Abstract
NK cells participate in the development of human multiple sclerosis (MS) and mouse experimental autoimmune encephalomyelitis (EAE), but the roles of different NK cell subsets in disease onset remain poorly understood. In this study, murine NK cells were divided into CD27high and CD27low/− subsets. The CD27high subset was decreased and the CD27low/− subset was increased in lymphoid organs during the pre-onset stage of EAE. Compared with the counterpart in naïve mice, the CD27high subset showed lower expression of Ly49D, Ly49H and NKG2D, and less production of IFN-γ, whereas the CD27low/− subset showed similar expression of the above mentioned surface receptors but higher cytotoxic activity in EAE mice. Compared with the CD27high subset, the CD27low/− subset exhibited increased promotion of DC maturation and no significant inhibition of T cells proliferation and Th17 cells differentiation in vitro. Additionally, adoptive transfer of the CD27low/− subset, but not the CD27high subset, exacerbated the severity of EAE. Collectively, our data suggest the CD27 NK cell subsets play different roles in controlling EAE onset, which provide a new understanding for the regulation of NK cell subsets in early autoimmune disease.
Keywords
Introduction
NK cells are large granular lymphocytes. 1 By acting early to identify and kill infected cells, these cells serve as the first line of defense in many diseases. 2 Upon activation, NK cells quickly produce various chemokines and cytokines, including MIP-1α/β, IP-10 (CXCL10), IFN-γ, TNF-α, IL-6, IL-10, TGF-β and GM-CSF. 3 These early source inflammatory mediators serve as a critical link between the innate and adaptive immune response. 4 The function of NK cells is regulated by surface activating and inhibitory receptors such as Ly49D, Ly49H, NKG2D, NKG2A and Ly49C/I. 5 The balance between activating and inhibiting signals determines the action of NK cells in the microenvironment. 6
Human NK cells are defined as CD3−CD56+ lymphocytes. 7 Like T cells and B cells, NK cells are highly heterogeneous cells with diverse receptor repertoires and distinct maturation levels.4,8 Based on the expression of CD56, human NK cells are divided into CD56bright and CD56dim subpopulations. 9 The CD56bright subset shows potent cytokine production capacity, whereas CD56dim subset shows a marked cytotoxic function. 10 Murine NK cells are identified as lymphocytes that express NK1.1 but lack CD3 and CD19 expression. 11 Mouse NK cells are classified into two subsets based on the surface expression of CD27. 12 The murine CD27high NK cell is equivalent to human CD56bright subset and exhibits potent cytokine production and high migratory capacity. The CD27low/− subset produces less cytokine, and is equivalent to human CD56dim subset. 8 The murine CD27high subset is mainly located in the secondary lymphoid organs, and produces IFN-γ for the polarization of Th1 cells.13,14 The CD27low/− NK cells are mainly located in solid organ tissues such as lung and liver, and have a high activation threshold for the high expression of killer immunoglobulin-like receptors such as Ly49C and Ly49I. 8 Despite these known differences, the function of NK cell subsets in disease remains largely unclear.
Multiple sclerosis (MS) is an inflammatory disorder characterized by demyelization and axonal damage. 15 In the blood of patients with active MS, both the percentage and number of CD56dim NK cells are increased. 16 The increased expression of CD94 on CD56dim NK cells suggests a possible role of this NK cell subset in MS pathogenesis. 17 Additionally, IFN-γ production of CD56bright NK cells are substantially diminished. 18 Treatment with daclizumab or IFN-β promotes disease recovery and CD56bright subset expansion in the cerebrospinal fluid (CSF) and blood of patients with MS.19−21 These data suggest the CD56bright subset may contribute to recovery of MS. So far, the effects of different NK cell subsets on MS onset remain unaddressed. We hypothesize that different subsets of NK cells have different functions on disease onset. However, testing of this hypothesis in people before MS onset would be a challenge.
Experimental autoimmune encephalomyelitis (EAE) is a mouse model that well mimics the clinical and neuropathological aspects of MS. 22 Some studies have reported the role of NK cells in the pathogenesis of EAE.23,24 Low disease severity is correlated with increased frequency of CD27high NK cells, and high disease severity is associated with increased frequency of CD27low/− NK cells in EAE. 25 EAE also includes the pre-onset stage which mimics the period before MS onset that involves the activation and expansion of auto-reactive T cells in the periphery.26,27 Thus, elucidation of the functions of NK cell subsets at the pre-onset stage of EAE may help to understand the roles of human NK cell subsets in MS onset.
In this study, the proportion and receptors pattern of the CD27high and CD27low/− NK cells were detected during the pre-onset stage of EAE. The effects of the two NK cell subsets on antigen-presenting cells (APCs) and T cells were then evaluated. In addition, adaptive transfer of the CD27low/− NK cells exacerbated mouse EAE disease progression.
Materials and methods
Mice
C57BL/6 mice (6–8 wk, females) were purchased from SLAC Laboratory Animal Co. Ltd. (Shanghai, China). All mice were housed in the animal facility under specific pathogen-free conditions. All animal experiments were performed under the guidelines and permission of Tongji Medical College Animal Care and Use Committee.
EAE induction and assessment
EAE was induced in mice by subcutaneous injection of 200 µg myelin oligodendrocyte glycoprotein (MOG35–55) peptide (CL Bio-Scientific Co. Ltd, Xi’an, China) emulsified with complete Freund's adjuvant (Sigma, St. Louis, MO, USA) containing heat-killed H37Ra Mycobacterium tuberculosis (5 mg/ml; Difco Laboratories, Detroit, MI, USA). On d 0 and 2 post-immunization, mice were also injected i.p with 200 ng pertussis toxin (Sigma). The clinical scores of EAE were blindly assessed every day as described previously: 28 0, no clinical signs; 0.5, weak tail; 1, complete loss of tail tone; 2, hindlimb paresis; 2.5, one hindlimb paralyzed; 3, both hindlimbs paralyzed; 3.5, forelimb weakness and hindlimb paralysis; 4, forelimbs paralyzed; 5, moribund or death.
Isolation of NK cell subsets and adoptive transfer
CD27 NK cell subsets were selected by a two-step magnetic separation. Briefly, NK cells were enriched from splenocytes of normal mice using the NK Cell Isolation Kit mouse (Miltenyi Biotec, Cologne, Germany). Next, 1 × 107 NK cells/100 μl were incubated with 2 μl biotin conjugated with anti-CD27 mAb (eBioscience, San Diego, CA, USA) for 15 min at 4℃. After washing, 10 μl anti-biotin microbeads (Miltenyi Biotec) were added and incubated for 15 min at 4℃. After washing, cells were magnetically separated into the biotin-labeled CD27high NK cells, and the biotin-unlabeled CD27low/− NK cells. The CD27 NK cell subsets were analyzed by flow cytometry (FCM; purity > 80%). To obtain sufficient numbers of CD27high and CD27low/− NK cell subsets for adoptive transfer, 5–7 mice were sacrificed and total splenocytes were collected and processed. In total, 3.5 × 105 CD27high or CD27low/− NK cells suspended in 200 μl PBS (containing 1% FBS) were transferred per mouse i.v before immunization (d –1). The control group was injected i.v with 200 μl PBS per mouse.
Induction of bone marrow-derived dendritic cells or bone marrow-derived macrophages maturation by NK cell subsets
Bone marrow-derived dendritic cells (BMDCs) or bone marrow-derived macrophages (BMDMs) were propagated from mouse bone marrow as described previously.29,30 The same numbers of CD27high or CD27low/− NK cells were added to the culture medium. About 4 × 104 CD27high or CD27low/− NK cells were co-cultured with 6 × 105 BMDCs or BMDMs per well in 24-well plates. After 48 h, cells were collected for flow cytometric analysis.
T-cell proliferation and Th17 induction in vitro
Naïve T cells (CD3+CD4+CD44lowCD62Lhigh > 90% purity) were selected from the splenocytes of normal mouse using the Naive CD4+ T cell Isolation Kit, mouse (Miltenyi Biotec). Naïve T cells were activated by plate-bound anti-CD3 mAb (Bio X Cell, West Lebanon, NH, USA) and anti-CD28 mAb (Bio X Cell). To explore the role of CD27 NK cell subsets in the proliferation of T cells, 1 × 104 CD27high NK cells or CD27low/− NK cells were co-cultured with carboxyfluorescein succinimidyl ester (CFSE)-labeled naïve T cells for 3 d per well in 96-well plates in complete RPMI medium 1640 (NK: T = 1:10). To determine the effects of CD27 NK cell subsets on Th17 expansion, IL-6 (40 ng/ml; eBioscience) and TGF-β (2 ng/ml; R&D Systems, Minneapolis, MN, USA) were added into the assay system. After co-culture, cells were collected for intracellular FCM, and the supernatants were used for ELISA detection of IL-17A (eBioscience).
FCM
PE/CY7 anti-mouse CD3ɛ (clone, 145-2C11), PE anti-mouse NK1.1 (clone, PK136), APC anti-mouse CD27 (clone, LG.7F9), FITC anti-mouse CD80 (clone, 16-10A1), PE anti-mouse CD86 (clone, GL1), PE/CY7 anti-mouse CD11c (clone, N418), APC anti-mouse MHC-II (clone, M5/114.15.2), APC anti-mouse F4/80 (clone, BM8), PE anti-mouse IFN-γ (clone, XMG1.2) anti-CD27 biotin (clone, LG.7F9) and Alexa Fluor®488 anti-Biotin (BK-1/39) were purchased from eBioscience. PercP/cy5.5 anti-mouse CD4 (clone, RM4-5), APC anti-mouse IFN-γ (clone, B27), APC anti-mouse CD107a (clone, 1D4B), PE anti-mouse IL-17A (clone, SCPL1362), FITC-anti-mouse Ly49H (clone, 3D10), FITC-anti-mouse Ly49D (clone, 4E5), PE-anti-mouse Ly49A (clone, A1) and FITC-anti-mouse Ly49C/I (clone, 5E6) were purchased from BD Biosciences. Nucleated cells were harvested from the spleen, draining lymph nodes (axillary lymph nodes and inguinal lymph nodes) and bone marrow respectively, and stained with fluorescence-conjugated Abs in staining buffer (PBS, 1% FBS). For the detection of IL-17A and IFN-γ, the cells were fixed using Cytofix/Cytoperm (BD Biosciences, San Jose, CA, USA) according to the manufacturer’s instructions. For the detection of the surface expression of CD107a, cells were stimulated with phorbol myfismte acetate and ionomycin and blocked with brefeldin A for 4 h in vitro, and then stained with fluorescence-conjugated Abs without damaging cell membranes. Data were collected using either a LSR II or a FACS Calibur flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA). Data were acquired using FACS Diva (Becton Dickinson) and analyzed using FlowJo 7.6.1 (Tree Star, Ashland, OR, USA).
Immunofluorescence
Frozen sections of spleen and lymph nodes from EAE mice (from d 1 to d 9) were incubated with goat anti-mouse NKp46 (1:200; R&D systems), Armenian Hamster anti-mouse CD11c eFlour® 625 (1:100; eBiosicence), and purified rat anti-mouse F4/80 (1:100; eBiosicence) at 4℃ for 12 h. Next, samples were washed three times with 1 × PBS, and then stained with Alexa Flour 488 rabbit anti-goat IgG (1:200; Invitrogen, Carlsbad, CA, USA) and Alexa Flour 594 donkey anti-rat IgG (1:200; Invitrogen) for 2 h at 37℃. Cells were examined using a laser scanning confocal microscope (Olympus Inc., Tokyo, Japan) and images were acquired using a FV10-ASW 2.1 image visualization system (Olympus).
Statistical analysis
Data were presented as the mean ± SEM. An ANOVA was used to compare the difference between groups. A two-tailed Student’s t-test was used to compare the mean fluorescence intensity (MFI) of CD80, CD86 and MHC-II on BMDCs. The analysis of the receptors expressed on corresponding NK cell subset on d 0 or d 5, and the accumulative scores of EAE were determined by two-way ANOVA. Values of P < 0.05 were considered significant.
Results
CD27low/− NK cells proportion increased and CD27high NK cells proportion decreased in lymphoid organs during the pre-onset stage of EAE
The actively induced EAE can be divided into four stages of pre-onset, onset, peak and chronic.
28
The period from d 1 to 10 after immunization is defined as the pre-onset stage. The frequency of NK cell subsets in lymphoid organs in this stage was examined by FCM. CD3−NK1.1+ lymphocytes were gated as NK cells, and then were classified as CD27high and CD27low/− subsets according to the expression level of CD27 (Figure 1A). The percentage of CD27low/− subset in total NK cells on d 0 (naïve mice) was about 50% in spleen, 30% in lymph nodes and 20% in bone marrow. During the pre-onset stage, the CD27low/− subset frequency increased and the CD27high subset decreased in the spleen, lymph nodes and bone marrow (Figure 1B). In addition, the MFI of CD27 continuously decreased on total NK cells in the three organs (Figure 1C). These data indicated that CD27low/− NK cells became the dominant subset during the development of EAE.
The percentage changes of CD27 NK cell subsets during the pre-onset stage of EAE. (A) Gate strategy of CD27high and CD27low/– NK cells. (B) The percentage of CD27high and CD27low/– subset in total NK cells in the spleen (SP), lymph nodes (LN) and bone marrow (BM). CD27hi: CD27high NK cells; CD27lo/-: CD27low/– NK cells. *p < 0.05, ***P < 0.001 vs. the percentage of CD27low/– subset on d 0, #p < 0.05, ###P < 0.001 vs. the percentage of CD27high subset on d 0. (C) The change of CD27 MFI on NK cells in the spleen (SP), lymph nodes (LN) and bone marrow (BM). *P < 0.05, **P < 0.01, ***P < 0.001 vs. CD27 MFI on d 0. These data were shown as mean ± SEM, n = 4/group and the data are representative of at least three independent experiments.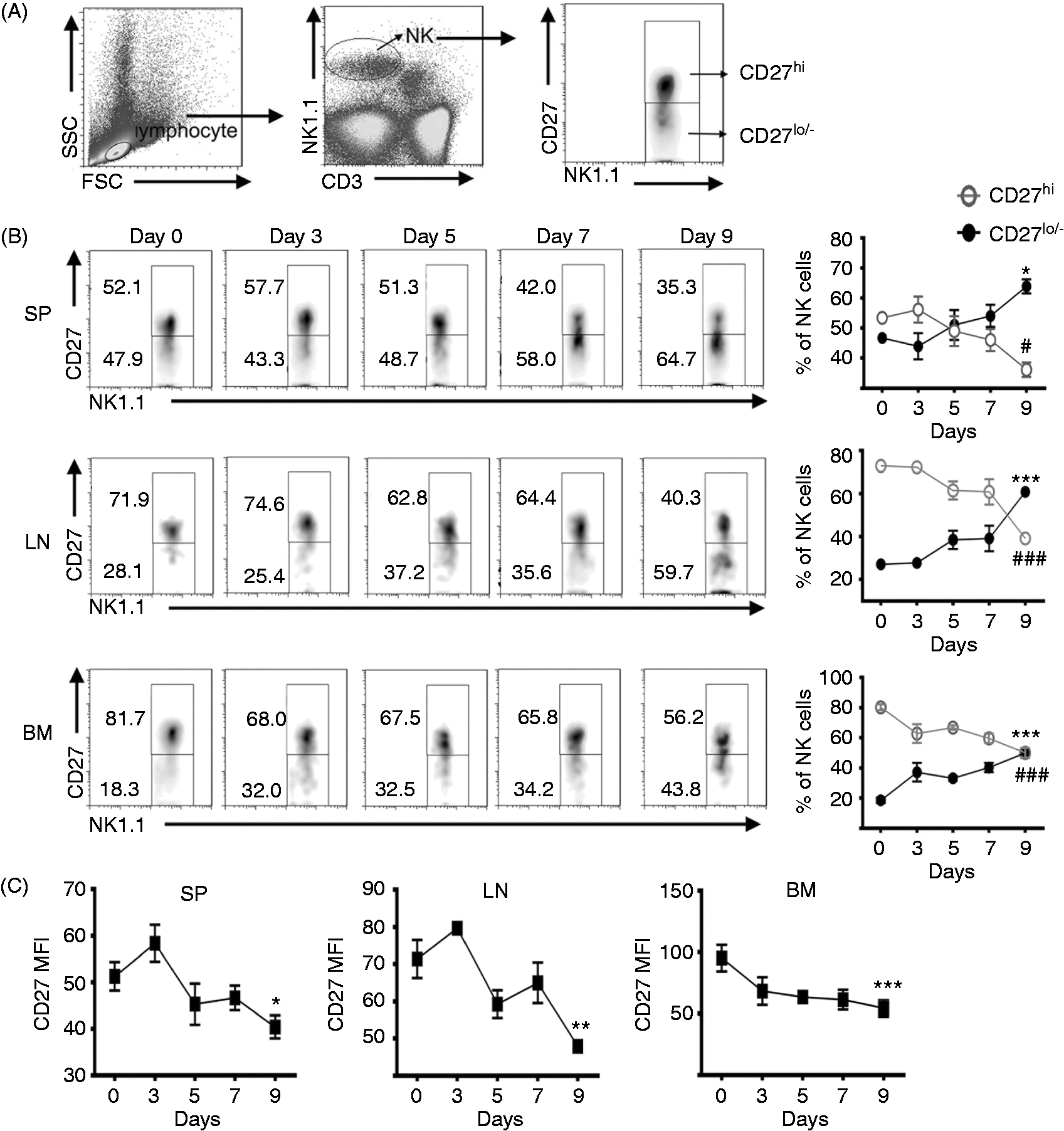
CD27low/− NK cells maintained stable receptors pattern and CD27high NK cells changed to an inhibitory receptor dominant pattern at the pre-onset stage of EAE
The function of NK cells is tightly linked to the pattern of receptors.
5
We therefore examined the expression of surface receptors on the CD27high and CD27low/− NK cell subsets in the spleen by FCM. On the CD27high subset, the activating receptors Ly49D, Ly49H and NKG2D, and inhibitory receptor Ly49C/I were down-regulated, and the activating receptor NKp46 and inhibitory receptors Ly49A and NKG2A had no significant change on d 5 of EAE compared with d 0 (Figure 2A, B). In contrast, receptors on the CD27low/− cells were unchanged between d 5 and d 0 (Figure 2A, B). These data suggested the CD27low/− subset maintained stable activity and the CD27high subset was biased towards less activity during the pre-onset stage of EAE.
Phenotypic changes of NK cell subsets in the spleen at the pre-onset stage of EAE. (A) The percentage of Ly49D, Ly49H, NKG2D, and NKp46-positive cells in CD27 NK cell subsets. (B) The percentage of Ly49C/I, Ly49A, and NKG2A-positive cells in CD27 NK cell subsets. (C) The percentage of IFN-γ+ and CD107a+ cells in CD27 NK cell subsets. (A–C) The data are representative of three independent experiments (mean ± SEM, n = 4/group; CD27hi, CD27high NK cells; CD27lo/-, CD27low/− NK cells; *P < 0.05, **P < 0.01, ***P < 0.001).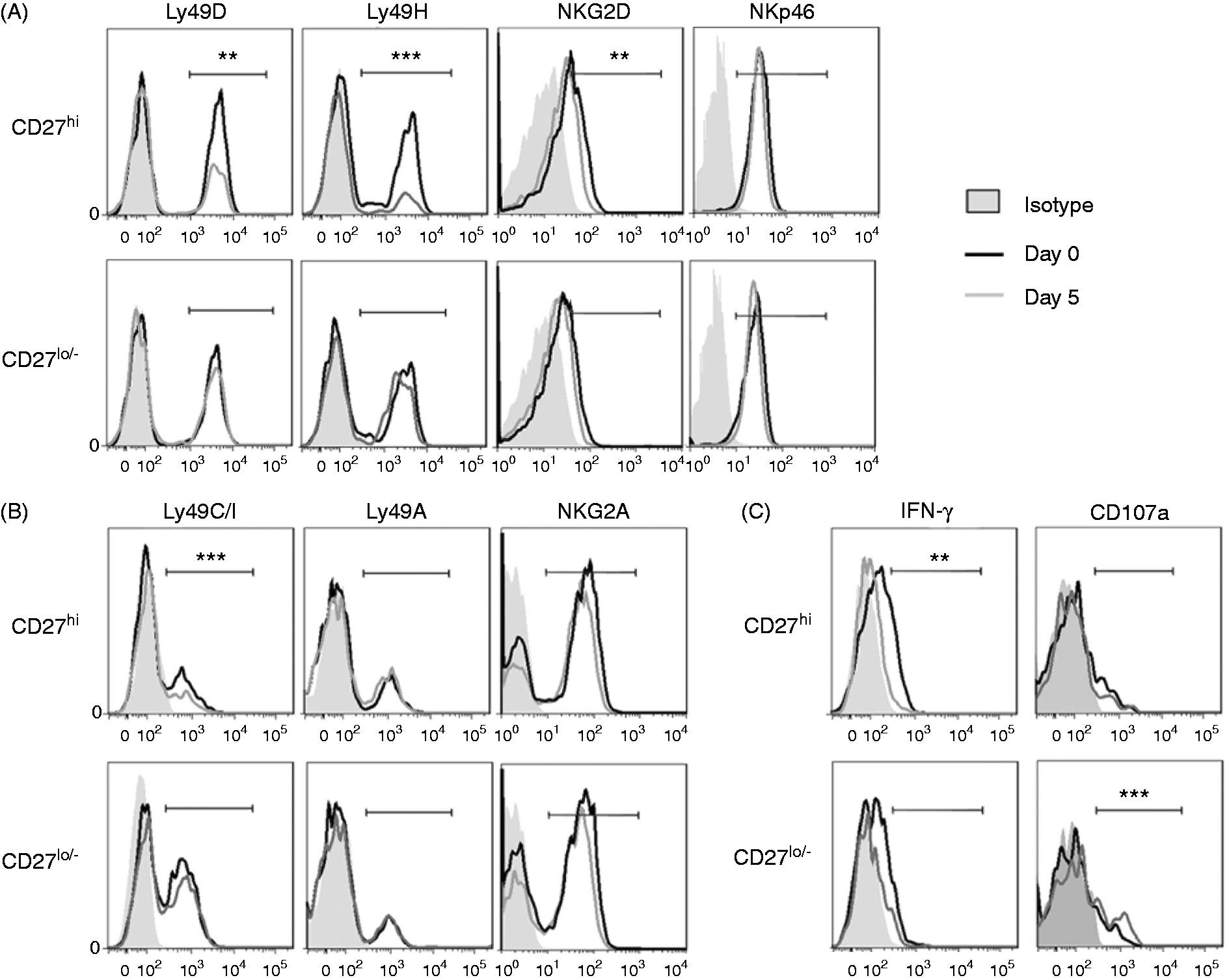
The cytotoxic ability of CD27low/− NK cells increased and the IFN-γ production of CD27high NK cells decreased during the pre-onset stage of EAE
The IFN-γ production and cytotoxicity of the CD27high and CD27low/− subsets in the spleen at the pre-onset stage of EAE were detected by FCM. Compared with d 0, the production of IFN-γ in the CD27high subset decreased, but had no change in the CD27low/− subset on d 5 (Figure 2C, left). Compared with d 0, the surface expression of CD107a, a functional marker that identified NK cells cytotoxic capacity, was not changed on the CD27high subset but was elevated on the CD27low/− subset on d 5 (Figure 2C, right). Thus, the cytokine production ability of CD27high subset was impaired, and the cytotoxic ability of CD27low/− subset was enhanced during the pre-onset stage of EAE.
The CD27low/− subset promoted more maturation of BMDCs compared with the CD27high subset
To investigate whether NK cells interact with DCs, sections of the spleen from EAE mice were stained with fluorescence-conjugated Abs and observed by confocal microscopy. The numbers of NK cells (NKp46 positive, green) and DCs (CD11c positive, red) were notably increased with EAE development. Direct contact between NK cells and DCs was observed from d 1 to d 9. NK cells were surrounded by DCs on d 9 (Figure 3A). These data indicated that there was direct interaction between NK cells and DCs during the pre-onset stage of EAE. To further elucidate the effects of the CD27high or CD27low/− subset on DCs, the same amounts of cells were separately cultured with BMDCs. The maturation of BMDCs was then evaluated by the expression of CD80, CD86 and MHC-II via FCM. The percentage of CD86+ or MHC-II+ cells in BMDCs increased when co-cultured with CD27high NK cells, but the percentage of CD80+ cells and MFI of the three molecules showed no change (Figure 3B, C). In contrast, co-culture with CD27low/− subset significantly increased the percentage of CD80+ or CD86+ or MHC-II+ cells in BMDCs and the MFI of the three molecules (Figure 3B, C). These data indicated that both subsets could promote the maturation of BMDCs in vitro; however, the CD27low/− subset stimulated more maturation of BMDCs.
The effects of CD27 NK cell subsets on the maturation of BMDCs in vitro. (A) The location of NK cells and DCs in the spleen from d 0 to d 9 of EAE (NK cells: NKp46 positive, green; DCs: CD11c positive, red; the magnification was 400×). The areas in white boxes were highly magnified. Arrows point to the contacting NK cells and DCs. Scale bars = 30 µm. (B) The percentage of CD80+, CD86+ and MHC-II+ CD11c+ cells. About 6 × 105 BMDCs were co-cultured with 4 × 104 CD27high or CD27low/– NK cells per well for 48 h in 24-well plates. (C) The histograms of the percentage (left) and MFI (right) of CD80, CD86 and MHC-II expressed on CD11c+ cells. Data in (A) are representative of three independent experiments; data for (B and C) are representative of seven independent experiments; Control, without NK cells; CD27hi, with CD27high NK cells; CD27lo/–, with CD27low/– NK cells; *P < 0.05, **P < 0.01, ***P < 0.001 vs. control group.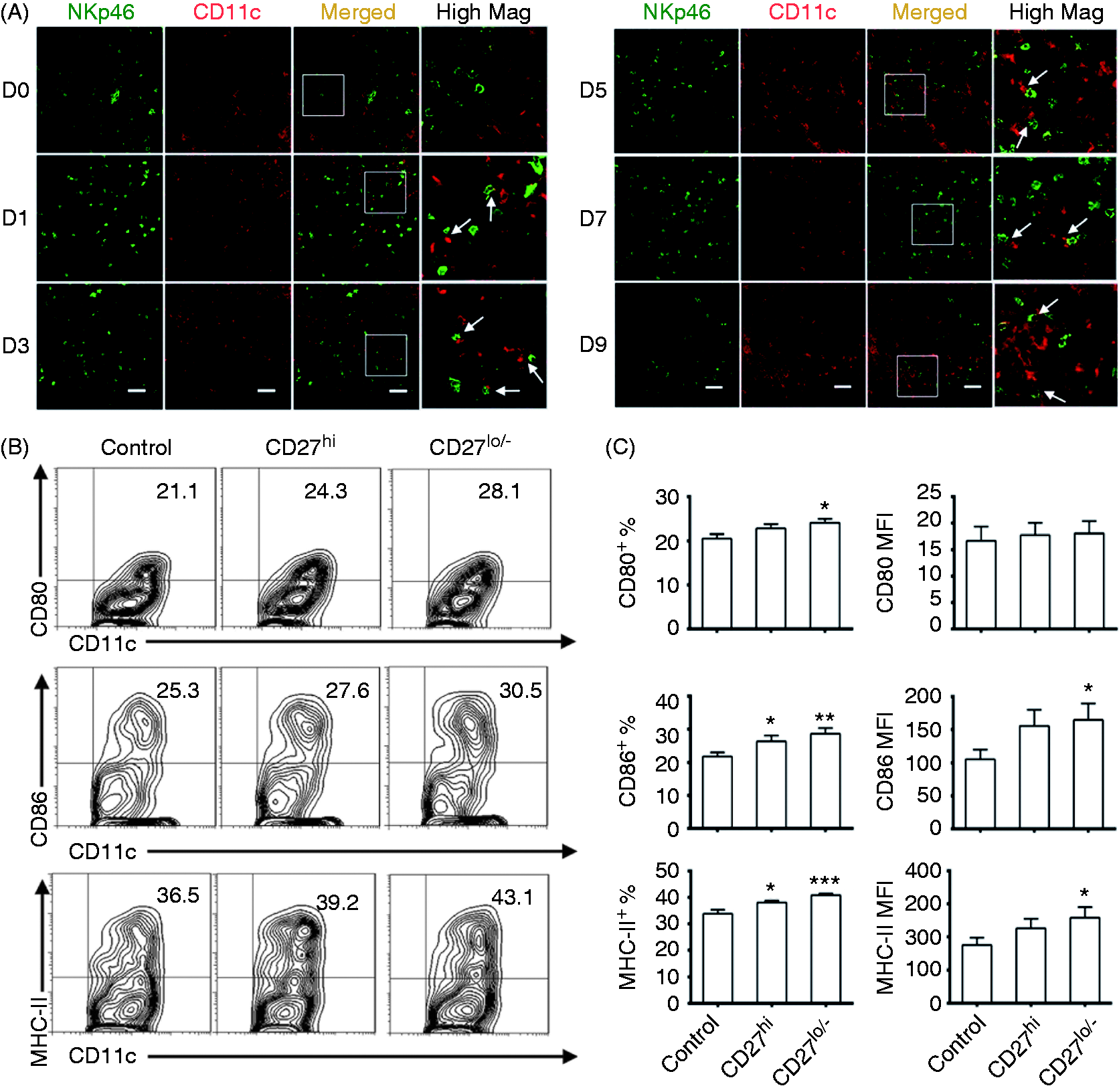
The CD27low/− subset was less able to promote the maturation of BMDMs than the CD27high subset
Macrophages are another important kind of APCs.
31
We next investigated the possible interactions between NK cells and macrophages during the pre-onset stage of EAE. There were only a few NK cells (NKp46 positive, green) and macrophages (F4/80 positive, red) in the spleen on d 0. The numbers of NK cells and macrophages gradually increased, and most NK cells were surrounded by macrophages from d 1 to d 9 (Figure 4A). These data indicated that NK cells interacted with macrophages during the pre-onset stage of EAE. BMDMs were then co-cultured with CD27high or CD27low/− NK cells for 48 h, and F4/80+ cells were analyzed by FCM for the expression of CD80, CD86 and MHC-II. Co-culturing with CD27high subset significantly increased the percentage of CD80+ or MHC-II+ cells in BMDMs, and also elevated the MFI of CD80 and CD86 (Figure 4B, C). However, co-culturing with the CD27low/− subset significantly increased only the percentage of CD80+ cells and MFI of CD80. These data indicated that both subsets promoted maturation of BMDMs, but the CD27low/− subset exhibited a less effect on the maturation of BMDMs in vitro.
The effects of CD27 NK cell subsets on the maturation of BMDMs in vitro. (A) The location of NK cells and macrophages in the spleen from d 0 to d 9 of EAE (NK cells: NKp46 positive, green; macrophages: F4/80 positive, red; the magnification was 400×). The areas in white boxes were highly magnified. Arrows point to contacting NK cells and macrophages. Scale bars = 30 µm. (B) The percentage of CD80+, CD86+, and MHC-II+ F4/80+ cells. In total, 6 × 105 BMDMs were co-cultured with 4 × 104 CD27high or CD27low/– NK cells per well for 48 h in 24-well plates. (C) The histograms of the percentage (left) and MFI (right) of CD80, CD86 and MHC-II expressed on F4/80+ cells. Data for (A) are representative of three independent experiments; Data for (B and C) are pooled from three independent experiments. Control, without NK cells; CD27hi, with CD27high NK cells; CD27lo/–, with CD27low/– NK cells. *P < 0.05, **P < 0.01, ***P < 0.001 vs. control group.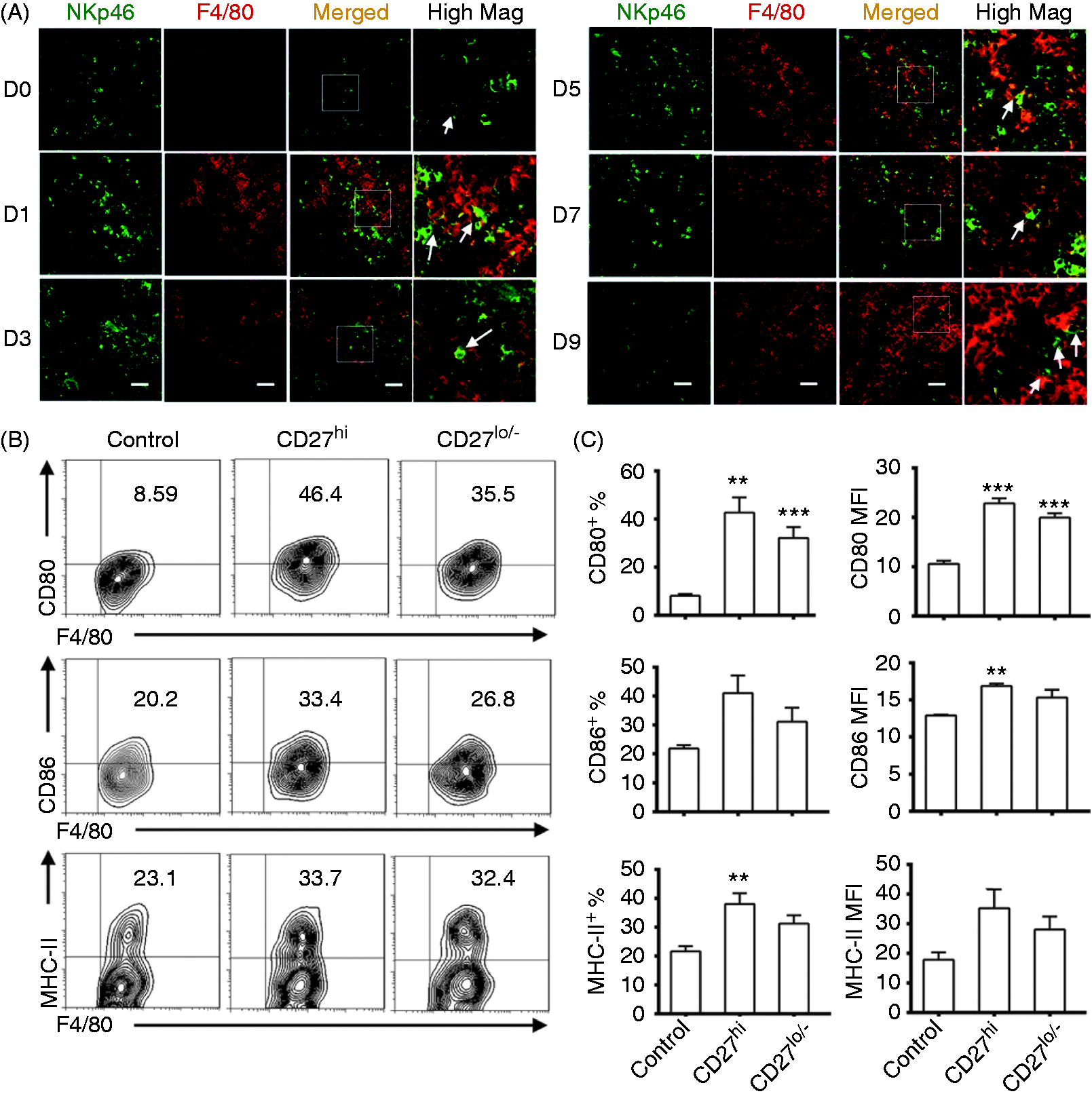
The CD27high subset but not the CD27low/− subset effectively inhibited the proliferation of CD4+ T cells
Auto-reactive CD4+ T cells are involved in the development of EAE.
26
In order to investigate the effects of NK cell subsets on T cells in EAE, CFSE-labeled CD4+ naïve T cells were activated by anti-CD3 mAb and anti-CD28 mAb, and then co-cultured with either CD27high or CD27low/− NK cells. The percentage of divided T cells in the total T cells (divided cells %) and the proliferation speed of T cells (proliferation index) were significantly decreased after 3 d of co-culturing with the CD27high subset, but did not change in response to co-culturing with the CD27low/− subset (Figure 5A, B). These data indicated the CD27high subset effectively inhibited the proliferation of T cells in vitro but the CD27low/− subset did not.
The effects of CD27 NK cell subsets on T-cell proliferation and Th17 differentiation in vitro and the disease score of EAE in vivo. (A) The effects of NK cell subsets on the proliferation of T cells. CFSE-labeled CD4+ naïve T cells were activated by plate-bound anti-CD3 and anti-CD28 Abs and then incubated with CD27high NK cells or CD27low/− NK cells per well for 3 d in 96 well plates (NK:T = 1:10). (B) The histograms of CD4+ T cells proliferation in (A). (C) The effects of NK cell subset on the production of Th17 cells. IL-6 (40 ng/ml) and TGF-β (2 ng/ml) were added to the culturing system of (A). (D) Histograms of Th17 cells of (E) and the IL-17A protein levels in the culturing medium. (E) The effects of transferred CD27 NK cell subsets on EAE. In total, 3.5 × 105 CD27high or CD27low/− NK cells (suspended in 200 μl PBS) were transferred to each mouse i.v on d –1. The clinical scores of EAE were assessed as described in ‘Material and methods’. (F) Clinical features of the mice of (E) were shown. Data for (A) and (C) represent one of three independent experiments. Data for (B) and (D) are pooled from three independent experiments (n = 12 per group; *P < 0.05, **P < 0.01, ***P < 0.001 vs. the group without NK cells; #P < 0.05, ##P < 0.01, ###P < 0.001 vs. the group with CD27high NK cells). Data for (E) and (F) were pooled from two independent experiments (n = 12). The accumulative clinical scores difference among three groups was evaluated using two-way ANOVA (***P < 0.001 vs. control group, ###P < 0.001 vs. CD27high subset transferring group).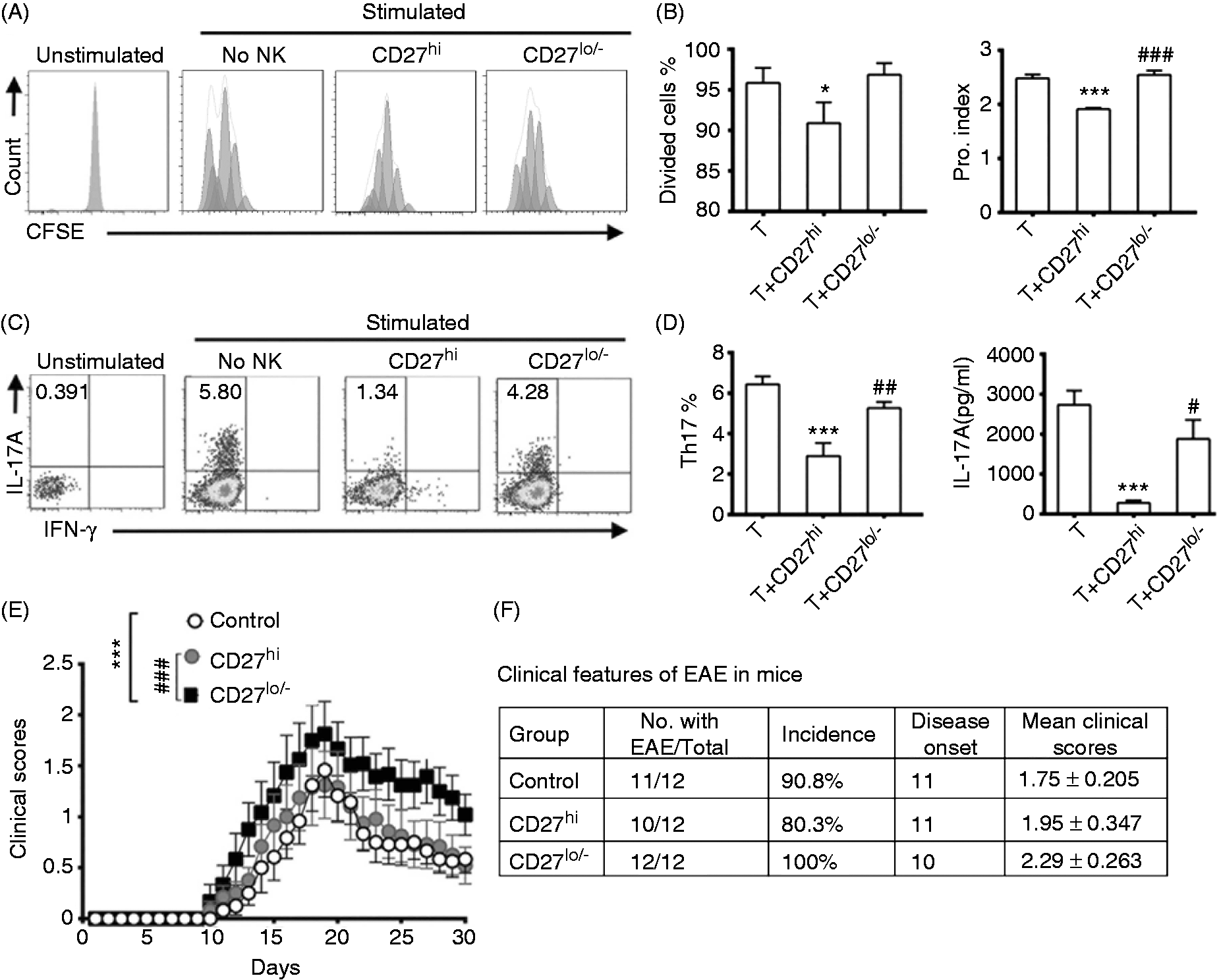
The CD27high subset but not the CD27low/− subset effectively inhibited the differentiation of Th17 cells
To investigate the effects of the two NK cell subsets on Th17 cells differentiation in vitro, IL-6 and TGF-β were added in the co-culture system to induce Th17 polarization.32,33 After culturing, CD3+CD4+ T cells were gated for FCM analysis. In the Th17 polarization condition, the percentage of Th17 cells and the levels of IL-17A in the culture supernatants were significantly decreased when co-cultured with the CD27high subset but were not changed with the addition of CD27low/− subset (Figure 5C,D). These results indicated that CD27high subset directly inhibited the differentiation of Th17 cells but the CD27low/− subset did not.
Adaptive transfer of CD27low/− NK cells aggravated EAE
Finally, to assess the effects of the two NK cell subsets on the disease scores of EAE, the equal numbers of both subsets were sorted by MACS, and were transferred to naïve mice 1 d prior to EAE induction. The mice transferred with the CD27high NK cells showed similar EAE symptoms with the control mice. The symptoms appeared on d 11–12 after EAE induction, and the clinical score peaked around d 19 and then gradually reduced (Figure 5 E,F). However, the mice transferred with CD27low/− cells showed an earlier initiation of EAE symptoms and higher clinical scores from d 10 to d 30 (Figure 5 E, F). These data indicated that the transfer of CD27low/− NK cells aggravated the development of EAE.
Discussion
EAE is an autoreactive Th17 and Th1 cells-involved disease. Myelin components specific Th17 cells are the critical pathogenic cells in disease induction and the inflammatory process. 34 During the pre-onset stage of EAE, APCs, especially DCs, lead to activation and expansion of Th17 cells by presenting the myelin peptides to T cells within secondary lymphoid organs.35,36 Therefore, we investigated the effects of NK cell subsets on APCs and T cells to elucidate the roles of NK cell subsets in the development of EAE.
It has been reported that depleting NK cells with Abs before immunization alleviated EAE. 37 Another report showed that NK cells exhibit an indispensable promoting role for the induction of EAE. 38 These studies suggest that NK cells play an important role during the pre-onset stage of EAE. In this study, we found that CD27high and CD27low/− NK cell subsets appeared discriminative characteristics and might play different roles during the pre-onset stage of EAE.
At the pre-onset stage of EAE, the CD27low/− subset became the dominant subset in the spleen, lymph nodes and bone marrow, and maintained a stable pattern of receptors. These results suggested the CD27low/− subset became more important during the development of EAE. In addition, the CD27low/− subset promoted DCs maturation more effectively. Therefore, the CD27low/− subset was involved in the development of EAE by increasing the number, promoting DC maturation, but not directly affecting the proliferation of CD4+T cell and the differentiation of Th17 cells. Based on the aggravation of EAE by CD27low/− NK cells transfer we concluded that the subset was pathogenic, although a previous report described the CD27low/− NK cells as resting cells. 8 Since the transferred mice still had endogenous NK cells in this study, the role of the CD27low/− NK cells in the development of EAE needed to be further confirmed using a passive EAE model, such as transferring encephalitogenic T cells together with NK cell subsets into cytokine receptor common gamma chain-deficient mice that lack NK cells.
Although the CD27high subset displayed greater inhibition of T-cell proliferation and Th17 differentiation in vitro in this study, as reported by other researcher CD27high NK cells mainly acted to suppress immune responses, 39 the number and the activity of this subset were decreased at the pre-onset stage of EAE. This might be why we observed no effects of the transfer of the CD27high subset on the clinical scores of EAE. Other reasons might include that the amounts of transferred cells were not sufficient, and the cells suppressive functions had been impaired in vivo.
NK cells are regarded as a therapeutic target of MS. 21 Importantly, the majority of human CD27− NK cells were found to be CD56dim, and a large proportion of CD27+ NK cells were CD56bright. 40 Murine CD27 NK cell subsets showed many similarities to their human CD56 counterparts. 8 We concluded that human CD56dim or CD27low/− NK cells were pathogenic cells in MS, which required future investigation.
In conclusion, this study identified different roles of CD27 NK cell subsets at the pre-onset stage of EAE, depending on their alterations, and their effects on pathogenic cells. Importantly, we provided new insights into NK cell subsets regulation in early EAE and MS, and new explanations for the diversity of roles of NK cells in EAE. Also, our study provides a certain reference value for the regulation of the human CD56 NK cell subsets in controlling MS onset.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Sciences Foundation of China to Fang Zheng (No.81072466) and the Major State Basic Research Development Program of China to Feili Gong (973 Program; Grant No. 2013CB530505).
