Abstract
Ganoderma lucidum (G. lucidum) is a medicinal mushroom long used in Asia as a folk remedy to promote health and longevity. Recent studies indicate that G. lucidum activates NK cells, but the molecular mechanism underlying this effect has not been studied so far. To address this question, we prepared a water extract of G. lucidum and examined its effect on NK cells. We observed that G. lucidum treatment increases NK cell cytotoxicity by stimulating secretion of perforin and granulysin. The mechanism of activation involves an increased expression of NKG2D and natural cytotoxicity receptors (NCRs), as well as increased phosphorylation of intracellular MAPKs. Our results indicate that G. lucidum induces NK cell cytotoxicity against various cancer cell lines by activating NKG2D/NCR receptors and MAPK signaling pathways, which together culminate in exocytosis of perforin and granulysin. These observations provide a cellular and molecular mechanism to account for the reported anticancer effects of G. lucidum extracts in humans.
Introduction
Ganoderma lucidum (G. lucidum)—also called reishi, lingzhi or mannentake—is a lamellae Basidiomycete fungus that grows naturally on decaying logs of many broad leaf trees. 1 G. lucidum has been recognized as a potent medicinal mushroom for more than 2000 years in China owing to its beneficial effects on health, healing and longevity. 1
Previous studies have indicated that G. lucidum produces various beneficial effects on cultured cells and laboratory animals. For instance, G. lucidum exhibits antioxidant activity against acute ethanol-induced heart toxicity. 2 This fungus also protects mice against colitis induced by trinitrobenzene sulfonic acid, 3 and prevents liver injury induced by Mycobacterium infection. 4 Notably, G. lucidum produces a beneficial effect in an animal model of type 2 diabetes mellitus by lowering serum glucose levels through suppression of phosphoenolpyruvate carboxykinase expression in the liver. 5
Several studies have shown that G. lucidum produces both stimulatory and inhibitory effects on immune cells. For instance, it enhances the activity of immune effector cells in immunosuppressed mice. 6 However, this fungus also suppresses cell proliferation and LPS-stimulated inflammatory responses in macrophages,7,8 and induces the phagocytic activity of human polymorphonuclear neutrophils 9 and cytokine expression in mouse splenocytes 10 by activating MAPK signaling pathways.
G. lucidum also inhibits the growth of leukemic cells and induces their differentiation into mature monocytes/macrophages. 11 It activates the proliferation and anti-tumor activity of cytokine-induced killer cells, 12 and it shows inhibitory effects on murine oral cancer, 13 bladder cancer cells, 14 hepatoma cells 15 and breast cancer cells. 16 G. lucidum produces anti-tumor effects by inducing cytokine release from activated macrophages and T lymphocytes. 17 Furthermore, intraperitoneal administration of the closely-related Ganoderma tsugae increases splenic NK cytotoxic activity in a dose-dependent manner in C3H/HeN mice. 18 Notably, preliminary evidence suggests that G. lucidum may provide beneficial effects in treating human cancers when used as an adjuvant treatment with conventional therapies.19–21
Even though numerous studies have been performed to understand the effects of G. lucidum on cultured cells and animals, the molecular mechanisms underlying the effects of this fungus on NK cells have not been examined so far. NK cells are important cells of the innate immune system that are involved in early host defense against infections and tumors.22–24 We, and others, have previously shown that the cytotoxicity of NK cells involves the secretion of cytolytic proteins, such as perforin, granzymes and granulysin towards the target cell.25–28 These cytolytic proteins are exocytosed by activated NK cells, and they mediate the cell-killing activity by inducing apoptosis in target cells.
NK cell cytotoxic activity is modulated by signals that bind to activating and inhibitory receptors on the cell surface. 29 The major activating receptors are natural cytotoxicity receptors (NCRs), which include the constitutively expressed receptors NKp46 30 and NKp30 31 , as well as the induced receptor NKp44. 32 Moreover, the natural-killer group 2D receptor (NKG2D), which is also found on human T cells, 33 also activates NK cell cytotoxicity. 34 The binding of ligands on the activating receptors leads to activation of a cascade of intracellular signals resulting in polarization and exocytosis of granules that lyse target cells.29,35 In NK cells, ERK, JNK and p38 are critical intermediates of the MAPK signaling pathways that mediate the reorientation of the microtubule organizing center, and induce granule polarization and secretion.36–38
In the present study, we examined the effects of a G. lucidum water extract on NK cell cytotoxicity. Our results indicate that G. lucidum activates NK cell cytotoxic activity by inducing NKG2D/NCR receptors, MAPK signaling pathways, and the release of perforin and granulysin.
Materials and methods
Chemical reagents and fungal strain
Cell culture media, including alpha minimum essential medium (α-MEM) without ribonucleosides or deoxyribonucleosides, RPMI 1640 medium without phenol red, horse serum and FBS were purchased from Invitrogen (Carlsbad, CA, USA). Inositol, 2-mercaptoethanol, folic acid, glutamine, JNK inhibitor (SP600125), ERK1/2 inhibitor (PD98059) and p38 inhibitor (SB203580) were obtained from Sigma-Aldrich (St. Louis, MO, USA). The MAPK inhibitors were stored in DMSO at –20℃ until use. The G. lucidum strain initially selected and characterized at Chang Gung Biotechnology (Taipei, Taiwan) was validated by sequencing of rDNA and internal transcribed spacer DNA, which showed 99.39% identity with a database strain [European Molecular Biology Laboratory (EMBL) accession number AF506371; EMBL, Hinxton, UK]. The water extract was prepared by mixing 400 g of dried G. lucidum mycelium into 10 l of double-distilled water, followed by agitation at a speed of 150 rpm for 30 min at 12℃. The solution was cooled to room temperature (22℃) and centrifuged at 5894 g for 30 min at 4℃ using a Sorvall RC 3C Plus centrifuge (Thermo Fisher Scientific, Waltham, MA, USA). The supernatant was concentrated to a final volume of 2 l by using a Büchi R220 Rotavapor vacuum concentrator (Buchi, Flawil, Switzerland) at 65℃. The extract was sterilized at 121℃ for 20 min, and stored at 4℃ in dark glass bottles prior to use.
Cell isolation and culture
The human NK cell line NK92 (ATCC CRL-2407) was derived from a case of non-Hodgkin’s lymphoma, as described previously. 39 NK92 cells were maintained in culture with 100 IU/ml of IL-2 (Peprotech, Rocky Hill, CT, USA). Primary NK cells (pNK) were collected from healthy individuals following the method of Favier et al. 40 Briefly, pNK cells were isolated from peripheral blood using the EasySep NK cell depletion kit, as described by the manufacturer (Stem Cells, Grenoble, France). The purity of pNK cell preparations was verified by flow cytometry based on CD16, CD56 and CD3 expression. The pNK cell preparations used contained more than 80% CD16+CD56+ cells, but no CD3+ cells. pNK cells were activated by adding 200 IU/ml IL-2 (Peprotech) to the culture medium for 36–48 h. All cell lines were cultured in α-MEM containing 2 mM L-glutamine, 1.5 g/l sodium bicarbonate, 12.5% horse serum and 100 IU/ml IL-2. The K562 cell line was obtained from the ATCC (Manassas, VA, USA), and was maintained in RPMI 1640 containing 10% FBS.
Abs
Mouse anti-human Abs raised against perforin, granulysin, NKG2D, NKp30, NKp44 or NKp46 were obtained from R & D Systems (Minneapolis, MN, USA). Rabbit polyclonal Abs against ERK, JNK, p38, phospho-ERK (Thr202/Tyr204), phospho-JNK (Thr183/Tyr185) or phospho-p38 (Thr180/Tyr182) were purchased from Cell Signaling Technology (Danvers, MA, USA). Mouse monoclonal anti-actin Ab was obtained from Chemicon International (Temecula, CA, USA).
MTT cell viability assay
pNK and NK92 cells (1 × 105/well) were seeded in 96-well plates in which 90 µl of culture medium containing various concentrations of G. lucidum water extract were incubated for 24 h. Then, 10 µl of MTT reagent (5 mg/ml; MTT In Vitro Toxicology Assay kit; Sigma) was added to the wells and the plates were incubated for 3 h at 37℃. Then, 100 µl of solubilization solution was added prior to incubation overnight (18 h). Cell viability was calculated based on instructions from the manufacturer. All experiments were performed in triplicate.
Cytotoxicity assay
Cytotoxicity was evaluated using the CytoTox 96 Non-Radioactive Cytotoxicity Assay (Promega, Madison, WI, USA) based on the colorimetric measurement of lactate dehydrogenase, a stable cytosolic enzyme released from target cells following lysis by NK cells. Briefly, NK effector cells (5 × 104, 2.5 × 104 and 5 × 103 cells/well in U-bottom 96-well plates (Corning, Corning, NY, USA) were treated with different doses of G. lucidum water extract for 24 h and pretreated with MAPK inhibitors for 30 min or with MAPK small interfering RNA (siRNA) overnight, followed by treatment with 0.1–5% G. lucidum for 4 h. NK cells were then washed and re-suspended in RPMI 1640. A constant number of K562, A549, HepG2, MCF-7 and HT-29 target cells (5 × 103/well) cultured in complete cell culture medium was then added at the E:T ratios of 1 : 1, 5 : 1 and 10 : 1, based on our previous experimental results. 38 The microplates were centrifuged for 4 min at 250 g to settle the cells before incubation for 4 h at 37℃ in 5% CO2. After this co-incubation, supernatant (50 µl) was collected from each well, added to 50 µl of the provided Substrate Mix and incubated for 10 min at room temperature in the dark. The enzymatic reaction was stopped using the Stop Solution (50 µl), and absorbance was measured at 490 nm. Maximum release (TM) was determined by lysing target cells with 10 µl of lysis solution. Spontaneous release by either the target cells (TS) or effector cells (ES) was determined following incubation alone at the respective cell concentrations. Results were calculated and expressed as percentage of cytotoxicity using the following equation: [(experimental – ES – TS)/(TM – TS)] × 100.
Flow cytometry analysis
Flow cytometry analysis was performed using a FACS CaliburTM (Becton Dickinson, Franklin Lake, NJ, USA). Fluorophore bound Abs were used to sort cells: anti-NKG2D-allophycocyanin, anti-NKp46-fluorescein- isothiocyanate, anti-NKp30-phycoerythrin (R & D Systems) and anti-NKp44-phycoerythrin (Becton Dickinson). In brief, the non-specific antigens of 5 × 105 NK92 cells were blocked using cold 1% BSA–PBS at 4℃ for 30 min, prior to treatment with the Abs in the dark at 4℃ for 30 min. The cells were washed twice in cold PBS and re-suspended in 1 ml cold PBS before analysis.
RNA isolation and real-time PCR
mRNA was isolated from pNK and NK92 cells using the Qiagen RNeasy kit (Qiagen, Valencia, CA, USA) based on the manufacturer’s instructions, and total RNA was converted to cDNA using the Taqman reverse transcriptase kit (Applied Biosystems, Foster City, CA, USA). Quantitative PCR was performed with 1/50 of the cDNA preparation in an Mx3000P (Stratagene, La Jolla, CA, USA) in a final volume of 25 µl with Brilliant QPCR Master Mix (Stratagene). Real-time PCR protocols included an initial denaturation at 95℃ for 10 min, followed by 50 cycles of 95℃ for 30 s, 55℃ for 1 min, 72℃ for 1 min and, finally, 95℃ for 1 min, 55℃ for 30 s and 95℃ for 30 s.
Western blot analysis
NK92 cells (106/well in six-well plates) were stabilized overnight in cultured medium, followed by addition of different doses of G. lucidum water extract. Washed cells were lysed in Mammalian Protein Extraction Reagent (Pierce Chemical, Rockford, MA, USA). Total lysate protein samples (40 µg per lane) were fractionated on a 10% SDS polyacrylamide gel and blotted onto polyvinylidene difluoride membranes (Immobilon-P; Millipore, Billerica, MA, USA). Membranes were blocked with 5% non-fat milk for 2 h at room temperature in TBS (Tris 10 mM, NaCl 150 mM, pH 7.6) containing 0.1% Tween 20, and probed with a primary Ab (1 : 1,000 for anti-perforin, anti-granulysin, anti-NKG2D, anti-ERK, anti-JNK, anti-p38, anti-phospho-ERK, anti-phospho-JNK and anti-phospho-p38; 1 : 10,000 for anti-actin) overnight at 4℃. Membranes were then incubated with the appropriate HRP-conjugated secondary Ab (1 : 10,000). Immunoreactive protein bands were developed by enhanced chemiluminescence (Amersham Pharmacia Biotech, Piscataway, NJ, USA).
siRNA knockdown and siRNA transfection
siRNA against human p38 (sc-29433), JNK-1 (sc-29380), ERK-2 (sc-35335), NKG2D (sc-42948), NKp44 (sc-72170), NKp46 (sc-63344), NKp30 (sc-42950) and control siRNA (sc-37007) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The siRNA was transfected into NK92 cells by electroporation using a commercial kit (Amaxa, Gaithersburg, MD, USA).
Data analysis
Statistical analysis was performed using ANOVA (SPSS 12.0 software; SPSS, Chicago, IL, USA) with a correction for multiple comparisons. A difference between the results of two assay conditions that gave a P-value < 0.05 was considered significant.
Results
Effect of G. lucidum water extract on NK cell viability and cytotoxic activity
We first verified whether the G. lucidum water extract may affect the viability of NK cells. The viability of pNK and the cultured NK92 cell line was not affected by treatment with 0.1% G. lucidum (Figure 1A, B). At 2 and 5%, we observed that G. lucidum instead increased the fraction of viable NK cells compared with untreated cells (Figure 1A, B). Next, we measured the cytotoxic activity of G. lucidum-treated NK cells against the human leukemia cell line, K562. NK cells were treated with G. lucidum at various concentrations (0.1, 2 and 5%), and the cytotoxic activity of the treated cells was measured against K562 cells at various effector:target (E:T) ratios (1 : 1, 5 : 1 and 10 : 1). We observed that treatment with G. lucidum stimulated the cytotoxicity of both pNK and NK92 cells in a dose-dependent manner (Figure 1C, D).
G. lucidum enhances perforin and granulysin expression and NK cell cytotoxic activity. G. lucidum water extract produced no cytotoxic effect on NK cells. At 2% and 5%, G. lucidum enhanced the viability of pNK (A) and NK92 cells (B). The cytotoxic effect of pNK (C) and NK92 cells (D) against K562 cells was induced by G. lucidum in a dose-dependent manner at E:T ratios of 1 : 1, 5 : 1 and 10 : 1. Cytotoxicity was measured based on the release of lactate dehydrogenase, and data were expressed as percent of cytotoxicity. NK92 cells were treated with G. lucidum at the dose indicated. mRNA (E) and protein (F) levels of perforin and granulysin were measured. The data represent means of three independent experiments performed in triplicate. (*P < 0.05, **P < 0.01, ***P < 0.001).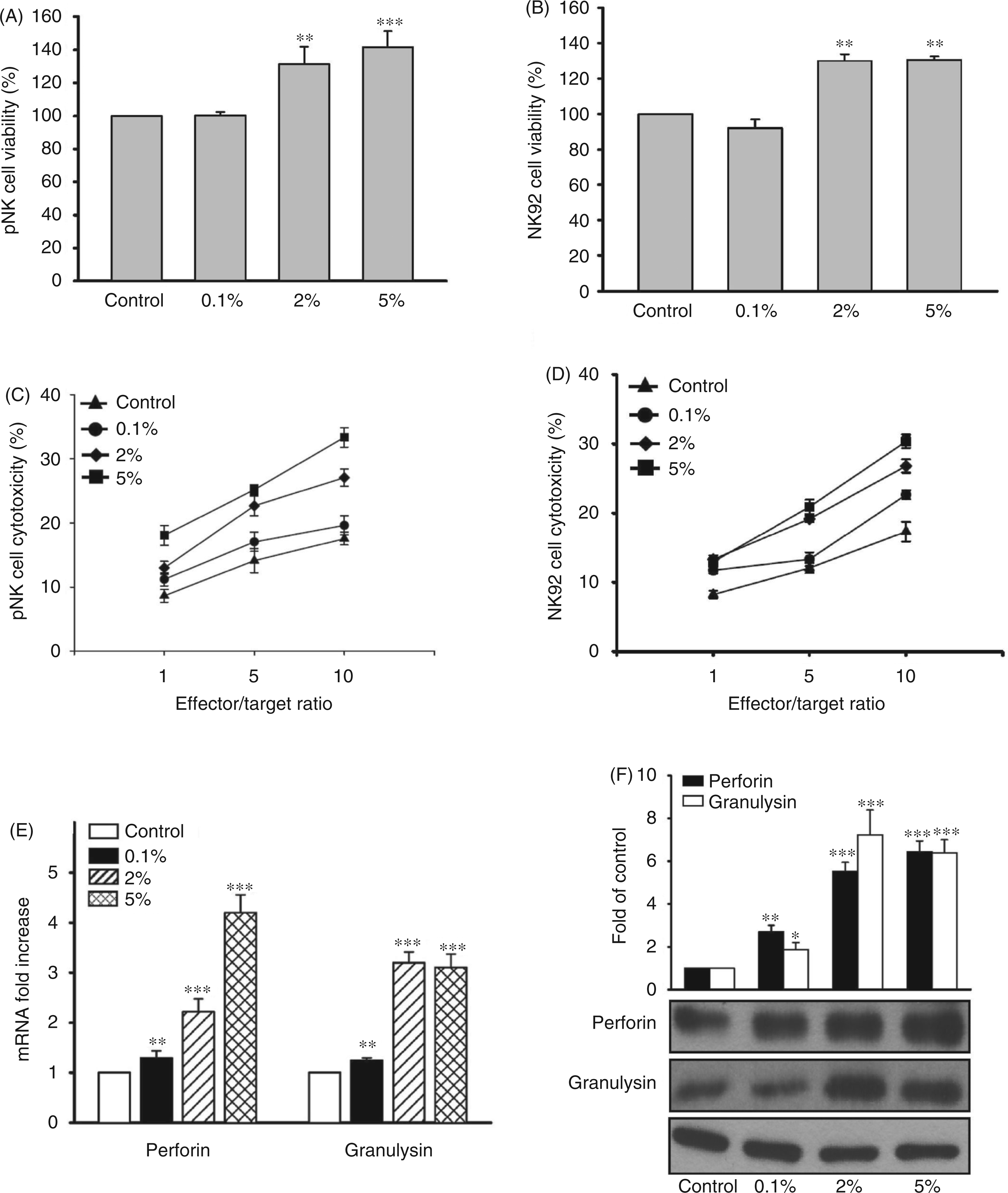
G. lucidum increases NK cell perforin and granulysin production
We next examined whether G. lucidum treatment may increase the production of the cytolytic proteins, perforin and granulysin in NK cells. We found that G. lucidum treatment increased the level of perforin and granulysin mRNA in a dose-dependent manner compared with untreated cells (Figure 1E). G. lucidum treatment also increased the production of perforin and granulysin protein in a dose-dependent manner (Figure 1F). These results suggest that G. lucidum treatment increases the cytotoxic activity of NK cells by inducing the expression and production of perforin and granulysin.
G. lucidum enhances expression of NKG2D and NCR cell surface receptors
NKG2D and NCRs represent cell surface receptors that activate NK cell cytotoxicity and induce the secretion of perforin and granulysin.22–24,27 Therefore, we examined the effects of G. lucidum on NKG2D and NCR gene expression. As shown in Figure 2A, the mRNA level of NKG2D, NKp30, NKp44, and NKp46 increased in a dose-dependent manner following G. lucidum treatment. In agreement with the level of mRNA expression, production of the corresponding proteins also increased following G. lucidum treatment compared with control cells (Figure 2B). To further confirm the level of activating receptors on NK cells, we performed a flow cytometry analysis of G. lucidum-treated NK cells using fluorophore-bound Abs. We observed that the level of cell surface NKG2D and NCR proteins was enhanced by treatment with G. lucidum compared with control, untreated cells (Figure 2C). These results indicate that G. lucidum not only affects the production of perforin and granulysin, but also the expression and production of upstream signaling receptors.
G. lucidum enhances NKG2D and NCR receptor expression. After treatment with G. lucidum (GL), total cellular mRNA from NK92 cells was prepared for quantification of NKG2D and NCR mRNA levels by real-time PCR (A) and receptor expression by Western blotting (B). Quantification of the receptors on NK92 cells was also monitored by flow cytometry (C). Data are representative of four independent experiments. APC: allophycocyanin; PE: phycoerythrin (*P < 0.05, **P < 0.01, ***P < 0.001).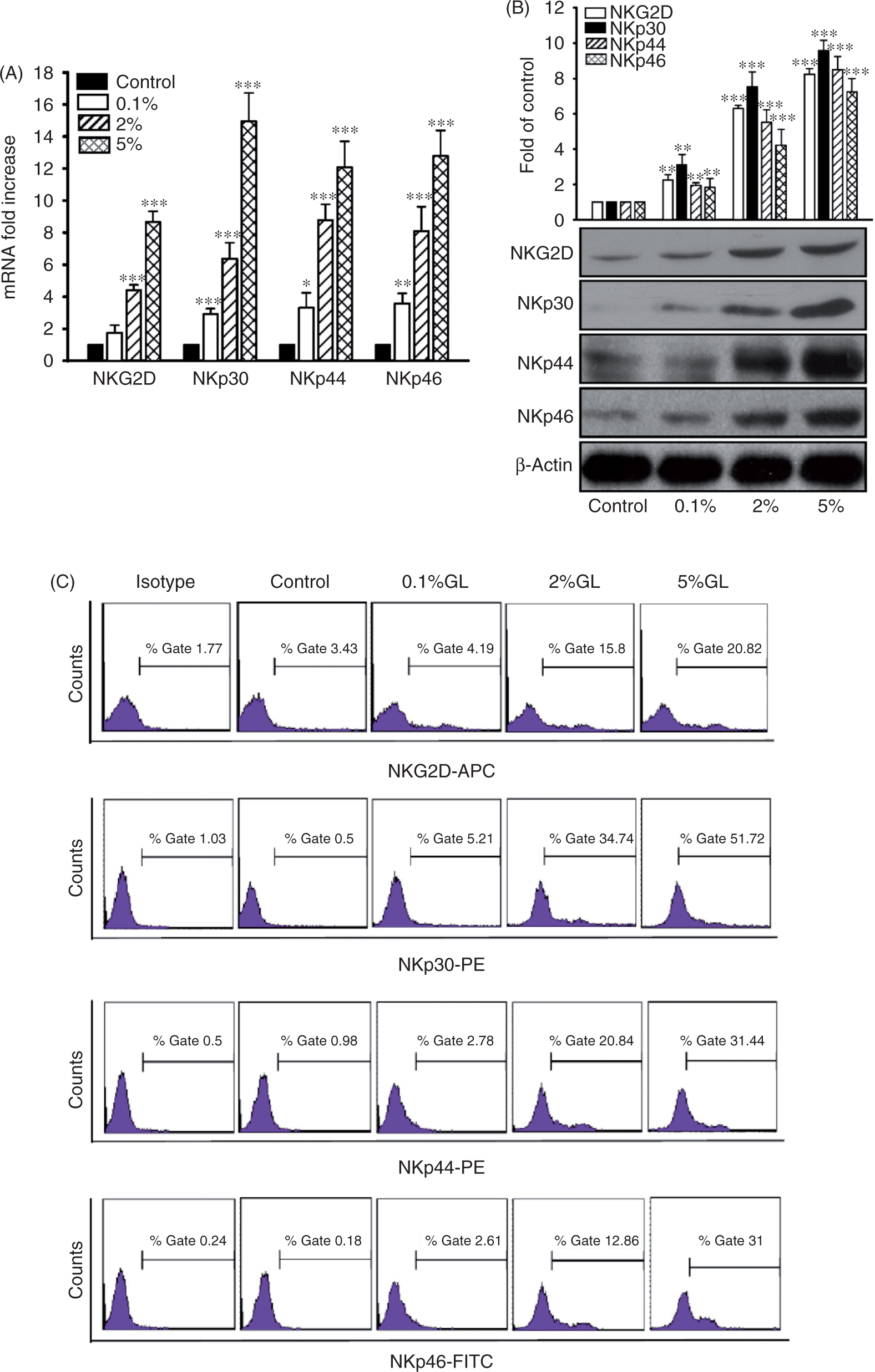
G. lucidum induces intracellular MAPK signaling pathways
Previous studies have shown that MAPK signaling is activated in NK cells during tumor cell lysis.37,41,42 In addition, inhibition of JNK and ERK-1/2 signaling has been shown to interfere with NKG2D-mediated NK cell cytotoxicity.
36
To identify the intracellular pathways modulated by G. lucidum in NK cells, we examined whether the mycelium extract regulated kinase signaling pathways. NK cells were treated with specific doses of G. lucidum prior to Western blotting analysis of MAPKs. As shown in Figure 3A, G. lucidum stimulated the phosphorylation of MAPKs in a dose-dependent manner. To confirm these results, we treated NK cells with specific MAPK inhibitors, and verified the cytotoxic activity of the treated cells. NK cells were treated or not with a MAPK inhibitor followed by treatment with G. lucidum, prior to challenge with target K562 cells at E:T ratios of 1 : 1, 5 : 1 and 10 : 1. Treatment with the MAPK inhibitors did, indeed, reduce the cytotoxicity of G. lucidum-treated NK cells (Figure 3B–D). Consistent with the reduced cytotoxicity, production of perforin and granulysin was reduced in NK cells treated with the MAPK inhibitors (Figure 3E–G).
G. lucidum activates MAPK pathways in NK cells. (A) NK92 cells were treated with G. lucidum (GL) water extract for 30 min before measuring phosphorylation (P) and total (T) protein levels of ERK, JNK and p38. The effects of (B) MAPK inhibitors against JNK (SP, SP600125, 10 µM), (C) ERK1/2 (PD, PD98059, 10 µM), or (D) p38 (SB, SB203580, 10 µM) on the cytotoxic activity of NK cells treated or not with 5% G. lucidum water extract are shown. (E–G) The effects of MAP kinase inhibitors on perforin and granulysin expression in NK cells treated or not with 5% G. lucidum are shown. The results are representative of four independent experiments.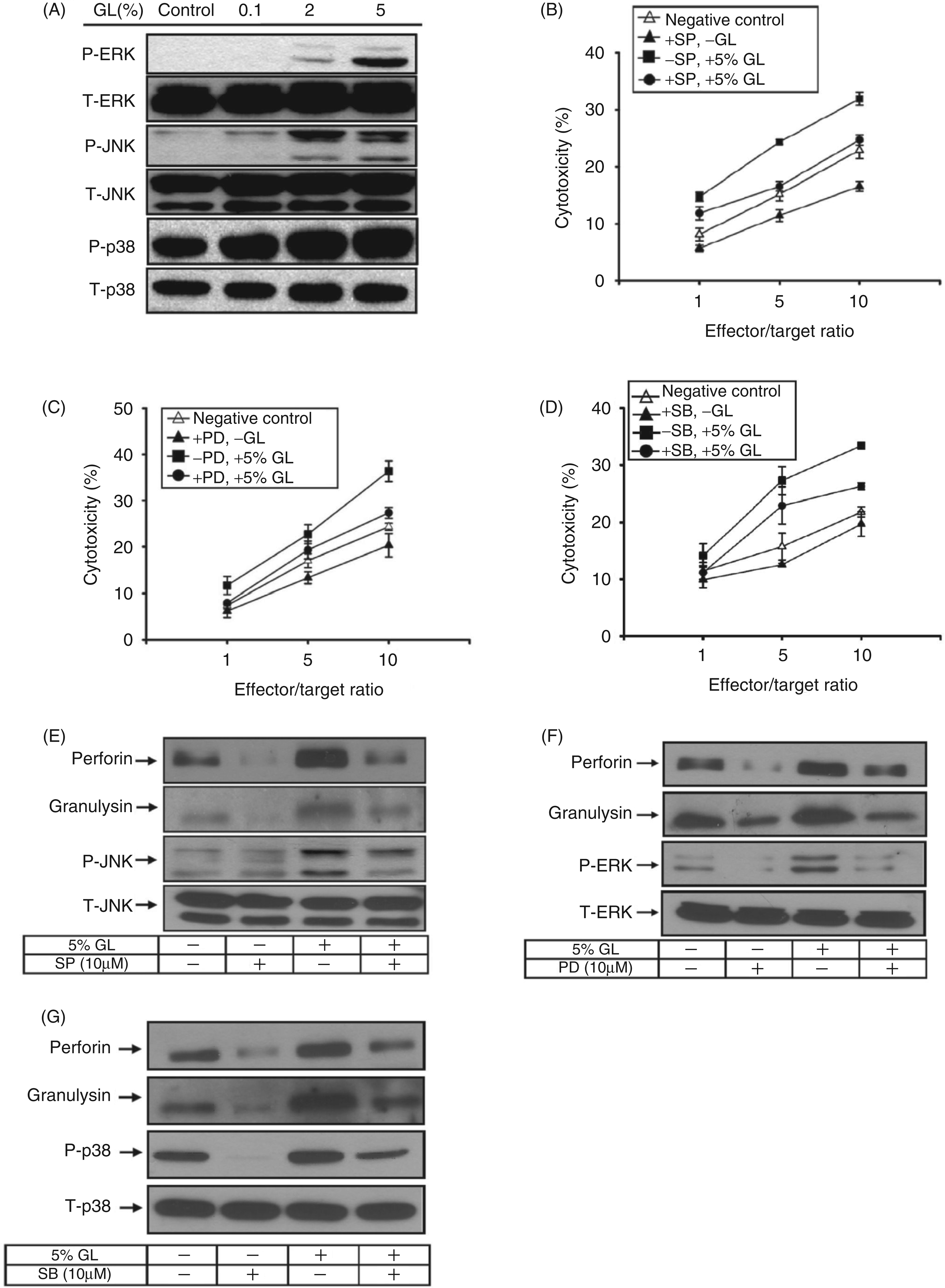
To further confirm that G. lucidum activates NK cell activity through MAPK signaling, we used specific siRNA to deplete MAPKs, and examined whether this treatment affected perforin and granulysin production. Reduced levels of p38, JNK and ERK proteins confirmed that the MAPKs selected were efficiently silenced following siRNA transfection (Figure 4A–C). Notably, the amounts of perforin and granulysin protein were also decreased in G. lucidum-treated NK cells expressing MAPK siRNA. In agreement with these results, transfection of MAPK siRNA in NK92 cells markedly reduced the cytotoxic activity of these cells against the K562 cell line (Figure 4D–F). Based on these findings, we concluded that G. lucidum activates NK cell cytotoxic activity and the release of perforin and granulysin by inducing ERK1/2, JNK and p38 MAPK signaling pathways.
G. lucidum stimulated MAPK activity leads to increased perforin and granulysin production and enhanced cytotoxic activity in NK cells. Effect of specific JNK (A), ERK (B) and p38 (C) siRNA on MAPK expression, perforin and granulysin production in NK cells treated or not with 5% G. lucidum (GL). NK cells were transfected with MAPK siRNA or scramble siRNA followed by stimulation with or without 5% G. lucidum water extract prior to cytotoxicity analysis (D–F). The results shown are representative of three independent experiments. (**P < 0.01, ***P < 0.001).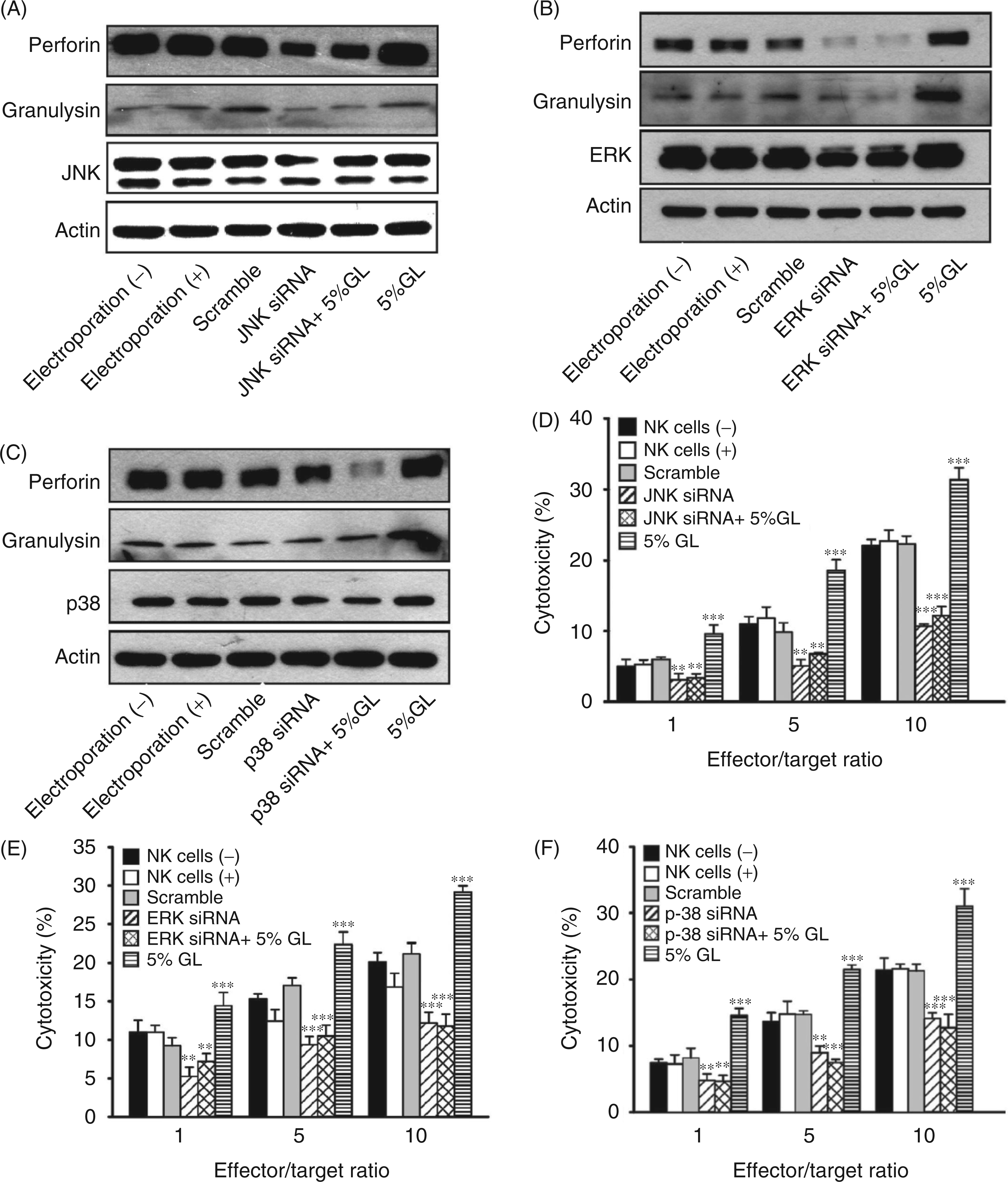
G. lucidum stimulates NK cell cytotoxicity against various cancer cell lines
To evaluate whether G. lucidum treatment increases NK cell cytotoxicity irrespective of the cell line used, we treated NK cells with various doses of G. lucidum, followed by co-culture with other cancer cell lines, including lung adenocarcinoma A549 cells, liver cancer HepG2 cells, breast cancer MCF-7 cells and colon cancer HT-29 cells. Our results showed that G. lucidum stimulated NK cell cytotoxic activity in a dose-dependent manner against all target cancer cell lines tested (Figure 5A–D, E:T ratios of 1 : 1, 5 : 1 and 10 : 1). G. lucidum thus induces NK cell cytotoxicity in a non-specific manner against a variety of tumor cell lines.
Effect of G. lucidum on NK cell cytotoxicity against various target cells. (A–D) NK cells treated with 5% G. lucidum (GL) were incubated with various human cancer cell lines (A549, HepG2, MCF-7 and HT-29) at E:T ratios of 1 : 1, 5 : 1 or 10 : 1. The results shown are representative of three independent experiments.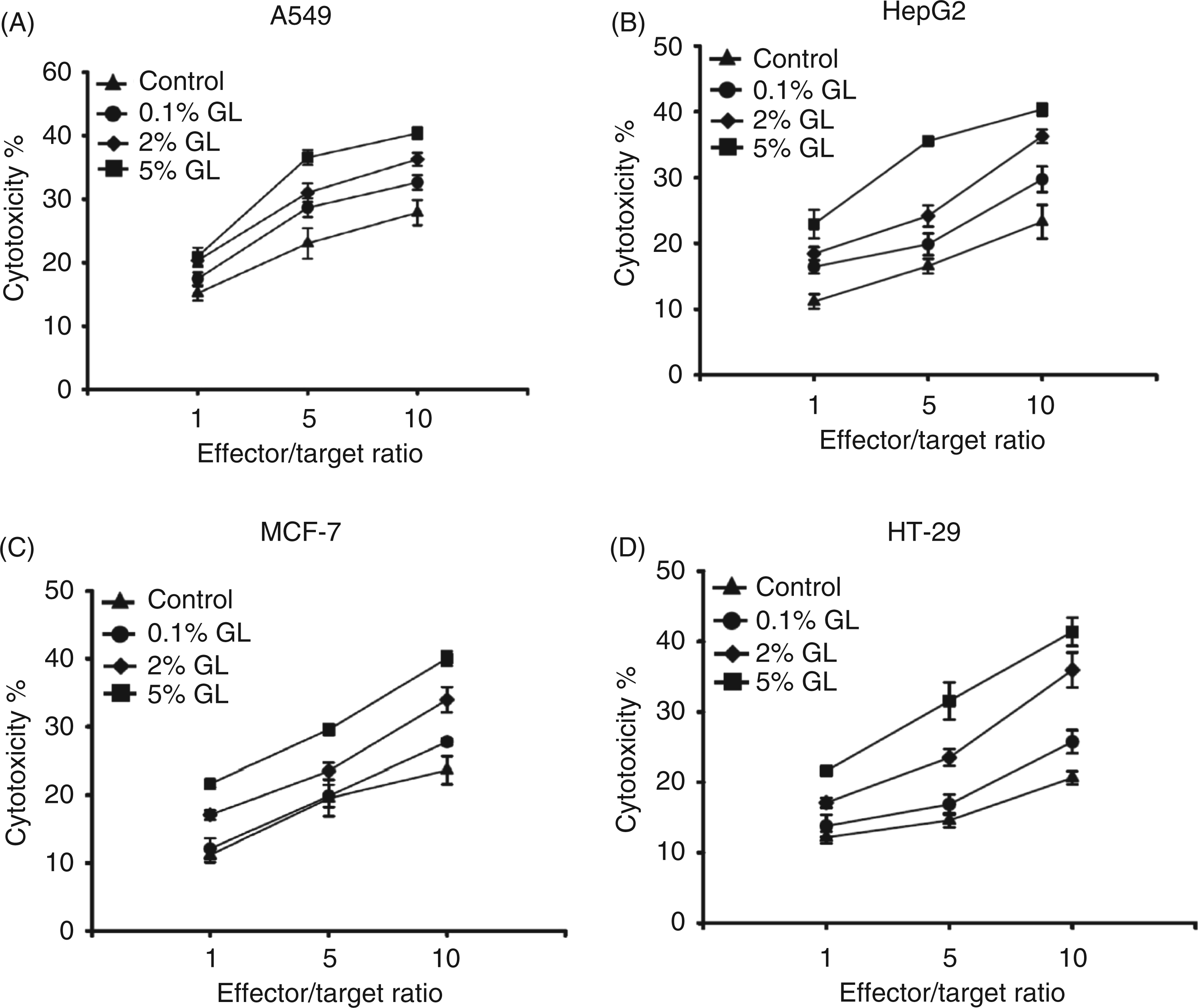
Discussion
In this study, we reported that the underlying mechanism of activation of NK cells by G. lucidum occurs through activation of the NGK2D/NCRs, and p38, JNK and ERK, and MAPK signaling pathways. The enhanced NK cytotoxic activity induced by G. lucidum is effective not only against the human leukemia K562 cells but also against various cancer cell lines (A549, HepG2, MCF-7 and HT-29), indicating that G. lucidum activates NK cells in a non-specific manner. Our results thus indicate that G. lucidum may generally enhance tumor clearance through activation of NK cells. These findings provide a rational basis for the use of G. lucidum water extract as a food supplement for cancer prevention and treatment.
The results of the knockdown assay on the NKG2D and NCRs in this study suggest that the active compounds present in the G. lucidum water extract may directly bind to the activating receptors. Nonetheless, we cannot rule out the possibility of indirect immunomodulation effects occurring from the activation of other signaling pathways that may cross-talk with the NKG2D and NCR pathways. Accordingly, activation of MAPK signaling pathways can be achieved through other interconnected signaling pathways. In order to address this issue, a reporter assay (such as the IFN-γ promoter-based bioluminescence system) may be designed to monitor the activation or inhibition of the NKG2D and NCRs pathways in real time. This reporter assay may also help to identify the active compounds present in the G. lucidum water extract.
We have previously reported that resveratrol, a polyphenolic compound found in red wine, and 2,3-butanediol, a metabolite produced by various organisms, also stimulates the cytotoxic activity of NK cells.38,43 An increase in the expression of NKG2D, MAPK activity and perforin production was observed when NK cells were treated with resveratrol, while NKG2D, NKp40, NKp46, MAPK activity and perforin production were activated by 2,3-butanediol. Compared with resveratrol and 2,3-butanediol, the G. lucidum water extract studied in the present study enhances NKG2D and NCR expression, and MAPK activity, as well as perforin and granulysin production. Thus, the mechanism of NK cell activation of G. lucidum, resveratrol and 2,3-butanediol may be different, an observation which is worthy of further studies.
The G. lucidum water extract may contain a variety of active components, and it may target different cellular components in NK cells. To clarify this issue, future experiments designed to isolate, identify and characterize single active compounds from the water extract will be necessary to obtain a detailed molecular and mechanistic view of G. lucidum compounds. Previous studies indicate that partially purified polysaccharides from G. lucidum enhance human CD56+ NK cells cytotoxicity in vitro 44 and in vivo 6 , including in advanced-stage cancer patients. 45 G. lucidum polysaccharides also inhibit tumor formation by activating host immune responses involving macrophages and T lymphocytes.10,12,13,46 Other studies also indicate that the main activating compounds mediating these effects in G. lucidum water extracts may be polysaccharides. 47 However, we cannot rule out the possibility that other active compounds may also exist in the extract used here. For example, a previous study showed that an active glycoprotein isolated from a water extract of G. lucidum, designated fraction 3 (F3), stimulates mice spleen cell proliferation and cytokine expression. 48 Another study demonstrated that incubation with F3 alone not only increased the cytotoxicity of CD56+ NK-cells against the NK-sensitive tumor-cell line K562 but also altered the expression of cell surface markers. 44 Fractionation of G. lucidum water extracts using various chromatography techniques is currently under way in our laboratory to identify the active compounds responsible for these effects. In addition, we are also examining the effects of ethanol extracts of G. lucidum on NK cell function in order to identify the full range of bioactivities associated with this medicinal mycelium.
In conclusion, we found that a G. lucidum water extract activates NK cell cytotoxicity by activating NKG2D/NCRs and MAPK signaling pathways, and secretion of the cytolytic proteins perforin and granulysin. These observations provide a cellular and molecular mechanism to account for the reported anticancer effects of G. lucidum extracts.19–21 Experiments are currently in progress to isolate, purify and characterize the active components responsible for this activity.
Footnotes
Funding
This study was supported by Primordia Institute of New Sciences and Medicine, grants NSC101-2325-B-182-005 and NSC101-2320-B-030-011 from the National Science Council of Taiwan, and grant CMRPD1B0052 from Chang Gung Memorial Hospital (Linkou, Taiwan).
Conflict of interest
Y.-F.K. is President and an employee of Chang Gung Biotechnology Corporation. J.D.Y. is Chairman of the Board of Chang Gung Biotechnology Corporation. The other authors have no potential conflicts of interest to declare.
