Abstract
Endotoxins (LPS) are highly potent immune stimulatory molecules and are mainly known for triggering Gram-negative sepsis. However, besides their toxic effects, this stimulatory function may be advantageous, for example when used as an adjuvant during vaccination. Thus, there is always a narrow range between the useful wake-up of the immune system and its overwhelming reaction, which can lead to diseases like sepsis. This raises the question of which conformational properties are responsible for making the LPS aggregates more or less potent. As described previously, the size, type and form of LPS aggregates play a major role in their immune stimulatory activity. In this study we investigate the role of these parameters. On the one hand, we use a peptide (Pep19-2.5; Aspidasept) that causes a change of the LPS aggregate structure into a less toxic state; on the other hand, we use a potent immune stimulating peptide (Hbγ-35), leading to higher toxicity. We have found opposing effects on LPS aggregate conformations allowing a better understanding of the processes of immune stimulation.
Introduction
LPS is a well-known trigger of Gram-negative sepsis. Various studies have focused on the role of inducing the generalised inflammation reaction, with often fatal outcomes.1,2 The size and form of the LPS aggregates are crucial for the immune-stimulatory effects of LPS. 3 They can range from ordered lamellar structures, as found within the outer membrane of Gram-negative bacteria, as well as in distinct LPS–protein or lipid mixtures to aggregates with cubic or tubular configurations. 4 Whereas the former structures are described as no or rather weak inducers of the immune system, the latter are known to exhibit strong endotoxic activity. It was shown that the accessibility of the lipid A part of the LPS molecule is of essential importance for the interaction with host cell receptors such as LPS-binding protein (LBP), CD14 and the TLR4/myeloid differentiation (MD)2 system responsible for the inflammatory onset. 5
Different endotoxin binding molecules that have an effect on LPS activity have been described, especially natural and synthetic antimicrobial peptides (AMPs), which are able to influence the outcome of an infection.6–10 Despite a great heterogeneity in origin and amino acid sequence, AMPs have in common a positive net charge in combination with an amphipathic secondary structure. It has been demonstrated that some of them have the potential to neutralise endotoxic effects, such as the peptide Pep19-2.5 (Aspidasept), by changing the LPS aggregate structure.11–13 Immunomodulating peptides have been a topic in the past, revealing an influence of the peptides on the host defence mechanisms independent of a direct interaction with microorganisms or toxins.14,15
Since the pioneering publications of Levin and co-workers,16–19 it is thought that molecules may also enhance the biological activity of endotoxins, in particular haemoglobin (Hb) and partial structures.16,20 Hb is the oxygen protein carrier in vertebrates and some invertebrates. In adult humans it is composed of two α and two β chains, with the haeme moiety held in a nonpolar cavity in each peptide chain. Previous evidence has shown that Hb intensifies the biological activity of LPS in the in vitro chromogenic Limulus amoebocyte lysate test, the production of TNF-α in human mononuclear cells (MNCs) and endothelial cell tissue factor assays.16,17,19,21 This enhancement of the biological response was associated with the conversion of LPS aggregates into a rather nonlamellar form, especially into cubic structures.22,23,24 Furthermore, Hb has been assessed as an important subject of clinical investigations in the resuscitation, cancer therapy, sickle cell anaemia, coronary balloon, angioplasty, stroke and myocardial infraction, and has an adjunctive therapeutic treatment.18,19 However, using cell-free Hb involves the risk of possible contamination with other blood components or even LPS, representing a barrier for the successful use of Hb as an adjuvant. The aim of the present work was to characterise a peptide derived from Hb that enhances the activity of the LPS and avoids the risk of impurities from natural compounds.
In this study, we investigated, using various biophysical methods, the interaction of the peptide Hbγ-35 derived from the Hb gamma chain with Salmonella enterica sv. Minnesota R60 LPS. The data showed that the addition of Hbγ-35 to LPS led to a change in the LPS aggregate structure, which is in contrast to the effect described for the peptide Pep19-2.5, suggesting a dissimilar way of binding and/or interaction. These differences have been translated to opposite biological consequences for both peptides. Hbγ-35 is able to enhance the endotoxin immune stimulatory property in contrast to the neutralising Pep19-2.5. Therefore, Hbγ-35 could be an appropriate candidate for use as an enhancer of LPS-derived adjuvants, as well as a stimulating booster of the immune system.
Material and methods
LPS and peptides
Extraction of the LPS from S. Minnesota R60 was performed using the PCP (aqueous phenol, chloroform, and light petroleum) method and analysed by matrix-assisted laser desorption/ionisation MS. 25 Synthesis and purification of Hbγ-35 (TVLAIHFGKEFTPEVQASWQKMVTAVASAL) was performed by EMC Microcollections (Tübingen, Germany) and Pep19-2.5 (GCKKYRRFRWKFKGKFWFWG) by BACHEM (Bubendorf, Switzerland) using procedures described previously. 26
Stimulation of human MNC by LPS
MNCs were isolated from heparinised blood of healthy donors as described previously. 27 The cells (5 × 106 cells/ml) were resuspended in medium (RPMI 1640) and 200 µl MNCs (1 × 106 cells) were transferred into each well of a 96-well culture plate for the stimulation studies. LPS, peptides and the LPS:peptide mixtures were preincubated for 30 min at 37℃ and added to the cultures at 20 µl per well. The cultures were incubated for 4 h at 37℃ under 5% CO2. After centrifugation of the culture plates (10 min, 400× g), supernatants were collected and TNF-α was measured as described in the protocol (OptEIA; BD, Heidelberg, Germany).
Fourier-transform infrared spectroscopy
The measurements were performed on an IFS-55 spectrometer (Bruker, Karlsruhe, Germany). LPS samples, dissolved in 20 mM HEPES buffer (pH 7.4), were spread on a Ge-attenuated reflection (ATR) plate for secondary structure analysis (see below) or were placed in a Ø 19.0–0.2 mm × 3.0 ± 0.1 mm CaF2 crystal disc (Korth Kristalle, Kiel, Germany). For phase transition measurements, temperature scans were performed between 10℃ to 70℃, with a heating rate of 0.6℃/min. In order to determine the peptide effect, it was added at a LPS:peptide ratio of 1:0, 1:0.3 and 1:1. At every step of 3℃, 50 interferograms were accumulated, apodised, Fourier-transformed and converted to absorbance spectra. The main vibrational bands used for the analysis are the symmetric stretching vibrations of the methylene groups versus (–CH2–) located in the range from 2850 to 2853 cm–1, as a measure for the order of the lipid A chains. 28 For the determination of the secondary structure of the peptides, the amide I [mainly v (C = O) stretching] band in the spectral range 1700 to 1600 cm–1 was evaluated.
Isothermal titration calorimetry
Microcalormetric measurements of peptide binding to endotoxins were performed on a ITC200 (GE Healthcare, Munich, Germany) as previously described. 29 For this, 1 mM Hbγ-35 or Pep19-2.5 in 0.9 % NaCl was titrated into 0.26 mM LPS. The enthalpy changes (ΔH) were measured versus time and were recorded against the concentration ratio of peptide to LPS.
Light scattering
In order to study the ability of the peptides to induce LPS aggregation, LPS in 20 mM HEPES pH 7.4 was placed in quartz cuvette at a final concentration of 0.01 mM in a volume of 2 ml and placed in the fluorescence spectrometer. Afterwards, 0.1 mM Hbγ-35 or Pep19-2.5 was added to the sample every 50 s. The temperature was kept constant at 37℃. For a homogeneous distribution of the peptides and LPS aggregates, a small magnet stirrer in the cuvette was added. Scattering intensity was recorded at a wavelength of 400 nm versus time. As controls, the pure peptides and LPS were also monitored.
Results
The two peptides differ in the cytokine secretion
As was demonstrated in previous studies, the addition of Hb to different LPS preparations enhanced the inflammatory cytokine production in mice and human MNCs.16,22 In order to investigate whether the peptide Hbγ-35 has similar effects, we used MNCs to evaluate the expression of TNF-α in the cell medium after stimulation with LPS in the absence and presence of different concentrations of peptide. The data in Figure 1 show that the addition of Hbγ-35 led to an increase of the TNF-α inducing capacity. No further increase was observable, enhancing the ratio from 1:1000 to 1:10,000 (LPS:Hbγ-35), probably owing to saturation of the system. In contrast, Pep19-2.5 reduced the induction of pro-inflammatory cytokines and inflammatory mediators triggered by LPS, also found in different cell types from the murine and human system.11,12 Moreover, neither Hbγ-35 nor Pep19-2.5 alone had an effect on the expression of TNF-α.
Influence of peptides Hbγ-35 and Pep19-2.5 on the ability of LPS to stimulate human MNCs. The cells were stimulated with S. Minnesota LPS R60 in the absence and presence of different concentrations of peptides Pep19-2.5 and Hbγ-35.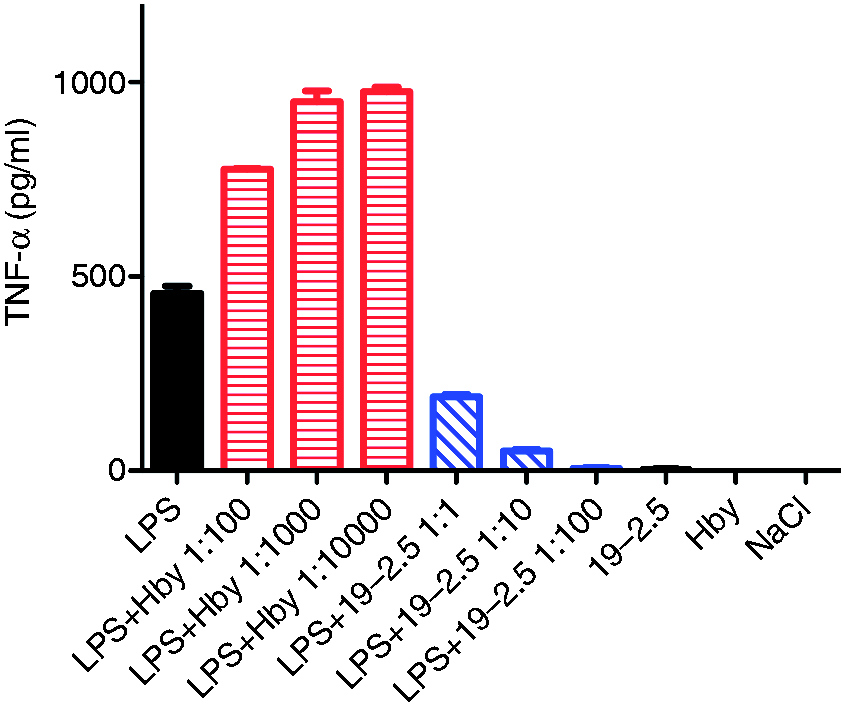
The two peptides lead to a different aggregation of LPS
LPS forms spontaneous aggregates in aqueous suspension,
30
which are strongly reduced in size by the addition of Hb.19,31 Taking into account the opposite immunomodulatory effect of the two peptides and to gain further insight into whether this effect might be related to the aggregation or disaggregation of LPS, we performed 90° light scattering experiments in a fluorescence spectrometer and recorded the signal of the titration of the peptides to LPS in dependence on time. The increase in scattering intensity at a wavelength of 400 nm is a sensitive measure of LPS aggregation. Figure 2 shows that LPS by itself scattered the light markedly (dot line) and the addition of both peptides had contrasting influence on the LPS scattering behaviour; although Hbγ-35 caused a considerable disaggregation of LPS, Pep19-2.5 led to an increase in aggregate sizes.
Change in the aggregation state of LPS after the addition of peptides. S. Minnesota LPS R60 was prepared as a 0.01-mM dispersion, and the influence of the Pep19-2.5 and Hbγ-35 on the light scattering signal at a wavelength of 400 nm was recorded versus time.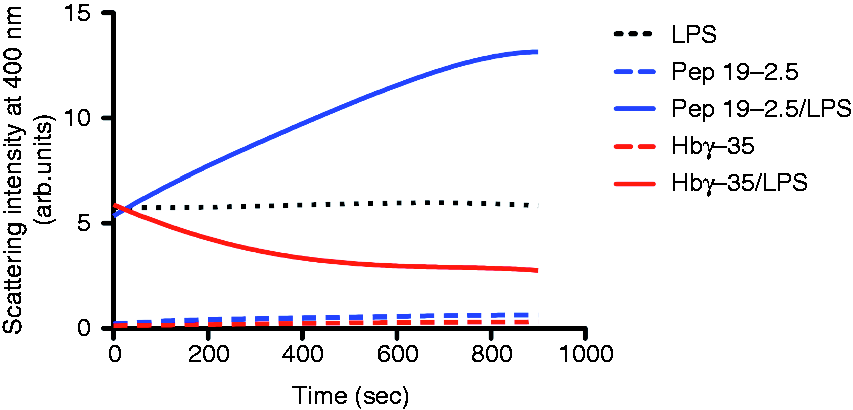
The two peptides differently influence the fluidity of the acyl chain of LPS
To obtain a better understanding of the influence of the peptides on the gel to liquid crystalline (β to α) phase transition of the acyl chains of LPS, Fourier-transform infrared spectroscopy was applied. In Figure 3, the peak position of the symmetric stretching vibrational band of the methylene groups from the lipid A moiety of LPS is plotted versus temperature for the two LPS:Hbγ-35 and LPS:Pep19-2.5 mixtures. As shown, the phase transition temperature (Tc) of LPS—from the ordered gel phase into an unordered liquid crystalline phase—was around 35℃, in accordance with published values.
32
The addition of Hbγ-35 induced a decrease in the acyl-chain fluidity (rigidification)—precisely increasing Tc by around 5℃. The effect induced by Pep19-2.5 was the opposite. At low concentrations the change of the wave number indicated a slight fluidisation of the LPS, but not a modification of Tc. At higher peptide concentrations, however, not only a fluidisation, but also a considerable decrease, in Tc took place.
Phase transition of LPS in the absence and presence of the peptides. (A) Hbγ35; (B) Pep19-2.5. Shown is the peak position of the symmetric stretching vibration of LPS R60, which is a sensitive measure of acyl chain order. In the gel phase, the peak positions are around 2850 cm−1, and in the liquid crystalline phase they are around 2852.5–2853.0 cm−1.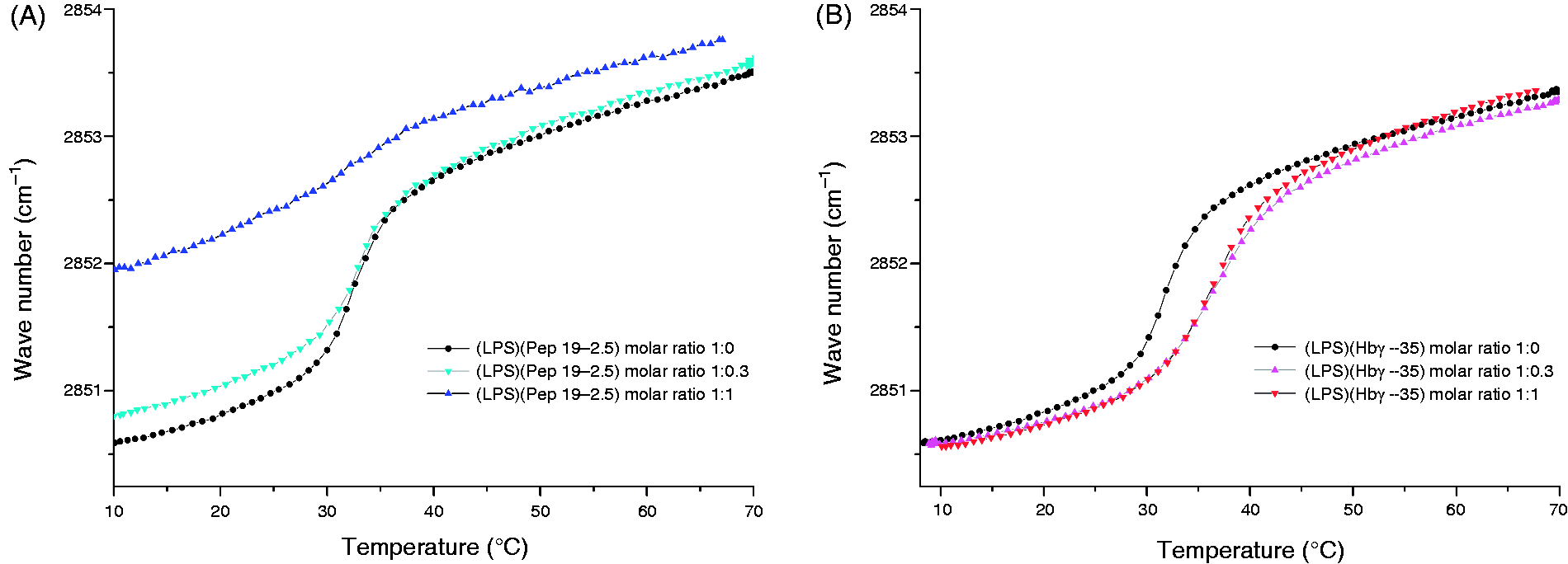
The two peptides differ in the peptide–LPS binding enthalpies
Given that the peptide charge and the hydrophobicity is important in the LPS interaction,
1
we determined whether the interaction reveals differences in LPS and the two peptides. We applied isothermal titration calorimetry (ITC) as a method to gain information about the strength of the interaction. As demonstrated in Figure 4, for both peptides the single titrations at 37℃ indicated negative enthalpy change ΔH values, corresponding to an exothermic reaction based on Coulomb interactions. For peptide Pep19-2.5, the binding curve describes a sigmoidal-shaped curve with clear saturation characteristics, implying a rather strong binding, whereas the interaction of Hbγ-35 led to a hyperbolic curve shape, indicating a weaker interaction.
Enthalpy change ΔH of the LPS:peptide interaction. (A) Pep19-2.5; (B) Hbγ35. LPS R60 was prepared at a concentration of 0.05 mM, and the peptides at a concentration of 1 mM were titrated in 5-µl portions to LPS. Recorded are the heat signals P (top) versus time (min), and the enthalpy changes ΔH versus [peptide]:[LPS] concentration ratios (bottom).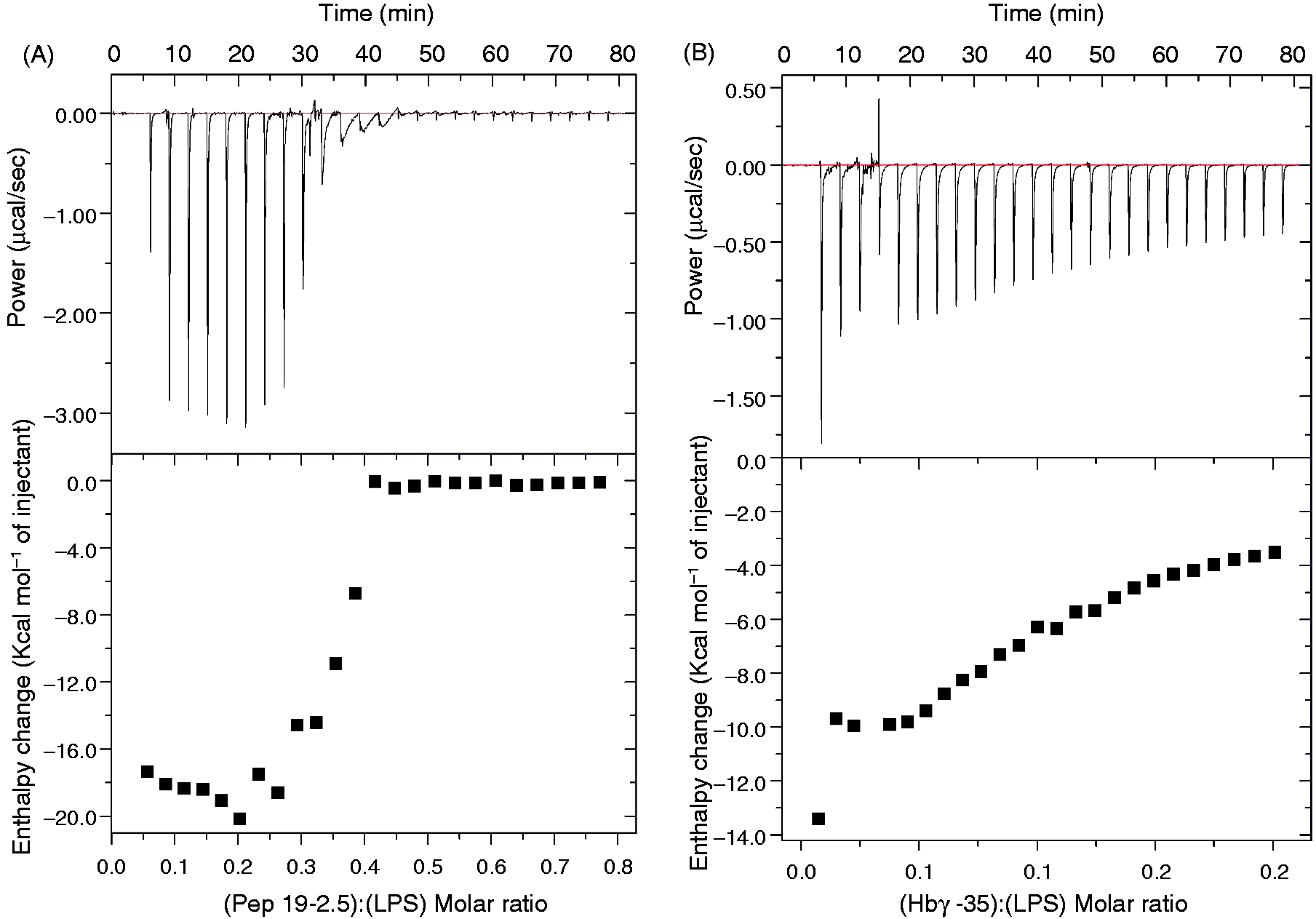
The two peptides differ in the secondary structure
As is known, the primary amino acid sequence of a peptide determines its folding. As presented above, Hbγ-35 and Pep19-2.5 differ in this sequence and in overall charge. Therefore, we determined the secondary structure of the peptides by using infrared ATR experiments of the peptides and LPS:peptide mixtures spread on a Ge crystal. The infrared spectra of proteins and peptides are commonly characterised by the absorbance bands of their amide groups. The commonly used amide I vibrational band is attributed to the combination of the C = O and C–N stretching vibrations.33–35 In Figure 5A, two distinct peaks are identified for both peptides. For Hbγ-35, α-helix and β-sheets were observed owing to the occurrence of bands at 1658.5 cm–1 and 1627.4 cm–1, respectively, while for Pep19-2.5 only β-structures were found [β-turns (1673.6 cm–1) and β-sheets (1629.5 cm–1). These secondary structures of the peptides were also observed in the presence of LPS (Figure 5B, C). The data clearly show that for Hbγ-35 (Figure 5B), as well as for Pep19-2.5 (Figure 5C), the secondary structures found in the absence of LPS are only slightly modified, that is their secondary structures are retained after binding to LPS in contrast to the drastic changes found for their reaction partner LPS.
Secondary structures of (A) the peptides alone and (B) Hbγ-35 or (C) Pep19-2.5 in the presence of LPS R60. Recorded are the infrared spectra in the range of the amide I-vibrational band between 1720 and 1600 cm−1, which show band components characteristic for α-helical structures in the range 1750–1760 cm−1, and for β-sheet structures from 1725 to 1740 cm−1 and β-turns from 1765 to 1775 cm−1.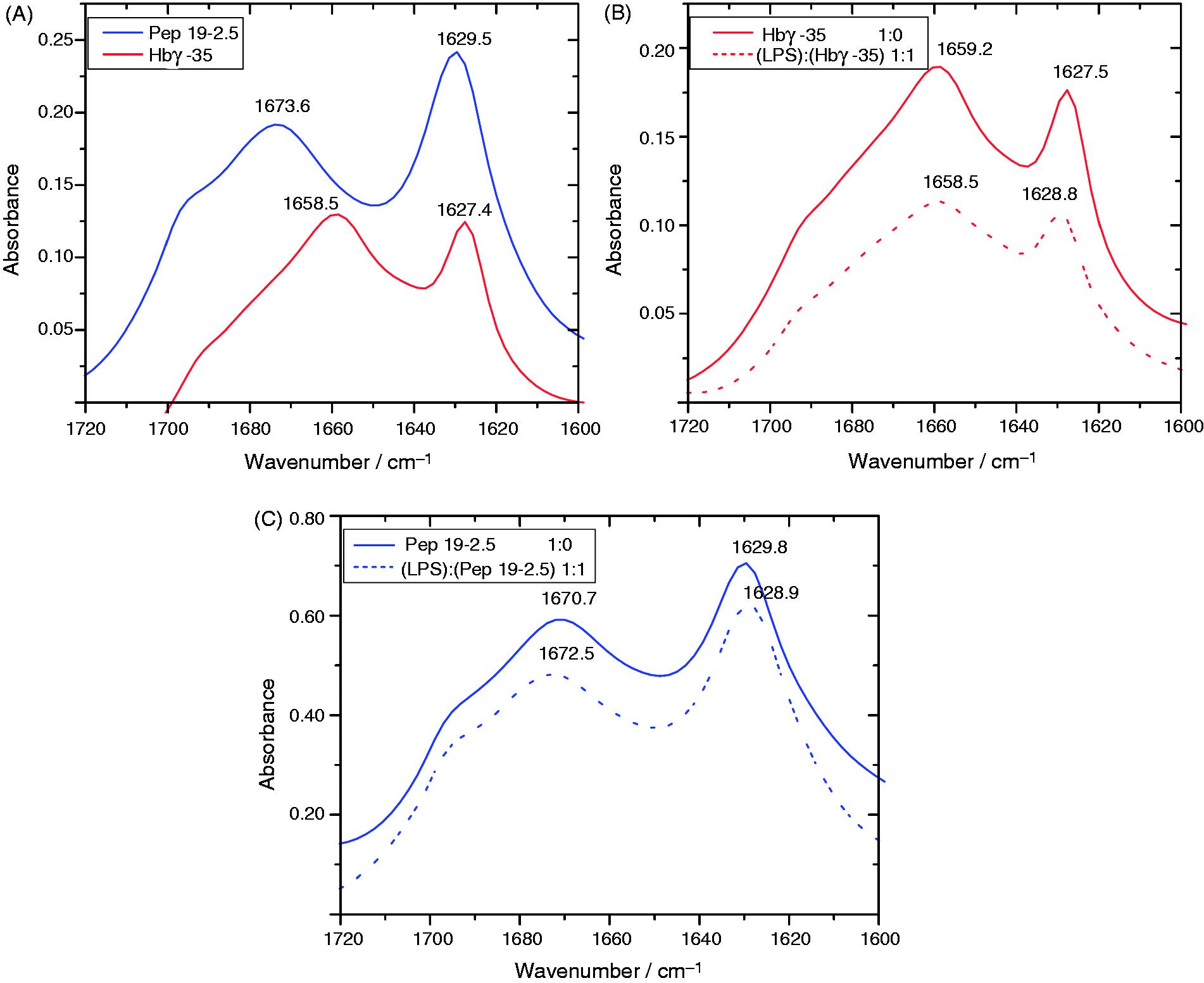
Discussion
We have characterised the biophysical mechanisms that are connected with the observation that the peptide Hbγ-35, derived from the Hb gamma chain, enhances the immune stimulatory property of LPS. A different peptide, Pep19-2.5, which has a similar length, neutralises LPS. It has been previously reported that the form of LPS aggregation, its endotoxin conformation, is responsible for the activation of the TLR4/MD2 signalling cascade and, consequently, determines its biological activity. In fact, the aggregate structure of LPS or lipid A with diverse chemical structures can adopt different types of aggregation, from multilamellar to unilamellar, mixed unilamellar/cubic and a pure nonlamellar cubic organisation. The latter has been described to be connected with the strongest activation of the human immune system. 3 It has been shown that highly biologically active LPS are converted from a cubic to a multilamellar form in the presence of various AMPs, such as polymyxin B, NK–lysin-derived peptide NK-2, lactoferrin (LF) and LF-derived peptides, as well as synthetic anti-LPS peptides SALP as Pep19-2.5,13,36–38 which directly corresponds to an inhibition of cytokine production.11,39,40 The interaction between Hb and LPS enhances the biological response, which was found to result from a conversion of LPS aggregates into a more pronounced nonlamellar, cubic aggregate structure and to a disaggregation.22,24 Furthermore, the diverging phase transition behaviour of bioactivity increasing and bioactivity decreasing compounds has also been reported for the biological weak or inactive pentaacyl lipid A and LPS preparations. Hb, for which a decrease in mobility of the hydrocarbon chains of LPS was also observed, stands in contrast to the system LPS and polymyxin B, which decreases the bioactivity and leads to a fluidisation at 37℃. 41 These findings are in direct accordance to the data presented here concerning the influence on the TNF-α production in MNCs by the two different peptides Hbγ-35 and Pep19-2.5 (Figure 1). The results from light-scattering experiments show that a disaggregation of the LPS samples by the former peptide takes place in contrast to the processes occurring with the latter, which clearly leads to an increase in aggregation (Figure 2). Both observations are in agreement with the described conformational concept of LPS activity, 42 that its increase correlates with nonlamellar small aggregates and its decrease with multilamellar large aggregates. 43
We found some differences in the secondary structures of both peptides. Hbγ-35 does not only adopt α-helices, as described for Hb in the literature, but it also adopts β-sheets.44,45 For Pep19-2.5, in contrast, only β-structures were observed, consisting of β-turns and β-sheets (Figure 5A). The peptides alone, irrespective of their secondary structure, did not cause expression of TNF-α when they were added to MNCs (Figure 1). These results coincide with those obtained by Kaconis et al. 13 for Pep19-2.5 and with those of Howe et al. 31 for Hb, which did not show any activity on the release of TNF-α. However, they are in contrast to result from another study by Carrillo et al., 21 where the addition of not only Hb plus LPS, but also Hb alone, enhanced TNF-α production in monocytes. These findings were not confirmed here, but may be explained by the observation that Hb purifications are more likely to be contaminated with LPS, which can be ruled out for the synthetic Hbγ-35 used here.
Interestingly, the addition of LPS to the peptides, which influences drastically the conformation of the endotoxin, only marginally influences the secondary structures of both peptides (Figure 5B, C), which was also found for other endotoxin binding partners such as LF and serum albumin.46,47
It is assumed that the negatively charged groups in the LPS inner core oligosaccharide and lipid A (mainly the phosphates in position 1 and 4’) interact with the cationic amino acids of MD-2, which can be an important step in the recognition of LPS by the TLR4/MD-2 system.48–50 Indeed, the negative LPS charges are targets for proteins and peptides, and modulate the endotoxin biological activity.51,52 As described, Pep 19-2.5,
13
NK–lysin-derived peptide NK-2 and lauryl-LF11 inhibit the LPS-induced cytokine release by the ability to overcompensate the surface charges of LPS aggregates.38,46 Although further research is required to determine whether Hbγ-35 directly binds to the phosphates or not, we can assume that there is an electrostatic binding between the negative charges of the LPS and the positive charges of the peptides due the exothermic reaction shown in the ITC experiments (Figure 4). However, the electrostatic interaction does not necessarily involve only the phosphates, but also the negatively charged 3-deoxy-
Beside the two important physical parameters, conformation and charge, the acyl chain fluidity and accessibility are crucial for the biological activity.42,53 These effects are superimposed and cannot be distinguished entirely. The fact that the addition of Hbγ-35 to LPS leads to a shift of Tc to higher temperatures (moderate rigidification; Figure 3) indicates that the peptide does not interact directly with the hydrophobic core of the LPS aggregate, as compared, for example, to endotoxin–neutralising peptides such as polymyxin B and Pep19-2.5, which have a strong influence on fluidity by decreasing the phase transition temperature and enthalpy (Figure 5).13,36 However, the parameter fluidisation of the acyl chains of the lipid A by itself does not correlate with the capacity of a molecule to block or enhance the stimulation capacity of LPS, but it can contribute to the modulation of the biological activity. In macrophages, the addition of ions such as Zn2+ reduces the acyl chain fluidity of LPS and the LPS-induced cytokine secretion (TNF-α and IL-1 b) is enhanced. 54 Also, the addition of human LF or LF-derived peptides leads to a reduction in the secretion of TNF-α, whereas the effect on the phase transition is different depending on the hydrophobicity of the peptides and the LPS used for the induction of fluidisation or rigidification of the acyl chains.46,55
In summary, we could demonstrate that the peptide Hbγ-35 is able to enhance the immune stimulatory effect of LPS, similar to the entire Hbγ chain and the complete Hb protein. Our results strongly support that LPS disaggregation induced by Hbγ-35 is the main factor in enhancing its biological activity, reinforcing the importance of LPS supramolecular structure in the cell activation process. In fact, when the disaggregation process is prevented, the macrophages can phagocytose the aggregates inhibiting the cytokine secretion. 56 Moreover, the LPS disaggregation by LBP or other proteins upon their transfer to CD14, and subsequently to the TLR4–MD2 complex or the MaxiK ion channel,57,58 is an important step. It is possible that the single LPS molecules within multilamellar stacks are more stable bound than within unilamellar/cubic structures. We have provided evidence supporting a model in which the disaggregation process by Hbγ-35 can facilitate the recognition structures in LPS necessary for receptor interaction. Another point to consider in this model is the binding affinity. Whereas Pep19-2.5 binds strongly to LPS aggregates, Hbγ-35 does not, supporting the idea that a strong binding of a peptide to LPS aggregates prevents LPS from binding to the cell receptors and hence inhibits cytokine secretion.
Although further work is needed to clarify how Hbγ-35 interacts with LPS, the development of Hbγ-35 may provide the opportunity to pave the way for this highly interesting immune stimulatory compound. A potential application could be the use of this peptide to design adjuvants for vaccination or resuscitation of the immune system.
Footnotes
Funding
Research at the laboratories of the authors is supported by the Else-Kröner-Fresenius Stiftung (project 2011_A140), the Clinique la Prairie, and the Department of Education of the Government of Navarra (Spain) for the Post-doctoral Fellowship within the “Anabasi+d Program (2010–2011), Outgoing model”.
Acknowledgements
The authors are indebted to Nina Hahlbrock and Christine Hamann for the excellent technical assistance with the cytokine production test system and light scattering experiments, respectively, as well as to Rainer Bartels and Volker Grote for the peptide synthesis. Pep19-2.5 patent OCT/EP2009/002565.
