Abstract
Cardiovascular diseases (CVDs) linked to atherosclerosis remains the leading cause of death worldwide. Atherosclerosis is primarily caused by the accumulation of oxidized forms of low density lipoprotein (LDL) in macrophages (MΦs) in the subendothelial layer of arteries leading to foam cell and fatty streak formation. Many studies suggest that LDL that is modified by myeloperoxidase (MPO) is a key player in the development of atherosclerosis. MΦs can adopt a variety of functional phenotypes that include mainly the proinflammatory M1 and the anti-inflammatory M2 MΦ phenotypes which are both implicated in the process of atherogenesis. In fact, MΦs that reside in atherosclerostic lesions were shown to express a variety of phenotypes ranging between the M1- and M2 MΦ types. Recently, we pointed out the involvement of MPO oxidized-LDL (Mox-LDL) in increasing inflammation in MΦs by reducing their secretion of IL-10. Since little is known about Mox-LDL-mediated pro-atherosclerostic responses in MΦs, our study aimed at analyzing the in vitro effects of Mox-LDL at this level through making use of the well-established model of human THP-1-derived Mφs. Our results demonstrate that Mox-LDL has no effect on apoptosis, reactive oxygen species (ROS) generation and cell death in our cell model; yet, interestingly, our results show that Mox-LDL is significantly engulfed at a higher rate in the different MΦ subtypes supporting its key role in foam cell formation during the progression of the disease as well as previous data that were generated using another primary MΦ cell model of atherosclerosis.
Introduction
The leading cause of cardiovascular disease (CVD) worldwide is the pathological phenomenon known as “atherosclerosis”.1,2 Atherosclerosis is a multi-factorial, polygenic disease that is initiated through the interaction of an array of risk factors, mainly dyslipidemia.3,4 Increased low-density lipoprotein (LDL) cholesterol levels, indeed, contribute to the elevation of the risk of heart disease and stroke. 5 Despite the fact that the assessment of LDL levels is considered as a valuable tool in atherosclerosis risk prediction, native LDL itself does not accumulate in macrophages (MΦs), nor does it trigger an inflammatory response. In fact, extensively modified forms of LDL reside in the bloodstream and arterial wall of atherosclerotic patients.6,7 The process of LDL modification can be catalyzed by various enzymatic systems such as myeloperoxidase (MPO). 8 In fact, the activation of MΦs and neutrophils during immune responses, in turn, activates the production of MPO. 9 MPO can either be released into phagosomes or into the extracellular space where it catalyzes the halide/pseudo-halide-dependent hydrogen peroxide (H2O2) conversion into reactive oxygen intermediates (ROI), including hypochlorous acid (HOCl). 10 MPO is a key player in the development of atherosclerosis and elevated MPO levels were reported to correlate with the risk of CVD development. 11 Moreover, MPO oxidized-LDL (Mox-LDL)-specific monoclonal antibodies were shown to recognize human atherosclerotic tissue, further confirming the key involvement of MPO in the modification of LDL during atherosclerotic processes.12,13 It is thought that Mox-LDL is recognized by scavenger receptors which will lead to its internalization via receptor mediated endocytosis. However, the specific receptors allowing for the internalization of Mox-LDL in MΦs remain undetermined.14–16 Mox-LDL induces inflammatory responses in endothelial cells as well as in MΦs.15,17 In fact, MΦs treated with Mox-LDL display an increase in tumor necrosis factor alpha (TNF-α) production. 18 It was shown that oxidized LDL (ox-LDL) in general accumulates within MΦs, ultimately leading to foam cell formation which is considered as the hallmark of atherosclerosis.15,19 MΦs display a variety of functional phenotypes, and are consequently capable of responding to diverse stimuli. 20 The two major macrophage phenotypes are designated as M1 and M2, otherwise known as classically activated and alternatively activated MΦs, respectively. Ox-LDL was shown to push polarization towards the M1 MΦ phenotype. M1 MΦs are characterized by their ability to mediate pro-inflammatory as well as Th-1 related responses. 6 Conversely, M2 MΦs are recognized for mediating anti-inflammatory responses and this phenotype is thought to support atherosclerotic plaque stabilization and regression via the promotion of tissue repair and remodeling, as well as angiogenesis, and matrix deposition. 21 Polarization of MΦs, inside atherosclerotic lesions, is managed by a diverse contrast of microenvironmental factors. In fact, lesion-resident MΦs express a continuum of phenotypes ranging between the pro-inflammatory, pro-atherosclerotic M1 state, and the anti-inflammatory M2 phenotype. 22 Interestingly, MΦs are capable of shifting their phenotype as a result of microenvironmental and intracellular stimuli, a phenomenon that is widely known as “repolarization”. 23 Recently, we reported that the treatment of THP-1 M1 MΦs with Mox-LDL results in a significant decrease at the level of their secretion of the anti-inflammatory cytokine, interleukin (IL)-10. These results suggest that Mox-LDL is involved in the induction and exacerbation of the pro-inflammatory state observed in lesion-resident MΦs. 17 By using the same THP-1 cell model, the present study aimed to analyze the potential mechanisms by which Mox- LDL might induce a MΦ-mediated pro-atherosclerostic response in the intima contributing to the progression of the atheroma plaque. In fact, our work is the first to employ this particular cell model in order to study the in vitro effects of Mox-LDL on lipid uptake, reactive oxygen species production, apoptosis and inflammation in human MΦs.
Material and methods
Maintenance of THP-1 monocytes
THP-1 cells (kindly provided by Dr Marwan El-Sabban, American University of Beirut) were cultured in growth medium (GM) made up of Roswell Park Memorial Institute medium (RPMI-1640, Sigma Aldrich), supplemented with 10% heat inactivated fetal bovine serum (FBS; Sigma Aldrich), 1% penicillin/streptomycin mixture, and 1% L-glutamine. Cells were maintained at 37 °C in a humidified 5% CO2 incubator at 37 °C. THP-1 cells were used between passages 10–30. Cells were counted for viability using the Trypan blue exclusion method.
THP-1 monocytes differentiation into uncommitted MΦs (M0-MΦs)
To differentiate THP-1 cells into unpolarized M0-MΦs, THP-1 cells were seeded in a 6-well culture plate at a density of 2 × 105 cells/well. Cells were either left untreated or treated with phorbol 12-myristate 13-acetate (PMA) at a concentration of 150 nM for a period of 48 h. Following differentiation, adherent PMA-treated THP-1 MΦs were washed twice with GM. Meanwhile, untreated THP-1 cells were replenished by the addition of GM. Then cells were subjected to a resting period for another 24 h in GM to obtain the resting M0-MΦs. 17
Polarization of THP-1 M0-MΦ into M1-MΦ and M2-MΦ types
THP-1 M0-MΦs were polarized into M1-MΦs by incubation with 20 ng/ml of IFN-γ (Invitrogen, #RIFNG100) and 100 ng/ml of LPS (Invivogen, #tlrl-3pelps) for 24 h. THP-1 M0-MΦ were polarized into M2-MΦ by incubation with 20 ng/ml of interleukin 13 (Gibco, # PHC0134) and 20 ng/ml of interleukin 4 (Invitrogen, # RIL4I) for 24 h.
Mox-LDL preparation
Mox-LDL was generated by mixing 1.6 mg of native LDL (Invitrogen; # L3486) [final concentration: 0.8 mg/ml in phosphate-buffered saline (PBS), pH 7.4], with 8 µl of hydrochloric acid (HCl) at a concentration of 1 M (final concentration: 4 mM), 45 µl of recombinant human MPO (final concentration: 250 nM; R&S Systems #3174-MP-250), and 40 µl of hydrogen peroxide (H2O2) at a concentration of 50 mM (final concentration: 1 mM). The volume was adjusted to 2 ml with PBS (pH 7.4) containing 1 g/l of ethylenediaminetetraacetic acid (EDTA). Afterwards Mox-LDL was desalted to remove impurities. 14
Mox-LDL treatment of M0, M1 and M2 MΦs
M0, M1 and M2 MΦs were left untreated or treated with Mox-LDL at the physiologically relative concentration of 100 μg/ml for 24 h. Untreated cells were exposed to vehicle control (PBS) for 24 h.
Detachment of different THP-1 derived MΦ types
To collect adherent untreated and Mox-LDL treated THP-1 M0, M1 and M2 MΦs, cells were washed twice with warm PBS and incubated with accutase solution (StemPro Accutase Cell Dissociation Reagent) for 5–10 min at 37 °C. Detached cells were collected by centrifugation at 1200 rpm at 4 °C for 5 min.
Enzyme-linked immunosorbent assay (ELISA) for the detection of TNF-α
Culture supernatants from untreated and Mox-LDL-treated M0, M1 and M2MΦs were collected and stored at -80 °C for later cytokine analysis. The levels of TNF-α in culture supernatants were determined using a commercially available sandwich ELISA kit (Invitrogen, #KHC3011). The samples were processed according to the manufacturer's instructions in duplicates and measured at 450 nm on a Biotek micro-plate reader.
Estimation of ROS production
After removing the supernatant, wells containing untreated and Mox-LDL-treated M0, M1 and M2 MΦs were washed with PBS. MΦs were then detached on ice using accutase. Following centrifugation, the pellet was resuspended in 5 μM (final concentration) of 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA) prepared in pre-warmed (37 °C) PBS and incubated at 37 °C for 60 min in the dark. Cell permeable H2DCFDA is normally cleaved by intracellular esterases to the non-fluorescent 2′,7′-dichlorodihydrofluorescein (H2DCF), which is further oxidized by intracellular released ROS to green fluorescent dichlorofluorescein (DCF). DCF fluorescence was detected using a FACSCalibur flow cytometer (BD Biosciences) and CellQuest Pro software 5.1 (BD Biosciences).
Apoptosis assay
Apoptosis in untreated and Mox-LDL-treated M0, M1 and M2 MΦs was assessed through staining with 5 µL of FITC-conjugated Annexin V (Annexin; BD Biosciences) and 1 µL of a prepared 100 µg/ml of propidium iodide (PI; BD Biosciences) working solution in each 100 µL of cell suspension containing 1 × 105 cells. Cells were incubated at room temperature in the dark for 15 min. After the incubation period, 400 µL of 1X annexin-binding buffer were added and samples were kept on ice until flow cytometry analysis. Stained cells were analyzed on a FACSCalibur flow cytometer (BD Biosciences) and data were analyzed using CellQuest Pro software version 5.1 (BD Biosciences). Annexin-/PI- were identified as viable cell populations, Annexin+/PI- were identified as early apoptotic cells, Annexin+/PI+ were identified as late apoptotic cells and Annexin-/PI+ were identified as dead cells.
Lipid (oil red O) staining
Oil Red O staining was performed according to the manufacturer's instructions (MAK194-1KT, Sigma-Aldrich). Untreated, native LDL- and Mox-LDL-treated M0, M1 and M2 MΦs were washed with PBS then they were fixed in 10% buffered formalin for 30 min at room temperature. The fixed cells were then rinsed with deionized water and 60% isopropanol. The stock solution of 0.5% Oil Red O (in isopropanol) was diluted 60:40 in water. The working solution was then added to fixed cells for 5 min at room temperature. Then, cells were rinsed with deionized water. Stain was then extracted in 250 μl isopropanol and 200 μl was used to measure Oil Red O stain in a 96 well Biotek micro-plate reader at 492 nm.
Statistical analysis
Statistical analysis was performed using GraphPad Prism software (version 6.0; GraphPad Software, Inc.). Data are expressed as the mean ± standard error of the mean (SEM). P < 0.05 was considered to show a statistically significant difference. The statistical significance analysis was conducted using the independent unpaired t-test or one-way ANOVA followed by Tukey's multiple comparison post hoc test.
Results
Effect of Mox-LDL treatment on TNF-α secretion by THP-1-derived M0, M1, and M2-MΦs
In order to study the effect of Mox-LDL treatment on inflammation in THP-1-derived M0, M1, and M2-MΦs, the concentrations of TNF-α secreted by the different THP-1-derived MΦ types were assessed by ELISA. Mean TNF-α levels secreted by untreated M0, M1, M2 MΦs were comparable to those secreted by Mox-LDL treated M0, M1 and M2 MΦs, respectively (Figure 1). Therefore, Mox-LDL treatment did not lead to a significant increase in TNF-α secretion in any of the different types of THP-1 derived MΦ populations when compared to their respective untreated controls.

Effect of Mox-LDL treatment on TNF-α release by THP-1-derived M0, M1 and M2-MΦs. TNF-α levels were assayed by ELISA in the culture supernatants of untreated and Mox-LDL-treated (at a concentration of 100 μg/ml for 24 h) M0-, M1-, M2-Mφs. Bar graphs presenting mean values of TNF-α levels (of 3 independent experiments n = 3; with each condition performed in duplicate). Error bars represent SEM.
Effect of Mox-LDL treatment on reactive oxygen species production in THP-1-derived M0, M1, and M2-MΦs
In order to assess the effect of Mox-LDL treatment on the overall oxidative stress in THP-1-derived M0, M1, and M2-MΦs, ROS generation assay was performed using the H2DCFDA probe whereby ROS levels were indirectly expressed as the MFI values of DCF in different untreated and Mox-LDL-treated M0, M1 and M2 Mφ populations. The MFI values of DCF in untreated M0, M1 and M2- MΦ populations were 91 ± 13, 109 ± 10, and 125 ± 25, respectively. On the other hand, the MFI values of DCF in Mox-LDL treated M0, M1 and M2 MΦ populations were 81 ± 18, 79 ± 19, and 95 ± 18, respectively. Mox-LDL treatment did not lead to a significant alteration in ROS generation across the different types of THP-1-derived MΦ populations (Figure 2).

Effect of Mox-LDL treatment on ROS generation in THP-1 derived M0, M1 and M2-MΦs. Different THP-1-derived MΦ types were cultured in 6-well plates and left untreated or treated with 100 µg/ml of Mox-LDL for 24 h. Cells were then harvested, labeled with H2DCFDA and analyzed via flow cytometry. Bar graph presenting the geometric MFI values (mean ± SEM) of DCF-stained MΦs (of 3 independent experiments n = 3; with each condition performed in duplicate). MFI, mean fluorescent intensity; H2DCFDA, 2′,7′-dichlorodihydrofluorescein diacetate; DCF, 2,7-dichlorofluorescein.
Effect of Mox-LDL treatment on apoptosis in THP-1-derived M0, M1, and M2-MΦs
Annexin/PI apoptosis assay was performed in order to assess the apoptosis-inducing and cytotoxic effects of Mox-LDL treatment on the different THP-1-derived MΦ types (Figure 3(a) and (b)).

Effect of Mox-LDL treatment on apoptosis and viability in M0, M1, and M2 MΦs. Different MΦ types were cultured in 6-well plates and left untreated or treated with 100 µg/ml of Mox-LDL for 24 h. Apoptosis was then assessed by Annexin V/ PI apoptosis assay followed by flow cytometric analysis. (a) Representative flow cytometry dot plots and (b) bar graphs representing the % (mean ± SEM) of viable, early apoptotic, late apoptotic and dead subpopulations with untreated and Mox-LDL-treated THP-1 derived MΦ types (of 4 independent experiments n = 4; with each condition performed in duplicate).
Mox-LDL treatment of THP-1-derived M0, M1, and M2 MΦs did not lead to significant alterations in any of the percentages of viable (Annexin-/PI-), early apoptotic (Annexin+/PI-), late apoptotic (Annexin+/PI+) and dead cell (Annexin-/PI-) MΦ subpopulations as compared to their respective control untreated MΦ subpopulations (Figure 3(b)).
Effect of Mox-LDL treatment on lipid uptake in THP-1-derived M0, M1, and M2-MΦs
In order to study the effect of Mox-LDL treatment on lipid uptake in THP-1-derived M0, M1, and M2-MΦs, the latter subtypes were left untreated or treated with native LDL or Mox-LDL (100 µg/ml), and the amount of lipid that was taken up by the different THP-1 derived MΦs was assessed by Oil Red O staining. The amounts of lipid that was accumulated by untreated, native LDL-treated and Mox-LDL-treated M0, M1 and M2 MΦs were measured by spectrophotometry. Analysis of generated optical densities strongly indicated that Mox-LDL treatment of M0, M1 and M2 MΦs significantly increased their lipid uptake when compared to untreated and native LDL-treated M0, M1, and M2-MΦs (Figure 4) which confirm previous data that were produced using both the murine RAW264.7 cell line and PBMC-derived MΦs. 24
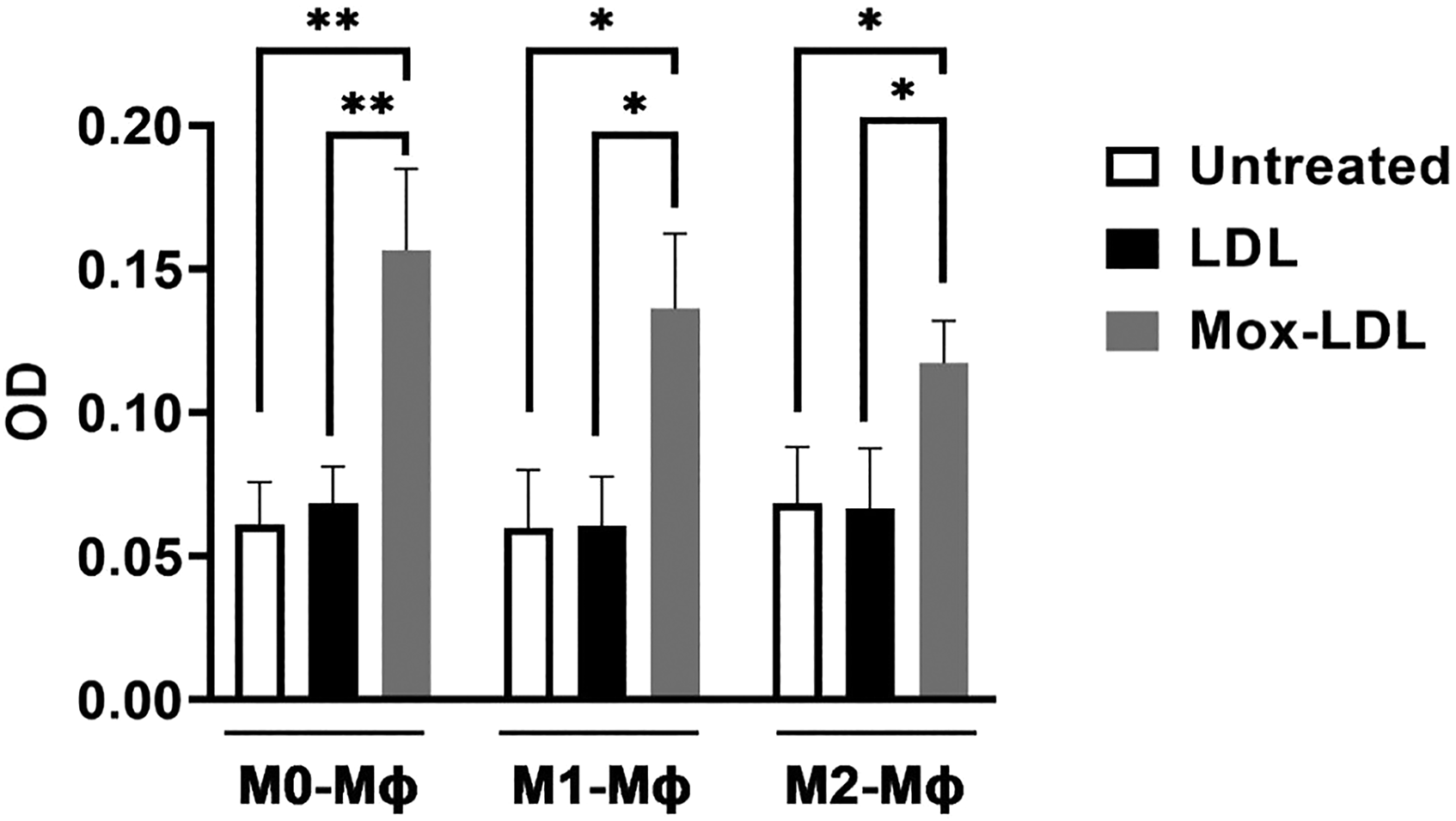
Effect of Mox-LDL treatment on lipid uptake in THP-1-derived MΦ types: M0, M1 and M2. Different THP-1 derived MΦ types were cultured in 6-well plates and left untreated or treated with 100 µg/ml of either native LDL or Mox-LDL for 24 h. Cells were then harvested and lipid uptake was assessed by Oil Red O staining and quantified via spectrophotometry analysis at 492 nm. Bar graph presenting the OD values (mean ± SEM) of Oil Red O stained MΦs (of 3 independent experiments n = 3; with each condition performed in duplicate). One-way ANOVA followed by Tukey's multiple comparison post hoc test was used to calculate statistical significance. * P < 0.05; ** P < 0.01.
Discussion
Atherosclerosis is best described as a progressive chronic inflammatory state affecting the tunica intima. Ox-LDL is a chief proatherosclerotic factor, inducing endothelial dysfunction (ED) and the formation and accumulation of foam cells within the intima, subsequently leading to the formation of lipid-laden plaques. Mox-LDL is the most physiologically relevant form of ox-LDL in the context of atherosclerosis, where it plays a major role in favoring the progression of atherosclerotic lesions.7,9 As previously mentioned, Mox-LDL was shown to increase the pro-inflammatory state in Mφs by reducing the secretion of the anti-inflammatory cytokine, IL-10. 17 Added to this well-documented pro-inflammatory role of Mox-LDL in Mφs, the reported shift from M2-MΦ to M1-MΦ phenotype accompanying plaque progression in atherosclerotic patients25,26 prompted us to investigate whether this phenomenon may be caused by a potential involvement of Mox-LDL in this particular context. In fact, we adopted the THP-1 derived Mφ model in order to explore any potential effects of Mox-LDL treatment (at a physiologically relevant concentration of 100 µg/ml) on TNF-α and ROS production, apoptosis, and lipid uptake in THP-1 derived M0, M1, and M2-MΦs. TNF-α is a major proinflammatory signature cytokine for THP-1 M1 Mφs. 27 In order to investigate any potential effect of Mox-LDL treatment on inflammation in THP-1 derived M0, M1, and M2-MΦs, we examined the secretion of pro-inflammatory cytokine TNF-α by the different THP-1 derived MΦ subtypes. We found that Mox-LDL treatment had no significant effect on TNF-α secretion among THP-1 derived M0, M1, and M2-MΦs. TNF-α secretion appeared to be higher in untreated and Mox-LDL-treated M1-MΦs as compared to untreated and Mox-LDL-treated M0 and M2-MΦs. This difference, however, held no statistical significance. In contrast to our observation, a previous study revealed that the incubation of THP-1 monocytes with the same physiological concentration of Mox-LDL employed in our study (100 µg/ml), induced a 2-fold increase in TNF-α secretion as compared to the control. 18 Nevertheless, other studies have demonstrated that THP-1 and U937 cell lines display enhanced resistance to ox-LDL exposure once they have differentiated into Mφs, as compared to their undifferentiated state.28,29 Meanwhile, we also investigated the effect of Mox-LDL on ROS production in THP-1 derived M0, M1, and M2-MΦs, and demonstrated that Mox-LDL treatment had no significant effect on ROS generation within the different THP-1 derived MΦ subtypes. Our findings are in agreement with previous reports demonstrating that Mox-LDL does not increase ROS generation in human aortic endothelial cells (HAECs), in contrast to cupper oxidized LDL (CuoxLDL), which was shown to have a positive effect at this level. 30 Furthermore, we tested for any potential cytotoxic effects that Mox-LDL treatment might have on THP-1 derived M0, M1, and M2-MΦs. We found that, despite an apparent overall increase in cell death in treated M1, and M2-MΦs as compared to their respective untreated controls, this variation held no statistical significance. In fact, Mox-LDL treatment did not significantly alter any of the percentages of viable, early apoptotic, late apoptotic and dead cell populations as compared to their respective controls. This observation is also comparable to multiple previous studies which reported that Mox-LDL has no cytotoxic effect in different cell models of atherosclerosis including HAECs and human umbilical vein endothelial cells (HUVECs).31–34 Another study showed that human primary monocyte-derived and THP-1 derived M2 polarized Mφs were significantly more sensitive to ox-LDL lipotoxicity when compared to non-differentiated monocytic cells or unpolarized MΦs. 34 However, this study employed a different form of modified LDL. Yet, in different study employing the THP-1 cell model, preliminary experiments using physiological concentrations of Mox-LDL showed limited toxicity and apoptosis, thus confirming our results. 35
On a different note, it was shown that Mox-LDL inhibits lysosomal protease activity in Mφs, and thus, might be involved in foam cell formation. 36 In order to test for any potential effects of Mox-LDL on lipid uptake in THP-1 derived M0, M1, and M2-MΦs, we treated these subtypes with either native- or Mox-LDL, and we assessed the amount of lipids taken up by the different THP-1 derived MΦ types by Oil Red O staining and spectrophotometric quantification. Exposure of M0, M1, and M2-MΦs to 100 μg/ml of Mox-LDL for 24 h significantly increased measured OD by ∼ 2.6-fold (p < 0.01), 2.3-fold, and 1.7-fold (p < 0.05) when compared to the control M0, M1, and M2-MΦs respectively. No significant alteration in OD was observed in native LDL- treated types when compared to their respective controls, nor was there any significant difference between different subtypes in any of the untreated, LDL- and Mox-LDL-treated conditions. These results suggest that Mox-LDL is taken up by M0, M1, and M2-MΦs at a significantly greater rate as compared to native LDL. This set of data is consistent with the line of research suggesting that only highly oxidized forms of LDL accumulate within MΦs, leading to foam cell formation. 19 Our data are also in agreement with what had been previously reported using human PBMC-derived MΦs where it was shown that these cells significantly accumulate more Mox-LDL than CuoxLDL or native LDL leading to lipid-laden MΦs. 24 The exact receptors mediating the internalization of Mox-LDL are not yet fully uncovered. 14 However, a recent study revealed that Mox-LDL treatment activates HAECs through the lectin-like oxidized low-density lipoprotein receptor-1 (LOX-1) receptor suggesting that Mox-LDL may drive ED through this scavenger receptor. 13 LOX-1 is a glycoprotein that binds to and recognizes oxidized forms of LDL; this receptor is known to be upregulated in the endothelial lining of atherosclerotic arteries both within a variety of animal species and in humans. 37 Meanwhile, M2 polarized MΦs were previously shown to exhibit an increased expression of the scavenger receptors CD36, SR-A1 and LOX1. M1 polarized MΦs; however, present reduced expression of cholesterol efflux proteins ABCA1, SR-B1 and 27 hydroxylase. 38 This goes hand-in-hand with our results, where no significant discrepancies were detected between the different subtypes at the level of Mox-LDL uptake. One might hypothesize that the reduced expression of cholesterol efflux proteins within M1 MΦs may have been compensating for the increased expression of the scavenger receptors within M2 MΦs. Nonetheless, a longer-lasting exposure to Mox-LDL may ultimately lead to a divergence in the rate of lipid uptake between the two subtypes.
Overall, our study demonstrates that Mox-LDL is taken up by THP-1 derived M0, M1, and M2-MΦs at a higher rate when compared to native LDL. Extensive research is still needed in order to better understand the complex mechanisms influencing atherosclerosis development. Treatment with higher concentrations of Mox-LDL for greater time intervals may be carried out as a means to bypass in vitro limitations, and to better replicate the phenomena occurring in vivo. Furthermore, a better understanding of the implication of LOX-1 in lipid uptake by Mφs, as well as the role of hypoxia in the hindering of apoptotic processes, 35 could contribute to the development of molecular drugs capable of attenuating, stopping, or even reversing the process of atherosclerosis.
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
All authors participated in the design, interpretation of the studies and analysis of the data and review of the manuscript; EJ, CF and SB conducted the experiments, EJ, SB and JD helped in the writing of the manuscript, SB and JD helped in the conceptualization of the research work and edited the final version of the manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been funded with the support of the University of Balamand (UOB RGA Grant ref. RGA/F AS/19- 20/007).
Patient consent for publication
Not applicable.
