Abstract
Background:
Oxidative stress and inflammation are associated with endothelial injury and coronary artery disease. Inflammatory factors that promote oxidative damage include endothelin-1 (ET-1), myeloperoxidase (MPO), and C-reactive protein (CRP). Current guidelines recommend the use of statins in patients with risk of atherosclerotic cardiovascular disease (ASCVD).
Aim:
To assess the impact of atorvastatin on plasma inflammatory and oxidant biomarkers in patients with moderate to very high risk of ASCVD.
Method:
Two hundred ten patients presented to the cardiology clinic were included and stratified into low, moderate, high, and very high risk of ASCVD. Moderate- (20 mg/d) to high-intensity (40 mg/d) atorvastatin was prescribed. Plasma levels of lipids, ET-1, CRP, MPO, total nitrite, lipid peroxides (thiobarbituric acid reactive substances [TBARS]), and superoxide dismutase (SOD) activities were measured at baseline and 12 weeks after treatment.
Result:
Relative to low-risk patients, baseline plasma inflammatory markers of CRP, MPO, ET-1, and nitrite were higher in patients with very high risk of ASCVD, whereas plasma SOD was lower (all
Conclusions:
Key markers of oxidative stress/inflammation such as CRP, ET-1, total nitrite, and MPO are associated with an increased risk of ASCVD. Moderate- and high-intensity atorvastatin use reduces plasma oxidative stress and inflammation regardless of ASCVD risk and independent of its lipid-lowering effect.
Keywords
Introduction
Coronary artery disease (CAD) is an important global cause of morbidity and mortality. 1 Dyslipidemia, characterized primarily by the elevation of serum concentrations of low-density lipoprotein (LDL) particle , is a major risk factor for the development of CAD.
An increasing evidence suggests that oxidative stress and inflammation are associated with the instability of atherosclerotic plaque and the development of acute coronary syndrome (ACS). 2 Reactive oxygen and nitrogen species (ROS/RNS) are unstable and highly reactive molecules that can rapidly interact and interfere with cell components such as lipids, proteins, and nucleic acids. 3
Reactive oxygen species induce inflammatory cascades and cause LDL oxidation, a key process in the formation of atherogenic plaques. 4 Several inflammatory factors promote oxidative damage of the vasculature and contribute to the development of CAD. Endothelin-1 (ET-1) is one of the most powerful vasoconstrictors and pro-inflammatory mediators that is synthesized by endothelial cells of the coronary arteries and macrophages. 5 We have shown previously that ET-1 is also produced by cardiac myocytes and fibroblasts 6 and is associated with myocardial infarction (MI) and poor clinical outcome in patients with CAD. 5
Myeloperoxidase (MPO) is a pivotal inflammatory modulator of coronary endothelial dysfunction, as well as plaque rupture. Increased plasma MPO (pMPO) in patients with CAD is associated with an increased neutrophil to lymphocyte ratio and an increased risk of MI. 7 C-reactive protein (CRP) is a sensitive but nonspecific inflammatory factor for which elevated levels are broadly predictive of coronary and other atherothrombotic events. 8,9
Low-density lipoprotein lowering is the primary goal of cholesterol-lowering therapy. 10 –12 Statins are the gold standard for lowering LDL 12,13 and have pleiotropic benefits such as improving endothelial function, stabilizing atherosclerotic plaques, and decreasing inflammation/oxidative stress. 14
The American College of Cardiology and the American Heart Association (ACC/AHA) guidelines have identified groups of patients who would benefit from statin therapy including those with established clinical ASCVD. 12 The National Lipid Association (NLA) expert panel classified patients into categories based on their risk of ASCVD: low, moderate, high, and very high risk groups. 10 However, it is unclear whether the ASCVD risk classification correlates with plasma levels of oxidative stress and inflammation. In addition, the effect of statin therapy on inflammatory biomarkers among various ASCVD risk categories, particularly those with low-to-moderate risk, is not fully characterized.
The primary aim of this study was to evaluate the impact of moderate- to high-intensity atorvastatin on key plasma levels of inflammatory and oxidative stress biomarkers (ET-1, MPO, CRP, nitrite, and thiobarbituric acid reactive substances [TBARS]) in patients with risk to develop ASCVD. We hypothesized that atorvastatin reduces plasma levels of inflammatory and oxidant biomarkers in patients with moderate to very high risk of ASCVD regardless of atorvastatin dose intensity and independent of lipid-lowering effect.
Methods
This is a prospective study of patients with different risk categories of ASCVD. All included patients were consecutive patients who presented with chest pain that requires clinical assessment. The study was approved by the institutional review board of Jordan University of Science and Technology and King Abdullah University Hospital (KAUH).
After an investigator explained the protocol and the potential hazards and benefits of the study, all patients provided written informed consent. A specific data sheet was extracted for each patient describing patient laboratory data and demographic and clinical characteristics, including the use of medications. Data were obtained from patient medical files available in the electronic data system of KAUH.
Inclusion criteria were newly diagnosed patients with dyslipidemia who would be eligible for statin therapy based on the ACC/AHA guidelines. 12 Eligible patients included those with established ASCVD, diabetes mellitus (DM; at ages of 40-75 years and LDL of 70-189 mg/dL), high LDL >190 mg/dL, and 10-year risk >7.5% (at the age of 40-75 years and LDL of 70-189 mg/dL). Other patients considered for statin therapy included those with low, moderate, and high risk of ASCVD with LDL levels ≥160, ≥130, ≥100 mg/dL, respectively, and those who might have other factors that require initiation of the therapy per the physician’s decision. No patients included in the study had received statin therapy before.
Exclusion criteria were patients with prior statin therapy, inflammatory disease or recent infection, recent surgery or trauma, and renal dysfunction (creatinine>1.5 mg/dL). Patients with contraindication to statins were also excluded, including those with hypersensitivity to statins, active liver disease, or unexplained persistent isolated elevations of serum transaminases, pregnancy, and lactation. Patients who received combined antilipidemic agents were also excluded.
Patient Groups and Procedure
Patients with varied risk of ASCVD were recruited. Major risk factors for atherosclerotic cardiovascular disease (ASCVD),
10
including age, family history of early coronary heart disease (first-degree relative at age <55 in men and <65 in women), current cigarette smoking, hypertension (HT; blood pressure ≥140/90 mm Hg), and low high-density lipoprotein (HDL<40 and <50 mg/dL for men and women, respectively), were evaluated. Patients were classified into 4 groups based on their primary risk of ASCVD according to the NLA criteria for ASCVD risk assessment.
10
Atorvastatin therapy was prescribed to eligible patients. At baseline, most patients received moderate-intensity atorvastatin (20 mg/d), while a subgroup of very-high-risk patients received high-intensity atorvastatin (40 mg/d) per the physician’s decision. Patients were followed up for 12 weeks for lipid and biomarker measurements. Patients were monitored after discharge by phone calls or by timely clinical visits to guarantee adherence to therapy.
Plasma Measurements of Lipid and Inflammatory Factors
Plasma levels of lipids, inflammatory, and oxidant markers were measured at baseline (before treatment) and at 12-week follow-up. Freshly blood samples were collected in plain and EDTA tubes and transferred on ice, then centrifuged at 2500 rpm for 15 minutes to separate plasma and serum. Plasma samples were stored at −80°C until analysis. Fresh serum samples were used to measure lipid levels at KAUH medical labs using standard techniques.
Plasma ET-1 levels were assessed using an ET-1 Quantikine enzyme-linked immunoassay (DET100 ELISA; R&D Systems, Inc, MA, USA), as previously described. 5 Plasma levels of CRP were measured using a human CRP ELISA (DuoSet DY1707; R&D systems, Inc). Plasma MPO levels were assessed using a human MPO instant ELISA (BMS2013INST; eBioscience, Vienna, Austria) as previously described. 7 Plasma TBARS (pTBARS) levels were evaluated using TBARS parameter assay according to the manufacturer’s instructions (TBARS KGE013; R&D systems, Inc). Enzymatic activity of superoxide dismutase (SOD) was measured using colorimetric assay according to kit instructions (19160 SOD kit; Sigma-Aldrich Corp, MO, USA). Plasma total nitrite levels were determined by nitric oxide (NO) parameter assay (KGE001; R&D systems, Inc)
Statistical Analysis
Data are expressed as mean ± standard error, unless otherwise specified. Normally distributed variables were analyzed using one-way analysis of variance; nonnormally distributed data were assessed using Kruskal-Wallis tests. Dunn and Tukey post hoc tests were used for multiple comparisons. Follow-up levels of plasma biomarkers were compared to baseline using Wilcoxon matched-pair signed rank test. χ2 tests were used to compare frequencies across study groups. Spearman correlation was used to evaluate correlation of variables that were not normally distributed. GraphPad Prism 6 was used for univariate analyses and preparation of figures. Multivariate analysis adjusting for possible confounders (including use of medications, LDL cholesterol, and heart failure [HF]) was performed to evaluate independent predictors of plasma biomarkers using JMP 11 software (SAS Institute). As age, gender, smoking history, DM, HT, and HDL levels were considered major risk factors to assess ASCVD risk category, they were omitted from the model to avoid collinearity with ASCVD risk status. As plasma biomarker levels were not normally distributed, a square root transformation was performed in the multivariate model. Values of
Results
Patients Characteristics
Plasma samples from 210 patients were selected for biochemical analysis of inflammatory/oxidant biomarkers and other lab measurements (eg, lipid profile, etc). Table 1 shows study groups stratified based on their primary risk of ASCVD. Study groups included (1) patients with lowest risk of ASCVD (low, n = 37), (2) patients with moderate risk of ASCVD (moderate, n = 15), (3) patients with high risk of ASCVD (high, n = 39), and patients with very high risk of ASCVD (very high, n = 119). Among patients with very high risk of ASCVD, 95 patients had coronary stenosis ≥50% (50 of whom had experienced an MI), 1 patient had previous history of stroke, and the remaining participants were diabetics with multiple major risk factors for ASCVD. Mean age ± standard error of the mean (SEM) of the patients was 51.03 ± 0.88. Most study patients had significant comorbidities; DM and HT were present in 36.45% and 42.36% of patients, respectively, whereas HF was present in 5.39% of patients. Ninety-four (46%) patients were smokers, and 91 (44.6%) had a family history of CAD (first- or second-degree relative with CAD). Patients with very high risk of ASCVD have significantly lower left ventricular ejection fraction (LVEF) relative to low-risk patients. Baseline LDL was 3.39 ± 0.077 mmol/L. Patients (n = 159) were found statin eligible and received atorvastatin. One hundred twenty-two patients received moderate-intensity statin (20 mg atorvastatin/day), while 37 patients with very high risk received high-intensity statin (40 mg atorvastatin/day). One hundred forty-eight patients (95 very high, 27 high, 12 moderate, and 14 low risk) were followed up after 12 weeks for biochemical evaluation of plasma inflammatory markers and lipid levels. Among those, 134 patients received statins. Low-to-moderate risk patients who were below the LDL level to consider drug therapy (<160 and <130 mg/dL, respectively) did not receive statin therapy and had no indication for follow-up. Fourteen of them accepted to be followed as untreated controls.
Baseline Patient Characteristics.a

Abbreviations: ACEi, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index (kg/m2); DM, diabetes mellitus; CAD, coronary artery disease; HDL, high-density lipoprotein; Hx, history; HT, hypertension; HF, heart failure as established by clinical symptoms and echocardiographic measurements; LDL, low-density low protein; LVEF, left ventricular ejection fraction; Non-HDL cholesterol, total cholesterol minus HDL cholesterol; T. Cholesterol, total cholesterol.
aData are presented as mean ± standard error of the mean (SEM) for continuous variables and percentage for categorical variables. Unit for serum lipids and glucose is mmol/L.
bPresence of significant differences between study groups compared to control using analysis of variance (ANOVA) or Kruskal-Wallis tests for continuous variables and χ2 test for categorical variables (
During follow-up, 10 patients with established ASCVD presented with recurrent angina requiring catheterization, and 4 of them were stented. Two of the stented patients received 40 mg atorvastatin, whereas 2 patients received 20 mg atorvastatin. None of the followed patients had MI attach, underwent coronary artery bypass surgery, or died.
Serum Lipids Levels
No significant differences in baseline serum lipids of LDL, triglycerides, total cholesterol, HDL, and non-HDL were found between ASCVD risk groups (
Effects of 12-Week Atorvastatin Therapy on Serum Lipids.a,b
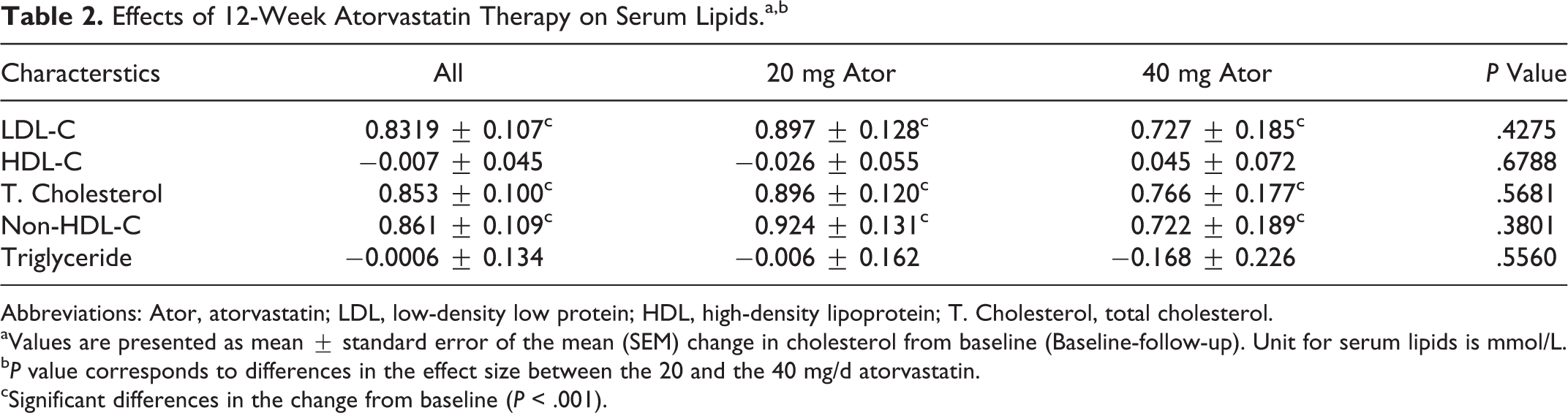
Abbreviations: Ator, atorvastatin; LDL, low-density low protein; HDL, high-density lipoprotein; T. Cholesterol, total cholesterol.
aValues are presented as mean ± standard error of the mean (SEM) change in cholesterol from baseline (Baseline-follow-up). Unit for serum lipids is mmol/L.
b
cSignificant differences in the change from baseline (
Plasma Levels of Inflammatory and Oxidant Biomarkers
Plasma levels of ET-1 (pET-1), MPO, CRP, TBARS, total nitrite, and SOD activity were evaluated as a function of ASCVD risk status (Figure 1A–F). Patients with very high risk of ASCVD had higher pET-1 than patients with high, moderate, or low risk (mean ± SEM = 1.19 ± 0.07, 0.71 ± 0.11, 0.69 ± 0.18, and 0.71 ± 0.14 pg/mL, respectively,

Association of plasma inflammatory/oxidant biomarkers with ASCVD risk status. Baseline plasma endothelin-1 (ET-1, A), myeloperoxidase (MPO, B), C-reactive protein (CRP, C) thiobarbituric acid reactive substances levels (TBARS, D), total nitrite (E), and superoxide dismutase (SOD, F) in patients with low risk of atherosclerosis cardiovascular disease (ASCVD), moderate risk, high risk, and very high risk (very high). **
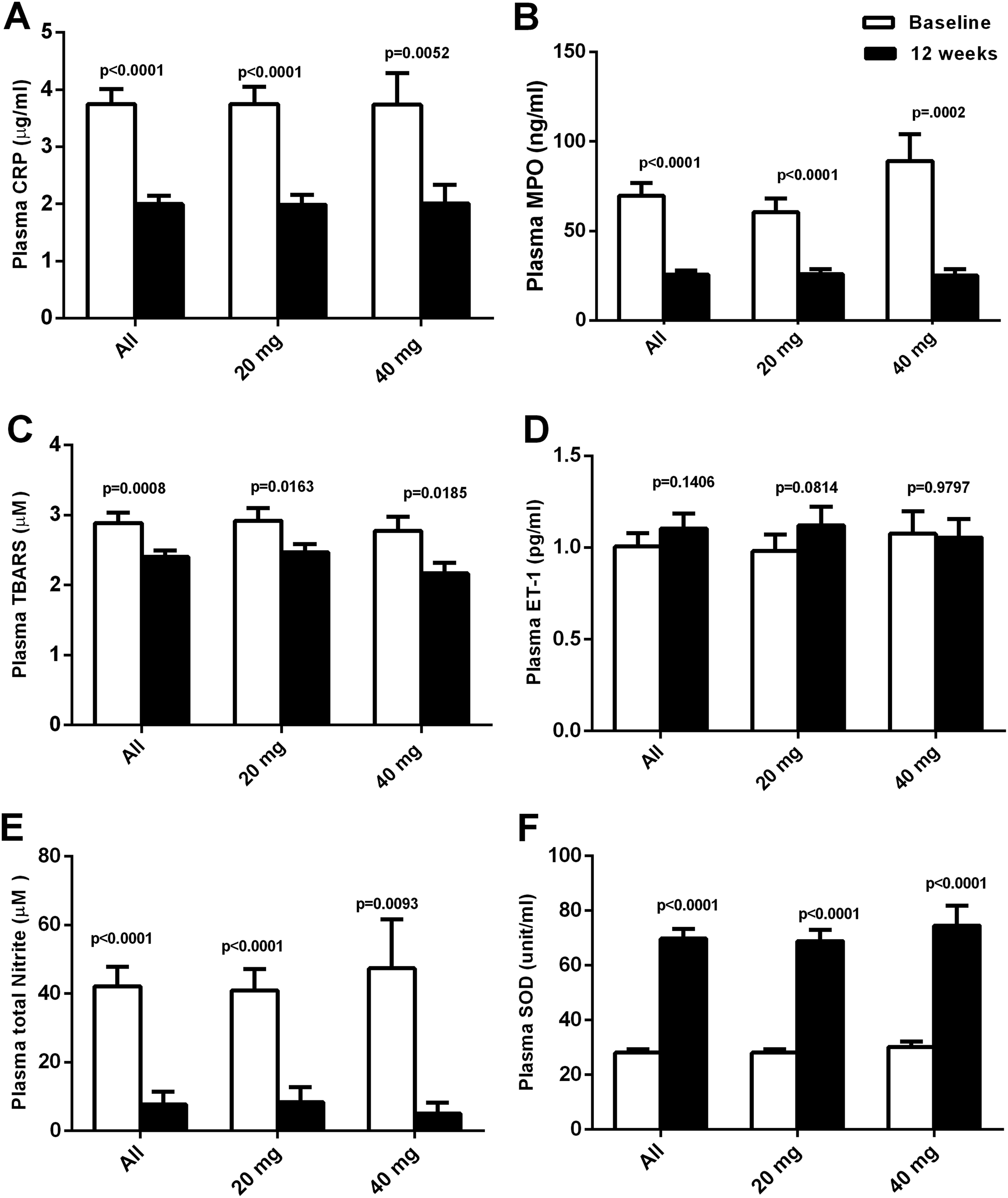
Effect of atorvastatin on plasma levels of inflammatory/oxidant biomarkers. Plasma levels of C-reactive protein (CRP, A), myeloperoxidase (MPO, B), thiobarbituric acid reactive substances levels (TBARS, C), endothelin-1 (ET-1, D), total nitrite (E), and superoxide dismutase (SOD, E) at baseline and at 12 weeks after atorvastatin treatment in all statin-treated patients (All), among patients who received moderate intensity atorvastatin (20 mg/d), and among those who received high-intensity atorvastatin (40 mg/d).
Similar findings were also observed for plasma CRP (pCRP) where patients with very high risk had higher baseline pCRP than patients with low risk (3.72 ± 0.24, 3.37 ± 0.42, 3.14 ± 0.75, and 1.88 ± 0.43 µg/mL, respectively,
Multivariate Analysis of Baseline Plasma Biomarkers With Risk of Atherosclerotic Cardiovascular Disease.a

Abbreviation: TBARS, thiobarbituric acid reactive substances.
aValues indicate adjusted correlation of atherosclerotic cardiovascular disease risk status (ASCVD) as the independent variable with square root of plasma biomarker as the dependent variable. Atherosclerotic cardiovascular disease was coded as 1= low risk, 2= moderate risk, 3= high risk, and 4 as very high risk. All models were adjusted to low-density lipoprotein levels (LDL), heart failure and pre use of medications including β blocker, angiotensin converting enzyme inhibitors or receptor blockers, diuretics, clopidogrel, and aspirin β is the slope/standard error.
Baseline plasma nitrite (pNitrite) was higher in patients with very high risk than patients with low risk of ASCVD (49.49 ± 6.97, 51.28 ± 9.19, 52.66 ± 12.77, and 15.49 ± 9.03 μM, respectively,
Effect of Atorvastatin on Plasma Levels of Inflammatory and Oxidant Biomarkers
Plasma levels of biomarkers were again evaluated 12 weeks after atorvastatin treatment (Figure 2A–F). Overall, pCRP content was reduced following atorvastatin treatment (change from baseline: −1.76 ± 0.27 μg/mL,
Plasma TBARS levels were also reduced following atorvastatin treatment (change from baseline: −0.49 ± 0.13 μM,
Plasma SOD activity was significantly increased following atorvastatin treatment (change from baseline: 39.39 ± 3.70 unit/mL,
Relative to baseline, no changes in levels of pCRP, pMPO, pET-1, pTBARS, pNitrite, or pSOD activities were detected in statin-untreated patients at follow-up (
Association of Plasma Inflammatory Biomarkers With Changes in Plasma Lipids
We further evaluated the association between baseline plasma oxidative stress biomarkers and changes in lipids relative to baseline. We found no correlation between biomarkers and lipid components, suggesting that baseline inflammatory status does not predict the lipid-lowering response to atorvastatin (all,
Discussion
Endothelial dysfunction is a critical mediator of atherosclerosis and plays a critical role in the development and progression of organ damages in patients with DM and HT. 15 Several mechanisms are involved in endothelial dysfunction and development of ASCVD including inflammation, vascular remodeling, increased generation of ROS/RNS, and increased production of vasoconstrictors or decreased vasodilators. 15 In the present study, we provide a strong evidence that elevated plasma levels of key inflammatory factors including CRP, MPO, and ET-1 are associated with an increased risk of ASCVD. Increased generation of total nitrite and lipid peroxides coupled with the decrease in SOD enzyme activity indicates that oxidative stress contributes to the risk of ASCVD in patients. Intriguingly, the use of atorvastatin therapy significantly reduced plasma pro-inflammatory and oxidant factors and increased pSOD activity in patients with different risk of ASCVD including those with moderate risk independent of dose intensity or lipid-lowering effects. Atorvastatin was used in this study because it is the most prescribed drug at our institution, has a marked LDL-lowering effect, and can be prescribed at both moderate- and high-intensity doses. As a lipophilic drug, it is associated with significant pleotropic effects when compared to other statins. 16
Statins are the most effective drugs to lower serum cholesterol levels and have proven their efficacy in primary and secondary prevention of CAD. These drugs also provide a number of pleiotropic effects on the cardiovascular system independent of their lipid-lowering effects. 17 Statins alter the expression of endothelial nitric oxide synthase (eNOS), the stability of atherosclerotic plaques, the production of pro-inflammatory mediators and ROS, and the reactivity of platelets, reducing the development and the progression of cardiac structural remodeling. 17 In the cholesterol biosynthesis pathway, statins inhibit the production of isoprenoid intermediates, the post-translational prenylation of small guanine nucleotide-binding proteins such as Rac and Rho, and other downstream effectors such as Rho kinase and nicotinamide adenine dinucleotide phosphate (NADPH) oxidases that are involved in vascular and cardiac inflammation and oxidative stress. 17
Current ACC/AHA guidelines strongly support the use of statin therapy in patients with risk of ASCVD for secondary prevention of MI, CAD, and stroke. 12 These guidelines were initially criticized as it is not clear yet whether or not to prescribe statin therapy for primary prevention in low-to-moderate risk individuals with no history of ASCVD and with a baseline 10-year risk of ASCVD >7.5%. 18 This decision requires a careful assessment of benefits versus risks as statin therapy is associated with increased risk of myopathy, cognitive impairment, DM, and liver toxicity. 18 Interestingly, no differences in plasma inflammatory and oxidant biomarkers were observed between the moderate- and the high-intensity atorvastatin therapies, suggesting that these pleiotropic effects are independent of lipid-lowering effect. Intriguingly, atorvastatin therapy reduced basal and elevated levels of plasma biomarkers in all statin-treated patients regardless of their ASCVD risk category, indicating that it can prevent the progression from 1 risk category to the next category. These findings support the use of moderate-intensity statin to reduce ASCVD risk in all patients including those with low-to-moderate risk without established ASCVD regardless of their serum LDL. This practice may minimize the side effects associated with high-intensity statin therapy. We have observed similar effects of atorvastatin on LDL and total cholesterol using the moderate and the high intensity. The lack of differences might be due to the smaller number of patients who received 40 mg atorvastatin as compared to the 20 mg atorvastatin, due to the differences between patients groups, or due to the short duration of the study, as maximum effects might require longer duration of treatment in some cases. 19
Several inflammatory factors are involved in the development of atherosclerosis including CRP, ET-1, and MPO. 14,19,20 Elevated levels of circulating CRP predict the development of CAD not only in patients with ischemic angina 20 but also in apparently healthy individuals. 8 Myeloperoxidase enzyme is an inflammatory mediator involved in LDL oxidation and conversion of the LDL lipid core into more atherogenic type, a rate-limiting step in atherosclerotic plaque formation. 21 We and others have shown that elevated circulating ET-1 levels are significantly associated with MI and poor clinical outcome in patients with CAD. 5,22 Here, we observed a trend for a stepwise increase in CRP levels to be associated with increasing risk of ASCVD, where patients with very high risk have significantly higher levels than low-risk patients, suggesting that pCRP may contribute to the development of atherosclerosis and might predict the risk of ASCVD in patients. Plasma MPO and ET-1 levels were also correlated with the risk of ASCVD, where patients with very high risk who have established CAD had higher pMPO than patients with low risk, indicating that MPO and ET-1 are primarily produced in response to coronary ischemia and hypoxia.
The concept of statin pleiotropy has provided a window of opportunity to evaluate other non-lipid-lowering mechanisms that may influence the risk of ASCVD. In type 2 DM, atorvastatin reduce pCRP and the degree of reduction in CRP levels is associated with the degree of improvement in endothelium-dependent vasodilatation. 23 In patients with ACS, CRP levels were also rapidly reduced with high-intensity atorvastatin (40 mg) over 30 days, indicating that the drug has rapid and early anti-inflammatory effects. 24 Previous studies showed that atorvastatin reduced pMPO levels in patients with ACS and inhibited MPO messenger RNA expression in human monocytes and macrophages. 25 Here, we also found that atorvastatin significantly reduced systemic MPO and CRP levels independent of lipid levels and regardless of the risk of ASCVD. Interestingly, we observed a greater reduction in pMPO in patients (primarily very-high-risk patients) who received high-intensity atorvastatin (40 mg/d) compared to those who received the moderate-intensity statin therapy (20 mg/d), suggesting that the atorvastatin effects on pMPO may be dose-dependent. As the increase in pCRP and MPO was most evident in very-high-risk patients, use of atorvastatin therapy may reduce the risk of ASCVD events for those patients. Attenuation of plasma inflammatory responses of CRP and MPO might be due to the activation of peroxisome proliferator-activated receptor γ 26 and NO generation. 27
A recent meta-analysis of 15 randomized controlled studies revealed that lipophilic statin therapy significantly reduced plasma ET-1 regardless of the duration or the dose. 28 In our study, we did not detect significant changes in plasma ET-1 following atorvastatin therapy with either the 20 or 40 mg doses. The variability in readings among patients may contribute. Further studies using larger sample sizes are required to better understand the effects of statins on pET-1 levels.
Hyperlipidemia promotes endothelial dysfunction and decreases synthesis of NO by increasing generation of multiple oxidants including peroxynitrite and lipid peroxidation products. 15 The increase in lipid peroxidation causes an increase in various different aldehydes, such as malondialdehyde (MDA). 29 Plasma MDA levels are increased in patients with familial hypercholesterolemia compared to controls. 30 Here, we did not observe any differences in baseline pTBARS levels among ASCVD risk categories. Lack of association with TBARS levels might be due to the lack of association of baseline lipid profile with ASCVD risk status. Plasma total nitrite levels (NO metabolites) were associated with an increased risk of ASCVD, documenting the role of NO signaling pathway and generation of RNS in the development and progression of ASCVD. Intriguingly, levels of total nitrite tend to increase in patients with moderate, high, and very high risk of ASCVD similarly, suggesting that ischemia is not the primary modulator of nitrite generation but modulated by other mechanisms or risk factors such as smoking, HT, and DM. In addition to generation of ROS/RNS, oxidative stress could occur in cases of decreased antioxidant defenses. Superoxide dismutase is an antioxidant enzyme that catalyzes dismutation of superoxide anion into hydrogen peroxide and water. 31 A stepwise decrease in the activity of SOD with the increase in ASCVD risk status was found in our study, clearly indicating that reduced antioxidant activity coupled with increased inflammatory and oxidants factors contribute to increased risk of ASCVD.
Previously, it has been demonstrated that low-dose atorvastatin (10 mg/d) and pravastatin (10 mg/d) reduced plasma MDA in patients with hypercholesterolemia. 32 The reduction in MDA levels may be due to the ability of atorvastatin to reduce systemic oxidants and inflammatory mediators and the expression of essential NADPH oxidase molecules that are the major sources of ROS in the vessel wall. 32 In addition to lipid peroxides, the decrease in NO bioavailability plays a primary role in endothelial dysfunction, suggesting that drugs with endothelium-protective properties may produce significant therapeutic benefits. 27 In patients with familial hypocholesteremia, oxidized lipids stimulate eNOS and generation of nitrite, and use of atorvastatin reduced plasma MDA, nitrite, 33 and nitrotyrosine levels. 34 Statins prevent the scavenging of NO by reducing the generation of toxic radicals, adhesion molecules, and neutrophils extravasation. 27 Here, we show that pTBARS and pNitrite content are significantly reduced following atorvastatin treatment. The reduction in pMPO and CRP coupled with the activation of SOD antioxidant enzyme might contribute to the reduction in pTBARS and pNitrite. These effects were evident in all statin-treated patients regardless of ASCVD risk, atorvastatin dose intensity, or baseline levels of inflammatory/oxidants biomarkers, supporting the use of moderate-intensity atorvastatin in patients with moderate to high risk without established ASCVD.
Conclusions
Our study suggests that key markers of vascular inflammation/oxidative stress such as CRP, MPO ET-1, and nitrite are associated with an increased risk of ASCVD. The increase in plasma inflammatory markers was most evident in very-high-risk patients who primarily have established CAD, suggesting that coronary ischemia and hypoxia are the main stimulatory factors for secretion of these factors. Plasma SOD were associated with reduced risk of ASCVD. Use of atorvastatin for 12 weeks reduced serum lipids as well as plasma markers of CRP, MPO, total nitrite, and TBARS and increased SOD activity using the moderate- and high-intensity therapy independent of changes in plasma lipids. Concordant with AHA/ACC guidelines, our study is supportive of the use of statin therapy in patients with increased risk of ASCVD including those with moderate risk without established ASCVD.
Footnotes
Acknowledgments
The authors thank Dr David Van Wagoner, PhD, Cleveland Clinic for helpful editorial assistance.
Author Contribution
Fadia Mayyas contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Duha Baydoun contributed to design and data collection, contributed to acquisition, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Rasheed Ibdah contributed to design and data interpretation, contributed to acquisition, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Khalid Ibrahim contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the deanship of research (grant number 292/2015) at Jordan University of Science and Technology, Irbid, Jordan.
