Abstract
The NO productivity of auditory cells in response to LPS was examined by using conditionally immortalized murine HEI-OC1 auditory cells. HEI-OC1 cells produced NO in response to LPS ranging from 0.1 µg/ml to 100 µg/ml in a concentration-dependent manner. LPS at 100 µg/ml exhibited no cytotoxic action against HEI-OC1 cells and led to the highest level of NO production. The NO output in LPS-treated HEI-OC1 cells gradually increased up to 72 h. LPS-induced NO production was mediated by the expression of an inducible NO synthase (iNOS) protein. TLR4 and CD14 was expressed on the cell surface of HEI-OC1 cells. LPS augmented the production of IFN-β in the MyD88-independent pathway of LPS signalling. HEI-OC1 cells produced NO in response to a TLR2 ligand but not TLR3 ligand. LPS was suggested to lead to NO production in auditory cells via iNOS expression. The immunological significance of NO production in auditory cells is discussed.
Introduction
Auditory hair cells are the sensory receptors of both the auditory system and the vestibular system in mammals. The auditory hair cells are located within the organ of Corti on a thin basilar membrane in the cochlea of the inner ear. Damage to these auditory cells results in decreased hearing sensitivity, i.e. sensorineural hearing loss. The chemotherapeutic agents in the treatment of a variety of solid tumours cause ototoxicity. The ototoxicity has been extensively studied by using the combination of cisplatin—a widely used chemotherapeutic agent—and the conditionally immortalized murine HEI-OC1 auditory cells.1–4 Production of pro-inflammatory cytokines, such as TNF-α, IL-1β and IL-6, in HEI-OC1 cells plays a critical role in cisplatin-induced auditory damage.4–7 Interestingly, cisplatin is reported to increase the expression of TLR4 as a LPS receptor in HEI-OC1 cells. 4 The augmented TLR4 expression leads to LPS-induced production of a series of pro-inflammatory cytokines, such as TNF-α, IL-1β and IL-6, and then affects the cisplatin-induced ototoxicity. 4 Thus, auditory cells have been studied as the target cells for chemotherapy-related ototoxicity. However, the LPS-induced cytokine production in auditory cells might be involved in immune response and host defence in the inner ear. The immunological significance of auditory cells in the inner ear is not fully elucidated. NO is also a key molecule in innate immunity, playing a role in a variety of immunological processes.8–10 In the present study, we studied the NO productivity of auditory cells in response to LPS by using a conditionally immortalized murine auditory cell line, HEI-OC1. Here, we report that HEI-OC1 auditory cells produce NO in response to LPS via an inducible NO synthase (iNOS).
Materials and methods
Reagents
LPS from Escherichia coli O55:B5 was purchased from Sigma Chemicals (St Louis, MO, USA). Rabbit anti-iNOS Ab was purchased from Upstate Inc. (Lake Placid, NY, USA). Pam3CSK4 and poly I:C were obtained from Calbiochem (San Diego, CA, USA) and Invivogen (San Diego, CA, USA) respectively.
Cell culture
The murine auditory cell line HEI-OC1 was established from long-term cultures of Immortomouse cochlea and used as conditionally immortalized auditory cells. 11 HEI-OC1 cells were maintained in DMEM containing 10% heat-inactivated FCS (GIBCO-BRL, Gaithersburg, MD, USA) and antibiotics at 33°C under 5% CO2.
Cell viability
HEI-OC1 cells were seeded in a 96-well plastic plate and allowed to grow for 12 h. The cells were treated with various concentrations of LPS. The cell viability was determined by the reduction of 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) (Promega, Madison, WI, USA).
Determination of nitrite concentration
The concentration of nitrite—the end product of NO metabolism—was determined with the Griess reagent, as described elsewhere.12,13 Briefly, HEI-OC1 cells were seeded in a 96-well plastic plate and then stimulated with LPS. To measure the nitrite concentration, 50 µl of culture supernatant was mixed with equal volume of Griess reagent. After 2 min incubation at room temperature, the nitrite concentration in the culture supernatant was measured at A570 nm with reference to the standard curve using sodium nitrite.
Determination of IFN-β
HEI-OC1 cells were stimulated with LPS for 24 h and then concentrations of IFN-β in the culture supernatant were determined by an ELISA kit (PBL Interferon Source, Piscataway, NJ, USA).
Immunoblotting
The immunoblotting method was as described previously.13,14 HEI-OC1 cells were cultured with LPS (100 µg/ml) for various time periods. The cell lysates were extracted by the lysis buffer containing 0.5 M Tris-HCL, 4% sodium dodecylsulfate and 2-mercaptoethanol, and boiled at 80°C for 5 min. The protein concentration of the samples was determined by the bicinchoninic acid protein assay reagent (Pierce, Rockford, IL, USA). An equal amount of protein (20 µg) was analysed by SDS-PAGE under reducing conditions and transferred to a membrane filter. The membranes were treated with an appropriately diluted anti-iNOS Ab for overnight. The immune complexes were detected with a 1:2000 dilution of HRP-conjugated anti-rabbit IgG Ab for 1 h and the bands were visualized with a chemiluminesent reagent (Pierce). Chemiluminescence was detected by a light capture system analyser AE6955 (Atto, Tokyo, Japan). The molecular masses of the Ags were determined by comparison with a pre-stained protein size marker kit (Invitrogen, Carlsbad, CA, USA).
Immunofluorescence staining and laser flowcytometric analysis
HEI-OC1 cells and RAW 264.7 macrophage-like cells were incubated with phycoerythrin (PE)-conjugated Ab to TLR4 (Biolegend, San Diego, CA, USA) or CD14 (PharMingen, San Diego, CA, USA) for 30 min on ice. An isotype control Ab was also used. The stained cells were analysed with a laser flowcytometer (FACS Calibur; Becton Dickinson, Palo Alto, CA, USA). The fluorescence intensity is expressed in a log scale.
Statistical analysis
Experimental values are represented as the mean of triplicates ± SD from at least three independent experiments. The significance of differences was determined by the two-way analysis of variance. A value of P < 0.05 was considered statistically significant.
Results
Induction of NO production in HEI-OC1 cells by LPS
The NO productivity of HEI-OC1 cells in response to LPS was examined. HEI-OC1 cells were incubated with various concentrations of LPS for 48 h and the concentration of nitrite in the culture supernatant was determined (Figure 1A). LPS at 0.1 µg/ml significantly induced NO production (P < 0.01). LPS at 100 µg/ml led to the highest level of NO production in HEI-OC1 cells. Next, the time course of LPS-induced NO production was examined (Figure 1B). The cells were incubated with LPS at 10 µg/ml or 100 µg/ml for various hours. LPS at 10 µg/ml gradually augmented NO production up to 72 h. There was no significant difference in NO output at 24 h between the cells stimulated with LPS at 10 µg/ml and those stimulated with LPS at 100 µg/ml. LPS at 100 µg/ml led to a higher NO output at 48 and 72 h than LPS at 10 µg/ml did. In addition, we confirmed that HEI-OC1 cells produced TNF-α in response to LPS at 100 µg/ml (data not shown), as reported by other groups.4–7
Induction of NO production in HEI-OC1 cells by LPS. (A) HEI-OC1 cells were incubated with various concentrations of LPS for 48 h. *P < 0.01,**P < 0.001 vs untreated control. (B) HEI-OC1 cells were incubated with LPS at 10 µg/ml or 100 µg/ml for various hours. The concentration of nitrite in the culture supernatant was determined. *P < 0.01, **P < 0.001 vs 0 h.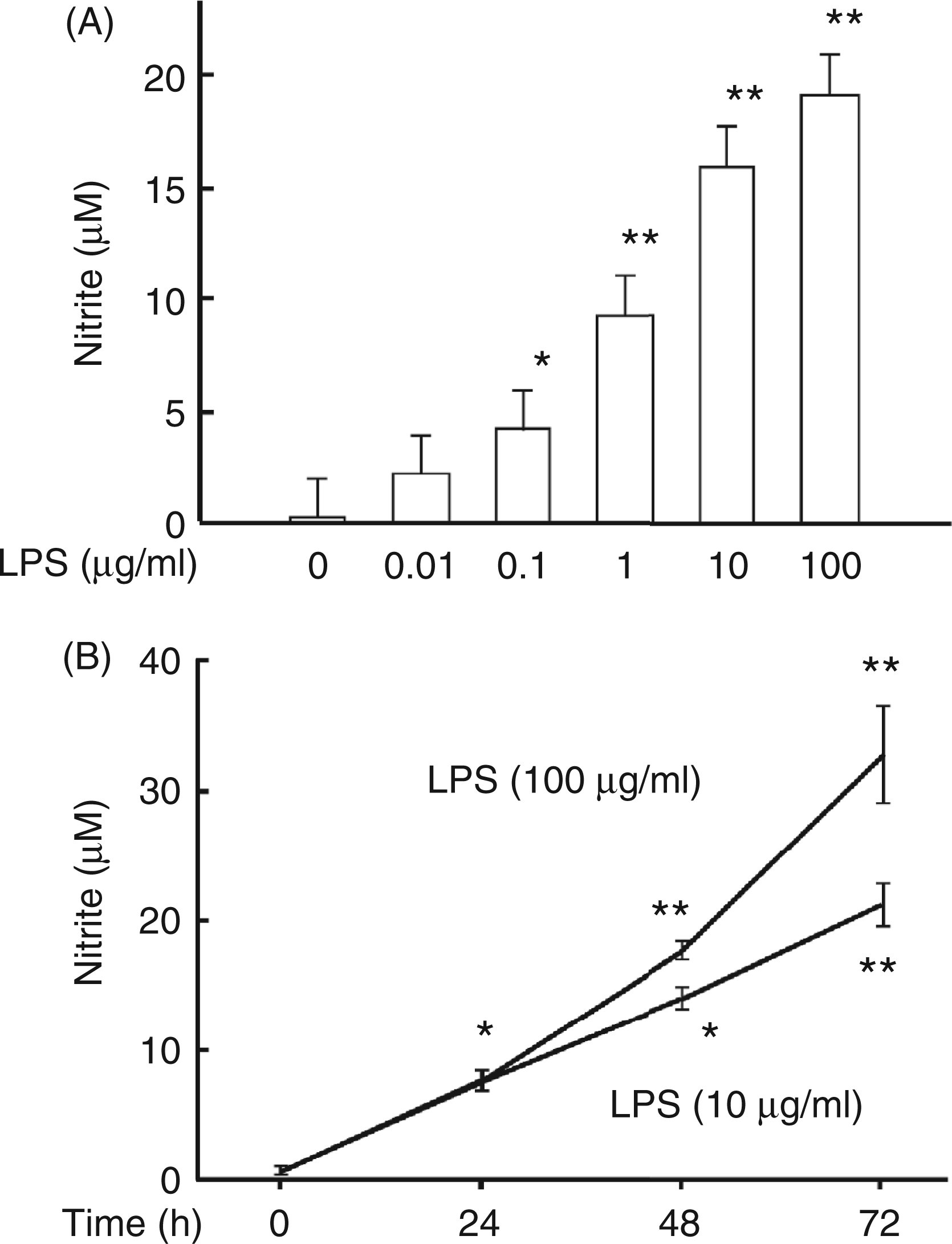
A cytotoxic action of LPS against HEI-OC1 cells was examined with a MTT assay. LPS at 100 µg/ml exhibited no cytotoxic action against the cells (data not shown). There was no significant morphological change between untreated and LPS-treated HEI-OC1 cells.
Induction of iNOS protein expression by LPS
A high amount of NO is produced by iNOS.15,16 Therefore, the effect of LPS on the expression of iNOS protein was examined in order to elucidate a high NO output in LPS-stimulated HEI-OC1 cells. HEI-OC1 cells were incubated with LPS at 10 µg/ml or 100 µg/ml for 48 h. Immunoblotting analysis demonstrated that the iNOS protein band was detected in LPS-treated cells but not in untreated cells (Figure 2). LPS at 100 µg/ml more markedly induced the iNOS expression than LPS at 10 µg/ml. A high NO output in LPS-stimulated HEI-OC1 cells was suggested to be mediated by the expression of iNOS protein.
Induction of iNOS protein expression by LPS. HEI-OC1 cells were incubated with LPS at 10 µg/ml or 100 µg/ml for 48 h. iNOS protein expression was determined by immunoblotting with anti-iNOS Ab. A typical result of three independent experiments is shown.
Cell surface expression of TLR4 and CD14 as LPS-related receptors
LPS is recognized by a series of LPS-related receptors, including TLR4 and CD14.17,18 The expression of TLR4 or CD14 on the cell surface of HEI-OC1 cells was examined in order to elucidate the LPS responsiveness of HEI-OC1 cells. HEI-OC1 cells and RAW 264.7 macrophage-like cells as a positive control were stained by PE-conjugated Ab to TLR4 or CD14. FACS analysis demonstrated that both TLR4 and CD14 were significantly expressed on the cell surface of the cells. The fluorescence intensity of RAW 264.7 cells shifted more compared with that of HEI-OC1 cells, suggesting a higher level of TLR4 and CD14 expression (Figure 3).
Cell surface expression of TLR4 and CD14 on HEI-O1 cells. HEI-OC1 cells and RAW 264.7 cells were stained with PE-conjugated anti-TLR4 or CD14 Ab. The fluorescent intensity is expressed on a log scale. A typical result of three independent experiments is shown.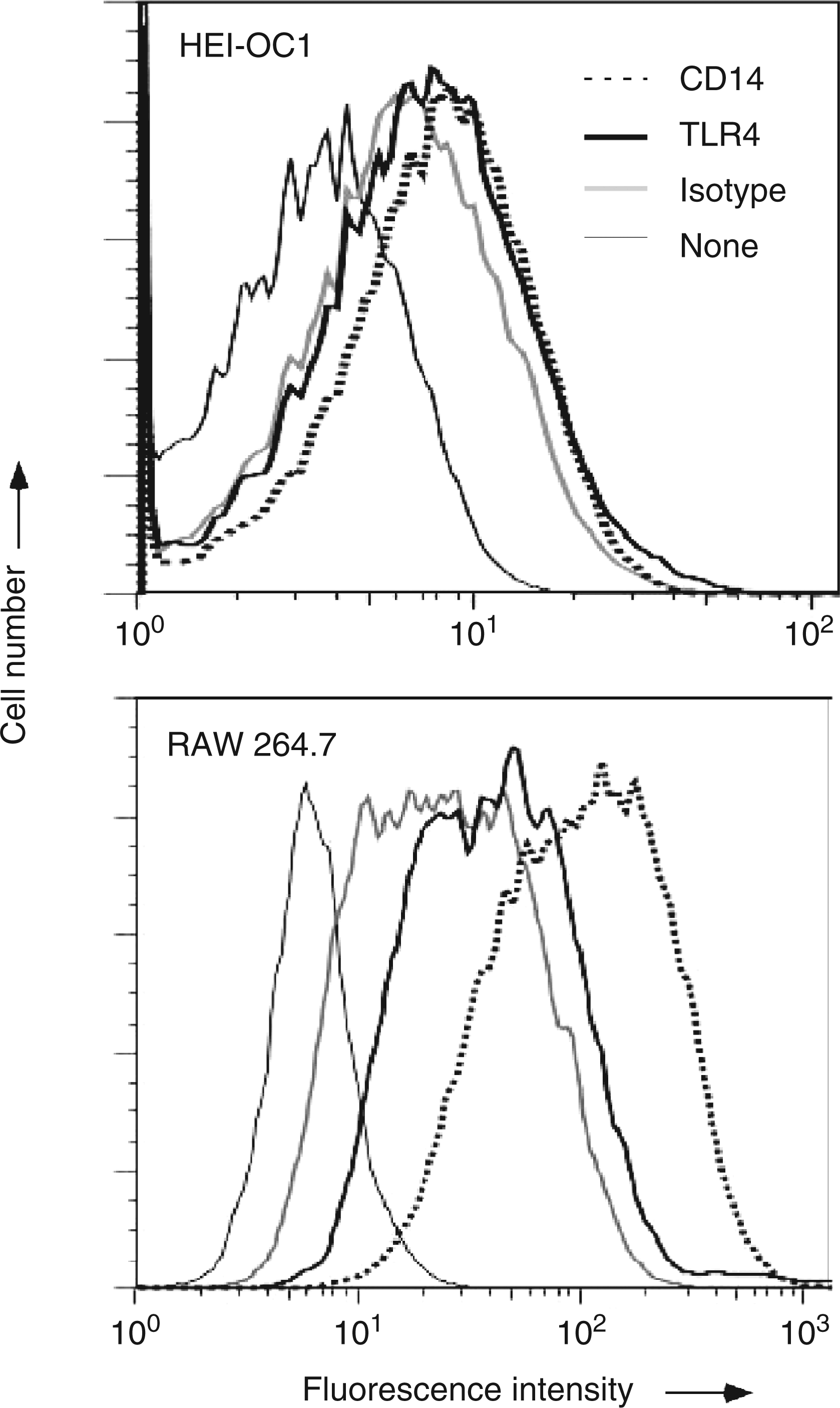
Induction of IFN-β production by LPS
LPS-induced NO production is triggered by the MyD88-dependent and independent pathways of LPS signalling.19–23 In particular, it is predominantly mediated with the MyD88-independent pathway, which is characterized by IFN-β production.20–23 In order to confirm a possibility that the MyD88-independent pathway was involved in LPS-induced NO production in HEI-OC1 cells, the effect of LPS on the production of IFN-β in HEI-OC1 cells was examined. HEI-OC1 cells were incubated with LPS at 10 µg/ml or 100 µg/ml for 6 h. LPS significantly induced the production of IFN-β (Figure 4). LPS at 100 µg/ml augmented the IFN-β production more markedly than LPS at 10 µg/ml.
Induction of IFN-β by LPS. HEI-OC1 cells were incubated with LPS at 10 µg/ml or 100 µg/ml for 6 h. The concentration of IFN-β in the culture supernatant was determined with an ELISA. *P < 0.05 vs untreated control. **P < 0.01 vs untreated control or LPS at 10 µg/ml.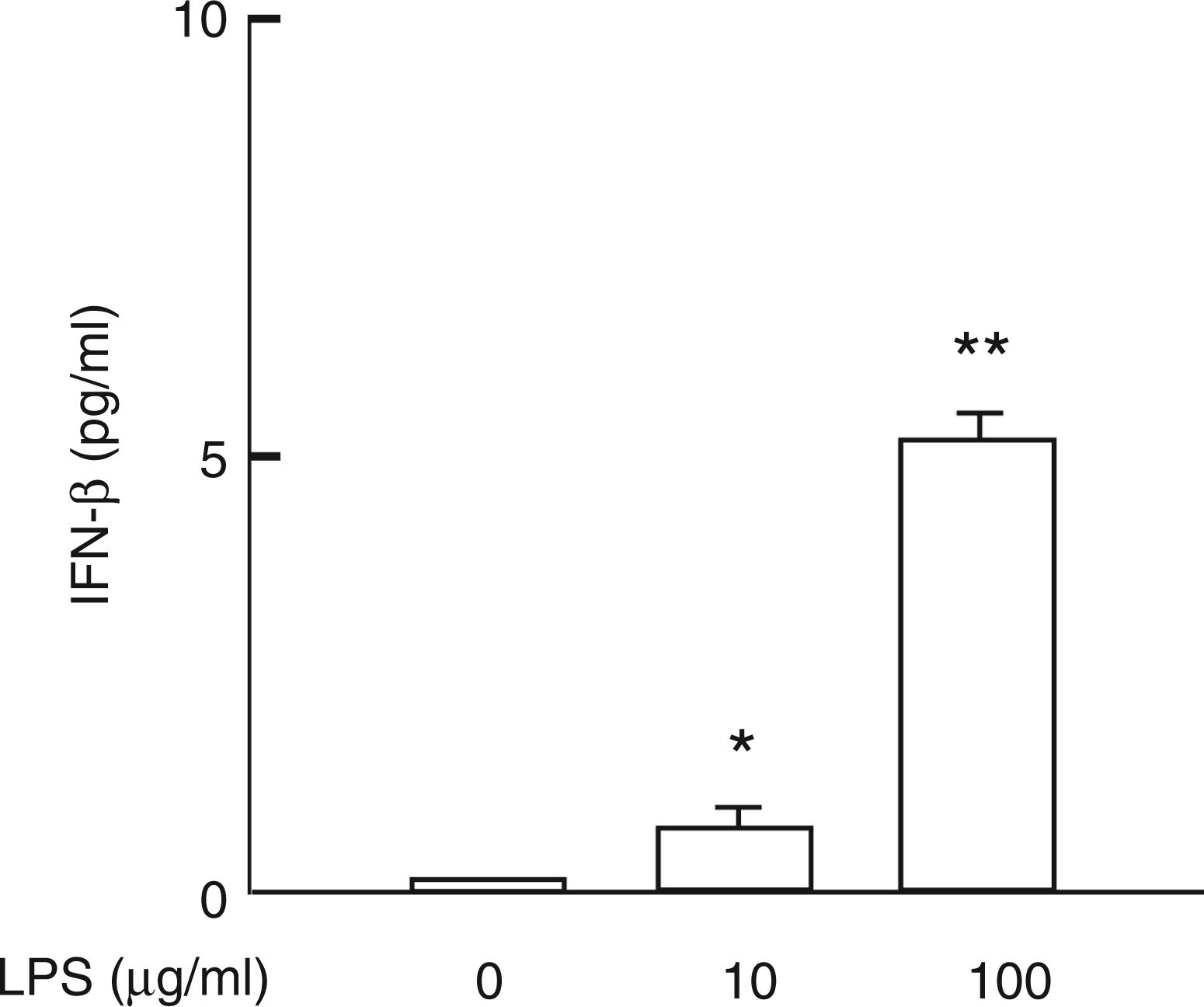
Induction of NO production by TLR2 ligand but not TLR3 ligand
The effect of several TLR ligands on NO production in HEI-OC1 cells was examined. HEI-OC1 cells were incubated with various concentrations of Pam3CSK4 as a TLR2 ligand or poly I:C as a TLR3 ligand24,25 for 24 h and 48 h. Pam3CSK4 induced NO production significantly whereas poly I:C did not (Figure 5).
Induction of NO production by TLR2 ligand but not TLR3 ligand. HEI-OC1 cells were incubated with various concentrations of Pam3CSK4 as a TLR2 ligand or poly I:C as a TLR3 ligand for 24 h or 48 h. The concentration of nitrite in the culture supernatant was determined. *P < 0.05, **P < 0.01vs untreated control.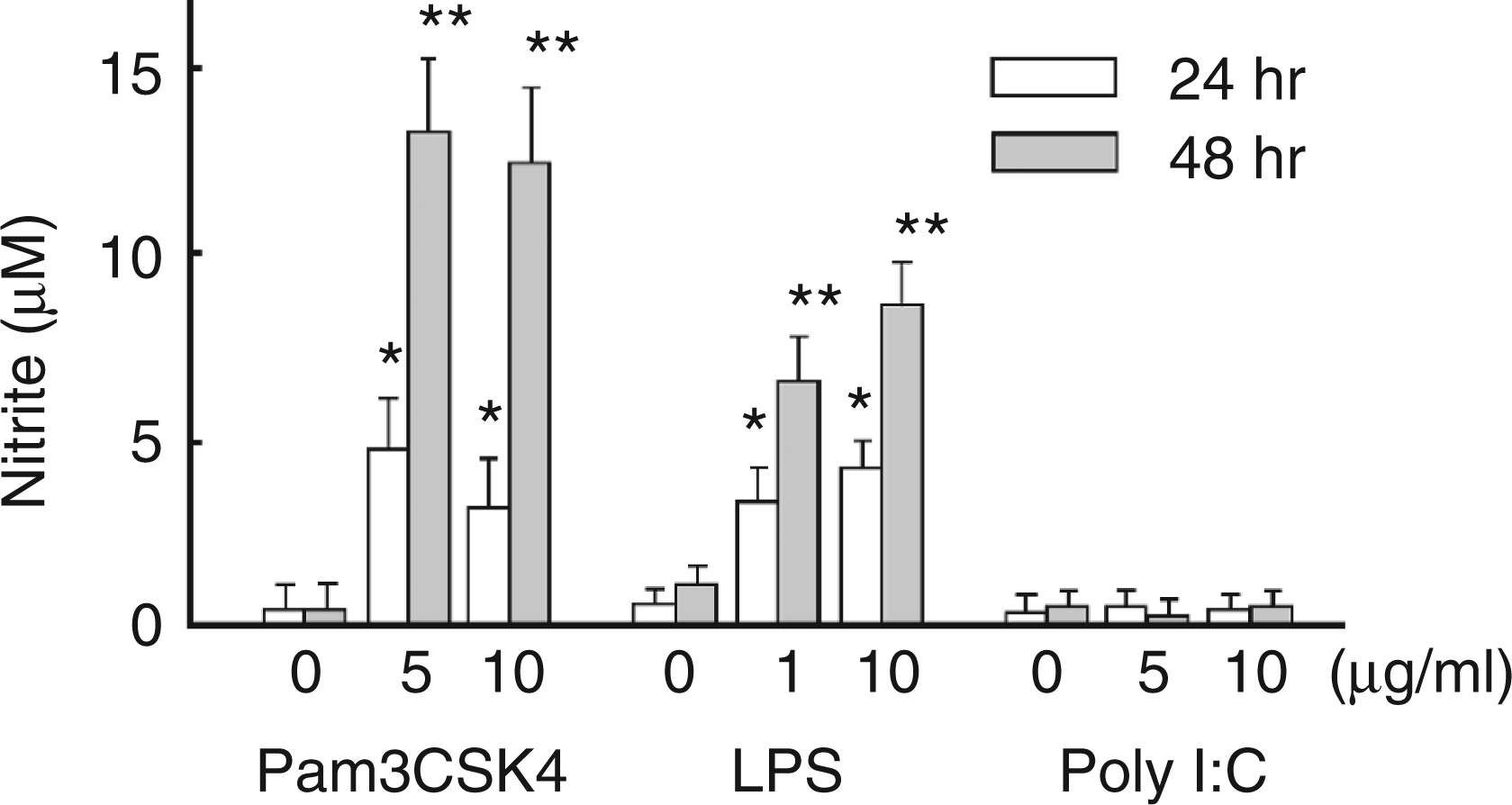
Discussion
In the present study, we demonstrate that murine HEI-OC1 auditory cells produce NO in response to LPS as a TLR4 ligand via iNOS protein expression. Moreover, those cells produce NO under stimulation with Pam3CSK4 as a TLR2 ligand. NO acts as a principal cytotoxic mediator of the immune system.8–10 Therefore, auditory cells might participate in killing infectious agents via NO production in the organ of Corti and play a role on the host defence in the inner ear.26,27 HEI-OC1 cells have been reported to produce various pro-inflammatory cytokines, such as TNF-α, IL-1β, IL-6, in response to LPS in the presence of cisplatin.4–7 Auditory cells may be involved in the regulation of inflammatory response in the organ of Corti via production of pro-inflammatory mediators, including cytokines and NO. Once again, it is suggested that auditory cells play an important role on innate immunity in the inner ear.
LPS-induced iNOS protein expression is triggered by the activation of NF-κB and IRF3.25,28 NF-κB and IRF3 are activated mainly by the MyD88-dependent and MyD88-independent pathways respectively. LPS has been already reported to trigger NF-κB activation in the MyD88-dependent pathway in HEI-OC1 cells. 4 For the first time, the present study demonstrates that, in HEI-C1 cells, LPS triggers the activation of the MyD88-independent pathway of LPS signalling via IFN-β production. Auditory cells seem to utilize both MyD88-dependent and independent pathways in response to LPS and lead to the production of various pro-inflammatory mediators. Interestingly, HEI-OC1 cells produce NO in response to Pam3CSK4 as a TLR2 ligand. TLR2 on the cell surface induces NF-κB activation and intracellular TLR2 induces the production of type I IFN. 29 Therefore, it is reasonable that Pam3CSK4 induces the expression of iNOS. However, HEI-OC1 cells do not produce NO in response to poly I:C as a TLR3 ligand, which triggers a signal pathway similar to the MyD88-independent pathway of LPS (TLR4) signalling. It is suggested that HEI-OC1 cells lack TLR3 expression, although the detailed mechanism awaits further characterization.
HEI-OC1 cells require a relatively high concentration of LPS, such as 10 µg/ml or 100 µg/ml, for NO production. However, murine RAW 264.7 macrophage-like cells produce NO in response to LPS at 10 ng/ml or 100 ng/ml. 30 HEI-OC1 auditory cells are approximately 1000 times less sensitive to LPS than RAW 264.7 macrophage cells. Auditory cells, unlike macrophages, might exclusively respond to LPS when exposed to high concentrations. HEI-OC1 cells may not respond to a low concentration of LPS because of a relatively low expression of TLR4 and CD14. However, IFN-β is normally produced in HEI-OC1 cells within 6 h after LPS stimulation, suggesting no delay in the intracellular events of LPS signalling.
HEI-OC1 auditory cells have been used extensively for characterization of ototoxicity with anti-cancer chemotherapy.1–7 Cisplatin, a commonly used chemotherapeutic agent, increases the TLR4 expression and up-regulates the production of pro-inflammatory cytokines, such as TNF-α, IL-β and IL-6, via NF-κB activation. 4 An excess of pro-inflammatory cytokines results in hearing impairment.4–7 NO is a major pathophysiological mediator of inflammation and host defence;8–10 NO secreted as an immune response is toxic to infectious agents.8–10 Furthermore, an excessive amount of NO is harmful for various cell types.8–10 Therefore, LPS-induced NO production may also cause injury of auditory cells. LPS-induced NO production might be involved in the ototoxicity of anti-cancer drugs.
In summary, HEI-OC1 auditory cells produced NO in response to LPS via iNOS expression. It was triggered by MyD88-dependent and independent pathways in LPS signalling. Auditory cells were suggested to carry not only the sensory receptors of the auditory system but also the sensory receptors of innate immunity. Auditory cells may play an important role in innate immunity of the inner ear.
Footnotes
Funding
This work was supported, in part, by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports and Culture of Japan, and a grant of Strategic Research Foundation Grant-aided Project for Private Universities from Ministry of Education, Culture, Sports, Science, and Technology, Japan (MEXT), 2011-2015 (S1101027).
Conflict of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the article.
