Abstract
In patients with active anti-neutrophil cytoplasmic Ab (ANCA)-associated vasculitis (AAV), there are high levels of circulating C-reactive protein (CRP), which can inhibit the alternative complement pathway by binding factor H and triggering the classical complement pathway by binding C1q. However, the alternative, not the classical, complement pathway has been proven to play an important role in AAV. We found that both purified myeloperoxidase (MPO) and MPO released from ANCA-stimulated neutrophils could bind modified CRP (mCRP), but not pentameric CRP. Furthermore, MPO could block the binding between mCRP and factor H, as well as the binding between mCRP and C1q. Binding with mCRP did not influence the enzymatic activity of MPO. Binding with mCRP also did not influence the binding between MPO and its physical inhibitor, ceruloplasmin, as well as the binding between MPO and MPO–ANCA. The results indicated that MPO might be a complement regulator and inhibit the negative regulatory effect of CRP on the alternative complement pathway.
Introduction
In the pathogenesis of anti-neutrophil cytoplasmic Ab (ANCA)-associated vasculitis (AAV), neutrophils and the complement system are two important participants besides ANCAs.1–6 ANCA-stimulated neutrophils can produce reactive oxygen radicals (ROS) and release granules containing ANCA auto-Ags, including myeloperoxidase (MPO) and proteinase 3 (PR3).7–13 The pathogenic role of MPO–ANCA has been demonstrated in the development of AAV. 10 Following immunization of MPO knockout (Mpo-/-) mice with mouse MPO, the direct pathogenic effect of anti-MPO IgG has been convincingly demonstrated by adoptive transfer of anti-MPO-positive splenocytes into Rag2-/- mice and passive transfer of anti-MPO IgG alone into wild type mice. 10 However, the existence of MPO is crucial in the pathogenesis of AAV. With hydrogen peroxide (H2O2) as the substrate, MPO can catalyze the oxidation of chlorine ion (Cl-) into hypochlorite (ClO-), which is harmful to vascular endothelial cells in AAV. It has been proven that neutrophils can regulate complement activation. ROS of activated neutrophils could activate the complement system in AAV, 6 but whether the released granules of neutrophils, especially MPO, also have complement-regulatory effects in AAV is controversial. 6
High levels of circulating C-reactive protein (CRP) are characteristic of patients with active AAV.14,15 Serum CRP is a highly soluble pentamer [pentameric CRP (pCRP)], which after binding to membranes, including liposomes and cell membranes, or in a denaturating or oxidative environment, dissociates into free subunits yielding modified/monomeric CRP (mCRP).16–21 mCRP has been proven to be more effective than pCRP in inhibiting the alternative complement pathway by binding factor H and triggering classical complement activation by binding C1q.22–25 However, the alternative, not the classical, complement pathway has been proven to play an important role in the development of AAV.1–5
Coexistence of the activated alternative complement pathway and the high levels of circulating CRP suggested that there might be some factors inhibiting the function of CRP in AAV. We speculated that contents from the granules released from ANCA-stimulated neutrophils also have complement-regulatory effects. In the current study, we found that PR3 could bind neither pCRP nor mCRP, while MPO could bind mCRP. We also demonstrated that such binding did not influence the enzyme activity of MPO, the binding between MPO and MPO–ANCA, or the binding between MPO and ceruloplasmin (the physical inhibitor of MPO), but could block the binding between mCRP and factor H and the binding between mCRP and C1q.
Materials and methods
Patients
Patient plasma was obtained from 10 AAV patients with positive MPO–ANCA and five AAV patients with positive PR3–ANCA. All 15 patients, diagnosed in Peking University First Hospital, fulfilled the 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. 26 Plasma from five healthy blood donors was obtained as normal control. The research complied with the declaration of Helsinki and was approved by the Ethics Committee of Peking University First Hospital. All participants provided written informed consent.
Preparation of materials
High-purity (>99%) human pCRP (C-4063) was purchased from Sigma (St. Louis, MO, USA). Recombinant cysteine (Cys)-mutated mCRP (rmCRP) was generously gifted by Dr. Wu Y, Institute of Biophysics, Lanzhou University, Lanzhou, China. 19 We also used three methods to generate mCRP by heating pCRP at 70°C for 30 min, by treating pCRP with 8 M urea in the presence of 10 mM EDTA for 1 h at 37°C 23 and by immobilizing pCRP on plates directly. 23 Once prepared, all the mCRP preparations were used immediately. Purified MPO was prepared as described previously. 27 PR3 was purchased from Merck Calbiochem (Darmstadt, Germany). IgG fractions from patients with positive ANCA and healthy blood donors were purified by protein G affinity column (GE Healthcare Lifesciences, Uppsala, Sweden). Ceruloplasmin and factor H were purchased form Merck Calbiochem. C1q was purchased from Dako (Carpinteria, CA, USA).
Detection Abs were obtained as follows: mouse anti-PR3 and anti-MPO Abs (ab91181 and ab25985, respectively; Abcam, Cambridge, MA, USA); rabbit anti-C1q (A013602; Dako); goat anti-factor H (341276; Merck Calbiochem); and mouse monoclonal anti-human mCRP (C1688, Clone anti-CRP 8), rabbit polyclonal anti-human CRP (HPA027396), goat anti-ceruloplasmin (C0911), alkaline phosphatase-conjugated goat anti-rabbit immunoglobulin (A3687), alkaline phosphatase-conjugated goat anti-mouse immunoglobulin (A3562), alkaline phosphatase-conjugated mouse anti-human IgG (A2064) and alkaline phosphatase-conjugated rabbit anti-goat immunoglobulin (A4187) were all from Sigma.
Activation of neutrophils by ANCA
Neutrophils from healthy human donors were isolated from heparinized whole blood as described previously. 28 Then, neutrophils (2.5 × 106/ml) were primed for 15 min at 37°C with 2 ng/ml TNF-α, and subsequently activated with 0.1 g/l ANCA-containing IgG or normal IgG for an additional 30 min. Cells were centrifuged and discarded. Supernatants were collected. The concentration of MPO in the supernatant was tested using a commercial ELISA kit (E90601Hu; USCNK, Wuhan, China) as described previously. 2
SDS-PAGE and Western blot
SDS-PAGE was performed as described by Taylor et al. 29 As pCRP was sensitive to SDS-induced monomerization, SDS levels in all solutions were reduced to 1/20 of normal levels (SDS concentration of electrophoretic buffer, 2 × sample buffer, 4 × separation gel buffer, 4 × stacking gel buffer were 0.015%, 0.3%, 0.02% and 0.02%, respectively). 29 pCRP, heated pCRP, urea/EDTA denatured pCRP and rmCRP were loaded (10 µg per lane). Electrophoresis was performed for 40–60 min. After PAGE, the proteins were transferred onto nitrocellulose (Schleicher & Schuell, Dassel, Germany) and blocked overnight at 4°C in TBSTB buffer [0.01 mol/l Tris-HCl, pH 7.2, 0.15 mol/l NaCl, 0.1% (v/v) Tween 20, 1.0% BSA]. To detect the migration of different forms of CRP, the membrane was cut into strips and incubated with Clone anti-CRP 8 (1:5000 in TBSTB solution) or rabbit anti-human CRP polyclonal Ab (1:1000 in TBSTB solution) for 1 h at 37°C. After washing three times with TBST (15 min each), the strips were incubated with alkaline phosphatase-conjugated goat anti-mouse Ab (1:5000 in TBSTB solution) or alkaline phosphatase-conjugated goat anti-rabbit Ab (1:5000 in TBSTB solution) for 1 h at 37°C, respectively. To detect the binding between MPO and CRP, and the binding between PR3 and CRP, the strips were incubated with MPO (2 µg/ml, in TBSTB solution) or PR3 (2 µg/ml, in TBSTB solution) for 1 h at 37°C. Then, strips were washed three times with TBST (15 min each). MPO was detected with mouse anti-human MPO mAb (1:5000 in TBSTB solution) for 1 h at 37°C. PR3 was detected with mouse anti-human PR3 mAb (1:5000 in TBSTB solution) for 1 h at 37°C. After three washes, the strips were incubated with alkaline phosphatase-conjugated goat anti-mouse Ab (1:5000 in TBSTB solution). Nitroblue tetrazolium (Sigma) and 5-bromo-4-chloro-3-indolyl phosphate (Sigma) wer used as the substrates for alkaline phosphatase for all Western blot assays.
Native PAGE
The assay was as described for SDS-PAGE, except that SDS was omitted and both the pH of separating gel and the pH of stacking gel were 8.8. pCRP and rmCRP were loaded on the gel separately. After electrophoresis, the gel was stained with rapid gel stain (Applygen Technologies, Beijing, China) and the binding between MPO and CRP was analyzed by Western blot. The result was revealed on autoradiographic film using ECL Plus Western Blotting Detection System (GE Healthcare, Piscataway, NJ, USA).
ELISA
Conditions for the different ELISA assays.
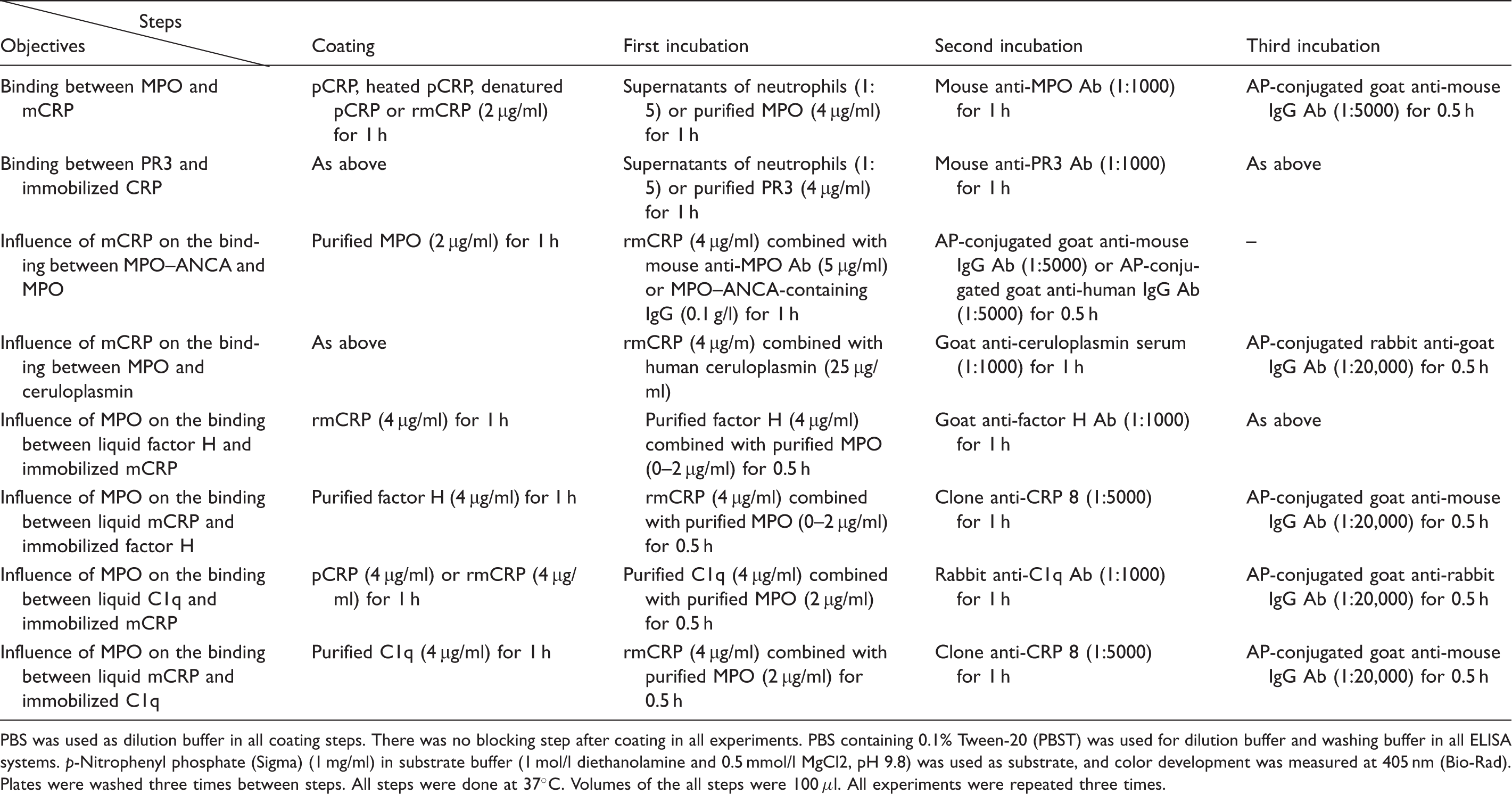
PBS was used as dilution buffer in all coating steps. There was no blocking step after coating in all experiments. PBS containing 0.1% Tween-20 (PBST) was used for dilution buffer and washing buffer in all ELISA systems. p-Nitrophenyl phosphate (Sigma) (1 mg/ml) in substrate buffer (1 mol/l diethanolamine and 0.5 mmol/l MgCl2, pH 9.8) was used as substrate, and color development was measured at 405 nm (Bio-Rad). Plates were washed three times between steps. All steps were done at 37°C. Volumes of the all steps were 100 μl. All experiments were repeated three times.
Influence of mCRP on the enzyme activity of MPO
The assay for MPO enzyme activity was adapted from the procedure described previously, 30 with some minor modifications. Purified MPO (2 µg/ml in PBS) was coated onto 96-well Costar microtiter plates (Data Packaging, Danvers, MA, USA) overnight at 4°C. After three washes with PBS, 100 µl of rmCRP (1–4 µg/ml) in PBS was added, and the plates were incubated at 37°C for 30 min. After three washes with PBS, 0.4 mg/ml o-phenylenediamine and 0.88 mM H2O2 in 0.05 M citrate buffer (pH 5.0) were added. Color development was measured at 450 nm (Bio-Rad, Tokyo, Japan). Ceruloplasmin (125 µg/ml) was added in as the positive control.
Statistical analysis
Data were shown to be normally distributed using the Kolmogorov–Smirnov test and variables were expressed as mean ± SD,and were evaluated using independent t-test or one-way ANOVA analysis as appropriate. A P-Value < 0.05 was considered significant. Analysis was performed with SPSS statistical software package (v. 15; SPSS, Chicago, IL, USA).
Results
MPO could bind mCRP whereas PR3 could bind neither pCRP nor mCRP
As shown in Figure 1A, in PAGE with 1/20 of standard SDS, pCRP ran as a single band with an apparent molecular mass > 250 ku, which was bigger than its expected molecular mass in PAGE with standard SDS (150 ku), while mCRP (heated pCRP, urea/EDTA denatured pCRP and rmCRP) ran at a molecular mass of 15 ku, which was close to its expected molecular mass in PAGE with standard SDS (23 ku). In Western blot analysis of PAGE with 1/20 of standard SDS (Figure 1B), pCRP ran as a smear, suggesting partial denaturation caused by SDS. Anti-CRP clone 8 recognized mCRP but did not recognize pCRP, while rabbit polyclonal anti-CRP Ab could recognize both pCRP and mCRP. MPO could not bind pCRP, but could bind mCRP. PR3 could bind neither pCRP nor mCRP.
Analysis of the binding between MPO and mCRP using PAGE. (A) Analysis of pCRP and mCRP with SDS-PAGE (1/20 of standard SDS, Coomassie brilliant blue staining). M: marker. (B) Binding between mCRP and various substances (Western blot analysis of PAGE with 1/20 of standard SDS). Polyclonal rabbit anti-CRP recognized both pCRP (lane 1) and mCRP (urea/EDTA denatured mCRP, lane 2). Mouse monoclonal anti-CRP (clone 8) did not recognize pCRP (lane 3), but recognized mCRP (lane 4). MPO did not bind pCRP (lane 5), but did bind mCRP (lane 6). PR3 recognized neither pCRP (lane 7) nor mCRP (lane 8). (C) Analysis of the binding between MPO and rmCRP with native PAGE. Gel staining indicated that both pCRP and rmCRP ran as a single band. In Western blot analysis, MPO bound to rmCRP, but not to pCRP.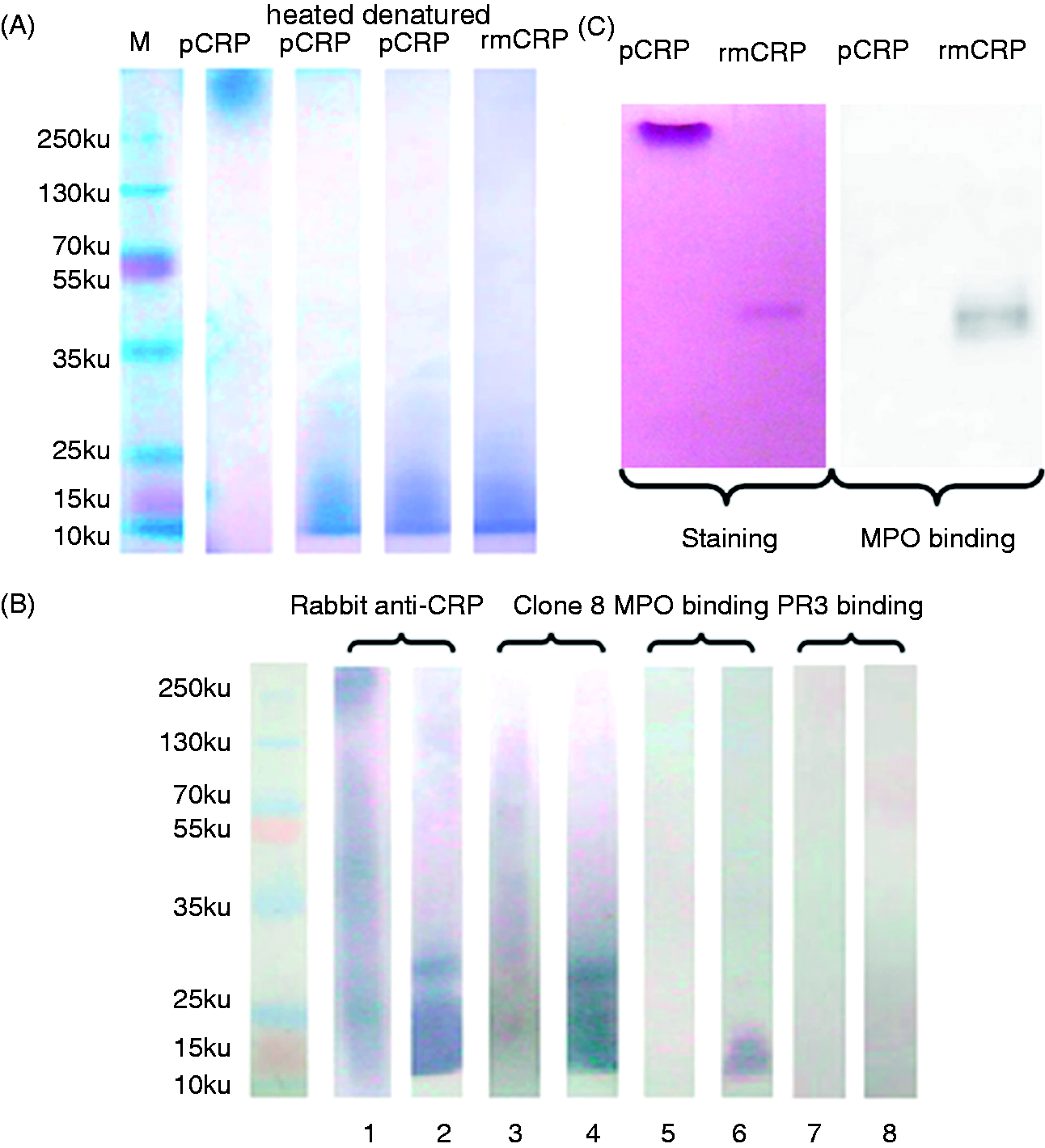
As shown in Figure 1C, in native PAGE, both pCRP and mCRP ran as single bands, and Western blot analysis showed that MPO could bind rmCRP, but not pCRP, indicating no denaturation of pCRP occurred in native PAGE.
In the ELISA, MPO showed strong binding to rmCRP and mCRP derived by the 3 different methods (immobilized pCRP, heated pCRP and urea/EDTA denatured pCRP) (Figure 2A). In contrast, PR3 showed no binding to either rmCRP or mCRP. Neither MPO nor PR3 bound BSA (Figure 2A).
Analysis of the binding between MPO and mCRP in ELISA. (A) MPO, but not PR3, could bind to immobilized mCRP in ELISA. Wells were coated with different forms of CRP, and binding of PR3 and MPO assessed. BSA was used as negative control. (B) Binding between the MPO released from neutrophils and mCRP. Plates were coated with recombinant mCRP and the binding of MPO contained within supernatants isolated from neutrophils incubated with PBS (1), IgG purified from five healthy donors (2), IgG purified from 10 patients with positive MPO–ANCA (3) and IgG purified from five patients with positive PR3–ANCA (4) determined. MPO levels in the supernatants of neutrophils were shown in Table 2.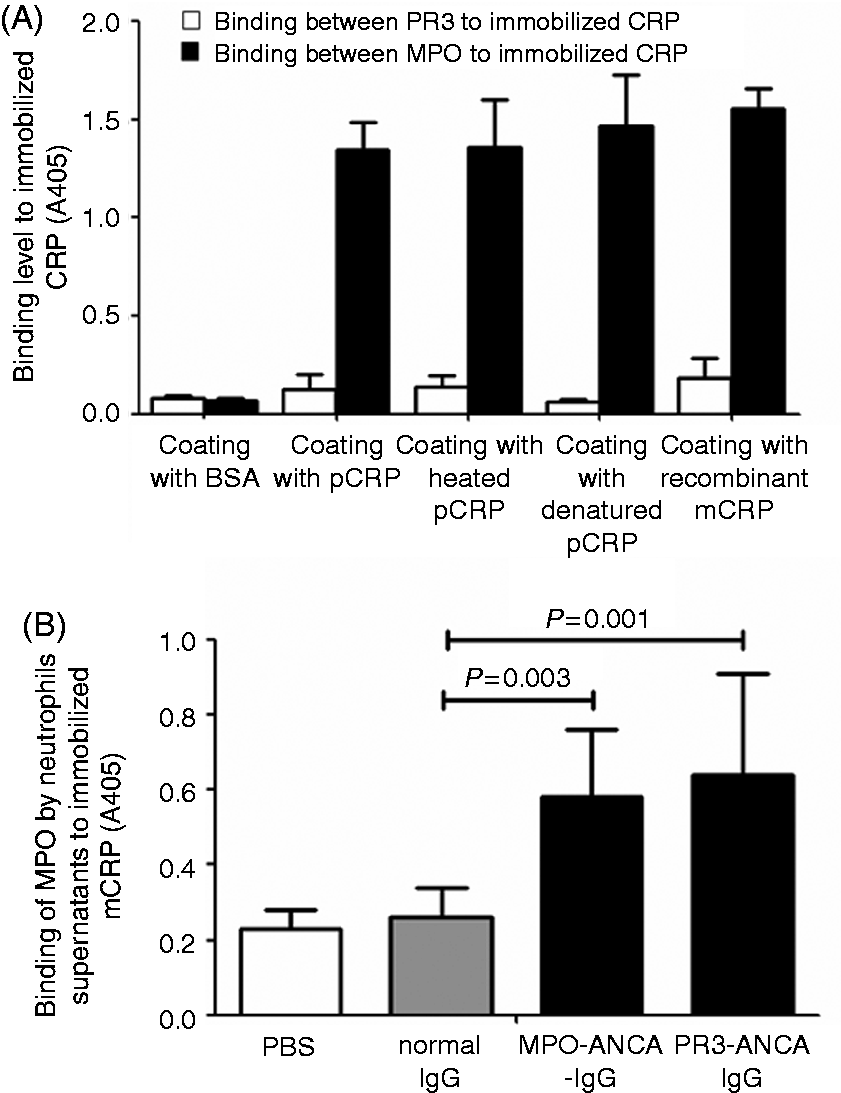
MPO released from ANCA-activated neutrophils could bind mCRP
MPO released from ANCA-activated neutrophils.
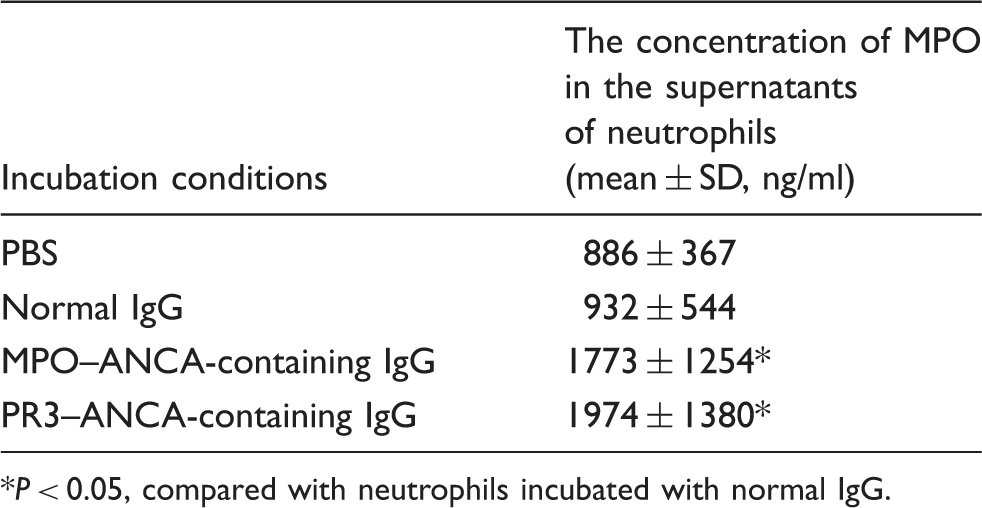
P < 0.05, compared with neutrophils incubated with normal IgG.
mCRP did not influence the enzyme activity of MPO, the binding between MPO and ceruloplasmin, or the binding between MPO and MPO–ANCA
As shown in Figure 3A, ceruloplasmin inhibited the enzyme activity of MPO, while mCRP did not. In addition, MPO–ANCA inhibited the binding between ceruloplasmin and immobilized MPO, while mCRP had no effect (Figure 3B). As excepted, mCRP did not influence the binding between MPO and MPO–ANCA (Figure 3C).
mCRP did not influence the enzyme activity of MPO and the binding between MPO and MPO–ANCA. (A) Recombinant mCRP did not influence the enzyme activity of MPO. Recombinant mCRP (1–4 µg/ml) or ceruloplasmin (125 µg/ml) was added to MPO and enzyme activity (A450) determined at 10 min. (B) mCRP did not influence the binding between ceruloplasmin and immobilized MPO. Recombinant mCRP (4 µg/ml) was added in the reactive system. The final concentration of both BSA and rmCRP was 4 µg/ml; MPO–ANCA-containing IgG purified from 10 patients were added as positive control (0.1 g/l). The concentration of ceruloplasmin was 25 µg/ml. (C) mCRP did not influence the binding between anti-MPO Abs and immobilized MPO. Moab: mouse anti-human MPO mAb; 1–10: IgG purified from 10 patients with positive MPO–ANCA. The concentration of MPO coated on wells was 2 µg/ml. Recombinant mCRP (1–4 µg/ml) was added in reactive system. Mouse anti-human MPO mAb was diluted 5 µg/ml, whereas the concentration of MPO–ANCA-containing IgG was 0.1 g/l.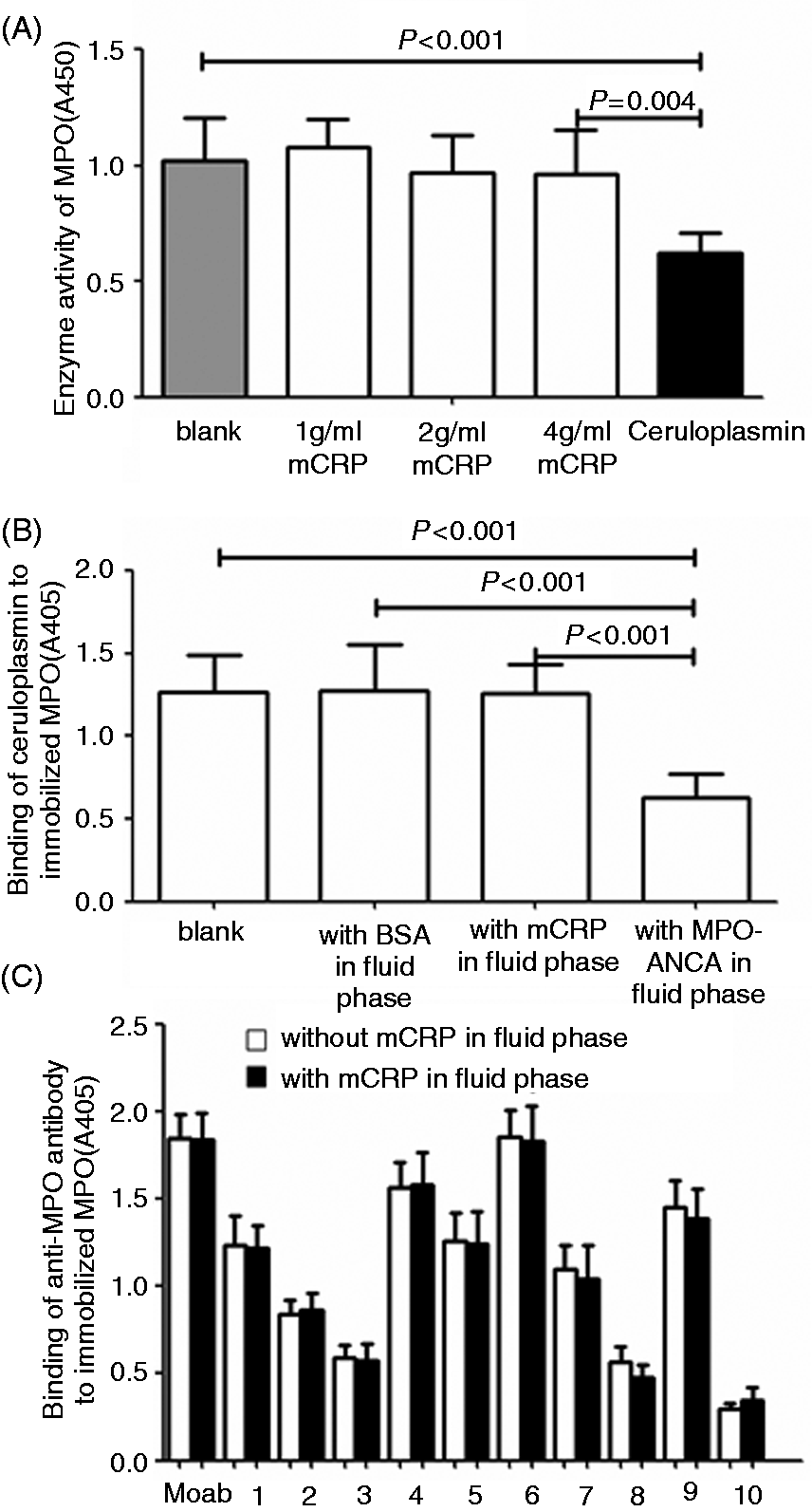
MPO could inhibit the binding between mCRP and factor H
MPO inhibited the binding between mCRP and factor H with 2 µg/ml by about 90% (Figure 4A). Alternatively, we analyzed the influence of MPO on the binding between liquid phase rmCRP to immobilized factor H with similar results. As shown in Figure 4B, the binding levels between liquid phase rmCRP and immobilized factor H decreased as the concentration of MPO increased.
Influence of MPO on the binding between mCRP and factor H. (A) Interaction between fluid phase factor H and immobilized rmCRP. (B) Interaction between fluid phase rmCRP and immobilized factor H.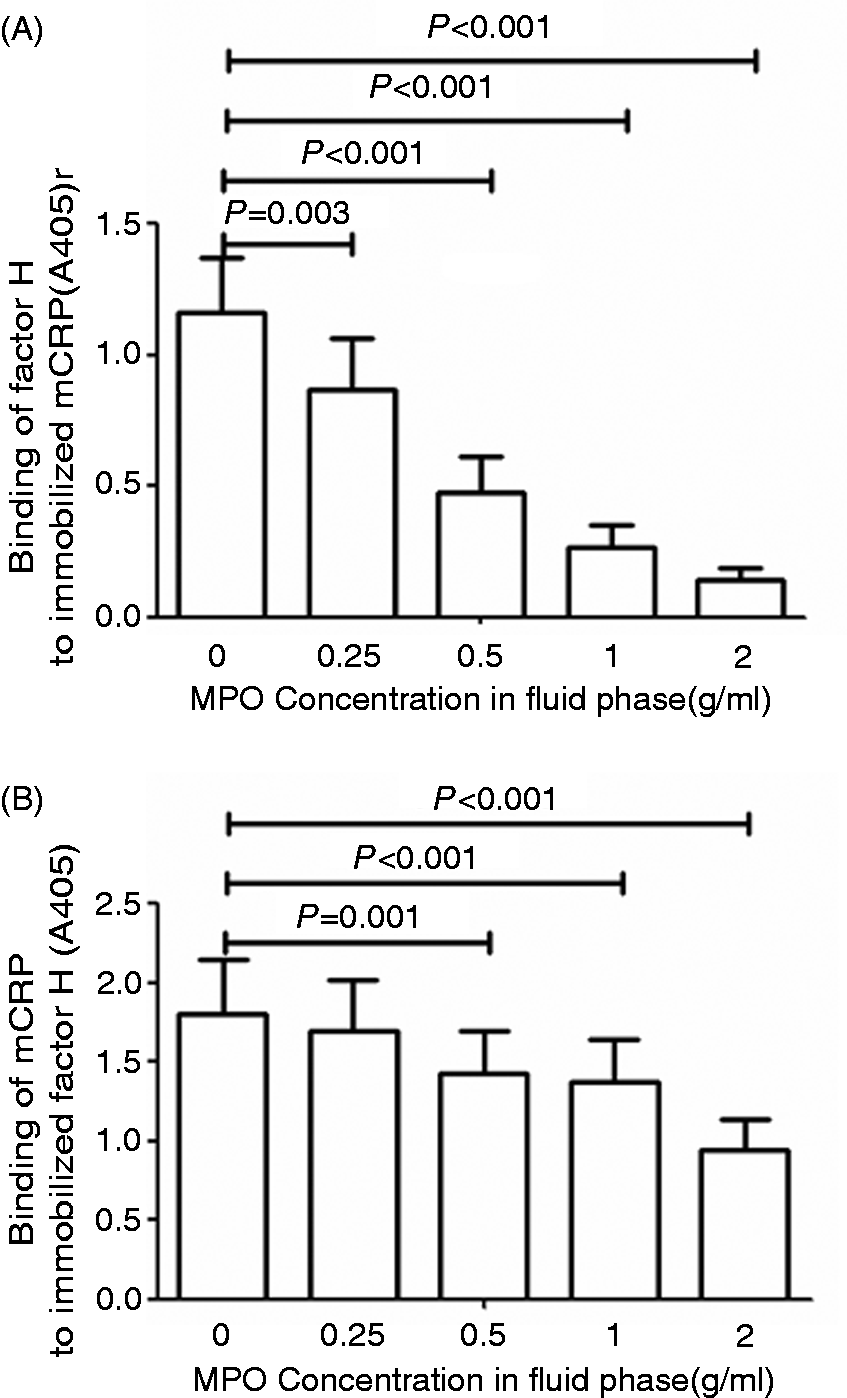
MPO could inhibit the binding between mCRP and C1q
We analyzed the influence of MPO on the binding between fluid phase C1q and immobilized mCRP and between fluid phase mCRP and immobilized C1q. The results indicated that mCRP, either immobilized on plastic or in the fluid phase, bound C1q avidly. MPO could inhibit the binding but the same concentration of BSA could not (Figure 5A, B).
Influence of MPO on the binding between mCRP and C1q. (A) Influence of MPO on the binding between C1q and immobilized mCRP. pCRP (4 µg/ml) or rmCRP (4 µg/ml) was coated on plates and the binding of C1q (4 µg/ml) assessed in the presence and absence of MPO or BSA (2 µg/ml). (B) Influence of MPO on the binding between mCRP and immobilized C1q. C1q (4 µg/ml) was coated on plates and the binding of rmCRP (4 µg/ml) assessed in the presence and absence of MPO or BSA (2 µg/ml).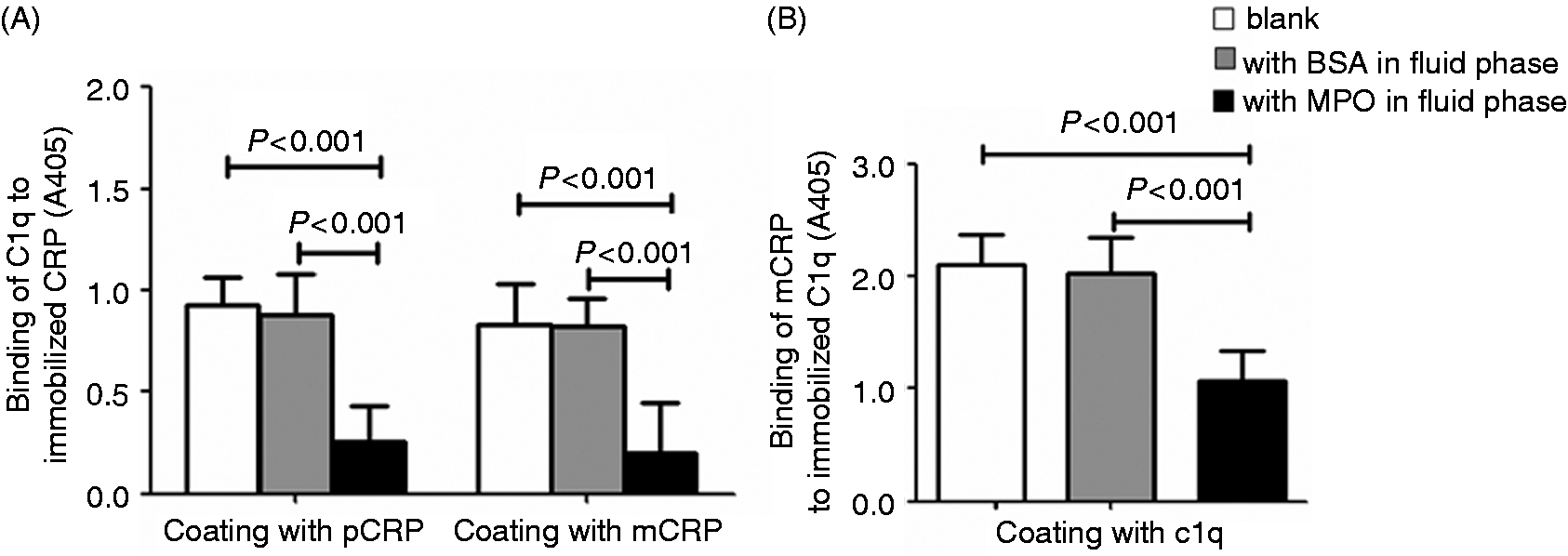
Discussion
ANCA can induce the release of neutrophil microparticles from primed neutrophils, which express a variety of ANCA auto-Ags.7–13 Recent studies have proved the existence of neutrophil extracellular traps that contain MPO and PR3, and can be released from ANCA-stimulated neutrophils and deposit locally in AAV. 31 We have demonstrated that PR3 could not bind mCRP, whereas MPO could bind mCRP. Furthermore, MPO could inhibit the binding between mCRP and factor H, which suggests that the alternative complement pathway would be amplified after MPO–mCRP binding. We also found MPO could inhibit the binding between mCRP and C1q, which might reasonably explain why activation of classical complement pathway is not found in AAV. 3 Besides, the mCRP-dependent clearance of apoptotic and necrotic cells might also be inhibited.
Both MPO–ANCA and PR3–ANCA could induce the release of MPO from neutrophils. However, ANCA stimulation is not the only origin of MPO release in AAV as it can also be released from neutrophils stimulated by high level of pCRP. Singh et al. 32 treated human neutrophils and monocytes with 25 and 50 mg/l pCRP for 6 h and found pCRP could induce the release of MPO from human neutrophils and monocytes. This phenomenon has also been proved by our previous study. 33 However, specific receptors for CRP on neutrophils have been documented by in vitro studies. 34
To the best of our knowledge, this is the first study demonstrating the direct binding between MPO and mCRP. Previous studies indicated that mCRP had lectin-like properties and could bind Gal-containing residues;35–37 however, mCRP did not bind PR3, which also had Gal-containing residues. Therefore, the exact binding site(s) between mCRP and MPO needs further identification. Although the binding site on mCRP for factor H was different from that for C1q, MPO could prevent both factor H and C1q binding to mCRP. We thought steric hindrance might be a reasonable explanation because MPO is a large molecule (140 ku) compared with mCRP (23 ku). Therefore, after binding by MPO, it is possible that binding sites for factor H and C1q on mCRP would no longer be accessible. We found MPO–mCRP binding did not influence the enzyme activity of MPO, the binding between MPO and MPO–ANCA, and the binding between MPO and ceruloplasmin. This indicated that the binding between mCRP and MPO did not influence the function of MPO.
Although the majority of circulating CRP is pCRP not mCRP, recent studies have suggested that the pathogenic role of CRP in diseases might be mainly attributed to mCRP not pCRP. 34 The existence of mCRP in AAV has been proven by our previous study, which found that CRP was observed in the cytoplasm of tubules and interstitium of renal biopsies from nine out of the 10 patients with AAV. 38 In the current study, we demonstrated anti-CRP clone 8 mainly recognized mCRP not pCRP, which was consistent with the results of some recent reports.39,40 So the CRP detected in situ with anti-CRP clone 8 in many previous studies is probably mCRP, and it is reasonable to speculate that the CRP detected in renal biopsies of AAV is also mCRP.
Our study indicates that MPO may have a complement-regulatory function. It is notable that the coexistence of neutrophil activation and an increased level of serum CRP is not unique in AAV. It can also be seen in many other inflammatory diseases, such as bacterial infection, burn wounds or even myocardial ischemic damage. Interestingly, unlike AAV, persistent activation of classical complement can be detected in these diseases.41–43 This apparent inconsistency may actually be an indication of the diversity of complement functions during the development and progression of various diseases. In the current study, we found that only mCRP could bind MPO, while intact pCRP could not. This is very crucial for the potential importance of MPO in modulating complement cascades in AAV and some other diseases. Without this condition, MPO might be bound by high levels of circulating pCRP, which will make it unavailable for binding to mCRP and having any effect on complement cascades. Although the data presented from PAGE/Western blot analysis suggests that MPO cannot bind pCRP, it needs further confirmation in other test systems.
Taken together, MPO, which can be released from ANCA-stimulated neutrophils, is a natural ligand for mCRP. MPO–mCRP binding can inhibit the binding between mCRP and factor H, which might inhibit the negative regulatory effect on alternative complement pathway of mCRP in AAV.
Footnotes
Funding
This study is supported by a grant of Chinese 973 project (No. 2012CB517702), the Research Fund for the Doctoral Program of Higher Education of China (No. 20120001110018) and two grants of the National Natural Science Fund (No. 81370829 and No. 81021004).
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
