Abstract
Innate immunity is triggered by a variety of bacterial molecules, resulting in both protective and potentially harmful pro-inflammatory responses. Further, innate immunity also provides a mechanism for the maintenance of homeostasis between the host immune system and symbiotic or non-pathogenic microorganisms. However, the bacterial factors that mediate these protective effects have been incompletely defined. Here, it was demonstrated that the lantiobiotic nisin Z is able to modulate host immune responses and mediate protective host immunity. Nisin Z induced the secretion of the chemokines MCP-1, IL-8 and Gro-α, and significantly reduced TNF-α induction in response to bacterial LPS in human PBMC. The results correlated with the ability of nisin Z to confer protection against both the Gram-positive organism Staphylococcus aureus, and the Gram-negatives Salmonella enterica sv. Typhimurium and Escherichia coli in murine challenge models. Mechanistic studies revealed that nisin Z modulates host immunity through similar mechanisms as natural host defense peptides, engaging multiple signal transduction pathways and growth factor receptors. The results presented herein demonstrate that, in addition to nisin Z, other bacterial cationic peptides and, in particular, the lantibiotics, could represent a new class of secreted bacterial molecule with immunomodulatory activities.
Introduction
The co-evolution of bacteria and their eukaryotic hosts has led to homeostatic mechanisms that provide mutual benefits to both the host and microbe. Indeed, it has been estimated that 100 trillion microbes may reside in the human gut, comprised of resident (autochthonous) and non-resident (allochthonous) members, and this symbiotic relationship relies on mutual recognition and tolerance. Indeed, the structural and immunological development of the gut correlates with the presence of intestinal bacteria. 1 However, few of the specific microbial biomolecules or mechanisms that mediate this homeostatic relationship with host immunity have been elucidated.
Initial evidence supporting a role for commensal and non-commensal bacteria in the modulation of host immunity was derived from the developmental defects observed in germ-free mice. These mice demonstrate defective development of the small intestine and mesenteric lymph nodes with associated immunological deficits, including reduced numbers of CD4+ and CD8+ T-cells, and marked reductions in the expression levels of secretory IgA, aniogenin 4, regenerating islet-derived 3γ (REG3γ) and IL-10. 1 Colonization of germ-free mice with the human symbiont Bacteroides fragilis, or treatment with purified polysaccharide A (PSA) from B. fragilis, promoted beneficial host immune responses in expansion and differentiation of splenic CD4+ T cells, restoring balance in Th cell subsets and directionality of lymphoid organogenesis. 2 Furthermore, the oral administration of purified PSA led to reductions in the pro-inflammatory cytokines TNF-α, IL-17 and IL-23, and reduced disease-associated mass loss and inhibition of epithelial cell hyperplasia and neutrophil infiltration to the gut. 2 Similarly, exopolysaccharides (EPS) isolated from the commensal Bifidobacterium lonhum BCRC 14 634 stimulated macrophage proliferation and IL-10 secretion, and suppressed LPS-mediated TNF-α release in vitro. 3 Analogously, short-chain fatty acids produced following fermentation of dietary fiber were able to attenuate inflammation in experimental colitis, arthritis and asthma following interaction with G-protein coupled receptor 43-expressing neutrophils. 4 Conversely, commensal organisms employing as probiotics can actually suppress potentially excessive inflammatory responses, 5 possibly as a mechanism for preventing excessive inflammation in response to these commensals, which themselves contain the bacterial signature molecules that trigger inflammation/innate immunity by interacting with TLRs.
Lactococcus lactis, a non-pathogenic lactic acid bacterium used in food fermentation and found commonly in milk products, has gained considerable attention recently for its use as a probiotic agent. 6 Indeed, the therapeutic application of genetically modified L. lactis has been documented as safe and effective, and it is presently being examined in Phase I and II clinical trials involving the treatment of inflammatory bowel disease, ulcerative colitis and oral mucositis. 6 In addition, pre-clinical trials are currently in development for the treatment of celiac disease, type I and II diabetes, allergies and a pneumococcal vaccine.6,7 Although there has been considerable investigation into the therapeutic potential of L. lactis, and suggestions that it modulates immune function, there is a paucity of information regarding the role of L. lactis biomolecules in the mediation of host immunity homeostasis.
Recently, there has been considerable interest in the Type A (I) lantibiotics, a group of bacterially-secreted cationic peptides, owing to their broad Gram-positive antimicrobial activities, high potency, low associated cytotoxicity and limited ability to promote antimicrobial resistance. 8 Such lantibiotics are produced in a broad range of organisms ranging from commensals like Streptococcus salivarius to pathogens like Staphylococcus aureus, S. epidermidis and Enterococcus spp. 8 Nisin Z is a 34-amino acid Type A (I) lantibiotic produced by L. lactis 9 and has been used as a broad-range food preservative for more than 50 years.10,11 In 1988, nisin was confirmed to be ‘generally recognized as safe’ in the USA with Food and Drug Administration (FDA) approval for use in pasteurized, processed cheese spreads 11 and is currently licenced in 48 countries. BiosynexusInc is developing nisin for human clinical applications and Immucell Corp. has licensed Mast Out, an anti-mastitis nisin-containing product, to Pfizer Animal Health.11,12 The antibacterial activity of nisin Z has been well studied; activity is due to its targeting of lipid II, an enzyme involved in peptidoglycan synthesis, as well as lipid II-mediated pore formation in the cytoplasmic membrane.13–15 The interaction between nisin and host immunity has remained largely undefined, though it has been demonstrated that short-term dietary administration resulted in increased CD4 and CD8 T-lymphocytes, and decreased B-lymphocytes, whereas prolonged administration resulted in a return to normal levels of both B- and T-lymphocytes. 16 More recently, it was demonstrated that nisin also stimulates neutrophil extracellular trap (NET) production through the activation of human neutrophils. 17
Despite profound structural differences to natural cationic host defense (HDP) and synthetic innate defense regulator (IDR) peptides, the similar physicochemical properties, ability to interact with negatively-charged biological membranes, and the fact that human commensal and pathogenic bacteria produce such cationic lantibiotics, led us to propose that lantibiotics would have immunomodulatory activities similar to those described recently for HDPs and IDRs. HDPs are ubiquitous in nature as an essential component of the immune system of all complex organisms. Although characterized initially by their often-weak broad-spectrum direct antimicrobial activities, many HDPs are pleiotropic with a potent ability to modulate immune responses.18,19 Indeed, HDPs have the ability, both in vitro and in vivo, to upregulate the synthesis of chemokines and their receptors, recruit leukocytes to sites of infection, suppress potentially harmful inflammatory responses, stimulate angiogenesis and wound healing, and upregulate dendritic cell activation markers and maturation.18,20–23 As a result of these broad immunomodulatory activities, there is increasing interest in the use of immunomodulatory HDPs as design templates for the development of novel IDR derivatives with enhanced activities.
Here, we demonstrate that the bacterial lantibiotic nisin Z has immunomodulatory properties analogous to those described recently for many HDPs, including LL-37,24–28 the defensins,29–31 and the synthetic peptides IDR-132 and IDR-1002. 33 Nisin Z was able to modulate innate immune responses through the induction of chemokine synthesis and suppression of LPS-induced pro-inflammatory cytokines. Interestingly, nisin Z appeared to utilize similar mechanism(s) of action for modulating host immune responses as those described recently for many HDPs and IDRs. Further, nisin Z administered prophylactically or therapeutically conferred protection in murine models of Gram-negative bacterial infection, indicating an in vivo immunomodulatory role for this microbial biomolecule. Overall, our results demonstrate that lantibiotics and, in particular nisin Z, represent a new class of bacterially-secreted peptides that manipulate host immunity.
Materials and methods
Reagents
All inhibitors used in this study were supplied by Enzo Life Sciences (Farmingdale, NY, USA). The inhibitors were re-suspended in DMSO (Sigma-Aldrich, Oakville, Canada) and stored at −20℃. The final concentrations of DMSO in cell culture never exceeded 0.02% (v/v) and all experiments included DMSO vehicle controls. The inhibitors were also checked against possible cytotoxic effects using a lactate dehydrogenase release colorimetric cytotoxicity detection kit (Roche, Basel, Switzerland).The purification of Pep534 and gallidermin 35 have been described previously.
Nisin expression and purification
The nisin-producing strain L. lactis was cultivated in tryptic soy broth (pH 6.5) (Merck, Darmstadt, Germany) without shaking at 30℃ for 24 h. The overnight growth was sub-cultured 1 in 4 into fresh media and incubated at 30℃ for 1 h. For autoinduction 1 µg/ml purified nisin was added to the culture and incubated at 30℃ for 5 h. Cells were harvested by centrifugation (10,000 g for 10 min). For isolation and purification of nisin, chloroform was added to the supernatant fluid (0.2:1, vol/vol), stirred vigorously overnight at 4℃ and centrifuged (10,000 g for 10 min) for phase separation. The precipitate formed at the interface between the chloroform and culture supernatant fluid was lyophilized. The crude extract was re-suspended in 20% acetonitrile −0.1% trifluoroacetic acid (TFA) and applied to a preparative HPLC column [Nucleosil 100-C18; 10 µm, 225 × 20 mm (inner diameter); Schambeck SFD, Bad Honnef, Germany]. The column was equilibrated with buffer A [H2O, 0.1% (vol/vol) TFA], and peptides were eluted using a linear gradient of 20–60% buffer B [acetonitrile, 0.1% (vol/vol) TFA] at a flow rate of 12 ml/min. For further purification, a semi-preparative [Nucleosil 100-5C18; 250 × 8.6 mm (inner diameter)] and/or analytical [Nucleosil 100-3C18; 10 µm, 250 × 4.6 mm (inner diameter)] column was used. MALDI-TOF spectrometry was used to confirm the correct mass and the purity of the peptide. Stock solutions were prepared in 0.05% acetic acid and stored at −20℃.
Cell isolation and nisin Z stimulation
Venous blood from healthy volunteers was collected in Vacutainer® collection tubes containing sodium heparin as an anticoagulant (BD Biosciences) in accordance with University of British Columbia ethical approval and guidelines. Blood was diluted with an equal volume of complete RPMI 1640 medium, supplemented with 10% (v/v) heat-inactivated FBS, 2 mM
Chemokine ELISAs
Following 24-h peptide exposure, the tissue culture supernatants were centrifuged at 16,000 g at 4℃ for 5 min in an IEC MicroMax centrifuge to obtain cell-free samples. Supernatants were aliquoted and then stored at −20℃ before assay for various cytokines/chemokines. Monocyte chemoattractant protein-1 (MCP-1), Gro-α and interleukin 8 (IL-8) secretion into the tissue culture supernatants was detected using sandwich ELISA kits (BioSource International, Burlington, Canada and eBiosciences, San Diego, CA, USA respectively). All assays were performed in triplicate. The concentration of chemokines in the culture medium was quantified by establishing a standard curve with serial dilutions of the recombinant human monocyte chemoattractant protein-1 (MCP-1), Gro-α or IL-8 respectively. Secretion of TNF-α was monitored in rested PMBC and cells exposed to peptide and/or LPS (Pseudomonas aeruginosa) by capture ELISA after 24 h (eBiosciences). For testing anti-endotoxic activities of nisin Z, human PBMC were stimulated with nisin Z (50 µg/ml) in the presence or absence of P. aeruginosa LPS (2 ng/ml). When the combination was used, the cells were pretreated for 45 min with Nisin Z before the addition of P. aeruginosa LPS. Secretion of human TNF-α was monitored by capture ELISA after 24 h of stimulation (eBiosciences).
Cytotoxicity assessment
To assess cell membrane permeabilization by nisin Z, human PBMC (1 × 105) were seeded into 96-well plates (Sarstedt, Newton, NC, USA) and incubated at 37℃ in 5% CO2 overnight. The enzyme lactate dehydrogenase (LDH) assay measuring the release of LDH from human PBMC was then performed after 24 h of incubation with nisin Z (50 µg/ml) in a medium containing 0.01% acetic acid and 0.2% BSA [used for minimum inhibitory concentration (MIC) testing]. Untreated control cells were used as a reference for normalization. All experiments were performed in triplicate.
Flow cytometry
All flow cytometry data was collected on FACSCalibur™ and analyzed using CellQuestPro software (Becton Dickinson). The stimulated cells were fixed in 2% (w/v) paraformaldehyde in PBS at room temperature (23℃) for 15 min, permeabilized in PhosphoPerm Buffer III (BD Biosciences, Mississauga, Canada) at 4℃ for 30 min and washed in 0.5% (w/v) BSA in PBS. The cells were stained for 45 min at room temperature for phospho-p38 (T180/Y182, clone 3D7), phospho-CREB (S133, clone 87G3) and phospho-AKT (S473, clone 193H12), all from Cell Signaling. Following washing, the cells were further stained with goat-anti rabbit IgG-AF647 (Invitrogen-Molecular Probes) at 2 µg/ml and with anti-CD14-phycoerythrin (clone HCD14; BioLegend).
Chemical inhibitor studies
Red blood cell-depleted splenocytes (5 × 105/well) from each mouse were cultured in 96-well, flat-bottom plates in a final volume of 0.1 ml of complete RPMI 1640 medium (RPMI 1640 supplemented with 10% FBS, 2 mM glutamine, 25 mM HEPES, 100 U/ml penicillin and 100 µg/ml streptomycin). Chemical inhibitors were diluted to 100 µM in PBS and added to a corresponding well at a final concentration of 10 µM for 1 h. Following this, nisin Z was added to each well at a final concentration of 100 µg/ml and incubated for 24 h. Murine IL-6 secretion into the tissue culture supernatants was detected using sandwich ELISA kits (eBiosciences). All assays were performed in triplicate. The concentration of murine IL-6 in the culture medium was quantified by establishing a standard curve with serial dilutions of the recombinant murine IL-6.
Phosphorylation Ab arrays and pathway over- representation analysis
Human THP-1 monocytes were treated for 1 h with nisin Z (100 µg/ml) and analyzed using the Human Phospho-Kinase Array kit (R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s instructions. Unstimulated THP-1 monocytes served as a control. In brief, cells were lysed, cleared of cellular debris by centrifugation and quantified for total protein concentrations. Equivalent protein amounts of cleared lysates from both conditions were loaded onto the membranes and the presence of bound phospho-proteins was determined. The signal intensities for all phosphorylation events were determined in triplicate by densitometry using the ImageJ software suite 36 and normalized on the provided positive control. The protein phosphorylation fold-changes were reported based on nisin Z stimulation relative to the unstimulated control. Pathway over-representation analysis was performed using InnateDB (www.innatedb.com) by loading phosphorylation fold-changes and standard errors along with the accompanying protein identifications. Pathways with P-values <0.053 were reported.
Animal infection models
All mouse experiments were conducted in accordance with the Animal Care Ethics Approval and Guidelines of the University of British Columbia. C57BL/6J, CD-1 or Balb/c female mice (from The Jackson Laboratory, Bar Harbor, ME, USA, or Harlan Breeders, Indianapolis, IN, USA respectively) were maintained under specific pathogen-free conditions. The mice were 6–12 wk old and age-matched in all experiments. Each infection model was repeated three times using five mice/group in each experiment. Nisin Z was administered i.p. in sterile saline and the mice infected i.p. with S. aureus (ATCC 25293, 2 × 108 CFU/mouse), E. coli Xen-14 (5 × 107 CFU/mouse; Caliper Life Sciences, Hopkinton, MA, USA) or Salmonella enterica sv. Typhimurium SL1344 (S. Typhimurium, 2 × 104 CFU/mouse). For the S. aureus challenge model, peritoneal lavage was collected from all the mice in 3 ml sterile saline, and the remaining bacterial numbers measured by serial dilution and plating on Mueller-Hinton (MH) or Luria-Bertani (LB) agar plates. Alternatively, for the S. Typhimurium and E. coli challenge models bacterial loads were attained from spleen homogenates and the remaining bacterial numbers measured by serial dilution and plating on MH or LB agar plates. The mice were anesthetized via inhalation of aerosolized isoflurane mixed with oxygen.
Statistical analysis
Prism 4.0 Software (GraphPad, San Diego, CA, USA) was used for statistical data analyses, with a two-tailed Student’s t-test used for comparisons of two datasets and significant results were those calculated as having a P value < 0.05. Two-way ANOVA was performed for the assessment of significant differences in chemokine induction between peptides at the concentrations listed.
Results
Selected Type A (I) lantibiotics induced potent chemokine production ex vivo in human PBMCs
The immunomodulatory activities of microbial biomolecules, natural HDPs and synthetic IDRs can be monitored by their ability to modulate chemokine production.2,3,33 As Type A (I) lantibiotics, like HDPs and IDRs, often have a net positive charge and are secreted naturally from bacteria we postulated that they may represent a novel class of bacterial immunomodulatory molecules. Therefore, a group of purified class A lantibiotics, including nisin Z, pep537 and gallidermin, 37 were investigated.
To screen for immunomodulatory activities we employed chemokine analysis in peptide-stimulated human PBMCs, as this assay is used routinely for the screening of peptide-mediated immunomodulatory activities.38,39 Further, it has also been demonstrated that immunomodulatory peptides are not inhibited by physiological concentrations of cations or serum, which is in contrast to the direct antimicrobial activities of cationic peptides.
20
In chemokine screens all of these cationic lantibiotics were able to induce chemokine production in a concentration-dependent manner in human PBMCs (Figure 1). Importantly, at most concentrations, the ability of the lantibiotics to induce chemokine production was greater than that of the natural human cathelicidin LL-37. Of the tested lantibiotics, nisin Z was the most potent inducer of MCP-1, IL-8 and Gro-α production in PBMCs, and was selected for further characterization (Figure 1). Further, two-way ANOVA demonstrated that the induction of MCP-1, Gro-α and IL-8 by nisin was significantly greater than LL-37 at all concentrations tested. Interestingly, LL-37 induced MCP-1 and IL-8 at 100 µg/ml. Although LL-37 has been reported to be cytotoxic at concentrations as low as 25 µg/ml,
40
it should be noted that this investigation employed epithelial cells as compared to the PBMCs used in our investigation. A previous investigation by Bowdish et al. demonstrated that LL-37 had only partial toxicity at concentrations of 50 µg/ml in human monocytes.
41
Chemokine-inducing activities of the class A lantibiotics gramicidin S, pep5 and nisin Z in human PBMC. Nisin Z was the most potent chemokine inducer of the three lantibiotics and more potent than the human cathelicidin LL-37. The chemokines MCP-1 (A), Gro-α (B) and IL-8 (C) were examined. Chemokine levels were measured by ELISA after 24 h of stimulation. Bars represent the means and SE from 3–6 independent experiments.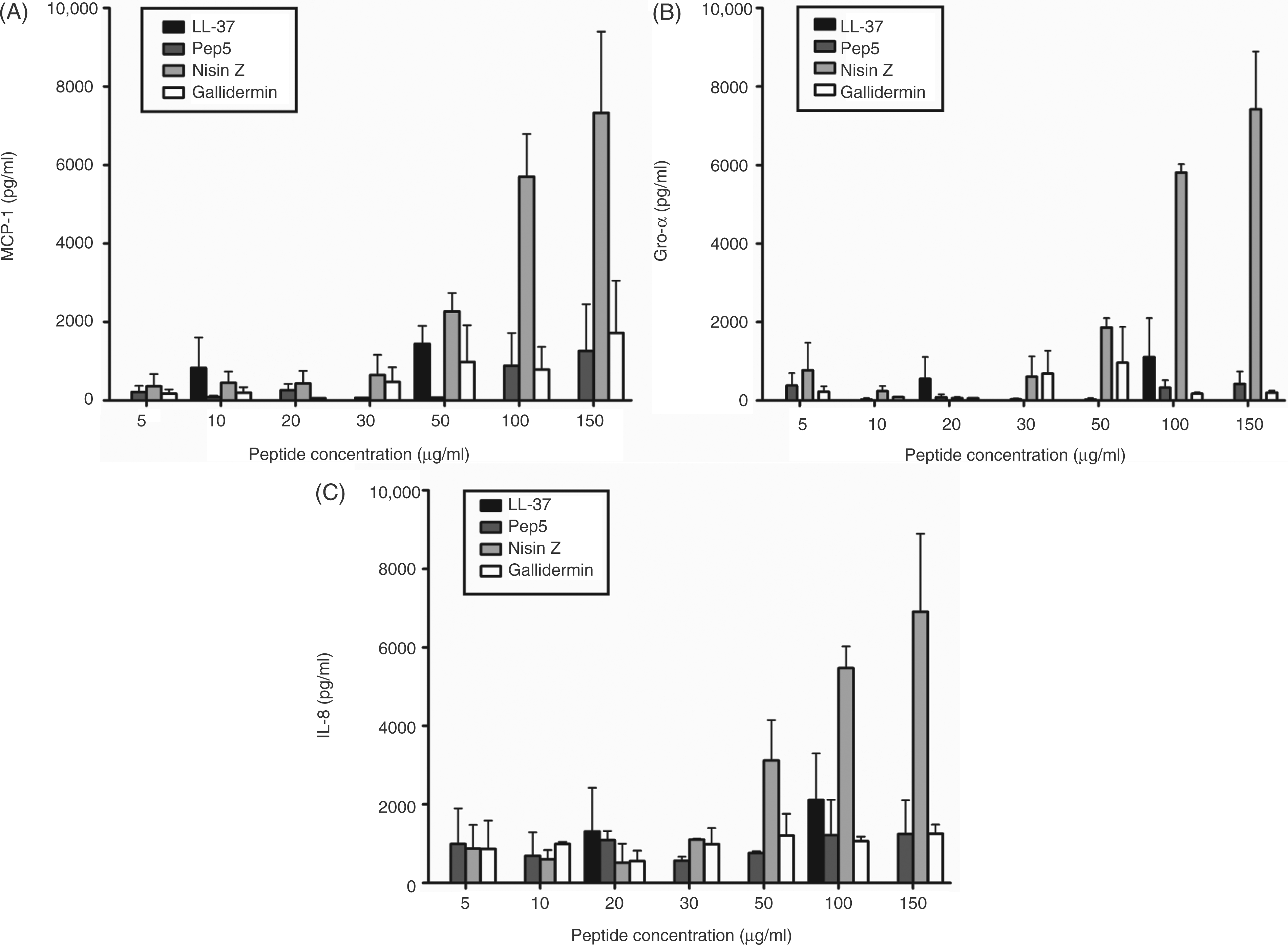
Lack of cytotoxicity of nisin Z in human PBMCs
It was demonstrated previously that nisin has limited cytotoxic activity in a variety of eukaryotic cells, including red blood cells
42
and cervicovaginal epithelial cells.
43
Also, long-term administration of nisin in mouse feed did not result in any significant toxicity or abnormalities.
16
Here, we addressed the cytotoxic activity of nisin Z by monitoring cell membrane permeabilization through the release of LDH following extended incubation with PBMCs. Nisin Z was devoid of cytotoxicity at concentrations of up to 200 µg/ml, which is in agreement with previous observations of negligible cytotoxicity for the peptide (Figure 2)16,42,43 in comparison to the natural host defense peptide LL-37, which demonstrates partial toxicity at concentrations as low as 25–50 µg/ml in human monocytes.
41
Nisin Z did not induce significant LDH release in human PBMC. Human PBMC were stimulated with a range of nisin Z concentrations for 24 h. Following stimulation, supernatants were collected and monitored for the release of LDH. The data represent three independent experiments.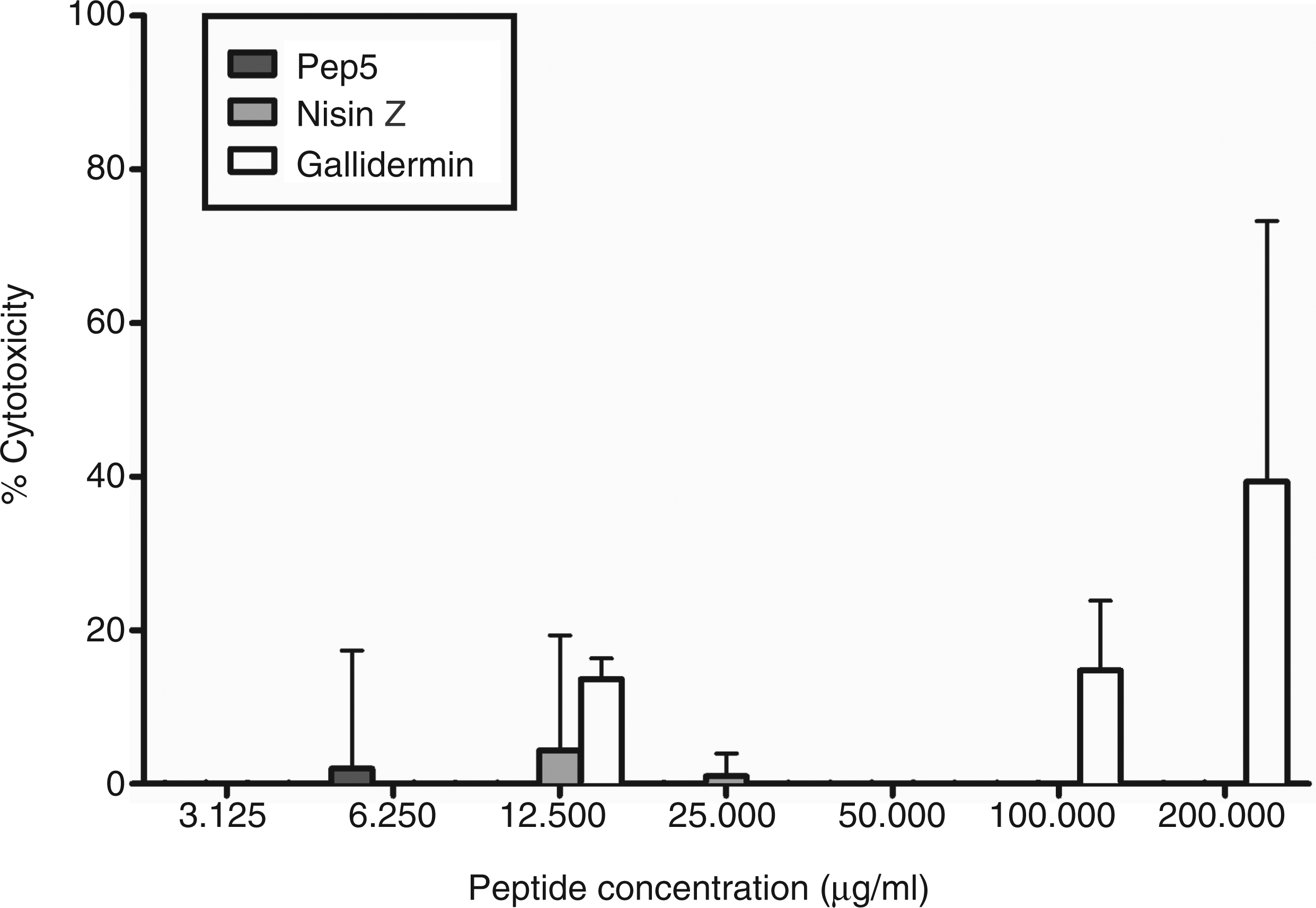
Modulation of pro-inflammatory responses
Many HDPs and synthetic IDRs are able not only to activate immune responses, but also to protect the host by reducing pro-inflammatory responses to microbial stimuli (TLR agonists), such as bacterial LPS. Thus, nisin Z was tested for its ability to dampen pro-inflammatory responses to LPS in human PBMCs. Nisin Z induced minimal or no TNF-α production when administered alone (Figure 3). When human PBMCs were pre-treated for 45 min with 50 µg/ml of nisin Z followed by washing and then subsequent stimulation with 2 ng/ml of LPS, nisin Z was able to repress TNF-α production by 79% when compared to treatment with LPS alone. This indicates that nisin Z was able to dampen pro-inflammatory responses.
Suppression of LPS-induced TNF-α by nisin Z in human PBMC. PBMC were incubated with nisin Z for 45 min before the addition of 2 ng/ml P. aeruginosa LPS or vehicle control. TNF-α secretion was measured after 24 h incubation at 37℃. The results represent mean ± SD of three or more biological replicates. Nisin Z did not induce significant production of TNF-α compared with the control (untreated). Percent reduction is indicated above the bars.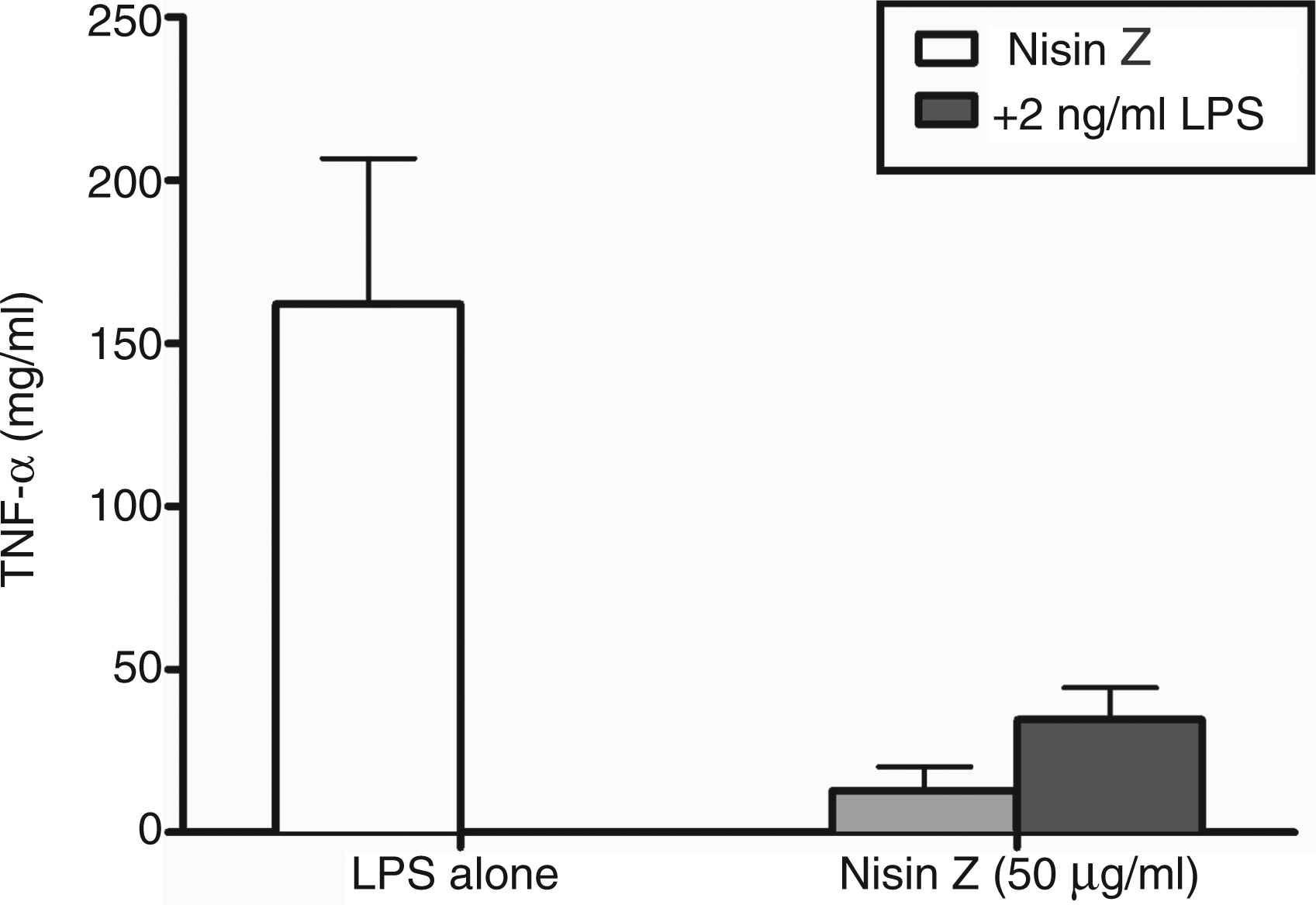
Protection in murine models of bacterial challenge
Investigations of the immunomodulatory properties of PSA in germ-free mice have demonstrated that administration of purified PSA directs immune system development and modulates pro-inflammatory responses. 2 We have also demonstrated that the ability of synthetic IDRs to modulate chemokine production ex vivo in human PBMC correlated with their ability to confer protection in vivo in murine models of bacterial infection. 33 Here, the ability of nisin to confer similar immunomodulatory responses was tested in Gram-positive (S. aureus) and Gram-negative (S. Typhimurium and E. coli) infection models. Recently, we have employed S. aureus and a luminescent strain of E. coli to investigate the ability of IDRs to confer protection against microbial challenge in vivo. 33 Our selection of Gram-negative challenge models for nisin was derived from the observation that nisin has no appreciable in vitro antimicrobial activity against most Gram-negative species (as confirmed here for the nisin used in this study). Thus, protection against microbial challenge in these models would likely indicate modulation of the murine immune response by nisin Z. We postulated that the induction of chemokine production ex vivo by nisin Z would correlate with enhanced protection in murine models of Gram-negative challenge.
In the S. Typhimurium infection model, nisin Z was able to significantly reduce the bacterial load in female BALB/c mice. Mice were pre-treated i.p. with a single administration of 200 µg of nisin Z in sterile saline and infected 4 h later with 1 × 104 CFU S. Typhimurium. Spleens were isolated 24 h post-infection and analyzed for bacterial counts. A single administration of nisin Z resulted in a significant reduction in bacterial counts in the mice (Figure 4A), despite a complete lack of direct antimicrobial activity against Salmonella. Similarly, an E. coli challenge model demonstrated that therapeutic administration of nisin Z, 1 h post-infection, conferred protection with a significant (3.7 log10) reduction in bacterial load (Supplementary Figure 1). In all animal model studies, protection was significant, but modest—as expected for an immunomodulatory therapy.
Protective activity of nisin Z against Gram-positive and Gram-negative bacterial pathogens. Nisin Z conferred significant protection when administered prophylactically in murine models of invasive (A) Salmonella Typhimurium and (B) S. aureus infections. Mice were pretreated i.p. with a single dose of nisin Z (200 µg/mouse) for 4 h and infected i.p. with 2 × 108 CFU/mouse S. aureus or 2 × 104 CFU/mouse S. Typhimurium. All mice were analyzed for bacterial counts in the peritoneal lavage (A) or from spleen homogenates (B) at 24 h post-infection. Bars represent means and SE. Comparisons by t-test. **P < 0.01; ***P < 0.001.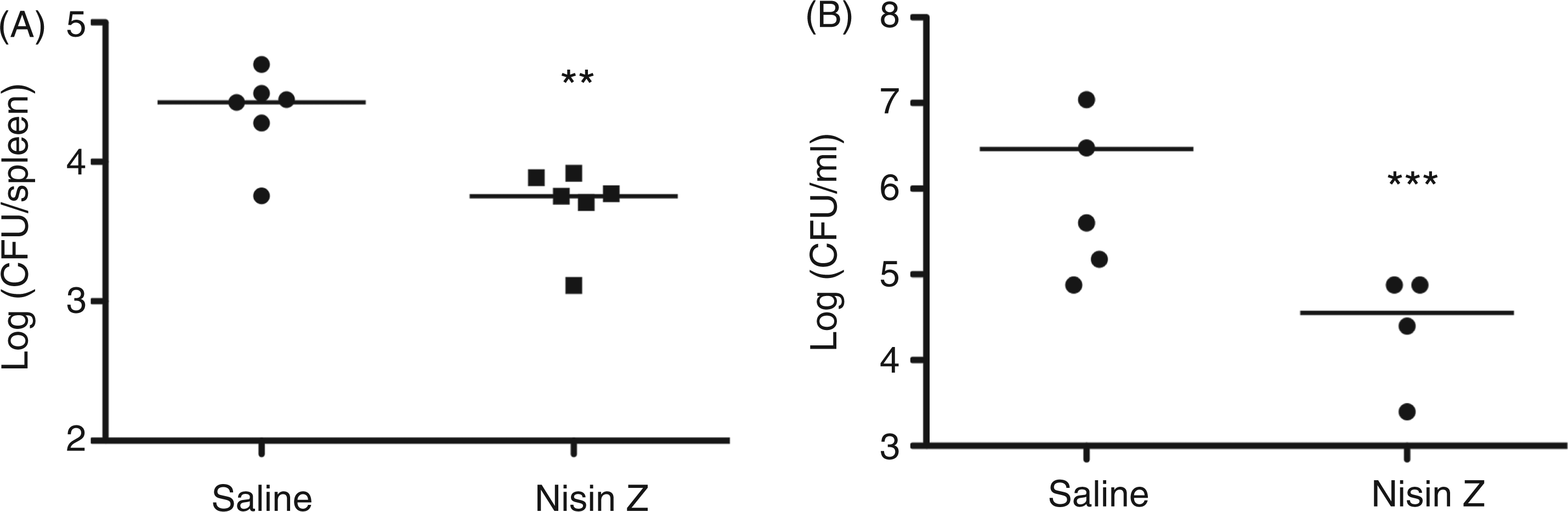
We also employed a Gram-positive murine challenge model using S. aureus. Female CD1 mice were administered 200 µg of nisin Z or saline and infected 4 h later with 2 × 108 CFU of S. aureus. Mice were euthanized 24 h later and the remaining S. aureus were assessed by colony count following peritoneal lavage. As in the Gram-negative challenge models, nisin Z administration resulted in a significant, 100-fold reduction in bacterial load (Figure 4B).
Phosphorylation analysis of nisin Z-mediated cell responses in human monocytes
To gain insight into the mechanisms by which nisin Z modulated host immune responses we employed phosphorylation analysis using flow cytometry and phospho-Ab arrays. Initial assays measured the activation of signaling pathways by nisin Z. Thus, the phosphorylation of p38, AKT and cAMP response element binding (CREB) were assessed in primary human monocytes by flow cytometry (Figure 5). Human PBMCs were stimulated with 100 µg/ml of nisin Z for 45 min and stained for phospho-p38 (T180/Y182), phospho-AKT (S473) and phospho-CREB (S133) following fixation and permeabilization, according to standard protocols. Monocytes were gated as CD14+ cells and represented 5–15% of the PBMC samples. Significant increases in phospho-AKT, phospho-p38 and phospho-CREB were observed in monocytes following stimulation with nisin Z (Figure 5). This suggests that, as for the cationic HDPs, PI3K, p38 MAPK and transcription factor CREB (downstream of AKT and MAPK) were all involved in nisin Z-mediated immunomodulatory responses in human monocytes.
Nisin Z-induced activation of p38 MAPK, AKT and CREB in human primary monocytes. Human blood mononuclear cells were stimulated with 100 µg/ml nisin Z over a 45 min time-course, stained for phospho-p38 T180/Y182, phospho-AKT S473 and phospho-CREB S133, and analyzed by flow cytometry gating on CD14+ monocytes. Percentage of monocytic CD14+ cells within the analyzed PBMC was 5–15% in the samples from different donors. (A) Fold induction in p38 MAPK, AKT and CREB phosphorylation in monocytes over the 45 min time-course. Fold induction in the phosphorylation of p38, AKT and CREB signaling mediators was calculated as a ratio of the mean fluorescence intensity of the nisin Z stimulated versus untreated sample. (B) Histograms of monocytes, unstimulated (grey) or stimulated with 100 µg/ml of nisin Z for 20 min (black), and stained for phosho- p38, AKT and CREB.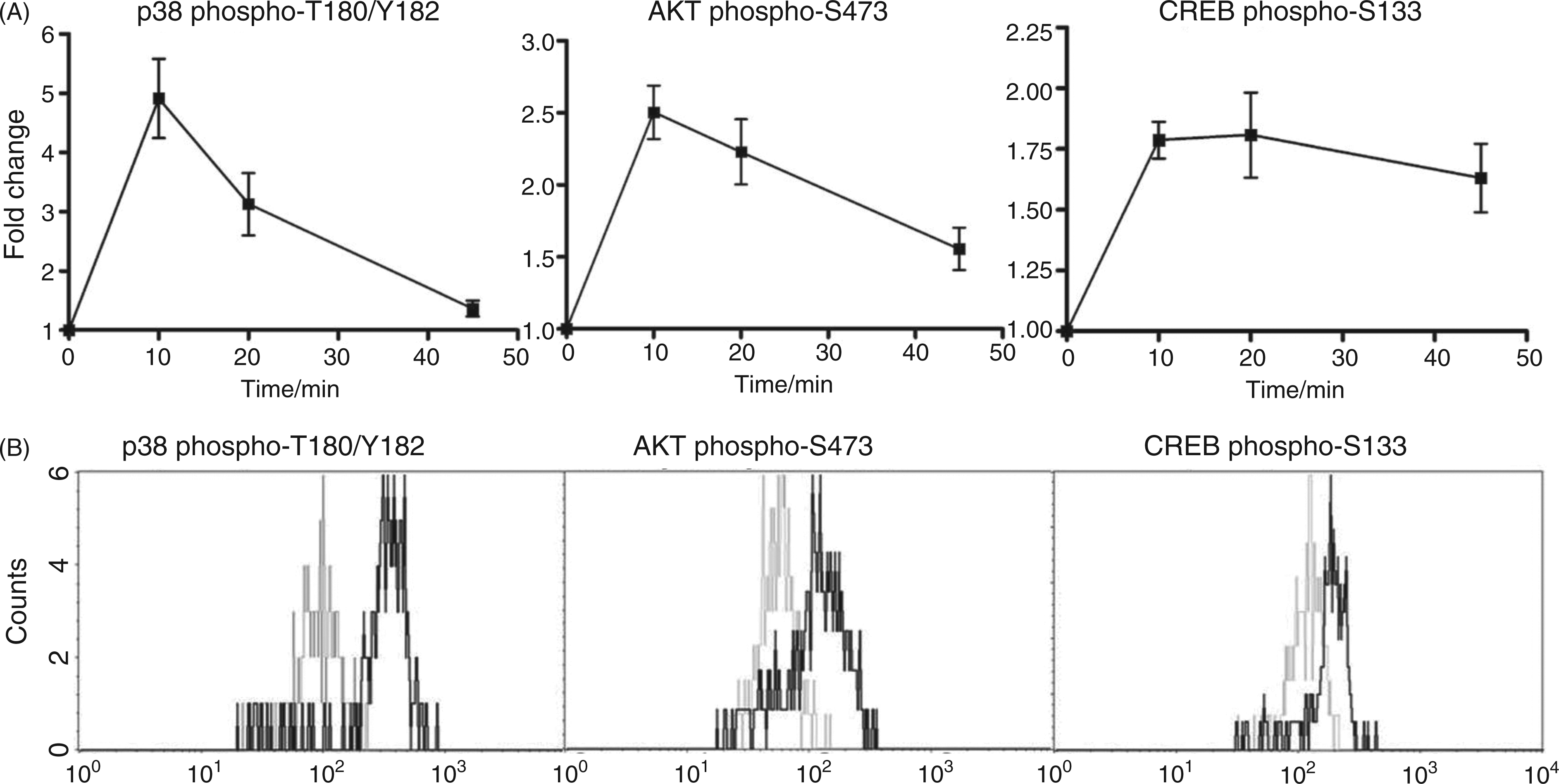
For further insight into the mechanisms through which nisin Z modulated host immunity we employed phospho-kinase Ab arrays and pathway over-representation analysis. Human THP-1 monocytes were stimulated for 1 h in the presence or absence of nisin Z and cell lysates were analyzed for phosphorylation of 46 specific kinase targets (Figure 6). Following image analysis, relative phosphorylation fold-changes were calculated by direct comparison of the nisin Z-stimulated spot densities to the unstimulated condition (Supplementary Table 1). As phosphorylation fold-changes are normally quite modest, we analyzed all proteins on the arrays and their accompanying relative phosphorylation fold-changes by pathway over-representation analysis (ORA) using the innate immune analysis platform InnateDB (Supplementary Table 2).
44
Upregulated phosphorylation events and pathways were largely related p38 signaling, and downstream activation of CREB, NF-κB and activator protein (AP)-1, consistent with the FACS studies. In addition, the pathway ORA suggested that nisin Z stimulation resulted in growth factor receptor responses [platelet derived growth factor receptor (PDGFR) and epidermal growth factor receptor (EGFR)] and in the engagement of integrin signaling. The upregulation of pathways related to focal adhesion kinase (FAK), PI3K and hypoxia-inducible factor 1α further correlated with a likely role for integrin signaling in nisin Z-mediated responses in monocytes.
Relative phosphorylation levels of kinase targets following nisin Z stimulation in human monocytes. Human THP-1 monocytes were stimulated with 100 µg/ml of nisin Z for 1 h. Cells were harvested, lysed and equal amounts of protein lysates were examined for kinase target phosphorylation with phospho-kinase Ab arrays. (A) Representative kinase activation from the analysis of 46 phosphorylated kinases in nisin Z-stimulated or non-stimulated THP-1 cells. (B) Comparison of overall pixel densities between both conditions. Each phospho-kinase Ab is spotted in duplicate. Sample layout is numbered in (A) and spot identities are listed in Supplementary Table 3.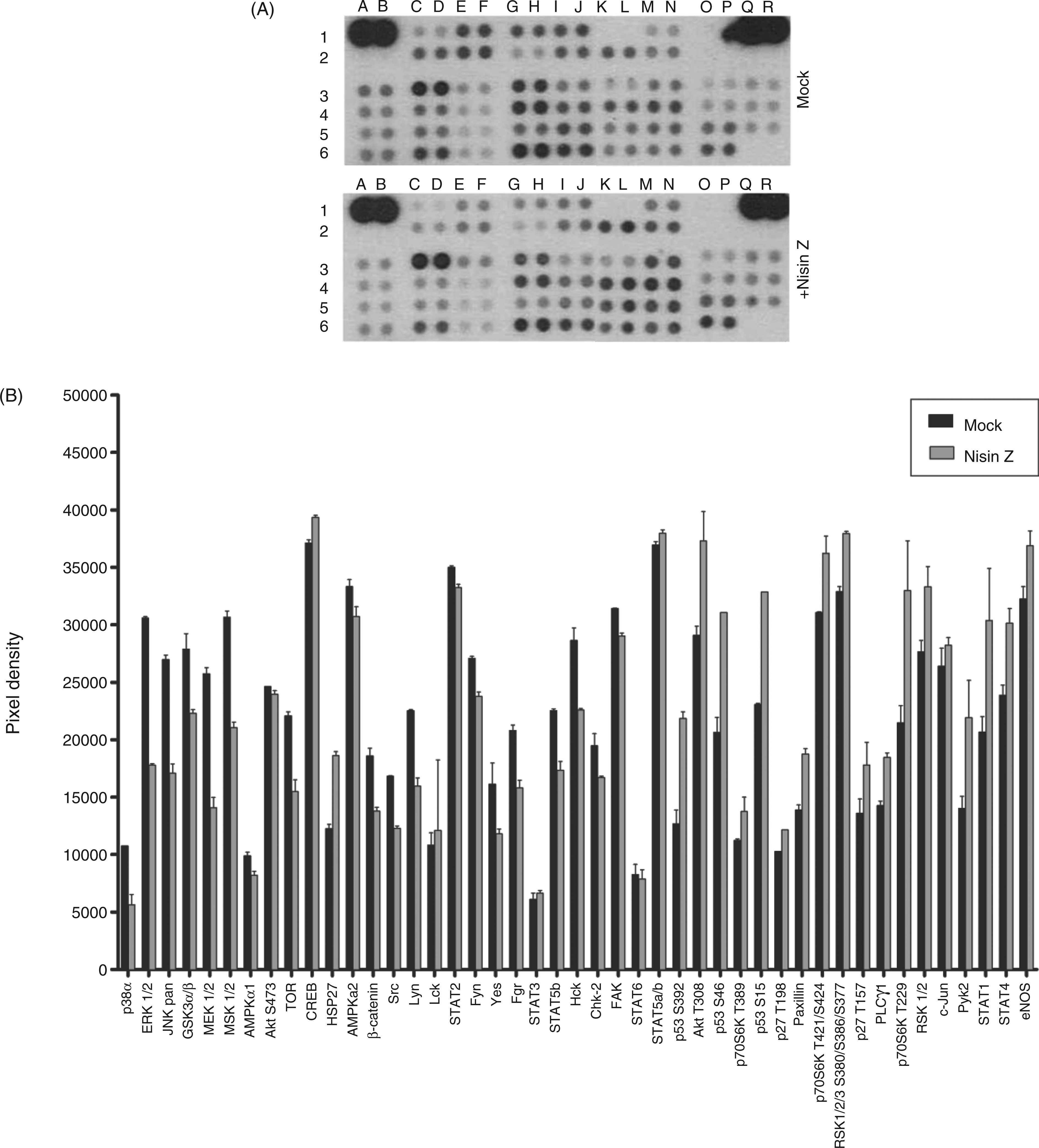
Role of growth factor receptors, ERK/MAPK, PKA and PKC signaling in nisin Z immunomodulatory activities
Based on the cues provided by the phosphorylation analyses, we employed chemical inhibitors against signaling pathway intermediates and cellular receptors to gain further insight into the molecular mechanisms through which nisin Z modulated immune responses. Murine splenocytes were pre-treated with various specific chemical inhibitors for 1 h followed by stimulation with 100 µg/ml of nisin Z for 18 h. Previously, we demonstrated that IDR peptides induced the secretion of IL-6 when administered to mice i.p.
33
The ability of the various inhibitors to repress nisin Z-mediated IL-6 production was assessed by ELISA (Figure 7). Control experiments revealed that none of the specific inhibitors utilized here was cytotoxic at the concentrations utilized
33
and DMSO vehicle controls were also included for all experiments at levels identical to those used as solvents for the inhibitors.
Signaling pathways mediating nisin Z induction of MCP-1 in murine splenocytes. Murine splenocytes were pretreated for 1 h with a broad range of kinase inhibitors targeting receptor tyrosine kinases, ERK/MAPK, PKA or PKC (10 µM) prior to stimulation with nisin Z at 100 µg/ml. Supernatants were collected after 24 h and IL-6 secretion was assessed by ELISA. All bars represent means and SE from three independent biological replicates.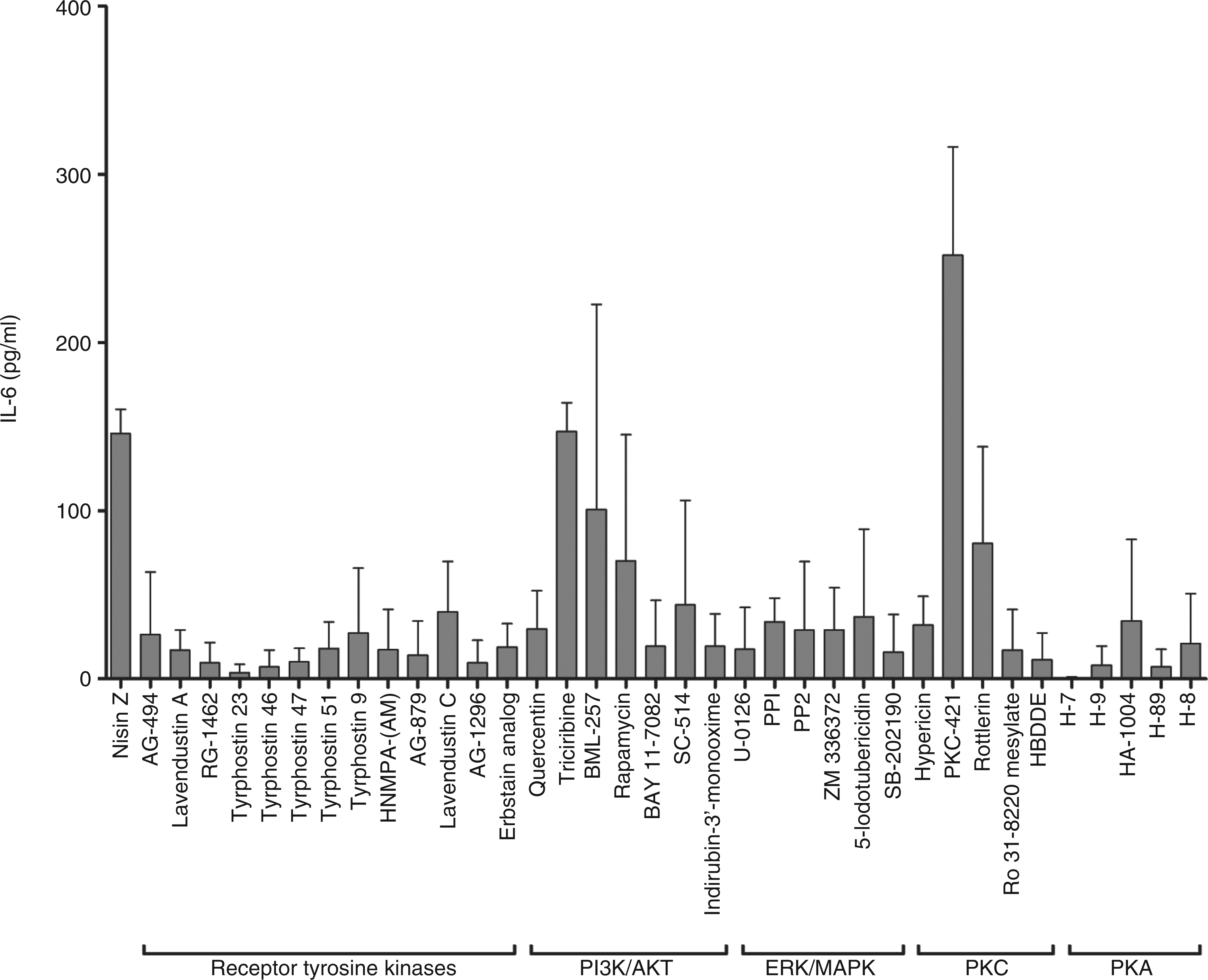
Previous investigations have demonstrated that HDPs mediate activation of diverse cellular responses, including cell migration, cytokine induction and angiogenesis, through a variety of cell signaling pathways, including MAPK.45–47 Thus, we investigated the role of these pathways in nisin Z immunomodulatory responses. Inhibition of nisin Z-mediated IL-6 secretion was effected by the inhibition of MAPK signaling and specific receptor tyrosine kinases (Figure 7). For example, inhibition of multiple elements of ERK/MAPK signaling significantly inhibited IL-6 secretion in nisin Z-stimulated splenocytes. Inhibition of p38 (SB-202190), MEK (U-0126), c-Raf (ZM 336372) and the Src family kinases (PP1 and PP2) all significantly inhibited IL-6 induction by nisin Z. This suggested a strong correlation between ERK/MAPK signaling and the induction of immunomodulatory responses by nisin Z.
LL-37-mediated activation of endothelial cells is dependent on PKC. 46 Ro 31-8220 mesylate, a PKC inhibitor with weak inhibitory activity against PKA, and 2,2′,3,3′,4,4′-hexahydroxy-1,1′-biphenyl-6,6′-dimethanoldimethylether (HBDDE), a selective inhibitor of PKCα and PKCγ, significantly inhibited IL-6 secretion following nisin Z stimulation. Inhibition of PKA also significantly inhibited the induction of IL-6 by nisin Z. Pre-treatment of splenocytes with inhibitors H-7, H-9, HA-1004, H89 and H-8 all had significant inhibitory effects on IL-6 induction. Although H-89 is broadly inhibitory to multiple cellular kinases, 48 the increased specificity of H-7, H-9, HA-1004 and H-8 for PKA indicated the involvement of PKA activation in nisin Z stimulation of IL-6 secretion.
In addition, inhibition of the IκB kinase (IKK)/NF-κB signaling pathway (Bay 11-7082; SC-514), glycogen synthase kinase (GSK)-3β and the cyclic dependent kinases 1 (CDK1) and CDK5 (indirubin-3′-monoxime) all resulted in significant inhibition of nisin Z-mediated IL-6 secretion.
We also examined the role of growth factor receptors based on the phosphorylation array data and the demonstration that keratinocyte migration and chemokine induction results from EGFR transactivation by the human cathelicidin LL-37.26,49 Multiple inhibitors of EGFR kinase activity (AG-494; RG-1462; Erbstatin analog; Lavendustin C; Tyrphostins 23, 46, 47, 51) and PDGFR kinase activity (Tyrphostin 46; AG-1296) resulted in significant inhibition of IL-6 secretion in nisin Z-stimulated cells. Furthermore, inhibition of the nerve growth factor receptor (NGFR) kinase (AG-879) and insulin receptor (IR) kinase [hydroxy-2-naphthalenylmethylphosphonic acid trisacetoxymethyl ester; HNMPA-(AM)] also inhibited IL-6 release in nisin Z-stimulated splenocytes. Thus, nisin Z appears to engage multiple growth factor receptors in the modulation of immune responses.
Taken together, these results indicate that the immunomodulatory activities of nisin Z and, in particular, induction of IL-6 secretion involve the specific engagement of the growth factor receptors, activation of NF-κB, PKC and PKA, and MAPK signaling.
Discussion
Recently, there has been an increasing appreciation of the role of symbiotic bacteria in the development and maturation of the mammalian immune system. Indeed, it has been demonstrated that microbial biomolecules, such as PSA and EPS, are beneficial to the development of host immunity.2,3,50,51 Here, we demonstrated that nisin Z, a lantibiotic from the non-pathogenic probiotic bacterium L. lactis also beneficially modulates host immunity. Purified nisin Z modulated host immune responses both ex vivo and in vivo. Nisin Z was selected from a panel of class A lantibiotic peptides owing to its superior ability to induce chemokine production in human PBMC. Interestingly, the extent of induction of the chemokine MCP-1 by nisin Z was similar to that reported for the optimized peptide IDR-1002. 33 Although previous investigations have demonstrated that the ingestion of lactic acid bacteria could modulate cytokine production in humans, 16 our study provides direct evidence for the modulation of chemokine responses by nisin Z. As nisin Z is conceptually analogous to natural HDPs, being an amphipathic peptide containing cationic and hydrophobic residues, we considered the possibility that many of the immunomodulatory properties associated with HDPs would be conserved in this lantibiotic. Consistent with this, nisin Z administration also inhibited LPS-induced pro-inflammatory responses, suggesting that nisin Z demonstrated selective immunomodulatory activity. This was in agreement with the recent demonstration that the cationic bacteriocin CBT-SL5 from Enterococcus faecalis was able to suppress IL-8 mRNA expression and production in response to Propionibacterium acnes in human keratinocytes. 52 Importantly, nisin Z was devoid of cytotoxicity within the concentration ranges required for inducing chemokine synthesis ex vivo, which is in agreement with previous studies. 42
The ability of nisin Z to modulate host immunity was confirmed in vivo as the administration of purified nisin Z resulted in the significant protection of mice from Gram-negative challenges, despite the fact that these organisms are completely resistant to the antibiotic action of nisin. 11 A previous investigation by de Pablo et al. 16 demonstrated that nisin (as nisaplin, a nisin preparation containing 2.5% nisin, 77.5% NaCl and non-fat dried milk 11 ) was able to modulate the adaptive immune response of mice when incorporated into their feed. The authors demonstrated increased CD4+ and CD8+ T-lymphocytes, and a concomitant decrease to B-lymphocytes. Although this previous study examined the adaptive response, it is consistent with the observations made here, as it is well established that adjuvants that stimulate innate immunity are able to promote adaptive responses, and that HDP and IDR peptides have adjuvant activity.39,53 In a subsequent investigation, Puertollano et al. 51 demonstrated that the incorporation of nisin into mouse feed did not affect the levels of pro-inflammatory cytokines TNF-α or IFN-γ, 54 consistent with the observation that such peptides do not induce pro-inflammatory cytokines,6,9 as confirmed here for TNF-α. Thus, these results lend support for a more widespread investigation of the role of lantibiotics and their host organisms in the development and maintenance of host immunity.
We demonstrated here that the immunomodulatory activities of nisin Z are similar to those described recently for many HDPs and IDRs. Therefore, we postulated that the mechanism(s) of action employed by these phylogenetically-diverse immunomodulatory molecules might overlap. The results obtained here indicate that despite profound compositional and structural differences of the lantibiotic nisin from other HDP and IDR peptides, they are both amphipathic polycations 55 and, perhaps because of this, they have many mechanistic features in common. The lanthionine bridges of nisin are required to maintain its amphipathic configuration and it would be interesting in future studies to determine if they were required for its immunomodulatory function. Recent investigations of HDP and IDR immunomodulatory activity have identified several signaling pathways that are involved in mediating peptide-induced immunomodulatory responses.28,32,33,56 We demonstrated here that nisin Z-mediated induction of IL-6 involved the engagement of multiple growth factor receptors, and activation of ERK and p38 MAPK, Src family kinases, and NF-κB, Akt/CREB, PKC and PKA signaling. These results are in agreement with previous investigations of LL-37 which demonstrated the involvement of growth factor receptor engagement26,47 and many of these pathways.46,47,56,57 It should also be noted that a recent investigation by Begde et al. demonstrated that nisin is also able to activate neutrophils, 17 and provides evidence that the immunomodulatory activities of nisin are multi-factorial and employ multiple subsets of host immune cells.
A variety of receptors appear to be involved in the activity of HDP and IDR peptides. Although there is no significant sequence and structural homology between various HDPs, IDRs and nisin Z, it is worth mentioning that other receptors of innate immunity, such as the TLRs, recognize a broad range of structurally and chemically diverse ligands to promote activation of innate immune responses. Thus, similar redundancies likely exist within the innate immune system for the recognition of structurally dissimilar immunomodulatory peptides of diverse amino acid sequences but general cationic amphipathic nature. Ultimately, this provides the opportunity for the initiation of the innate immune responses through the recognition of a broad-range of host or microbe-derived peptides. Phosphorylation analysis of intracellular kinase targets provides a mechanism for defining global host responses to stimulation or insult. In agreement with our chemical inhibitor studies and flow cytometry data, nisin Z stimulation of human monocytes appeared to upregulate multiple pathways related to growth factor receptors, and activation of p38 MAPK, CREB, NFκB and AP-1, consistent with other studies on human HDP LL-37. 56 Interestingly, the pathway ORA also suggested a role for the activation of integrin-related signaling events following nisin Z stimulation. As previous investigations have demonstrated that HDPs can increase integrin expression on host cells 58 and LL-37 has been demonstrated to activate angiogenesis directly, 43 our results suggest that integrin-mediated signaling events may represent a broad immunomodulatory response to both HDPs and bacterial antimicrobial peptides. Indeed, we demonstrated recently that integrin activation and signaling is involved in the mediation of chemotactic responses by IDR-1002. 59
In summary, we have demonstrated directly that the lantibiotic nisin Z is a multi-functional immunomodulatory peptide. We hypothesize that the lantibiotics are, indeed, a general player in the interactions of bacteria and their hosts, modulating innate immune responses to provide a selective advantage. Intriguingly, nisin Z has similar in vitro and in vivo immunomodulatory and anti-infective activities to those described previously for HDPs and the synthetic IDRs, and suggest a possible role for nisin Z as a novel immunomodulatory therapeutic. Correspondingly, nisin Z appears to employ similar growth factor receptors and signaling pathways as many HDPs demonstrate at least partial conservation of mechanism of action amongst these structurally diverse peptides. Our results indicate that bacteria can modulate host immunity through their secreted bacteriocins and suggest that lactic acid bacteria may play a more general role in the homeostasis and maintenance of host gastrointestinal immunity.
Footnotes
Funding
We gratefully acknowledge financial support from the Canadian Institutes for Health Research to REWH. REWH was the recipient of a Canada Research Chair. JK was supported by a fellowship from the Canadian Cystic Fibrosis Foundation. HGS acknowledges support by the BONFOR program of the University of Bonn.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
