Abstract
Interactions among major risk factors associated with bacterial infections were assessed in a model system using surrogates for virus infection; IFN-g, and exposure to cigarette smoke; cigarette smoke extract (CSE), nicotine and cotinine. Cytokine responses elicited by LPS from THP-1 cells in the presence of these components, or combinations of components, were assessed by multiplex bead assay, i.e. IL-1β, IL-6, IL-8, IL-10, TNF-α and IFN-γ. IFN-γ-priming significantly increased pro-inflammatory cytokines induced by LPS. CSE suppressed production of pro-inflammatory cytokines IL-1β, TNF-α and IFN-γ, but enhanced production of IL-8. Nicotine and cotinine suppressed all cytokine responses. In combination, IFN-γ masked the inhibitory effects of CSE. In relation to the objectives of the study, we concluded that (a) IFN-γ at biologically relevant concentrations significantly enhanced pro-inflammatory responses; (b) CSE, nicotine and cotinine dysregulated the inflammatory response and that the effects of CSE were different from those of the individual components, nicotine and cotinine; (c) when both IFN-γ and CSE were present, IFN-γ masked the effect of CSE. There is a need for clinical investigations on the increase in IL-8 responses in relation to exposure to cigarette smoke and increased pro-inflammatory responses in relation to recent viral infection.
Introduction
Both virus infections and exposure to cigarette smoke are risk factors for susceptibility to bacterial infection or severity of the host’s responses to infections. The combined effects of virus infection and smoking have not been explored thoroughly. Some studies assessed the effects of cigarette smoke in relation to viral or bacterial infections,1–4 but not in relation to virus-mediated effects on bacterial diseases. Raza et al. 4 assessed in vitro the effects of cigarette smoke and respiratory syncytial virus (RSV) infection on induction of TNF-α and NO responses from human peripheral blood monocytes. Cigarette smoke had no effect on TNF-α responses induced by RSV and toxic shock syndrome toxin (TSST). Three other studies investigated the effects of viral infection and cigarette smoke on inflammatory responses;5–7 IFN-γ was used as a surrogate for virus infection.
The objective of this study was to develop a model to assess effects of components of cigarette smoke and virus infection on inflammatory responses to a bacterial antigen LPS. These included IL-1β, IL-6, IL-8, IL-10, TNF-α and IFN-γ. In vitro stimulation of the THP-1 cell line was used as the model. The advantage of a cell line was the reduced effect of environmental factors (recent infection, diet, age or exposure to cigarette smoke) and genetic factors (cytokine gene polymorphisms, sex) on the inflammatory responses. 8
THP-1 cells were used as they are functionally similar to resident macrophages and peripheral blood monocytes and widely assessed for cytokine responses elicited by LPS. THP-1 cells do not normally express the CD14 antigen, which, in conjunction with TLR4, binds to LPS to initiate stimulation. To induce CD14, THP-1 cells were pre-treated with 1α,25-dihydroxyvitamin D3 (VitD3).9,10
Concentrations of LPS in other studies ranged from 1 µg/ml LPS to 10 pg/ml LPS.8,11,12 In animal models, virus infections have been demonstrated to enhance inflammatory responses to bacterial endotoxin 13 and exotoxin. 14 IFN-γ is reported to differentiate and activate THP-1 cells. 15 Pre-treatment with IFN-γ prior to stimulation enhanced inflammatory responses to LPS.16,17
Previous studies reported IFN-γ responses to viral infection in PBMCs ranging from ∼ 0.2 to 20 ng/ml at 5 × 105 PBMCs ml.15,18
IFN-γ concentration data in the literature is abundant; however, it could not be translated to the system as cell concentration is rarely reported. Studies that reported cell concentration varied significantly in their methodologies. Adjusted per 5 × 105 cells/ml, the highest concentration of IFN-γ used was 170 ng/ml (10-fold above biologically relevant range); the lowest was 0.02 ng/ml.15,19,20
The effects of cigarette smoke on inflammatory responses has been examined utilising cigarette smoke extracts (CSE). Concentrations of CSE used in previous studies ranged from 5 × 10−2 cigarettes (cig)/ml to 5 × 10−6 cig/ml. 21 Owing to significant variation in the toxicity of CSE, concentrations of chemicals in the preparation, duration of cell stimulation and other conditions, previous studies were used only as a guide. CSE can differ significantly in composition as a result of differences in preparation. 22 The chemical composition of CSE can vary with differences in the rate of smoking, type of cigarette(s) used, type of solvent and other downstream processes (i.e. filtration and freezing). CSE prepared in this study was partly quantified by measuring nicotine concentration, which allowed for comparison with other studies that reported the concentration of nicotine in the CSE. 23
Nicotine is one of approximately 4000 chemicals in cigarette smoke and has been widely assessed. It is associated with altered inflammatory responses. In studies assessing the effect of nicotine on inflammatory responses, concentrations ranged from 8 ng/ml to 8 µg/ml (adjusted for 5 × 105 cells/ml).24–27 Cotinine, the metabolite of nicotine produced in the liver, is often detected in body fluids as a marker of exposure to cigarette smoke and was included in the assessments.
The objective of the study was to develop a model for examining interactions between the surrogates for cigarette smoke and virus infections on inflammatory responses using non-toxic and biologically relevant concentrations of the components to be tested. The model was developed to test two hypotheses: (1) the surrogate for virus infection, IFN-γ, would—at biologically relevant concentrations—enhance pro-inflammatory cytokine responses to LPS; (2) the surrogate for exposure to cigarette smoke, CSE, would dysregulate cytokine responses to LPS, and the pattern of dysregulation would differ from that induced by nicotine alone.
Materials and methods
Approval for this study was obtained from the University of Newcastle Human Research Ethics Committee (Australia) (H3410597, H4601102).
All cell culture media and reagents were supplied by Invitrogen (Melbourne, Australia). FCS was heat-inactivated at 56℃ for 30 min. THP-1 cell growth medium ‘RPMI 10%’ consisted of RPMI 1640 supplemented with FCS (10% v/v); 2 mM glutamine; 1 mM sodium pyruvate; filter sterilised (0.22 µM; Millipore, Sydney, Australia); and stored at 4℃ for a maximum of 2 wk and pre-heated to 37℃ immediately before use.
Unless otherwise stated, all reagents were supplied by Sigma (St Louis, MO, USA) and were of analytical grade. All solutions were made up or diluted using MilliQ water (Millipore) and autoclaved if heat stable. Reagents were thawed immediately before use in the stimulation assays and diluted as required in RPMI 10%.
LPS
LPS from Escherichia coli 026:B6 was made up to 1 mg/ml in E-Toxate Water (Sigma, St Louis, MO, USA). Subsequent dilutions of LPS were made in RPMI 10% and stored in aliquots at −80℃. The highest concentration of LPS assessed for toxicity was 100 µg/ml; 10-fold serial dilutions were tested for toxicity and induction of cytokines from THP-1 cells.
Stimulation of THP-1 cells with vitamin D3
The method described previously was employed. 8 CD14 expression of vitamin D3 (VitD3)-differentiated THP-1 cells was confirmed by flow cytometry (Hunter Area Pathology Service, Newcastle, Australia).
Human recombinant IFN-γ
IFN-γ (1 mg/ml) (Oxoid #BR0014) was dissolved in PBS (PBS adjusted to pH 8.0 with NaOH according to manufacturer’s instructions). Subsequent dilutions were made in RPMI 10% and stored at −20℃. Ten-fold dilutions (10 µg/ml to 1 pg/ml) were assessed for toxicity to THP-1 cells.
CSE
A solution containing soluble (aqueous) components of mainstream cigarette smoke was prepared. The mainstream smoke of 20 cigarettes (16 mg tar) was bubbled through RPMI 1640 (200 ml) at room temperature using a vacuum pump. Five common commercial brands were combined to produce the final quantity. Each cigarette was lit and ‘smoked’ within 1 cm of the filter within 5 min using suction provided from a vacuum pump. The final solution of CSE (0.1 cig/ml) was filter-sterilised (0.22 µM; Millipore) and stored in aliquots at −80℃. Filtered CSE (0.1 cig/ml) was assessed for nicotine (20 µg/ml) and cotinine (0 µg/ml) levels by Leeder Consulting, Melbourne, Australia. Samples were frozen and shipped to Leeder Consulting on dry ice to avoid chemical degradation during transportation. To reduce the possibility of variation in the composition of CSE between experiments, a large batch was produced for all experiments and stored in aliquots at −80℃. Ten-fold dilutions of the stock CSE solution (0.1 cig/ml to 0.000001 cig/ml) were tested for toxicity.
Nicotine and cotinine
(-)–Nicotine free base was diluted to 10 mg/ml in molecular grade absolute ethanol. (-)–Cotinine was dissolved to 10 mg/ml with molecular grade absolute ethanol. Subsequent dilutions of nicotine and cotinine were made in RPMI 10%, stored at −80℃, and protected from UV and fluorescent lights to avoid degradation. Ten-fold serial dilutions from 1 mg/ml to 10 µg/ml nicotine and cotinine were assessed for toxicity in THP-1 cells.
THP-1 cell line
THP-1 cells were kindly provided by Dr Philip Hansbro, University of Newcastle, Australia. Cells provided by Dr Hansbro were originally purchased from the American Type Culture Collection, Manassas, VA, USA and were stored in 90% (v/v) FCS, 10% (v/v) DMSO (Ajax Finechem, Sydney, Australia), penicillin (100 U/ml) and streptomycin (100 µg/ml) at a concentration of 1 × 107 cells/ml.
THP-1 maintenance
A batch of cells sufficient to complete all experiments was harvested and stored in 1-ml aliquots in LN2. Cells were washed by centrifugation at 250 g for 5 min and resuspended in pre-warmed (37℃) RPMI 10%. Cells were maintained at log-phase growth (1 × 104 – 1 × 106 cells/ml) incubated in 5% CO2 at 37℃. Medium was renewed with pre-warmed RPMI 10% every 2–3 d. Cells were discarded after 18 wks of growth in log phase. Cells were washed and resuspended in fresh, pre-warmed RPMI 10%, and were plated into cell culture well-plates immediately prior to stimulation assays.
Mycoplasma contamination detection
THP-1 cells were tested for Mycoplasma contamination at intervals (or if contamination was suspected) using the MycoSensor PCR Assay Kit (Stratagene, La Jolla, CA, USA). Contamination was assessed in both cells and cell culture supernatants according to the manufacturer’s instructions.
Cell viability
A cell viability of >85%, determined by Trypan blue exclusion, was considered acceptable for all experiments.
Assessment of toxicity
Prior to optimisation, 10-fold dilutions of all stimulants were assessed for toxicity (<85% viable cells) with VitD3-stimulated THP-1 cells. Toxicity was assessed at 0, 6, 12, 24 and 48 h after stimulation. The lowest concentration that induced toxicity was noted. Toxicity of stimulants was also assessed in combinations; however, this was only performed for optimised conditions.
IFN-γ induced adherence of cells. Attached cells were removed by incubation for 20–25 min at 37℃ with 10× trypsin-EDTA (0.5% v/v trypsin with EDTA 4Na). Cells were washed in RPMI 10% and resuspended in original cell volume and assessed for viability.
Stimulation assay
All experiments were carried out with VitD3-stimulated THP-1 cells at a final concentration of 5 × 105 cells/ml. Negative control wells contained equivalent volumes of RPMI 10% to replace individual components: LPS, IFN-γ or CSE (or nicotine or cotinine). At the end of each experiment, supernatants were collected by centrifuging cell culture plates at 250 g for 5 min. The supernatants were collected and immediately stored at −20℃. Assays were performed in triplicate.
Optimisation of LPS
The highest non-toxic concentration of LPS (100 ng/ml) was initially assessed over 48 h to observe cytokine responses. The time point that best represented the peak of the response was used for dose response experiments. Ten-fold dilutions of LPS (100 pg/ml–100 µg/ml) were assessed for cytokine responses at 6 h post-stimulation. The concentration that produced the 50% maximal cytokine response was selected for subsequent experiments. A time course assay over 48 h was carried out with the optimum concentration of LPS. The peak time point of the LPS-induced cytokine response was selected as the end-point for the experimental model.
Optimisation of IFN-γ and CSE
Optimal IFN-γ and CSE concentrations were determined with optimal concentrations for LPS. Biologically relevant concentrations of IFN-γ (0.1 ng/ml, 1 ng/ml and 10 ng/ml) were added to cells at 0, 6 and 24 h prior to LPS stimulation to investigate the effect of priming. Concentration and duration of IFN-γ stimulation that represented optimal enhancement of LPS-induced cytokine responses and cell adherence were selected.
To test the effect of CSE on responses to LPS, CSE (10−5 cig/ml, 10−4 cig/ml or 10−2 cig/ml) was added to cells at 24, 6 and 0.5 h prior to LPS stimulation. Before addition of LPS, cells were washed with fresh RPMI 10% to remove CSE. Co-stimulation was also investigated by adding CSE and LPS at the same time point.
Conditions of stimulation for nicotine and cotinine were not optimised for the experimental model. They were added to the system based on levels of nicotine in CSE.
Analysis of cytokine response
Cell culture supernatants were measured for IL-1β, IL-6, IL-8, IL-10, TNF-α and IFN-γ using a six-plex bead-based immunoassay kit (Bio-Rad, Hercules, CA, USA) performed according to manufacturer’s instructions. Bead median fluorescence intensity was detected with the Luminex 200 (Luminex, Austin, TX, USA) using the high RP1 setting. Cytokine concentrations (pg/ml) of unknowns were calculated from the standard curve using the Luminex 2.3 Software (Luminex). Duplicates with a co-efficient of variance (CV) higher than 20% were excluded from the analysis.
The minimum detection limit of the assay was assigned by the Luminex analyser and varied for each assay for each cytokine measured. For samples in which cytokine was not detected, i.e. ‘<0.19’, a value of zero was assigned. Samples with a CV >20% with an average value <10 pg/ml were considered acceptable for comparative analysis.
To eliminate variability between assays, only two different lots of beads were used for data presented. To assess variability of each run, an internal control was included. This was generated by stimulating THP-1 cells (5 × 105 cells/ml) with LPS (100 ng/ml) and IFN-γ (10 ng/ml) for 6 h.
Variability of stimulation assay
Variability of the experimental model was assessed by examining cytokine degradation over time, variability of cytokine detection method, and inter-well, inter-plate and inter-day variability of cytokine production. To assess cytokine degradation and inter-well variability, triplicate samples of THP-1 cells were stimulated with LPS (100 ng/ml) for 6 h. The supernatant from each well was collected and aliquoted into four separate tubes labelled 0 min, 30 min, 60 min and 90 min. Each tube was stored at −20℃ at the assigned time points and assessed for cytokine responses. Variability between triplicate wells for each of the time points was also assessed to investigate inter-well variability.
Statistical analysis
Comparisons were made only for samples tested on the same Luminex run. Data were analysed using STATA v. 10.0. The Wilcoxon matched pairs signed ranks test was used to assess differences in cytokine responses between treatment groups. The Kruskall–Wallis test was used to assess differences between more than two treatments groups. The significance level for all tests was set at P < 0.05. Statistical tests were not performed on dose and duration response data as there was not sufficient sample size. Linear regression analysis on the log-transformed data was used to assess the effects and interactions of LPS, IFN-γ, CSE, nicotine and cotinine on cytokine responses. The significance level for all tests was set at P < 0.05. To enable linear regression on log-transformed data, samples with undetectable levels of cytokine were assigned a value other than zero, equivalent to the lowest detectable concentration for that cytokine.
Results
VitD3 differentiation of THP-1 cells
Flow cytometry confirmed THP-1 cells incubated with VitD3 expressed CD14 (96.1%); the ethanol control was negative (0.0%). As in previous studies, 8 analysis of cytokine response to LPS confirmed that VitD3-stimulated THP-1 cells had significantly higher cytokine responses elicited by LPS compared with undifferentiated cells. IL-10 was not detected. All further experiments were carried out with VitD3-stimulated THP-1 cells.
Toxicity of stimulants
Toxicity was defined as fewer than 85% viable cells. LPS induced toxicity at 1 µg/ml after 6 h. IFN-γ at the highest concentration tested (10 µg/ml) did not induce toxicity. CSE was toxic at 0.01 cig/ml after 24 h. The addition of 1 ng/ml LPS to 0.01 cig/ml CSE accelerated the toxic effect from 24 h to 6 h. There was no toxicity observed for 0.001 cig/ml CSE, 100 ng/ml LPS or 100 ng/ml IFN-γ.
The combination of LPS (100 ng/ml), IFN-γ (100 ng/ml) and CSE (0.001 cig/ml) were not toxic at 48 h. IFN-γ (100 ng/ml) on its own induced cell attachment at 48 h post-stimulation. The time needed for attachment of cells to the wells was reduced by addition of LPS (100 ng/ml) from 48 to 6 h. The addition of CSE (0.001 cig/ml) delayed attachment by LPS and IFN-γ-treated cells to 24 h.
Nicotine and cotinine were assessed for toxicity at concentrations equivalent to the level of nicotine in the optimised concentration of CSE (0.0001 cig/ml and 0.001 cig/ml). Nicotine and cotinine (20 ng/ml and 200 ng/ml) were not toxic.
Cytokine degradation
There was no difference in cytokines detected in supernatant aliquots frozen within the 30 min intervals tested following collection (up to 90 min).
Inter-plate Luminex variability
Luminex plate variability was assessed by internal control samples. Internal control variability, reported as CV (%), ranged from 27% (IL-1β) to 56% (IFN-γ). CVs were reduced when controlled for assay lot number.
Optimisation of dose and duration of stimulation
The cytokine response of THP-1 cells to LPS (100 ng/ml) was originally assessed at 0, 6, 12, 24 and 48 h post-stimulation. The time point for optimal detection of the cytokine response was 6 h and was selected for the end-point of the LPS dose response assays.
The lowest concentration of LPS that initiated a cytokine response was 1 ng/ml with the response peaking at 100 ng/ml. The 50% maximal cytokine response was detected between 1 and 100 ng/ml for cytokines tested.
Cytokines were assessed over a 48-h period: every 2 h for the first 14 h then at 18, 22, 24, 28, 32, 36, 42 and 48 h. With the exception of TNF-α, which peaked at 2 h and declined rapidly (6 h), the other cytokines remained elevated: IFN-γ peaked at 6 h (50% at 48 h); IL-1β peaked at 24 h (75% at 48 h); IL-6 peaked at 10 h (50% at 48 h); IL-8 peaked at 8 h (50% at 24 h).
IFN-γ time course and dose response
Pre-treatment of cells with IFN-γ (10 ng/ml) for 24 h elicited the highest cytokine responses to LPS (50 ng/ml).
CSE time course and dose response
CSE alone produced no detectable cytokine response. When CSE was incubated with LPS there was little variation in cytokine responses between the different incubation periods. The longer CSE stimulation (i.e. 24 h pre-treatment and 12 h co-stimulation with LPS) produced the highest cytokine response. Cells pre-treated with CSE for 0.5 h and 6 h produced cytokine responses similar to LPS alone. There was a weak dose response with CSE across the different conditions tested. IL-8 was suppressed at higher CSE concentrations, while IL-1β and IFN-γ were increased. There was no clear trend with TNF-α. IL-6 was not detected in response to LPS or stimulation with LPS and CSE.
Effect of LPS, IFN-γ and CSE on cytokine responses
The effects of LPS, IFN-γ and CSE on cytokine responses were assessed in three separate experiments. Although triplicate samples were collected for each condition for all three experiments, only one supernatant for the first two experiments and two supernatants for the final experiment were tested; four supernatants were tested for each experimental condition. All data for the effects of nicotine and cotinine were produced from the third experiment. Figures depict mean values with SEM. Interactions of LPS and IFN-γ, LPS and CSE, IFN-γ and CSE, and the combination of all three LPS and IFN-γ and CSE were investigated. Two concentrations of IFN-γ, low (L) (1 ng/ml) and high (H) (10 ng/ml), and CSE, low (L) (0.0001 cig/ml) and high (H) (0.001 cig/ml), were assessed in the experimental model.
Compared with control cells incubated with growth medium alone, LPS induced IFN-γ, IL-1β, IL-8 and TNF-α. IL-6 and IL-10 were not detected.
Compared with unstimulated control cells, IFN-γ and IL-8 were elicited by the lower concentration of IFN-γ. The high concentration of IFN-γ enhanced IFN-γ detected in the samples and elicited IL-1β, IL-6, IL-8 and TNF-α. Responses from cells treated with higher level of IFN-γ were significantly higher than those treated with the lower concentration: IL-6 (P = 0.02), IFN-γ (P = 0.02), IL-8 (P = 0.02).
CSE alone at either high or low concentration elicited no cytokine responses. The baseline level of IL-8 (∼50 pg/ml) produced by THP-1 cells was slightly inhibited (30–40 pg/ml) by both concentrations of CSE; the lower concentration of CSE elicited significantly lower IL-8 responses (P < 0.05).
Effect of IFN-γ on cytokine responses of THP-1 cells to LPS
When LPS was added to cells pre-treated with either high or low concentrations of IFN-γ, there was a significant increase in production of IL-1β, IL-6, IL-8 and TNF-α compared with LPS alone (P < 0.02 for all cytokines for both concentrations of IFN-γ) (Figure 1). In contrast to stimulation with LPS alone, IL-6 was elicited from cells exposed to both IFN-γ and LPS.
Effect of priming with IFN-γ (1 or 10 ng/ml) on cytokine responses of THP-1cells to LPS. *P ≤ 0.05–0.01*.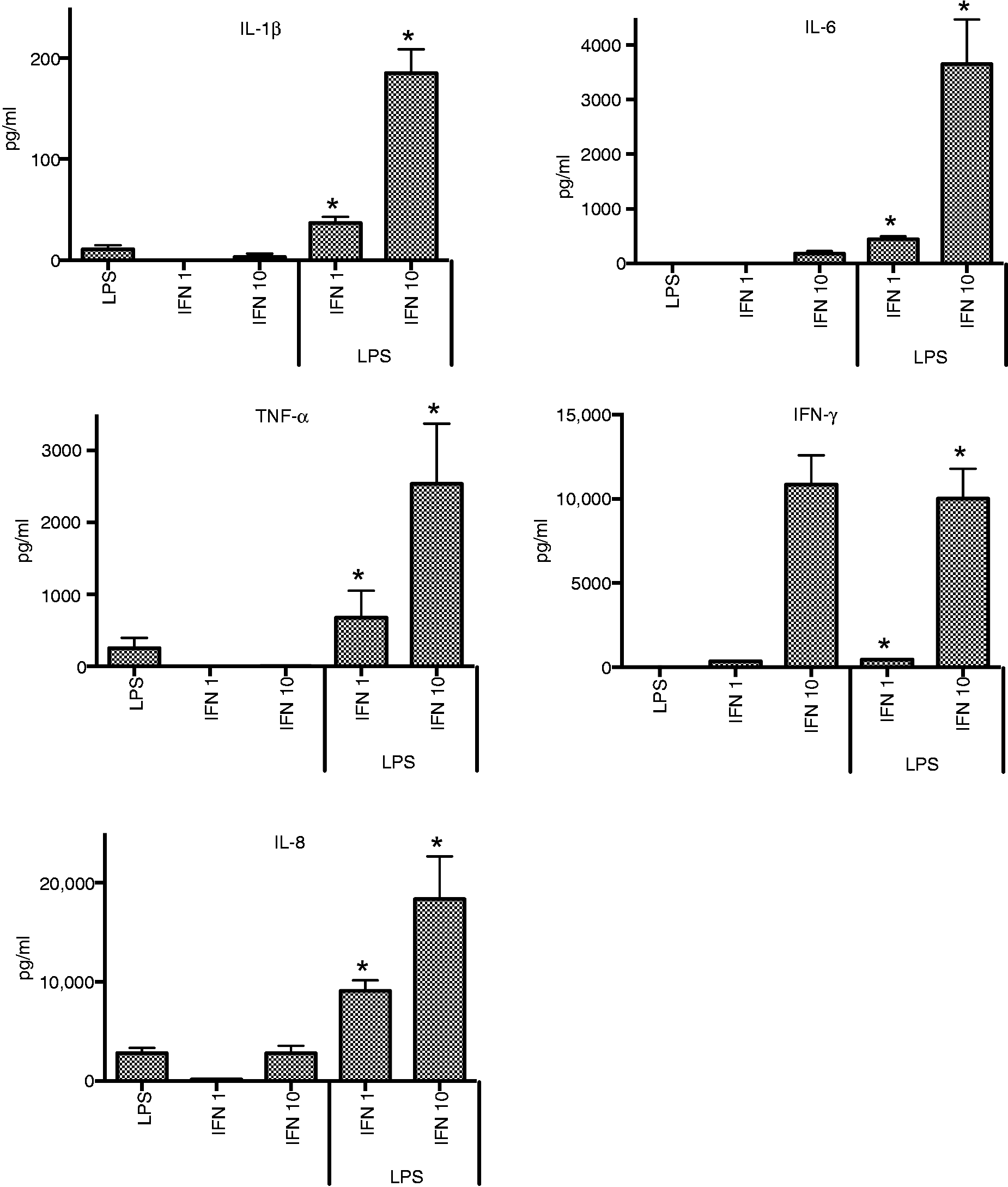
Linear regression analysis determined that there was a significant interactive effect between LPS and low level IFN-γ treatment: IL-1β (1.45, P < 0.001); IL-6 (4.55, P < 0.001); TNF-α (2.48, P < 0.001); and IL-8 on the additive scale (4,555.50, P = 0.001). IL-8 responses had a negative coefficient on the log-scale. For cells treated with LPS and high level IFN-γ, the interactions were also enhanced for IL-1β (3.09, P < 0.001), IL-6 (3.46, P < 0.001), TNF-α (4.22, P < 0.001) and IL-8 (interaction only detected on the additive scale 12,358.46, P < 0.001).
Effect of CSE on cytokine responses of THP-1 cells to LPS
LPS-induced cytokine responses from THP-1 cells were not significantly affected by CSE; IL-8 production was increased, but IFN-γ, IL-1β and TNF-α responses were decreased (Figure 2). Effects were greater for the higher dose of CSE. IL-6 was not detected. Linear regression analysis determined that there was a significant interactive effect between LPS and CSE cytokine responses. Interactive effects were observed for IL-8 for the low dose of CSE (0.91, P < 0.001).
Effect of CSE (L = 0.0001 cig/ml, H = 0.001 cig/ml) on cytokine responses of THP-1 cells to LPS.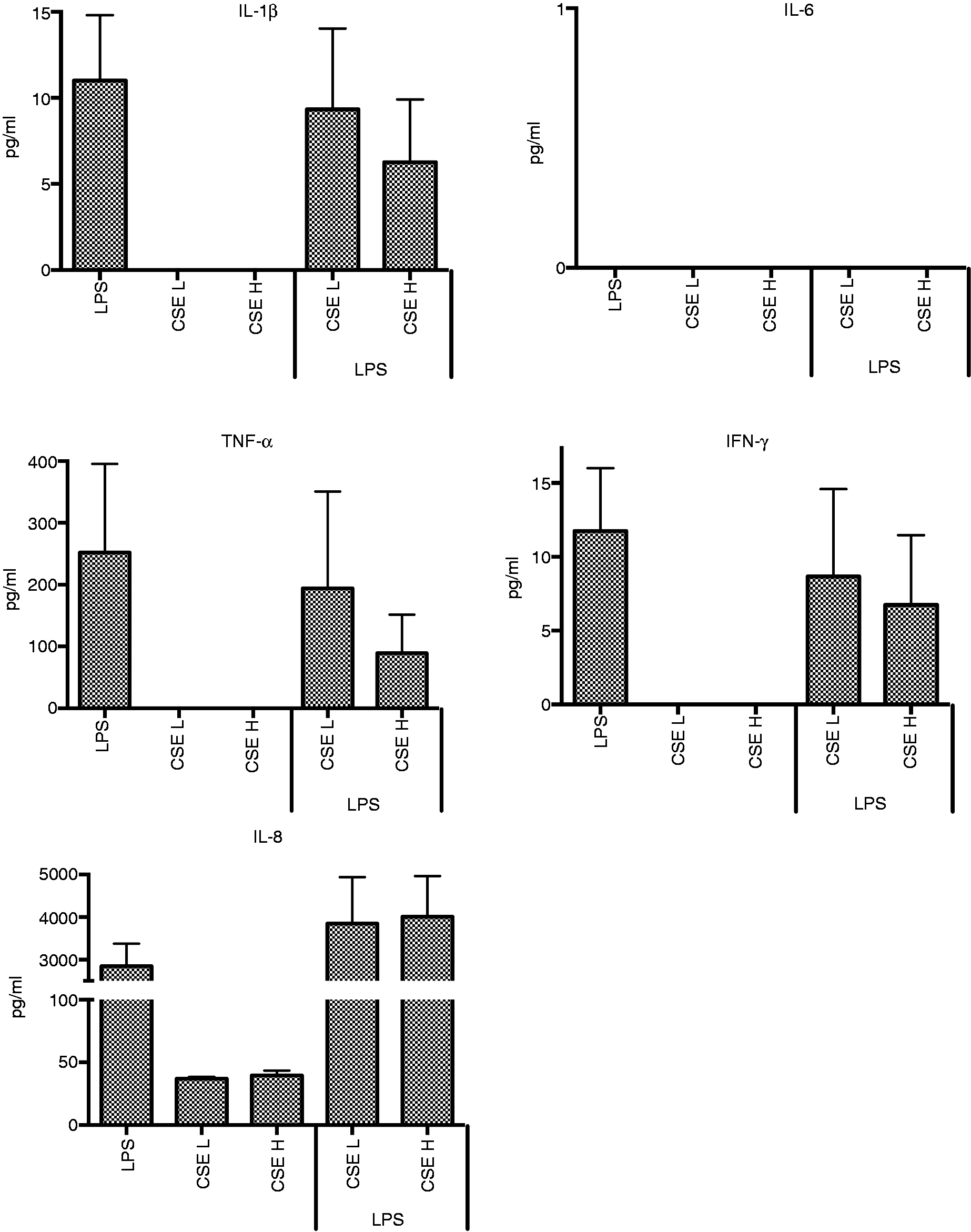
Effect of CSE on cytokine responses of THP-1 cells to LPS and IFN-γ
The only effect of CSE on cytokine production by THP-1 cells to LPS following pre-treatment with low IFN-γ was observed for IL-8; IL-8 was increased for the high dose of CSE (P = 0.03). There were no significant effects of CSE on cytokine responses to LPS following pre-treatment with the high dose IFN-γ. Although not statistically significant, a trend for increased IL-8 responses was observed for the high dose of CSE.
Effect of IFN-γ on cytokine responses of THP-1 cells to LPS and CSE
The effects of IFN-γ on cytokine responses to LPS and either high or low levels of CSE were similar to those observed for cytokine responses to combinations of LPS and IFN-γ (Figure 3). The effects of low IFN-γ on cytokine responses elicited by LPS and CSE were significant for all cytokines except TNF-α (P < 0.05).
Effect of CSE (C) (0.001 cig/ml) and IFN-γ (L = 1 ng/ml, H = 10 ng/ml) on cytokine responses of THP-1 cells to LPS. *P ≤ 0.05–0.01*.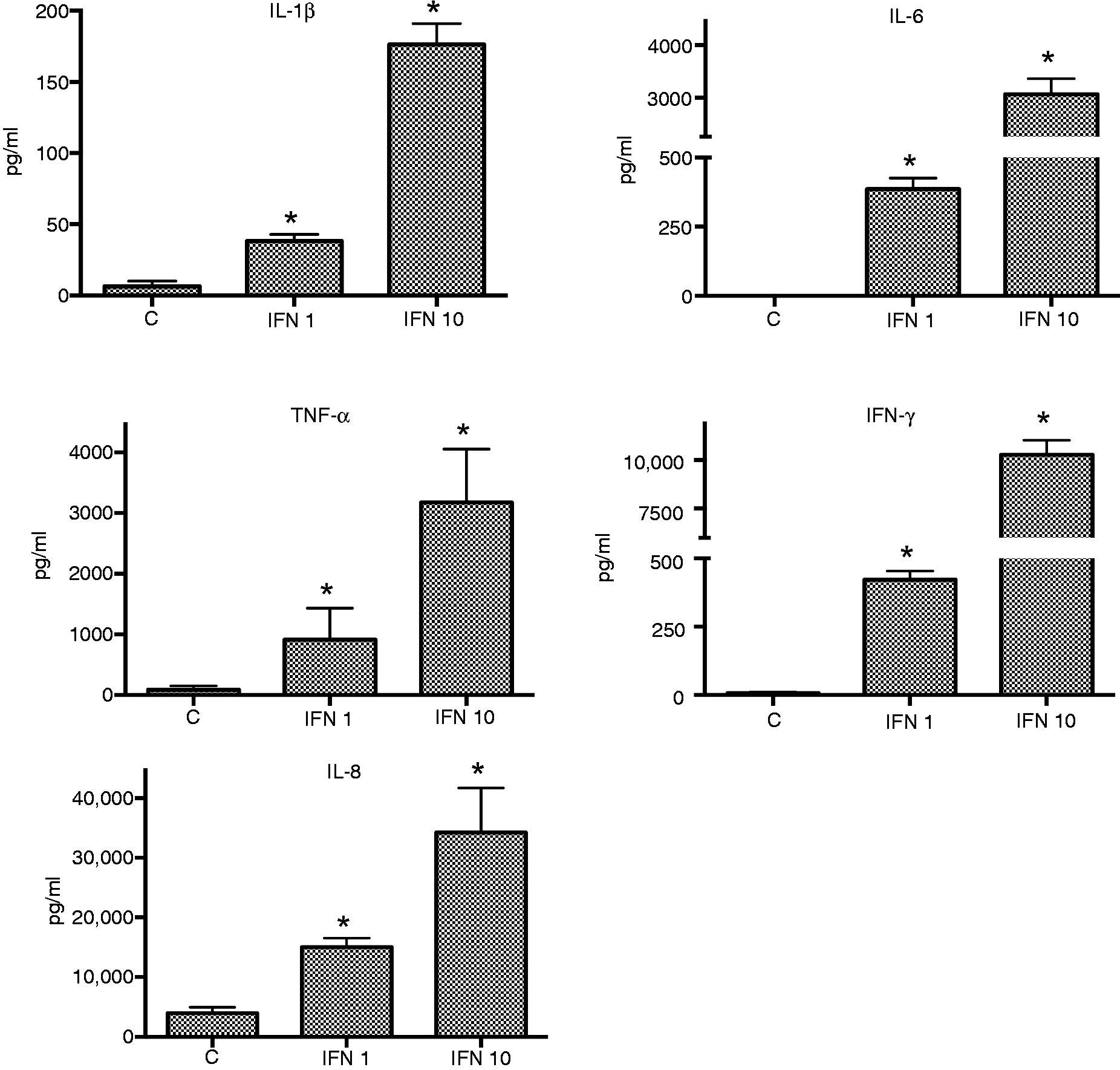
Effect of nicotine or cotinine on cytokine responses from THP-1 cells
The effect of nicotine and cotinine were assessed in comparison to CSE. The data for nicotine, cotinine and CSE were obtained from samples from the same experimental plate and day. Only one experiment was performed and tested in triplicate. Two of the triplicate samples were assessed for cytokine production. High variation was observed between duplicates for some samples, resulting in high SEM. The effect of nicotine and cotinine on LPS and IFN-γ induced cytokine responses differed from those observed with CSE.
As observed for CSE, nicotine and cotinine alone did not induce detectable levels of cytokine. Nicotine and cotinine inhibited the cytokine response to LPS in most experimental conditions, and this effect was enhanced at the higher dose.
Effect of nicotine or cotinine on cytokine responses of THP-1 cells to LPS
Nicotine and cotinine completely inhibited LPS-induced IFN-γ, IL-1β, and TNF-α responses; IL-8 was partially inhibited. A weak dose response for IL-8 suppression was also observed.
In comparison, CSE only partially inhibited the production of LPS-induced IFN-γ, IL-1β and TNF-α. IL-8 production was increased by CSE, but decreased by nicotine and cotinine. The effects of CSE, nicotine and cotinine observed for IL-8 production were greater at the higher doses.
In combination with data from the previous experiments, linear regression analysis determined that nicotine (200 ng/ml) was a significant predictor in the linear model: IL-1β (–0.65, P = 0.013); TNF-α (–0.97, P = 0.036). No significant predictors were identified for cotinine (20 ng/ml and 200 ng/ml) and the lower dose of nicotine. No interactive effects were observed for nicotine (200 ng/ml).
Effect of nicotine or cotinine on cytokine responses of THP-1 cells to LPS and IFN-γ
There was very high variation among the results for the effects of nicotine and cotinine on cytokine responses of THP-1 cells to LPS and pre-treatment with the lower IFN-γ dose. Despite the variation, distinct differences between the effects of nicotine/cotinine and CSE were identified. Nicotine and cotinine suppressed IL-1β and IL-8; CSE had no or little effect on IL-1β, but it enhanced IL-8 production. For cells pre-treated with the higher level of IFN-γ, at the higher doses nicotine and cotinine had a clear suppressive effect on IL-1β and IL-8 responses. In contrast, CSE enhanced IL-8 responses; IL-1β responses were unaffected (Figure 4).
Effects of nicotine (Nic) (L = 20 ng/ml, H = 200 ng/ml), cotinine (Cot) (L = 20 ng/ml, H = 200 ng/ml) or CSE (L = 0.0001 cig/ml, H = 0.001 cig/ml) on cytokine responses to THP-1 cells primed with IFN-γ (10 ng/ml) to LPS.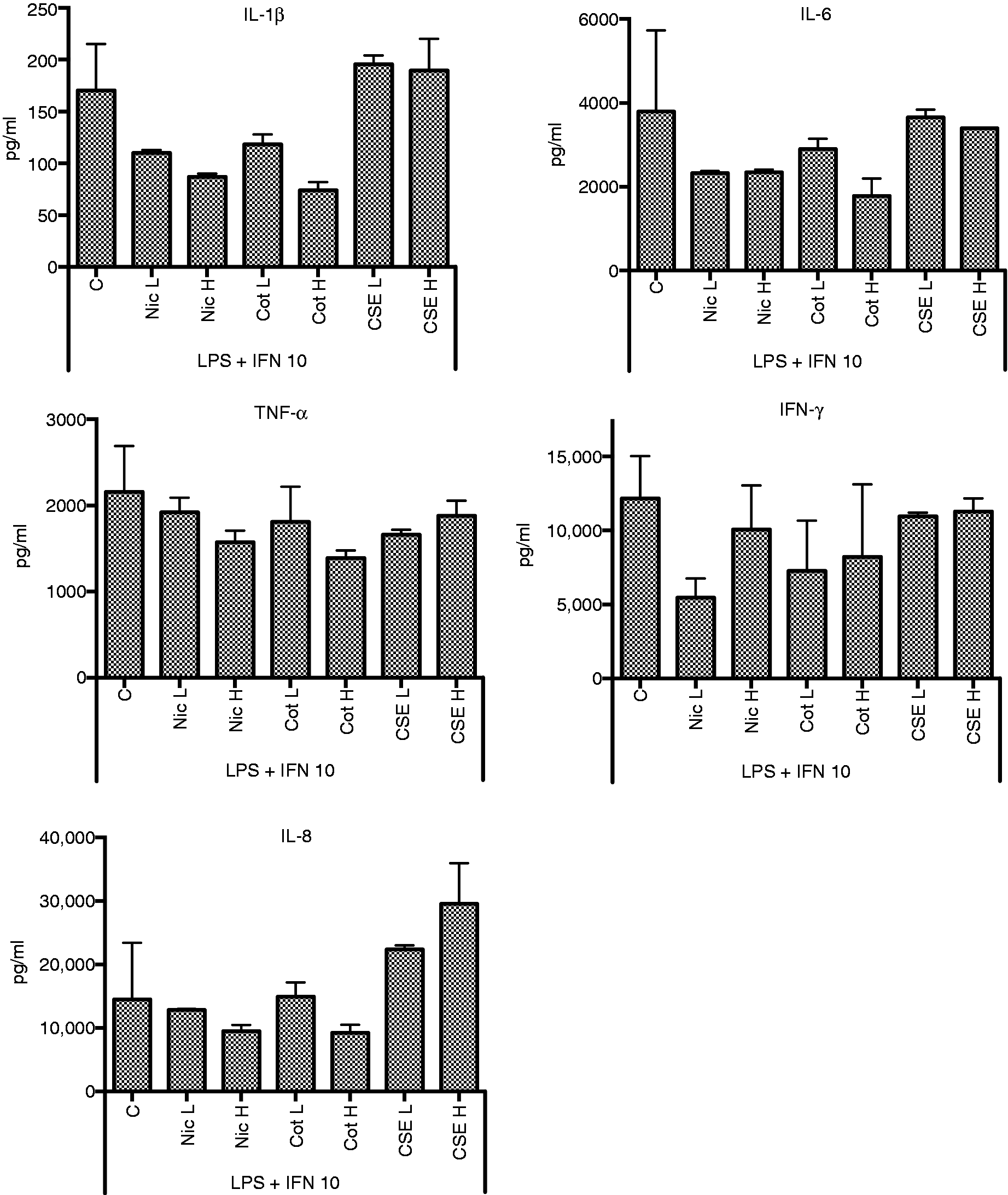
Discussion
The aims of the study were to develop an in vitro experimental model for assessment of inflammatory responses to LPS to examine the interactions between the surrogates for cigarette smoke and virus infection on these responses; to determine the reliability of the model by assessing cytokine stability and variation within the model; and to determine the optimal biologically relevant doses of the components of the system and duration of stimulation.
Limitations of the model system
Optimisation of the experimental model was performed with small numbers of samples; however, trends observed for the effect of treatment dose and time responses were confirmed in subsequent experiments. A limiting factor of the experimental model was the reduced ability to detect low levels of cytokine. When assessing the dose and time response, LPS stimulated approximately 10 pg/ml of IL-6. When LPS-induced IL-6 responses were assessed in additional experiments, this cytokine was not detected at the dilution (1 in 4) tested. The use of the THP-1 cell line in the experimental model also proved to be limited. Despite the stimulation by LPS, IFN-γ and/or CSE, the cell line did not produce the anti-inflammatory cytokine IL-10.
Optimal dose and duration of stimulation
As reported earlier, VitD3 induced expression of CD14 and significantly enhanced the cytokine response to endotoxin stimulation of cytokines from THP-1 cells. 8
LPS (50 ng/ml) was selected as the optimum concentration to initiate detectable production of all cytokines. Although this concentration approached the maximum cytokine response induced by LPS, priming the cells with IFN-γ elicited significantly higher cytokine production. The highest cytokine responses were induced by LPS (50 ng/ml) at 12 h from cells primed with IFN-γ (10 ng/ml) for 24 h prior to stimulation with LPS. These experimental conditions were adopted for all subsequent experiments.
Dose–response effects were observed with CSE with LPS. Previous articles indicate that short stimulation periods are necessary to mimic the effects of smoking; cells were exposed for 15 min–1 h and washed.28,29 This approach did not take into account the effects of long-term smoking or passive exposure to cigarette smoke. Other studies incubated cells for up to 72 h.23,30 Regardless of duration of stimulation or removal of CSE, most studies in which CSE was added prior to the addition of LPS indicated a possible priming effect of CSE. In our model, CSE was added at 3 h prior to LPS. While there was a dose-dependent effect of CSE on cytokine responses, the higher concentration of CSE was utilised in the experimental model. Concentrations of CSE within the optimised range have been frequently assessed in other studies assessing the effects of cigarette smoke on inflammatory responses.
The effects of surrogates for virus infection and exposure to cigarette smoke on cytokine responses of THP-1 cells to LPS
This was the first study to investigate the interactions between IFN-γ and CSE on inflammatory responses induced from THP-1 cells by endotoxin. Only one other study that has assessed the effect of virus infection and exposure to cigarette smoke on cytokine responses elicited by TSST; and only TNF-α and NO were assessed. 31
The cytokine responses induced by LPS and IFN-γ detected in this study were similar to those previously reported.32,33 IFN-γ significantly enhanced the production of LPS-induced pro-inflammatory cytokines, and responses were higher compared to IFN-γ or LPS alone. Linear regression analysis also confirmed an interactive effect between the predictors LPS and IFN-γ. In relation to viral infection, these findings confirm that concurrent or recent virus infection can significantly enhance pro-inflammatory responses to bacterial antigens.
This study identified that CSE non-significantly suppressed the production of pro-inflammatory cytokines IL-1β, TNF-α and IFN-γ; it enhanced significantly the production of IL-8. The suppression of IL-1β, TNF-α and IFN-γ responses,21,28 and increased IL-8 production34,35 by CSE have been reported previously. Similar results have also been detected in other cell lines or THP-1 cells using stimulants other than LPS. Paul-Clark et al. 36 are the only other group to identify increases in LPS induced IL-8 production from THP-1 cells.
Increased production of IL-8 has been previously reported in smokers in vivo34,37,38 and has been reported to result in increased neutrophil recruitment to the lung epithelial tissue. Reduced activity of recruited neutrophils and circulating leukocytes of smokers have been reported. 39 It is unknown if suppression of other pro-inflammatory cytokines (IL-1β, TNF-α, IFN-γ) is linked to leukocyte inactivity and/or the reduced ability to fight infection. It is unknown if the increased production of IL-8 could result in increased severity of infection in smokers. In this study, it was demonstrated that cytokine responses to LPS are dysregulated in the presence of components of cigarette smoke.
The effect of both IFN-γ and CSE on responses to LPS was of particular interest in this study. The addition of IFN-γ to the model resulted in statistically significant increases in cytokines induced by LPS and CSE. The addition of IFN-γ masked the inhibitory effects of CSE on LPS responses. The impact of IFN-γ on cytokine responses induced from the cells by LPS and CSE were similar to the effect of IFN-γ alone on LPS-induced responses. The addition of CSE to the model did not have any major effects on cytokine responses induced by LPS and IFN-γ. Only one statistically significant effect was observed by the addition of high-dose CSE to low-dose IFN-γ and LPS: IL-8 production was increased. A similar trend for increased IL-8 was also observed for the high-dose CSE and high-dose IFN-γ and LPS. These findings are also consistent with the effects of CSE on responses to LPS (without IFN-γ) reported in this and other studies.34–36 The results suggest a need for clinical investigations on increased IL-8 responses in relation to exposure to cigarette smoke and increased pro-inflammatory cytokines in relation to previous or concurrent viral infection.
The addition of nicotine or cotinine to the model resulted in complete or partial inhibition of cytokine production, including suppression of IL-8. In contrast, CSE partially inhibited some pro-inflammatory cytokines and enhanced the production of IL-8. Similar effects of nicotine and cotinine were observed in relation to responses induced by LPS from IFN-γ primed cells; all cytokines examined were reduced, including IL-8. The enhanced IL-8 responses in the presence of CSE require further investigation in relation to its role as a chemoattractant for polymorphonuclear leukocytes in relation to smoking-related lung disease.
In conclusion, the model developed was used to assess the two hypotheses: that environmental factors, such as virus infection, and exposure to cigarette smoke could significantly alter pro-inflammatory responses to bacterial antigens. In relation to the infant rat model of Blood-Siegfried et al., 13 animals with the asymptomatic virus infection were significantly more susceptible to a dose of LPS that was not lethal for the control group unexposed to the virus infection. If our studies reflect the processes occurring in the mouse model, the findings indicate that IFN-γ produced in vivo in response to minor virus infections can significantly enhance pro-inflammatory responses to bacterial antigens. The findings in relation to nicotine and cotinine are consistent with previous reports of suppressed cytokine responses.40,41 It is recommended that not only nicotine but also CSE be used in to assess the effects of cigarette smoke in vitro. There are advantages and limitations of CSE compared with nicotine. CSE contains more components of cigarette smoke, and reflects more closely the conditions of exposure of humans to the effects of both passive and active smoking. Maintaining consistency of the chemical composition of CSE can prove to be a limiting factor in experimental design.
Footnotes
Funding
The majority of this work was funded by the Foundation for the Study of Infant Death (FSID) (UK) and by other smaller grants from Hunter Medical Research Institute (HMRI), the University of Newcastle and the John Hunter Children's Research Foundation.
