Abstract
In the present study we used a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced Parkinson's disease (PD) mouse model to analyze resveratrol neuroprotective effects. The MPTP-induced PD model is characterized by chronic inflammation, oxidative stress and loss of the dopaminergic (DA) neurons in the substantia nigra pars compacta (SNpc). We observed that resveratrol treatment significantly reduced glial activation, decreasing the levels of IL-1β, IL-6 and TNF-α, as well as their respective receptors in the SNpc of MPTP-treated mice, as demonstrated by Western blotting, RT-PCR and quantitative PCR analysis. This reduction is related to possible neuroprotection as we also observed that resveratrol administration limited the decline of tyrosine hydroxylase-immunoreactivity induced in the striatum and SNpc by MPTP injection. Consistent with these data, resveratrol treatment up-regulated the expression of the suppressor of cytokine signaling-1 (SOCS-1), supporting the hypothesis that resveratrol protects DA neurons of the SNpc against MPTP-induced cell loss by regulating inflammatory reactions, possibly through SOCS-1 induction.
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by tremor, rigidity and bradykinesia. The symptoms of PD are due to a loss of at least 50% of the dopaminergic (DA) neurons in the substantia nigra pars compacta (SNpc), leading to a reduction by over 80% in dopamine levels in the striatum.1,2 The observation that the degree of terminal loss in the striatum appears to be more pronounced than the magnitude of SNpc DA neuron loss 3 suggests that striatal DA nerve terminals are the primary target of the degenerative process and that neuronal death in PD may result from a ‘dying back’ process. 4 Activated astroglia and microglia have been found in the striatum and the SNpc in PD.5,6 In particular, microglia are resident immunocompetent and phagocytic cells in the central nervous system (CNS), and are thought to mediate the innate defense system, thus playing a critical role in normal CNS function. 7 Classically, activated microglia secrete high levels of the pro-inflammatory cytokines TNF-α and IL-1β, and potent reactive radicals, including the superoxide anion and NO. In this respect, pro-inflammatory cytokines, such as TNF-α, IL-1β and IL-6, are increased in cerebrospinal fluid (CSF) and in the striatum of patients with PD.8,9 In addition, up-regulation of inducible NO synthase and cyclooxygenase-2 (COX-2)-containing amoeboid microglia has been observed in the SNpc of PD patients. 10
Suppressor of cytokine signaling (SOCS) proteins are intracellular, cytokine-inducible proteins that inhibit cytokine signaling in numerous cell types, including cells of the immune system and CNS. Several
In a previous study conducted in an experimental 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) PD mouse model, we observed an increased expression of IL-1β, TNF-α, IL-6 and their respective receptors (IL-1βRI, TNF-αRI, IL-6Rα) in the SNpc in comparison with untreated mice, suggesting a role for both pro-inflammatory cytokines and their receptors in the pathogenesis of PD. 20
In the present study, we report that in MPTP-treated mice, resveratrol administration significantly reduced glial activation, the levels of pro-inflammatory cytokines and their receptors in SNpc, protecting mice from MPTP-induced substantia nigra tyrosine hydroxylase (TH)-immunoreactive neuron loss.
Consistently with these data, resveratrol treatment up-regulated the expression of SOCS-1, supporting the hypothesis that resveratrol protects SNpc DA neurons against MPTP-induced cell death by modulating inflammatory reactions.
Materials and methods
Animals and treatment
This study was carried out in strict accordance with the European Council Directive 86/609/EEC and the Italian animal welfare legislation (art. 4 and 5 of D.L. 116/92).
Twenty-four adult male C57BL/6 N mice (22–24 g body mass, 8–10 wks of age) purchased from Harlan, Italy, were kept under environmentally controlled conditions (ambient temperature: 20 ± 2℃; humidity: 50 ± 5%) on a 12 h light/dark cycle with food and water
MPTP is oxidized in the brain to produce its biologically active metabolite MPP+, which is selectively taken up into dopaminergic neurons by dopamine transporters. Administration of MPTP in mice leads to selective destruction of substantia nigra DA neurons and consequent dopamine depletion in the nigrostriatal pathway.24,25
On d 8, six mice of each group were given four i.p. injections of the neurotoxin MPTP (20 mg/kg), for a total of four doses over an 8 h period, and killed 14 d later. 26 Control mice were treated with sterile saline solution. Mice were deeply anesthetized and killed by cervical dislocation for immunoblotting and RT-PCR assays.
Immunohistochemistry
For immunohistochemical staining, mice were transcardially perfused with tris-buffered saline (pH 7.6) followed by 4% paraformaldehyde in PBS pH 7.4 at 4℃. Brains were subsequently post-fixed in the same fixative, then transferred to 30% sucrose in 0.1 M PBS until they sank, for cryoprotection. Finally, the tissues were kept at −80℃ in sucrose solution until sectioning. The fixed tissues were embedded in a cryostat cutting medium, frozen at −20℃ and coronal sections of 20 µm thickness were cut on a cryostat. Immunohistochemistry was performed following a standard avidin–biotin complex procedure. Briefly, specimens were incubated with a mouse primary mAb anti-TH at a ratio of 1 : 1500 (Millipore, Milan, Italy), a mouse mAb anti-glial fibrillary acidic protein (GFAP) at a ratio of 1 : 1000 (Millipore), or a mouse mAb anti-CD11b at a ratio of 1 : 20 (Developmental Studies Hybridoma Bank, Iowa City, IA, USA) overnight and then with an anti-mouse biotinylated secondary Ab (Dako, Milan, Italy) at a 1 : 1000 dilution for 1 h. To visualize the formation of the antigen–Ab complex, sections were incubated for 1 h with extravidin peroxidase (Sigma, Milan, Italy) diluted 1 : 1500, and color development was obtained with 3,3′-diaminobenzidine.
Immunoblotting assays
Following sacrifice, each animal’s brain was rapidly removed and the ventral midbrain containing the SNpc was dissected out with the aid of a stereomicroscope according to Jackson-Lewis and Przedborski. 26 Tissues were stored at −80℃ until analysis.
SNpc from mice was minced in ice-cold PBS, washed and then homogenized in a buffer containing lysis buffer (50 mM Tris pH 8, 0.02 g/ml NaCl, 0.2% SDS, 1% Triton-X, 4 U/ml aprotinin, 2 μM leupeptin, 100 μM phenylmethanesulfonylfluoride). Lysates were centrifuged at 13,000
RT-PCR and real-time PCR
Specific primers.
One microliter of cDNA was amplified in 25 μl of PCR solution (11.5 μl of cDNA solution in water, 1 μl of primer sets and 12.5 μl of PowerSYBR Green PCR Master Mix; Invitrogen) in a 7500 Real-time PCR System (Applied Biosystems, Monza, Italy), and fluorescence was monitored at each cycle. Cycle parameters were 95℃ for 15 min to activate Taq, followed by 40 cycles of 95℃ for 15 s, 55℃ for 1 min and 72℃ for 1 min. Serial dilutions of cDNA from the same source as samples were used to obtain a standard curve. The individual targets for each sample were quantified by determining the cycle threshold and by comparison with the standard curve. The relative amount of the target mRNA was normalized to the level of GAPDH mRNA.
Densitometric analysis
The visualized bands, obtained after immunoblotting and RT-PCR experiments, were submitted to densitometric analysis using the id image analysis software (Kodak Digital Science). β-Actin and GAPDH were used for normalization of immunoblotting and RT-PCR products respectively. Results (means ± SD) were expressed as relative optical measured density.
Statistical analysis
Statistical analysis of experimental data was performed using Student’s
Results
Resveratrol reduced glial activation and neuronal damage
TH immunoreactivity
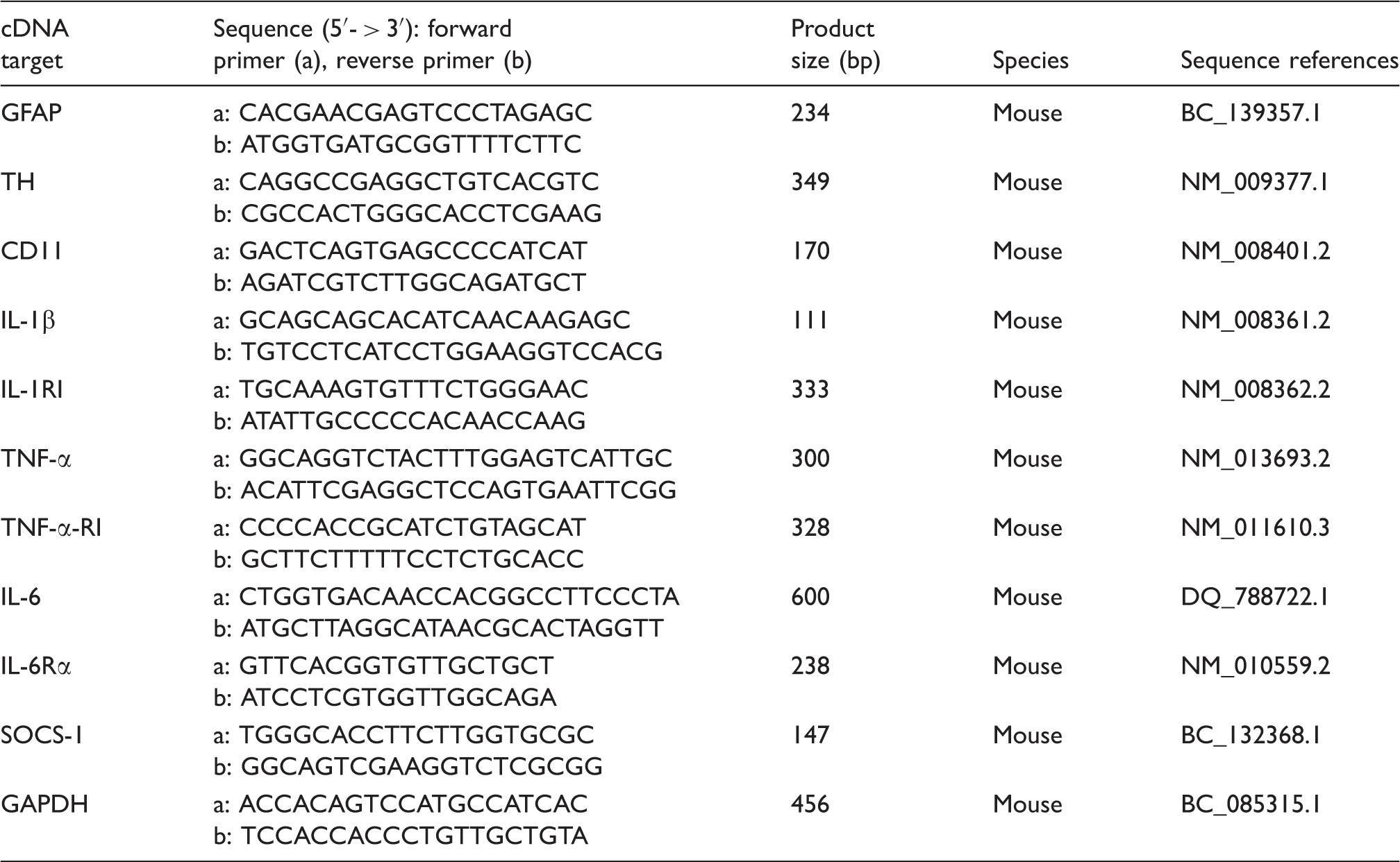
The neuroprotective action of resveratrol on SNpc DA neurons was assessed by mapping TH, the rate-limiting enzyme for DA biosynthesis (Figure 1). Results of TH staining (Figure 1A) showed a marked TH-immunoreactivity decrease in striatum and SNpc following MPTP administration in comparison to controls. Conversely, TH immunostaining in MPTP-treated mice that received resveratrol by intragastric gavage was more pronounced than in those that received MPTP and vehicle, suggesting that resveratrol protects, in part, SNpc DA neurons against MPTP-induced cell death. Resveratrol-treated animals exhibited a TH immunoreactivity pattern similar to controls. Results of the densitometric analysis of TH protein expression, reported in Figure 1B showed that TH loss was significantly reduced ( (A) TH immunoreactivity in the striatum (upper) and SNpc (lower) in control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. Scale bars = 1 mm (upper) and 500 µm (lower). (B) Densitometric analysis of TH protein expression in the SNpc of control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. (C) RT-PCR analysis of TH transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene. (D) Real-time PCR analysis of TH mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. For TH protein expression analysis values represent the relative OD (optical density) after normalization against β-actin expression. For TH mRNA real-time PCR analysis values represent the TH mRNA fold changes relative to GAPDH used as resident control. All values are expressed as means ± SD of five independent experiments. *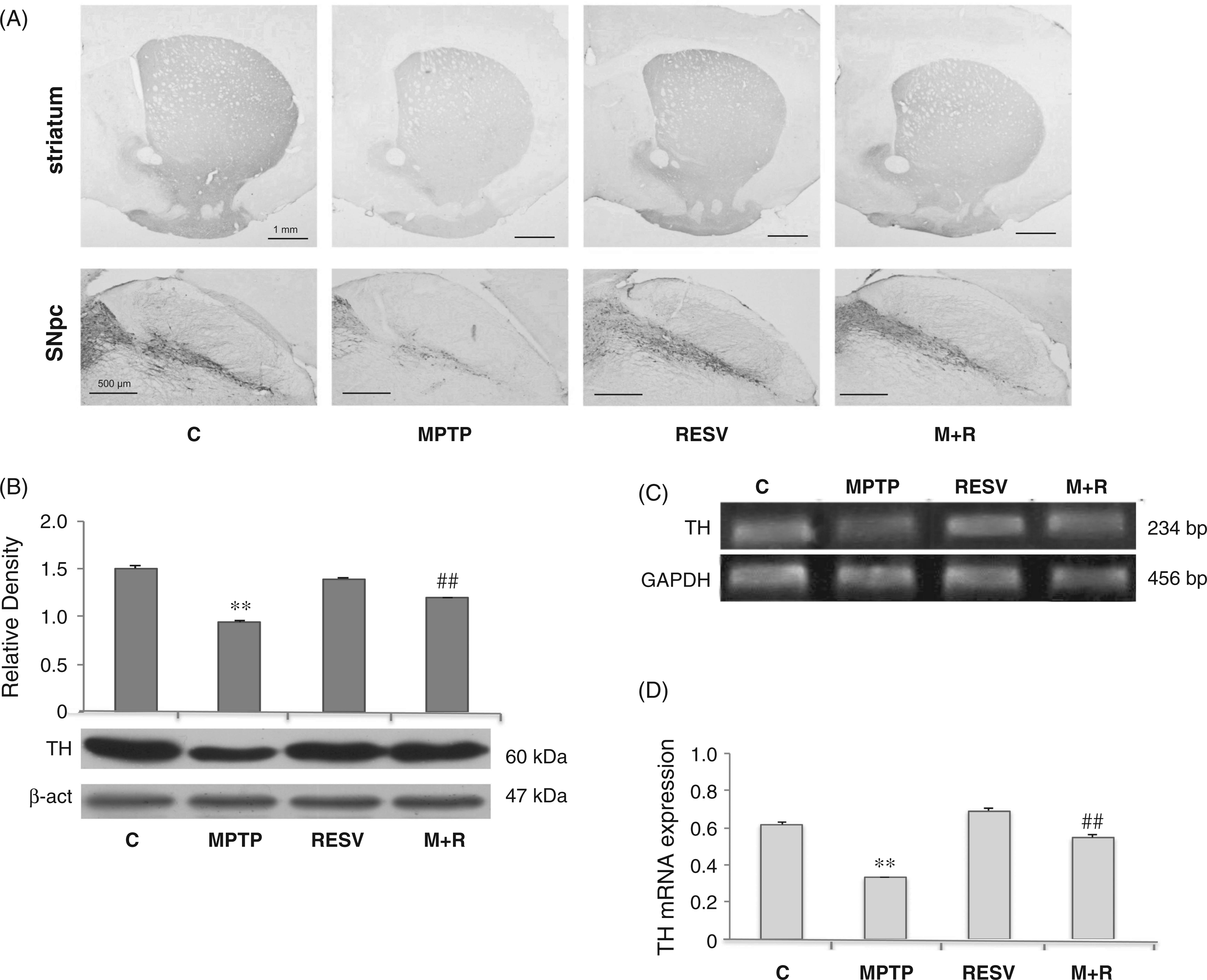
GFAP expression
Astroglial activation was characterized by means of GFAP expression analysis (Figure 2). MPTP treatment determined an increase of immunoreactive cell bodies (Figure 2A), suggesting astrocyte activation, while the administration of resveratrol reduced the presence of GFAP immunoreactive cells; in fact, GFAP expression was augmented after MPTP administration (Figure 2B, (A) GFAP immunoreactivity in the brain of control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. Scale bars = 1 mm (upper) and 200 um (lower). (B) Densitometric analysis of GFAP protein expression in the SNpc of control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. (C) RT-PCR analysis of GFAP transcripts from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene. (D) Real-time PCR analysis of GFAP mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. For GFAP protein expression analysis values represent the relative OD after normalization against β-actin expression. For GFAP mRNA real-time PCR analysis values represent the GFAP mRNA fold changes relative to GAPDH used as resident control. All values are expressed as means ± SD of five independent experiments. *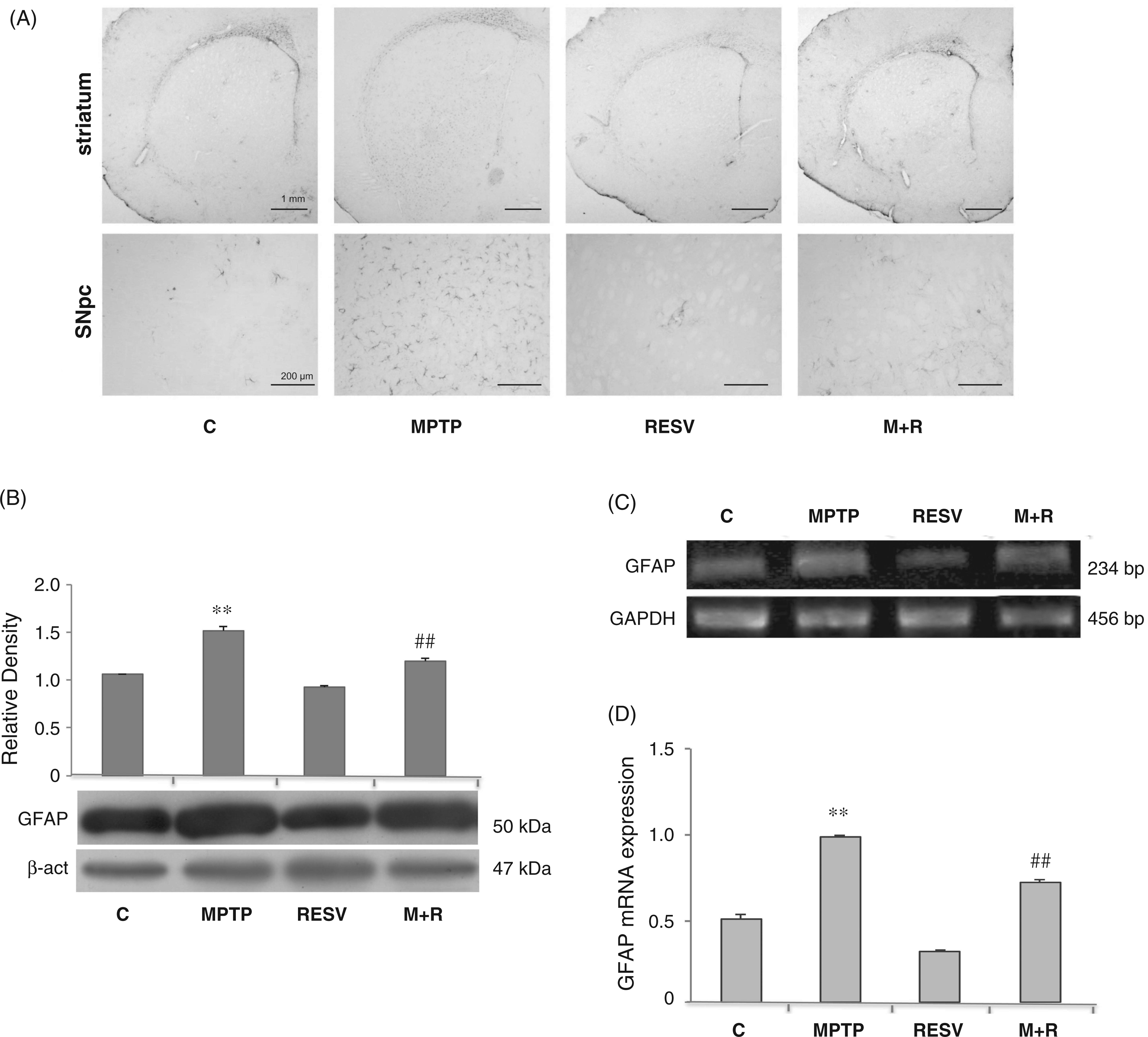
CD11 expression
In Figure 3, CD11 immunoreactivity, a marker of activated microglia, in the SNpc of control and MPTP-treated mice, is shown (Figure 3A). The intensity of CD11-immunoreactivity was augmented following MPTP administration, suggesting microglial activation. In MPTP-treated mice SNpc CD11-positive cells were more numerous, showing a more intense immunoreactivity, as well as a ramified phenotype. Resveratrol administration determined the reduction of CD11-immunoreactivity in MPTP-injected mice, suggesting that resveratrol reduces, at least in part, the microglial activation induced by the neurotoxic insult. CD11 expression was also detected by immunoblotting and RT-PCR analysis at the level of the SNpc. Densitometric analysis of the protein bands obtained shows that CD11 levels were significantly reduced in the SNpc of MPTP-treated mice, which were administered resveratrol in comparison to MPTP-treated mice (Figure 3B). Similarly, CD11 transcripts expression was reduced in animals that received resveratrol in comparison to animals treated with MPTP alone (Figure 3C). These data were confirmed by real-time PCR analysis; in fact, the mRNA levels were significantly ( (A) Representative microphotographs of CD11 brain slices immunostaining in control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. Scale bars = 200 μm (upper) and 10 μm (lower). (B) Densitometric analysis of CD11 protein expression in the SNpc of control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. (C) RT-PCR analysis of CD11 transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene. (D) Real-time PCR analysis of CD11 mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. For CD11 protein expression analysis values represent the relative OD after normalization against β-actin expression. For CD11 mRNA real-time PCR analysis values represent the CD11 mRNA fold changes relative to GAPDH used as resident control. All values are expressed as means ± SD of five independent experiments. *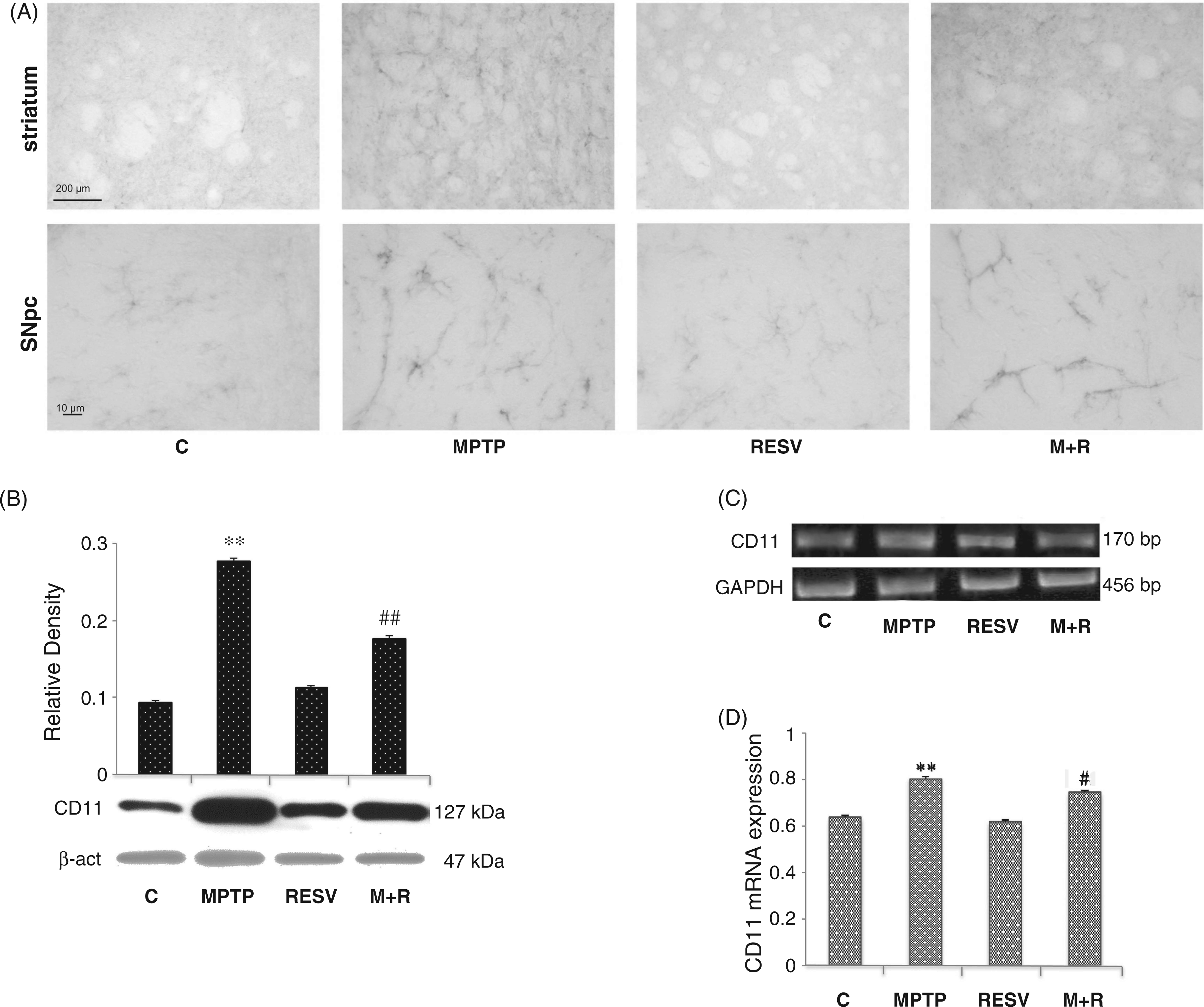
Resveratrol down-regulated the expression of pro-inflammatory cytokines in MPTP-treated mice
IL-1β analysis
IL-1β was detectable by RT-PCR analysis of the transcripts at the level of the SNpc in all animals tested. In the MPTP-treated mice there was an increase of mRNA transcripts in the SNpc (Figure 4A, upper). Real-time PCR revealed that resveratrol administration in MPTP-treated animals determined a significant reduction ( Analysis of IL-1β, TNF-α and IL-6 pro-inflammatory cytokine mRNA expression. (A) RT-PCR analysis of IL-1β transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene (upper). Real-time PCR analysis of IL-1β mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (lower). (B) RT-PCR analysis of TNF-α transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene (upper). Real-time PCR analysis of TNF-α mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (lower). (C) RT-PCR analysis of IL-6 transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene (upper). Real-time PCR analysis of IL-6 mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (lower). Values represent the IL-1β, TNF-α and IL-6 mRNA fold changes relative to GAPDH used as resident control and expressed as means ± SD of five independent experiments. *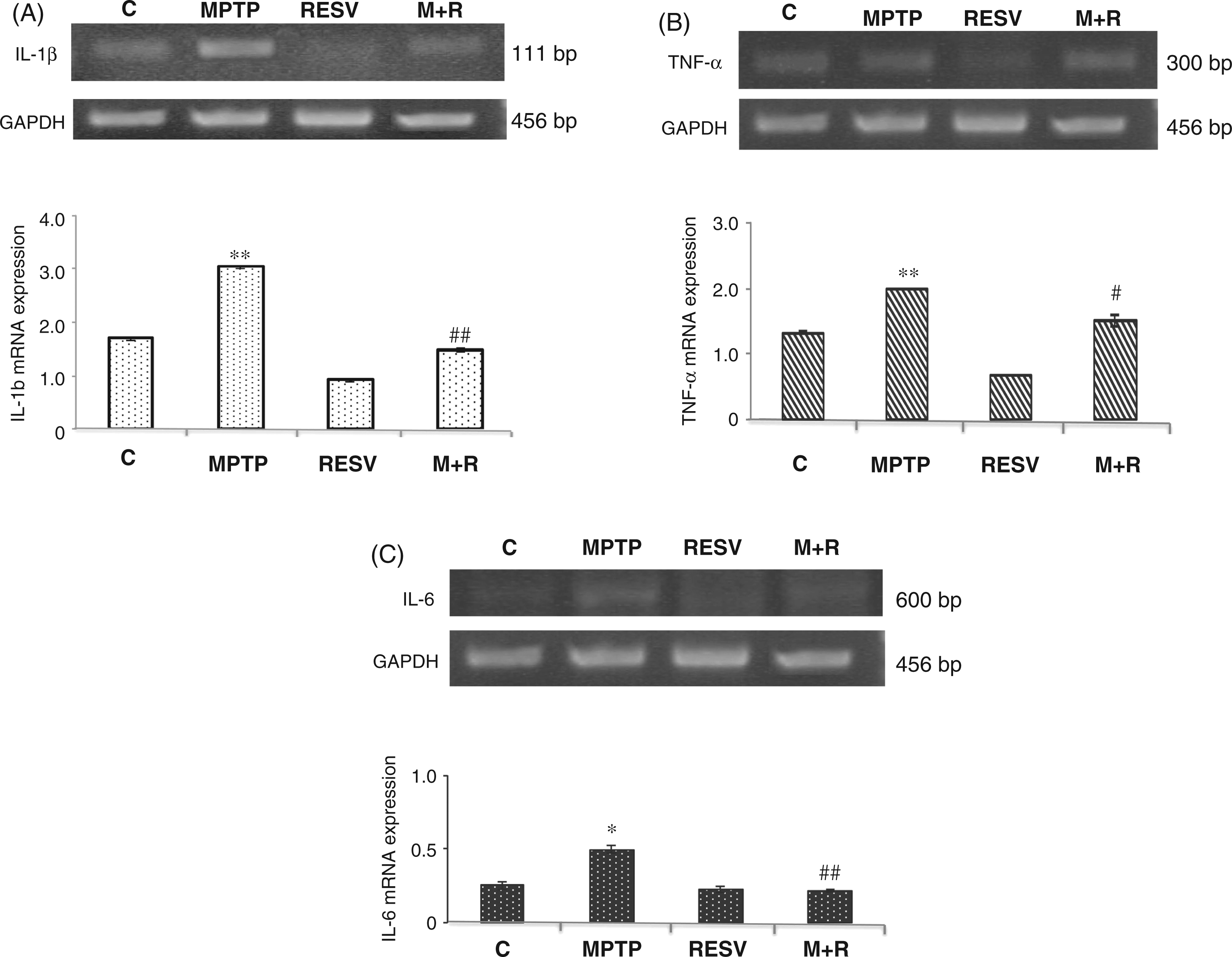
TNF-α analysis
Results of TNF-α analysis are reported in Figure 4B. RT-PCR revealed a reduction of mRNA transcripts in MPTP-treated mice that received resveratrol (Figure 4B, upper). This result was confirmed by quantitative PCR showing that resveratrol was able to reduce the expression of mRNA levels of TNF-α in MPTP + resveratrol-treated mice compared with animals treated only with MPTP (Figure 4B, lower).
IL-6 analysis
Similar results were obtained for IL-6 expression in SNpc. Also in this case, real-time PCR revealed that in MPTP-treated animals resveratrol was able to significantly (
Resveratrol down-regulated the expression of pro-inflammatory cytokine receptors in MPTP-treated mice
IL-1RβI, TNF-αRI and IL-6Rα analysis
IL-1βRI, TNF-αRI and IL-6Rα expression was detected by RT-PCR and immunoblotting analysis at the level of the SNpc in all animals tested. As reported in Figure 5 (A, B, and C, right), densitometric analysis revealed that resveratrol treatment in MPTP-treated animals determined a significant reduction ( Analysis of IL-1βRI, TNF-αRI and IL-6Rα pro-inflammatory cytokine receptor mRNA and protein expression. (A) RT-PCR analysis of IL-1βRI transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene (left, upper). Real-time PCR analysis of IL-1βRI mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (left, lower). Densitometric analysis of IL-1βRI protein expression in the SNpc of control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (right). (B) RT-PCR analysis of TNF-αRI transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene (left, upper). Real-time PCR analysis of TNF-αRI mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (left, lower). Densitometric analysis of TNF-αR protein expression in the SNpc of control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (right). (C) RT-PCR analysis of IL-6Rα transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene (left, upper). Real-time PCR analysis of IL-6Rα mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (left, lower). Densitometric analysis of IL-6Rα protein expression in the SNpc of control (C), MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (right). For IL-1βRI, TNF-αRI and IL-6Rα mRNA real-time PCR analysis values represent the IL-1βRI, TNF-αRI and IL-6Rα mRNA fold changes relative to GAPDH used as resident control. For IL-1βRI, TNF-αRI and IL-6Rα protein expression analysis values represent the relative OD after normalization against β-actin expression. All values are expressed as means ± SD of five independent experiments. *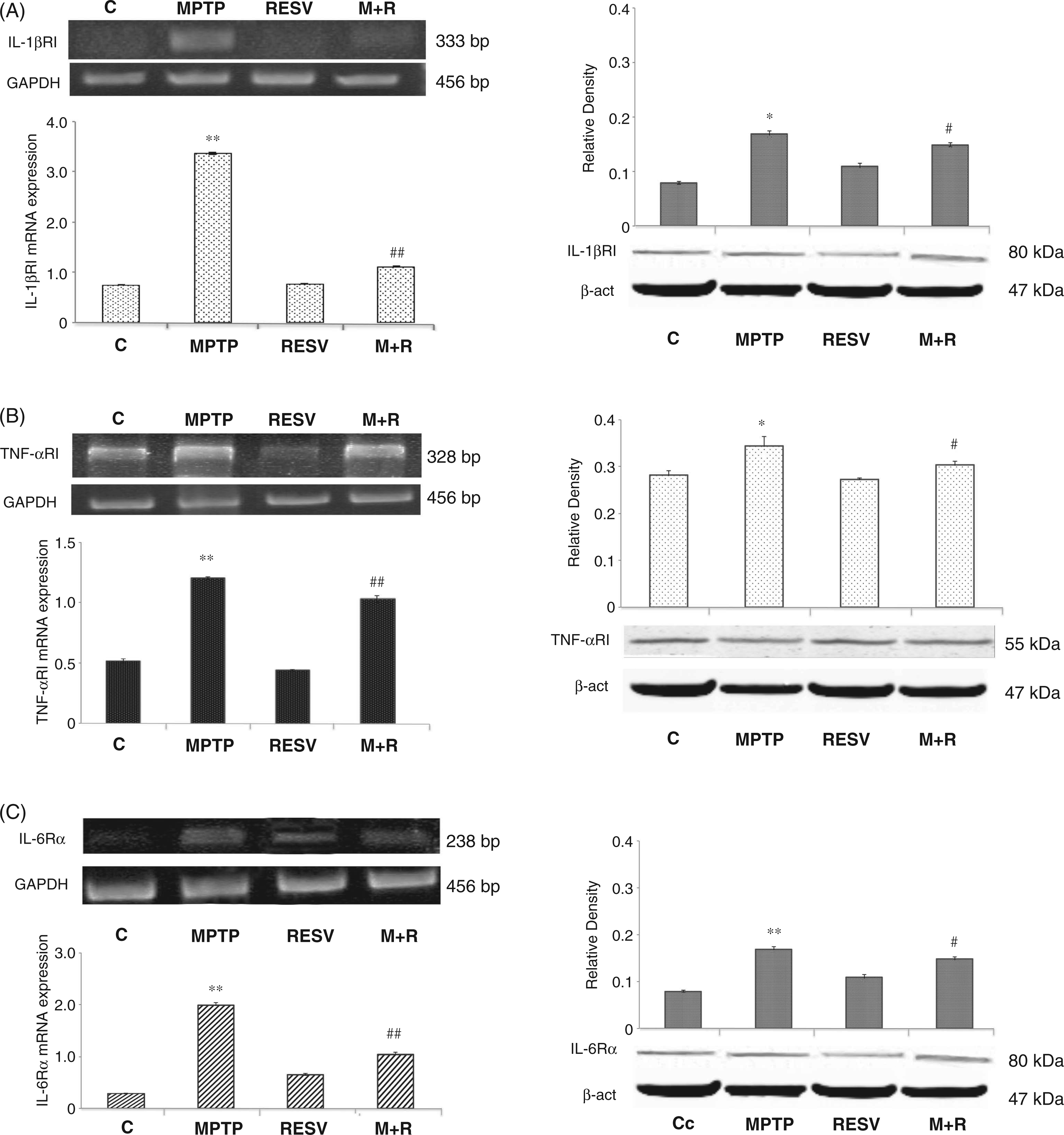
Effect of resveratrol on SOCS-1 expression
We tested the effect of resveratrol on both SOCS-1 protein and gene expression (Figure 6). In this context, resveratrol was able to up-regulate the expression of SOCS-1 transcripts in MPTP-treated animals in comparison with MPTP-treated mice without resveratrol pretreatment (Figure 6A, upper) These results were confirmed by quantitative PCR analysis (Figure 6A, lower). Moreover, SOCS-1 protein levels were significantly ( Analysis of SOCS-1 mRNA and protein expression. (A) RT-PCR analysis of SOCS-1 transcripts extracted from SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice. GAPDH was used as housekeeping gene (upper). Real-time PCR analysis of SOCS-1 mRNA expression levels in SNpc of untreated (C) and MPTP, resveratrol (RESV) and MPTP plus resveratrol (M + R)-treated mice (lower). (B) Densitometric analysis of SOCS-1 protein expression. For SOCS-1 mRNA real-time PCR analysis values represent the SOCS-1 mRNA fold changes relative to GAPDH used as resident control. For SOCS-1 protein expression analysis values represent the relative OD after normalization against β-actin expression. All values are expressed as means ± SD of five independent experiments. *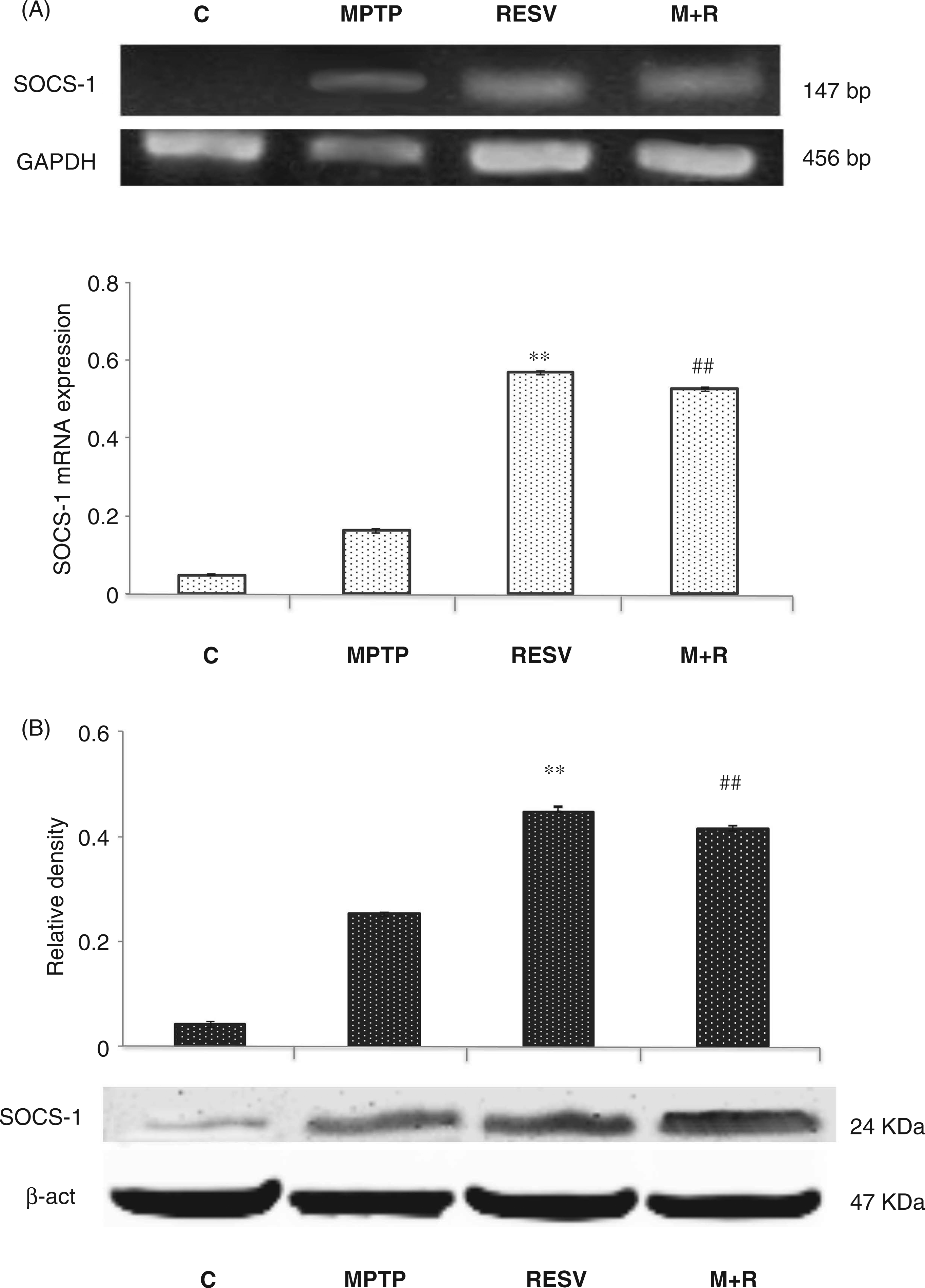
Discussion
Resveratrol is reported to be a neuroprotective agent against neurodegenerative diseases. 28 The neuroprotective potential of resveratrol is dependent on its entry and bioavailability in the brain. In this context, it has been reported that resveratrol is able to cross the blood–brain barrier to enter the brain. 29
In this study, mice were treated with resveratrol daily for 3 weeks in order to maintain the bioavailability of resveratrol, according to a previous report. 29
We observed that
More interestingly, our results demonstrated that in MPTP-treated mice resveratrol was able to down-regulate the pro-inflammatory cytokines IL-1β, TNF-α and IL-6, as well as reduce the expression of the corresponding cytokine receptors, namely IL-1βR, TNF-αRI and IL-6RI, supporting the hypothesis that the protection of DA neurons observed is probably owing to the down-regulation of both cytokines and their relative receptors by this polyphenol.
It is well known that cytokines mediate their functions via specific cell surface receptors that initiate intracellular signaling pathways and gene expression, 31 and that the down-regulation of the pro-inflammatory cytokine and relative receptors by resveratrol, by reducing inflammatory reactions of microglia, could result in neuroprotectivity. These findings are concordant with previous studies in which resveratrol was reported to inhibit the expression of pro-inflammatory cytokines (e.g. TNF-α) and mediators (e.g. COX-2).32–34 Moreover, it has been described in a rat model that resveratrol exerts a neuroprotective effect on 6-hydroxydopamine-induced PD by reducing the levels of COX-2 and TNF-α mRNA in the SNpc. 23
IL-1β is a potent inflammatory cytokine involved in many important cellular functions, such as proliferation, activation and differentiation, and is an important component of the innate immune response. The release of IL-1β is a critical step in inflammation through the induction of other inflammatory cytokines, such as IL-6 and TNF-α.35,36 In our model we detected, apart from an IL-1β increase in MPTP-treated mice, moderate levels of IL-1β in untreated animals. In this regard, it has been reported that in the physiological CNS IL-1β is expressed at low levels, playing a role in some physiological processes, including sleep and synaptic plasticity.37–39 Alternatively, it has been reported that elevated levels of IL-1β are able to induce the production of other cytokines, such as TNF-α and IL-6, by microglia and astrocytes.38,39 Therefore, it can be postulated that in MPTP-treated mice, the elevated levels of IL-1β produced are able, in turn, to up-regulate the production of TNF-α and IL-6, both notoriously implicated in neuroinflammation and in chronic CNS diseases.37–40 Interestingly, we observed that resveratrol was able to significantly reduce not only cytokines and relative receptors expression, but also CD11 levels in the SNpc of MPTP-treated mice, suggesting that the reduced glial activation induced by this polyphenol may be related to the reduced pro-inflammatory reaction observed.
There is growing evidence that an overproduction of pro-inflammatory cytokines by CNS cells contributes to pathophysiological changes seen in various neurologic diseases, including PD, and that the major cellular source for these pro-inflammatory cytokines is the microglia. 41 It is well accepted that in the brain, activated microglia produce chronically elevated pro-inflammatory factors, and induce delayed and progressive loss of DA neurons in the Snpc, thus providing valuable insight into the potential pathogenesis and self-propelling nature of PD. 42 Therefore, elevated inflammatory cytokines in the brain, CSF and plasma of PD patients support the existence of functional interconnections between the immune response and the nervous system.43,44
In this regard, the SOCS proteins are included among the possible effectors engaged in the control of innate immunity.45,46 Emerging evidence supports the activation of negative regulatory mechanisms during neurodegenerative disorders, such as Alzheimer's and Parkinson's diseases, in order to prevent prolonged and extensive inflammation. 47 Therefore, in the present study we tested the possible involvement of SOCS-1 protein in the protective action of resveratrol. Interestingly, we observed significant levels of SOCS-1 in MPTP-treated animals, and that resveratrol is able to up-regulate both the SOCS mRNA transcript and protein levels in MPTP-treated mice. This result is also correlated with the reduced expression of cytokines and their receptors, as well as with the reduction of neuronal damage, in MPTP + resveratrol treated mice, not detectable in mice subjected to treatment with MPTP in the absence of resveratrol. Evidently, although a significant increase of SOCS-1 in mice treated with MPTP alone is observed, it is not sufficient to reduce the inflammatory reaction and the resulting neuronal damage as in MPTP + resveratrol-treated mice.
Expression of many of the
Finally, it has been reported that administration of the SOCS-1 mimetic form, tyrosine kinase-inhibitor peptide (Tkip), prevents EAE and that this protection is correlated with a decreased expression of IL-2, IL-5, TNF-α and IFN-γ in the CNS of Tkip-treated mice, 54 indicating that SOCS-1 can attenuate neuroinflammatory responses and might, therefore, have therapeutic value in neurodegenerative diseases. Because of their biological importance, the SOCS proteins have been the subject of intense investigation, including the development of strategies to utilize these proteins to control cytokine-induced signal transduction for therapeutic purposes.
Overall, based on our results we hypothesize that resveratrol may act in a protective manner against MPTP-induced neurotoxicity through up-regulation of SOCS-1 which, in turn, may be responsible for the pro-inflammatory responses attenuation, thus suggesting a new potential mechanism of neuroprotection for this polyphenol.
Further investigations, for example in knockout animals, are needed to elucidate the beneficial effect of resveratrol in experimental PD, and to specifically define the protective role of SOCS regulation by polyphenols during inflammatory responses in the brain.
Footnotes
Funding
This work was supported by the project PRIN2009YRH27R_003 and a grant of the
Acknowledgements
We are grateful to Mary Victoria Candace Pragnell, B.A., for reviewing the language of the manuscript.
