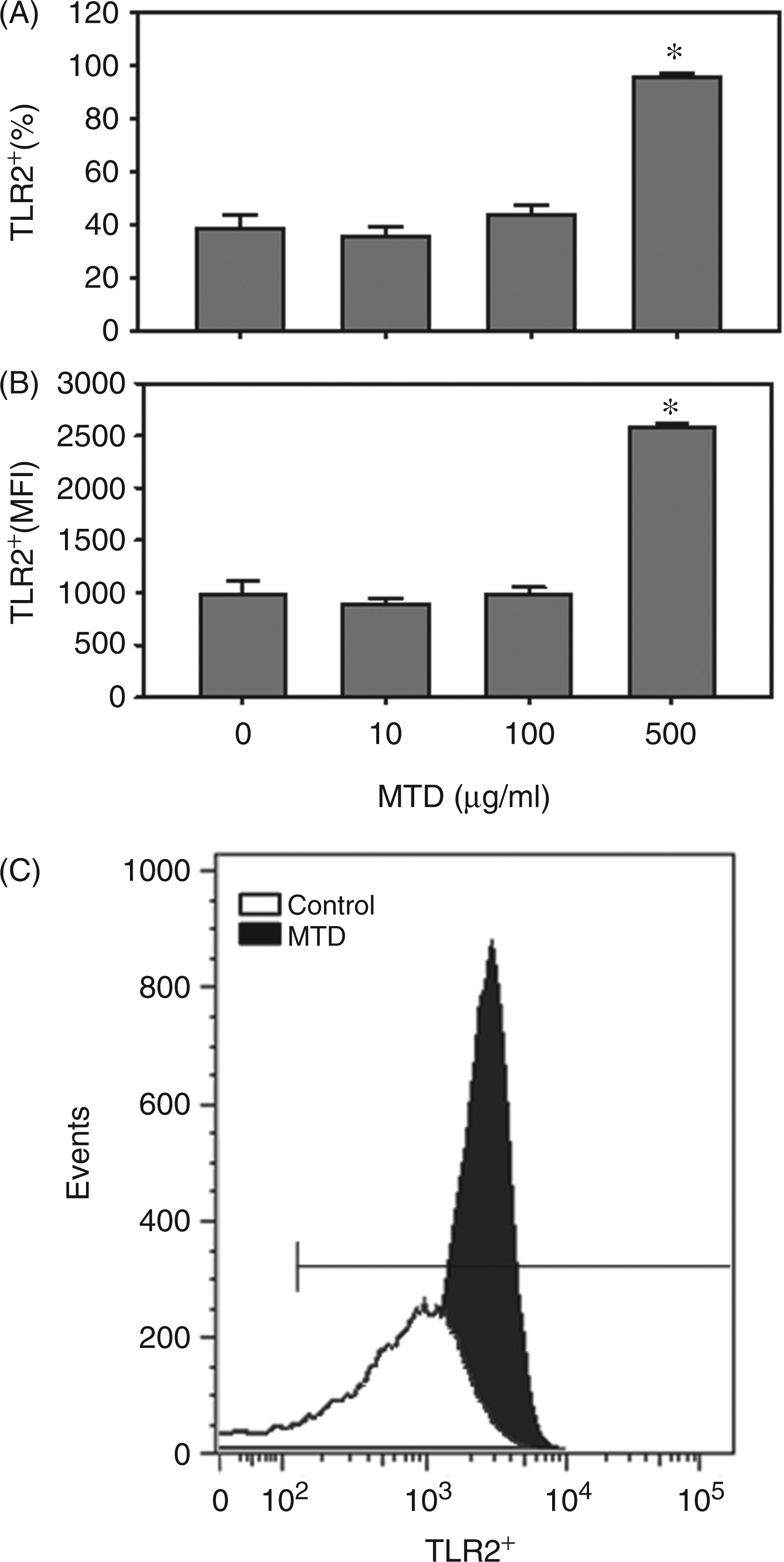
TLR4 expression by γδ T-cells under naive conditions was approximately 60% and significantly greater than that of TLR2 (57 ± 2% vs 38 ± 5%, n = 5, P < 0.05). MTD at a concentration of 500 µg/ml increased TLR4 expression to approximately 75% (Figure 2A). Similar to TLR2, MTD (500 µg/ml) enhanced the MFI for TLR4 by approximately 65% (Figure 2B). In contrast to TLR2, the range of TLR4 expression on γδ T-cells after MTD remained consistent with that of naive cells (Figure 2C).
Effect of MTD on TLR4 expression by γδ T-cells. Splenic γδ T-cells were isolated and cultured for 24 h with various concentrations of MTD, as described in the ‘Materials and methods’. The lymphocyte population was identified by forward and side scatter, and analyzed for CD3, γδ TCR and TLR4 expression. Data in (A) show the percentage of γδ T-cells positive for TLR2 and the data in (B) show the MFI for TLR4 in the CD3+ γδ TCR+ cell population. (C) Representative data of the effect of MTD (500 µg/ml) on TLR4 expression by the CD3+ γδ TCR+ cells. Data are mean ± SEM for n = 5/group. *P < 0.05 vs control (0 µg/ml MTD).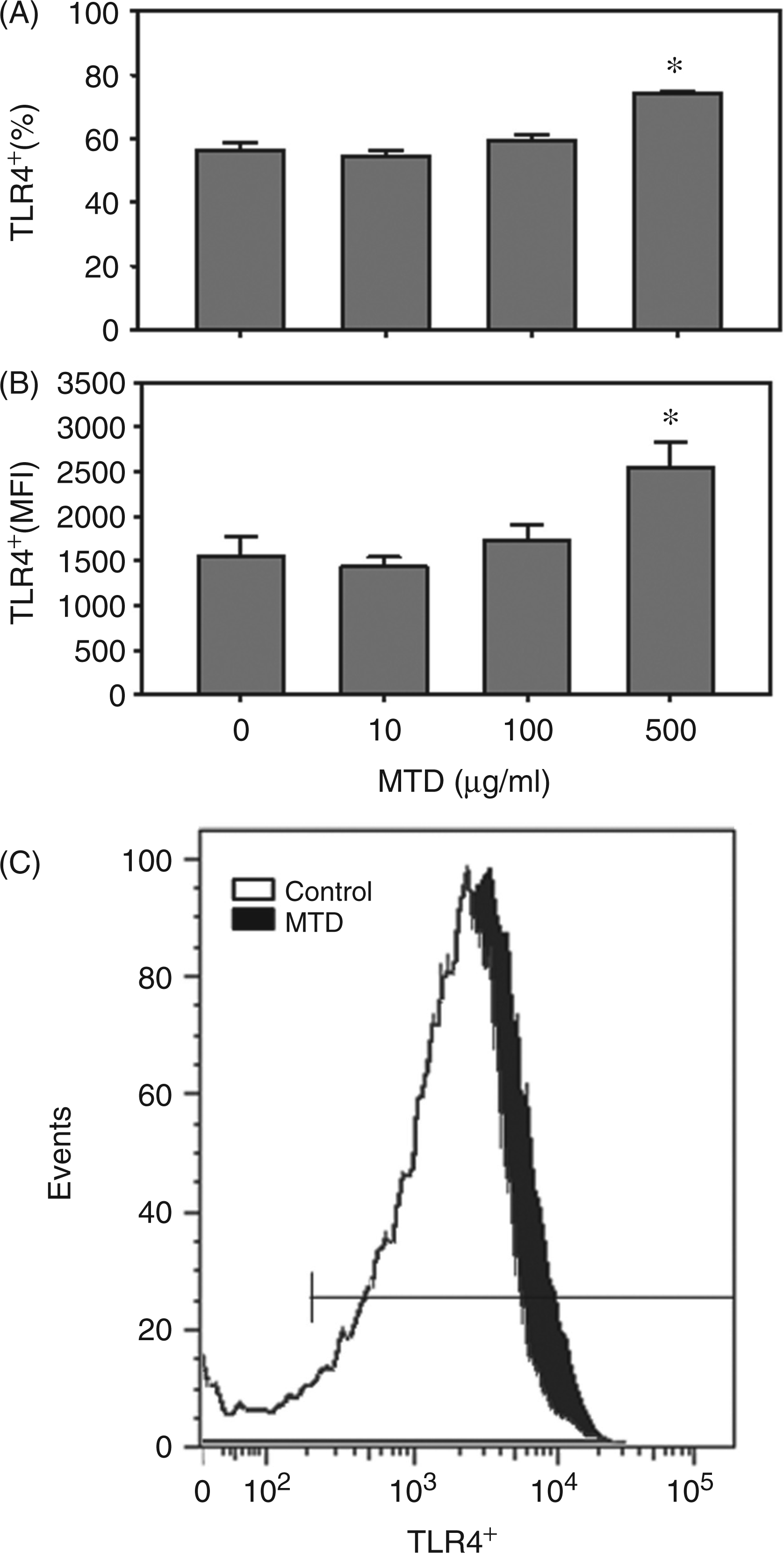
In vitro activation by MTDs resulted in a marked increase in the percentage of cells positive for both TLR2 and TLR4 (Figure 3A). Further analysis of TLR2 and TLR4 expression revealed that under naive conditions comparable percentages of γδ T-cells expressed either TLR2 alone, TLR4 alone or both TLR receptors (Figure 3B). While a significant percentage of the cells following MTD activation remained TLR2+TLR4−, only a minor percentage (5%) of the cells were positive for only TLR4 expression under such conditions (Figure 3B).
Effect of MTD on the co-expression of TLR2 and TLR4 by γδ T-cells. Splenic γδ T-cells were isolated and cultured for 24 h with various concentrations of MTD, as described in the ‘Materials and methods’. The lymphocyte population was identified by forward and side scatter, and analyzed for CD3, γδ TCR and TLR4 expression. Data in (A) show the percentage of γδ T-cells positive for both TLR2 and TLR4. (B) Dot plots of a representative experiment of the effect of MTD on TLR2 and TLR4 in the CD3+ γδ TCR+ cell population. Data are mean ± SEM for n = 5/group. *P < 0.05 vs control (0 µg/ml MTD).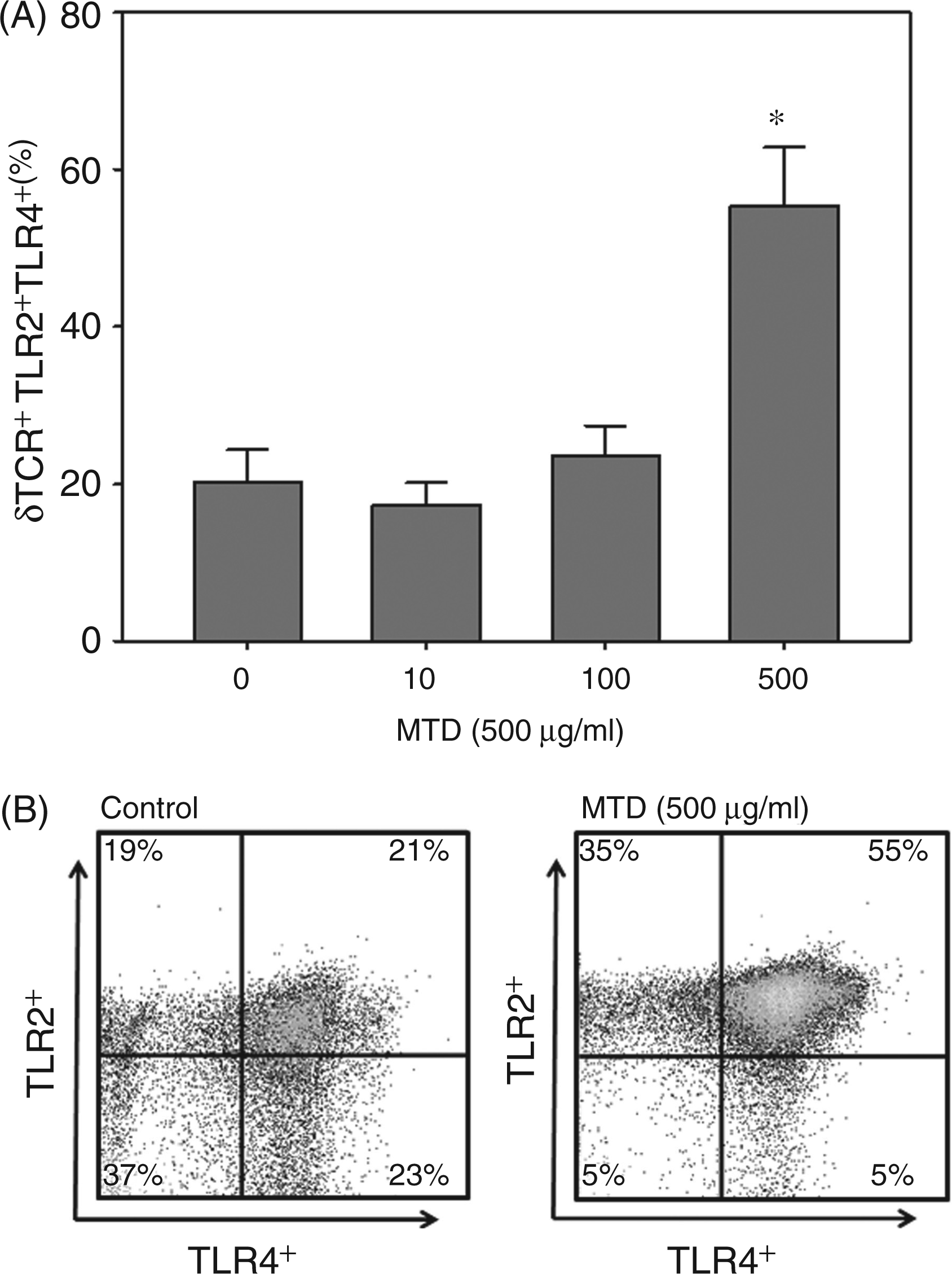
MTD-induced activation of γδ T-cells is associated with increased production of selected cytokines and growth factors
MTD activation of γδ T-cells induced a significant increase in the production of IL-1β, IL-6, IL-10 and RANTES (Figure 4). IL-1β, IL-10 and RANTES production was increased approximately three- to fivefold by MTD activation (500 µg/ml), whereas IL-6 levels were increased up to eightfold compared with the control group (i.e. 0 µg/ml MTD). However, IL-6 levels, while detectable, were quite low and likely to be of little biological significance. Lower concentrations of MTD (100 µg/ml) also induced significant increases in IL-6 and IL-10 production, but not IL-1β and RANTES. The basal release of TNF-α (in the absence of MTD) was relatively high (400 pg/ml) and was not further enhanced by MTD-induced activation. IL-17 levels were barely detectable and unaffected by MTD treatment.
The effect of MTD on cytokine release by γδ T-cells. Supernatants from splenic γδ T-cells cultures after incubation with various concentrations of MTD for 24 h were assayed for the cytokines TNF-α, IL-10, IL-17 and RANTES, as described in the ‘Materials and methods’. Data are mean ± SEM for n = 5/group. *P < 0.05 vs control (0 µg/ml MTD).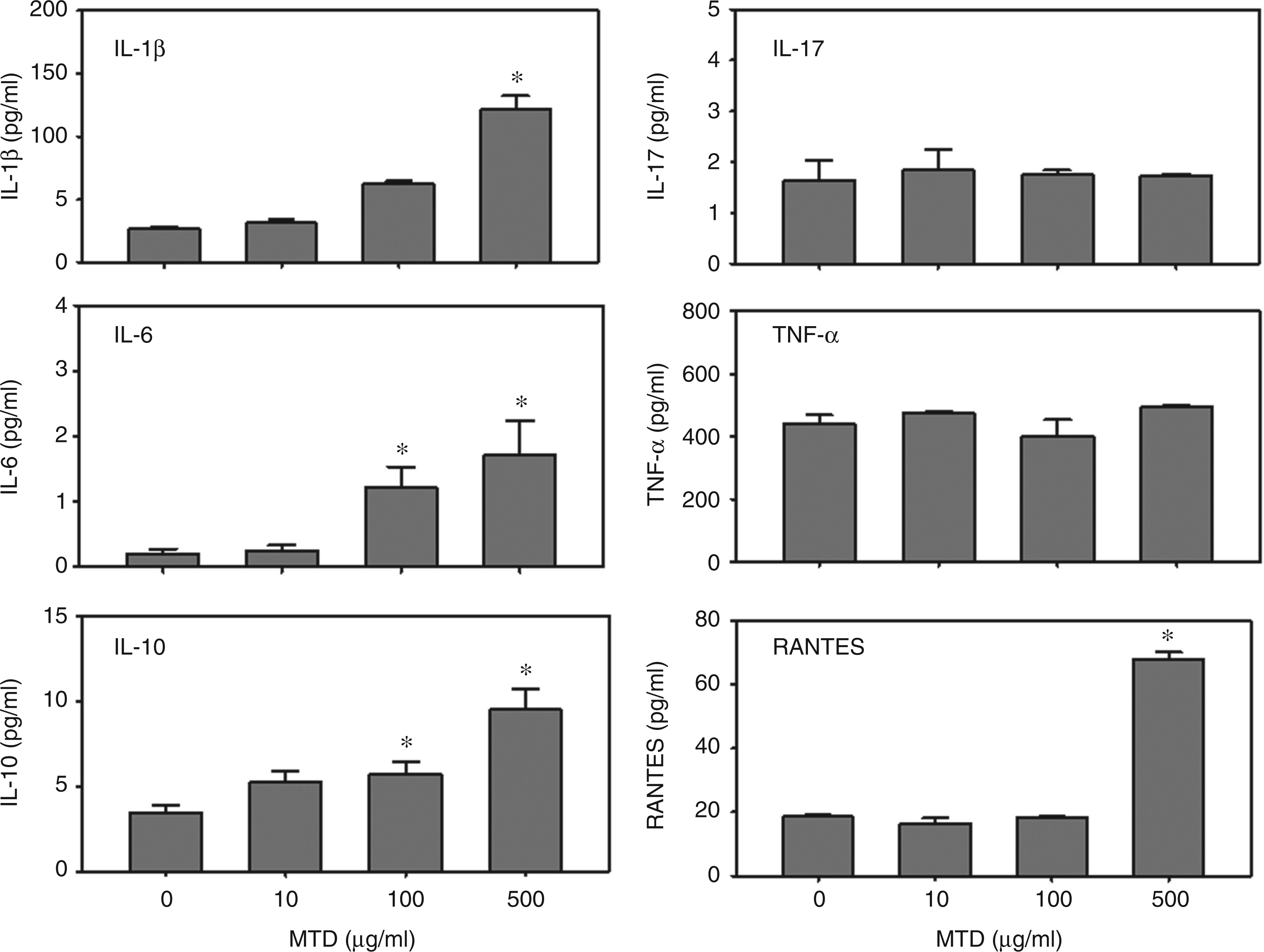
The effect of MTD activation was much greater for growth factors than cytokines (Figure 5). FGF-basic levels were significantly elevated by MTD at the concentrations of 100 and 500 µg/ml. A 20-fold increase in FGF-basic was observed after treatment with 500 µg/ml MTD. VEGF levels were increased more than 30-fold in cells treated with 500 µg/ml MTD. Other concentrations of MTD did not significantly alter VEGF levels.
The effect of MTD on growth factor release by γδ T-cells. Supernatants from splenic γδ T-cells cultures after incubation with various concentrations of MTD for 24 h were assayed for the growth factors FGF-basic and VEGF, as described in the ‘Materials and methods’. Data are mean ± SEM for n = 5/group. *P < 0.05 vs control (0 µg/ml MTD).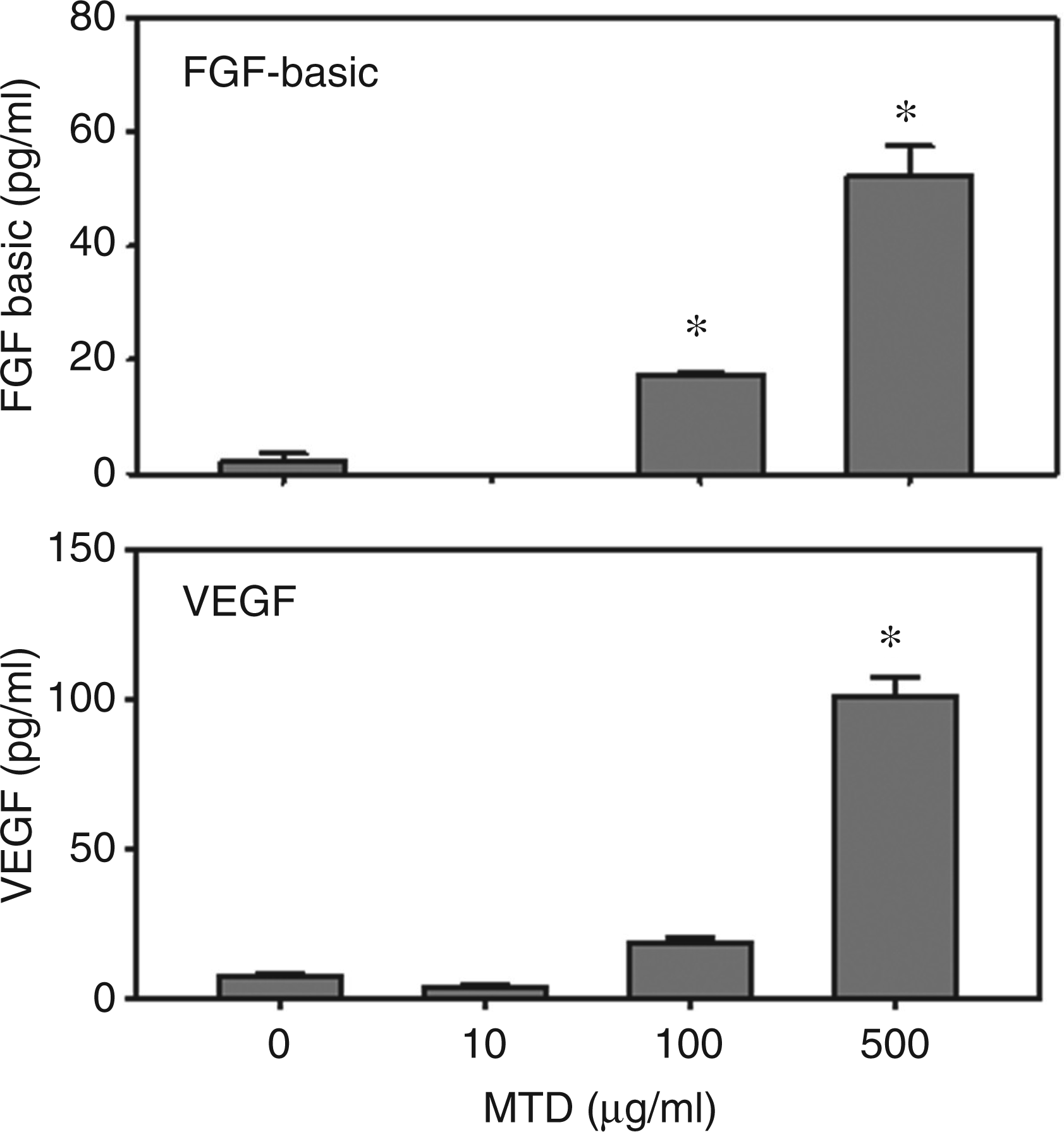
Discussion
Innate immunity activation, while an essential step in the early response to infection, is also important in the clearance of injured tissue and the initiation of tissue repair. Activation of the innate immune system involves multiple immune cells that initiate and sustain the inflammation. While macrophage and neutrophils are traditionally thought of as the primary cells of the innate immune system, a unique T-cell population, γδ T-cells, has been shown to be important in innate immunity and to have a critical role in inflammation and tissue repair.4,5,7,21 While previous experimental and clinical studies have shown that this T-cell population is activated following injury7,22,23 these studies did not identify the mechanism(s) by which this occurs. Our findings herein support the concept that the MTDs released after tissue injury may play an important role in γδ T-cell activation under such conditions.
Cellular injury can lead to release of intracellular molecules called DAMPs.24,25 DAMPs are derived from a large array of cellular components, including the plasma membrane, nucleus, endoplasmic reticulum and cytosol. Interestingly, mitochondria have recently emerged as other organelles that function as a source of DAMPs. 26 This is consistent with the endosymbiotic theory that mitochondria are of prokaryotic origin evolving through symbiosis with microorganisms. It is further supported by the existence of common recognition patterns for DAMPs and PAMPs on TLRs. A wide range of molecules of mitochondrial origin, including mtDNA, N-formyl peptides, cardiolipin, cytochrome C, carbamoyl-phosphate synthase 1 and ATP have been identified as DAMPS, which are recognized by a number of different receptor types, including TLRs.19,26–29 DAMPs are immunostimulatory leading to inflammation in the absence of infection (i.e. SIRS). SIRS, while well recognized, remains poorly understood. From a clinical perspective, its presentation can be indistinguishable from that of infection/sepsis. Inflammatory complications, such as acute lung injury, adult respiratory distress syndrome and multiple organ dysfunction syndrome are often common with SIRS. 30
The majority of studies have focused on the role of DAMPs in activating innate immune cells, such as monocytes, neutrophils and dendritic cells. However, in addition to these traditional innate immune cells, a T-cell subset, γδ T-cells also express TLRs.31–33 In our recent study 22 we observed an increase in the number of circulating γδ T-cells with elevated TLR2 and TLR4 expression early after burn injury, a time when elevated levels of MTDs due to the massive tissue trauma associated with burn would be likely. Interestingly, later post-injury (7 d) γδ T-cell TLR expression was not elevated, a time when elevated levels of DAMPs would not be likely. Recent studies have demonstrated activated γδ T-cells in the circulation of patients with SIRS, demonstrating an important role for γδ T-cells in the early response to injury. 23 This T-cell subset is also an important source of chemokines early post-burn, as plasma/tissue chemokine levels were attenuated in γδ T-cell deficient mice. 9 Nonetheless, while tissue injury is known to release mitochondrial peptides into the circulation,34,35 the concept that changes in circulating levels of MTDs are responsible for the activation of TLR expression after burn are merely speculative at this time.
In the current study we chose to focus on the expression of TLRs. TLR2 and TLR4 are present on the cell membrane surface, and would be the initial receptors to respond to MTDs. Previous studies have demonstrated an up-regulation of TLR2 and TLR4 on circulating γδ T-cells after burn—a condition associated with massive tissue damage, which would generate large amounts of MTDs. 22 A wide range of DAMPs, including HMGB1, are believed to act through TLR2 and TLR4. 36 The fact that we observed a difference in TLR2 and TLR4 expressions after exposure to MTDs that correlated with elevation in specific cytokine responses is highly suggestive of a causative relationship. Thus, MTDs may possess new novel DAMPs that act through TLR2 and TLR4. Nonetheless, the presence of mtDNA in MTDs suggests that the activation of γδ T-cells by MTDs could also be through TLR9. TLR9 has been shown to recognize unmethylated CpG motifs in DNA molecules. CpG sequences are common in bacterial genomes, and thus common in mtDNA. Unlike TLR2 and TLR4, TLR9 is expressed intracellularly within the endosomal compartments. Zhang et al. 19 have shown that MTD-mediated activation of p38 mitogen-activated protein kinase in neutrophils was TLR9-dependent, but that MTD was ineffective in inducing cytokine release in the absence of other activators (i.e. N-formyl-methionyl-leucyl-phenylalanine (fMLP)). Thus, TLR9 activation alone may not be sufficient to induce a biological response by the cell. Nonetheless, future studies will need to evaluate TLR9 expression by γδ T-cells after MTD exposure, as the presence/role of TLR9 in γδ T-cell activation is unknown.
The inflammatory process is believed to be instrumental in supplying growth factors, cytokines and chemokines that orchestrate the cell movement necessary for wound repair.37,38 Thus, inflammation is a critical and necessary component of successful wound healing. Previous studies have led to the development of the concept that epithelial γδ T-cells survey for general signs of epithelial injury or stress rather than recognizing specific antigens. Studies have demonstrated that epidermal γδ T-cells recognize antigen expressed by damaged or stressed keratinocytes and produce chemokines and growth factors upon stimulation.39,40 Jameson et al. 21 have shown that mice deficient in γδ T-cells had defects in keratinocyte proliferation and tissue re-epithelialization. Our findings have shown that mice deficient in γδ T-cells have a delayed healing of the burn wound site that was associated with suppressed growth factor levels and cellular infiltration.10,11
Wound healing is a dynamic process that involves complex interactions between the extracellular matrix, soluble factors, a variety of resident cells and infiltrating immunoinflammatory cells. The initial goal in repair is to achieve tissue integrity and homeostasis. 41 The cellular responses to injury are multi-faceted, involving cell–cell and cell–matrix interactions, as well as the crosstalk between different cell populations through soluble factors. With major tissue trauma, such as burn, the release of high local levels of MTDs, as well as other DAMPs, would be anticipated. Our findings herein show that MTDs activated the release of cytokines and growth factors by γδ T-cells. We observed a degree of specificity with regard to the MTD mediated cytokine production. In particular, IL-1β, IL-10, RANTES and growth factors were up-regulated, whereas other pro-inflammatory cytokines, such as TNF-α and IL-17, were not, and the low levels of IL-6 observed (∼2 pg/ml) are likely of little biological significance. It can be speculated that RANTES release serves as a chemokine for the recruitment of other T-cells to an injury site. IL-10 acts to quell the initial massive pro-inflammatory response to injury that includes IL-1β, and the growth factors FGF-basic and VEGF facilitate early tissue remodeling. In support of this concept, our preliminary findings show that the burn wound is heavily infiltrated with αβ T-cells early after injury 42 and other studies show that γδ T-cells are important in the regulation of growth factors in the burn wound. 10 We observed that basal levels of TNF-α were already high in the absence of MTDs and it can be speculated that further induction was not possible. The inability of MTDs to activate the cells for IL-17 is unclear, as γδ T-cells are potent producers of IL-17.43–45 Nonetheless, multiple signals are required for differentiation of Th-17 T-cells,46,47 which might not be present in a purified cell culture system, such as the one we employed. γδ T-cells are also heterogeneous, 44 and the splenic γδ T-cells may not be of the Th-17 lineage. Future studies will need to address this question, possibly through the use of more complex in vivo models of MTD-induced inflammation.
Conclusions
Our findings presented herein show that γδ T-cells can be activated by MTDs, which are likely to be in high concentrations at a site of tissue injury. Thus, the activation of this unique T-cell population can lead to the elaboration of chemokines and growth factors, which orchestrate the early phases of tissue remodeling. Moreover, the up-regulation of TLRs on γδ T-cells by DAMPS is likely to make them more responsive to activation by MTDs, as well as other DAMPs, present at a site of tissue injury. We hypothesize that release of MTDs from injured tissue is a critical link between tissue injury and a γδ T-cell dependent initiation of sterile inflammation, and subsequent tissue remodeling response.
Footnotes
Funding
Support was provided by NIH grant GM079122.
Acknowledgements
These findings were presented in part at the 99th annual meeting of the American Association of Immunologists, Boston, MA, USA, May 2012. The opinions or assertions contained herein are the private views of the author and are not to be construed as official or as reflecting the views of the Department of the Army or the Department of Defense. The authors declare that they have no competing interests.
