Abstract
The immunomodulatory activity of Leonurus cardiaca L. polyphenol-rich extract (LCE) was tested in vitro on HUVECs to explore its potential therapeutic usefulness in the treatment of inflammatory lesions. The phytochemical composition of LCE, its antioxidant and cytotoxic activity, and the influence of LCE on NO and platelet-activating factor (PAF) secretion by HUVECs and platelet aggregation were all assessed. Total polyphenol contents in LCE reached 137.0 ± 0.8 mg/g, with hydroxycinnamic acid derivatives as the predominant phenolic compounds. LCE expressed antioxidant capacity, which was, however, 13- to 16-fold lower than the antioxidant activity of ascorbic acid. The plant extract was not cytotoxic up to a concentration 4500 µg/ml and did not exhibit proapoptotic activity. LCE significantly increased NO production in HUVECs in a concentration-dependent manner and led to the inhibition of PAF secretion induced by staphylococcal peptidoglycan. The extract used at the concentration of 100 µg/ml significantly reduced platelet aggregation in the presence of arachidonic acid. We provide in vitro data demonstrating the immunomodulatory potential of LCE, which may be beneficial in preventing the development of difficult-to-treat inflammatory lesions within chronically infected tissues.
Introduction
The most prominent physiological effects of soft tissue damage (either infectious or not) include changes in the endothelium, platelets and immunocompetent cells. The non-thrombogenic endothelial surface switches to a pro-coagulant and pro-inflammatory phenotype, leukocyte–endothelial interactions intensify, leading to diapedesis and chemotaxis, and powerful vasoactive mediators [e.g. NO and platelet-activating factor (PAF)] are released.1–5 These activities are intended to restore homeostasis in the body, which depends on the relevant repair processes in damaged tissues. Therefore, the reasons for the development of many chronic lesions, such as infective endocarditis (IE), diabetic foot ulcerations (DFU) or chronic wound infections (CWI), include both microbial infection and pathological changes within the host tissue, which disturb these physiological repair processes.6–9 Thus, thinking about new therapeutic strategies, the multidirectional activity of potential drugs should be considered to achieve a combined favourable effect. In this context, plant secondary metabolites (e.g. polyphenols, essential oils and saponins) seem to be promising as novel medicaments supporting classic therapy. Their wide, direct antimicrobial activity (against bacteria, fungi, viruses and parasites) and diverse biological effects to host eukaryotic cells (e.g. antioxidant, antimutagenic, anticancer, antiallergic, antiatherosclerotic, surface-active anaesthetic, antioedematous, proliferative and immunomodulatory) have often been reported.10–17
The subject of our research was a polyphenol-rich extract from Leonurus cardiaca L. (LCE), which is considered as a supplementary therapeutic product in chronic soft tissues infections. Studies in murine macrophages demonstrated that flavonoids, including flavonols (quercetin, kaempferol) and flavones (apigenin), affect the activity of the enzymes involved in the synthesis of inflammatory mediators, such as NO, prostanoids and leukotrienes. Apigenin, galangin, kaempferol and quercetin reduced the expression of ICAM-1, vascular cell adhesion molecule 1 (VCAM-1) or E-selectin on the surface of endothelial cells, reducing diapedesis of phagocytes. Resveratrol and procyanidins intensified lymphocyte apoptosis.18,19 However, some polyphenols, such as those present in the extract from Cassia auriculata, stimulate the immune system by increasing the percentage of T cells and B cells in the total number of immunocompetent cells or enhancing the proliferation of splenocytes (studies on a rat model). 20 Understanding the mechanisms of activity for novel medicaments is necessary prior to use in practice. Moreover, the pathophysiological situation in damaged host tissues is typically complex and requires prudent action. For example, exposure of extracellular matrix protein neoepitopes, deposition of platelets and fibrin, and up-regulated expression of various molecules in endothelial cells and leukocytes (e.g. ICAM-1, VCAM-1, selectins, tissue factor) lead not only to the development of a proper local inflammatory process, but also to the creation of the surface suitable for the attachment of the pathogens.5,8,9,21 Therefore, the main aim of this study was to assess the influence of LCE, a phytocompound with potential therapeutic utility, on the physiological properties and condition of endothelial cells, including their viability, apoptosis and secretory activity based on NO and PAF production, as well as platelet aggregation. These studies also included phytochemical analysis of LCE and the assessment of its antioxidant activity.
Materials and methods
LCE preparation
A motherwort (L. cardiaca L.) sample (50 g; Figure 1) was commercially available from KAWON - HURT Nowak Sp.j. (Gostyń, Poland), and the herbarium voucher is stored at the producer. Air-dried (60℃, 4 h) material was ground to a fine powder using a coffee mill (Moulinex type A505) and extracted with acetone-water (70:30, v/v) at a solid to liquid ratio 1:10 (w/v) under stirring for 30 min at 22℃. After centrifugation (2500 g, 15 min), the pellet was re-extracted twice with 70% aqueous acetone for 15 min. Then, the combined supernatants were evaporated at < 40℃ under reduced pressure (Rotavapor RII, Büchi, Switzerland) to remove acetone. The remaining water phase was defatted nine times with chloroform (1:1, v/v). Finally, the water fraction was concentrated in vacuum and freeze-dried (Alpha 1-2 LD plus; Martin Christ Gefrierttrocknungsanlagen GmbH, Osterode am Harz, Germany). The extraction yield was 15.97%.
Commercially available L. cardiaca L. herb sample used in the study.
Phytochemical analysis of LCE
The dry matter content of LCE was determined by drying at 105℃ to a constant mass. The differences in mass before and after incineration at 550℃ for 16 h enabled the assessment of the ash content. The total phenolic content was determined using a Folin–Ciocalteu reagent based on the method described by Bordonaba and Terry. 22 The results were expressed as gallic acid equivalents. The phenolic profile of LCE was analysed using an HPLC method. A analytical reversed-phase HPLC system (Waters, Milford, MA, USA) with an autosampler 2707 and a binary HPLC pump 1525 coupled to a 996 photodiode array detector (2998) controlled by the Waters Breeze 2 software was used as described previously. 16 According to Dyrby et al., 23 the binary mobile phase consisted of water and formic acid in the ratio of 90:10 (v/v) (solvent A), and water, acetonitrile and formic acid in the ratio of 49:50:10 (v/v/v) (solvent B). The separation of phenolics was performed using the following gradient programme with a flow rate of 1 ml/min: 0 min, 88% A + 12% B; 26 min, 70% A + 30% B; 40–48 min, 0% A + 100% B; and 48–50 min, 88% A + 12% B. Spectral identification and the maximum of UV-Vis absorption were the basis for phenolic qualification in three subclasses: hydroxybenzoic acids and flavanols (detection at 280 nm; expressed as mg of gallic acid equivalents/g of the extract), hydroxycinnamic acids (detection at 320 nm; expressed as mg of chlorogenic acid equivalents/g of the extract) and flavonols (detection at 360 nm; expressed as mg of quercetin galactoside equivalents/g of the extract).
Antioxidant activity of LCE
Antioxidant capacity of LCE was evaluated by three in vitro assays. The scavenging effect of stable ABTS•+ [2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid)] radicals was tested according to the procedure described by Re et al., 24 and DPPH• (2,2-diphenyl-1-picrylhydrazyl) free radical scavenging activity was determined using the method of Kim et al. 25 The ferric reducing antioxidant power (FRAP assay) was performed according to the protocol reported by Benzie and Strain. 26 Distilled water was used as a blank, and Trolox (water-soluble analogue of alpha-tocopherol) was used as a standard. For comparison to the LCE antioxidant activity, ascorbic acid was tested as a positive control. The results for all three methods were expressed as µM of Trolox equivalents per gram of the extract or ascorbic acid (Trolox-equivalent antioxidant capacity (TEAC). Two independent experiments were performed for each assay.
Cells
The HUVECs (Cascade Biologics, Portland, OR, USA) used in the study were cultured according the manufacturer’s instructions in Medium 200 (Gibco, Grand Island, NY, USA) supplemented with Low Serum Growth Supplement (LSGS; Gibco) at 37℃ in a humidified atmosphere of 5% CO2. Peripheral venous blood (as the source of the platelets) was freshly collected from volunteers with the consent of the Human Research Ethics Committee at the University of Lodz. Hirudin (at the final concentration 25 µg/ml) was used as an anticoagulant. This protein is the most potent natural inhibitor of thrombin that does not interfere with the biological activity of other serum proteins and is recommended by the manufacturer to test platelet functions.
Cytotoxicity of LCE
The cytotoxicity of LCE on HUVECs was tested using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction assay based on a previous description. 27 Briefly, HUVECs in the exponential growth phase were seeded (1 × 104/well) on 96-well plates (Nunc, Roskilde, Denmark) and cultured for 24 h at 37℃. Stock solution of LCE was freshly prepared in 50% DMSO (POCh, Poland, Gliwice), diluted with complete culture medium and added to the cells to obtain the final concentration range of 10–4500 µg/ml. The final concentration of DMSO did not exceed 0.5%, which was non-toxic for HUVECs. Untreated cells were used as a positive control (100% of cells viability). HUVECs were incubated with LCE for 1 h and 24 h under the same conditions. Next, 50 µl MTT (5 mg/ml in PBS) was added, and the incubation was continued for 2 h. After aspiration of the MTT-containing medium, 100 µl 20% SDS and 50% dimethylformamide at pH 4.7 was added to dissolve blue formazan crystals produced by the metabolically active cells. Then, the absorbance was read (λ = 550 nm) using the multifunctional plate reader Victor2 (Wallac, Turku, Finland) and compared with the positive control to calculate the percentage of viable cells. Two independent experiments with four replicates in each were performed.
Induction of cell apoptosis by LCE
HUVECs (2 × 106 cells/ml) were incubated for 24 h at 37℃ in 5% CO2 in a 75 cm2 cell culture flask (Nunc) in Medium 200 supplemented with LSGS. The cell monolayer was washed with PBS, and trypsin was used to recover HUVECs from the flask bottom and to obtain cell suspension at the density of 7.5 × 106 cells/ml. A stock solution of LCE was freshly prepared in 50% DMSO, diluted with complete culture medium and added (1:1) to 200 µl cell suspension (1.5 × 106 cells in an Eppendorf tube) to the final concentration range of 10–4500 µg/ml. After 24 h of co-incubation of HUVECs with LCE at 37℃, the cells were centrifuged (150 g, 10 min), and the culture medium was removed. Then, the cell pellet was lysed using a cell lysis buffer included in the commercial test (50 µl buffer/2 × 106 cells). The lysates were centrifuged and supernatants were collected and stored at –80℃ until testing. The activity of caspase-3 in HUVEC lysates was assessed on 96-well plates using Caspase-3 Colorimetric Assay (R&D Systems, Minneapolis, MN, USA), which is based on the colorimetric proteolytic reaction of a specific substrate (DEVD-pNA), as recommended by the manufacturer. Caspase-3 activity, which relates to cell apoptosis, was compared between untreated control cells (spontaneous apoptosis) and LCE-treated cells. Two independent experiments were performed, each in triplicate.
Measurement of NO production under the influence of LCE
HUVECs cultured in Medium 200 supplemented with LSGS were seeded on 96-well culture plates (3 × 104 cells/well) and incubated for 24 h at 37℃ in 5% CO2. A stock solution of LCE, which was freshly prepared as described above, was diluted in culture medium and added to the cells at the final concentrations of 50, 100 and 350 µg/ml. HUVECs in culture medium alone were used as control cells. The cells were treated with LCE for 4 and 24 h. After incubation, the culture medium was collected and stored at –80℃ until testing. The concentration of NO (nmol/ml) in the samples was assessed by measuring the levels of oxidized forms (nitrates and nitrites) using the Nitric Oxide Colorimetric Assay Kit (Abcam, Cambridge, MA, USA), as recommended by the manufacturer. Two independent experiments were performed.
Effect of LCE on PAF secretion
The cultures of HUVECs prepared on 96-well plates as described above were treated with media containing LCE at the final concentrations of 50, 100 and 350 µg/ml and peptidoglycan from Staphylococcus aureus (Sigma-Aldrich, Fluka, Buchs, Germany) at the final concentration of 10 µg/ml for 4 and 24 h. HUVECs stimulated by S. aureus peptidoglycan alone were used as control cells. After incubation, the culture medium was collected and stored at –80℃ until testing. The concentration of PAF (pg/ml) in the samples was assessed using the ELISA Kit for Platelet Activating Factor (Uscn Life Science Inc., Houston, TX, USA) as recommended by the manufacturer. Two independent experiments (one in duplicate, one in triplicate) were performed.
Aggregation of platelets in the presence of LCE
Blood was obtained from healthy donors (11 individuals: five men and six women; mean age: 35.1 ± 10.7 yr). Hirudin was used as a safe anticoagulant. The studies were approved by the Human Research Ethics Committee at the University of Lodz. None of the donors had taken aspirin or other drugs affecting platelet function for at least 10 d prior to blood collection. A stock solution of the plant extract was freshly prepared in 50% DMSO as described above, diluted and used at the final concentrations of 50, 100 and 350 µg/ml. The incubation of whole blood with LCE was performed at 37℃ for 15 min. Whole blood without the plant extract with the respective concentration of DMSO was used as the control. Prior to measurement, the platelets were activated with 6.4 µM ADP, 3.2 µg/ml collagen (type I native collagen fibrils from equine tendons suspended in isotonic Glc solution pH 2.7) or 0.5 mM arachidonic acid (all reagents from Dynabyte Medical, Munich, Germany). Platelet aggregation in whole blood was determined using a Multiplate–Multiple Platelet Function Analyzer (Roche, Basel, Switzerland) and a five-channel impedance aggregometer (multielectrode aggregometer). The measurements were performed according to the manufacturer’s protocol. The total area under the aggregation curve expressed in arbitrary units was recorded.
Statistical analysis
The results are provided as the means ± SD. For the analysis of the results of NO and PAF secretion, significant differences were evaluated using the Mann–Whitney U-test and the program STATISTICA 10.0 (Stat Soft Inc., Tulsa, OK, USA). P < 0.05 was considered significant. Statistical differences between extract-treated and control blood platelets in the assessment of aggregation were estimated using ANOVA with repeated measures and paired Student’s t-test with Bonferroni’s correction. P < 0.01 was considered significant.
Results
Phytochemical analysis and antioxidant activity of LCE
Phytochemical analysis of LCE with respect to phenolic contents.

Values are provided as mean ± SD, n ≥ 3. aDetermined by Folin–Ciocalteu reagent as gallic acid equivalents; bdetermined by HPLC at 280 nm as gallic acid equivalents; cdetermined by HPLC at 320 nm as chlorogenic acid equivalents; d determined by HPLC at 360 nm as quercetin galactoside equivalents.
The antioxidant activity of LCE was also assessed. Scavenging activity towards synthetic free radicals (ABTS•+ and DPPH•) and reducing power in the FRAP assay (Fe3+ reduction to the Fe2+) of LCE are presented in Figure 2 as TEAC (µM/g). The antioxidant capacities of LCE were in the range of 350 ± 20–455 ± 17 µM Trolox/g. To compare the antioxidant activity of the LCE with positive controls, ascorbic acid was also tested. TEAC values of this natural antioxidant were 4896 ± 168, 7180 ± 208 and 4793 ± 98 µM of Trolox/g for the ABTS, DPPH and FRAP methods, respectively.
Total antioxidant capacity of LCE determined by three in vitro methods. ABTS: the scavenging effects on stable ABTS•+ radicals; DPPH: the scavenging effects on stable DPPH• radicals; FRAP: ferric reducing antioxidant power (FRAP assay). The results for all three methods were expressed as µM Trolox equivalents per gram of LCE (TEAC). Two independent experiments for each method were performed with error bars denoting SD from the means.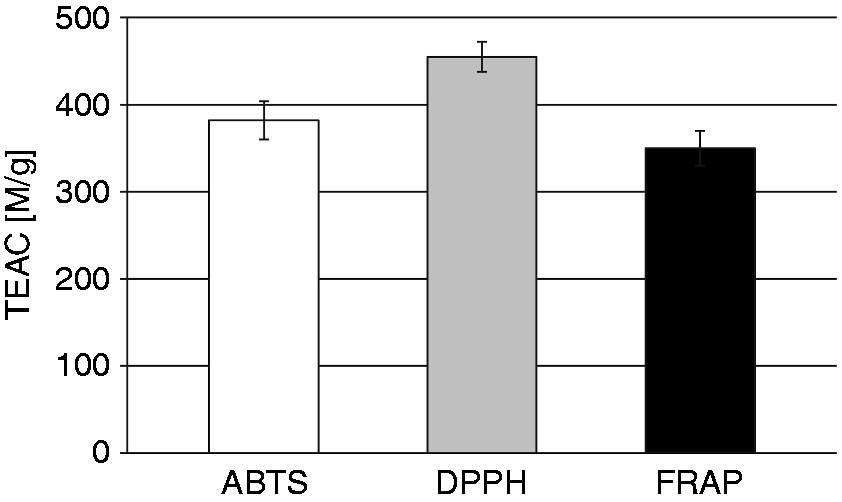
Cytotoxic and proapoptotic activity of LCE
The effect of LCE on HUVEC viability during 1 h and 24 h co-incubation was tested using an in vitro method involving the reduction of MTT by metabolically active cells. At all concentrations tested (10–4500 µg/ml), LCE did not reduce HUVEC viability compared with untreated control cells (Figure 3).
The effect of LCE on HUVEC viability tested by the MTT reduction assay. The percentage of viable HUVECs after 1 h and 24 h of co-incubation with LCE was calculated based on absorbance values compared with the viability of untreated (control) cells, which was set at 100%. Two independent experiments were performed with error bars denoting SD from the means.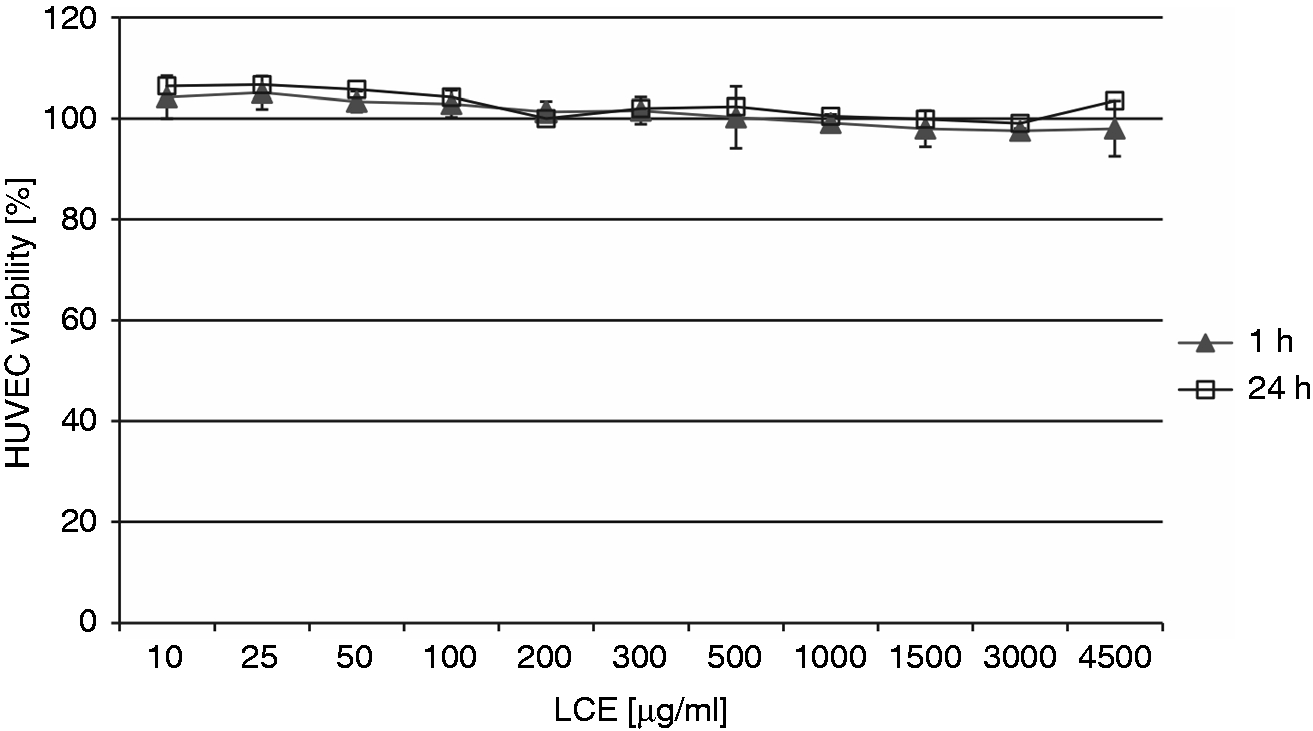
The cell viabilities were in the ranges of 98.74 ± 5.56%–111.53 ± 8.4% and 94.1 ± 5.97%–110.29 ± 9.41% after 1 h and 24 h co-incubation, respectively, with various concentrations of LCE. The non-cytotoxic properties of LCE were also confirmed by the assessment of HUVEC apoptosis based on caspase-3 activity. After 24 h, HUVECs exposed to LCE at 10, 100, 1000 and 4500 µg/ml had caspase-3 activity levels, indicating apoptosis, of 111.1 ± 6.8% (P = 1), 104.2 ± 3.6% (P = 1), 83.4 ± 3.6% (P = 0.4216) and 84.8 ± 2.2% (P = 0.7299), respectively, compared with the levels of caspase-3 activity in untreated (control) cells that underwent spontaneous apoptosis, which was considered as 100%. Thus, LCE did not induce apoptosis in these cells over the range of concentrations applied. Moreover, the extract used at higher concentrations (1000 and 4500 µg/ml) seemed to inhibit spontaneous apoptosis of HUVECs.
LCE impact on NO production by HUVECs
NO secretion by HUVECs subject to LCE.

The results are presented as the mean NO concentration (in nmol/ml ± SD) secreted by HUVECs in the presence or absence (control) of LCE used at 50, 100 and 350 µg/ml after 4 h and 24 h incubation. Two independent sets of experiments were prepared, each in duplicate. Statistical differences between extract-treated and control HUVECs were estimated with the Mann-Whitney U-test. *Significant differences, P < 0.05.
HUVECs exposed to LCE for 4 h and 24 h produced a significantly higher level of NO (P = 0.03) compared with control cells. NO secretion reflected a dose-dependent but not a time-dependent trend, with the strongest stimulation observed at 350 µg/ml LCE.
PAF secretion under the influence of LCE
To determine the effect of LCE on the production of PAF by HUVECs in the presence of S. aureus peptidoglycan as a PAF secretion stimulator, an ELISA Kit for PAF was used. The obtained results are presented in Figure 4.
The effect of 50, 100, and 350 µg/ml LCE on the production of PAF by HUVECs stimulated with S. aureus peptidoglycan after 4 and 24 h of co-incubation. Two independent sets of experiments were prepared with error bars denoting SD from the means. Statistical differences between extract-treated and control HUVECs were estimated with the Mann–Whitney U-test. *Significant differences, P < 0.05.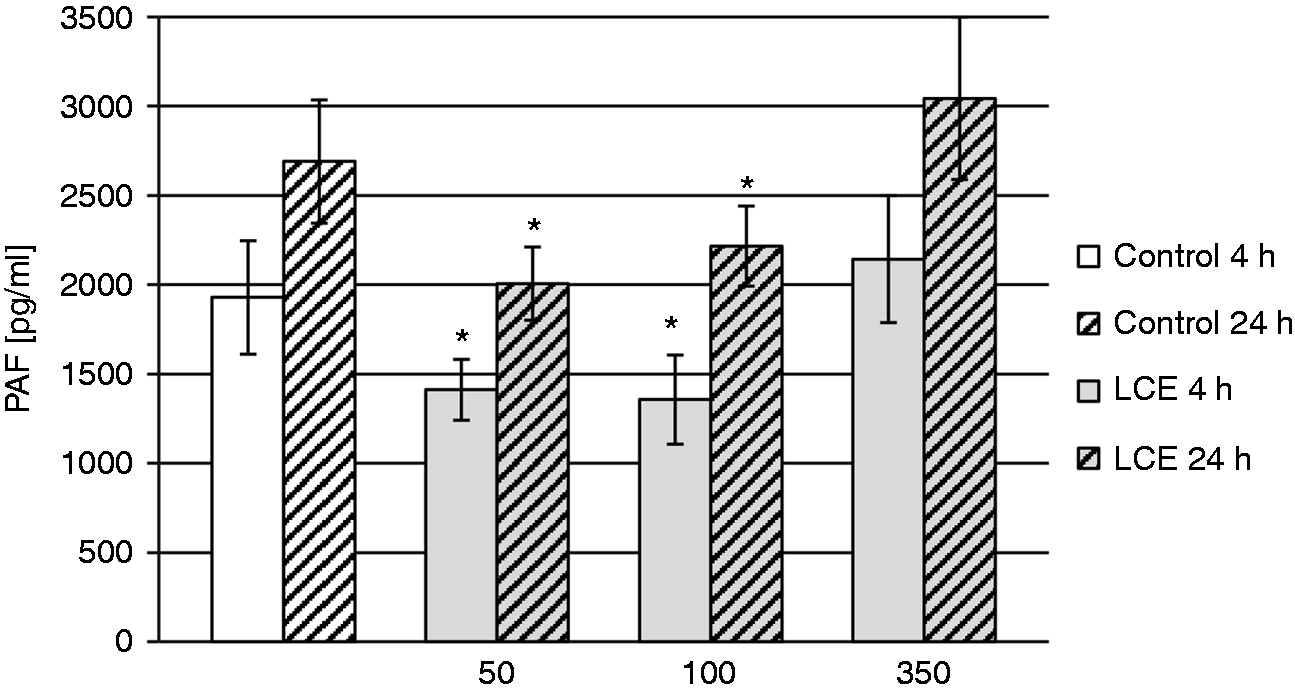
A significant reduction was found in PAF secretion by the cells treated with 50 and 100 µg/ml LCE for 4 h and 24 h compared with control (untreated) cells. The PAF concentration produced by HUVECs was 1.36-fold (P = 0.0084) and 1.4-fold (P = 0.012) lower after 4 h treatment with 50 and 100 µg/ml LCE, respectively, and 1.34-fold (P = 0.0059) and 1.2-fold (P = 0.0235) lower after 24 h treatment with 50 and 100 µg/ml LCE, respectively. No effect was observed for 350 µg/ml LCE.
LCE effect on human blood platelet aggregation
Effect of LCE on human blood platelet aggregation measured in whole blood.
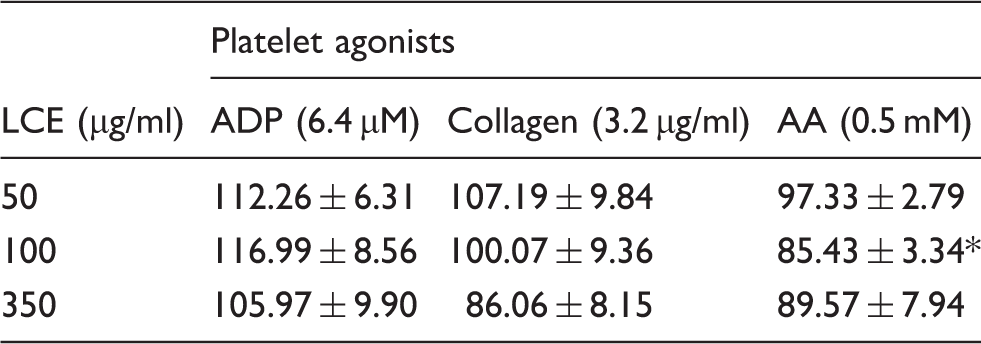
The results are presented as a percentage of control platelet aggregation, and expressed as the mean % ± SD (n = 11). Statistical differences between extract-treated and control blood platelets (without extract) were estimated with ANOVA with repeated measures and paired Student’s t-test with Bonferroni’s correction. *Significant differences, P < 0.01.
Discussion
Both antimicrobial and health-promoting (including immunomodulatory) properties of natural-origin polyphenols could be useful in supporting therapy of numerous chronic, difficult-to-treat diseases. These compounds are an important component of our diet and have existed in folk medicine for thousands of years.10,13,18,28,29 One of their known sources is L. cardiaca L., which is popularly referred to as motherwort, belonging to the mint family Lamiaceae. It is a perennial herb commonly found in rural areas in Europe, central and eastern Asia, northern Africa and North America. Since ancient times, motherwort has been used to treat heart conditions, such as palpitation and tachycardia associated with stress, depression or hyperthyroidism, as an agent stabilizing heartbeat and temporarily decreasing blood pressure. According to traditional Chinese medicine, L. cardiaca herb promotes the recovery of the uterus after childbirth. Motherwort application in digestive disorders, bronchial asthma, climacteric symptoms, menstruation irregularities, or wounds and skin inflammations has also been reported.30,31 Nevertheless, only sedative and hypotensive activities of motherwort (probably due to the presence of such alkaloids as leonurine and stachydrine, which mildly dilate blood vessels and have a relaxing effect on smooth muscles) have been confirmed in clinical studies, and there is no real evidence to support the use of this herb in other cases. Therefore, for the first time, we proposed the assessment of the biological activity of L. cardiaca polyphenolic extract in the context of its potential suitability for supporting the treatment of tissue dysfunctions accompanying infections, such as IE, DFU or CWI. To date, very little is known about the effects of LCE on the endothelium, blood platelets or other host cells involved in tissue repair processes and immunity, so we wanted to fill this gap.
LCE tested in the present study was obtained from dried herb using an acetone/chloroform extraction with a yield of 15.97%, which was reduced compared with the yield of the extraction with 80% methanol, which reached 23.2–31.8%.32,33 The differences may be due mainly to the transition of some components into the chloroform phase and the diversity of plant material. However, the total polyphenols content in LCE (137.0 ± 0.8 mg/g) was similar to that reported by Matkowski et al. at 150.1 ± 8.9 mg/g, 32 and twice as high as the content obtained by Jafari et al. for total extract, 62.85 ± 3.7 mg/g. 33 Thus, regardless of the method and the yield of the extraction, the total content of phenolic compounds in LCE seems to be quite stable. The HPLC analysis of LCE showed that hydroxycinnamic acid derivatives were predominant phenolic compounds. This group of phenolics includes chlorogenic, caffeic, p-coumaric and ferulic acids, as well as caffeic acid ester and glycoside. 31 However, it is worth noting that many factors, such as growth conditions of the plants or the methods of extraction and analysis, can cause differences in the quantitative and qualitative composition of the extract.
According to the literature data, polyphenols (within a specified range of concentrations) possess strong antioxidant properties.32–34 The ability to neutralize and inhibit the production of ROS, such as free radicals, could be beneficial within inflamed tissues by preventing cellular damage induced by oxidative stress. Therefore, in the present study, we assessed the antioxidant activity of LCE using ABTS and DPPH methods, which are based on the scavenging of synthetic radicals (ABTS•+ or DPPH•), and the FRAP assay (Fe3+ reduction to the Fe2+). The antioxidant capacity of LCE was 13- to 16-fold lower than the antioxidant activity of pure ascorbic acid. These differences may be due to the low concentration (13.7%) of phenolic antioxidants and the presence of non-active components in the extract. However, it is worth noting that we consider the natural food product used as a dietary supplement and not a chemical compound. Moreover, antioxidant activity seems to be a constant and quite stable feature of LCE as similar results were reported by Yi and Wetzstein, 35 where TEAC values varied from 200 to 400 μM/g of motherwort extract, depending on the extraction and drying conditions of the herb. Matkowski and Piotrowska also demonstrated that the methanolic extract from L. cardiaca possesses scavenging potential toward DPPH• radicals, 34 and the reducing power was tested by phosphomolybdenum complex formation and lipid peroxidation inhibiting activity.
The spectrum and potency of the antimicrobial activity of plant natural products (including polyphenols) often depends on the range of concentrations used leading to multidirectional changes, such as an increase in the cell wall permeability followed by the leak of intracellular constituents, impairment of microbial enzyme systems or intracellular accumulation of reactive oxygen species.10–13,16 Similar cytotoxic effects can also be observed in eukaryotic cells. In an in vitro model, polyphenolic compounds from apple, strawberry and blackcurrant extracts were incorporated into the hydrophilic area of the erythrocyte membranes, resulting in the modification of their properties. As the concentrations of the extracts used were quite low (10–60 µg/ml), the observed effects were beneficial (e.g. an increase in the membrane osmotic resistance) without the induction of haemolysis. 36 However, assuming the medical use of the plant extracts (above all the direct application on host tissues) and being aware of their interaction with eukaryotic cell membranes, it is necessary to test the cytotoxicity of such preparations. 37 We assessed the effect of LCE on HUVECs during short (1 h) and long (24 h) co-incubation using the MTT reduction method. LCE at a wide range of concentrations tested (10–4500 µg/ml) did not reduce HUVEC viability compared with untreated control cells. The non-cytotoxic properties of LCE were also confirmed through an inability to induce apoptosis in HUVECs. However, some literature data point to natural and commercial polyphenols as the compounds supporting anticancer therapy by inducing programmed cell death. For example, green tea polyphenols (e.g. epigallocatechin-3-gallate), curcumin (a spice derived from Curcuma longa) and resveratrol found in red wine or grapes all inhibit the proliferation and metastasis of numerous types of cancer cells (e.g. lung, prostate, breast, leukaemia, colon).38,39 Of note, proapoptotic activity is beneficial only in the case of certain pathological changes and should be applied selectively with respect to chosen target cells. Considering chronic inflammatory lesions complicated by infections, induced death of normal cells, such as epithelial or endothelial cells, could even delay repair processes within infected tissues. Thus, the non-cytotoxic and anti-apoptotic activity of LCE on HUVECs demonstrated in our study enabled us to consider this extract as harmless for normal eukaryotic cells. It can be assumed that LCE can be safely used not only as orally administered dietary supplements, which minimizes its bioavailability, but also in other forms of medicaments (e.g. as a component of ointments) dispensed directly on host tissues.
NO, which is a major effector molecule included among the chemically simplest hormones, neurotransmitters and intracellular messengers, is produced by many types of cells, but its secretion by the endothelium seems to be particularly important given the regulatory function. Through guanylate cyclase activation, NO leads to the relaxation of smooth muscles in vessels that regulate blood flow. NO inhibits blood platelet activation and aggregation, indicating its significant role in limiting the development of some pathological changes, such as IE.40–42 IE is one the most dangerous, often fatal, diseases related to microbial infection (predominantly streptococcal and staphylococcal) of the endocardium or native/artificial valves. Both microorganisms and processes occurring in the host tissues (inflammation and coagulation) are responsible for pathological changes. Excessive platelet activation by bacterial products leads to the development of inflammatory lesions (vegetations) associated with the deposition of platelet aggregates, fibrin, blood leukocytes, erythrocytes and bacteria, which are extremely difficult to treat.21,43–45 Similar lesions can occur within other chronically infected tissues during diabetic foot ulcerations or chronic wound infections.14,46,47 In such cases, the overproduction of NO could be beneficial. NO produced in very high concentrations via iNOS activity participates in the immune response to infection and exerts mainly anti-inflammatory effects. NO inhibits leukocyte adhesion to the vascular endothelium and their migration, T-cell proliferation and NF-κB activation, while NO also exhibits antioxidant activity by interrupting lipid peroxidation.42,48,49 However, NO participates in oxidative-dependent intracellular killing of microorganisms by phagocytes. The reaction between the free radicals NO and superoxide (O2–) leads to the formation of other toxic compounds, such as peroxynitrite, and also neutralizes NO immunosuppressive activity.42,49 Thus, controlling the production of NO could be a potential weapon against some pathological processes. To the best of our knowledge, no study has assessed the effect of LCE on NO production to date. The present study demonstrated that exposure of HUVECs to LCE significantly increased the production of NO in a dose-dependent manner. Similar results on the positive regulation of endothelial NO synthase expression and subsequent NO synthesis by an alcohol-free red wine polyphenol extract were shown by Leikert et al. 50 The stimulatory effect of LCE on NO production by HUVECs could be useful in medical practice to limit immune- and infection-mediated tissue damage. The protective role of NO was previously demonstrated in mouse models of nephritis, gut inflammation, thermal injury and graft versus host disease. 42
PAF is another potent signalling mediator acting at very low concentrations, which causes platelet activation and aggregation followed by inflammation and allergic response. This natural phospholipid that is formed primarily through lipid re-modelling by phospholipase A2 and lyso-phosphatidylcholine acetyltransferase is derived not only from stimulated leukocytes, but also from endothelial cells and platelets. PAF is produced continuously at low levels and moves within and between cells playing a messenger function by binding to a specific G protein-coupled receptor for PAF on target cells. Under some stimuli, for example those released during microbial infections, PAF secretion increases rapidly leading to inflammation, which is often excessive and harmful in the course of sepsis, IE or DFU.43,51 Numerous studies have demonstrated that tumours and cancer cell lines also synthesize and secrete PAF, indicating an important role of this phospholipid in cancer cell growth and motility in at least some types of neoplastic changes.52–54 For the first time, we present the results of a study on the impact of LCE on PAF production. LCE significantly reduced PAF secretion by HUVECs stimulated with S. aureus peptidoglycan. The effect was exclusively observed for lower concentrations of LCE (50 and 100 µg/ml) and was time independent. These results confirmed the anti-inflammatory and indirect antiplatelet properties of LCE applied in strictly defined concentrations. The inhibition of PAF secretion is believed to be a possible mechanism of such activity. Given that PAF is a potent inducer of platelet activation and thrombosis in vivo with the concentration growing rapidly during microbial infections, the inhibitory effect of motherwort could be beneficial in patients with acute ischaemic stroke, IE, and valvular and heart transplants.
In this study, the direct effect of LCE on human blood platelet aggregation was also investigated for the first time. The applied impedance technique of measuring aggregation in whole blood allows for studying aggregation of platelets in their natural environment without isolation of platelet-rich plasma or washed platelets. Therefore, the method used should limit a risk of undesirable artificial platelet activation during preparation. 55 ADP, collagen and AA were used in the study as platelet agonists. The effect was strongly depending on the agonist used, and only in the case of AA, 100 µg/ml LCE significantly reduced platelet aggregation. Thus, out of the three different pathways of platelet activation, namely, purinergic receptors for ADP, several platelet receptors for collagen, and metabolism of AA to thromboxane (TX), only the latter seems to be affected by the studied plant extract.
ADP is regarded as a weak agonist of blood platelets but remains an important mediator of platelet activation evoked by other agonists, which induces massive ADP release from dense granules, where it occurs in molar concentrations. Thus, ADP action underlies a positive feedback that facilitates further platelet aggregation and leads to platelet plug formation. Platelets express two types of P2Y ADP receptors: P2Y1 and P2Y12. ADP-dependent platelet aggregation is initiated by the P2Y1 receptor, whereas P2Y12 receptor augments the activating signal and promotes platelet release reaction.56,57
Two receptors are potentially involved in the platelet response to collagen: integrin α2β1 promotes platelet adhesion to collagen, allowing these cells to interact with the lower-affinity receptor glycoprotein VI, which is responsible for platelet activation. Collagen-mediated platelet activation proceeds through activation of the phospholipase Cγ2 pathway leading to inositol 1,4,5-trisphosphate and 1,2-diacylglycerol formation. Inositol 1,4,5-trisphosphate induces the release of calcium from the dense tubular system, which is accompanied by the entry of extracellular calcium, whereas 1,2-diacylglycerol activates protein kinase C. In addition, 1,2-diacylglycerol and calcium mediate platelet activation responses, such as shape change, granule secretion and aggregation. 58
Activated phospholipase A2 releases AA by cleaving fatty acids, especially phosphatidylcholine and phosphatidylethanolamine. AA is a precursor for thromboxane A2 synthesis. In the first step, enzyme cyclooxygenase 1 (COX-1) located in blood platelets catalyses the transformation of AA into cyclic endoperoxide PGG2 and PGH2. These substrates are then converted by thromboxane A2 (TXA2) synthase into TXA2, which is a platelet agonist acting via TX receptor coupled to Gq-protein. 59
Our observation that LCE inhibited AA-induced aggregation only suggests that the plant extract could interfere with the COX-1-related platelet activation pathway. Obviously, we should keep in mind that lower concentrations of collagen could also exert the same effect as AA on platelets by stimulating TXA2 production. 58 However, collagen is a much stronger agonist than AA, acting (as it was described above) also via other molecular pathways. In light of these facts, only the inhibitory effect of LCE on AA-induced aggregation is understandable. This observation could be regarded as an assumption for further studies, possibly testing the effects of LCE on the TX generation in the serum or plasma. Noteworthy, the observed inhibiting effect of LCE is unlikely to be attributed to the cytotoxicity of polyphenols towards blood platelets given that we have recently demonstrated no cytotoxic effects for a couple of polyphenolic extracts tested in a similar range of concentrations. 60
To date, there are no reports indicating the immunomodulatory activity of LCE. According to the studies described herein, LCE influences the secretion of the factors involved in the regulation of immune responses by human endothelial cells. The stimulatory effect on NO production, together with the significant reduction of induced PAF secretion, place LCE among natural-origin products possessing anti-inflammatory activity. Furthermore, the inhibitory effects of LCE on the AA-evoked aggregation of human platelets reported in our study suggest its potential interference with AA metabolism and TX generation. Moreover, LCE applied in a wide range of concentrations is non-cytotoxic and does not induce cells apoptosis. Thus, we want to pay attention to the possibility of a new use for LCE in preventing the development of difficult-to-treat inflammatory lesions within chronically infected tissues through its immunomodulatory activity. Nevertheless, there is no doubt that future in vivo studies on the effect of LCE are necessary.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for the project was received from The National Science Center (Grant No 2013/09/N/NZ6/00826).
