Abstract
Severe traumas are associated with hypercortisolemia due to both disruption of cortisol secretion rhythm and increase in its total concentration. Understanding the effects of altered cortisol levels and rhythms on immune function is of great clinical interest, to prevent conditions such as sepsis from complicating the recovery. This in vivo study assesses the responses of circulating leukocytes to coupled dose and rhythm manipulation of cortisol, preceding an immune challenge induced by endotoxin administration. Through continuous infusion, plasma cortisol concentration was increased to and kept constant at a level associated with major physiologic stress. In response, transcriptional programming of leukocytes was altered to display a priming response before endotoxin exposure. Enhanced expression of a number of receptors and signaling proteins, as well as lowered protein translation and mitochondrial function indicated a sensitization against potential infectious threats. Despite these changes, response to endotoxin followed very similar patterns in both cortisol and saline pre-treated groups except one cluster including probe sets associated with major players regulating inflammatory response. In sum, altered dose and rhythm of plasma cortisol levels engendered priming of circulating leukocytes when preceded an immune challenge. This transcriptional program change associated with stimulated surveillance function and suppressed energy-intensive processes, emphasized permissive actions of cortisol on immune function.
Introduction
Healthy function of the immune system is intrinsically tied to the biological rhythms running in harmony. There is a bi-directional information flow between immune and neuroendocrine systems such that while a wide range of immune parameters goes through daily fluctuations under the control of autonomic and endocrine rhythms, immune effectors, such as cytokines, can, in turn, influence the phase of these rhythms. 1 In the case of acute stress, such as trauma or infection, an inflammatory response is mounted by the coordinated action of immune and neuroendocrine systems. The hypothalamus–pituitary–adrenal axis is a crucial mediator of this action because of its control on circadian and ultradian glucocorticoid (GC) secretion patterns. 2 These fluctuating patterns are hypothesized to provide the basis for a ‘continuous dynamic equilibration’ that is fundamental to the establishment of homeostasis and allostasis. 3 Cortisol is the major circulating human GC which, through feedforward and feedback regulatory loops, maintains this dynamic state that is essential for proper response to stressors. Through mathematical modeling of these regulatory mechanisms, our group previously showed that the ultradian oscillations of GC concentration are critical for maintaining the homeostatic balance on downstream responses. Mean levels of GC responsive mRNA synthesis was shown to be greater when the pulsatility in plasma cortisol levels was diminished despite a constant total amount of GC exposure. 4 Besides cortisol secretion frequency, parameters such as concentration and the timing with respect to a stressor, also affect the characteristics of stress response, contributing greatly to the emergence of inter-individual variations in reaction to similar stressors. 5
GCs are mostly involved through their immunosuppressive and anti-inflammatory actions, which have been clinically utilized for the treatment of autoimmune and inflammatory disorders for decades; however, the very first recognized view on GC physiology in the 1940s by Selye suggested that release of endogenous GCs enhances and mediates the response to external stressors, rather than limiting it. This early statement was later reshaped and extended by emerging anti-inflammatory actions of GCs 6 and a view that synthesizes both permissive and suppressive actions of GCs on immune function was put forward. This view proposes that GCs can have disparate effects on the individual’s response to a stressor depending on the timing, duration or concentration of the exposure. These can include permissive, suppressive, stimulatory or preparatory effects. With regard to the concentration of GCs, only permissive effects on immune function are typically associated with basal levels of GCs, whereas the other three effects (suppressive, stimulatory or preparatory) are commonly observed when GC concentration is at high or stress-induced levels. However, permissive actions can also be associated with higher cortisol levels if these instances precede an acute stressor. 7 Concentration-dependent specific actions of GCs are exemplified by an in vivo study in which low-dose corticosterone administered to rats stimulated an inflammatory phenotype in macrophages, while high-dose corticosterone acted in a purely immunosuppressive fashion. 8 Related to the temporal dependence of a GC surge and the incidence of an acute stressor; research has shown contrasting effects even in the same concentration range. When GC administration preceded an immune challenge (i.e. LPS exposure) in rats, both peripheral and central pro-inflammatory responses were potentiated as reflected by increased cytokine secretion. However, when the same concentration of GCs was administered after the inflammatory stimulus, attenuation of the inflammatory reaction was observed. 9
As GCs are not only major endogenous hormones regulating the stress response but also pharmacological agents that are used clinically for the treatment of many inflammatory diseases, building a solid understanding of their effects on immune cells is of primary importance. Studies have previously investigated the effects of varying doses of GCs on the response to LPS challenge in healthy humans, establishing both permissive and stimulatory roles of GCs, as determined by the measurement of circulating cytokines.10,11 Study of a single immunological parameter is not adequate to define the complexity of immune system. System-wide approaches, such as global transcriptional profiling, provide a wider view of the physiological alterations in response to stimuli, enabling better definition of complex biological phenomena. 12 This approach has been used to profile transcriptional responses of blood cells stimulated by GCs ex vivo.13,14 Importantly, major physical and psychological traumas are associated with hypercortisolemia, which emerges as a result of the disruption of the cortisol secretion rhythm and the increase in total cortisol concentration.15,16 Understanding how this alteration challenges the immune system and its function is of great clinical interest, and is particularly relevant to the management of critically ill or injured patients, in order to prevent complications. The present in vivo study seeks to assess the transcriptional responses of whole blood leukocytes [white blood cells (WBCs)] to coupled dose and rhythm manipulation of cortisol preceding an immune challenge modeled by LPS administration. A healthy human model of LPS administration has been used as a reproducible experimental platform providing mechanistic insights into how cells and organs respond to systemic inflammation. The model transiently alters many physiologic and metabolic processes in a manner that is qualitatively similar to those observed after acute injury and systemic inflammation. 17 It therefore serves as a suitable platform for investigating the changes in immune response due to hormonal manipulations. The current study design involves blunting the natural circadian rhythm of cortisol in healthy volunteers by continuous cortisol infusion for 30 h at a concentration that mimics hypercortisolemia induced by a major physiologic stressor. At hour 24 of this infusion period, LPS was administered as a bolus injection and transcriptional responses of leukocytes were investigated by gene expression analysis for 24 h periods both before and after the LPS challenge. Differential gene expression and clustering analyses were done separately for these two periods to allow focusing both on the response to GC infusion alone and on the effects of this treatment to subsequent LPS exposure.
Materials and methods
Human participants
Nine healthy participants aged between 18 and 40 yr were recruited by public advertisement for participation. All participants provided written, informed consent under guidelines approved by the Institutional Review Board of Robert Wood Johnson Medical School. Inclusion criteria were being 18–40 years of age and having ‘normal general health’, as demonstrated by medical history and physical examination, as well as laboratory testing. Women of child-bearing potential were screened for pregnancy risk and were included if utilizing reliable contraception. Participants underwent an initial screening visit that included the comprehensive history, physical and laboratory testing to establish suitability for inclusion; eligible participants returned within 3 wk for admission to the clinical research center.
Volunteers were randomized to receive intravenous saline (n = 4) infusion or continuous cortisol infusion at a dose of 3 µg/kg (n = 5) for a total of 30 h starting 24 h before endotoxin administration. Participants in both groups were intravenously administered 2 ng/kg endotoxin (CC-RE, lot 2) as a bolus dose to induce moderate systemic inflammation at 0 h (09:00 clock time), as previously described.17,18 Vital signs and blood samples were obtained and recorded at various times both pre- and post-endotoxin challenge. Throughout admission, all participants were attended by nursing staff on a 24-h basis. Subjects were precluded from any physical activity other than walking about the study unit.
Blood-derived plasma was analyzed by radioimmunoassay for plasma cortisol concentration, as previously described.
19
Whole blood total leukocyte levels and differentials were measured by standard laboratory procedures at the Robert Wood Johnson University Hospital clinical pathology laboratories. Blood samples were collected and leukocytes were isolated at multiple time points throughout a 48-h period (indicated in Figure 1) starting with the cortisol/saline infusion and ending 24 h after LPS administration. Following RNA extraction and cRNA synthesis, total cellular RNA was hybridized on to a GeneChip Human Genome Focus Array (Affymetrix, Santa Clara, CA, USA), as described previously.
20
Study design: nine healthy participants received continuous cortisol (or saline) infusion starting 24 h before the bolus intravenous endotoxin (LPS) injection and continuing until 6 h after the injection. Blood samples were collected and leukocytes were isolated at multiple time points over 24 h both before and after LPS administration.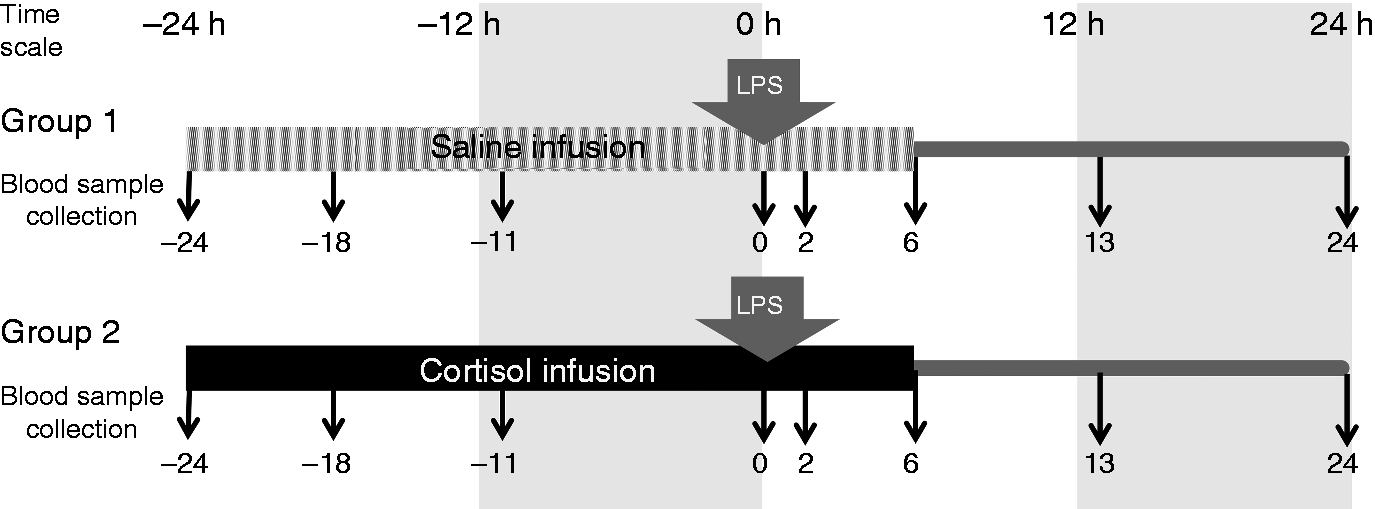
Microarray data analysis
Data analysis included extraction of expression values, identification of treatment group members, filtering for probe sets of interest and clustering. DNA chip analyzer (dChip) software was used to generate expression values from the microarray data, using invariant-set normalization and perfect match modeling. Probe sets of interest were identified using software for the extraction and analysis of gene expression (EDGE). 21 EDGE was used to determine probe sets differentially expressed over time for all participants. 22 Principal component analysis (PCA) was then performed using these probe sets, and the first principal component (PC1) was plotted versus time for each participant. The resulting projections were clustered in MATLAB using wavelet model-based clustering to identify the members of the two treatment groups. EDGE was then used to identify probe sets differentially expressed between the two treatment groups. Finally, the filtered data sets were clustered using ‘consensus clustering’, to identify subsets of the probe sets with common coherent expression patterns. 23 Functional annotation of these probe sets was done by using the pathway enrichment function in Ingenuity Pathway Analysis (IPA) tools (Ingenuity Systems, www.ingenuity.com), as well as analyzing functions of each individual gene corresponding to the probe sets, using databases of IPA and Kyoto Encyclopedia of Genes and Genomes.
Promoter analysis
Promoters of genes were extracted from a rich database of promoter information (Genomatix). In order to identify cortisol-responsive genes, we examined the presence of GC response elements (GREs) on gene promoters. Conserved regions across orthologous promoters of the selected genes were first identified and then the GRE binding site motif (TGTTCT 24 ) was scanned against those conserved promoter regions (detailed methodology in Nguyen et al. 25 ). The routine was applied to all genes associated with the clusters obtained by analyzing the −24 h to 0 h time period.
Results
This study aims to elucidate the effects of coupled dose and rhythm manipulation of plasma cortisol levels on the transcriptional changes that occur in response to acute LPS exposure in humans. The study design, which is schematically shown in Figure 1, involves two groups of healthy participants treated either with cortisol or saline infusion for 30 h. Both groups were administered a bolus dose of LPS 24 h after the initiation of infusion. The blood samples were collected from the participants at multiple time points and transcriptional response of leukocytes were determined via microarray analysis. In addition, blood cell counts and plasma cortisol concentrations were monitored.
Plasma cortisol levels, total number of leukocytes and leukocyte differentials
Baseline (before −24 h) plasma cortisol concentrations were 10–15 µg/dl for both the saline + LPS and cortisol + LPS groups. After cortisol infusion commenced at −24 h, plasma cortisol levels increased to ∼ 35 µg/dl in the cortisol + LPS group, but fluctuated between 2 to 15 µg/dl levels in the saline + LPS group, as expected from its natural circadian rhythm (Figure 2). After LPS administration at 0 h, there was no further elevation in the plasma cortisol concentrations for cortisol + LPS group; however, for the saline + LPS group, there was a steep increase in the cortisol concentration from a mean of 10 µg/dl up to 23 µg/dl within the first 3 h of LPS administration, after which plasma cortisol levels started to return to normal. For the cortisol + LPS group, cortisol concentration fluctuated around 30–35 µg/dl until the infusion period was ended 6 h after LPS administration and returned to normal in the following 7 h.
Change in the level of plasma cortisol (A) and total number of WBCs (B) and percentage of WBC subpopulations (C–E) in response to continuous cortisol infusion and LPS administration (dashed line: saline + LPS; solid line: cortisol + LPS).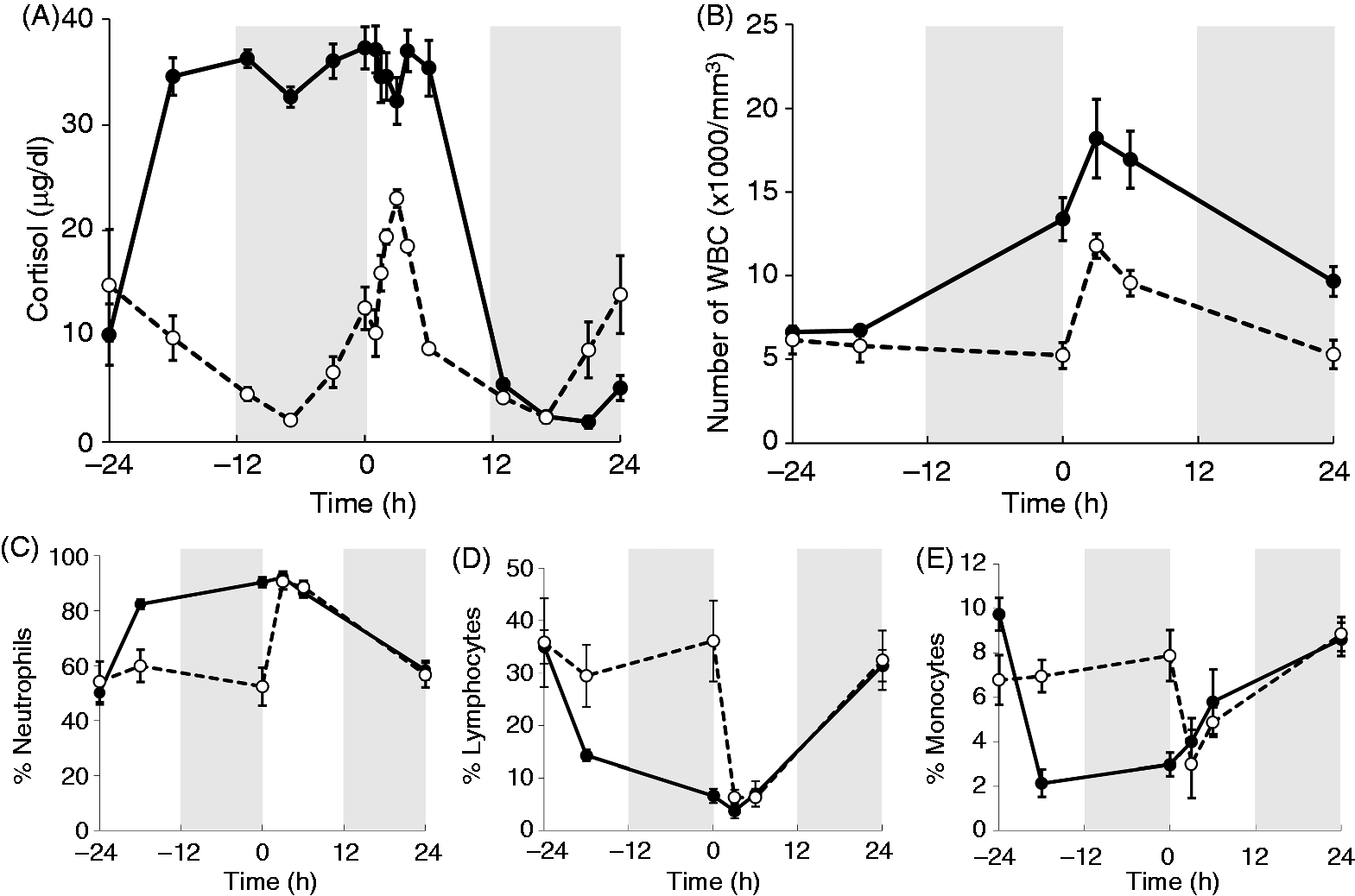
There was a significant difference between saline- and cortisol-treated groups in the total number of leukocytes at time 0. High concentration and altered rhythm of cortisol by continuous infusion resulted in a significant alteration in both the total number of leukocytes (WBCs) and the WBC differentials. The total number of cells at 0 h (before LPS administration) was at a level comparable to that observed after LPS administration for the saline-infused control group with a relative increase in the percentage of neutrophils, and a significant decrease in the percentage of lymphoctyes and monocytes (Figure 2C–E). Following LPS administration, both groups demonstrated an increase in the number of WBCs at a similar rate (Figure 2B). Interestingly, the leukocyte subpopulations as a percentage of the total leukocyte count were not significantly different by 3 h post LPS administration.
Identification of pre-treatment groups from gene expression data
We performed an initial evaluation of the microarray data in which we allocated each participant into one of the two groups according to the gene expression patterns, while initial assignments of the participants were kept anonymous. For this purpose, first, the probe sets, which were differentially expressed over time for all participants, were identified (P < 0.01, q < 0.01), resulting in 799 probe sets of interest out of 8793 total. Then, PCA was performed on the differentially expressed probe sets. The first principal component (PC1), which accounted for ∼ 50% of the total variance, was plotted over time for each of the nine participants and the resulting profiles were then clustered. Averaged PC1 profiles of two resulting clusters are shown in Figure 3. The final assignment according to these clusters, which was later verified by the physicians who undertook the study, placed four participants in one group (saline pre-treated, dashed line) and five in the other (cortisol pre-treated, solid line).
Averaged first principal components (PC1) for saline- (dashed line) and cortisol- (solid line) treated groups.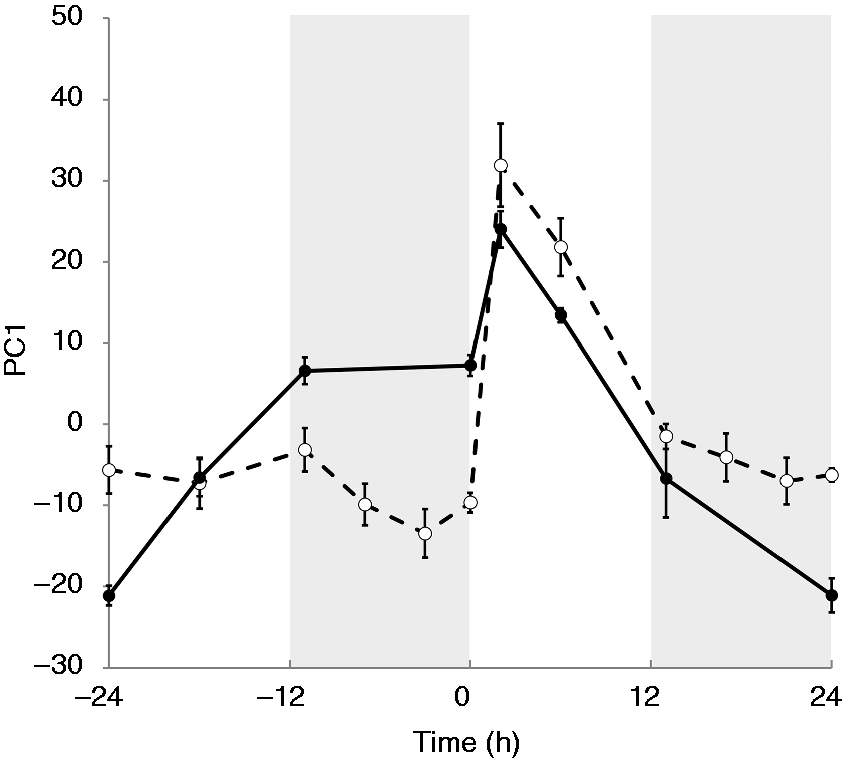
Transcriptional effects of manipulated plasma cortisol levels
After the initial evaluation of the data, the focus of the analysis was narrowed down to the first 24 h, where the response to cortisol infusion was independent from LPS exposure. In the time period between −24 to 0 h, we identified 332 out of total 8793 probe sets to be differentially expressed between cortisol- and saline-treated subjects (P < 0.05 and q < 0.05). According to their temporal expression profiles, 200 out of 332 of the probe sets were classified into six clusters by consensus clustering analysis.
25
Resulting clusters were further grouped according to whether cortisol group shows up- or down-regulation with respect to saline group, as shown in Figure 4.
Transcriptional responses of leukocytes to cortisol prior to endotoxin administration in time period between −24 h and 0 h.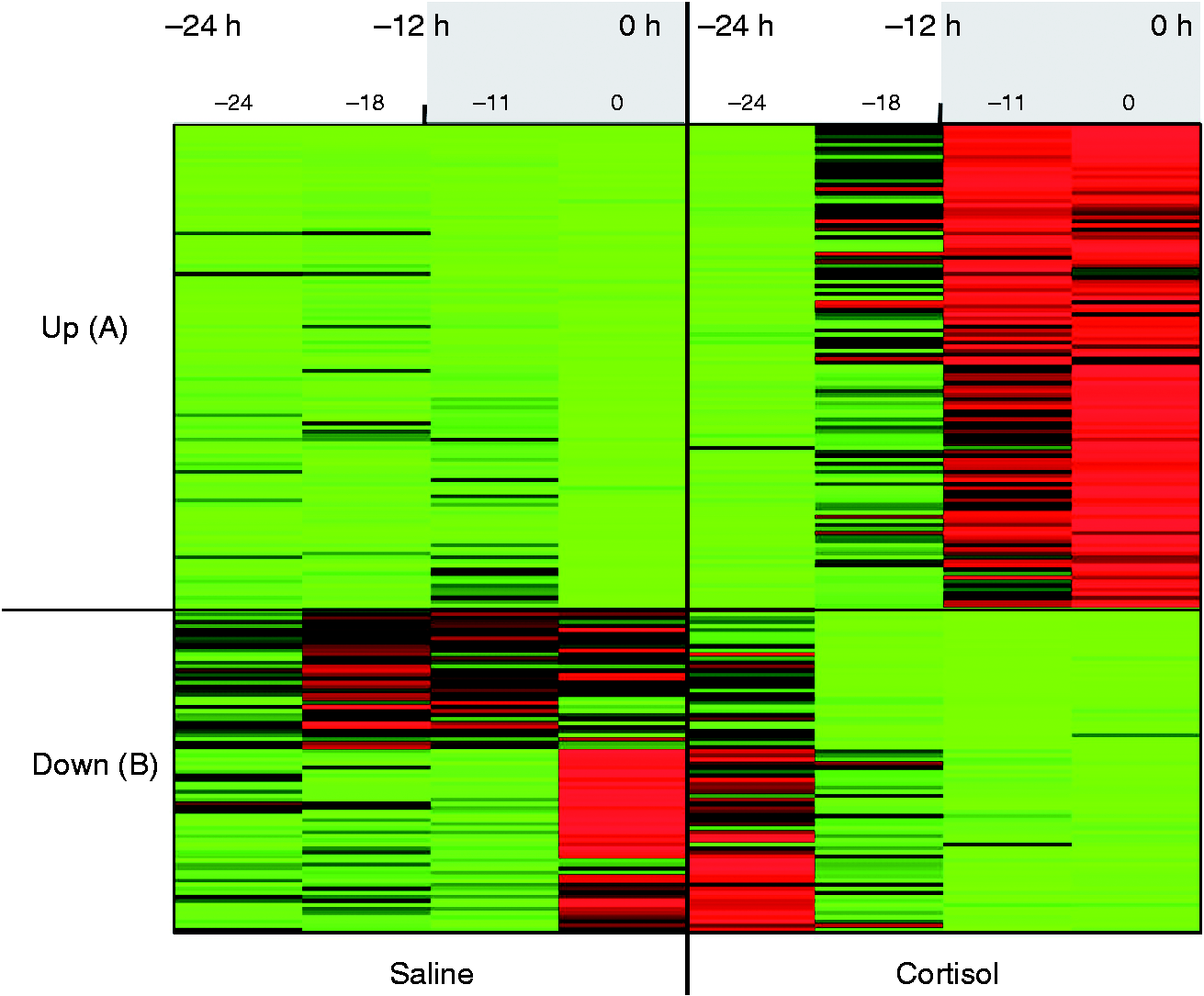
Functional annotation and representative probe sets associated with up- and down-regulated clusters in response to continuous cortisol infusion in the −24 h to 0 h time period.

Within the genes associated with these clusters in the −24 h to 0 h period, we performed a promoter analysis by scanning for the GRE binding site (TGTTCT motif) on conserved regions across sets of orthologous promoters. Among 200 genes, there were 82 genes that contained a GRE-binding site, 50 genes with no GRE-binding site and 68 genes that had to be excluded owing to limitation of (orthologous) promoter information. Genes that are identified to have GRE binding sites within their promoter regions did not show any particular directionality in regulation. Distribution of the genes to up- and down-regulated clusters had almost equal weights. The genes included in the promoter analysis are listed in Supplementary Tables S2 S3, together with their distribution to up- and down-regulated clusters and basic classifications of corresponding proteins.
Overall response to LPS
After concentrating on the effects of cortisol on leukocyte transcription dynamics, the analysis was extended to cover the entire duration of the study in order to comprehensively evaluate the response to LPS and how it is affected by preceding cortisol infusion. One hundred and ninety-nine out of 8793 probe sets were differentially expressed between time points −24 h and 24 h for the two treatment groups (P < 0.05 and q < 0.05). One hundred and fifty-seven out of 199 probe sets were distributed among five clusters according to their expression patterns, which were further grouped according to the direction of the response in the first 24 h of the study as LPS-dominant, cortisol-enhanced and cortisol-suppressed (Figure 5A–C).
Overall differential response to LPS between cortisol + LPS and saline + LPS groups. Five clusters obtained by consensus clustering were grouped into three based on response to cortisol between −24 h and 0 h as (A) LPS-dominated (17 probe sets), (B) cortisol-enhanced (74 probe sets) and (C) cortisol-suppressed (66 probe sets).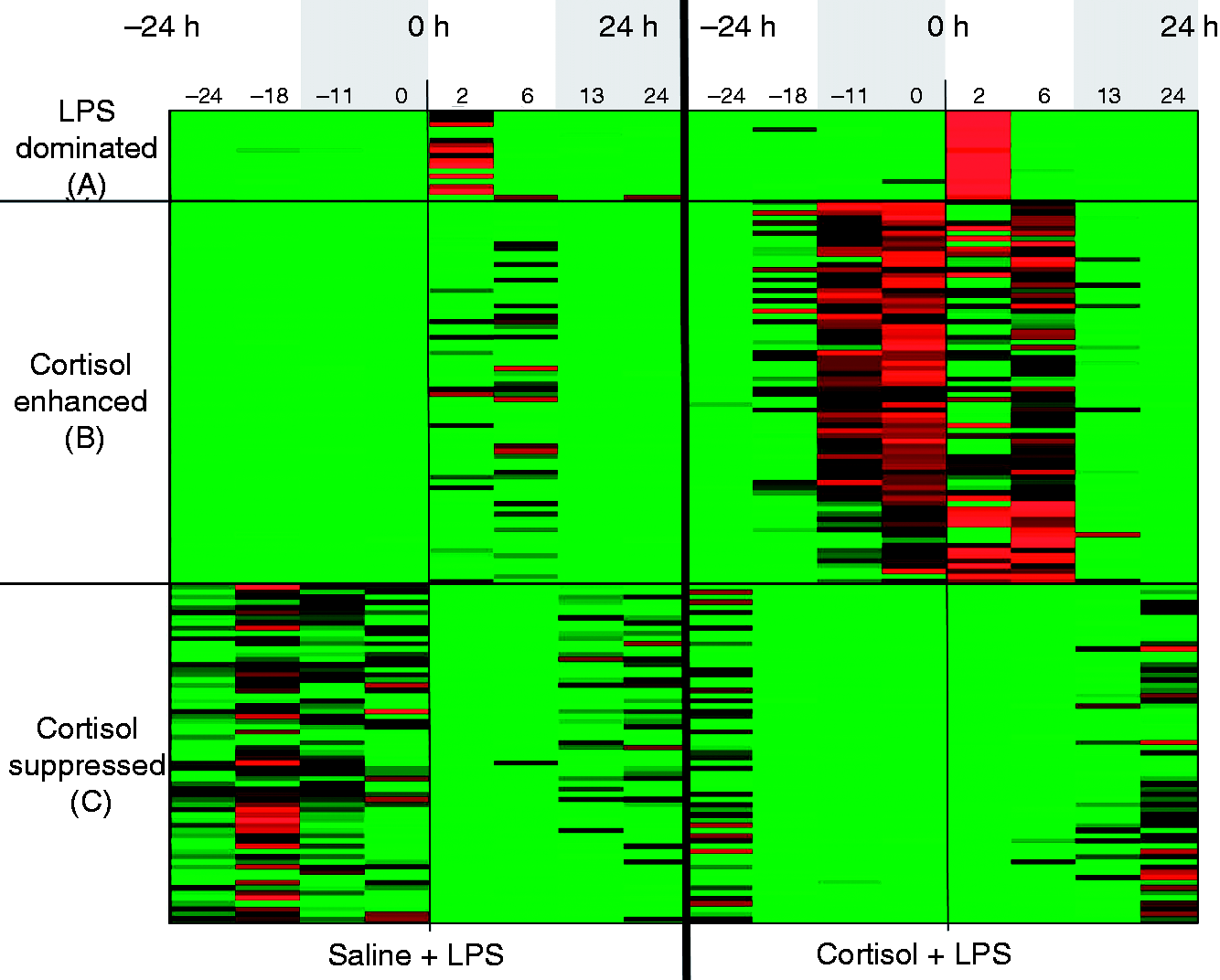
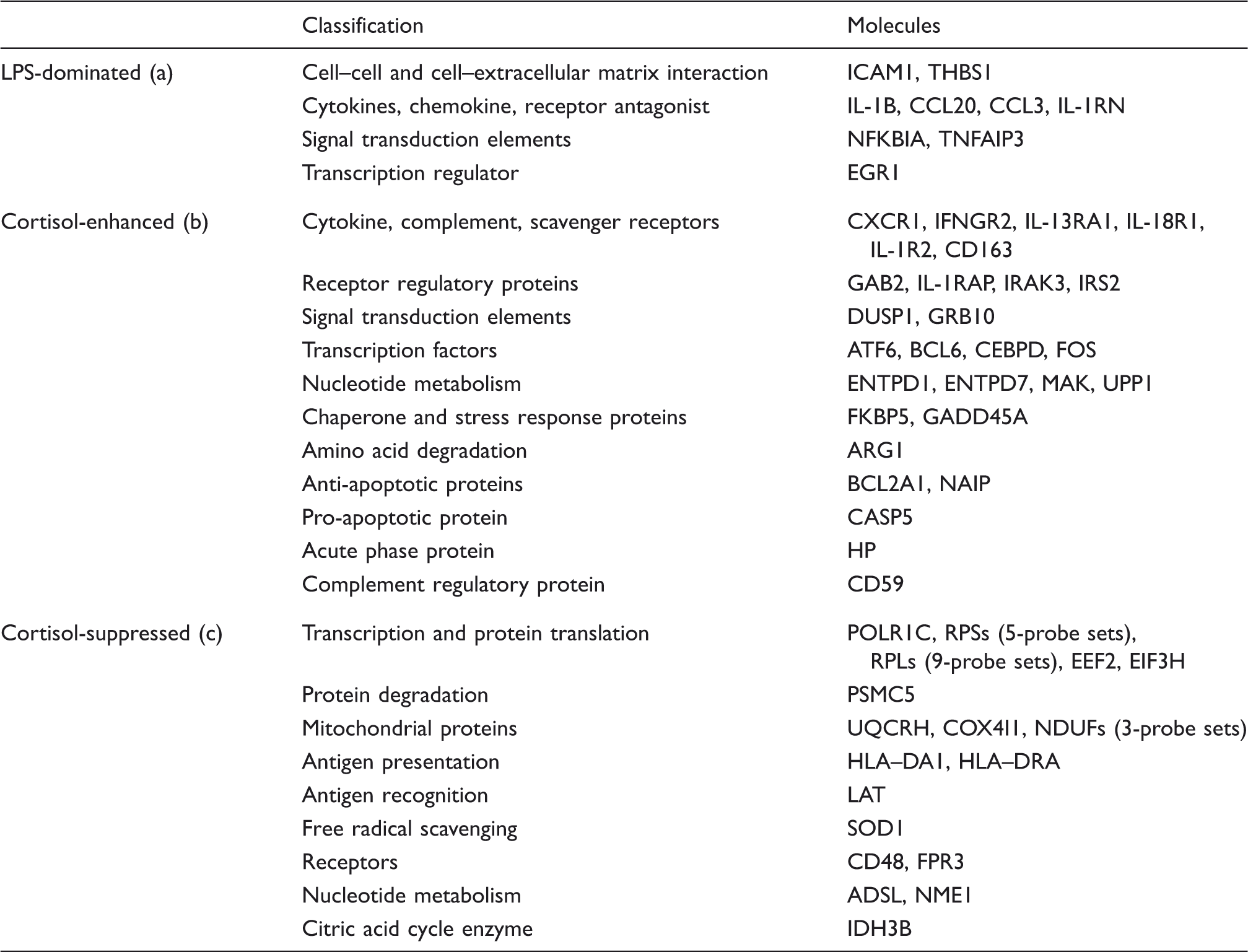
Functional annotation and representative probe sets associated with up- and down-regulated clusters in response to continuous cortisol infusion within the full time period of the study (–24 h to 24 h).
Discussion
The design of the current study involved blunting the natural circadian rhythm of cortisol in healthy volunteers by continuous cortisol infusion for 30 h. This increased and maintained plasma cortisol concentrations at a level associated with major physiologic stress. 26 At hour 24 of this continuous infusion period, LPS was administered as a bolus injection. Transcriptional response of leukocytes was monitored by microarrays, and data were analyzed specifically for cortisol infusion period in conjunction with the whole time period. Both GCs27–29 and LPS17,30–32 are known to alter the total and differential peripheral blood leukocyte counts. As gene expression analysis in this study was conducted on peripheral blood leukocytes without any process for separating specific cell subtypes, observed transcriptional changes represent an omnibus response that reflects both cellular responses and changes in subpopulations of cells in peripheral blood.
Plasma cortisol levels showed the expected circadian pattern 26 in control participants and rapidly increased following LPS administration. In cortisol-infused participants, cortisol levels were steady throughout the 30 h infusion period around twice the normal daily peak level of control participants (∼ 35 µg/dl), and did not show any further increase upon LPS administration. Cortisol concentration returned to normal levels by 12 h in both groups. This pattern shows that the followed dosing strategy successfully blunted the natural rhythm of cortisol and clamped the concentration at a level associated with stress-induced physiologic conditions. The number of leukocytes was significantly increased in response to continuous cortisol treatment before LPS administration, and there was an additional increase immediately following LPS administration at a similar extent observed in the control participants. This contrasts with previous stress-related studies on rats showing an inverse relationship between plasma corticosterone levels and total number of leukocytes.27,28 One reason for this, apart from obvious difference in species, might be related to the way in which GC levels are manipulated. Animal studies are based on increasing endogenous corticosterone levels by exposing the animals to restrain stress for a short amount of time (1–2 h) to study the acute changes in leukocyte distributions. However, in our study design, cortisol concentrations were fixed at a level associated with stress by exogenous infusion of cortisol. As it has been already shown that not only the concentration, but also pulsatility, duration and timing of cortisol can affect physiological processes in virtually all the organs, 7 this major difference in study designs can be the reason for observing contradictory results.
Transcriptional responses within the first 24 h of the study reflect a number of cortisol-induced changes, the most important of which are likely to be the changes in functional gene regulation and in the leukocyte subpopulations. As a consequence of the continuous cortisol infusion, we observed enhanced expression of genes encoding for a number of pattern recognition and cytokine receptors, receptor regulatory elements and signal transduction elements, as well as reduced expression of the genes encoding for elements of protein translation process, and mitochondrial proteins. Increased receptor and signal transduction protein expression were indicative of a priming effect of the cortisol on the immune function, where cells were sensitized to recognize potential infectious threats or endogenous danger signals. These observations agree well with the previous studies investigating the effects of stress or direct cortisol delivery on the immune function and response to LPS challenge which demonstrated amplified pro-inflammatory receptor expression and function on immune cells33–35 and subsequently enhanced inflammatory response to LPS.8,9,13,36–38
Up-regulation of cytoskeletal proteins and extracellular matrix degradation enzymes also imply an increased mobility and enhanced ability for extravasation to penetrate the tissues or engulf pathogens. These are key events for the inflammatory function of leukocytes and the enhancing effect of cortisol on these functions supports its permissive role on inflammatory response as supported by earlier studies.13,39
Tuning down the protein translation process can also be regarded as part of the priming effect as it points toward re-prioritization of cellular resources and energy, which would presumably allow better energy management when the anticipated threat is encountered. Decreased protein synthesis together with the increased expression of protein degradation enzymes indicates a catabolic state that is characteristic of cortisol action and in line with its role as a stress hormone.7,40,41 Overall, cortisol infusion seems to induce an anticipatory state in the immune cells and have preparatory effects to increase the competency in combatting with the anticipated threat. Although this might seem counter-intuitive considering the fame of GCs for their immunosuppressive and anti-inflammatory actions utilized in clinic, depending on the timing, duration and concentration of the exposure, cortisol is shown to also have permissive and enhancing effects on immune function.7,8,13,35,42,43
Promoter analysis within these genes associated with altered regulation due to constant cortisol concentration indicated that, mechanistically, only less than half of differential expression patterns might have been as a direct consequence of GC receptor (GR)-DNA binding. However, a quarter of the genes do not contain any GRE-binding site in their promoter regions, pointing to the previously recognized regulatory function of GR on other transcription factors via mechanisms other than direct DNA binding, such as tethering.44–46 Furthermore, distribution of these genes with or without GRE-binding site in their promoters within up- or down-regulated clusters indicated that there is possibly no preferential directionality in different modes of transcriptional regulation through GR. This underscores the complexity of mechanisms that govern the transcriptional programming through GR,47,48 which is unsurprising considering the elicited vastly different functional consequences of GCs in the cellular milieu.7,49,50
Despite the significant transcriptional program changes in the leukocytes prior to 0 h, the overall response to LPS did not significantly differ between cortisol- and saline-treated groups, as reflected in the clusters obtained from differentially expressed genes throughout the full study period. Both cortisol- and saline-treated groups displayed similar up- and down-regulation responses as reflected in the 0–24 h periods of the heatmap shown in Figure 5(B, C). These groups of clusters correspond well to the first part of the study, and are therefore named according to the observed response to cortisol as ‘cortisol enhanced’ (Figure 5B) and ‘cortisol suppressed’ (Figure 5C). However, the expression pattern in the cluster shown in Figure 5A substantially differs from the rest. Intensity change within the first 2 h of LPS administration is so steep in this cluster that, even if cortisol effect was present prior to LPS administration, it is overridden by this steep change. The important pro-inflammatory NFKBIA gene, which was in the up-regulated cluster in Figure 4 (and listed in Table 1) for the previous clustering analysis, appears as neutral in this analysis in the same time period. The reason for this is that NFKBIA is a gene predominantly controlled by pro-inflammatory stimuli and although its expression is moderately up-regulated as a result of continuous cortisol infusion, the intense up-regulation after LPS is predominant. Furthermore, the fact that LPS response was much higher for this cluster in the cortisol group when compared with control suggests that while cortisol might not have directly affected transcription of the genes in this cluster, it may have had an indirect effect, perhaps through the modulation of up-stream signaling cascades. The net result is that the response to the subsequent inflammatory challenge (i.e. LPS) was intensified.
As noted above, at the 0 time point, there were significant differences in both the total leukocyte count and the composition of the leukocyte subpopulations. In particular, the subpopulation of neutrophils was larger in the cortisol-treated group (90% of the total leukocyte count) when compared with the control group (55%). This is an important consideration because studies from our own group20,31 and from others 36 indicate that the reported change in whole blood inflammatory gene transcription/translation at early time points (2–4 h) after LPS administration can be attributed mainly to circulating neutrophils. In the present study, however, the leukocyte subpopulations (as percent of the total leukocyte count) were not significantly different by 3 h post-LPS when cortisol and control groups are compared, making it unlikely that the robust LPS response observed in this gene cluster (Figure 5A) can be ascribed simply to the altered cell differential numbers. Rather, the changes may reflect alterations in the maturity of circulating neutrophils (e.g. CD16dim 51 ) or the bone marrow recruitment of neutrophils following LPS challenge in the presence of cortisol. One of the challenges of this study is that the subpopulations of cells were not further investigated or isolated to examine their unique transcription signatures.
The probe sets associated with the LPS-dominant cluster include the major players known to regulate the inflammatory response, such as cytokines and chemokines (Il1B, CCl20, CCL3 and IL1RN), signal transduction elements that negatively regulate pro-inflammatory signaling (NFKBIA and TNFAIP3), and receptors mediating recruitment of leukocytes to sites of inflammation (ICAM, THBS1). Together, these results suggest that continuous cortisol infusion primes circulating and bone marrow leukocytes, increases the efficiency of their surveillance function and dampens their energy-intense cellular processes so that they can more rapidly and efficiently respond to a subsequent inflammatory challenge. Although these changes in the transcriptome coherently point out to the permissive roles of cortisol on circulating leukocytes, this study is limited to describe the resulting phenotype and how inflammatory processes are actually controlled and implemented. It is worth noting that tuning down the protein translation processes by continuous cortisol treatment also inherently implies that produced transcripts might not exactly mirror the protein end products, such as inflammatory cytokines. Therefore, to fully describe the effect of cortisol on immune response, further studies focusing on proteomic and phosphoproteomic profiles of leukocytes, as well as on plasma metabolomics are essential.
Conclusions
Natural daily fluctuation of plasma cortisol concentration in healthy volunteers was blunted by continuous cortisol infusion prior to an immune challenge with a bolus dose of LPS. Transcriptional responses of leukocytes, as well as the change in total leukocyte count in blood and plasma cortisol levels, were monitored over 24 h both before and after LPS administration. By continuous infusion, plasma cortisol concentration was increased to, and kept constant at, around 35 µg/dl, which is a level associated with major physical stress and almost twice as much as the normal daily peak. This treatment induced a significant increase in the total number of leukocytes and altered their transcriptional programming. A priming response of leukocytes at the end of the 24-h infusion period associated with enhanced expression of a number of pattern recognition and cytokine receptors, receptor regulatory and signal transduction elements, as well as tuned-down protein translation and mitochondrial function was observed. Promoter analysis within cortisol-responsive genes indicated that only less than half of this response might have been as a direct consequence of GR–DNA binding; therefore, transcriptional regulation mechanisms other than DNA binding, such as tethering, plays a significant role. Furthermore, absence of any correlations between the direction of the response and different modes of transcriptional regulation underscored the complexity of mechanisms that govern the transcriptional programming through GR. Despite these significant changes prior to LPS administration, response to LPS followed very similar patterns in both cortisol and saline pre-treated groups except one significantly different cluster including probe sets associated with major players known to regulate the inflammatory response such as cytokines and chemokines, anti-inflammatory signal transduction elements and receptors mediating recruitment of leukocytes to the sites of inflammation. The much more intense response observed in the cortisol group in this cluster indicated that although continuous cortisol infusion did not directly affect the transcription of major inflammatory players, it indirectly affected their regulation, possibly through modulating their up-stream signaling cascades. Overall, coupled dose and rhythm manipulation of plasma cortisol levels promoted the priming of circulating leukocytes associated with sensitization of their surveillance function and tuning down of energy-intensive processes which may be linked to increasing their efficiency in responding to subsequent infectious threats.
Footnotes
Funding
The authors acknowledge support from NIH GM082974 and NIH GM34695.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
