Abstract
The lung is constantly exposed to immune stimulation by LPS from inhaled microorganisms. A primary mechanism to maintain immune homeostasis is based on anti-inflammatory regulation by surfactant protein A (SP-A), a secreted component of lung innate immunity. The architecture of LPS aggregates is strongly associated with biological activity. We therefore investigated whether SP-A affects the physico-chemical properties of LPS. Determination of the three-dimensional aggregate structure of LPS by small-angle X-ray scattering demonstrated that SP-A induced the formation of multi-lamellar aggregate structures. Determination of the acyl-chain-fluidity of LPS aggregates by Fourier transform infrared (FTIR) spectroscopy showed that the phase transition temperature of LPS was reduced in the presence of SP-A. The phosphate groups at the diglucosamine backbone of LPS represent important functional groups for the bioactivity of LPS. FTIR analysis revealed changes in the vibrational bands νas
Keywords
Introduction
The mammalian immune system has developed highly sensitive recognition systems for LPS (endotoxin) 1 of Gram-negative bacteria, providing an immediate immune response to pathogens. The resulting inflammatory process is the basis of antimicrobial immunity; however, it can also lead to detrimental effects in case of an overbordering systemic immune response leading to sepsis and septic shock. The lipid A part of the LPS molecule represents the minimal active structure inducing the immune response and has therefore been termed its endotoxic principle. The recognition of microbial LPS is tightly regulated and involves a specific set of transport and recognition receptors, as well as immune modulatory proteins. 1 LPS-binding protein (LBP) and glycosylphosphatidylinositol (GPI)-anchored or soluble CD14 are important cofactors for the recognition of the amphiphilic bacterial lipid, which together provide transport of LPS from aggregates to the surface receptors on phagocytes. TLR4, together with the associated protein MD-2, forms the primary cellular signaling receptor for LPS.
In contrast to the sterile vascular compartment, the lung is constantly exposed to environmental noxae, such as dust and microorganisms. To account for this, lung innate immunity has developed specific mechanisms of adaptation combining, under physiological conditions, efficient pathogen removal with an anti-inflammatory immune status. Surfactant protein A (SP-A) is the major secreted hydrophilic protein in the lung providing central immune-regulatory functions. 2 SP-A efficiently attenuates the pro-inflammatory response of the primary resident phagocytes in the lung—the alveolar macrophages—to LPS in vitro and in vivo.3,4 Immune modulation by SP-A has been related to different mechanisms. SP-A has been demonstrated to modulate the LPS-induced canonical NF-κB pathway, as well as the activation of pro-inflammatory signaling cascades in alveolar macrophages by stabilizing the cytoplasmic inhibitor protein IκBα 5 .This regulatory route depends on clathrin-mediated endocytosis and subsequent activation of the atypical kinase PKCζ. 6 A number of potential receptors for SP-A in pro- or anti-inflammatory immune modulation has been suggested, such as SPR-210, SIRPα, GP-340, and the general collectin receptor complex of CD91 and calreticulin. 7 Binding of SP-A to a number of proteins involved in the cellular recognition of LPS has been demonstrated to inhibit pro-inflammatory responses, including CD14, 8 TLR4 and MD-2. 9 In addition, interference of SP-A with LBP and CD14 has also been shown to contribute to immune modulation by the collectin.10–12 From these findings it can be concluded that several different activities contribute to the immune regulation by SP-A. The respective mechanisms underlying SP-A-mediated inhibition of LPS-induced cell activation in vivo and in vitro are only partially understood.
Binding of SP-A to LPS represents a primary step in collectin-mediated immune modulation. LPS is an amphiphilic glycolipid that forms aggregates in aqueous environment. The supramolecular structure of LPS aggregates has been demonstrated to critically determine its immune-stimulatory activity observed in biological systems. However, only limited information is available about the molecular consequences of SP-A binding to LPS. SP-A has been shown to bind to LPS from a variety of microorganisms; however, the capacity of SP-A to interact with LPS appears to depend on the degree of glycosylation. Thus, in solid phase binding assays SP-A binding is strongest to LPS with low saccharide content as deep rough mutant (Re) LPS.8,13 In accordance with this, strains with rough-type LPS, such as lipooligosaccharides, are more common among pathogens colonizing the pulmonary compartment. 14 The aim of the current study was to analyze SP-A-induced changes in the physico-chemical characteristics of LPS aggregates in relation to their capacity to induce immune stimulation.
Material and methods
Reagents
Deep rough mutant LPS (Re LPS) was extracted from Salmonella enterica serovar Minnesota strain R595 and Escherichia coli strain WBB01 according to the phenol/chloroform/light petroleum procedure.
15
LPS preparations were used in the natural salt form. LPS from E. coli strain WBB01 was further purified by a deoxycholate-modified phenol–water method to extract lipoproteins.
16
The LPS preparations were tested on HEK293–TLR2 cells to be devoid of TLR2-activating components. Both types of LPS showed TLR4 activity, as tested on HEK293-TLR4/MD2 cells. Mass spectrometric analysis demonstrated for the LPS from E. coli strain WBB01 a major mass peak in accordance with a hexa-acylated diglucosamine, carrying two 3-deoxy-
For biological experiments, LPS samples were prepared as aqueous suspensions in 20 mM HEPES, pH 7.0 at 1 mg/ml stock by sonication for 30 min in an ultrasonic bath, temperature-cycled twice between 4 and 56℃ to enable the formation of stable aggregates, and then stored at 4℃ for at least 12 h for equilibration. SP-A or CaCl2 were added as indicated in the respective experiment. For biophysical experiments, 4–8 mg LPS from S. Minnesota strain R595 were dissolved in 50–80 µl of 20 mM HEPES, pH 7.0 without CaCl2, or with 2 or 5 mM CaCl2 and further treated as described above. Prior to measurement, 250 µg SP-A were added to the aggregate preparations to a final sample volume of 120 µl corresponding to a final SP-A concentration of about 4 µM. These preparations were analyzed by small-angle X-ray scattering, infrared-spectroscopy and cytokine induction in human macrophages.
SP-A purification
Human SP-A was isolated from bronchoalveolar lavage of patients with alveolar proteinosis using a sequential butanol and octyglucoside extraction as described in detail previously. 17 Procedures were approved by the Ethical Commission of the University of Lübeck and patients gave informed consent. In brief, the lavage fluid was extracted with butanol and the resulting SP-A containing pellet was sequentially solubilized in octylglucoside and 5 mM Tris (pH 7.4). The SP-A fraction was solubilized in tris buffered water (TBW, 5 mM Tris HCl, Tris-Base, pH 7.4) and treated with polymyxin B agarose (Sigma-Aldrich, Seelze, Germany) beads to reduce endotoxin contamination. SP-A preparations were tested for the presence of bacterial endotoxin using a Limulus amebocyte lysate assay (Cambrex, Wiesbaden, Germany) and contained < 0.2 pg endotoxin/µg SP-A. For quality control, SP-A preparations were run on a 12% SDS gel under reducing conditions and subsequently stained with Coomassie blue to show SP-A monomers and dimers. Biological activity of SP-A was evaluated by the effect on LPS-stimulation of human macrophages.
Activation of human macrophages
Human mononuclear cells (MNC) from healthy donors were isolated from heparinized peripheral blood by the Hypaque–Ficoll gradient method. Procedures were approved by the Ethical Commission of the University of Lübeck and the volunteers gave informed consent. MNC were harvested, washed and cultivated for 7 d at 37℃, 5% CO2 atmosphere in Teflon bags in RPMI 1640 medium containing 100 U/ml penicillin, 100 µg/ml streptomycin, 2 mM
Stimulation of HEK293–TLR4/MD-2 cells
The HEK–TLR4/MD2 cell line was established by transfection of HEK293 cells with expression plasmids coding for human TLR4 and human MD2. Stable clones were selected in the presence of hygromycin and geniticin. Cells were maintained at 37℃ under an atmosphere of 5% CO2 in DMEM (Biochrom AG, Berlin, Germany) containing 10% FCS (Linaris, Werthein, Germany), 2%
X-ray scattering
Small angle X-ray scattering measurements of LPS aggregate preparations and LPS/SP-A complexes were performed at the European Molecular Biology Laboratory outstation at the Hamburg synchrotron radiation facility HASYLAB using the SAXS camera X33.
19
Scattering patterns in the range of the scattering vector 0.1 < s < 1.0 nm−1 (s = 2 sin θ/λ, 2θ scattering angle and λ the wavelength = 0.15 nm) were recorded between 20℃ and 80℃ with exposure times of 120 s using an image plate detector with online readout (MAR345; MarResearch, Norderstedt, Germany).
20
The s-axis was calibrated with Ag-Behenate. The scattering patterns were evaluated as described previously
21
assigning the spacing ratios of the main scattering maxima to defined three-dimensional (3D) structures. The lamellar and cubic structures are most relevant here. They are characterized by the following features:
Lamellar—the reflections are grouped in equidistant ratios, i.e. 1, 1/2, 1/3, 1/4, etc. of the lamellar repeat distance dl Cubic—the different space groups of these non-lamellar 3D structures differ in the ratio of their spacings. The relation between reciprocal spacing shkl = 1/dhkl and lattice constant a is shkl = [(h2 + k2 + l2)/a]1/2 (hkl = Miller indices of the corresponding set of plane).
Fourier-transform infrared spectroscopy
Infrared spectroscopic measurements were performed on an IFS-55 spectrometer (Bruker, Karlsruhe, Germany). For phase transition measurements, LPS or LPS/SP-A was placed in a CaF2 cuvette with a 12.5 -µm Teflon spacer. Temperature scans were performed automatically between 5℃ and 70℃ with a heating rate of 0.6℃/min. Every 3℃, 50 interferograms were accumulated, apodized, Fourier-transformed and converted to absorbance spectra. For the evaluation of the gel to liquid crystalline phase behavior, the peak position of the symmetric stretching vibration of the methylene band νs (CH2) around 2850 cm−1 was analyzed, which is a sensitive marker of acyl chain order.
Attenuated total reflectance infrared spectroscopy
Lipid samples were prepared as hydrated oriented thin multilayers, as described previously, 22 by spreading LPS or LPS/SP-A aggregate preparations on a ZnSe attenuated total reflectance (ATR) crystal. Infrared ATR spectra were recorded on a IFS-55 spectrometer (Bruker) using a mercury–cadmium–telluride detector with a scan number of 1000 at a resolution of 2 cm−1. Anti-symmetric stretching vibrations from the negatively-charged phosphate groups νas(PO2–) were recorded at 28℃ in the wave number range 1300–1180 cm−1. The vibrational band around 1220 cm−1, assigned to the 1-phosphate group and around 1260 cm−1, assigned to the 4’-phosphate group, were analyzed to evaluate the state of hydratization of the phosphate residues at the diglucosamine backbone.
Results
SP-A inhibits LPS-induced activation of HEK293-TLR4/MD-2 cells
SP-A purified from bronchoalvealar lavage was analyzed by separation on a 12% SDS gel under reducing conditions. Staining with Coomassie blue showed the typical appearance of SP-A monomers and dimers at about 35 and 70 kDa, respectively (Figure 1a). Cell activation by LPS depends on the presence of the TLR4/MD-2 receptor complex. We used a HEK293–TLR4/MD-2 cell line as a model to demonstrate the anti-inflammatory effect of SP-A. The ability of SP-A to inhibit the production of IL-8 in HEK293–TLR4/MD-2 cells was tested after stimulation with LPS alone or with LPS pre-incubated for 30 min with SP-A. Inhibition of the IL-8 secretion from the cells was observed in the presence of 20 and 40 µg/ml SP-A compared with stimulation with LPS in the absence of SP-A (Figure 1b). To get more information on the function of SP-A, we investigated the kinetics of SP-A-mediated inhibitory effects. The cells were either pre-incubated with SP-A between 30 min and 24 h and subsequently stimulated with LPS, or SP-A was added at the time of LPS stimulation without pre-incubation of the cells. In these experiments, short pre-incubation times of 30 and 60 min, and also simultaneous addition of LPS and SP-A, were sufficient to mediate strong inhibitory effects on the LPS-induced production of IL-8 (Figure 1c). These results derived from HEK293–TLR4/MD-2 cells demonstrate that the attenuation of the cell activation by SP-A is a very immediate effect and suggest that SP-A may inhibit biological activity by direct interaction with LPS.
SP-A inhibits LPS-induced activation of HEK293–TLR4/MD-2 cells. (a) Comassie blue staining of SP-A isolated from bronchoalveolar lavage. Gel shows representative preparations. (b) Re LPS from E. coli was pre-incubated for 30 min at 37℃ with buffer or SP-A and subsequently added to HEK293–TLR4/MD-2 cells. (c) For the investigation of the time dependence of SP-A-mediated effects on LPS stimulation, HEK293–TLR4/MD-2 cells were pre-incubated with SP-A between 30 min and 24 h before stimulation with 1 ng/ml LPS, or SP-A and LPS were added simultaneously to the cells. Cell-free supernatants were collected after 24 h of LPS stimulation and analyzed for the chemokine IL-8. Data shown are mean and SD of triplicate samples of one experiment representative of three independent experiments.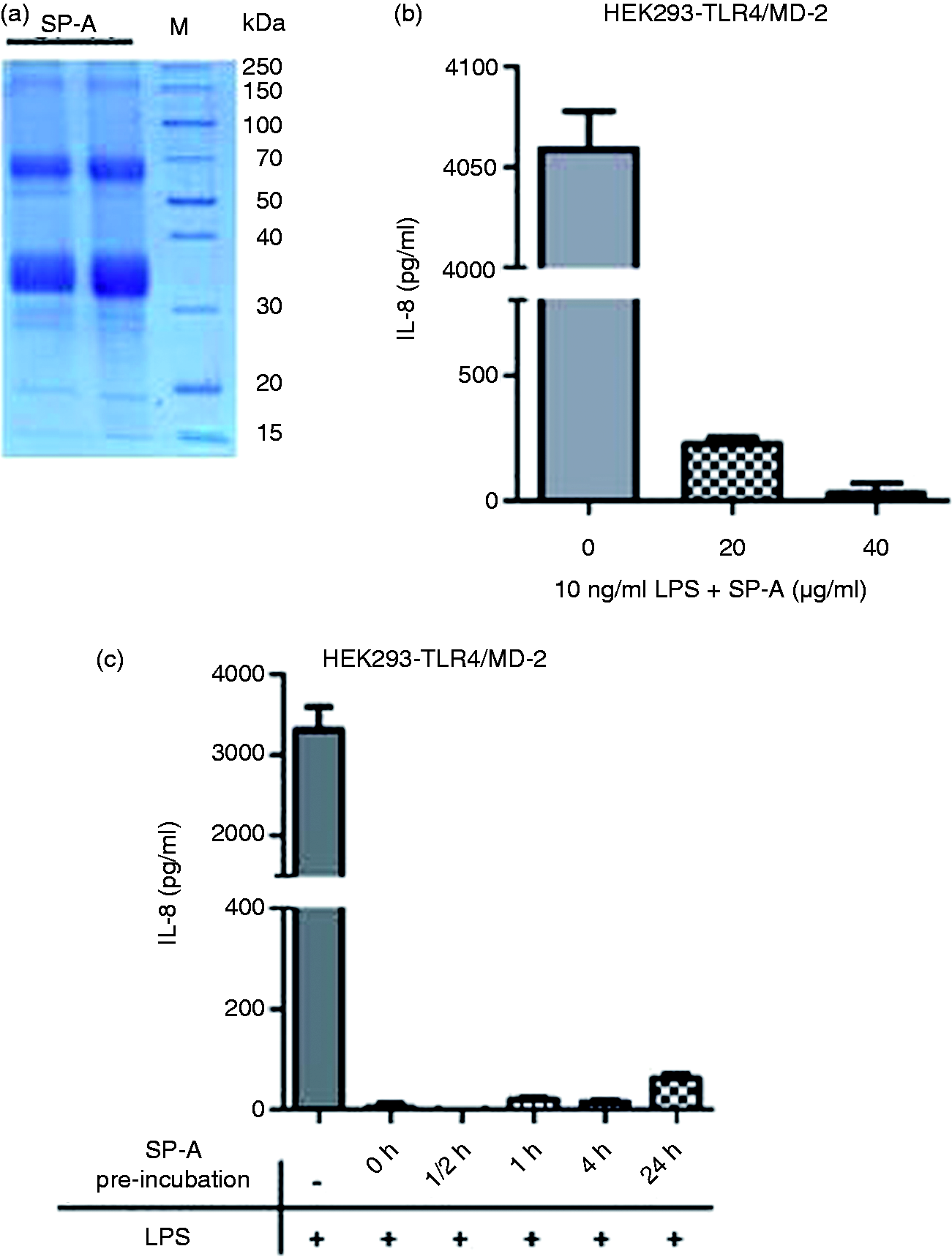
Response of human macrophages to SP-A/LPS complexes
Macrophages are the primary target cell of SP-A immune modulatory function. Therefore, we next investigated SP-A effects on the response of human macrophages to LPS. In order to avoid priming of macrophages by SP-A of the physiological environment, we performed experiments with macrophages derived from peripheral blood. We stimulated macrophages with LPS or LPS pre-incubated with SP-A for 30 min at 37℃ to allow interaction of LPS aggregates with the protein and determined the production of TNF-α as an important early pro-inflammatory mediator induced by LPS. Activation of macrophages by LPS/SP-A complexes in the presence of 5 mM CaCl2 was strongly attenuated compared with stimulation with LPS in the absence of SP-A (Figure 2a, left panel). TNF-α-production was reduced in the presence of SP-A by about 85–90% compared with LPS control at SP-A concentrations between 10 ng/ml and 10 µg/ml. Surprisingly, higher concentrations of SP-A in the range of 20–60 µg/ml were less active and at 60 µg/ml SP-A no inhibitory effect was observed at all. Ca2+ binding has been demonstrated to be crucial for many of the biological functions of SP-A. In accordance with this, when SP-A and LPS were pre-incubated in the absence of CaCl2, the attenuation of TNF-α-production from macrophages was much less pronounced. The optimal inhibitory concentration of SP-A was around 1 µg/ml (Figure 2a, right panel), leading to an inhibition of about 45% of the TNF-α production observed without SP-A. Also under these conditions the inhibitory affect was reduced with increasing concentrations in the range of 20–60 µg/ml. Results similar to the above presented with LPS from S. Minnesota were also obtained with LPS from E. coli (data not shown), supporting the fact that the observed effects are also valid for Re LPS from another bacterial species.
Incubation of Re LPS from S. Minnesota with SP-A dose-dependently inhibits TNF-α-production from human macrophages. (a) LPS was pre-incubated with SP-A for 30 min at 37℃ in HEPES buffer with 5 mM CaCl2 (left panel) or without CaCl2 (right panel). Human macrophages were stimulated with the LPS/SP-A complexes at the indicated final concentrations. (b) Human macrophages were pre-incubated with SP-A for 30 min, 1, 2 or 4 h, and subsequently stimulated with 1 ng/ml LPS in the presence of 5 mM CaCl2. TNF-α was determined in cell-free supernatants after 4 h of LPS stimulation (mean and SD are from duplicate determinations). The data are representative of three independent experiments with cells from different donors.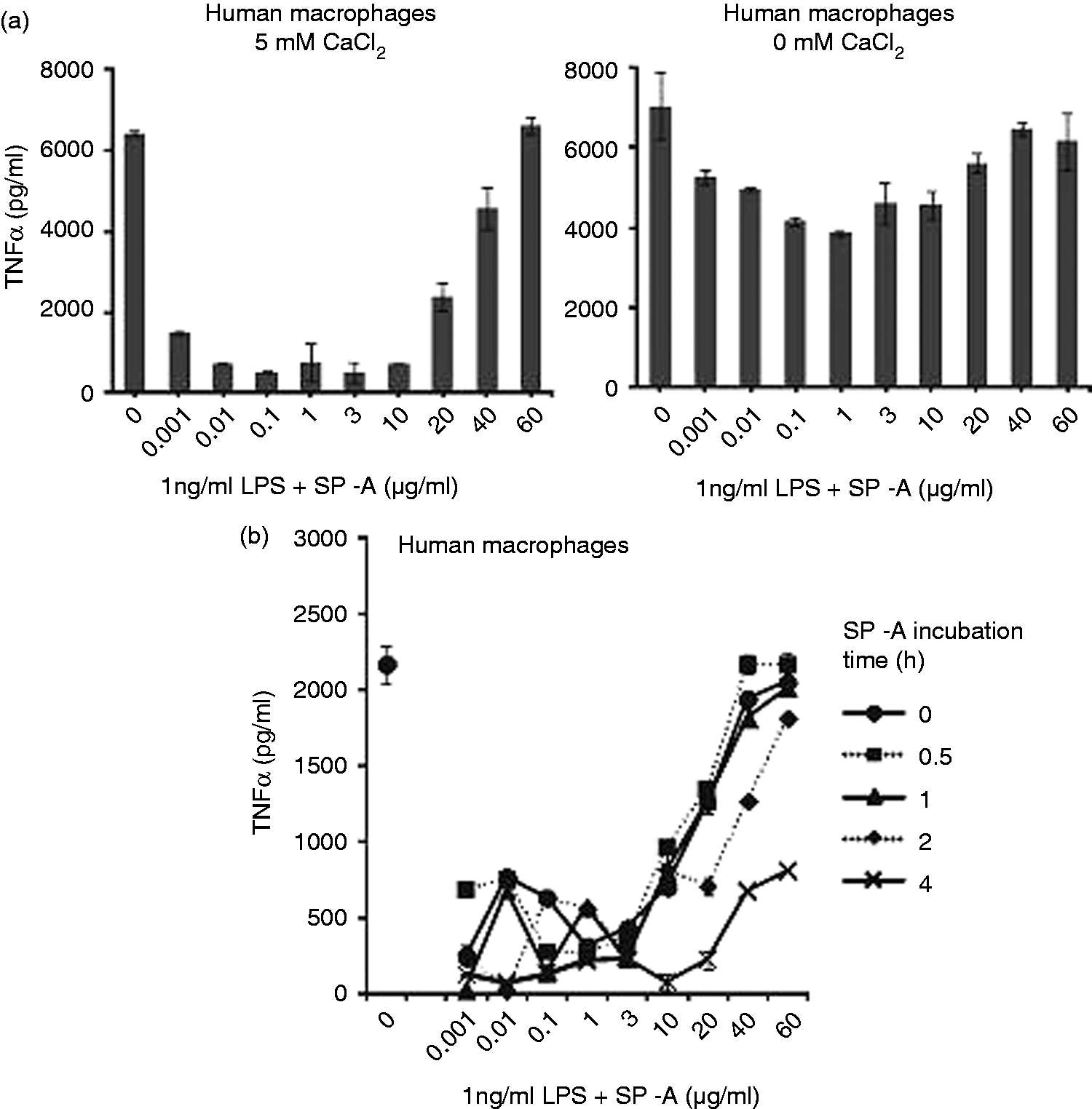
To compare the effects observed upon pre-incubation of SP-A and LPS with a setting where SP-A can act on the macrophage cell before LPS stimulation, we performed a set of experiments where macrophages were first pre-incubated with SP-A for various times and subsequently stimulated with LPS, or SP-A and LPS were added simultaneously. Pre-incubation of macrophages with SP-A for 30 min and 1 h led to strong attenuation of the TNF-α-production upon LPS stimulation at concentrations between 1 ng/ml and 10 µg/ml of SP-A (Figure 2b). In accordance to what we observed upon pre-incubation of SP-A and LPS to form complexes, at increasing concentrations of SP-A from 20 to 60 µg/ml its inhibitory effect was strongly reduced. However, at longer times of pre-incubation of the macrophages with SP-A, the inverse dose effect of SP-A-mediated attenuation of LPS stimulation was less pronounced and at 4 h pre-incubation TNF-α-production was attenuated by about 65% also at the higher SP-A concentrations applied.
Effect of SP-A on the 3D structure of LPS aggregates
Physico-chemical properties of LPS have been shown to critically determine its biological activity. Thus, conical-shaped LPS molecules, which form cubic inverted aggregate structures, exhibit high biological activity, whereas lamellar aggregate structures, which are formed by cylindrical LPS molecules, exhibit no (or low) biological activity on human immune cells.
23
We therefore investigated SP-A/LPS complexes by synchroton radiation small-angle X-ray scattering to elucidate the influence of SP-A on the 3D structure of LPS aggregates. In Figure 3a, scattering patterns of LPS from the deep rough mutant strain S. Minnesota R595 are presented at temperatures between 20℃ and 80℃. LPS aggregates in the presence of SP-A show a temperature-dependent appearance of individual scattering peaks (Figure 3a, left panel) at 40℃ and 60℃. At 40℃, reflections with equidistant spacing ratios were observed at 7.04 nm, and the first-order periodicities d/2 = 3.55 nm, and 5.35 nm and d/2 = 2.65 nm (Figure 3a, right panel), which are indicative of a multi-lamellar aggregate structure formed by molecules with a cylindrical molecular conformation with similar cross-sections of the hydrophilic backbone and the hydrophobic hydrocarbon chains within the aggregate. Reflections showed increased intensity at 60℃ and were slightly shifted towards lower values, indicating the higher fluidity of the acyl chains at temperatures above the phase transition temperature Tc. At 80℃, the aggregate structure was dominated by a major reflection at 4.81 nm, which is consistent with the completion of acyl chain melting in the liquid crystalline (α) phase. Control experiments with LPS aggregates in the absence of SP-A did not show any indication of multi-lamellar reflections (data not shown).
SP-A affects the 3D structure of aggregates. (a) Aggregates from Re LPS of S. Minnesota were prepared in 20 mM HEPES and 5 mM CaCl2 by ultrasonication. SP-A was added to a final concentration of ∼ 4 µM. X-ray scattering was performed at temperatures between 20 and 80℃. The scattering patterns are presented as relative intensities of the scattering vector(s). The numbers indicated are the spacing ratios of the reflections d = 1/s (nm) and are indicative of the aggregate structure. (b) To determine the biological activity of the preparations used for SAXS measurements, the samples were diluted in 20 mM HEPES and human macrophages were stimulated at the indicated final concentrations. TNF-α was determined in cell-free supernatants in duplicate determinations. Data of three independent experiments with cells from different donors were normalized to the sample 100 ng/ml LPS in the absence of SP-A. Data were evaluated by a one-tailed Students’ t-test and considered statistically significant (*) if P ≤ 0.05.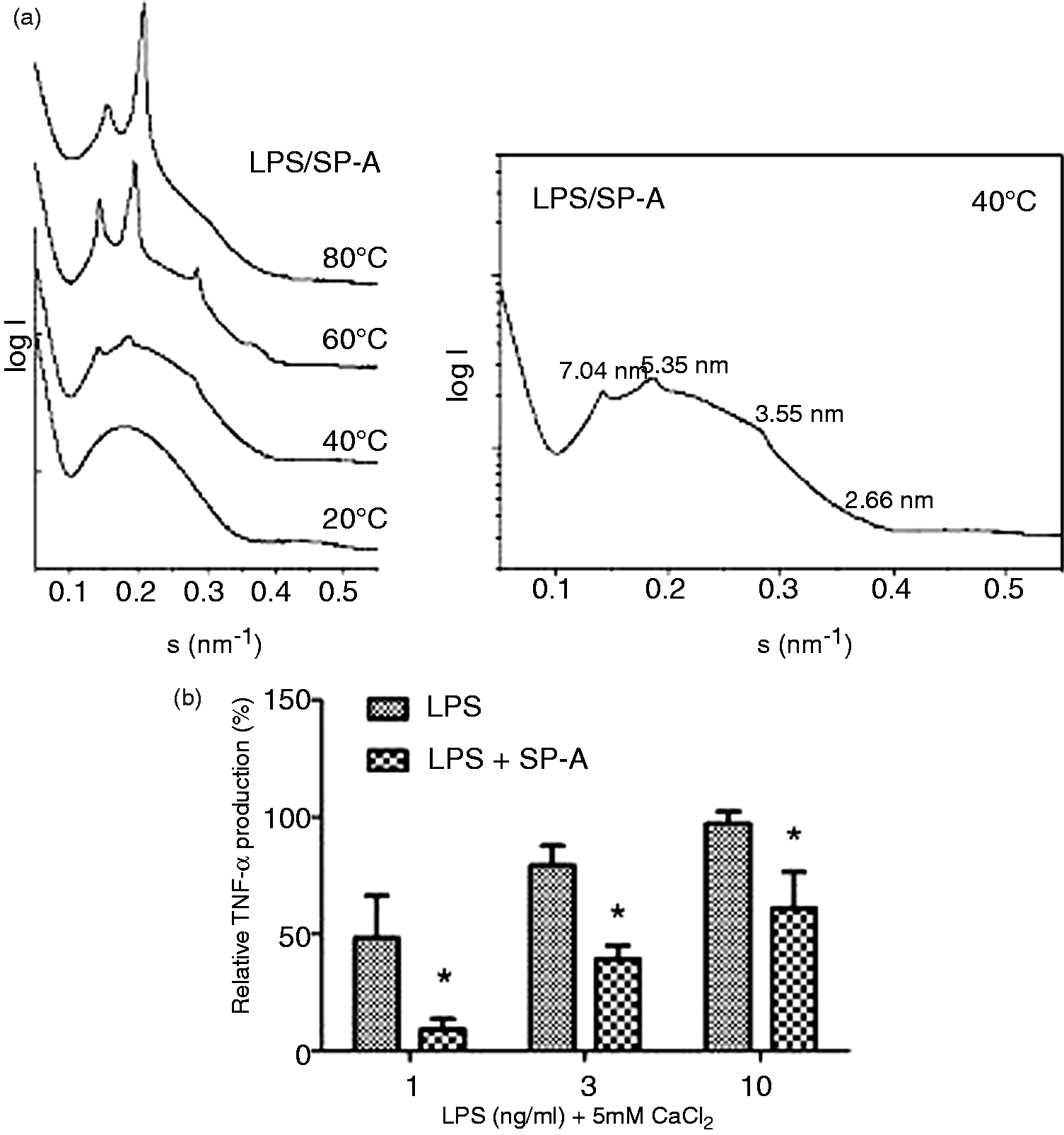
The concentrations of LPS necessary for small-angle X-ray scattering experiments were in the range of several mM LPS, which is extremely high compared with the concentrations used in biological test systems. To demonstrate the biological function of SP-A at these concentrations of LPS, we diluted LPS aggregates and LPS/SP-A complexes prepared for SAXS measurements in buffer to biologically relevant LPS concentrations and determined the activation of human macrophages. Macrophages showed a typical dose-dependent production of TNF-α when stimulated with the LPS diluted from the highly concentrated aggregate preparation with sensitive activation starting at LPS concentrations around 100 pg/ml. Dose-dependent activation was observed up to a concentration of 10–100 ng/ml LPS. At higher concentrations TNF-α production was maintained at a donor-specific maximal level (data not shown). The presence of SP-A significantly (P ≤ 0.05) attenuated TNF-α-production by LPS (Figure 3b), indicating that the preparations used for the determination of aggregate structures exhibited biologically relevant effects.
SP-A decreases the acyl chain fluidity in LPS aggregates
An important physico-chemical characteristic of LPS is the fluidity of the hydrocarbon chains. At low ambient temperatures the acyl chains are in a solid gel (β) phase, which is characterized by low fluidity of the acyl chains, whereas at high temperatures the acyl chains are in a liquid crystalline (α) phase, with high fluidity of the hydrocarbon chains. Between these phases, a main gel-to-liquid crystalline phase transition can occur at a characteristic phase transition temperature Tc.
To further characterize the interaction of SP-A and LPS, we investigated the fluidity of acyl chains in LPS aggregates in the absence and in the presence of SP-A by evaluating the symmetric stretching vibrational band νs (CH2) by infrared spectroscopy. Aggregates from Re LPS of S. Minnesota in the presence of 5 mM CaCl2 exhibited a phase transition from the gel (β) phase to the fluid (α) phase around 34℃ (Figure 4). In the presence of SP-A, the chain melting was shifted to lower temperatures with a transition temperature Tc at 30℃. In the fluid (α) phase, the CH2 stretching vibrations were observed at higher wave numbers for the LPS/SP-A complexes, suggesting that the binding of SP-A led to a higher degree of mobility of the acyl chains also in the fluid phase. At a concentration of 2 mM CaCl2, the lowering of the phase transition temperature by SP-A was less pronounced with a shift to 31℃, and, in the absence of CaCl2, no effect of SP-A on Tc was observed (data not shown).
SP-A increases the acyl-chain fluidity of LPS aggregates from Re LPS of S. Minnesota. The acyl chain fluidity of LPS aggregates was determined by Fourier transform infrared spectroscopy in dependence on temperature in the presence or absence of SP-A. The vertical line indicates the physiological temperature of 37℃. The phase transition temperature of the gel to liquid crystalline phase transition of LPS is Tc = 30℃ and of LPS/SP-A Tc = 34℃, respectively. The data shown are representative of two experiments with independent LPS and LPS/SP-A preparations.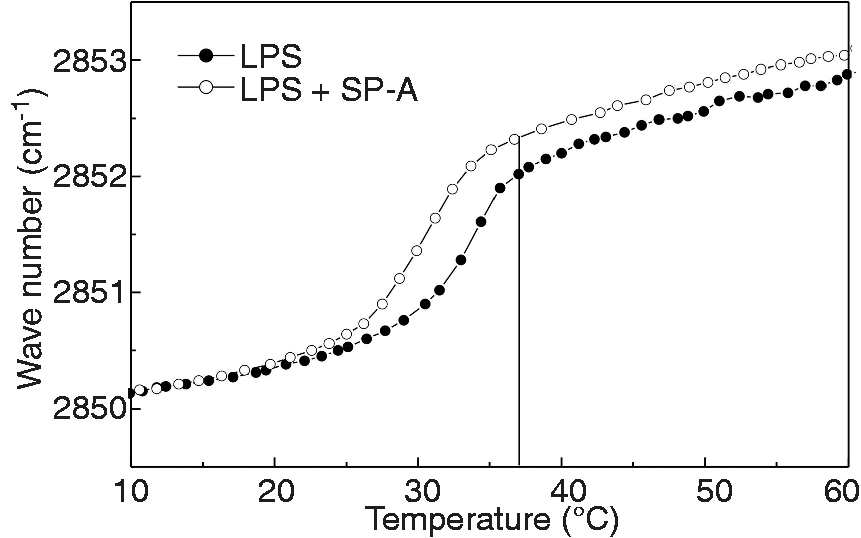
Interaction of SP-A with the phosphate groups of LPS
The phosphate groups bound to the diglucosamine backbone of the lipid A moiety are of central importance for the immunological activity of LPS. They represent major recognition sites for the interaction with LPS binding proteins, such as LBP and CD14, which are responsible for transport and highly sensitive recognition of LPS by cellular receptors. However, little is known about the affinity and interaction of SP-A with phosphate groups. To obtain information about potential interactions of SP-A with phosphate groups of LPS-aggregates, we studied the spectral region of the anti-symmetric stretching vibration νas PO2– (1220–1260 cm−1) of LPS aggregates and LPS/SP-A complexes by ATR spectroscopy. This vibration is sensitive to hydration changes of the phosphate groups, indicated by changes in the wave number of the phosphate-specific absorption peaks. In Figure 5a, the infrared absorption spectrum of LPS is shown. Analysis of the absorption intensities of the phosphate-specific wave numbers resulted in the specific absorption spectra shown in Figure 5 (c, d). The assignment of the phosphate groups to their position at the diglucosamine backbone of LPS is shown in Figure 5b. The wave numbers at 1220 cm−1 and 1260 cm−1 can be related to the absorption of the 1 - and the 4’-phosphate group, respectively. Clearly, in the presence of SP-A a shift of the absorption maximum of the 1-phosphate group to higher wave numbers was observed in the presence of 5 mM Ca2+ (Figure 5c). In contrast, the absorption peak of the 4’-phosphate was not affected by SP-A. The shift in the absorption peak was observed in the presence, as well as in the absence, of Ca2+ (Figure 5d), demonstrating that Ca2+ is not a necessary cofactor for this effect.
Interaction of SP-A with the phosphate groups of LPS. The influence of SP-A on the phosphate groups of LPS was determined in LPS aggregates from Re LPS of S. Minnesota in the presence or absence of SP-A. (a) Absorption spectrum of LPS aggregates indicating the wave number ranges relevant for the vibrational absorption of the CH2 groups of the acyl chains and the anti-symmetric stretching vibrations from the negatively-charged phosphate groups νas(PO2−). (b) Assignment of the phosphate groups of LPS. (c) The vibrational band around wave number 1220 cm−1, assigned to the 1-phosphate group and around 1260 cm−1, assigned to the 4’-phosphate group, were analyzed to evaluate the state of hydratization of the phosphate residues at the diglucosamine backbone in the presence of 5 mM CaCl2 and (d) in the absence of CaCl2. The data shown are representative of two experiments with independent LPS and LPS/SP-A preparations.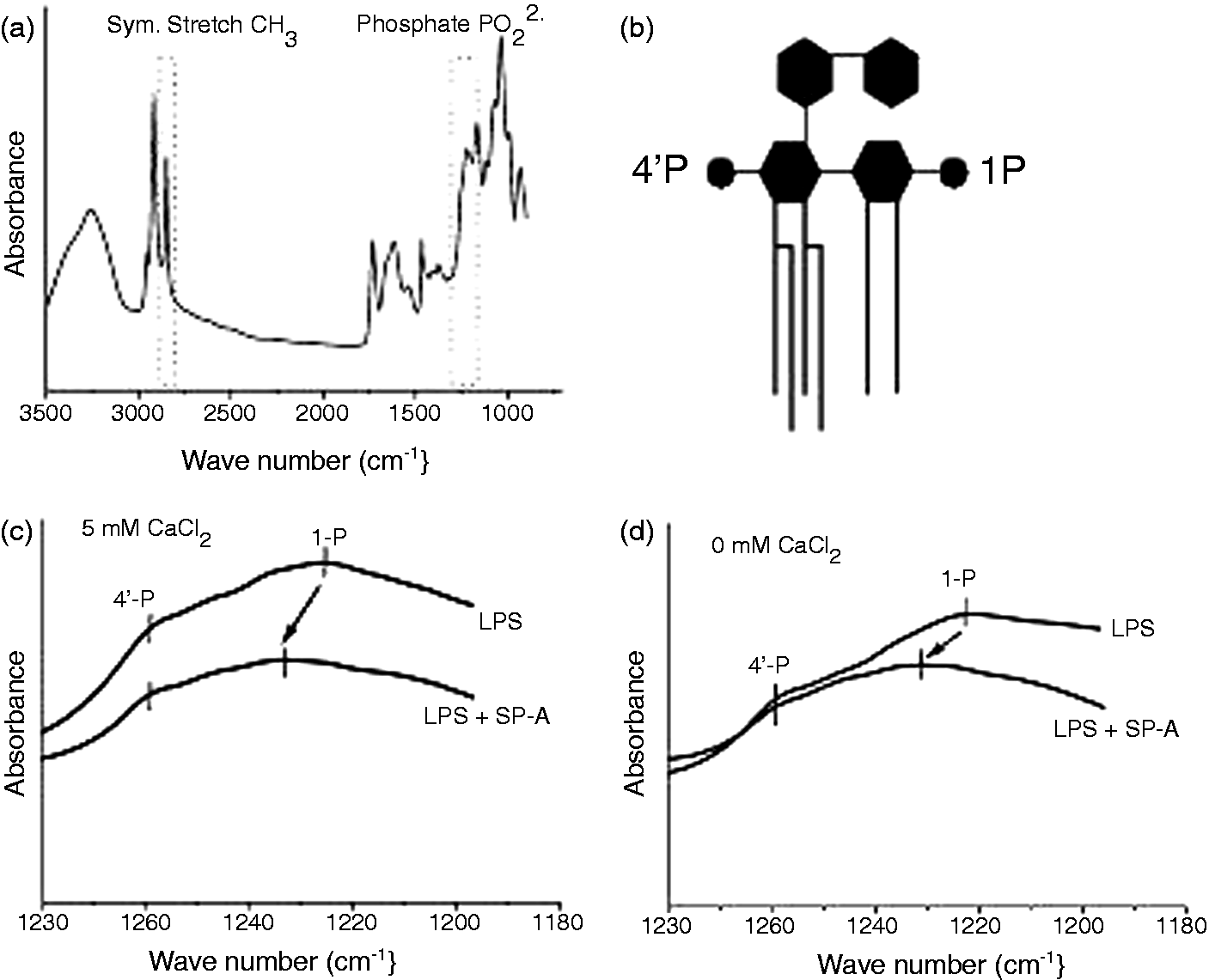
Discussion
In aqueous environments like the aqueous alveolar lining, LPS released from the surface of dividing or dying microorganisms undergoes aggregation above the critical aggregate concentration, which can be estimated to be in the nanomolar range or even lower.23,24 Therefore, recognition by pattern recognition receptors does not only depend on the chemical composition of LPS, but also includes the recognition of the 3D supramolecular structure of aggregates. Using small-angle X-ray scattering to determine the structure of Re LPS aggregates we found that SP-A profoundly affected the organization of LPS aggregates. LPS from enterobacterial origin, such as from deep rough mutant strains of E. coli or S. Minnesota, have been shown to form cubic or hexagonal inverted aggregate structures, which are associated with high endotoxic activity.25,26 Our data demonstrate that SP-A induced a re-arrangement of the aggregate structure of LPS and induced the formation of multi-lamellar aggregate systems characterized by X-ray scattering maxima of first and second order with equidistant spacing ratios (Figure 3a). These effects were most pronounced at a temperature of 40℃, which is close to the physiological body temperature. The occurrence of two different major scattering peaks at 7.04 nm and 5.35 nm observed at 40℃ (Figure 3a, right panel) indicate the co-existence of two major structural entities. The different membrane spacing observed may result from association of different amounts of SP-A to the LPS aggregates. At 80℃, the structure is dominated by a peak at 4.81 nm, indicating that the membrane thickness above the phase transition is reduced. The formation of multi-lamellar aggregate structures is associated with low biological activity, which has been demonstrated for lipid A from Enterobacteriaceae, but also from other microorganisms, such as Chromobacterium violaceum and Rhodobacter capsulatus, 26 for synthetic lipid A mimetic structures, 27 and has also been shown to be valid for other microbial membrane components, such as lipopeptides. 28 A surprising effect in the opposite direction has been demonstrated for hemoglobin, which induces the formation of cubic and hexagonal inverted aggregate structures of LPS aggregates, thereby increasing the biological activity of LPS, 29 strikingly demonstrating the effect of physico-chemical characteristics on the recognition process. Thus, the SP-A-induced changes clearly induce the formation of aggregate structures expressing low biological activity on human immune cells.
Contribution of a physico-chemical mechanism to the SP-A-mediated attenuation of the biological activity of LPS is supported by the kinetics of SP-A immune regulation observed in experiments using a HEK293-TLR4/MD-2 cell line. The HEK epithelial cell line is a well-investigated model for the function of the LPS receptor TLR4/MD-2. Stimulation of these cells with LPS pre-incubated with SP-A demonstrated strong inhibitory effects of SP-A on IL-8 production (Figure 1b), supporting the conclusion that the observed effects of SP-A are not only applicable to macrophages, but also apply to other LPS-responsive cell types. The inhibitory effect of SP-A observed on the HEK293–TLR4/MD-2 did not require time for cellular interaction prior to stimulation with LPS. Addition of SP-A to the cells at the same time as addition of LPS was sufficient to completely block IL-8 production under these conditions (Figure 1c), underlining the importance of direct LPS/SP-A interaction for the regulation of cell activation. Attenuation of the immune response by LPS/SP-A complexes of human macrophages was dose-dependent, with optimal inhibitory effects observed at a SP-A concentration around 0.1 µg/ml. In accordance with the described importance of Ca2+-binding to the Ca2+-coordination sites in the carbohydrate recognition domain (CRD) on a variety of SP-A functions, the inhibitory function of SP-A was strongly increased in the presence of 5 mM CaCl2 (Figure 2a). These experiments showed an unusual dose response of SP-A with decreasing efficiency of inhibition on LPS activation with increasing concentrations of SP-A (Figure 2a). One might speculate that SP-A self-aggregation, which occurs depending on Ca2+ and salt concentrations, pH and temperature, 30 might contribute to a loss of activity at high SP-A concentrations. However, the experiments presented in this article do not provide an explanation of the underlying mechanisms. As several pathways of cellular immune regulation by direct interaction of SP-A with alveolar macrophages have been described, we compared the dose response observed upon stimulation with pre-incubated SP-A/LPS complexes with a setting where cells were incubated for varying times in the presence of SP-A before stimulation with LPS. Under these conditions, an increase in SP-A function with incubation time was observed specifically at high SP-A concentrations of 20–60 µg/ml, and after 4 h of pre-incubation of the macrophages with SP-A these concentrations also exhibited strong inhibitory activity (Figure 2b). This biphasic effect of the inhibitory function of SP-A may actually be an indication of the different modes of action of the collectin at the level of ligand interaction and at the cellular level.
The diglucosamine backbone of LPS is acylated by ester- and amide-linked hydrocarbon chains that determine the amphiphilic nature of the molecule. Changes in the fluidity of the acyl chains have been shown to modulate the biological activity of LPS.31–33 SP-A induced a Ca2+-dependent decrease of the phase transition temperature by 2℃ in the presence of 2 mM CaCl2 (data not shown) and by about 4℃ at 5 mM CaCl2 (Figure 4). The decrease in Tc, as well as the higher state of fluidity observed above Tc, indicate higher mobility of the acyl chains in the presence of SP-A at the physiological temperature of 37℃. Experiments on lipid A monolayers showed an increase in the space requirement of lipid A molecules upon interaction of SP-A from the sub-phase, which was interpreted as integration of SP-A molecules into the monolayer. 34 However, the observed effect may be alternatively explained by an effect of SP-A on the acyl chain mobility leading to a fluidization. Thermodynamic analysis of LPS/SP-A complexes by differential scanning calorimety demonstrated a decrease in the transition enthalpy of LPS aggregates undergoing the gel-to-fluid phase transition. 35 The acyl chain fluidity is likely to influence the aggregate stability and therefore can be assumed to be a parameter also affecting biological activity. Both aggregates and monomers have been shown to be important for the biological recognition process.36,37 It has been proposed that the acyl chains are involved in the binding of LPS to crystallized TLR4/MD-2 complexes with the acyl chains of LPS tightly buried in the hydrophobic pockets of the binding receptor MD-2. 38
The phosphate groups belong to the most important functional groups of LPS. Reduction of the number of phosphates from two to one is accompanied by a strong attenuation in biological activity, and dephosphorylated lipid A is devoid of biological activity. 39 The phosphate groups adopt different orientations within the plane of LPS membranes. Thus, the 1-phosphate has been shown to be highly hydrated and is surrounded by an aqueous environment sticking out of the membrane plane. In contrast, the 4’-phosphate is adjacent to the hydrophobic region of the lipid A backbone. 40 The observed shift of the absorbance peak of the 1-phosphate at 1220 cm−1 to higher wave numbers clearly indicates a dehydration of the 1-phosphate group in the presence of SP-A (Figure 5c, d). Notably, the presence of substituents at the lipid A backbone, as determined by mass spectrometry analysis, does not appear to interfere with the observed effect of SP-A on the hydration of the phosphate residue. The negative charges presented by the phosphate groups are important for the binding to LBP, 41 as well as for the activation of the TLR-4/MD-2 receptor complex. 38 Binding of SP-A to the exposed 1-phosphate could lead to a shielding of this functional group thereby preventing the interaction with proteins important for the biological activity of LPS. In line with this conclusion would be the observation that SP-A can inhibit the binding of LPS to LBP. 12 Other reports did not observe effects of SP-A on LPS binding to LBP, but suggest an interference with the transport activity of LBP leading to diminished functional transport of LPS to CD14. 10
SP-A is associated with the phospholipid-rich alveolar surfactant and a major part of the collectin is bound to dipalmitoylphosphatidylcholine (DPPC). LPS binding has been demonstrated to occur via the CRD of SP-A; however, mechanisms of binding and contributions of the specific functional groups in LPS/SP-A binding are not very well defined. Crystallographic analysis of human SP-A revealed structural information on monosaccharide binding sites in the CRD. 42 The binding pockets and mechanism of binding to DPPC of the lung surfactant and to microbial ligands such as LPS are still to be defined.
In conclusion, we have demonstrated that the interaction of SP-A with LPS aggregates induces specific changes in the 3D conformation of LPS molecules leading to a rearrangement of the supra-molecular aggregate structure. The formation of multi-lamellar aggregate structures has been associated with a decrease in biological activity of LPS and therefore provides a molecular mechanism of immune regulation by SP-A. Our findings demonstrate a new mode of action contributing to the important anti-inflammatory functions of SP-A and underline the concept that SP-A acts at different molecular levels (a) by direct effects on microbial-derived molecules such as LPS; (b) by effects on LPS-binding proteins and cellular receptors and (c) by immune regulatory effects on the alveolar macrophages, which together provide anti-inflammatory immune regulation as a basis to immune homeostasis in the lung.
Footnotes
Funding
This study was carried out with financial support from the Deutsche Forschungsgemeinschaft (grant SCHR621/3-1 to AS).
Acknowledgements
We acknowledge the excellent technical assistance of Sabrina Groth, Nina Hahlbrock and Irina von Cube. We are indebted to Professor Stamme, Research Center Borstel, for providing bronchoalveolar lavages and SP-A preparation for our initial experiments.
