Abstract
Although diminazene aceturate (Berenil) is widely used as a trypanolytic agent in livestock, its mechanisms of action remain poorly understood. We previously showed that Berenil treatment suppresses pro-inflammatory cytokine production by splenic and liver macrophages leading to a concomitant reduction in serum cytokine levels in mice infected with Trypanosoma congolense or challenged with LPS. Here, we investigated the molecular mechanisms through which Berenil alters pro-inflammatory cytokine production by macrophages. We show that pre-treatment of macrophages with Berenil dramatically suppressed IL-6, IL-12 and TNF-α production following LPS, CpG and Poly I:C stimulation without altering the expression of TLRs. Instead, it significantly down-regulated phosphorylation of mitogen-activated protein kinases (p38, extracellular signal-regulated kinase and c-Jun N-terminal kinases), signal transducer and activator of transcription (STAT) proteins (STAT1 and STAT3) and NF-кB p65 activity both in vitro and in vivo. Interestingly, Berenil treatment up-regulated the phosphorylation of STAT5 and the expression of suppressor of cytokine signaling 1 (SOCS1) and SOCS3, which are negative regulators of innate immune responses, including MAPKs and STATs. Collectively, these results show that Berenil down-regulates macrophage pro-inflammatory cytokine production by inhibiting key signaling pathways associated with cytokine production and suggest that this drug may be used to treat conditions caused by excessive production of inflammatory cytokines.
Keywords
Introduction
Inflammation is the earliest response of the host defense system to injury or insult, including infection. During infection, an inflammatory response may be beneficial or detrimental. Inflammation is beneficial to the host if it provides protection against infection. However, uncontrolled and dysregulated inflammation can be detrimental, as is the case in septic shock. 1 Thus, inflammatory processes must be tightly regulated by highly coordinated signals that initiate, maintain and ultimately shut down the cascade when the insult is eliminated. Cellular and tissue damage occur when there is an imbalance that results in excessive inflammation. In addition, prolonged inflammation results in a chronic inflammatory state, which is the major pathogenic mechanism in a number of chronic diseases, including, for example, rheumatoid arthritis, inflammatory bowel disease and psoriasis. 2
TLRs are a family of evolutionarily conserved receptors that play a crucial role in early host defense against microbial pathogens. 3 They are major PRR receptors present on macrophages and other innate immune cells that act as the first line of defense against pathogens. Upon recognition of their respective ligands, which are molecular patterns expressed on pathogens, a cascade of intracellular signaling events is activated leading to the activation of NF-κB, which ultimately results in the transcription of pro-inflammatory genes and the production of inflammatory cytokines. 4
An inflammatory response is orchestrated by the interaction between several inflammatory mediators and immune cells. 5 Macrophages play a critical role in inflammation via Ag presentation, phagocytosis and the production of pro-inflammatory cytokines [e.g. IL-6, IL-12, TNF-α (TNF)] and other factors that result in immunomodulation. 6 Additionally, macrophages also respond to cytokines, free oxygen radicals and complement products produced as result of an inflammatory process. 7 The production and autocrine effects of inflammatory cytokines on macrophages results in the activation of transcription factors (e.g. NF-кB) and protein kinases (e.g. MAPKs), which, in turn, regulate the expression of many target genes, including those that are involved in inflammatory cytokine production. 8
Diminazene aceturate (Berenil) has been the drug of choice for treatment of animal trypanosomiasis since 1955. It is an aromatic diamidine consisting of two amidinophenyl moieties linked by a triazene bridge. 9 In addition to its trypanocidal activity, Berenil also has excellent anti-babesial activity, which has increased its field applicability. 10 Although the compound has been in use for more than half a century, the exact mechanism of action and its effect on the host immune system are still poorly understood. Some studies suggest that the compound binds to trypanosomal kinetoplast DNA in a non-intercalative manner through specific interaction with sites rich in adenine–thymine base pairs. 11 This causes irreversible damage resulting in the development of akinetoplast parasites.12,13 Additionally, Berenil induces condensation of heterochromatin during the G2 phase of cell cycle, which causes the DNA to become completely unfolded. 14 Berenil also alters the confirmation of DNA by interfering with binding of DNA topoisomerases. 15
Although many of the anti-parasitic effects of Berenil have been attributed to its biochemical properties enumerated above, few reports suggest that the compound may have some effect on the host immune system. In Trypanosoma congolense-infected cattle, Berenil treatment at the time of vaccination abolishes immunosuppression and enhances the immune response to Leptospira biflexa. 16 Treatment of T. congolense-infected highly susceptible BALB/c mice with Berenil leads to control of parasitemia and a dramatic increase in plasma levels of parasite-specific IgG2a and IgG3 Abs. 17 In addition, Berenil treatment leads to complete cure of infected mice and results in resistance to homologous challenge. 18 In a seminal study, Arowolo et al. 19 showed that in addition to its trypanocidal action, Berenil treatment blocks histamine-induced responses in tissues and exert some anti-histaminic and anti-inflammatory effects in vivo. Recently, we showed that Berenil treatment of T. congolense-infected mice dampens T-cell and macrophage hyperactivation, and lowers serum levels of pro-inflammatory cytokines by directly altering the production of these cytokines by splenic and hepatic macrophages. 20 Collectively, these results indicate that, in addition to its trypanolytic effect, Berenil also modulates the host immune system.
In this study, we performed in vitro and in vivo experiments to more clearly investigate how Berenil down-regulates pro-inflammatory cytokine production in macrophages. We show that Berenil does not mediate its anti-inflammatory properties by altering TLR expression on macrophages. Rather, the compound strongly down-regulated phosphorylation of MAPKs [extracellular signal-regulated kinase (ERK), p38 and c-Jun N-terminal kinase (JNK)], signal transducers and activators of transcription (STAT1 and STAT3) and NF-кB p65 subunit, and directly interferes with IL-6 promoter activity. In addition, Berenil also up-regulates the expression of suppressor of cytokine signaling (SOCS) in macrophages, suggesting that it could exert both direct and indirect effects on pro-inflammatory cytokine production in macrophages.
Materials and methods
Reagents
LPS from Escherichia coli was purchased from DIFCO Laboratories (Detroit, MI, USA). Diminazene aceturate (Berenil) was purchased from Sigma Aldrich (Mississauga, ON, Canada). Phycoerythrin (PE)-conjugated mAb against murine TLR4 (TLR4/MD-2 complex) and TLR2 and APC-conjugated mAb against CD14 were purchased from BD Biosciences (Mississauga, ON, Canada). FITC anti-mouse TLR9 mAb was purchased from eBioscience (San Diego, CA, USA). Rabbit anti-mouse p38 MAPK and ERK1/2 mAb, affinity purified rabbit anti-phospho p-38 MAPK (Thr180/Tyr182), affinity purified mouse anti-phospho ERK1/2 (Thr 202/Tyr204), rabbit anti-total and phosphor-specific SAP/JNK (Thr183/Tyr 185) mAbs, rabbit polyclonal anti-STAT1, STAT3, STAT5, rabbit anti-phospho and total NF-κB mAb, and rabbit anti-SOCS1 and SOCS3 Abs were all purchased from Cell Signalling Technology (Danvers, MA, USA). A fast-activated cell-based ELISA NF-кB p65 profiler kit was purchased from Active Motif (Carlsbad, CA, USA). RPMI media, antibiotics and cell culture reagents were purchased from HyClone Laboratories (Logan, UT, USA).
Mice
Six to 8-wk-old female C57BL/6 mice were purchased from the Central Animal Care Services, University of Manitoba (Winnipeg, Canada). All mouse experiments were approved by the University of Manitoba Animal Care Committee in accordance with the regulation of the Canadian Council on Animal Care.
Cell lines and cell cultures
The origin of retrovirus-immortalized bone marrow-derived macrophage (BMDM) cell line (ANA-1) from C57BL/6 mice has been previously described. 21 The immortalized cell lines were grown in complete medium (RPMI 1640 medium supplemented with 10% FBS [Hyclone, Fisher Scientific, Ottawa, Canada], 10 U/ml penicillin/streptomycin and 50 µM 2-mercaptoethanol). The cells were cultured in 24-well plates, treated overnight (12 h) with different concentrations of Berenil (0.01–10.00 µg/ml) and then stimulated with LPS (1 µg/ml) for an additional 12–16 h. The culture supernatant fluids were collected and stored at −20℃ until used for cytokine assays.
BMDM and bone marrow dendritic cells
BMDM were differentiated from marrow cells as previously described. 22 Briefly, bone marrow cells were isolated from the femur and tibia of C57BL/6 mice and differentiated into macrophages using complete medium supplemented with 30% L929 cell culture supernatant (conditioned media). After 7 d, the cells were harvested, washed, re-suspended in complete medium and used for experiments as described below. To differentiate bone marrow dendritic cells (BMDC), marrow cells were seeded in 100 × 15 mm Petri dishes at 2 × 105/ml in the presence of recombinant murine granulocyte–macrophage colony stimulating factor (GM-CSF) (20 ng/ml; PeproTech, Rocky Hill, NJ, USA). The culture medium was changed twice on d 3 and 6, and on d 7, the non-adherent cells (DC) were collected, washed twice with complete medium and used for in vitro experiments as described below.
Assessment of Berenil toxicity to cells at different concentrations
BMDM were grown in complete RPMI medium, treated overnight with different concentrations of Berenil (0.01–100.00 µg/ml) and assessed for cell survival by MTT, Trypan blue exclusion and propidium iodide assays. The MTT assay was done as described previously.23–25 Briefly, the cells (in microplates) were treated overnight with different concentrations of Berenil and 20 µl of MTT solution (Sigma Aldrich) was added to each well and incubated for 4 h at 37℃. Thereafter, the supernatants were discarded and 200 µl of DMSO was added to the wells to dissolve the insoluble formazan crystals. Formazan quantification was performed using an automatic plate reader with a 570-nm test wavelength and a 690-nm reference wavelength. The Trypan blue dye exclusion test and propidium iodide assay for cellular viability were performed as explained previously.26,27
Assessment of TLR expression by flow cytometry
The effects of Berenil on TLR expression on macrophages was assessed by flow cytometry. Briefly, differentiated BMDMs were pre-incubated with varying concentrations of Berenil for 12–24 h and stained with PE-conjugated mAb against murine TLR4 and TLR2, and APC-conjugated mAb against CD14. TLR9 expression was detected by intracellular staining using the FITC-conjugated mAb against murine TLR9. The relative expression of TLR4, TLR2, CD14 and TLR9 was assessed by flow cytometry and quantified by determining the mean fluorescence intensities (MFI).
Cytokine ELISA
The levels of IL-6, IL-12p40 and TNF in culture supernatants from BMDM and ANA cells treated with Berenil (10 µg/ml), LPS (1 µg/ml) or CpG (5 µg/ml) were determined by ELISA using paired Abs and appropriate recombinant cytokine standard (eBioscience) according to the manufacturer’s suggested protocols. The sensitivities of the ELISA were 15, 125 and 50 pg/ml for IL-6, IL-12p40 and TNF, respectively.
Western blot
Assessment of MAPKs, STATs and NF-кB p65 subunit phosphorylation, and the induction of SOCS1 and SOCS3 were determined by Western blot, as previously described. 28 Briefly, fully differentiated BMDMs were grown in Petri-plates and, once confluent, the cells were serum starved for about 24–48 h. The cells were then treated with Berenil (10 µg/ml) overnight and stimulated with LPS (1 µg/ml) for different time points as indicated. Thereafter, the cells were washed twice with ice cold PBS, lysed with NP40 lysis buffer and the total protein in the lysate was determined by BCA protein assay. Ten μg of protein lysate was resolved in 10% SDS-PAGE, transferred onto a nitrocellulose membrane, blocked with 5% BSA and probed overnight at 4℃ with primary Abs (rabbit anti-mouse mAb) specific for SOCS1 and SOCS3, phosphorylated ERK, p38, JNK, STAT1, STAT3, STAT5 and NF-кB p65 subunit. The blots were washed rigorously with Tris-buffered saline with Tween 20 (TBST), incubated with anti-rabbit HRP-conjugated secondary Ab and the bands were revealed by enhanced chemiluminescence (ECL) (Amersham, GE Healthcare Biosciences, Pittsburgh, PA, USA) reagents. Thereafter, the blots were routinely stripped and total ERK, p38, JNK, STAT1, STAT3, STAT5 and NF-кB p65 subunit were detected and used as loading controls. In addition, the membranes were also stripped and probed for β-actin as additional loading control. Densitometric analysis was performed and the integrated density values were presented as ratio of phosphorylated over the total protein compared with media control.
Peritoneal macrophage isolation after treatment with Berenil
Groups of mice were treated with or without Berenil i.p. (14 mg/kg). After 24 h, the mice were either challenged with PBS or LPS (5 mg/kg), sacrificed at different times (3–12 h) and the peritoneal lavage fluid was collected as previously explained. 29 Macrophages were isolated from the peritoneal lavage and either directly lysed with lysis buffer or further stimulated in vitro with LPS (1 µg/ml) for different time periods. The cells were then routinely lysed and the lysates were used for Western blot as above. In addition, the peritoneal wash fluids were assessed for IL-6 and TNF by ELISA.
Quantification of phosphorylated and total NF-кB p65
NF-кB p65 phosphorylation was detected by using Fast Activated Cell-based ELISA NF-кB p65 profiler kit (Active Motif) according to the manufacturer’s suggested protocols. Briefly, the cells were cultured in 96-well plates and stimulated with Berenil (10 µg/ml) overnight and then with LPS (1 µg/ml). At the indicated times, the cells were fixed rapidly, washed and incubated with phospho and total NF-кB p65 primary Ab for overnight at 4℃. The plates were then washed, incubated with HRP-conjugated secondary Ab, developed and the relative number of cells in each well was determined by using crystal violet. The phospho and total NF-кB p65 signals were normalized for corresponding cell numbers and the ratio of phosphorylated NF-кB p65 to total NF-кB p65 over the controls was determined.
Luciferase reporter constructs and transient transfection
Dr Gail Bishop, University of Iowa, generously provided the IL-6 promoter luciferase constructs used in this study. ANA cells in complete RPMI medium were seeded into 24-well plates and at 70–80% confluency, transient transfection of the cells was performed by using Turbofect (Fermentas Canada, Ottawa, ON, Canada) according to the manufacturer’s suggested protocol. Into each well, cells were co-transfected with 0.8 µg IL-6 promoter luciferase DNA vector and 0.2 µg Renilla luciferase vector pRL-TK for 24 h. The cells were then washed and treated with Berenil (10 µg/ml) and LPS (1 µg/ml) for 12 h. Following additional washings, the cells were lysed with lysis buffer and the cell lysates were collected. Luciferase activity was assessed by a luminometer using the Dual Luciferase Assay System Kit (Promega, Madison, WI, USA) according to manufacturer’s suggested protocol. All values were normalized to Renilla luciferase activity and expressed relative to transfected unstimulated control cells.
Statistical analysis
Cytokine and densitometric data are presented as mean ± SEM. A two-tailed Student’s t-test was used to compare differences in cytokine production. Two-way ANOVA was used to compare differences in phosphorylation between different groups after densitometry. Significance was considered if P < 0.05. All analyses were carried out using GraphPad Prism software.
Results
Berenil down-regulates LPS-induced pro-inflammatory cytokines production in BMDM and BMDC
We previously showed that injection of Berenil dramatically lowers serum levels of pro-inflammatory cytokines (including IL-6, IL-12p40 and TNF) in mice infected with T. congolense or following LPS challenge.
20
This effect was mediated by direct modulatory effect on splenic and hepatic CD11b+ cells from treated mice. To more clearly determine the molecular mechanisms through which Berenil suppresses pro-inflammatory cytokines production, we pre-treated immortalized macrophage cell line (ANA-1) or primary BMDMs and BMDCs overnight with varying concentrations of the compound (ranging from 10 ng/ml to 10 µg/ml) and then stimulated them with LPS (1 µg/ml) for 12–16 h. Berenil treatment suppressed LPS-induced IL-6, IL-12p40 and TNF production in a dose-dependent manner in both immortalized cell line (Supplementary Figure S1A–C) and primary BMDMs (Figure 1A–C). To determine whether the effect of Berenil was restricted to macrophages only, we also assessed LPS induced pro-inflammatory cytokine production by BMDC following pre-treatment with Berenil. As shown in Figure 1D–F, and consistent with the effects observed in BMDMs, Berenil also down-regulated LPS-induced pro-inflammatory cytokine production by BMDCs, suggesting that the effect is not restricted only to macrophages. Interestingly, Berenil did not down-regulate the production of MCP-1 in macrophages (data not shown), suggesting that it does not exhibit global inhibitory effect on inflammatory cytokine release. In addition, at < 10 µg/ml, the drug was non-toxic to the cells, as assessed by MTT, trypan blue dye exclusion and propidium iodide assays (Supplementary Figure S2A–C). These results indicate that, as observed in vivo,
20
Berenil directly and significantly reduced the production of pro-inflammatory cytokines by macrophages and DC in vitro.
Berenil pre-treatment down-regulates LPS-induced cytokine production in macrophages and DC. BMDM and BMDC were differentiated from marrow cells isolated from C57BL/6 mice, treated with Berenil (10 µg/ml) overnight and stimulated with LPS (1 µg/ml). After 12 h, the culture supernatant fluids were collected and assayed for IL-6 (A, D), IL-12 (B, E) and TNF (C, F) by sandwich ELISA. Shown are IL-6, IL-12 and TNF production by BMDM (A–C) and BMDC (D–F). The data presented are representative of three independent experiments with similar results. *P < 0.05; **P < 0.01; ***P < 0.001.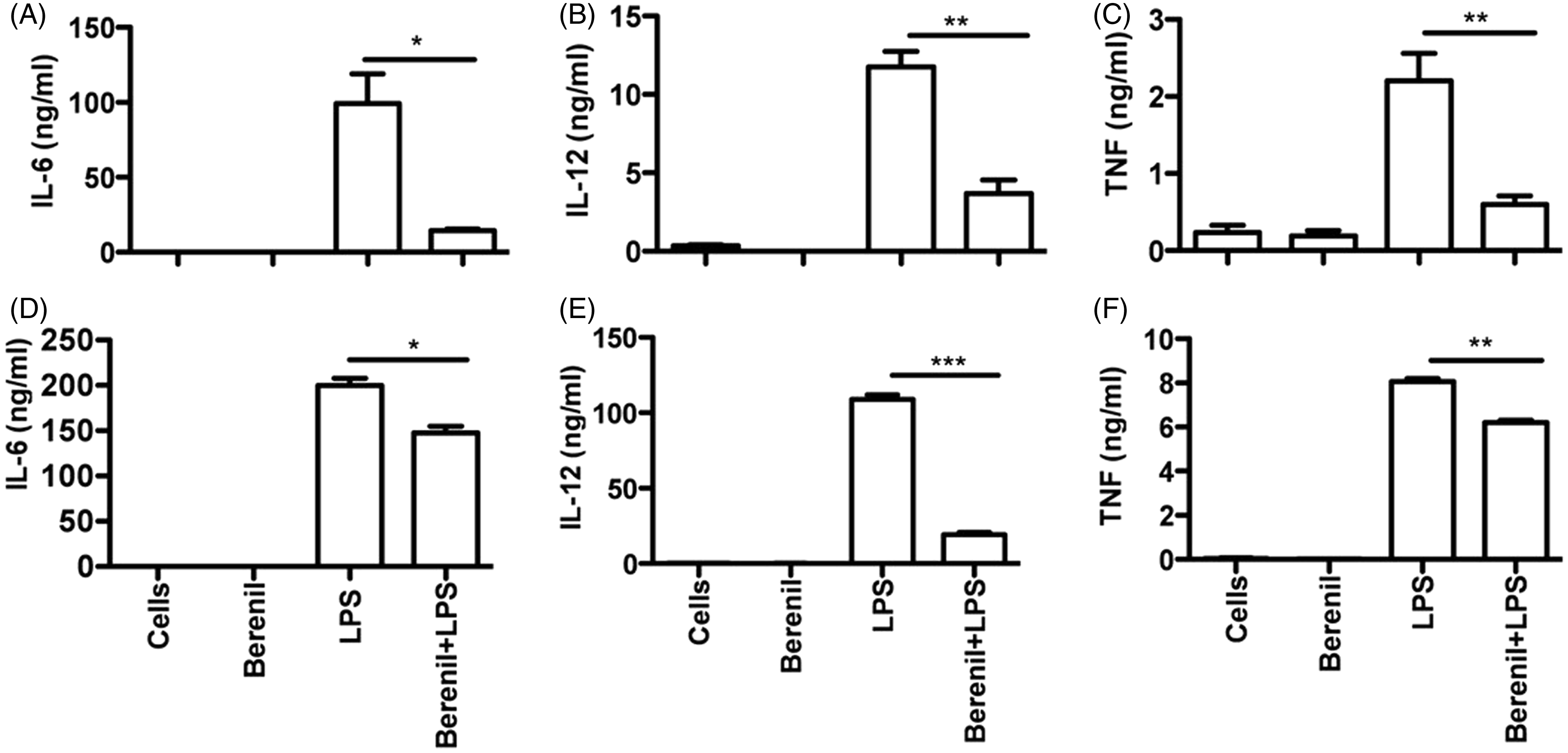
Berenil down-regulates CpG induced-production of pro-inflammatory cytokine production in macrophages and DC
Next, we assessed whether the down-regulatory effects of Berenil on pro-inflammatory cytokine production was global or specific to LPS stimulation. Bacterial DNA and synthetic oligonucleotides (ODN) containing unmethylated CpG motifs (CpG–ODN) can stimulate cells of innate and adaptive immune system (including macrophages) leading to increased pro-inflammatory cytokine production.30–32 Because CpG DNA receptor (TLR 9) shares common signaling pathways with the LPS receptor (TLR4),
33
we compared the cytokine production in Berenil pre-treated BMDM and BMDC following CpG ODN stimulation. Pre-treatment of BMDMs and BMDCs with Berenil significantly down-regulated CpG-induced IL-6, IL-12p40 and TNF production (Figure 2A–F). In addition, Berenil treatment also down-regulated Poly I:C (a TLR3 agonist) induced IL-6 production by BMDMs (Supplementary Figure S3).
Berenil pre-treatment down-regulates CpG-induced cytokine production in macrophages and DC. BMDM and BMDC were treated with Berenil (10 µg/ml) overnight, stimulated with CpG ODN (5 µg/ml) for an additional 12 h and the culture supernatant fluids were assayed for IL-6 (A, D), IL-12 (B, E) and TNF (C, F) by sandwich ELISA. Shown are IL-6, IL-12 and TNF production by BMDM (A–C) and BMDC (D, E). The data presented are representative of three independent experiments with similar findings. *P < 0.05; ***P < 0.001.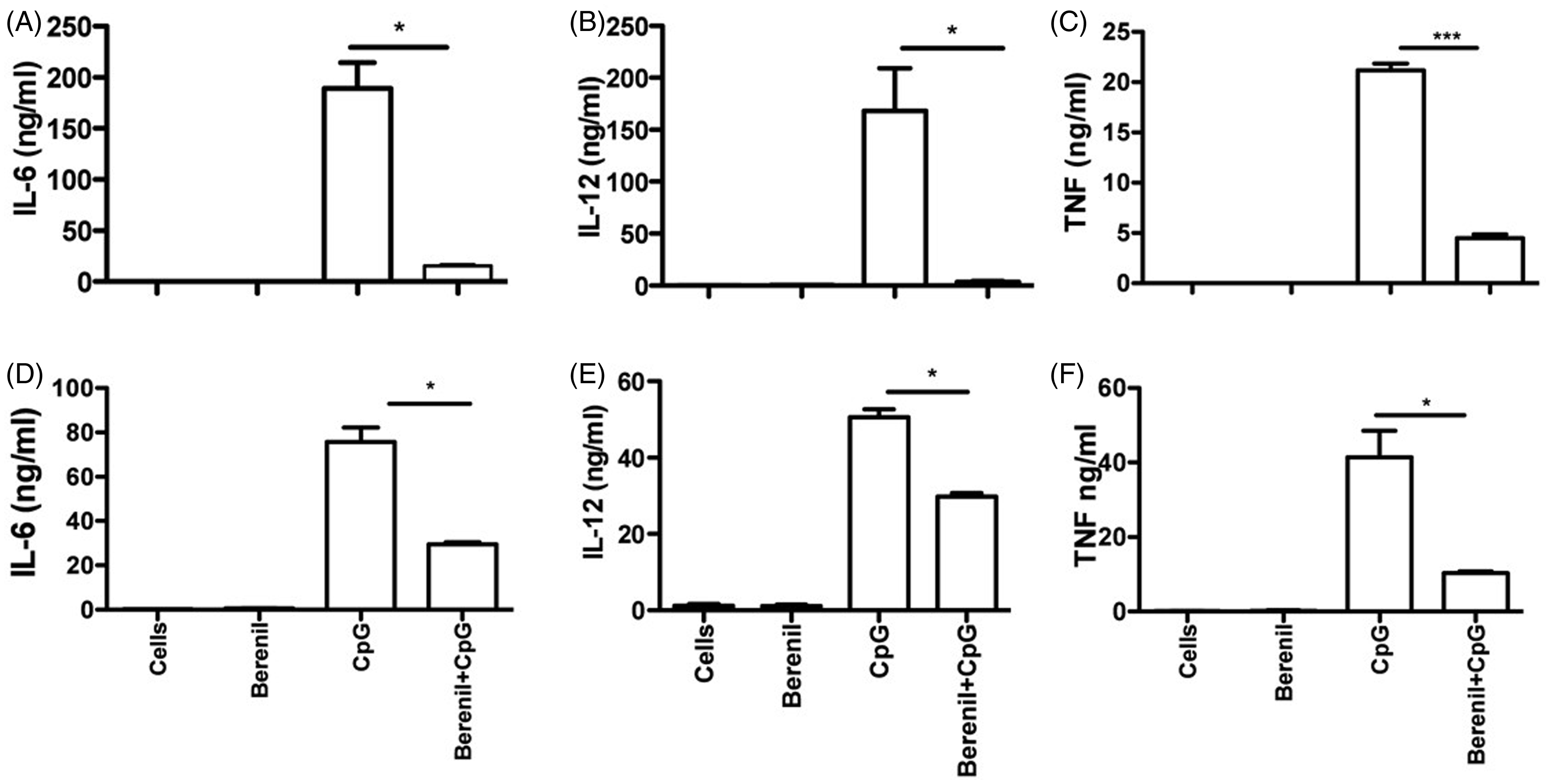
Berenil does not alter TLR expression on macrophages
TLRs are innate immune sensors that recognize pathogen-associated molecular patterns (PAMPs). Binding of specific ligands/agonists to their respective TLRs activates cell signaling pathways leading to up-regulation of co-stimulatory molecules and the production of pro-inflammatory cytokines.
34
Because we found that pre-treatment of BMDMs with Berenil significantly suppressed their production of IL-6, IL-12p40 and TNF, we hypothesized that Berenil alters TLR expression on macrophages. Therefore, we pre-treated BMDMs with varying concentrations of Berenil (0.01–10.00 µg/ml) and assessed the expression of TLR4–MD2 complex and CD14, which is involved in LPS recognition and signaling.
35
Pre-treatment with Berenil did not alter the surface expression of TLR4–MD2 complex and CD14 on macrophages (Figure 3A–D). Some reports show that signaling via TLR2 is required along with TLR4 for optimal LPS-induced intracellular signaling that leads to pro-inflammatory cytokines production in macrophages.
36
Therefore, we considered the possibility that Berenil might be altering the TLR2 expression. As seen for TLR4, Berenil pre-treatment did not affect TLR2 expression (Figure 3E, F). Similarly, Berenil pre-treatment did not alter TLR9 expression on macrophages (Figure 3G, H). Collectively, these results show that the down-regulation of pro-inflammatory cytokines production by Berenil is not due to alteration in TLR expression on macrophages, suggesting that it may be altering downstream intracellular signaling pathways following ligation of TLRs by their respective ligands.
Berenil treatment does not affect the expression of TLR and other key LPS binding molecules on macrophages. BMDM were treated with or without Berenil (10 μg/ml) overnight, stained with PE-labeled mAb against TLR4 and TLR2, APC-conjugated mAb against CD14 and FITC-conjugated mAb against TLR9 and analyzed by flow cytometer. Shown are representative histograms of TLR4 (A), CD14 (C), TLR2 (E) and TLR9 (G). The bar graphs represent the corresponding MFI (B, D, F, H). The data presented are representative of three independent experiments with similar findings.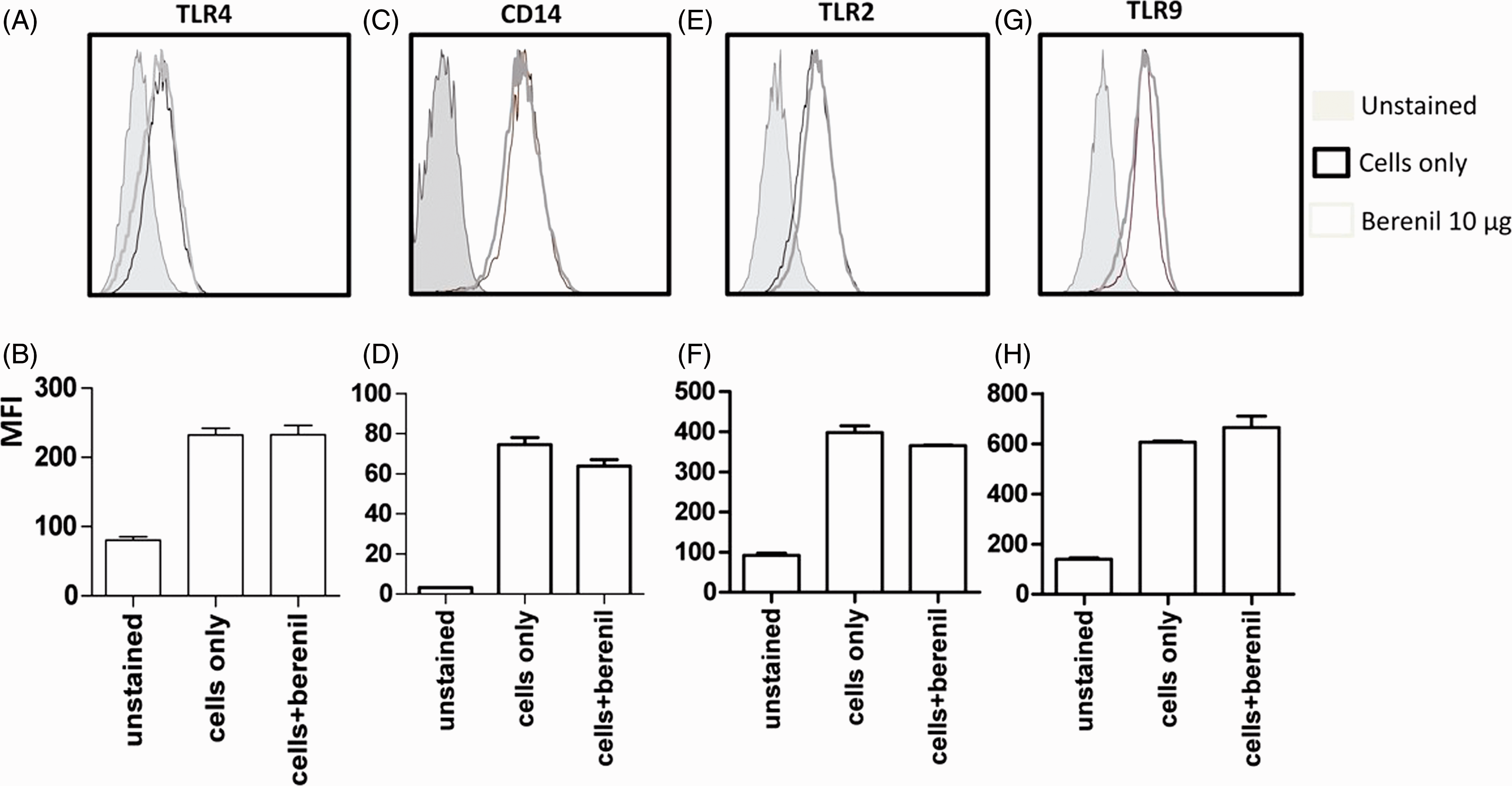
Berenil pre-treatment down-regulates the phosphorylation of MAPKs and STATs in macrophages
MAPK and STAT are critical signaling molecules that regulate pro-inflammatory cytokine production in immune cells.37,38 As we found that Berenil does not mediate its inhibitory effects on pro-inflammatory cytokines production by altering TLR expression, we speculated that the drug might be altering intracellular signaling molecules and pathways that regulate pro-inflammatory cytokine gene expression in macrophages. Therefore, we used Western blot to investigate the effect of Berenil pre-treatment on phosphorylation of BMDMs in vitro. Pre-treatment with Berenil inhibits LPS-induced phosphorylation of ERK, JNK and P38, (Figure 4A–D; JNK data not shown) and STATs (STAT1 and STAT3; Figure 5A–D). Interestingly, pre-treatment with Berenil did not suppress, but rather enhanced, STAT5 phosphorylation (Figure 5E, F), suggesting the compound does not globally affect intracellular phosphorylation events in macrophages. Collectively, these results suggest that the inhibitory effect of Berenil on cytokine production is mediated by the down-regulation of MAPKs and STATs signaling.
Berenil down-regulates the phosphorylation of MAPKs in macrophages. BMDM were treated with Berenil (10 µg/ml) overnight and then stimulated with LPS. At indicated times, the cells were lysed and the lysates were assessed by Western blot for phosphorylation of ERK and p38 (A–D). The same blots were stripped and re-probed with Abs against total ERK and p38 and used as loading controls. β-Actin was also done as loading control. The Western blot results represent one of three independent experiments. Densitometric analysis was performed from three blots and presented as ratio of phosphorylated proteins over total (unphosphorylated) controls compared to media. The data presented are representative of three independent experiments with similar findings. *P < 0.05; ***P < 0.001. Berenil down-regulates the phosphorylation of STATs in macrophages. BMDM were treated with Berenil (10 µg/ml) overnight and then stimulated with LPS (1 µg/ml). At indicated times, cells were lysed and the lysates were assessed by Western blot for phosphorylation of STAT1, STAT3 and STAT 5 (A–F). The same blots were stripped and re-probed with Abs against total STAT1, STAT3 and STAT5, and used as loading control. The Western blot results represent one of three independent experiments. Densitometric analysis was performed from three blots and presented as ratio of phosphorylated proteins over total (unphosphorylated) controls and compared to the media control. The data presented are representative of three independent experiments with similar findings. *P < 0.05; **P < 0.01; ***P < 0.001.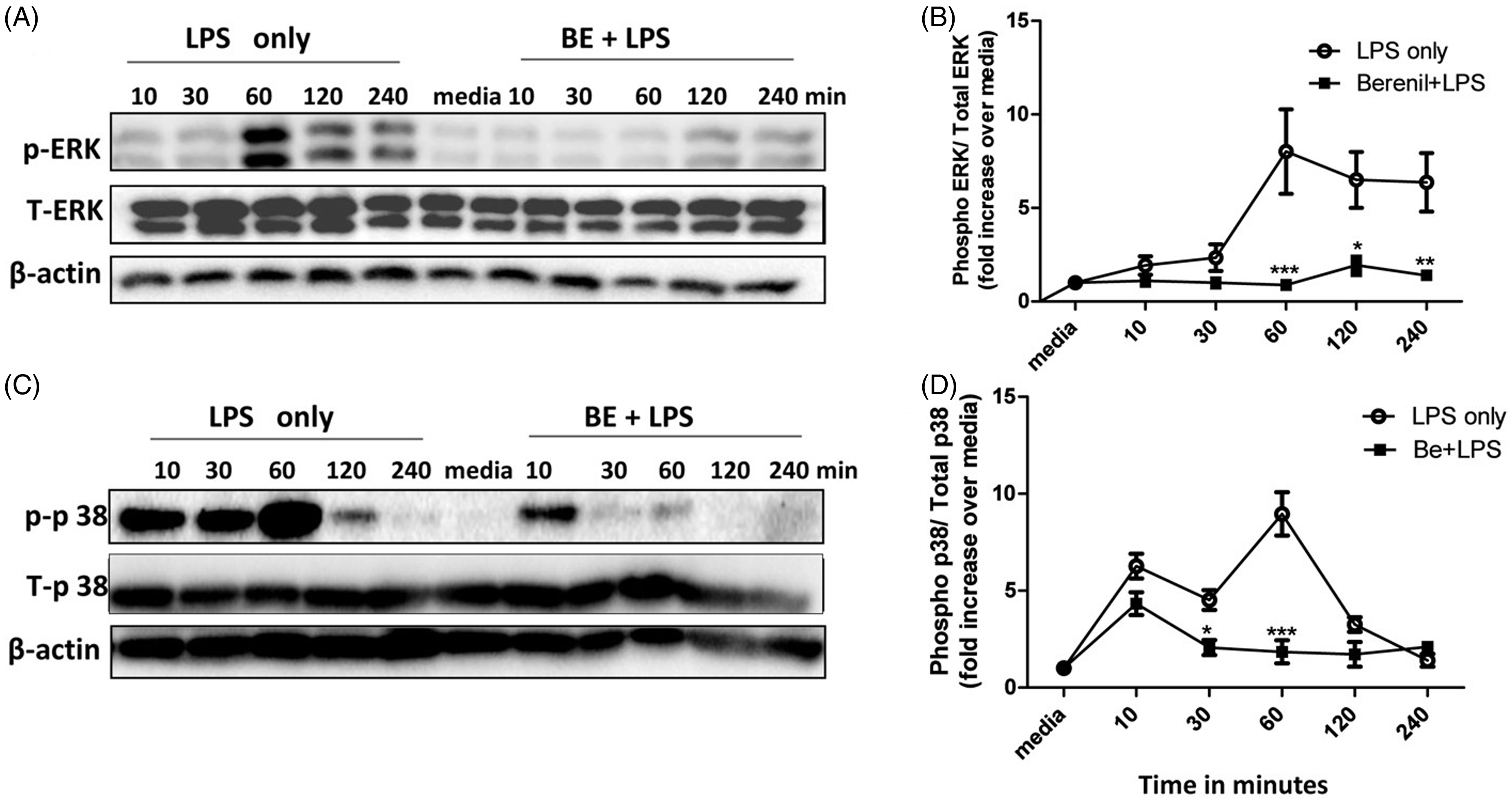
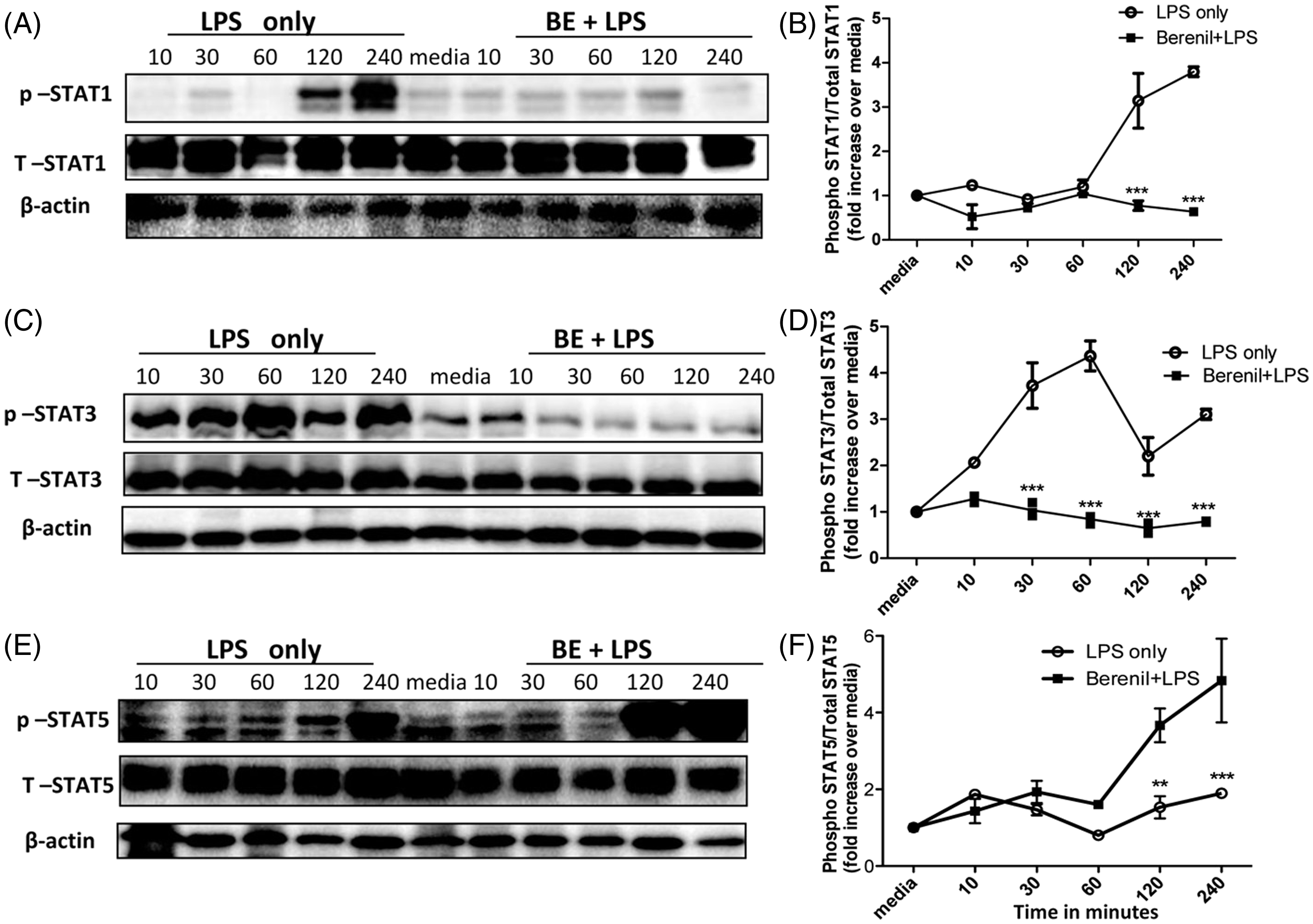
Berenil down-regulates the phosphorylation of MAPKs and STATs in peritoneal macrophages
Next, we evaluated whether Berenil-induced down-regulation of MAPK and STAT phosphorylation was reproducible in vivo. We pre-treated mice with Berenil (14 mg/kg), and, after 16–24 h, isolated the peritoneal macrophages and re-stimulated them in vitro with LPS (1 µg/ml). As shown in Figure 6A, B, and consistent with our in vitro observations, treatment with Berenil down-regulated LPS-induced phosphorylation of ERK (data not shown), p38, STAT1 (data not shown) and STAT3 (Figure 6A, B) at different times tested.
Berenil down-regulates MAPK and STAT phosphorylation in peritoneal macrophages. Groups of mice were treated with or without Berenil (14 mg/kg) overnight and sacrificed to isolate peritoneal macrophages. The cells were further stimulated in vitro with LPS (1 µg/ml) for 30, 60 and 120 min, lysed and the lysates were assessed by Western blot for phosphorylation of p38 (A) and STAT3 (B). The same blots were stripped and re-probed with Abs against total p38 and STAT3 and used as loading control. β-Actin was also done as loading control. In another set of experiments, mice treated with or without Berenil (14 mg/kg) overnight were injected with LPS (10 mg/kg, i.p.) and, at indicated times, sacrificed and lysates of peritoneal macrophages were assessed directly for phosphorylation of p38 (C) and STAT3 (D) by Western blot. In addition, the levels of IL-6 (E) and TNF (F) in the peritoneal wash fluids were assayed by ELISA. The data presented are representative of two independent experiments with similar results (n = 3 mice per group). **P < 0.01; ***P < 0.001.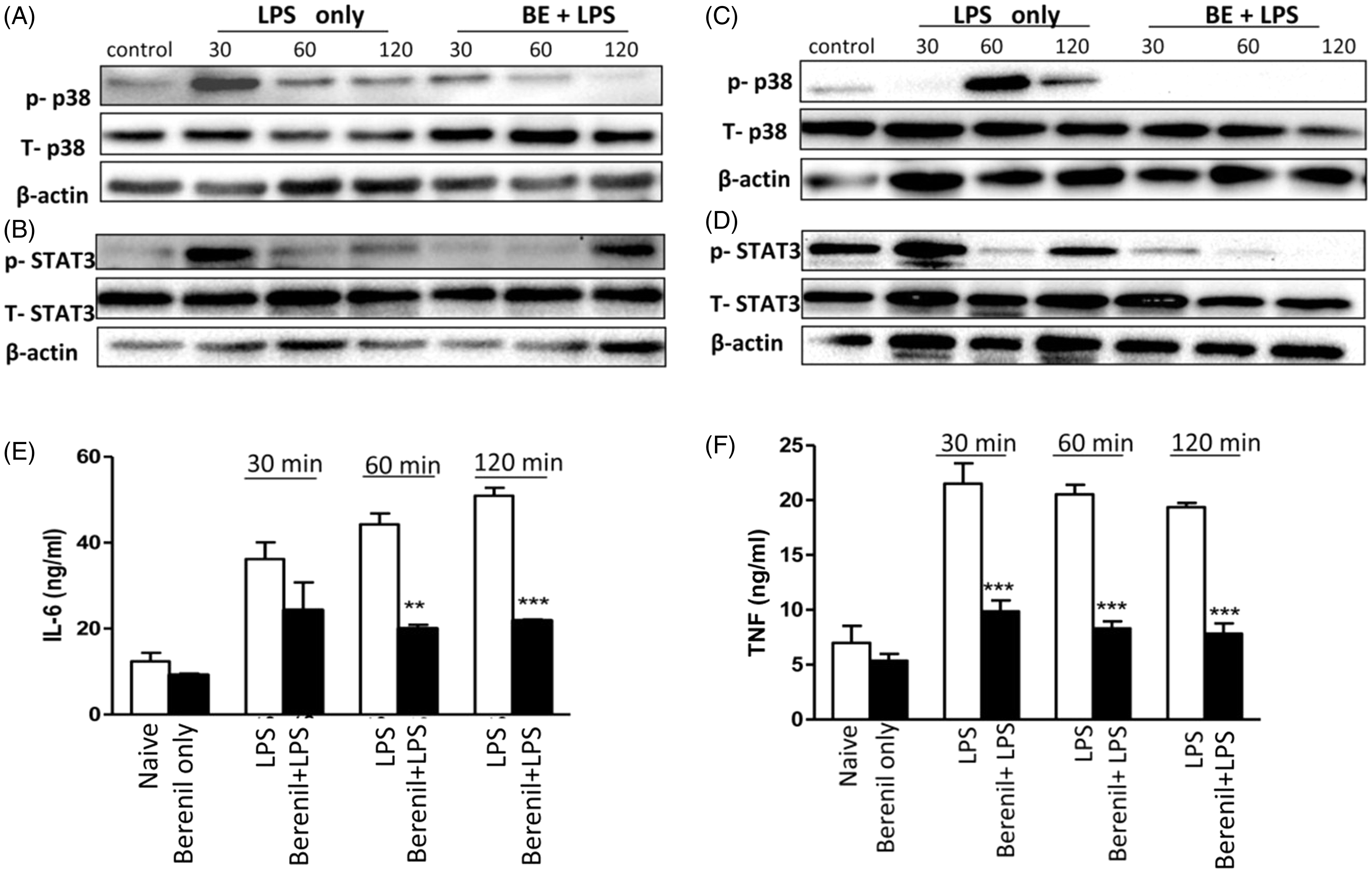
To further confirm the effect of Berenil on MAPK and STAT phosphorylation in a more relevant in vivo setting, we pre-treated mice with Berenil for 24 h, injected them with LPS (5 mg/kg) and, at different times, sacrificed them and assessed phosphorylation of MAPKs and STATs in the peritoneal macrophages directly ex vivo. Berenil treatment significantly down-regulated LPS-induced phosphorylation of p38 and STAT3 (Figure 6C, D). Consistent with this, Berenil treatment caused significant reduction in the levels of IL-6 and TNF in the peritoneal lavage fluids of LPS treated mice (Figure 6E, F). Collectively, these results further confirm that Berenil down-regulates LPS-induced production of pro-inflammatory cytokines by inhibiting phosphorylation of MAPKs and STATs in vitro and in vivo.
Berenil down-regulates LPS-induced phosphorylation of NF-кB p65 and IL-6 promoter activity
The NF-кB/Rel family of transcription factors play a crucial role in inflammation by their ability to induce transcription of pro-inflammatory cytokine genes. The most activated form of NF-кB is a heterodimer consisting of p65 subunit and p50 subunit as they have the transactivation domains necessary for gene induction.
39
In order to evaluate whether Berenil suppresses pro-inflammatory cytokine production by altering NF-кB activation and thus gene transcription, we performed Western blot and ELISA to detect NF-кB p65 phosphorylation. Pre-treatment of BMDMs with Berenil inhibited LPS-induced phosphorylation of NF-кB p65 subunit as assessed by Western blot (Figure 7A) and by ELISA (Figure 7B).
Berenil pre-treatment down-regulates LPS-induced phosphorylation of NF-кB p65 subunit and IL-6 gene promoter activity in macrophages. BMDM were treated with Berenil (10 µg/ml) overnight and then stimulated with LPS (1 µg/ml). At indicated times, the cells were lysed and the lysates were assessed by Western blot (A) or ELISA (B) for phosphorylation of NF-кB p65 subunit. The same blot was stripped and re-probed with Ab against total NF-кB and used as loading control (A). ANA-1 cells were transiently transfected with IL-6 promoter construct containing luciferase gene and then pre-treated with Berenil (10 µg/ml) for 12 h. Thereafter, the cells were stimulated with LPS (1 µg/ml) for another 12 h and the luciferase reporter activity was measured as described in the ‘Materials and methods’. Data are presented as fold increase over unstimulated controls are representative of three independent experiments with similar findings. *P < 0.05; **P < 0.01.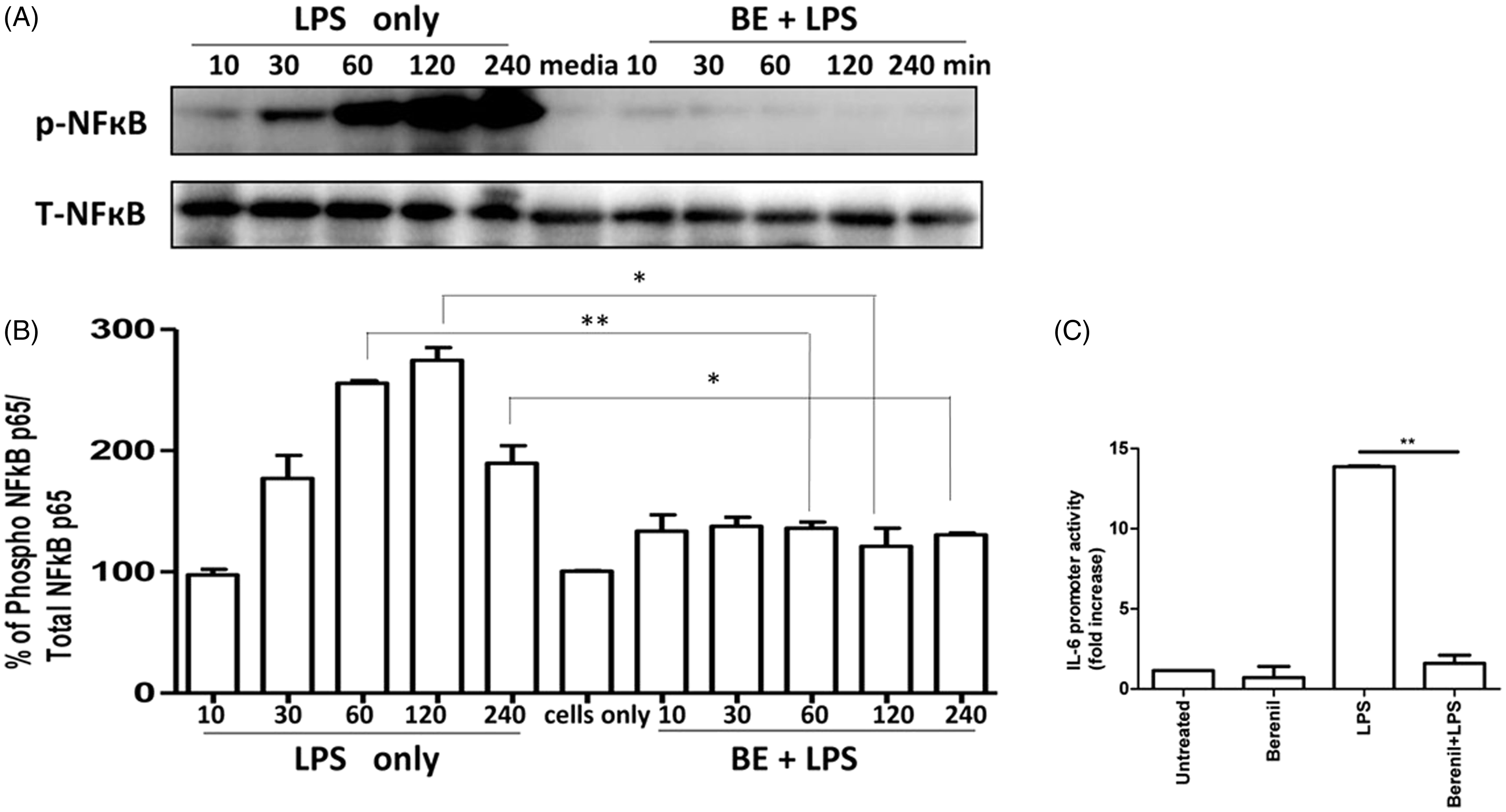
To further determine the mechanisms by which Berenil suppresses pro-inflammatory cytokine production, we investigated whether Berenil directly affects the promoter activity of the IL-6 gene. ANA cells were transiently transfected with wild type (WT) IL-6 luciferase reporter constructs, pre-treated overnight with Berenil and stimulated with LPS. ANA cells transfected with IL-6 promoter construct showed a significant increase (P < 0.01) in luciferase activity in response to LPS (1 µg/ml) stimulation. In contrast, pre-treatment with Berenil dramatically down-regulated LPS-induced promoter activity (Figure 7C). Collectively these data suggest that Berenil directly affects transcriptional mechanisms in macrophages leading to down-regulation of pro-inflammatory cytokine production.
Pre-treatment with Berenil up-regulates SOCS1 and SOCS3 expression in macrophages
The SOCS are very important regulators of the immune system and have been shown to regulate activation and cytokine production by T-cells, macrophages and DC. Forced expression of SOCS3 in a mouse arthritis model suppresses pro-inflammatory cytokine production and the induction or the development of the disease.
40
In addition, SOCS1 has been reported to block the initial steps involved in TLR signaling in macrophages.
41
Therefore, we investigated the possibility that Berenil might be suppressing pro-inflammatory cytokine production by up-regulating SOCS expression. As shown in Figure 8A–D, pre-treatment of BMDMs caused increased expression of SOCS1 and SOCS3 starting as early as 30 min and lasting up to 3 h. These results suggest that the inhibitory effects of Berenil on MAPK and STAT phosphorylation and the suppression of pro-inflammatory cytokine production may be owing to its effect on SOCS induction.
Berenil induces SOCS1 and SOCS3 expression in macrophages. BMDM were treated with Berenil (10 µg/ml) and at designated time points the cells were lysed and SOCS1 and SOCS3 expression was assessed by immunoblotting of total cell lysates using anti-SOCS1 and SOCS3 Abs. The same blot was stripped and re-probed with Ab against β-actin as loading control. The data presented are representative of two independent experiments with similar findings.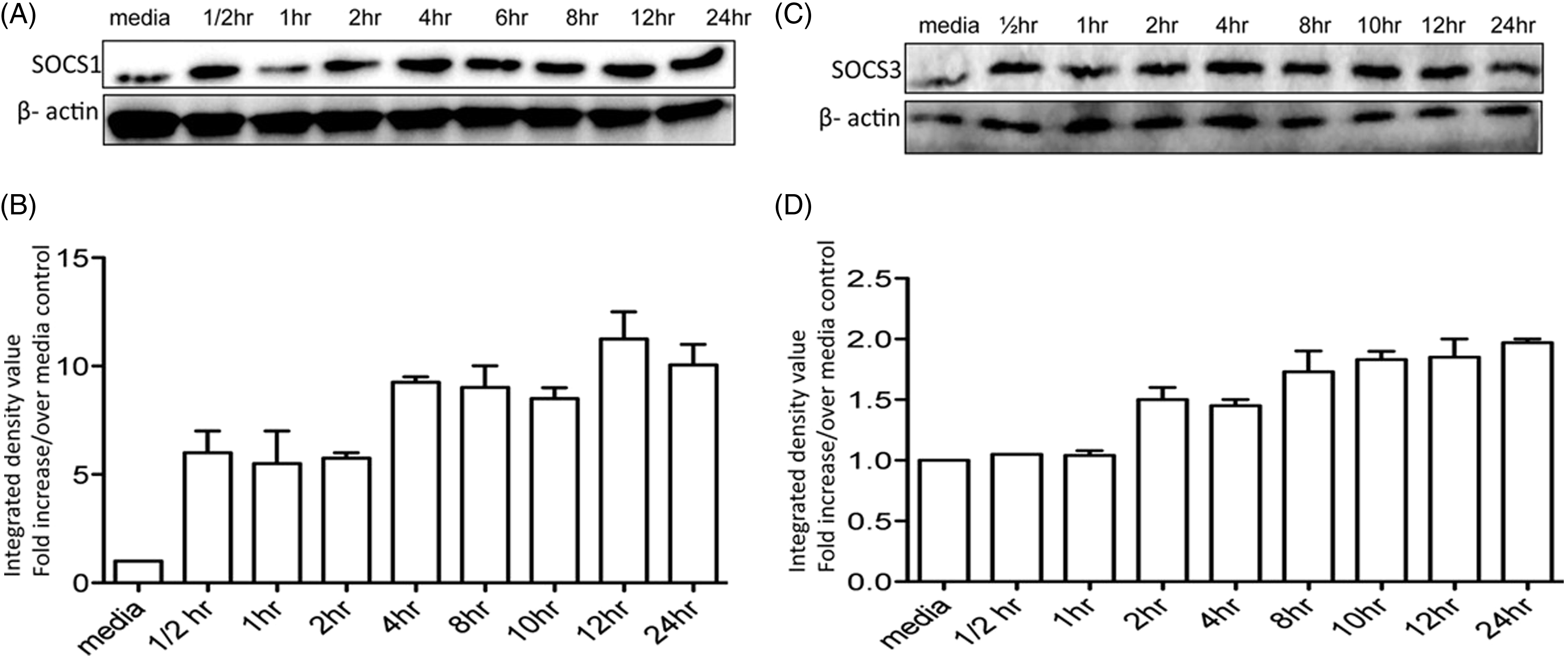
Discussion
We previously showed that Berenil treatment dramatically suppressed the production of pro-inflammatory cytokines (IL-6, IL-12p40 and TNF) by splenic and liver macrophages leading to a concomitant reduction in serum levels of these cytokines following T. congolense infection or LPS challenge. Therefore, the primary aim of the present study was to investigate the molecular mechanisms through which Berenil alters pro-inflammatory cytokine production by macrophages. Using more controlled in vitro conditions, we showed that pre-treatment with Berenil down-regulated LPS-, CpG- and poly (I:C)-induced cytokine production in macrophages and DC. Because these PAMPs are recognized by innate immune cells via TLRs, we hypothesized that Berenil down-regulates TLR expression on macrophages. Surprisingly, Berenil did not alter the expression of any of the TLRs (TLR2, TLR4 and TLR9) on macrophages. Hence, we considered the possibility that Berenil interferes with signaling molecules and transcription factors that mediate the expression of pro-inflammatory cytokines. Indeed, we found that Berenil down-regulates the phosphorylation of MAPKs (ERK, p38, JNK), STATs (STAT1 and STAT3) and NF-кB p65 subunit, key signaling molecules and transcription factors involved in the production of pro-inflammatory cytokines by immune cells. In addition, we found that Berenil down-regulated IL-6 promoter activity, indicating that the compound also directly affects transcription of pro-inflammatory genes. Similar to the in vitro effects, we showed that Berenil also inhibits MAPK and STAT phosphorylation in vivo. Interestingly, we found that Berenil up-regulated SOCS1 and SOCS3 expression, molecules that act as negative regulators of cytokine signaling by inhibiting MAPKs and STATs.
The hyper-activation of macrophages and a concomitant overproduction of pro-inflammatory cytokines has been proposed as the leading cause of acute death in T. congolense-infected highly susceptible BALB/c mice. 42 T. congolense-infected BALB/c have significantly higher serum levels of IL-6, IL-12p40 and TNF than infected, relatively resistant C57BL/6 mice. 17 The overproduction of pro-inflammatory cytokines in infected BALB/c mice leads to systemic inflammatory response syndrome-like phenomenon leading to death. We have found that treatment of T. congolense-infected mice with Berenil clears parasitemia and dramatically lowers serum levels of pro-inflammatory cytokines, including IL-6, IL-12p40 and TNF. 20 The dampening effect on pro-inflammatory cytokine production was mediated by the effect of Berenil on CD11b+ cells, including splenic and liver macrophages. 20 Furthermore, pre-treatment of mice with Berenil significantly ameliorated LPS-induced sepsis and pro-inflammatory cytokine production in vivo. 20 In the present study, we found that Berenil down-regulated LPS-, CpG-, and Poly (I:C)-induced cytokine production, strongly suggesting that the effect of the drug is global and is not pathogen-specific.
Macrophages are key innate immune cells that contribute to efficient destruction of intracellular pathogens via the production of free radicals and other potent cytotoxic molecules, including NO. In addition, they also act to shape the overall immune response through the production of inflammatory and immunomodulatory cytokines. Once pathogens are recognized by macrophages, they are taken up by phagocytosis, which initiates a cascade of intracellular events that may lead to the production of pro-inflammatory cytokines. Various PRR (including TLRs), are constitutively expressed by macrophages and play important roles in the recognition of specific microbial components. 43 In response to various stimuli, the TLRs are activated and this initiates the activation of powerful intracellular signaling pathways through their cytoplasmic Toll/interleukin-1 receptor (TIR) domains.44,45 Once the TLR signaling is initiated, several adaptor protein families are recruited in a finely coordinated manner, which, in turn, activates the downstream protein kinases ultimately leading to the activation of key transcription factors such as NF-кB or activation protein 1 (AP-1). 46 These transcription factors then enter the nucleus and bind to their specific binding sites on the gene promoters leading to transcription of pro-inflammatory genes. We found that despite suppressing LPS-, CpG- and poly (I:C)-induced production of pro-inflammatory cytokines, Berenil did not alter the expression TLR4, TLR9 and TLR2 on macrophages. In addition, it did not alter MD2 and CD14 expression on macrophages. MD2 is intimately associated at the cell surface with TLR4, 47 and plays a critical role in recognition and regulation of cell activation by LPS. 48 Similarly, CD14 is a key component of LPS recognition because it binds and transfers LPS to LPS binding protein, an important step that is crucial for LPS interaction and activation of TLR signaling events. Thus, our results show that the down-regulation of pro-inflammatory cytokine production by Berenil is not due to its alteration of TLR expression on macrophages.
In response to inflammatory stimuli or following ligation of TLRs by their specific agonists, a series of intracellular signaling cascades are initiated that ultimately lead to activation of macrophages and their release of pro-inflammatory cytokines. Prominent among these intracellular signaling events include the phosphorylation of MAPK and STAT pathways leading to the activation of NF-кB, 49 a crucial transcription factor for pro-inflammatory cytokines production. Indeed, LPS-induced production of inflammatory cytokines by macrophages involves activation of MAPK.50,51 In addition to activating the MAPK pathway, binding of LPS to TLR4 also activates the JAK–STAT signal transduction pathways, particularly STAT1.52–54 Several studies indicate that the anti-inflammatory effect of various naturally occurring, as well as synthetic, compounds are due to their effect on ERK, JNK, p38 and STAT phosphorylation. For example, silibinin treatment inhibits inducible nitric oxide synthase expression in tumor cells and TNF-induced production of pro-inflammatory cytokines in lung epithelial carcinoma cells by inhibiting phosphorylation of STAT1, STAT3 and ERK1/2 leading to a concomitant suppression of NF-κB and AP-1 activation. 55 Another study indicates that curcumin suppresses the JAK–STAT pathway and thus attenuates inflammatory responses of brain microglial cells. 56 Furthermore, it has been reported that estradiol down-regulates LPS-induced cytokine production in macrophages and this effect was mediated via alteration of intracellular signaling pathways that results in impaired NF-кB binding activity. 57 In line with these, our studies show that Berenil significantly impairs the phosphorylation of ERK, p38, JNK, STAT1, STAT3 and NF-кB, suggesting that it inhibits MAPKs and JAK–STAT signaling pathways. Such a wide range of inhibitory targets could explain the global effects of this compound in suppressing pro-inflammatory cytokines, including those associated with CpG and Poly (I:C) stimulation. Interestingly, we found that when macrophages were pre-treated with Berenil overnight and stimulated with LPS (1 µg/ml) for only 1–2 h, there was a down-regulation of STAT1 and STAT3 phosphorylation without a concomitant down-regulation of pro-inflammatory cytokine production. The down-regulation of pro-inflammatory cytokine production was observed only after prolonged (>12 h) LPS stimulation, strongly suggesting that the reduced phosphorylation of STAT1 and STA3 is not likely due to reduction in cytokine production, but more likely related to the direct effect of Berenil on the cells. In contrast, we found that Berenil pre-treatment up-regulated STAT5 phosphorylation in macrophages. Previous studies have shown that GM-CSF and IL-15 up-regulate STAT5 phosphorylation in several cell types.58–60 It is conceivable that the increased STAT5 phosphorylation following Berenil pre-treatment may be indirect, due, in part, to increased production of IL-15 and GM-CSF by macrophages.
How does Berenil inhibit MAPKs and STATs leading to suppression of pro-inflammatory cytokine production in macrophages? TLR signaling must be tightly regulated to maintain a balance between activation and inhibition of the immune system in order to avoid inappropriate inflammatory responses. Following the interaction of TLRs with their respective ligands, the ensuing intracellular signaling events are tightly controlled by intracellular regulators, including SOCS proteins, TRIAD3A and A20, TOLLIP, etc. 61 In particular, SOCS proteins have been shown to inhibit many intracellular signaling pathways, including those mediated by MAPKs and STATs. 62 SOCS proteins inhibit signal transduction by acting in a negative feedback loop to dampen excessive activation. 63 Indeed, it has been shown that SOCS proteins inhibit LPS-induced IL-6 production by regulating the JAK-STAT pathway. 64 Another study showed that SOCS3 inhibits TNF-induced MAPK phosphorylation in rat islets. 65 We found that Berenil pre-treatment up-regulates SOCS1 and SOCS3 expression in macrophages. We speculate that this up-regulation of SOCS proteins by Berenil has a negative effect on STAT and MAPK signaling pathways, partially contributing to the down-regulation of pro-inflammatory cytokine production in macrophages. In addition to up-regulating SOCS1 and SOCS3, we also found that Berenil significantly inhibits LPS-induced NF-кB and IL-6 gene promoter activity, suggesting that, in addition to its effects on MAPKs and STATs, Berenil also directly affects transcriptional activities in macrophages. It is also conceivable that Berenil might, in addition to up-regulating SOCS, affect the activities of other negative regulators in macrophages.
In summary, our results demonstrate that Berenil modulates a pro-inflammatory immune response by inhibiting key signaling pathways and transcription factors associated with inflammatory cytokine production. Thus, our studies point to a novel effect of Berenil. The suppression of pro-inflammatory cytokines production in macrophages by Berenil in vitro and in vivo suggests that this compound might have anti-inflammatory properties and could therefore be used for the treatment and control of chronic inflammatory diseases such as rheumatoid arthritis, inflammatory bowel disease, artherosclerosis and sepsis. Unveiling the molecular mechanisms of action of Berenil could result in ways to increase the efficacy of the drug and prevent its unwanted side effects.
Footnotes
Acknowledgements
We thank members of the parasite vaccine laboratory and Vidyanand Anaparti for providing some technical assistance. We also thank Dr. Sandrine Lafarge for help with NF-кB ELISA.
Funding
Funding for this study was provided by the Natural Sciences and Engineering Research Council (NSERC) of Canada and the Manitoba Health Research Council (MHRC). Shiby Kuriakose is supported by the Manitoba Health Research Council Studentship Award.
Conflicts of interest
The authors do not have any potential conflicts of interest to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
