Abstract
Endotoxin contaminations of engineered nanomaterials can be responsible for observed biological responses, especially for misleading results in
Introduction
The development and production of nanomaterials and their applications are steadily increasing. There is a large diversity of nanomaterials available because they can be produced from virtually every material,
1
made in different shapes and sizes, and can be functionalized by additional surface coatings. Especially for medical applications, engineered nanomaterials have to be tested for toxicologic and immunologic effects on living cells and organisms.2–4 In addition to thorough physicochemical characterization of the nanomaterials, the detection of contaminations with pyrogenic substances, especially bacterial endotoxins, is of major importance. Since at least the 1990s it has been known that endotoxin contamination of commercial particles can be responsible for observed biological responses.5,6 Just recently, it was demonstrated7–9 again that undetected endotoxin contaminations in particle suspensions can cause misleading results in
Here, we tested the traditional LAL gel clot assay for the detection and semi-quantification of endotoxin in dispersions of silica nanoparticles synthesized for different biological and biomedical applications. In contrast to other studies that compared the effects of different kind of nanomaterials on the assay systems,18,19 we systematically investigated the effects of nanomaterials made of the same outer material. Six different dispersions of bare or PEGylated silica nanoparticles, as well as of four dispersions of iron oxide–silica core shell nanoparticles, were tested. The gel clot assay—the only LAL-based assay which is not based on an automatic optical read out—was selected owing to the strong absorbance of the tested iron oxide–silica core shell particles at the typical read-out wavelength (405 nm) of the chromogenic LAL assays. In addition, the gel clot assay is considered to be less prone to interfering substances.
21
According to the
Materials and methods
Particle synthesis
Silica particles were prepared by a modified Stöber process
23
or according to a modified method described by Hartlen et al.
24
through
Particle characterization
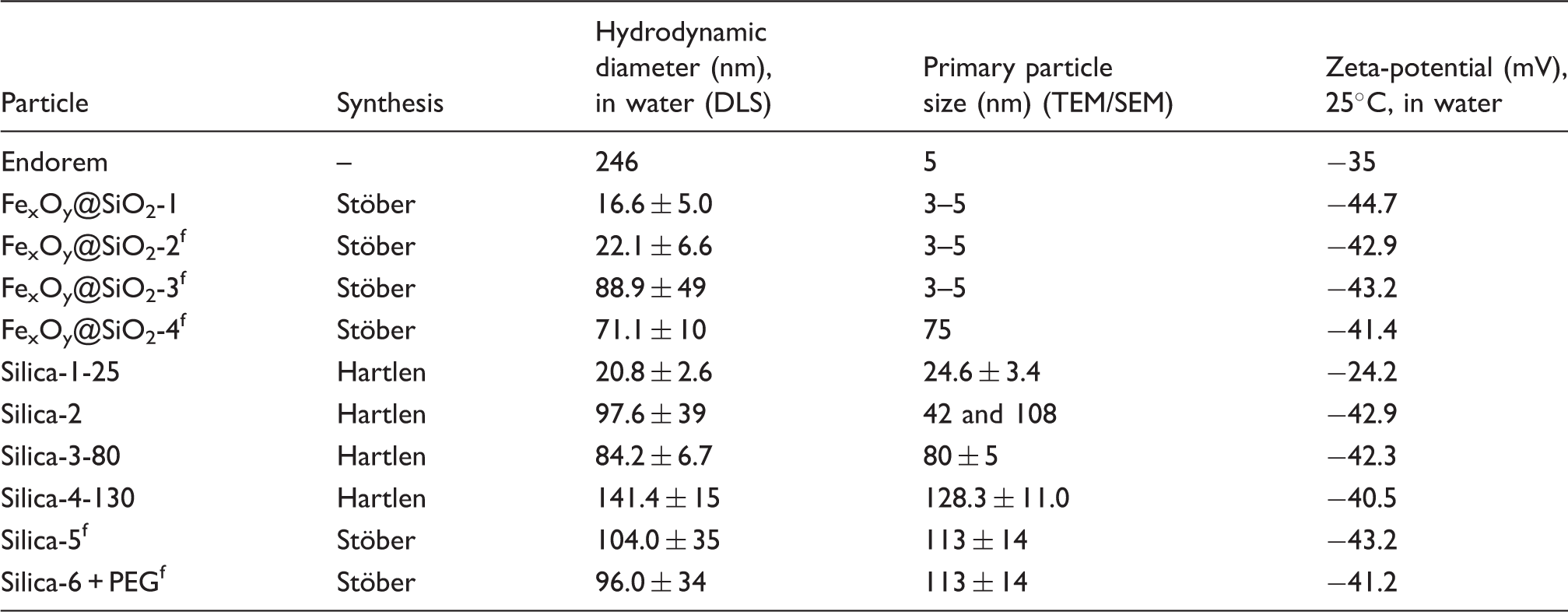
Aliquots of the sterile filtered nanoparticle suspensions were applied for particle characterization. Particle size and morphology were determined by scanning or transmission electron microscopy (SEM/TEM). SEM samples of undiluted nanoparticle suspensions were prepared by vacuum drying onto silicon wafers. The hydrodynamic particle diameter was recorded by dynamic light scattering (DLS; Dyna Pro Titan, Wyatt Technology Europe, Dernbach, Germany). Zeta-(ζ)-potential measurements of 1-ml samples were carried out in disposable cuvettes at 25℃ with a Zetasizer Nano (Malvern, Germany). UV and visible light spectroscopy (UV-vis) measurements in the wavelength range of 350–800 nm were performed with a Cary 5000 spectrophotometer (Varian, Darmstadt, Germany). The pH values of all tested samples were controlled and in the required pH range of the assay (pH 6–8). No adjustment of the pH was necessary.
Endotoxin tests
Endotoxin tests were performed with the LAL Gel Clot Assay Lonza (Pyrogent™ Plus N294-03; Lonza Walkersville, Walkersville, MD, USA). All materials used were sterile and labeled to be pyrogen-free by the manufacturers. Endotoxin detection was performed according to the protocol ‘Nanokon SOP 2.2.2: Detection and semi-Quantification of Endotoxin Contaminations in Nanoparticle Suspensions –
Results
Physicochemical properties of the tested particle suspensions.
IEC results for all tested particles.
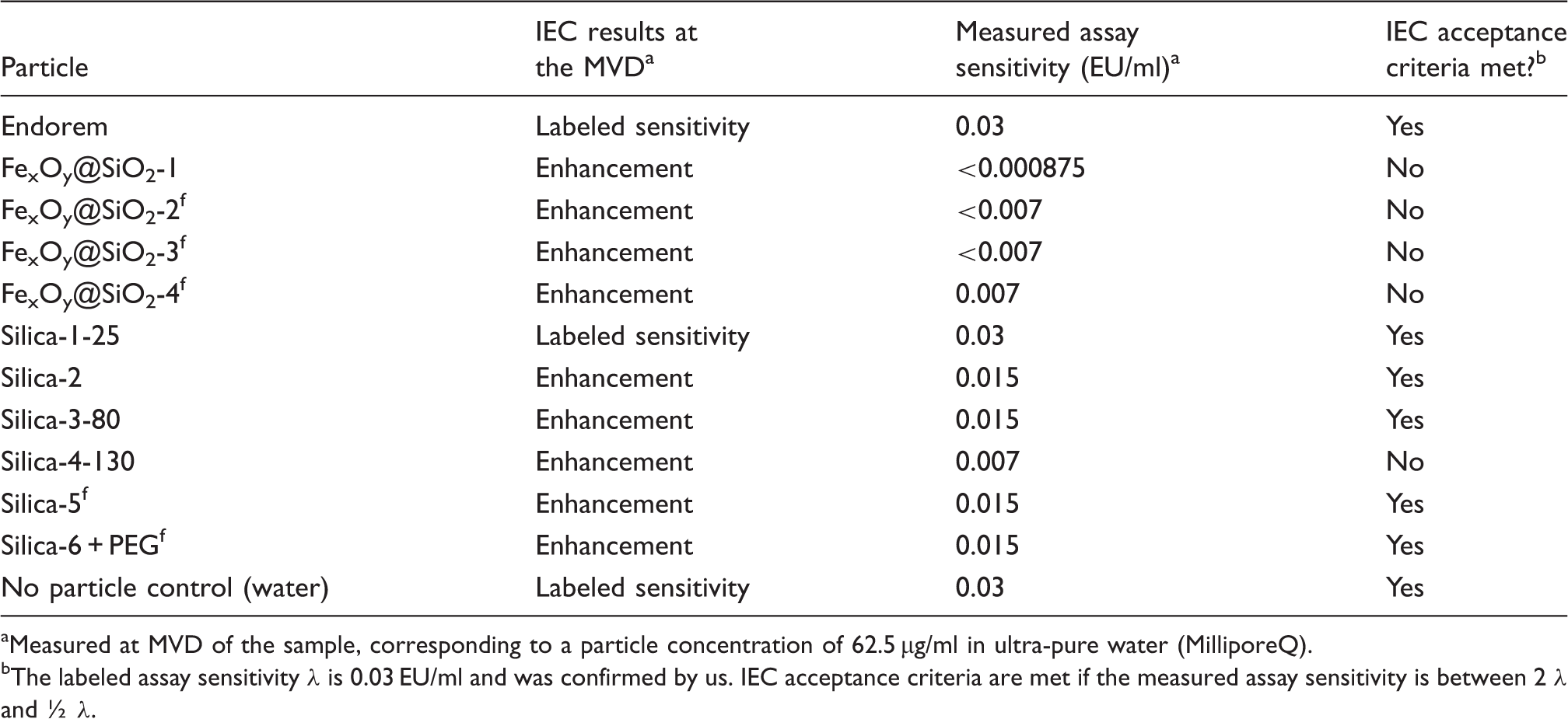
Measured at MVD of the sample, corresponding to a particle concentration of 62.5 µg/ml in ultra-pure water (MilliporeQ).
The labeled assay sensitivity λ is 0.03 EU/ml and was confirmed by us. IEC acceptance criteria are met if the measured assay sensitivity is between 2 λ and ½ λ.
Silica particles: bare and PEGylated
For the majority of silica particles no endotoxin contamination (>0.03 EU/ml) was detected for all particle concentrations tested (1 mg/ml to 62.5 µg/ml particles). Only one of six silica nanoparticle samples contained low amounts of endotoxin. For all particles, IEC were performed at the MVD, as well as at least one other particle concentration. Generally, all silica particles showed coagulation enhancement activity. If tested at the MVD, which corresponds to a concentration of 62.5 µg/ml particles, only Silica-1-25 did not change the sensitivity of the assay. In contrast to the other silica nanoparticle suspensions, Silica-1-25 showed a zeta-potential of −24.2 mV instead of −40 to −45 mV. Four silica particle suspensions (Silica-2, Silica–3–80, Silica-5f and Silica-6-PEGf) showed slight enhancement activity at a concentration of 62.5 µg/ml particles, which was within the limits of acceptance (0.5–2λ) given by the SEM image of monodisperse silica particles silica-4-130 with a primary particle size of 128.3 ± 11 nm. The spherical particles were obtained by a modified method described by Hartlen et al.
24
through Concentration-dependent IEC for Silica-2. +: positive reaction, clot formation; −: negative reaction, no clot formation. The labeled assay sensitivity (λ) is 0.03 EU/ml. The limit of acceptance for assay enhancement is 0.015 EU/ml (enhancement factor 2).
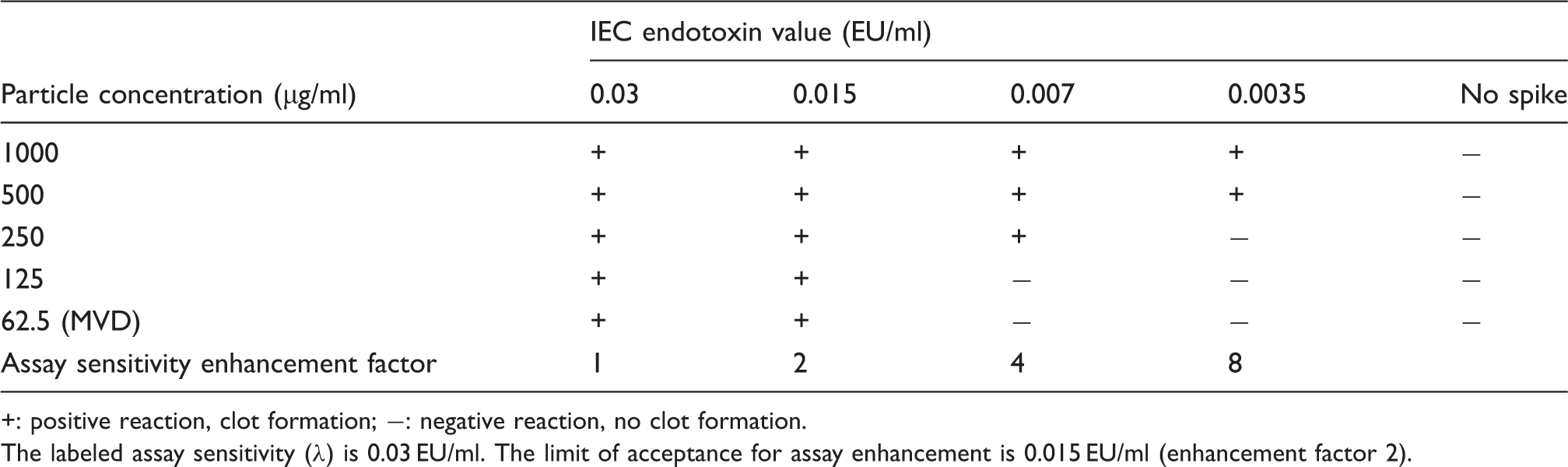
To investigate the influence of surface modification on the assay results and enhancement, silica particles modified with PEG were included and compared with particles without PEGylation. At a high particle concentration (500 µg/ml SiO2), the results obtained with Silica-5f and Silica-6 + PEGf diverged, and the detected assay enhancement was of different intensity. PEGylated particles (Silica-6 + PEGf) showed a lower degree of enhancement, which was within the acceptance range, whereas the enhancement by the non-PEGylated particles (Silica-5f) was beyond the acceptance range of the assay. The results indicate that PEGylation reduces the enhancement activity of silica nanoparticles.
Iron oxide–silica core shell particles (FexOy@SiO2)
Results of the endotoxin tests for FexOy@SiO2-2.
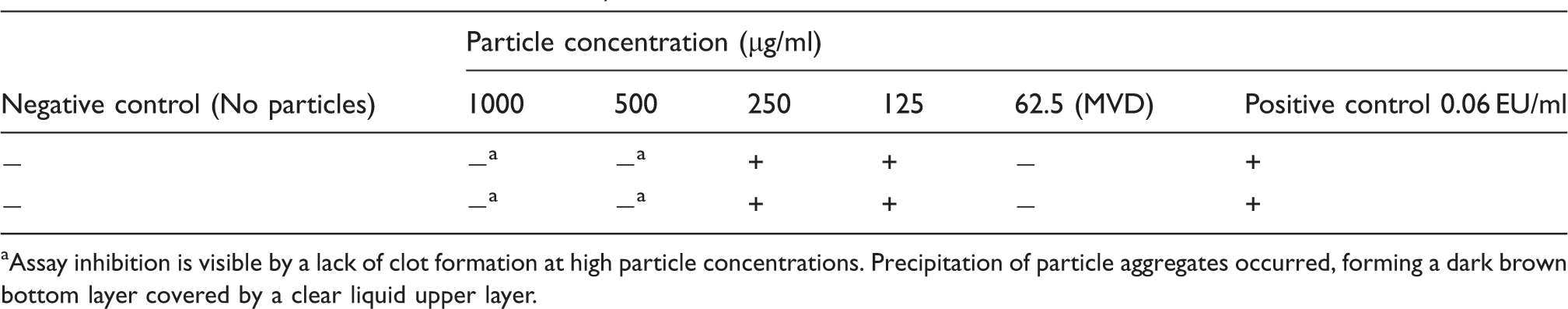
Assay inhibition is visible by a lack of clot formation at high particle concentrations. Precipitation of particle aggregates occurred, forming a dark brown bottom layer covered by a clear liquid upper layer.
Concentration-dependent IEC for FexOy@SiO2-1.
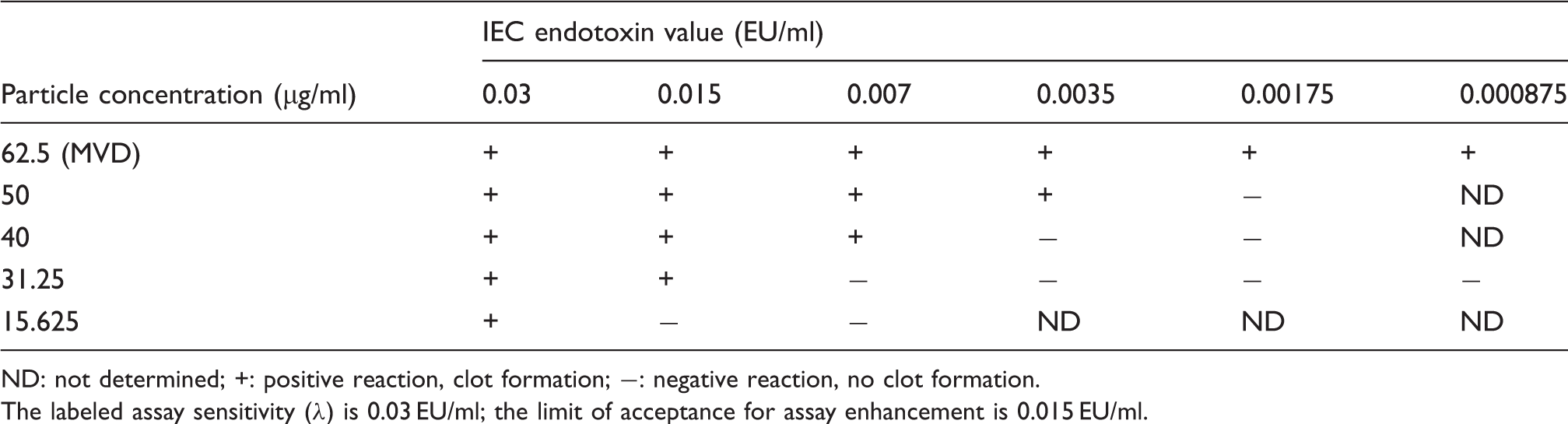
ND: not determined; +: positive reaction, clot formation; −: negative reaction, no clot formation.
The labeled assay sensitivity (λ) is 0.03 EU/ml; the limit of acceptance for assay enhancement is 0.015 EU/ml.
Endorem (FexOy)
Endorem, a formulation based on super paramagnetic iron oxide nanoparticles with a primary particle size of 5 nm, was tested as a reference to the applied iron oxide–silica core shell nanoparticles with a primary particle size of around 3 nm. Endorem (AMI 25) has been approved as contrast agent for the detection of liver metastases by MRI and is applied via intravenous injection. As expected, no endotoxin contamination was detected in diluted and undiluted Endorem. Endorem showed no interference with the LAL gel clot assay if applied in the lowest tested particle concentration (62.5 µg/ml particles). Even in undiluted state, at a particle concentration of 11.2 mg/ml spiked with endotoxin, no full inhibition of the LAL gel clot assay was detected. A stable and homogenously colored brown gel clot was formed, without any sign of visible particle precipitation.
Summary
Detailed inhibition/enhancement screening revealed that in the presence of endotoxins, all tested particles with a silica surface showed enhancement activity in the
At high particle concentrations, iron oxide–silica core shell particles with a small primary particle size of around 3–5 nm led to inhibition of the assay, visible by a lack of clotting. This was not observed using larger iron oxide–silica core shell particles if tested at similar mass concentration. In contrast, dextran-coated iron oxide nanoparticles, Endorem, which have a primary particle size smaller than the tested iron oxide–silica core shell particles, did not lead to a full inhibition of the assay, even if the particle concentration was elevated by a factor of 10 compared with the iron oxide–silica core shell particles.
Reliability of the test results
For most tested silica particles a dilution of the sample within the MVD was sufficient to overcome non-valid enhancement, enabling semi-quantification of the endotoxin contamination. In contrast, for one of the tested silica particle suspensions and all tested iron oxide–silica core shell nanoparticles, enhancement at the MVD was still beyond these limits. For these nanoparticle suspensions, other endotoxin detection methods must be evaluated.
Discussion
The present study shows that nanoparticles with silica surface can interfere with the LAL gel clot assay. Assay interference was detectable as either enhancement or inhibition. The degree of enhancement was variable between nanoparticle samples made of the same material. As mentioned before, assay interference by nanoparticles is a common problem to diverse assay formats.14–16 However, to date, only a few studies have investigated the effects of nanomaterials on the endotoxin detection systems, and showed that several organic and inorganic nanoparticle suspensions interfere with various LAL formats.9,17,18 In this context a systematic study of nanoparticles made of the same material is missing. It is important to note that most tested conventional substances also show some degree of interference with LAL assays, 20 for example by masking the presence of endotoxin, alteration of the endotoxin aggregate structure or coordination divalent cations (Ca2+, Mg2+). Owing to their anionic and amphiphilic character, endotoxins have the potential to bind to virtually every organic and inorganic material. In addition to accumulation on the surface of nanoparticles especially designed for endotoxin binding,29–31 endotoxins were found to bind to several non-functionalized surfaces, for example aluminum silicates, 32 polyethylene, titanium and silica.5,6,33 In case of the here-tested nanoparticle samples, it would be expected that a tight binding of the LPS to the silica surface would reduce the detected endotoxin values. But the presence of a LPS spike within the nanoparticle suspension does not prevent recognition by the factor C. This might be based on the fact that endotoxin binding to certain molecules is dependent on the ionic strength of the surrounding medium. Through contact of the nanoparticle suspension with the LAL, the ionic strength of the surrounding medium can be changed significantly. The nanoparticles become coated with proteins once they enter a biological fluid and various proteins are introduced that compete for the available particle surface. Both the ionic strength and the presence of proteins has the potential to affect binding of the LPS to the silica particle surface. In addition, it cannot be excluded that LPS is bound to the silica particle surface in a way that makes the LPS more accessible for the LPS-sensitive factor C.
Another explanation for the observed assay enhancement activity goes into the mechanism of coagulation. The human coagulation and immune system is very different from the ancient system of the horseshoe crab. The coagulation cascade of the horseshoe crab is one of the best studied invertebrate coagulation systems, and several defense factors and mechanisms have been identified within the last decades.
34
Just a few years ago it was found that factor C, next to its role as pattern recognition receptor for LPS on amebocytes
35
and initiator of the LPS-triggered coagulation cascade, acts as an LPS-responsive C3 convertase.
36
Factor C contains a sushi domain found in human complement factors.
37
Despite the differences, it might be possible that observations made on the human coagulation and complement system can be used to interpret the data obtained here. Studies on the protein corona of different sized silica particles in human plasma showed a high percentage of bound coagulation factors and members of the complement system.
38
As these factors contain highly conserved structural motifs, it is likely that factors of the
At least since the beginning of the twentieth century it has been known that exposure of human blood to foreign surfaces can result in blood coagulation.
39
These observations and further studies led to the discovery of the contact activation pathway, also known as the intrinsic pathway of the human coagulation system. Blood in contact with negatively-charged surfaces, for example amorphous silica, quartz and kaolin, triggers the intrinsic pathway by activation of factor XII, the Hageman factor,
40
which can also be activated by the presence of endotoxins.
41
This contact activation by silica is used to shorten coagulation times in blood collection tubes.
42
In the LAL gel clot assay, shorter coagulation times would cause clotting of samples that would normally not result in clot formation by the end of the assay incubation time. Experimental proof of the above-mentioned assumptions must be made in future experiments, especially measurement and comparison of the coagulation times of the LAL in the presence of the different nanoparticle suspensions. The pro-coagulant properties of several silica nanoparticles on human whole blood and plasma were demonstrated a few years ago.
43
It was shown that coagulation enhancement relies on an immediate activation of factor XII of the intrinsic coagulation pathway. Even if a contact activation mechanism similar to the human coagulation system has not yet been reported in Sensitivity of the LAL gel clot assay in dependance on the applied concentration of the iron oxide-silica core shell particles FexOy@SiO2-1 and calculated surface area respectively. Labeled sensitivity of the assay is 0.03 EU/ml. Elevation of the particle concentration leads to an increase in surface area available for protein–particle interactions, which becomes noticeable in enhancement of the assay sensitivity.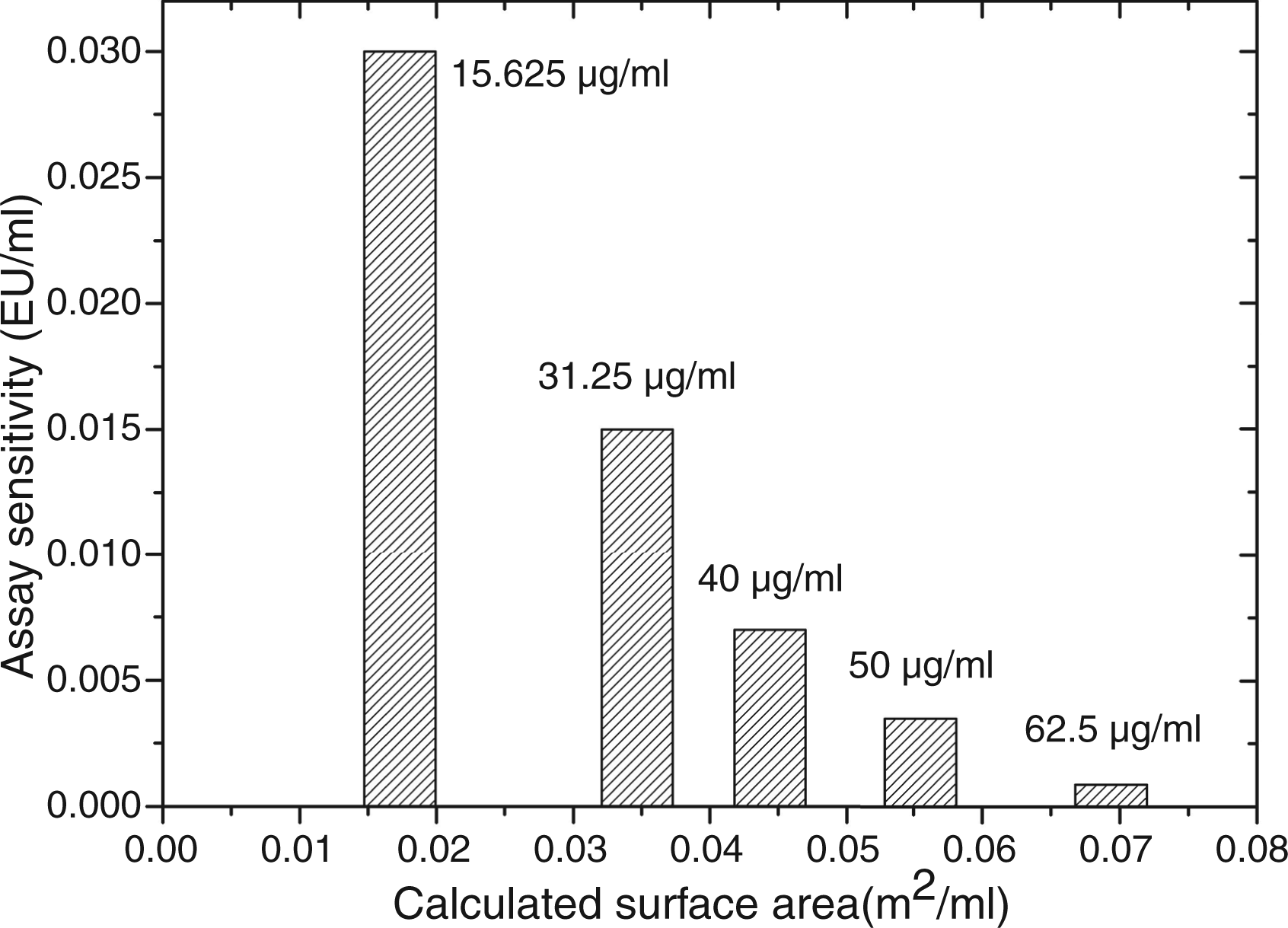
The assumption that protein–particle interactions are the driving force for the assay enhancement is strengthened by the results obtained from the comparison of silica particles with and without PEGylation. As expected, the PEGylated particles differ in enhancement intensity compared with the non-PEGylated particles. This can be explained by the protein-repelling characteristics of PEGylated materials. PEGylation of titanium surfaces, known to initiate the intrinsic pathway, was shown to enhance blood compatibility by reducing protein–surface interaction and contact activation, resulting in prolonged coagulation times of human blood in contrast to non-modified titanium surfaces. 46 Similar observations were made by Tavano et al. 43 by comparison of PEGylated and non-PEGylated ORMOSIL nanoparticles.
In this study, next to enhancement we also observed assay inhibition. Inhibition was caused by small-sized nanoparticles (∼ 3–5 nm), which have an extremely large surface-to-volume ratio, a strong curvature and high number of atoms on the particle surface. At a comparable mass concentration, the available surface area for interaction with proteins is much larger for small particles than for large particles (see Figure 3).
1
The ratio of the available surface to the amount of proteins present in the LAL is different for small and large particles. There are two possible explanations for the observed inhibition. First, on the one hand, the small particles might destabilize the gel clot by binding to coagulin monomers to a high extent, impairing the formation of a firm gel clot. On the other hand, low particle concentrations might lead to a stabilization of the gel clot, resulting in enhancement. Second, concerning only the available surface area for protein binding, enhanced adsorption of a significant amount of coagulation factors or other proteins involved in coagulation could reduce the coagulation rate up to a rate that does not lead to the formation of a firm gel clot within the assay incubation time. An excess of surface area over the amount of proteins available for binding can lead to binding of proteins that have lower binding affinity to the particles,
47
and are normally not able to compete with high-affinity proteins. Coagulogen is a possible candidate to bind to the particle surface. Coagulogen has functional, but not structural, similarity to vertebrate fibrinogen, and the coagulin monomer is a functional analogue to the fibrin monomer.11,48,49 In single protein studies, as well as in studies on human blood samples, it has been shown that fibrinogen can adsorb to silica particles,50,51 and that its secondary structure changes when adsorbed onto small silica particles with a radius < 30 nm.
52
In the 1980s Vroman et al.53,54 observed that on clot-promoting surfaces (glass or oxidized metals) fibrinogen is initially deposited from plasma, but is displaced by high molecular mass kininogen and other molecules within a few minutes—an effect called the ‘Vroman effect’. The increased surface area of the small iron oxide silica particles might reduce the competition with high affinity molecules and enable the adsorption of a significant amount of coagulogen/coagulin molecules onto the particle surface, which, in turn, would lead to a reduction of free coagulogen/coagulin molecules within the lysate. In addition, a change in secondary structure would decrease the amount of intact coagulogen molecules. Both effects could lead to a significant reduction of the coagulation rate and clot forming ability by the small iron oxide–silica core shell particles, but not by the large iron oxide–silica core shell particles with a radius >30 nm. In our study, small dextran-coated iron oxide nanoparticles (5 nm in size) in a 10-fold higher mass concentration than the iron oxide–silica nanoparticles did not lead to a full inhibition of the assay. This might be due to the presence of 10% low molecular dextran within the formulation, in part covering the nanoparticle surface, or simply in altered surface characteristics due to the different material. In preliminary studies, high concentrations of citrate–stabilized iron oxide nanoparticles of comparable particle size also showed inhibition of the LAL gel clot assay. But the possibility remains that this effect is based on another mechanism, the anti-coagulation activity of citrate, which is known to affect LAL detection systems if applied in certain concentrations. Owing to the complexity of the LAL, as well as the multiple factors that influence protein–particle interaction, thus far only assumptions can be made about the mechanisms of the observed assay interference. Further information is needed, especially on the protein composition of the commercial LAL reagent. As the LAL reagent is a biological product, fluctuations in the composition of different batches might also lead to different results. Lot-to-lot variability of the results must be investigated in future studies.
Calculated particle surface area as a function of primary particle size. A decrease in particle size is connected to an exponential increase in particle surface area. Squares correspond to the size of the tested particle samples.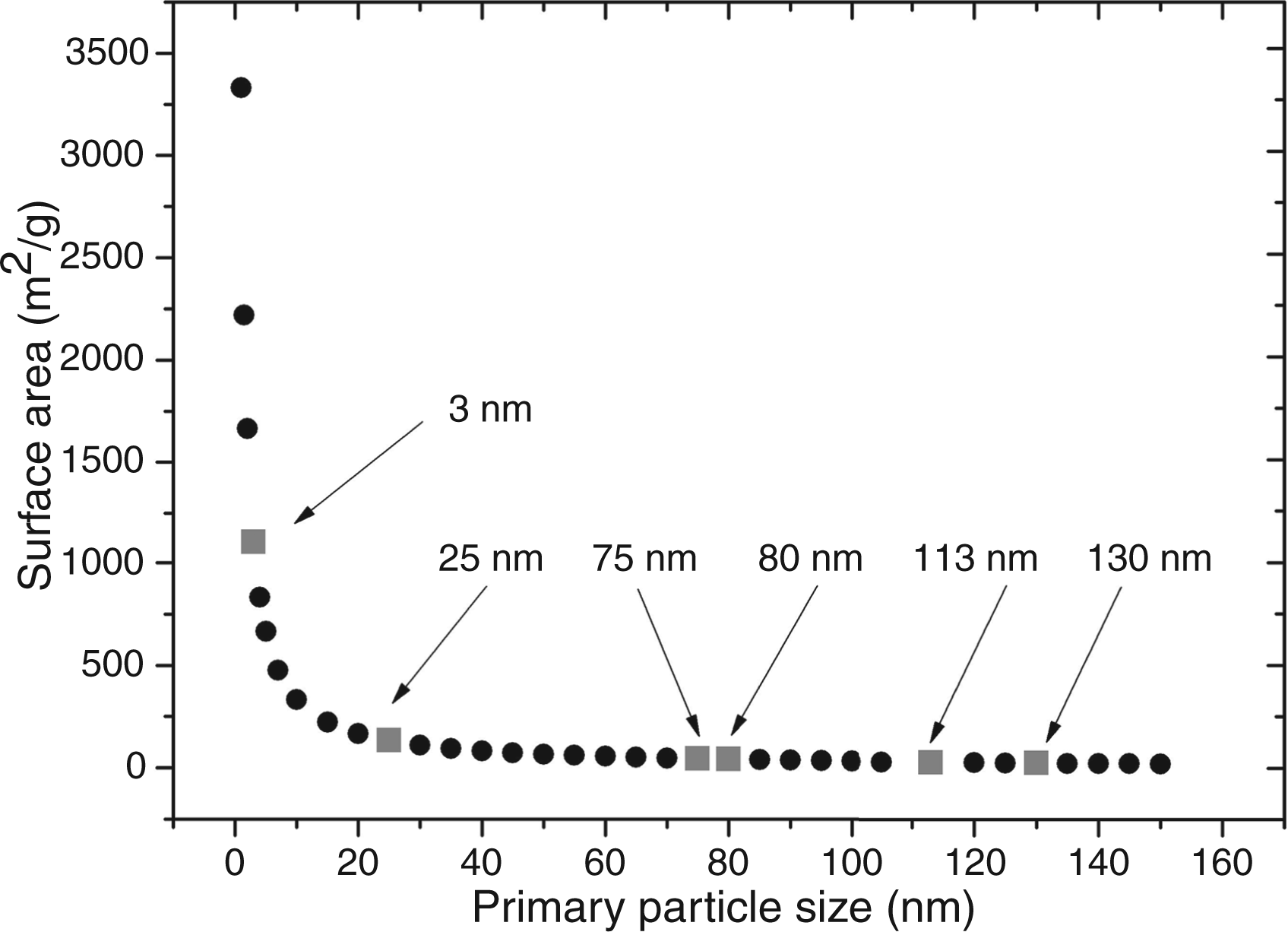
Conclusions
The results, once again, show that every nanoparticle type and individual batch has to be carefully tested for inhibition and enhancement of a biological assay. Results obtained with one particle type cannot be necessarily applied to a second particle type made of the same material. Even minor changes of the physicochemical properties of the nanoparticle suspension can render the assay results invalid. The size, composition, zeta potential, surface curvature, accessible surface area, and aggregation behavior of the nanoparticles, as well as the protein–surface interaction seem to be crucial factors that influence the outcome of the LAL gel clot assay. As biological assays consist of complex media, it is difficult to draw conclusions from the results to the underlying mechanisms. For nanoparticle suspensions with strong inherent optical properties, for example light absorption and scattering, as well as strong interference with the LAL gel clot assay, other alternative methods without optical read out must be found to obtain reliable results concerning endotoxin contamination.
Footnotes
Acknowledgements
We thank Thomas Jung, Nanogate AG, Germany, for providing iron oxide nanoparticles. We also thank Aljosha Rakim Jochem, Sarah Schmidt, Anika Schwarz and Anika Weber for assistance in particle synthesis and physicochemical characterization; Marcus Koch for SEM/TEM investigation; and Henrike Peuschel, Jennifer Atchison and Julia Callender (all INM) for proofreading the manuscript. We thank Eduard Arzt for continuing support of our work at INM.
Funding
This work was funded by the Federal Ministry of Education and Research (BMBF), in frame of the project ‘NanoKon - Systematic evaluation of health effects of nanoscale contrast agents’ [grant number 03X0100C].
Conflict of interest
The authors do not have any conflicts of interest to declare.
