Abstract
The peak age at which sudden infant death syndrome (SIDS) occurs corresponds to the developmental period in which infants are dependent on their innate responses to infection. There is a growing body of evidence indicating that dysregulation of inflammatory responses might contribute to the physiological changes leading to these sudden deaths. This study examined the effects of three important risk factors for SIDS on inflammatory responses: cigarette smoke, virus infection and male sex. Cytokine responses of peripheral monocytic blood cells of healthy, non-smoking males and females to endotoxin were measured. Surrogates for virus infection or cigarette smoke were assessed using IFN-γ or water-soluble cigarette smoke extract (CSE). For most conditions, cells from males had lower pro-inflammatory cytokine responses than those of females. An opposite trend was observed for IL-10. Significantly lower levels of some cytokines were noted for cells from male donors exposed to CSE. In females, there were significant correlations between testosterone levels and levels of pro-inflammatory cytokines, but none for males. Testosterone levels in females correspond to those among male infants in the age range at greatest risk of SIDS. The effects of the testosterone surge in male infants need to be examined in relation to changes in cortisol levels that occur during the same period of infant development.
Introduction
The peak in sudden infant death syndrome (SIDS) occurs during the developmental period in which infants have low levels of specific Ab protection, either maternal or actively acquired immunity. They are dependent on their innate responses to deal with new infectious agents they encounter in their environment. Infection and inflammatory responses have been implicated in many of these deaths and we have examined the hypothesis that some SIDS deaths occur as a result of dysregulation of inflammatory responses, which can affect other physiological systems suggested to be involved in triggering these deaths.
Mage and Donner 1 examined a mathematical model of SIDS, concluding that SIDS had distinctive characteristics that needed to be explained: (i) the age distribution rising from 0 at birth to mode at 2 months, median at 3 months and an exponential decrease towards 0 at 1 yr or more; (ii) the decrease in SIDS following the campaigns to reduce prone sleeping position; (iii) the seasonal variation with the peak of deaths in winter; (iv) the characteristic higher proportion of males among all post-neonatal deaths.
Our previous studies, and those of others, have indicated that infection and infants’ inflammatory responses to these infections might trigger physiological dysregulation involved in the various hypotheses proposed to explain sudden death in infancy: hypoxia; cardiac arrhythmia; hypoglycaemia; shock; and hyperthermia.2,3 There is significant evidence of infection among these infants,4,5 the presence of bacteria or their toxins 6 and inflammation.5,7
In relation to the infectious trigger hypothesis, the age distribution corresponds to the lowest levels of maternal Abs and the infants’ immunisations are not yet complete.8,9 These are conditions in which non-immune infants are dependent on inflammatory responses to deal with new microorganisms in their environments. The decrease in the proportion of infants in the 4 and 5 month age range following the change in infant immunisation schedules in the UK appeared to reflect a protective booster effect.10,11 The age range at greatest risk of SIDS also corresponds to physiological changes that result in low night-time cortisol levels, 12 and these levels were unable to control inflammatory responses in a model system. 13
In relation to the effect of the campaigns to reduce prone sleeping, there are two factors associated with infections that are relevant. In the prone position, the number and variety of microorganisms in the nasopharynx of both adults and children are increased. Among older infants with respiratory virus infections who slept in the prone position, their nasopharyngeal flora was similar to that found among SIDS infants. 11 Staphylococcus aureus was the most common isolate from both SIDS and healthy children during the age range in which most SIDS deaths occur,11,14 and many of these isolates are capable genetically of producing pyrogenic toxins, such as those involved in toxic shock syndrome.15,16 Staphylococcal pyrogenic toxins have been identified in sera or tissues of over half of SIDS infants from five different countries. 6 The toxins are produced only between 37 ℃ and 40 ℃ and the mean temperature range of the nasopharynx of children in the non-prone position was 34.53 (95% CI 34.36–35.00). 17 In the prone position, the mean temperature was significantly higher; 5/30 (16.6%) recorded temperatures ≥37 ℃. 17 Prone position and evidence of infection were common findings for SIDS infants in a large Scandinavian study. 18
Seasonal variation noted by the winter peak could be associated with an increase in upper respiratory viral infections often reported by parents of SIDS victims. It has also been reported that in experimental models, viral infections significantly enhanced the lethality of staphylococcal toxins and endotoxin.8,19,20
The higher proportion of males observed among SIDS, sudden unexpected death in infancy (SUDI) and infants who died of respiratory infections 1 was explored in the current study. Review of the literature confirmed that there are significant immunological differences between genders. Compared with males, females have higher levels of Ig, mount greater humoral responses to vaccination and infection, respond better to microbial and non-microbial antigens, and have a higher incidence of autoimmune disease.21–23 Males also are reported to have increased risk of sudden unexpected deaths, SIDS, sepsis mortality and trauma-associated deaths; susceptibility for each of these conditions has been associated with altered cytokine responses. Differences in these responses have been attributed to 5α-dihydrotestosterone and 17β-estradiol. 24 Sex hormones have been associated with altered cytokine production in healthy donor PBMCs. 25 Evidence for the effect of gender on the inflammatory cytokine response is conflicting. Studies in relation to trauma suggest that males have reduced levels of inflammatory cytokines than oestrus females (i.e. high oestrogen levels), 26 but other studies indicate decreased pro-inflammatory cytokines in females. 27
The effect of testosterone on inflammatory responses has been controversial. For studies using blood samples from hypogonadal men, in vitro stimulation with LPS indicated testosterone increased IL-10 responses; however, a dose–response effect was not observed with increasing testosterone concentrations. 28 In vivo, testosterone replacement in hypogonadal men reduced TNF-α and IL-1β, but it increased IL-10. 29 In mice, testosterone reduced expression of TLR4. Castrated mice were significantly more susceptible to endotoxic shock, and this effect was reversed with testosterone replacement. 30
Many studies on the effect of gender have not controlled for important risk factors, such as smoking or exposure to cigarette smoke or concurrent infections. In the current study, responses of cells from healthy male and female blood donors to LPS were assessed in relation to the effects of cigarette smoke and virus infection. Because there is an increase in testosterone in male infants during the period in which in which infants are at greatest risk of SIDS, 31 these responses were also analysed in relation to testosterone concentrations in the sera of the donors.
Materials and methods
Buffy coats from 14 male (n = 14) and 14 female (n = 14) donors, aged 20–55 yr, were purchased from the Australian Red Cross Blood Service (ARCBS) (Sydney, Australia). Ethical permission was obtained from University of Newcastle Human Research Ethics Committee (H-229-0606) and ARCBS Ethics Committee (07-11NSW-07) for the purchase and use of human buffy coats for the purposes of the study. PBMCs were collected from each donor for in vitro cytokine stimulation assays and plasma was collected for the assessment of cotinine for evidence of exposure to cigarette smoke, a confounding variable in assessment of cytokine responses. 32 Donors with detectable levels of cotinine were excluded from the analysis. Only ARCBS donor samples that were cleared for infectious agents were received. Buffy coats were processed within 24 h of collection.
Analysis of exposure to cigarette smoke
ARCBS donors were assessed for exposure to cigarette smoke by analysing the level of cotinine in the plasma. Plasma was stored in 0.5 ml aliquots at −20 ℃ until required for cotinine testing. Cotinine was assessed by a semi-quantitative commercial competitive enzyme immunoassay kit according to manufacturer’s instructions (OraSure Technologies Inc., Bethlehem, PA, USA). To prevent false-negative classification of exposure to cigarette smoke, the qualitative cut-off of the assay was lowered from the recommended 25 ng/ml to 10 ng/ml. Donors with detectable levels of cotinine were excluded from stimulation assays.
Analysis of testosterone levels
Total testosterone levels in the plasma of the 28 donors was assessed by Clinical Biochemistry, Hunter Area Pathology Service using the Access immunoassay system (Beckman Coulter, Brea, CA, USA).
Stimulation assay
PBMCs from 28 donors (n = 28) were assessed for in vitro cytokine responses to a common bacterial antigen, LPS, identified in a significant proportion of SIDS and SUDI infants. 33 IFN-γ was used as a surrogate for virus infection and a cigarette smoke extract (CSE) as a surrogate for exposure to cigarette smoke. Conditions for the assays were optimised previously in experiments with a human monocytic cell line (THP-1).
PBMCs were collected by density gradient separation with Ficoll-Paque Plus (GE Healthcare, Buckinghamshire, UK) and Leucosep tubes (Greiner Bio-One, Frickenhausen, Germany) according to the manufacturer’s instructions. Cells were resuspended in pre-chilled freezing media containing 90% FCS (v/v) and 10% DMSO (v/v), at a concentration of 1 × 107 cells/ml. Cells were transferred into cryogenic vials in 1-ml aliquots (Nalgene, Rochester, NY, USA) and stored in liquid nitrogen.
Cryopreserved PBMCs were thawed quickly and diluted in pre-warmed RPMI containing FCS (10%, v/v) (RPMI 10%); 0.5 ml drop wise, followed by 1 ml, 2.0 ml and 4.0 ml at 1 min intervals and centrifuged for 10 min at 150 g. Cells were washed twice in RPMI 10%, counted and assessed for viability using the trypan blue exclusion method. A cell viability of >85% was considered acceptable.
PBMCs were plated into tissue culture well-plates (Greiner Bio-One) (500 µl/well) at a final concentration of 5 × 105cells/ml. Stimulants were added immediately to the cell culture system. Concentrations and durations of stimulation were as follows: 24 h pre-treatment with IFN-γ (10 ng/ml); 3 h pre-treatment with 0.001 cig/ml CSE, followed by 12 h incubation with LPS (50 ng/ml).
Cell culture supernatants were collected at 12 h after stimulation with LPS and stored immediately at –20 ℃ until assessed for cytokine production. Supernatants were collected by centrifuging cell culture plates at 250 g for 5 mins.
Analysis of cytokine response
Supernatants were measured for IL-1β, IL-6, TNF-α, IFN-γ, IL-8 and IL-10 using Bio-Rad 6-plex assays (Bio-Rad, Hercules, CA, USA) and the Luminex 200 analyser (Luminex Corporation, Austin, TX, USA). Cytokine concentrations (pg/ml) were calculated from the standard curve using Luminex 2.3 software.
Statistical analysis
Data were analysed using STATA™ version 10.0. Mean donor cytokine measurements were used for statistical analysis. The Wilcoxon matched-pairs signed ranks test was used to assess differences in cytokine responses between treatment groups. The significance level was set at P < 0.05. Testosterone concentrations were assessed in relation to cytokine responses by Pearson’s correlation.
Results
In addition to sex, the effects of virus infection (IFN-γ) and exposure to cigarette smoke (CSE) on donor inflammatory responses to LPS stimulation were assessed.
The effect of gender on inflammatory responses
Mean cytokine responses from cells of male (
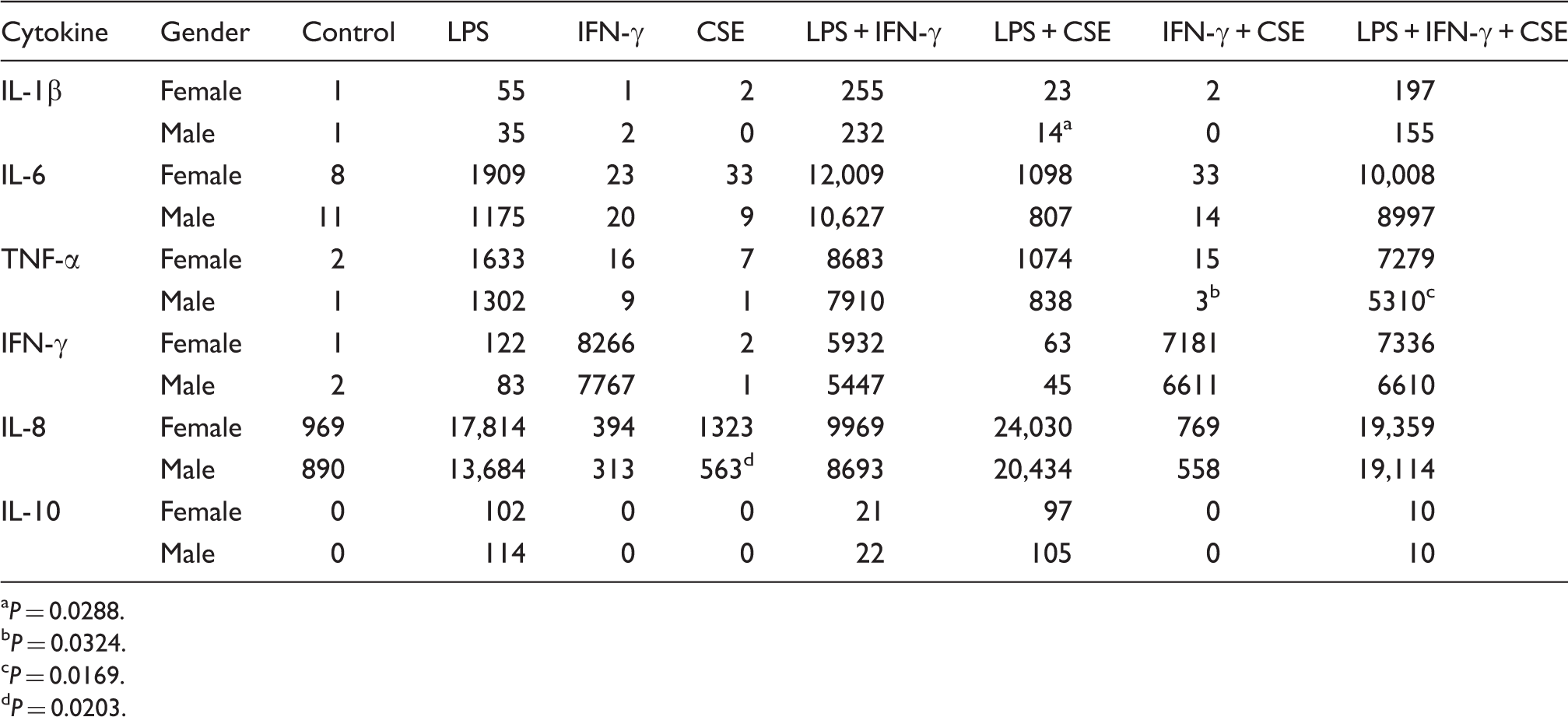
P = 0.0288.
P = 0.0324.
P = 0.0169.
P = 0.0203.
The effect of testosterone level on inflammatory responses
Correlation between testosterone levels and cytokine responses in males and females (Pearson coefficient).
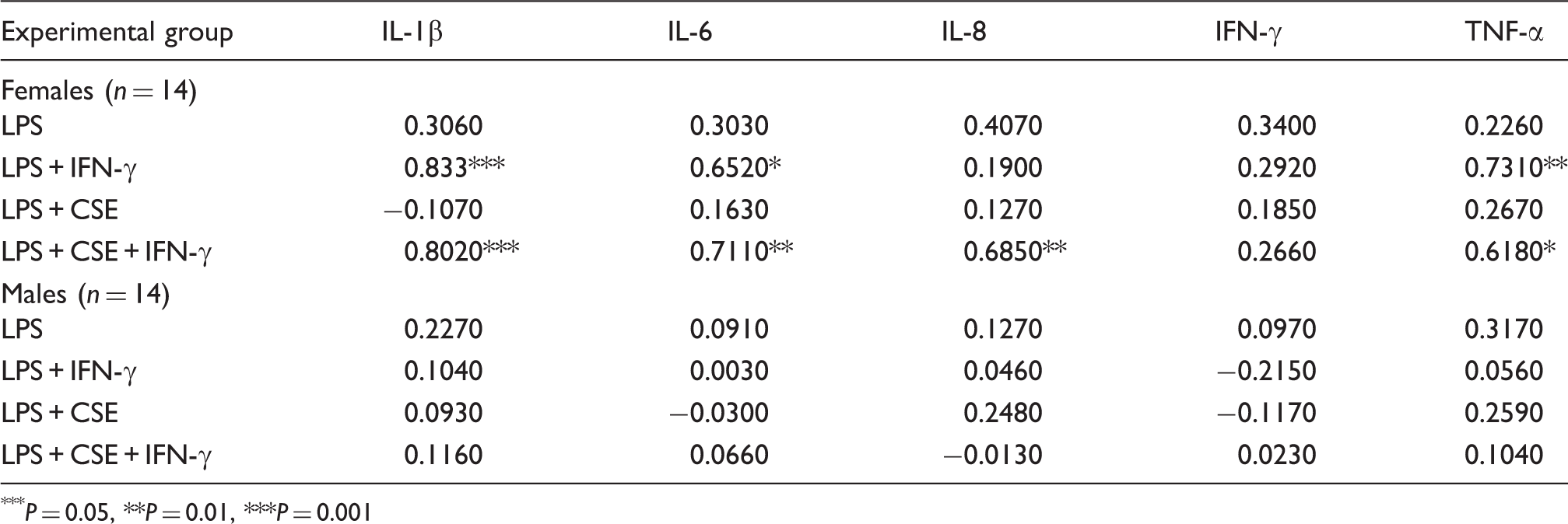
P = 0.05, **P = 0.01, ***P = 0.001
Discussion
Gender differences in inflammatory responses are well documented. Studies of the effect of gender on the inflammatory cytokine response are conflicting and often not controlled for recent or concurrent infections, or exposure to cigarette smoke.
The results of this study indicate that there are differences in inflammatory responses associated with gender; cells from males produced lower pro-inflammatory responses than those from females. The differences were most obvious when cells were exposed to components of cigarette smoke. Significant differences were observed only when cells were exposed to CSE or in combination with LPS or IFN-γ; however, a general trend was observed across all experimental conditions for all pro-inflammatory cytokines assessed. A reduction in pro-inflammatory responses could reduce damage due to responses to infectious stimuli, but they could also increase susceptibility to the initial stages of invasion/infection.
It is unclear if there is an interaction between cigarette smoke exposure and gender in inflammatory responses. Interactions between gender and smoking on cytokine responses have been reported previously. Seymour et al. 34 reported that female BALB/c mice had higher serum IgE and Th2 cytokine (including IL-10) production than male mice; when exposed to environmental tobacco smoke, these levels were increased significantly for females, but only increased significantly for Th2 responses in males.
There is a rise in testosterone production associated with the period during which SIDS is most prevalent. Between 1 and 5 months, testosterone levels range from 0.03 to 6.14 nmol/l for males and from 0.03 to 0.17 for females. In males these levels decrease to 0.07–0.24 at 6–11 months. 31 The ranges of testosterone in the adult females tested in our studies were within the range for males in the 1–5-month age range. There was a positive correlation between testosterone levels and pro-inflammatory responses to LPS when the cells were pre-treated with IFN-γ or IFN-γ and CSE (Table 2). No correlations were observed for levels found in adult males. The range of testosterone in adult males might be plateau levels in which concentrations of the hormone were too high to observe a positive correlation. The effect of testosterone on inflammatory responses has been controversial. For studies using blood samples from hypogonadal men, in vitro stimulation with LPS indicated that testosterone increased IL-10 responses; however, a dose–response effect was not observed with increasing testosterone concentrations. 28
These findings indicate that in addition to low levels of night-time cortisol present in infants during the age range at greatest risk of SIDS, 13 an additional factor affecting dysregulation of the inflammatory responses to apparently ‘mild’ infections might be the increase in testosterone in male infants. As this does not occur in female infants, the rise in testosterone could be an additional factor contributing to the higher proportion of males among SIDS infants.
Conclusion
This study confirms that PBMCs of males produce lower levels of pro-inflammatory cytokines than females and the responses were further reduced with exposure to the surrogate for cigarette smoke. A reduction in pro-inflammatory cytokines could reduce tissue damage or effects on physiological homeostasis during responses to infection. Reduced pro-inflammatory responses might also increase susceptibility to the initial stages of invasion/infection. Further investigations of the differences in cytokine responses and hormone levels in males and potential interaction of exposure cigarette smoke might provide further understanding of the effects of risk factors identified in epidemiological studies of SIDS and SUDI.
Footnotes
Funding
The work was funded by the Foundation for the Study of Infant Death (FSID) (UK), Babes in Arms (UK), Hunter Medical Research Institute (HMRI), the University of Newcastle and the John Hunter Children's Research Foundation.
