Abstract
Objective:
Type 1 diabetes mellitus (T1DM) is a pro-inflammatory state characterized by high C-reactive protein (CRP) levels. Previously, we showed that CRP accentuated a macrophage (MO) activity in spontaneously diabetic biobreeding (BB) rats and increased the MO activity of protein kinase C-alpha (PKC-α) and p47phox. In this report, we tested the effects of molecular inhibition of CRP effects on MO activity using antisense oligonucleotide (ASO) to both PKC-α and p47phox.
Methods:
Prior to administration of human C-reactive protein (hCRP) daily for 3 days, ASO or scrambled ASO to either PKC-α or p47phox was also delivered for 3 days and after killing on day 4, peritoneal MOs were isolated.
Results:
The increase in the levels of superoxide anion, interleukin (IL)-1, monocyte chemoattractant protein-1 (MCP-1), tumour necrosis factor-alpha (TNF-α) and IL-6 release in MOs with hCRP compared to human albumin was significantly attenuated by antisense to either PKC-α and p47phox (p < 0.01 vs. scrambled ASO; n = 5 per group).
Conclusion:
Our novel data suggest that antisense to either PKC-α or p47phox attenuates the pro-inflammatory effects of human CRP on MOs in diabetic rats.
Type 1 diabetes mellitus (T1DM) confers an increased propensity to both microvascular and macrovascular complications. 1 Inflammation plays a crucial role in the pathogenesis of both microvascular and macrovascular complications of diabetic patients.2,3 C-reactive protein (CRP) is the prototypic marker of inflammation and has been shown to predict cardiovascular events and cardiovascular mortality in diabetic patients.4,5 Also, CRP has been shown to induce pro-inflammatory effects in endothelial cells and macrophages (MOs) both in vitro and in vivo.6,7 CRP levels are increased in patients with T1DM8–10 along with other biomarkers of inflammation. Previous studies have shown a significant association between increased intima–media thickness (IMT) and decreased flow-mediated vasodilatation (FMD) with CRP in patients with T1DM, and recently, CRP levels predicted progression of nephropathy in patients with T1DM.11–14
The biobreeding diabetes-prone (BB-DP) rats spontaneously develop T1DM through a T-cell-mediated autoimmune destruction of pancreatic β-cells. 15 The rat appears to be the preferred model to test the effects of human C-reactive protein (hCRP). 16 Also, we showed that hCRP increases oxidized low-density lipoprotein (LDL) uptake, reactive oxygen species (ROS) generation, and tissue factor (TF) release and induces matrix metallopeptidase 9 (MMP-9) activity in MOs and impairs vasoreactivity and the endothelial glycocalyx of non-diabetic Wistar rats. 6 Recently, we showed that hCRP accentuates MO activity in biobreeding (BB) rats and induces both protein kinase C-alpha (PKC-α) and membrane p47phox. 17 Hence, in this report, we tested the effect of in vivo molecular inhibition of both these pathways using antisense therapy in the BB rat model.
Materials and methods
BB-DP rats (males; weighing 125–150 g) were obtained from BRM Inc. (Boston, MA) and were housed in a controlled sterile environment (pathogen-free conditions) at an ambient temperature of 21°C ± 2°C on a 12-h light: 12-h dark cycle. 17 Food and water were provided ad libitum. The animals were weighed periodically, and diabetes condition was confirmed by measuring tail vein blood glucose levels using a commercial glucometer (Accu-Chek, Roche, USA). The rats were considered diabetic when blood glucose levels exceeded 300 mg/dL (non-fasting) on three readings and their weights were maintained in hyperglycaemic condition (300–450 mg/dL) by subcutaneous insulin pellets (Linplant, Linshin, Canada) (1–2 U/day) to prevent the weight loss.
Two weeks after persistent hyperglycaemia, hCRP or human serum albumin (huSA) control was administered by intraperitoneal (IP) injection (20 mg/kg body weight for 3 days, n = 5 rats in each group), as previously described.16,17 The rats were killed on day 4 by overdose of pentobarbital. Resident peritoneal MOs were isolated following lavage with 5.0 mL of warm phosphate-buffered saline (PBS) and showed F4/80 positivity (85%) when analysed by flow cytometry, as described previously,16,17 and were used for further analysis. Serum levels of hCRP were measured using enzyme-linked immunosorbent assay (ELISA) that only recognized hCRP and not rat CRP. 17 The protocol was approved by the Animal Committee of University of California at Davis, which regulates the principles of laboratory animal care.
CRP was purified from human pleural or ascites fluid using the method devised by Du Clos et al. 18 Lipopolysaccharide (LPS) was removed from CRP preparations with a Detoxi-Gel column (Pierce Biochemicals, Rockford, IL) and found to contain <0.125 EU/mL (<12.5 pg/mL) by the Limulus assay (BioWhittaker, NJ, USA), as described previously. 17 CRP was stored at 4°C in Tris-buffered saline with 2 mM calcium. This CRP preparation has been shown to exert pro-inflammatory effects in toll-like receptor 4 (TLR4) knock-down cells.6,7
We measured MO-released interleukin-1 beta (IL-1β), IL-6, KC/IL-8, monocyte chemoattractant protein-1 (MCP-1) and tumour necrosis factor-alpha (TNF-α) using a multiplex cytokine assay (Millipore, St. Charles, MO), as described previously. 17 Intra- and inter-assay coefficients of variation (CVs) were determined to be <11%. Superoxide anion release from peritoneal MOs was assessed by dihydroethidium (DHE) fluorescence, as described previously. 17
Since in previous studies, we have shown under hyperglycaemic conditions that both PKC-α and p47phox are critical in mediating pro-inflammatory effects,19,20 we focused on these signalling pathways using antisense oligonucleotide (ASO). The animals received ASO injections (50 mg/kg body weight) that targets PKC-α mRNA, nt: −13 to +7 regions (GenBank X07286.1) or rat p47phox (phagocyte oxidase; GenBank AY029167) subunit of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, nt: 5–26 regions. Scrambled sequences were used as negative controls. The sequence of PKC-α ASO is 5′ CAG CCA TGG TTC CCC CCA AC 3′, and the sequence of p47phox ASO is 5′ GGT GTC CCC CAT GGC TGG GCA G 3′. The oligonucleotides were synthesized with phosphorothioate-modified bases to limit degradation, to achieve improved specificity of binding to mRNA and nuclease resistance and high-performance liquid chromatography (HPLC) purified to limit contamination by incomplete synthesis products. ASOs were manufactured at IDT (San Jose, CA). The rats received ASO 4–6 h intraperitoneally prior to hCRP injection every day for 3 days (n = 5 rats per group). The rats were killed on day 4, and peritoneal MOs and serum were collected for measurement of biomediators and hCRP. Western blots for both PKC-α and p47phox were performed, as previously described. 17
All assays were performed in duplicate. The comparisons between group means were analysed using analysis of variance (ANOVA). The experimental results are presented as the mean ± SD (standard deviation). Paired t-tests were used to compute differences in the variables, and the level of significance was set at p < 0.05.
Results
The average body weight of the BB rat was 187 ± 41 g, and the average glucose level was 496.8 ± 110 mg/dL. Administration of hCRP compared with huSA in diabetic rats led to significantly higher circulating levels of hCRP (Table 1). Previously, we reported that CRP in BB rats induced MO activity resulting in increased circulating and MO release of numerous biomediators including superoxide, plasminogen activator inhibitor-1 (PAI-1), IL-1, IL-6, IL-8, TNF, MCP-1 and TF and increased activity of both PKC-α and membrane p47phox component of NADPH oxidase. 17 We conducted dose response and time course experiments with antisense oligodeoxynucleotides (ASO) to PKC-α and p47phox in vivo in hCRP-injected diabetic rats. A dose of 50 mg/kg body weight ASO was not toxic to rats (there were no significant changes in complete blood count (CBC), creatinine and liver function, Table 1). While ASO administration to PKC-α and p47phox significantly abolished PKC-α and p47phox expression, respectively, this was not affected with the scrambled ASO (Figure 1). As shown in Figure 2, compared to scrambled ASO, IP administration of PKC-α or p47phox ASO prior to hCRP administration significantly decreased CRP-induced superoxide anion release and decreased biomarkers of inflammation: IL-1β, MCP-1, TNF-α and IL-6 release in the MOs of rats (Figure 2) (p < 0.01, n = 5 per group).
Biochemical characteristics.
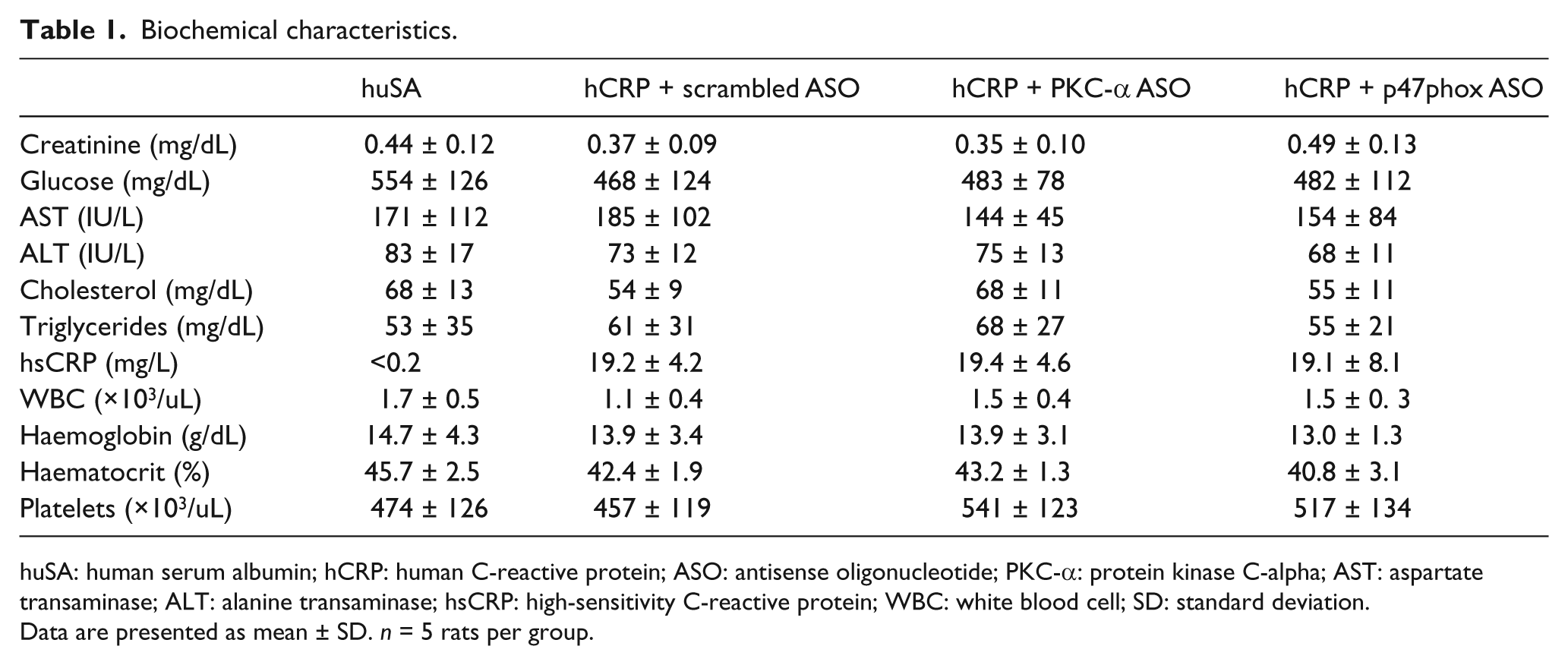
huSA: human serum albumin; hCRP: human C-reactive protein; ASO: antisense oligonucleotide; PKC-α: protein kinase C-alpha; AST: aspartate transaminase; ALT: alanine transaminase; hsCRP: high-sensitivity C-reactive protein; WBC: white blood cell; SD: standard deviation.
Data are presented as mean ± SD. n = 5 rats per group.
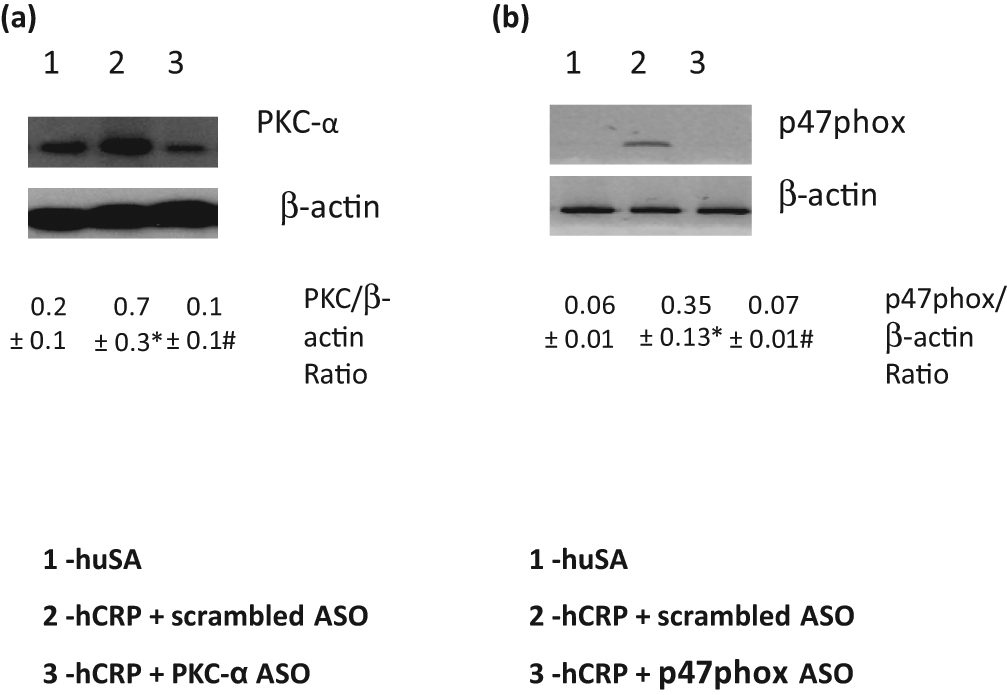
Effect of PKC-α and p47phox ASO on (a) PKC and (b) p47phox expression in macrophages of BB rats: BB rats were treated with hCRP or huSA or hCRP + ASO to PKC-α or p47phox as described in section ‘Materials and methods’ (n = 5 per group). Scrambled ASOs were used as control. Representative Western blot of PKC-α and membrane p47phox from isolated macrophages are shown. *p < 0.05 compared to huSA and #p < 0.05 compared to hCRP + scrambled ASO.
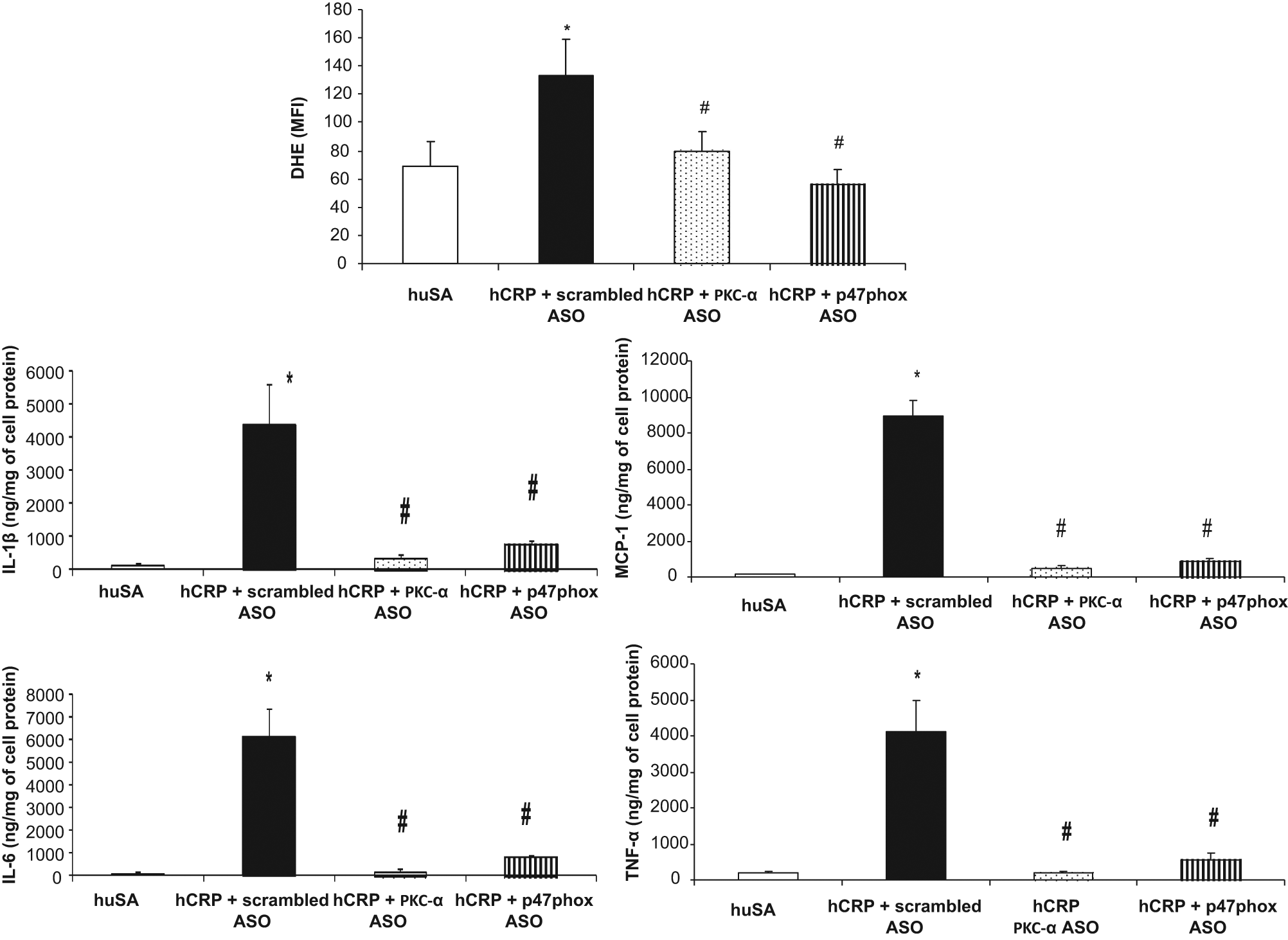
Effect of PKC-α and p47phox ASO inhibition on superoxide anion and IL-1β, MCP-1, TNF-α and IL-6 release from macrophages of BB rats treated with hCRP or huSA (20 mg/kg body weight for 3 days, n = 5 per group). Superoxide anion release was assessed by DHE fluorescence. Scrambled ASOs were used as control. The results are expressed as mean ± SD MFI for superoxide and nanogram per milligram of cell protein for cytokines and MCP-1. *p < 0.01 versus huSA. #p < 0.01 versus hCRP + scrambled ASO.
Discussion
Recently, we presented novel data showing that hCRP exacerbates the pro-inflammatory and pro-oxidant and procoagulant states of T1DM using the BB rat 17 confirming our findings in the non-diabetic Wistar rats.6,16
It is generally accepted that mice are not the optimum model to study the biological effects of hCRP. The rat model is preferred to mice to document the pro-inflammatory and vascular effects of hCRP.6,7,17 hCRP has been shown to increase myocardial infarct size in a coronary artery ligation model and stroke volume following middle cerebral artery occlusion.6,7 With respect to mechanistic insights, we have shown that hCRP delivery in non-diabetic rat models stimulated TF activity, NF-κB activity, ROS and MMP-9 activity, and oxidized low-density lipoprotein (Ox-LDL) uptake impairs vasoreactivity by uncoupling endothelial nitric oxide synthase (eNOS), perturbs the endothelial glycocalyx and promotes polarization of the MOs to the pro-inflammatory M1 phenotype.6,7,17 Also, hCRP in rats activates complement.6,7 In fact, in a rat model, a small molecular inhibitor to hCRP was shown to prevent the induction of an increase in myocardial infarct size with hCRP.6,7 Thus, several lines of evidence support rat models to study in vivo effects of hCRP.
Recent studies have implicated both PKC and NF-κB in the pro-inflammatory state of diabetes. We showed for the first time that CRP induces PKC-α expression in a diabetic rat model leading to downstream activation of NF-κB and consequent inflammation. 17 Furthermore, we now show that ASO to PKC-α attenuates the CRP-induced inflammation in diabetic rats providing key mechanistic details. In future studies, we will explore the significance of the expression of other PKC isoforms such as delta. However, it needs to be emphasized that in the present report, PKC-α appears to be pivotal with respect to the effects of CRP in diabetic rats since ASO to PKC-α reversed the pro-inflammatory phenotype. p47phox is a critical member of NADPH oxidase and translocates to the membrane to activate NADPH oxidase and produce superoxide. We and others have shown that CRP induces superoxide anion in the diabetic milieu. 17 In the present report, we show that ASO to this critical subunit attenuates the effects of CRP on MO cytokine and chemokine secretion, suggesting that ROS are upstream in the regulation of these biomediators and NF-κB. Thus, inhibitions of both PKC-α and p47phox reduce the pro-inflammatory effects of hCRP and could be relevant targets for anti-inflammatory strategies in reducing the inflammatory burden of diabetes in addition to targeting CRP.
Footnotes
Acknowledgements
We thank Dr R. Dasu and B. Syed for care of the animals and assisting with experimentation.
Funding
This study was supported by National Institutes of Health (grant no. ROI-HL074360).
Conflict of interest
The authors declare that they have no conflicts of interest.
Authors’ contribution
Ishwarlal Jialal: Designed experiments and helped with manuscript preparation and discussion;
Sridevi Devaraj: Performed rat experiments and manuscript preparation.
