Abstract
Corneal Langerhans cells (LCs) offer the opportunity to gain insight into the activity of the innate immunity. We examined the density and the distribution of LCs and compared the results with dry-eye parameters in rheumatoid arthritis (RA). Fifty-two RA patients with various degrees of disease activity and 24 healthy subjects were enrolled. Peripheral and central LC number and morphology were assessed with in vivo laser confocal microscopy. In addition, ocular surface disease index (OSDI), lid parallel conjunctival folds, Schirmer test, and tear break-up time (TBUT) were evaluated. The prevalence of central and peripheral LC, and the central LC morphology values (LCM) were higher than normal in RA. Within the RA group, LC prevalence and morphology were not affected by disease activity. However, patients on anti-TNF or glucocorticosteroid (GCS) therapy exhibited normal LCM, and normal central and peripheral LC density. OSDI was higher and TBUT was lower than normal in RA. The alteration of LC in RA suggests an active inflammatory process in the cornea, which may reflect an increased activation state of the innate immune system—even in inactive stages of RA and without ocular symptoms. The results also indicate ocular effects of GCS therapy in RA.
Introduction
Although the predominant feature of rheumatoid arthritis (RA) is joint involvement, further extra-articular manifestations, including ocular involvement, may also be present. The most common ocular problems in RA are sicca symptoms and about 10% of RA patients develop secondary Sjögren’s syndrome. 1 Episcleritis, scleritis and a variety of corneal changes, including keratitis and keratoconjunctivitis sicca, may also occur. The cells participating in these processes, however, are poorly clarified. The highly potent antigen-presenting dendritic cells, the so-called corneal Langerhans cells (LCs), act as potent inducers and regulators of local corneal immune processes.2–4 They can stimulate both primary and secondary T- and B-cell responses. The distribution and role of LCs in the central cornea is an issue of ongoing discussion. Several investigators have demonstrated the presence of LCs in the epithelium and stroma of the central cornea ex vivo.5–7 Moreover, the presence of LCs in the central corneal epithelium of healthy volunteers has also been documented. 6 Detailed information concerning the maturation and migration of LCs in response to cytokines and chemokines is not yet fully available,3,8,9 but some data suggest the principal role of pro-inflammatory cytokines, including TNF-α.10,11 In recent years new techniques that directly visualize microscopic changes of ocular structures in vivo have become available for clinical use. Confocal microscopy, first described by Minsky, 12 now applies a laser beam to obtain coronal optical sections of the cornea and to collect data even on the prevalence of certain cell subtypes. With this new tool we characterized the presence and distribution of LCs in the corneal epithelium of RA patients with various disease severities.
Materials and methods
Patients
Clinical characteristics of healthy individuals and RA patients. Data are expressed as median [interquartile range].

Eye examinations
All examinations were carried out on the right eye only in the same room among unchanged conditions.
Dry eye examinations
An ocular surface disease index (OSDI) questionnaire was used to evaluate the subjective ocular discomfort. 15
Lid parallel conjunctival folds (LIPCOF) are subclinical folds of the conjunctiva, situated at the temporal and nasal aspect of the bulbar conjunctiva. Loosened conjunctiva creating folds, detectable by slit lamp are thought to reflect dryness of the ocular surface. According to the height and morphology, grading can be made on a 0–3 scale reflecting the severity of dry eye. LIPCOF was measured at the temporal aspect of the lower eyelid margin during routine slit lamp examination, according to the protocol by Pult. 16
The Schirmer-strip (Haag-Streit, UK, Ref: 4701001) was used to evaluate tear production and was placed at the temporal aspect of the lower conjunctival sac. Values were read after 5 min with the help of a ruler.
Tear break up time (TBUT) was determined after instillation of one drop of fluorescein dye into the lower conjunctival sac.
In vivo confocal microscopy investigations
All patients were examined with a digital corneal confocal laser-scanning microscope (HRT II RCM Heidelberg Engineering Inc., Heidelberg, Germany, Rostock Cornea Module), equipped with the in-built software Heidelberg Eye Explorer version 1.5.10.0.
In vivo confocal microscopy (IVCM) uses helium neon diode laser source with a wavelength of 670 nm. The two-dimensional images captured by the IVCM have a definition of 384 × 384 pixels over an area of 400 µm × 400 µm, with lateral spatial resolution of 0.5 µm and a depth resolution of 1–2 µm.
Data collection
Topical anaesthetic drops (Oxybuprocaine-Humacain 0.4%, Human Pharmaceuticals, Gödöllő, Hungary) were applied to the right eye and confocal cornea microscopy was carried out. Proper alignment and fixation of the eye was maintained with a target mobile red light for the contralateral eye. Sterile packaging disposable plastic cap (TomoCap®, Heidelberg, Germany) was used to keep the distance from the cornea to the microscope head stable. Carbomer gel (Vidisic; Dr Mann Pharma, Berlin, Germany) was used as coupling medium. Positioning of the objective lens was monitored with a digital camera tangential to the eye.
LCs were seen as hyper-reflective corpuscular particles with or without dendrite-like protrusions situated at the level of the basal epithelial cells and the sub-basal nerve plexus. We found LCs to have an average diameter of up to 15 µm in the corneal centre. LC densities were examined both at the centre of the cornea and its periphery at the 6 o’clock position at a depth of 40–60 µm from the corneal surface at the level of the basal epithelial cell layer and sub-basal nerve plexus. Forty images were taken and the five best-focused images were considered for the analysis in a masked fashion. After selection of the region of interest, cells could be marked and the system calculated cell density automatically (cell number/mm
2
). LC morphology (LCM) was evaluated on a 0–3 scale, where score 1 represented LCs without dendrites, score 2 was given to LCs with small processes and score 3 was given to LCs with long processes. Score 0 described the condition when the cornea was devoid of LCs. The average LCM was calculated in each of the figures selected and was used to describe the maturation of the LCs at both locations of the cornea (Figure 1).
Confocal microscopic image of the corneal centre (A) and the periphery (C) in the control group. Note that central area is without obvious signs of the presence of Langerhans cells (LC) [LC morphology (LCM): score 0], while in the periphery some non-activated LCs (white arrow indicating a score 1 LC) can be seen accompanied by sub-basal nerves. (B) Corneal centre with gathering LCs with small processes (white arrowhead shows a score 2 LC). (D) Long and interdigitating LCs (empty arrow pointing at a score 3 LC) situated in a network-like fashion in the periphery indicating mature form of this cell type in the rheumatoid arthritis (RA) group.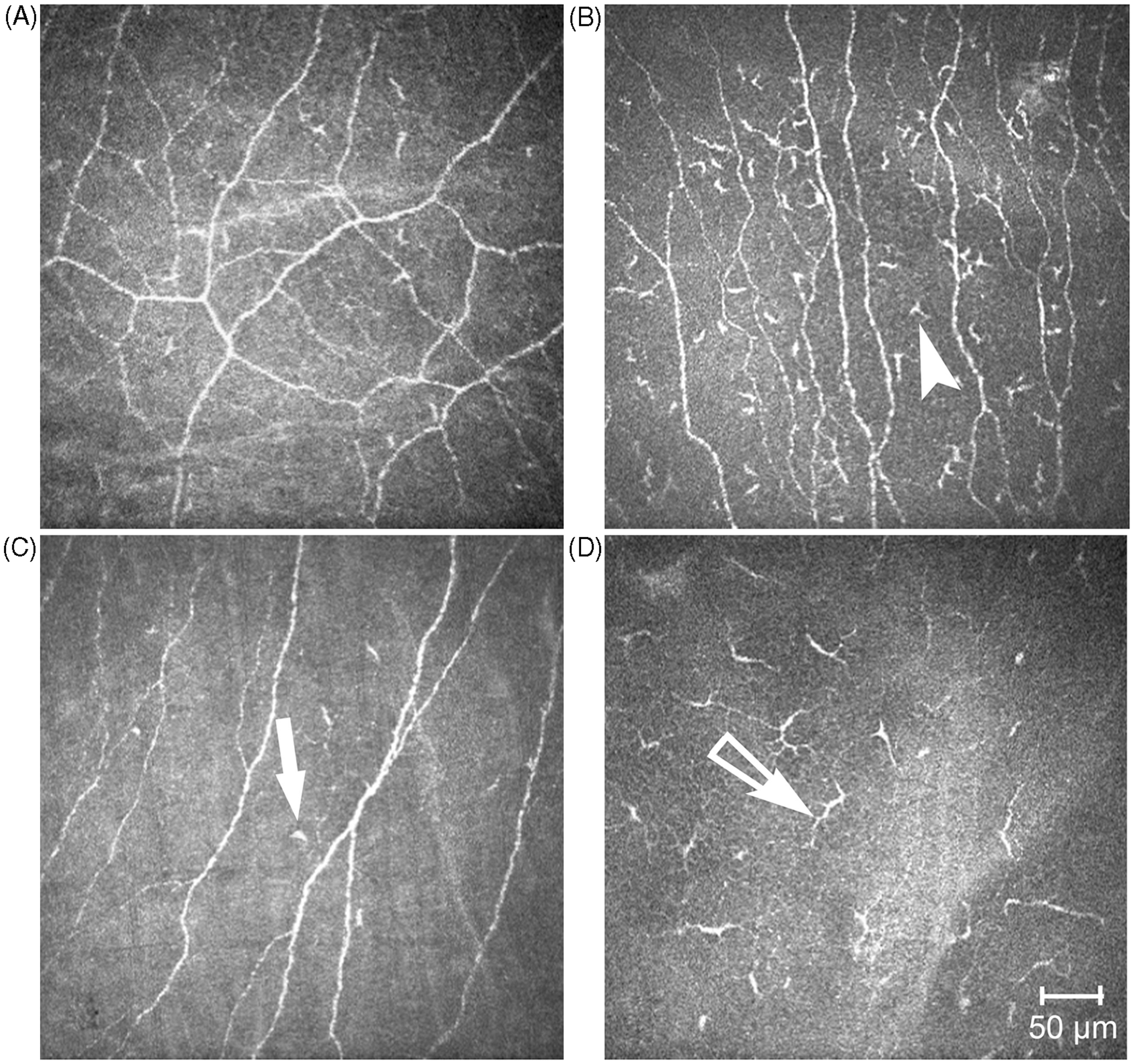
Statistics
Data are expressed as mean ± SD. Comparisons between healthy individuals and RA patients, as well as between RA patient subgroups, were made with Cochran-Armitage trend tests in case of ordinal variables (LCM and LIPCOF). Continuous variables were compared using t-tests. Prior to analysis, the distribution of variables were checked graphically and, if necessary, variables were Box-Cox transformed prior to the analysis. We tested all possible two-way interactions (anti-TNF-α therapy*DAS28, DAS28*glucocorticosteroid, anti-TNF-α therapy *glucocorticosteroid) in the group of RA patients. None of these interactions were significant (details not shown). We used Spearman’s rank correlation test to investigate correlations within groups. P-values <0.05 were considered significant. Statistical analysis was performed using STATISTICA version 8.0 (StatSoft, Tulsa, OK, USA).
Results
Dry eye-related parameters in different subgroups of RA patients. Data are expressed as mean ± SD. Comparisons between healthy individuals and RA patients, as well as between RA patient subgroups, were made with Cochran-Armitage trend tests in the case of LIPCOF values. Continuous variables were compared using t-tests. Prior to analysis the distribution of variables was checked graphically and, if necessary, variables were Box-Cox transformed prior to the analysis.
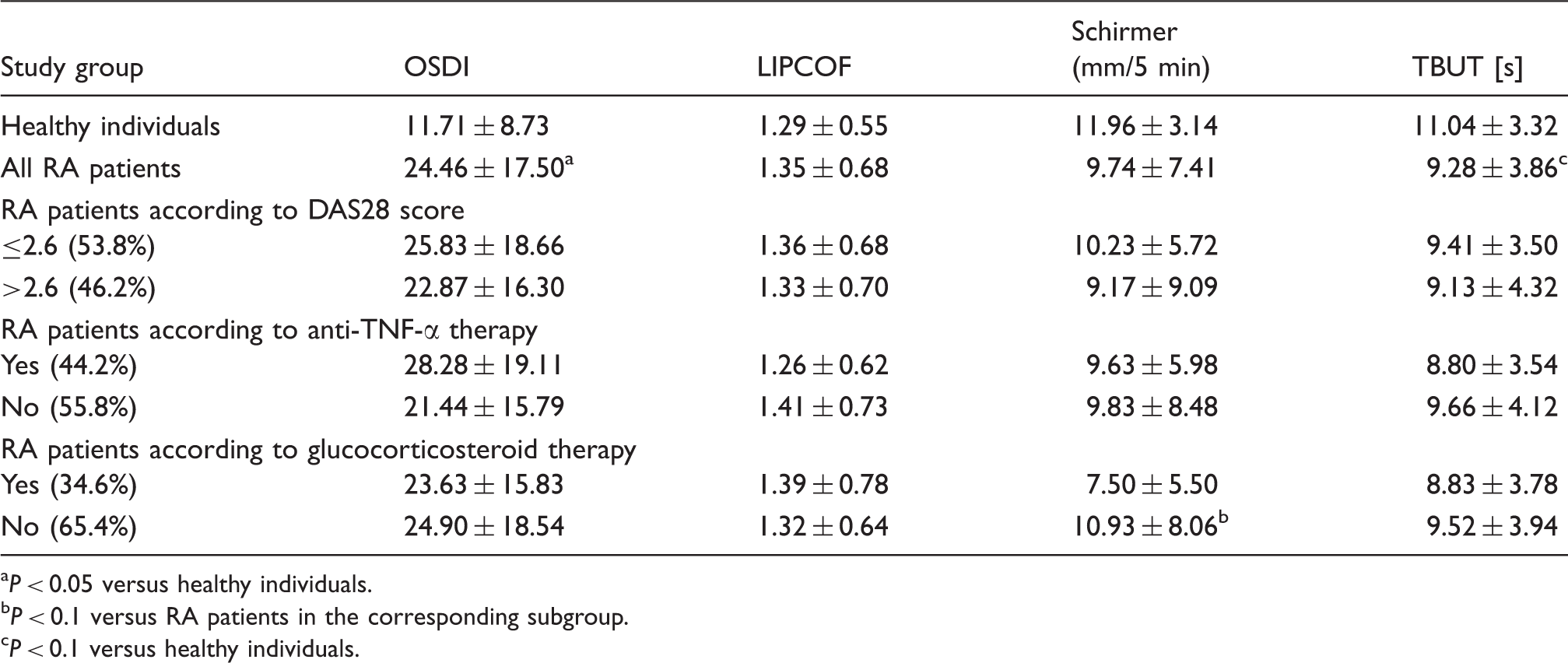
P < 0.05 versus healthy individuals.
P < 0.1 versus RA patients in the corresponding subgroup.
P < 0.1 versus healthy individuals.
Confocal microscopy data
Confocal microscopy results in different subgroups of RA patients. Data are expressed as mean ± SD. Comparisons between healthy individuals and RA patients, as well as between RA patient subgroups, were made with Cochran-Armitage trend tests in case of LCM values. Continuous variables were compared using t-tests. Prior to analysis the distribution of variables was checked graphically and, if necessary, variables were Box-Cox transformed prior to the analysis.
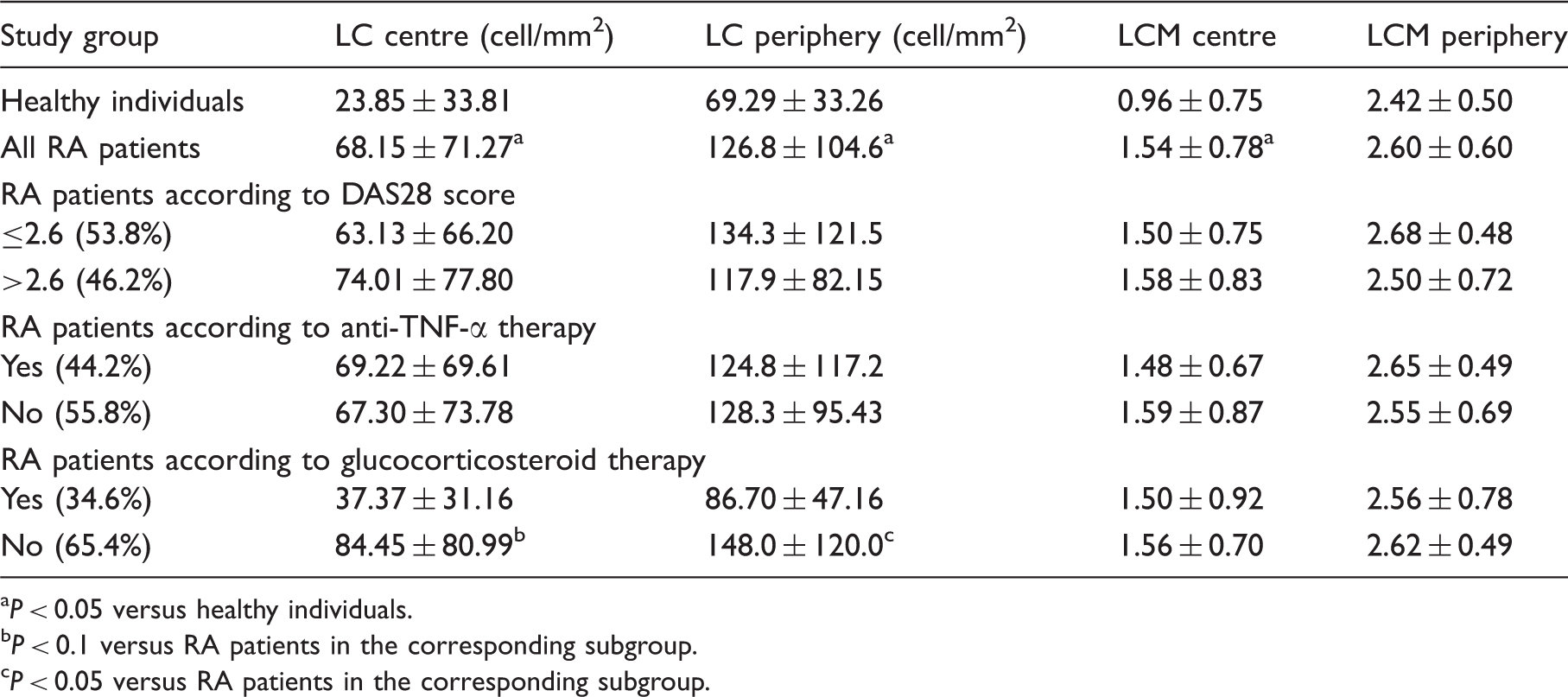
P < 0.05 versus healthy individuals.
P < 0.1 versus RA patients in the corresponding subgroup.
P < 0.05 versus RA patients in the corresponding subgroup.
We also compared the LC density and morphology between RA subgroups formed according to therapy. LC density values were higher in RA patients without GCS therapy. In other words, GCS therapy restored the increased LC density observed in RA patients without GCS.
Discussion
To the best of our knowledge, this is the first study to investigate the characteristics of corneal LC density and morphology in RA. We have demonstrated increased density and activation of dendritic cells in the cornea of RA patients in the absence of clinically manifest ocular inflammation. This increased LC presence and activation did not appear to be associated with articular inflammatory disease activity, but correlated with the severity of hypolacrimation, and was abrogated by GCS therapy.
With the development of histochemistry and electron microscopy, a huge body of evidence was accumulated on corneal immunology, which required the revision of the tenet on the cornea as an immune privileged tissue. 17 Various immune cells were demonstrated to reside in the cornea, including LCs. 7 In healthy conditions, LCs are scarce in the centre of the cornea. Upon different triggers, however, LCs migrate to the centre of the cornea and transform into an active participant of the corneal immune response by the formation of dendrite-like processes and expression of co-stimulatory molecules, including CD80 and CD86. 18 LCs are found in a greater number in several inflammation-related conditions, including contact lens wearers 3 and Thygeson’s superficial punctate keratitis,8,9 and also in experimental conditions.19,20 The elevation of LC and leukocyte density both in the centre and the periphery of the cornea has already been described in patients with Sjögren’s syndrome. 21 In this study we have used confocal microscopy, a recently developed device that is non-invasive and capable of visualizing the ultrastructure of the ocular surface, including the meibomian glands at a cell level. 22 Reliability and specificity of the device have been acknowledged in a study by Imre et al.; 23 however, there are some limitations when it is used on corneas with decreased transparency. We aimed to investigate the density and morphology of corneal LCs for their presumed role in ocular surface diseases and their properties to indicate the corneal homeostasis. LCs reside almost exclusively in the superficial cornea and can be distinguished from leukocytes through their appearance. They have an extraordinary capacity to stimulate naïve T cells and are recognized as essential regulators of both the innate and acquired arms of the immune system.7,21 We have observed that, in contrast with healthy subjects, LC density was elevated markedly and found to reside in the corneal centre of RA patients, even without overlapping Sjögren’s syndrome or in the absence of overt eye symptoms. Additionally, more than half of these LCs showed an activated phenotype with small or long projections.
It has been shown that the recruitment and maturation of LCs can be induced by several pro-inflammatory cytokines, including IL-1α, TNF-α, IL-6, IL-8 and IL-12.10,23 Both IL-1 inhibitors, anti-TNF-α therapy and GCS suppressed the LC migration in vitro effectively. 24,25 Our clinical results also suggest this possible inhibitory effect of GCS therapy on the activation of LCs in RA.
Corneal immune status may be strongly interrelated with dysfunctional tear production, but the causative relationship has still not been established. It has long been known that dry eye is more common among patients with autoimmune diseases, 1 but the precise pathophysiologic mechanism is still unclear. Hyposecretory conditions, such as Sjögren’s syndrome, through the reduced moisturization of the ocular surface, may initiate or contribute to the development of dry eye. Corneal morphological changes like thinning of the cornea, lower density of superficial epithelial cells, fewer sub-basal nerves and greater nerve tortuosity have been described in association with dry eye 26 and in RA subjects. 27 Inadequate moisturization leads to increased apoptosis and further induction of pro-inflammatory cytokines, including IL-1, 6, 8 and TNF-α, which have been found to be overexpressed and to accumulate in the ocular surface. In turn, these cytokines are thought to have the capacity to decrease tear production via neuronal and hormonal effects.28,29 Indeed, we found decreased tear production in RA patients who had at least moderate RA activity. Theoretically, higher clinical activity in RA results in higher systemic inflammation, which may also increase the secretion of pro-inflammatory cytokines in tears. The possible higher amount of these cytokines in the tears of RA patients with higher disease activity might impair tear production and it might partly explain the dry eye mechanism without the presence of true overlapping Sjögren’s syndrome. It is of importance to emphasize that none of the RA patients had overlapping Sjögren’s syndrome. Our results are in accordance with the results of Villani et al., 27 which is of particular interest because some reports suggest that there are profound aetiologic and pathogenic differences between keratoconjunctivitis sicca in patients with RA with secondary Sjögren’s syndrome, and patients with RA without secondary Sjögren’s, though they lack manifest differences in the clinical indicators. 30 Rather, our data, along with that of Villani et al., 27 suggest that these alterations of the cornea are associated with the systemic inflammatory effect of the disease than a specific pathogenic difference in KCS in patients with or without Sjögren’s syndrome. Also, our data, together with that of Villani et al., 27 may lend itself to the identification of some indices of disease activity on the ocular surface that could be compared with the indices of activity of the systemic disease. Our results are also in agreement with previous results, which showed that ocular surface LCs are essential for T cell-mediated dry eye. 31 Our results also support the systemic multi-organ-related feature of RA affecting the eye. Recent data showed an increased accumulation of dendritic cells (DCs) in the synovial fluid of patients with active RA and the decrease in DCs in the peripheral blood. Because DCs migrate to sites of inflammation, lack of them in the peripheral blood of patients with active RA may be owing to the recruitment of DCs at the synovium. According to our results a similar process might act also in the cornea of patients with mild or moderate active RA. 32 We have also demonstrated that low-dose, continuously-used oral GCS therapy can significantly reduce the density of LCs, both in the central and peripheral parts of the cornea. Low-dose, continuously-used oral GCS therapy also inhibits the dendrite-forming ability of central LCs. These results also suggest that the immunosuppressive effects of GCS also affect the corneal immune mechanisms in RA.
Footnotes
Funding
Attila Balog and Gergely Toldi were supported by the Magyary Zoltán Hungarian Scientific Scholarship (Grant no. TÁMOP-4.2.4. A/2-11-1-2012-001).
