Abstract
This study was designed to characterize potential sexually dimorphic stress and immunological responses following a corticotropin-releasing hormone (CRH) challenge in beef cattle. Six female (heifers) and six male (bulls) Brahman calves (264 ± 12 d of age) were administered CRH intravenously (0.5 µg of CRH/kg body mass) after which serum concentrations of cortisol increased from 0.5 h to 4 h. From 1 h to 4 h after CRH administration, serum cortisol concentrations were greater in heifers than in bulls. In all cattle, increased serum concentrations of TNF-α, IL-6 and IFN-γ were observed from 2.5 h to 3 h after CRH, with greater concentrations of IFN-γ and IL-6 in heifers than bulls. Heifer total leukocyte counts decreased 1 h after CRH administration, while bull leukocyte counts and percent neutrophils decreased 2 h after CRH administration. Heifers had greater rectal temperatures than bulls, yet rectal temperatures did not change following administration of CRH. There was no effect of CRH administration on heart rate. However, bulls tended to have increased heart rate 2 h after CRH administration than before CRH. Heifer heart rate was greater than bulls throughout the study. These data demonstrate that acute CRH administration can elicit a pro-inflammatory response, and cattle exhibit a sexually dimorphic pro-inflammatory cytokine and cortisol response to acute CRH administration.
Introduction
Cattle morbidity and decreased performance are often associated with stressful events, such as weaning, transportation, handling and commingling.1–4 Not all stressors cattle encounter, however, may negatively affect innate immune function. The interaction between the hypothalamic–pituitary–adrenal (HPA) axis and the innate immune system is complex; the type of stressor and how an animal perceives and responds to the stressor determines the subsequent influence on immune competence.5,6
The neuroendocrine system plays an important role in responding to stressors and maintaining homeostasis by mediating the response of the immune system. 5 When stress is perceived by an animal, corticotropin-releasing hormone (CRH) is released from the hypothalamus, causing the release of adrenocorticotropic hormone (ACTH), which signals the release of glucocorticoids from the adrenals. In addition, after stress is perceived, the sympathetic response increases heart rate and body temperature. Glucocorticoids contribute to the homeostasis of the HPA axis and stress response by providing a negative feedback to the hypothalamus and pituitary.7,8 Therefore, glucocorticoid sensitivity is very important in maintaining homeostasis. 5 Glucocorticoids can greatly influence and modulate many aspects of the immune system; thus, an animal's sensitivity to glucocorticoids is also very important in the regulation of the immune response. 9 Although researchers have traditionally associated glucocorticoids with immune suppression, glucocorticoids can act in a permissive manner and cause pro-inflammatory cytokine release.6,9 In fact, circulating glucocorticoids before a stressor (basal concentrations) can cause pro-inflammatory responses to stressors, which may help prime the animal’s immune system for potential insults or injury from the stress response. 6
There is evidence that the HPA response differs between female and male cattle.10,11 Angus bulls (7–12 months of age) had lower cortisol concentrations than heifers of the same age, breed and management. 12 In addition, Arthington et al. 13 reported that Angus heifers consistently had greater cortisol concentrations than bulls before, during and after periods of transportation and commingling. In a companion study, Brahman heifers had a greater cytokine response to an endotoxin challenge than bulls; 14 therefore, one could speculate that there may also be a sexually dimorphic role in the HPA response. In rodents, female rats have greater basal glucocorticoid concentrations, lower pituitary glucocorticoid receptors, 15 and prolonged elevated peripheral concentrations of glucocorticoids following adrenocorticotropic hormone (ACTH) challenge compared with male rats.15–17 In humans, males are more sensitive to glucocorticoids and are less susceptible to autoimmune disease than women.18,19 Some researchers speculate that female stress responses may be hard-wired long before puberty, and sex differences in immunity might have evolved because of physiological and behavioral differences among males and females. 20 These factors may influence how the HPA axis reacts to stressors, causing a sexual dimorphism of the HPA axis and, consequently, immune regulation. The sexually dimorphic cortisol response in cattle could influence pro-inflammatory cytokine secretion; therefore, the objective of this study was to determine the effect of a CRH challenge on purebred Brahman heifer and bull physiological parameters, cortisol responses, pro-inflammatory cytokine responses and changes in peripheral leukocytes.
Materials and methods
Animals and housing
Animal procedures were reviewed and approved by the Texas Tech University Animal Care and Use Committee. Six pure-bred Brahman (Bos indicus) heifers and six bulls (average age = 245 ± 8.6 and 280 ± 50.3 d; average body mass = 202 ± 9 and 246 ± 23 kg for heifers and bulls respectively) from the Texas AgriLife Research Center’s herd in Overton, TX, were transported approximately 770 km (∼9 h) to the cattle facility at the Texas Tech University’s Beef Center in New Deal, TX. Previous research has shown that temperament may be a source of variation to consider when designing experiments or assigning animals to treatment groups;21,22 therefore, heifers and bulls of intermediate (moderate) temperament classification (compared with their herd mates; n = 110) were selected for this study using methods described previously.21,22 Typically, literature values for age at puberty in Brahman cattle exceed 365 d of age for both heifers and bulls.23,24 Therefore, the cattle in this study were considered pre-pubertal. Cattle were housed in individual stanchions (2.13 m long × 0.76 m wide) following transportation for the duration of the CRH challenge. During the experiment, the ambient temperature in the morning ranged between 2.0℃ and 11.4℃, afternoon temperatures ranged from 11.4℃ to 18.3℃ and evening temperatures ranged from 7.8℃ to 11.8℃. Cattle were provided unlimited access to water and were fed a diet ad libitum that consisted of 30% cotton seed hull pellets, 25% cotton seed hulls, 12.5% dehydrated alfalfa, 10% rice bran, 10.5% soybean meal, 7.5% soybean hulls, 2% crimped-corn and 2.4% mineral supplement (Cargill Animal Nutrition, Minneapolis, MN, USA). Two d after transportation all cattle were fitted with rectal temperature (RT) monitoring devices and indwelling jugular vein catheters, as described previously.25,26 The following day, all cattle were challenged intravenously with bovine CRH at a dose of 0.5 µg/kg body mass (Bachem Chemicals, Torrance, CA, USA). Blood samples (6 ml) for serum were collected at 30-min intervals, and whole blood samples (3 ml) for blood leukocyte counts and differentials were collected at 1-h intervals from -2 h to 8 h, and at 12 h and 24 h relative to the CRH challenge (0 h). Catheter patency and cattle fluid volume were maintained by flushing 10 ml of saline, followed by 5 ml of heparinized saline (10 IU/ml) into the catheters following each sample collection.
RT and heart rate
RT monitoring devices consisted of automated indwelling temperature loggers (Tidbit®v2 Temp Logger UTBI-001; Onset Corp., Pocasset, MA, USA) that collected and stored RT at 1-min intervals. 26 The Polar RS400 monitor (Polar Electro Öy, Kempele, Finland) was used to collect heart rate (1000 Hz, 5-s intervals) and store data from a transmitter with two probes that were placed close to the sternum and the right scapula. Some heart rate monitor probes malfunctioned during the experiment; therefore, the heart rate data reported was derived from four heifers and five bulls.
Processing and analysis of blood samples
Whole blood was allowed to clot for 30 min at room temperature (20–22℃) and serum was collected after centrifugation at 1250 g for 20 min at 4℃. Serum was collected and stored at −80℃ until analyzed for cortisol and cytokine concentrations. All serum samples were analyzed in duplicate. Serum concentrations of cortisol were determined using a single Ab radioimmunoassay (MP Biomedicals, LLC Diagnostic Division, Orangeburg, NY, USA) utilizing rabbit anti-cortisol antiserum coated tubes according to the manufacturer’s instructions. 1 The minimum detectable cortisol concentration was 1.2 ng/ml and the intra- and inter-assay coefficients of variation were 9.2% and 8.0% respectively. Serum cortisol concentrations were determined by comparison of unknown samples to a standard curve generated with known concentrations of cortisol and are presented as the concentration in ng/ml.
Cytokines for bovine IL-6, TNF-α and IFN-γ were measured using the multiplex sandwich-based Chemiluminescence-ELISA kit (Searchlight-Aushon BioSystems, Inc. Billerica, MA, USA) as described previously.25,27 The intra-assay coefficient of variation was less than 10% and inter-assay coefficient of variation was less than 15% for all cytokine assays.
Within 30 min of collection, whole blood was analyzed for total leukocyte counts and differentiation analyses into neutrophils, lymphocytes and others (monocytes, eosinophils, basophils) using a Cell Dyn 3600 with automated 50-sample loader and vet-package software (Abbot Laboratories, Abbot, IL, USA). Additionally, the neutrophil : lymphocyte ratio was calculated.
Statistical analysis
All repeated continuous data were analyzed by restricted maximum likelihood ANOVA using the MIXED procedure of SAS (v.9.1.3; SAS Inst. Inc., Cary, NC, USA). A linear, mixed model with the fixed effects of sex (heifer or bull), sampling time and the interaction of sex × time was fitted. The anti-regression covariance structure for the within-subject measurement was used. Repeated data were tested for normality of the residuals by evaluating the Shapiro-Wilk statistic using the UNIVARIATE procedure of SAS. Data that were not normally distributed were either log- or root-transformed prior to mixed model analysis. Pair-wise differences were performed at each time point using a sliced-effect multiple comparison approach with a Tukey–Kramer adjustment. Rectal temperature was measured in 1-min intervals and heart rate in 5-s intervals, but data were averaged over 10-min intervals prior to analyses to define the febrile and physiological responses. The response interval from −2 h to 10 h was analyzed for RT and heart rate. Least squares means (±SEM) are reported throughout. A treatment difference of P ≤ 0.05 was considered significant.
Results and discussion
Mean (±SEM) measurements of the overall (all time periods) sex interaction for heifers and bulls.
RT was collected continuously by indwelling rectal temperature monitoring devices.
Heart rate, beats per min (bpm) collected continuously.
cTotal circulating leukocyte counts.
dNeutrophil to lymphocyte (N:L) ratio.
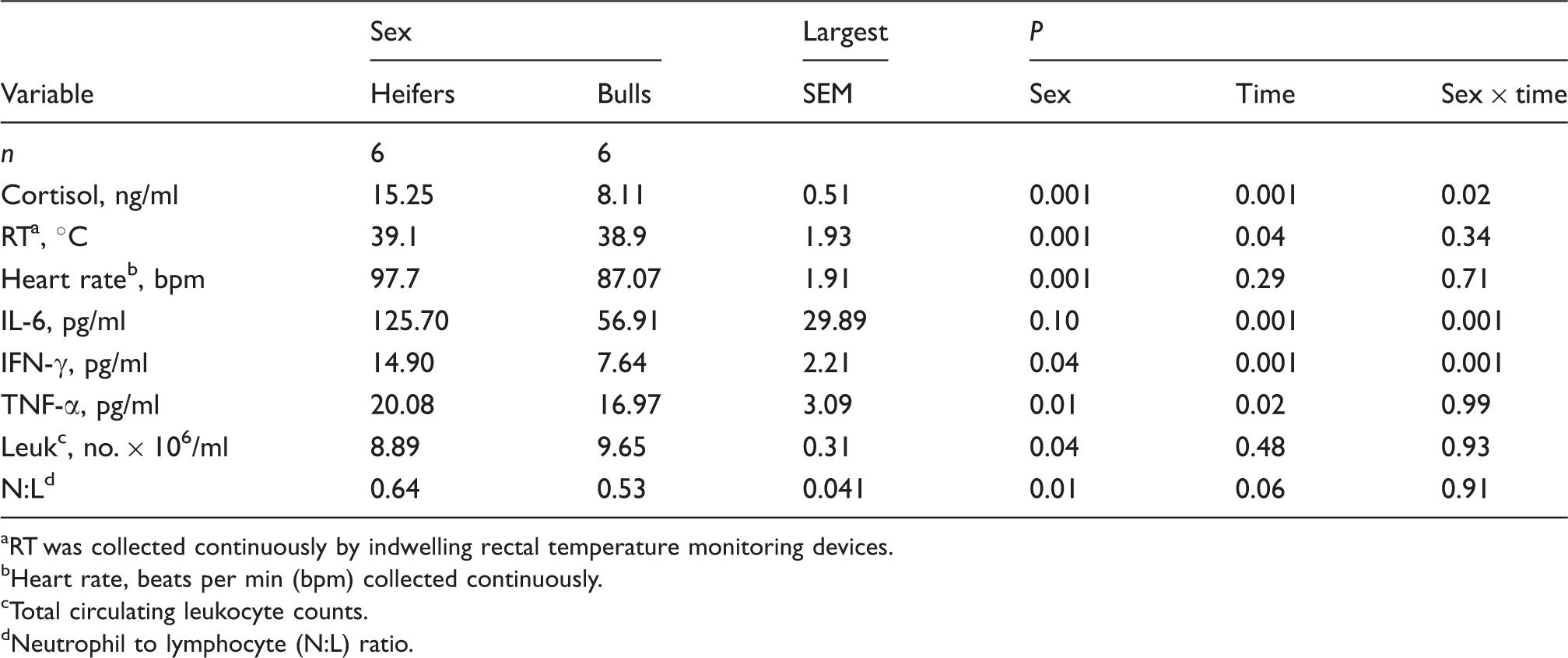
Mean (± SEM) cortisol concentrations from Brahman heifers (n = 6; solid-circle) and bulls (n = 6; open-circle) following an i.v. bolus-dose challenge with bovine corticotropin-releasing hormone (CRH; 0.5 µg/kg body mass) at 0 h. *P < 0.05, **P < 0.01.
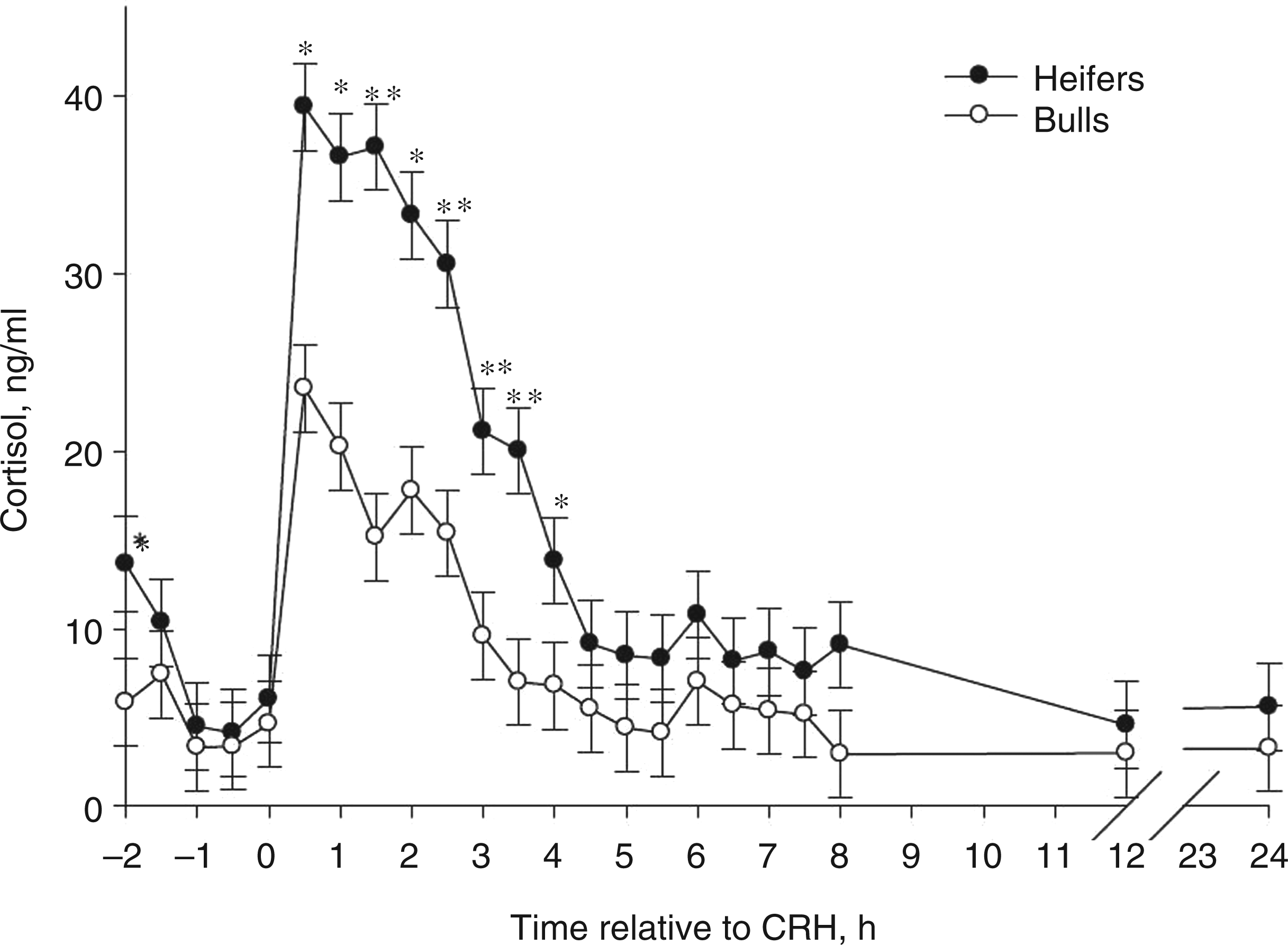
Increases in pro-inflammatory cytokine concentrations were observed from 3 h to 3.5 h following CRH challenge (Figure 2). Heifers had greater (P < 0.001) IL-6 and IFN-γ concentrations from 3 h to 3.5 h, and greater TNF-α at 3.5 h than bulls. Twenty-four h after the CRH challenge, bulls had elevated IL-6 concentrations when compared with heifers (Figure 2A; P < 0.01). This increase in pro-inflammatory cytokines may be owing to direct effects of CRH on circulating leukocytes, as well as indirect effects due to the actions of glucocorticoids on circulating leukocytes. Heifers had more cortisol and cytokine secretion than bulls, suggesting that the bull cytokine response after CRH challenge may have been suppressed by the direct effects of CRH or indirectly by cortisol. Researchers have reported that Holstein calf mononuclear leukocyte proliferation and IL-2 cytokine receptor expression was reduced by an in vivo glucocorticoid challenge.
36
It is well documented that glucocorticoids suppress many other cytokine secretions, including IL-6, TNF-α and IFN-γ.36–39 Nonetheless, there is evidence that CRH can directly influence cytokine secretion from murine macrophages, where CRH augments endotoxin-induced cytokine secretion.
39
Mean (± SEM) circulating concentrations of IL-6 (A), IFN-γ (B) and TNF-α (C) in Brahman heifers (n = 6; solid-circle) and bulls (n = 6; open-circle) following an i.v. bolus-dose challenge with bovine corticotropin-releasing hormone (CRH; 0.5 µg/kg body mass) at 0 h. *P < 0.05, #P < 0.10.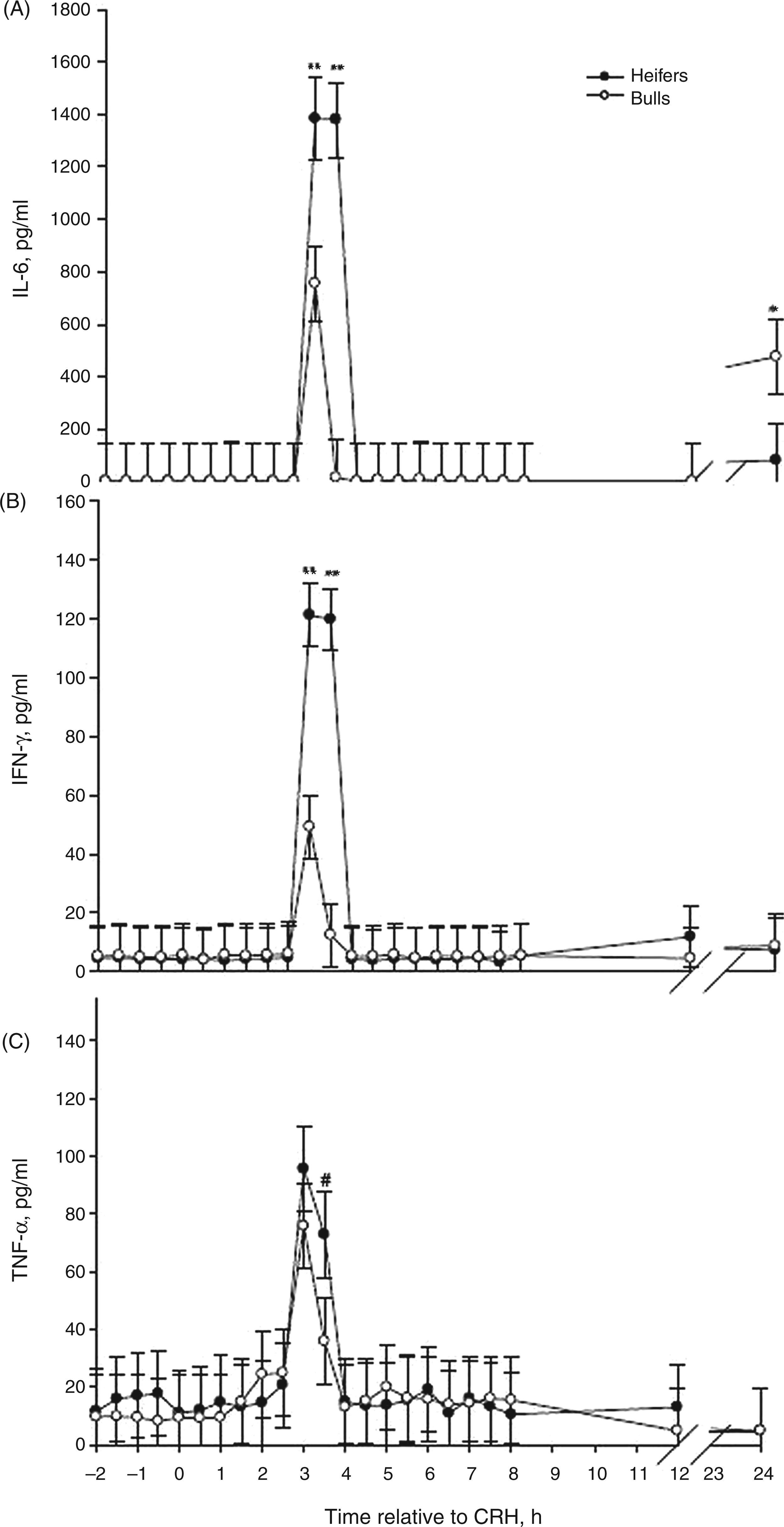
Similar to current findings, mouse models and ex vivo research have also shown that glucocorticoids can enhance the immune system by causing up-regulation of cytokine receptors and also by acting synergistically with cytokines, such as IL-6, to induce acute phase protein production by hepatic cells.5,40 Therefore, sexually differential secretion of cortisol may have influenced the differential release of cytokines and we speculate that the pro-inflammatory cytokine regulation by cortisol was more apparent in bulls. In contrast to the bovine response in the current study, in humans exposed to a psychosocial stressor, women had a lesser cortisol response than men. 19 Moreover, in humans, dexamethasone inhibited endotoxin-induced cytokine secretion to a lesser degree in women than men, suggesting that glucocorticoid sensitivity is greater among men. 19 However, similar to our findings, IL-6 and TNF-α plasma concentration in women before and after a psychosocial stressor were greater than cytokine concentrations in men. 18
Heifers might not only be less sensitive to glucocorticoids, but also more responsive to the peripheral injection of CRH. Peripheral CRH has been shown to increase the pro-inflammatory cytokines in rodent models of inflammation.41,42
Lymphocytes and monocytes have CRH receptors, 33 and CRH can act as an autocrine or paracrine pro-inflammatory cytokine. 42 Likewise, human mast cells have CRH receptors and can secrete CRH, IL-6, IL-8 and TNF-α.42–44 The degree of the cytokine response observed in the present study after the peripheral CRH challenge suggests that CRH may have stimulated leukocytes directly and to a greater degree in heifers than bulls. This increase in pro-inflammatory cytokine secretion in heifers after CRH suggests that heifers may have more priming of the immune system than bulls following acute stress.6,9,36
CRH, glucocorticoids, cytokines and chemokines can influence the migration of leukocytes. A decrease in peripheral leukocytes by 1 h following CRH administration was observed among heifers and by 2 h among bulls (P < 0.05; Figure 3A). The percentage of neutrophils decreased in bulls, but not heifers, 2 h after CRH administration (P < 0.05; Figure 3B). Heifers also had less total leukocytes (P < 0.05, Table 1), but a greater neutrophil : lymphocyte ratio (P < 0.05) than bulls. This is similar to our findings in heifers and bulls from the same herd and age group challenged with endotoxin.
14
Neutrophils do not express CRH receptors; therefore, CRH had indirect effects on bull neutrophil : lymphocyte in the periphery.
45
Stress responses in animals can result in gradual and chronic down-regulation of L-selectin (CD62L), an important adhesion molecule expressed on neutrophil surfaces. This down-regulation is associated with neutrophilia and increased susceptibility to infection.2,3,45,46 Glucocorticoids also stimulate the release of new neutrophils from bone marrow stores and these neutrophils can be immature.45–48 In this study, heifers had greater percentages of neutrophils prior to CRH administration than bulls (P < 0.01; Table 1) and all cattle displayed a decrease in neutrophil : lymphocyte ratio 1–5 h after CRH, but neutrophil : lymphocyte ratios were above baseline at 6–8 h, as well as 24 h after CRH (Figure 3B). This suggests that neutrophils from bulls were more sensitive to changes in glucocorticoids than were neutrophils from heifers.
Mean (±SEM) total leukocytes (A) and neutrophil:lymphocyte ratio (B) in Brahman heifers (n = 6; solid-circle) and bulls (n = 6; open-circle) following an i.v. bolus-dose challenge with bovine corticotropin-releasing hormone (CRH; 0.5 µg/kg body mass) at 0 h. *P < 0.05.
There was not an obvious change in RT or heart rate following CRH (time × sex P > 0.11; Figure 4). However, overall, heifers had greater heart rates and RTs compared with bulls (P < 0.01; Table 1). This is similar to what others have reported.
48
Specifically, heart rate in heifers was greater than bulls immediately after 14 h of transportation.
48
In addition, after an endotoxin challenge, heifers had a greater febrile response and increased heart rate, but heifers had fewer time periods where they appeared sick.
14
Intracerebral ventricular (i.c.v.) administration of CRH increased rat heart rates and cardiac output, whereas i.v. delivery of CRH decreased blood pressure followed by reflex tachycardia because of vasodilation of certain vascular beds.38,49 Centrally-administered CRH may elicit a stronger sympathetic nervous system response than peripheral CRH, which was administered in the current study. Likewise, core body temperature can also be influenced by centrally-administered CRH,
50
but RTs were not influenced by the peripheral CRH challenge in the present study. In rats, CRH may mediate increased core body temperature among exercised, but not sedentary, rats.
50
The cattle in the present study were placed in stanchions allowing for limited movement; therefore, dramatic changes in heart rate and RT from the CRH administration may have been overshadowed by the animal’s inability to respond to the stressor by a full ‘flight’ reaction.
Mean (±SEM) heart rate [beats per minute (bpm); (A) of 9 Brahman cattle (4 heifers = solid-circle, 5 bulls = open-circle) and rectal temperature (B) of 12 Brahman cattle (6 heifers = solid-circle, 6 bulls = open-circle) after an i.v. bolus-dose challenge with bovine corticotropin-releasing hormone at time 0 (CRH; 0.5 µg/kg body mass) at 0 h. *P < 0.05, **P < 0.01, #P < 0.10.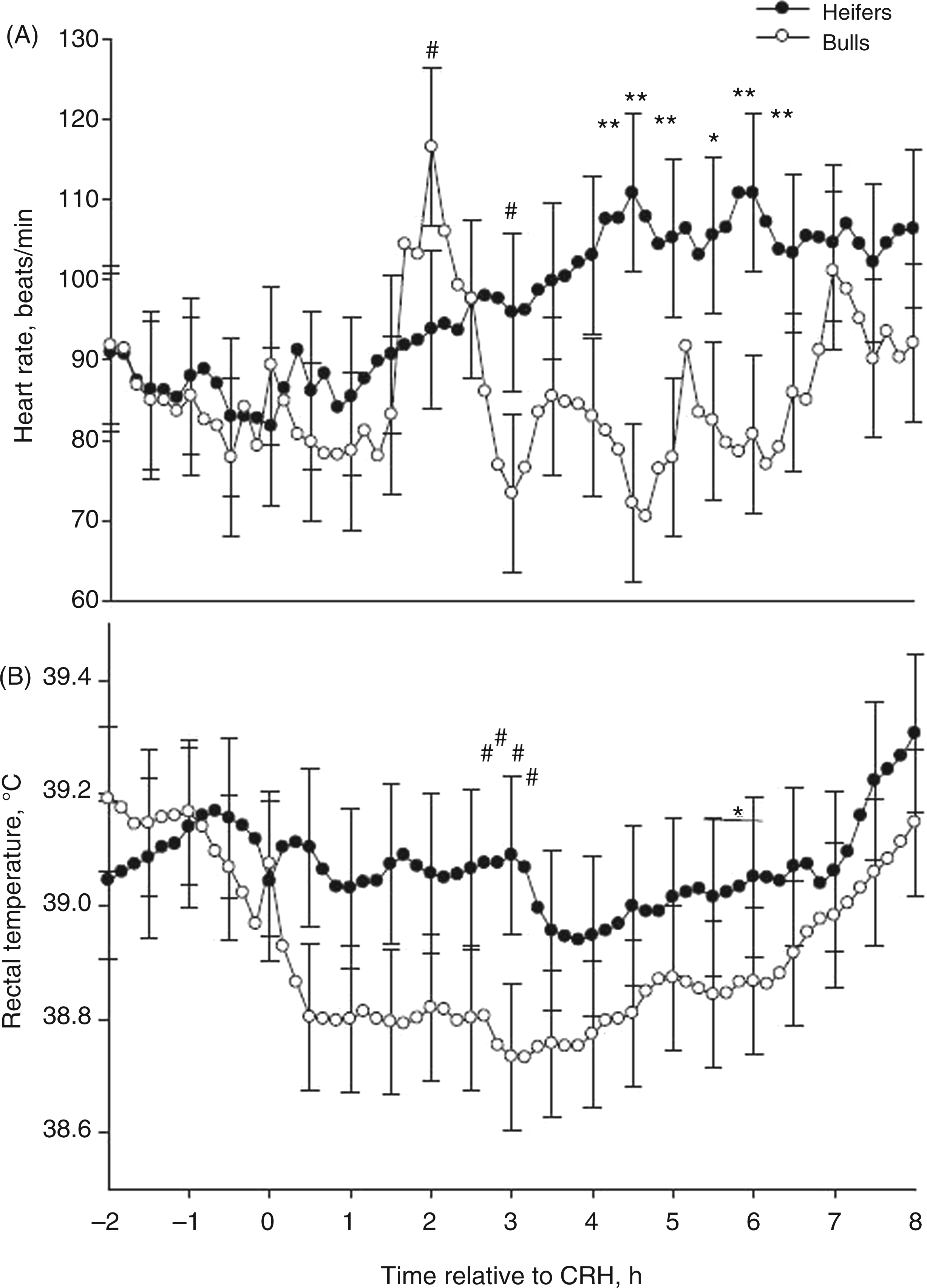
Conclusions
Results from this study demonstrate that sexually dimorphic cortisol, cytokine and leukocyte responses to CRH exist among Brahman cattle. Heifers had greater circulating cortisol and cytokine concentrations, and greater neutrophil : lymphocyte, which suggests that heifers are less sensitive to glucocorticoids than bulls. Likewise, CRH may have acted both directly and indirectly on certain leukocytes and mast cells to cause the greater secretion of pro-inflammatory cytokines that was observed. The sexually dimorphic features of Brahman cattle responses to CRH suggest inherent sexual differences in HPA function and regulation of the immune system. Therefore, during stressful periods (e.g. weaning, handling, feedlot induction) different managerial practices for heifer and bull calves may need to be considered. For example, because bulls may be more sensitive to stress they might be more susceptible to immunosuppression from stress, thus requiring more prophylactic approaches or intervention methods than heifers during stressful periods.
Footnotes
Acknowledgements
Mention of trade names or commercial products in this article is solely for the purpose of providing information and does not imply recommendation or endorsement by the U.S. Department of Agriculture (USDA). The USDA prohibits discrimination in all its programs and activities on the basis of race, color, national origin, age, disability and, where applicable, sex, marital status, familial status, parental status, religion, sexual orientation, genetic information, political beliefs, reprisal, or because all or part of an individual's income is derived from any public assistance program. (Not all prohibited bases apply to all programs.) Persons with disabilities who require alternative means for communication of program information (Braille, large print, audiotape, etc.) should contact USDA's TARGET Center at (202) 720-2600 (voice and TDD). To file a complaint of discrimination, write to USDA, Director, Office of Civil Rights, 1400 Independence Avenue, S.W., Washington, D.C. 20250-9410, or call (800) 795-3272 (voice) or (202) 720-6382 (TDD). USDA is an equal opportunity provider and employer. The authors acknowledge the technical support of Don Neuendorff, Adam Lewis, Luke Schwertner, Jessica Carroll, ChunFa Wu and Samantha Werth.
Funding
This study was supported, in part, by Texas A&M AgriLife Research, USDA-NRI-CSREES Grant 2005-01671 and USDA SG 2006-34564-170.
