Abstract
Bacteroides fragilis is a member of the normal intestinal flora and is involved in host immunostimulation via TLR2. On the bacterial cell surface, glycoconjugates, such as LPS and capsular polysaccharide A (PSA), have been reported to participate in host immunostimulation via TLR2. Previously, we identified a TLR2-stimulating lipoprotein in B. fragilis cells. In this study, we demonstrated that TLR2-stimulating principal molecules in glycoconjugate fractions prepared from B. fragilis are contaminating proteinous molecules, which may also be lipoproteins. The glycoconjugate fractions were prepared by phenol–hot water extraction of B. fragilis wild type and PSA-deficient strains, followed by hydrophobic interaction chromatography. TLR2-stimilating activities of the fractions were not affected by PSA deficiency. By in-gel TLR2-stimulation assay, molecules in high-molecular-mass area, where capsular polysaccharides were migrated, were found not to stimulate TLR2, but those in the range of 15–40 kDa were active. Further, proteinase K could digest the latter molecules and the TLR2-stimulating activities were migrated to the area of below 15 kDa. These results support that proteinous molecules, which are estimated to be lipoproteins, are responsible for almost all TLR2-stimulating activity in the glycoconjugate fractions prepared from B. fragilis.
Introduction
Bacteroides fragilis is an anaerobic commensal bacterium that is present in the microflora of the human gastrointestinal tract, and can cause intra-abdominal infections, including abscess formation and bacteremia. The cell-surface glycoconjugate fractions of B. fragilis have been shown to be pathogenic factors that modulate the host immune system.1,2 Among them, zwitterionic polysaccharide fractions, including polysaccharide A (PSA) and polysaccharide B (PSB),3,4 which are dominant components of capsular polysaccharides (CPS) produced by B. fragilis, have been extensively characterized. These fractions have been shown to possess the ability to induce intra-abdominal abscesses and to modulate the CD4+ T-cell-dependent immune response. 5 In addition to CPS, LPS are also shown to activate macrophages and induce them to secrete cytokines. 6
TLRs are key receptors of the mammalian innate immune system. They recognize PAMPs and initiate the host response to bacterial invasion. 7 In common Gram-negative bacteria, TLR4 is an important receptor for innate immune recognition of bacterial LPS. 8 In contrast, TLR2 is reported to be a critical receptor for the innate immune response to B. fragilis components. For example, TLR2- but not TLR4-transfected cells recognized an LPS preparation from B. fragilis.9,10 In addition, wild type (WT) and TLR4-hyporesponsive mice formed abscesses in response to a challenge with B. fragilis PSA fractions or bacteria, but TLR2–/– mice exhibited a reduced ability to form abscesses. 5 It is also reported that B. fragilis PSA fractions induced signaling through TLR2 on regulatory T cells to promote immunologic tolerance. 11 These observations suggest that B. fragilis LPS and PSA molecules themselves are the TLR2 ligands. However, the structures of B. fragilis LPS and PSA do not appear to include the typical TLR2 ligand structure.
TLR2 is the predominant receptor for lipoproteins derived from various bacteria. The typical structure of lipoproteins is a protein whose N-terminal cysteine is substituted with a diacylglyceryl group at the thiol group through a thioether linkage, and is sometimes acylated with a fatty acid at its amino group. 12 In Gram-negative bacteria, the inflammatory responses to membrane lipoproteins from Borrelia burgdorferi, Escherichia coli, Neisseria gonorrhoeae and Porphyromonas gingivalis are mediated by TLR2.13–16 In several bacteria, small amounts of lipoproteins are known to contaminate LPS fractions prepared from bacterial cells by conventional extraction methods, and lead to activation of TLR2 in addition to TLR4 stimulation. In several enterobacteria, the contaminating lipoproteins are able to be removed by phenol re-purification of LPS fractions to eliminate TLR2 activity. 17 However, removal of lipoprotein contaminants by phenol re-purification from several non-enterobacterial LPS fractions has been shown to be impossible.16,18 In the latter cases, it is difficult to obtain immunologically purified LPS fractions.
Previously, we identified a TLR2-stimulating lipoprotein in B. fragilis cells by Triton X-114 phase separation. 19 These findings suggest that the glycoconjugate fractions prepared from B. fragilis also contain lipoproteins as contaminants. In the present study, we investigated the possibility that TLR2-stimulating components in the B. fragilis glycoconjugate fractions are lipoproteins.
Materials and methods
Bacterial strain
B. fragilis JCM 11019 (NCTC 9343) was provided by Japan Collection of Microorganisms, RIKEN BRC, which is participating in the National BioResource Project of the MEXT, Japan. This strain is designated as WT.
Deletion of PSA biosynthesis genes of B. fragilis
Two kb upstream and downstream of the PSA biosynthesis locus were amplified separately using primers PSA-1 (5’-ATTACGATTCG
Bacterial components
The bacteria were cultured in GAM broth (Nissui, Tokyo, Japan) under anaerobic conditions at 37℃ for 6 h. The bacterial cells were harvested by centrifugation and then washed three times with saline. B. fragilis cells were suspended in PBS and digested with lysozyme (100 µg/ml) for 2 h at 37℃, and then with DNase and RNase (10 µg/ml) for 2 h at 37℃. The digested cells were subjected to phenol–hot water extraction, according to a previously described method, 21 to obtain crude glycoconjugate extracts. The extracts were then subjected to hydrophobic interaction chromatography on an OctylSepharose 4FF column (GE Healthcare, Little Chalfont, UK) using 0.1 M of acetate buffer (pH 4.5) with a linear gradient of 1-propanol (15–60%), and fractionated into 80 tubes. The selected eluents were then combined, dialyzed, and lyophilized to obtain three fractions.
Bacteroides fragilis WT lipoprotein fractions (BF-WT-TX) were obtained by Triton X-114 (TX-114)-water phase partitioning of the bacterial cells according to a previously described method. 19
FSL-1, a synthetic diacylated lipopeptide, was purchased from EMC Microcollections (Tübingen, Germany). Escherichia coli O111:B4 ultrapure LPS (InvivoGen, San Diego, CA, USA) was used as a TLR4-specific ligand.
Chemical and enzymatic treatments
Specimens were digested with proteinase K (1/100, w/w; Takara, Shiga, Japan) in a buffer system consisting of 10 mM Tris-HCl (pH 7.8), 10 mM EDTA and 0.5% SDS for 16 h at 37℃, to digest proteinous molecules. Phenol re-purification using a sodium deoxycholate–phenol–water system was performed as previously described. 17 Components in the aqueous and the phenolic phase were precipitated with ethanol. To cleave the lipid A part from LPS, the specimens were hydrolyzed with 0.6% acetic acid at 100℃ for 2.5 h.
Analytical methods
Hexose and phosphorus contents were measured using anthrone–sulfuric acid assay and the molybdenum blue method, respectively.22,23 Analytical SDS-PAGE was performed with the Tris–glycine method.
24
The gels were visualized with Coomassie Brilliant Blue (CBB) or periodic acid–silver (
Sugar compositions were analyzed by the alditol acetate method. 26 Specimens were hydrolyzed with 2 M trifluoroacetic acid at 100℃ for 3 h or 6 M HCl at 100℃ for 18 h, derivatized as alditol acetates and then analyzed with an GCMS-QP2010 system (Shimadzu, Kyoto, Japan) equipped with a DB-5 capillary column (0.25 mm × 30 m; Agilent, Santa Clara, CA, USA) or a SP-2330 column (30 m × 0.25 mm; Sigma-Aldrich, St. Louis, MO, USA).
Protein identification was performed by in-gel or on-membrane tryptic digestion followed by MALDI-TOF–MS analysis using an Autoflex speed mass spectrometer (Bruker Daltonics, Billerica, MA, USA) as previously described.19,27 Lipoproteins were identified by a DOLOP database search (http://www.mrc-lmb.cam.ac.uk/genomes/dolop/). The proteins were designated according to the locus tags for the B. fragilis NCTC 9343 genome.
Luciferase assay
Ba/F3 cells that stably expressed p55IgκLuc, an NF-κB/DNA binding activity-dependent luciferase reporter construct (Ba/κB), a murine TLR2 and p55IgκLuc reporter construct (Ba/mTLR2), and a murine TLR4/MD-2 and p55IgκLuc reporter construct (Ba/mTLR4/mMD-2) were kindly provided by Professor K. Miyake (Institute of Medical Science, University of Tokyo, Japan). The NF-κB-dependent luciferase activities in these cells were determined as previously described. 28 The results are shown as the relative luciferase activity, which was determined as the ratio of stimulated to non-stimulated activity.
In-gel TLR-stimulation assay
Specimens were separated by SDS-PAGE and visualized by CBB staining. The resolved components in the gel were cut into 4-mm strips, and each strip was transferred to 1.5-ml microtubes separately and broken down mechanically with a BioMasher II homogenizer (Nippi, Tokyo, Japan) in 100 µl RPMI-1640 medium. The resultant suspensions were applied to a luciferase assay using Ba/mTLR2 cells.
Results
Separation of the glycoconjugate fractions from B. fragilis
The crude glycoconjugate extract from B. fragilis WT cells was obtained by phenol–hot water extraction. Accordingly, 127 mg of the extract was obtained from about 5 ml wet bacterial cells, which were cultured in 800 ml GAM broth. The extract was then fractionated by hydrophobic interaction chromatography. The elution profile is shown in Figure 1. Two peaks, hexose-containing and phosphorous-containing peaks, were observed (Figure 1a), indicating at least two components existed in the extract. The TLR-stimulating activities of eluents were determined using Ba/mTLR2 and Ba/mTLR4/mMD-2 cells (Figure 1b). Ba/κB cells were used as a negative control. The eluted components possessed strong TLR2-stimulating activity but weak TLR4-stimulating activity. SDS-PAGE profiles of the eluents showed that fractions 46–54 contained CBB-positive high-molecular-mass components (Figure 1c) and Separation of crude glycoconjugate extract from B. fragilis WT. (a) Elution profile of the extract on an OctylSepharose column. The dotted line represents the 1-propanol concentration. (b) The NF-κB activation of the fractions eluted from the column. Each fraction was combined with medium to dilute to 1/10 for Ba/κB (KB) and Ba/mTLR4/mMD-2 (TLR4) cells or 1/3000 for Ba/mTLR2 (TLR2) cells. The cells were incubated with the diluted fractions for 4 h, and their NF-κB activation levels were measured in a luciferase assay. (c, d) SDS-PAGE profiles of the fractions eluted from the column. The fractions were separated on a 15% gel and visualized by (c) CBB and (d) SDS-PAGE profiles of each glycoconjugate fractions. (a) intact glycoconjugate fractions, (b) the LPS fractions with or without weak acid hydrolysis. The fractions were separated on a 15% gel and visualized by 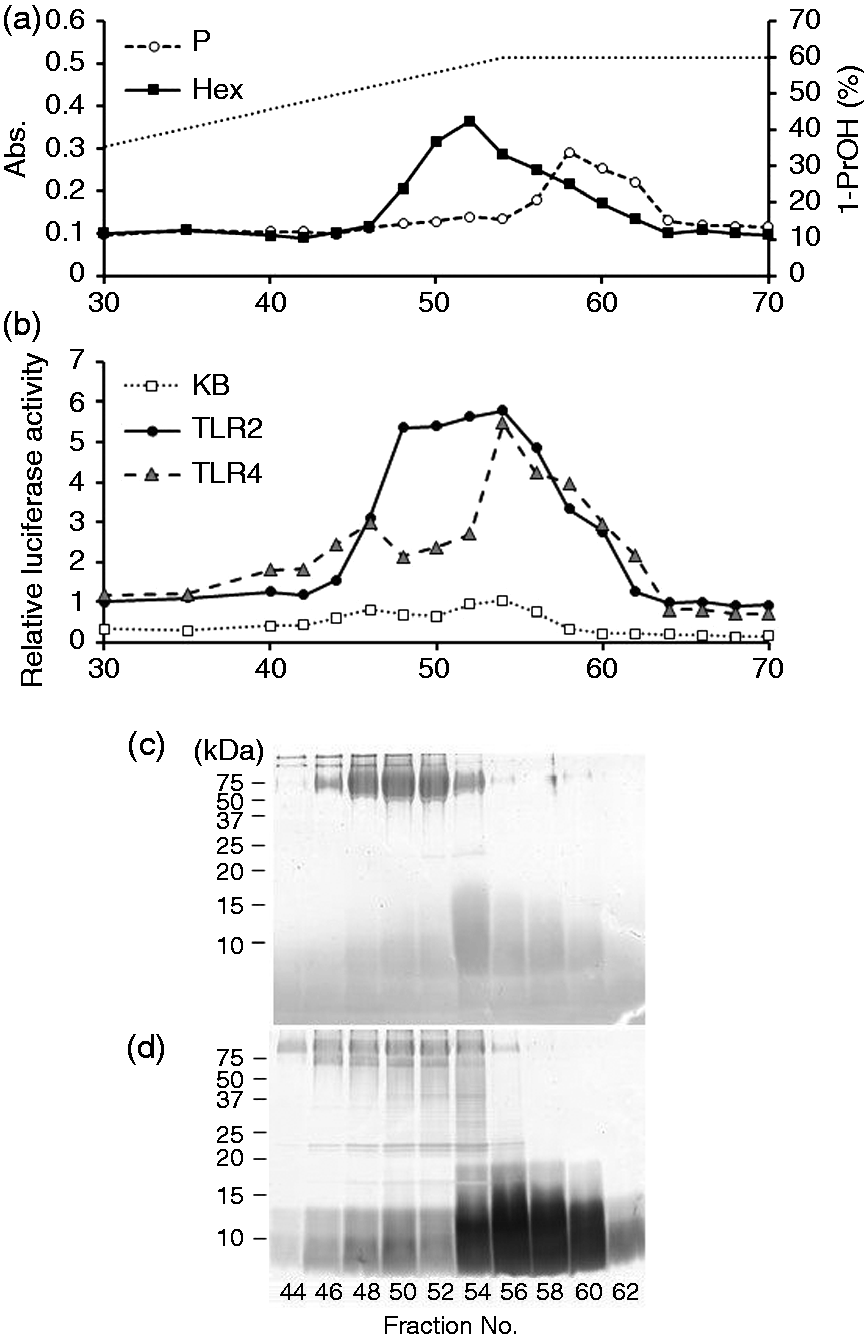
Bacteroides fragilis ΔPSA cells were also subjected to phenol–hot water extraction, and 109 mg extract was yielded. The extract was fractionated similarly (Figure 3). Hexose-containing and phosphorous-containing components were not eluted similarly, indicating that several components were present in the eluents (Figure 3a). The components also stimulated TLR2 strongly and TLR4 weakly (Figure 3b). SDS-PAGE profiles showed that CBB-positive high-molecular-mass components were observed at those in fractions 46–56 (Figure 3c), and that the eluents in fractions 56–62 contained Separation of crude glycoconjugate extract from B. fragilis ΔPSA. (a) The elution profile of the extract on an OctylSepharose column. The dotted line represents the 1-propanol concentration. (b) NF-κB activation of the fractions eluted from the column. Each fraction was combined with medium to dilute to 1/10 for Ba/κB (KB) and Ba/mTLR4/mMD-2 (TLR4) cells or 1/3000 for Ba/mTLR2 (TLR2) cells. The cells were incubated with the diluted fractions for 4 h, and their NF-κB activation levels were measured in a luciferase assay. (c, d) SDS-PAGE profiles of the fractions eluted from the column. The fractions were separated on a 15% gel and visualized by (c) CBB and (d) 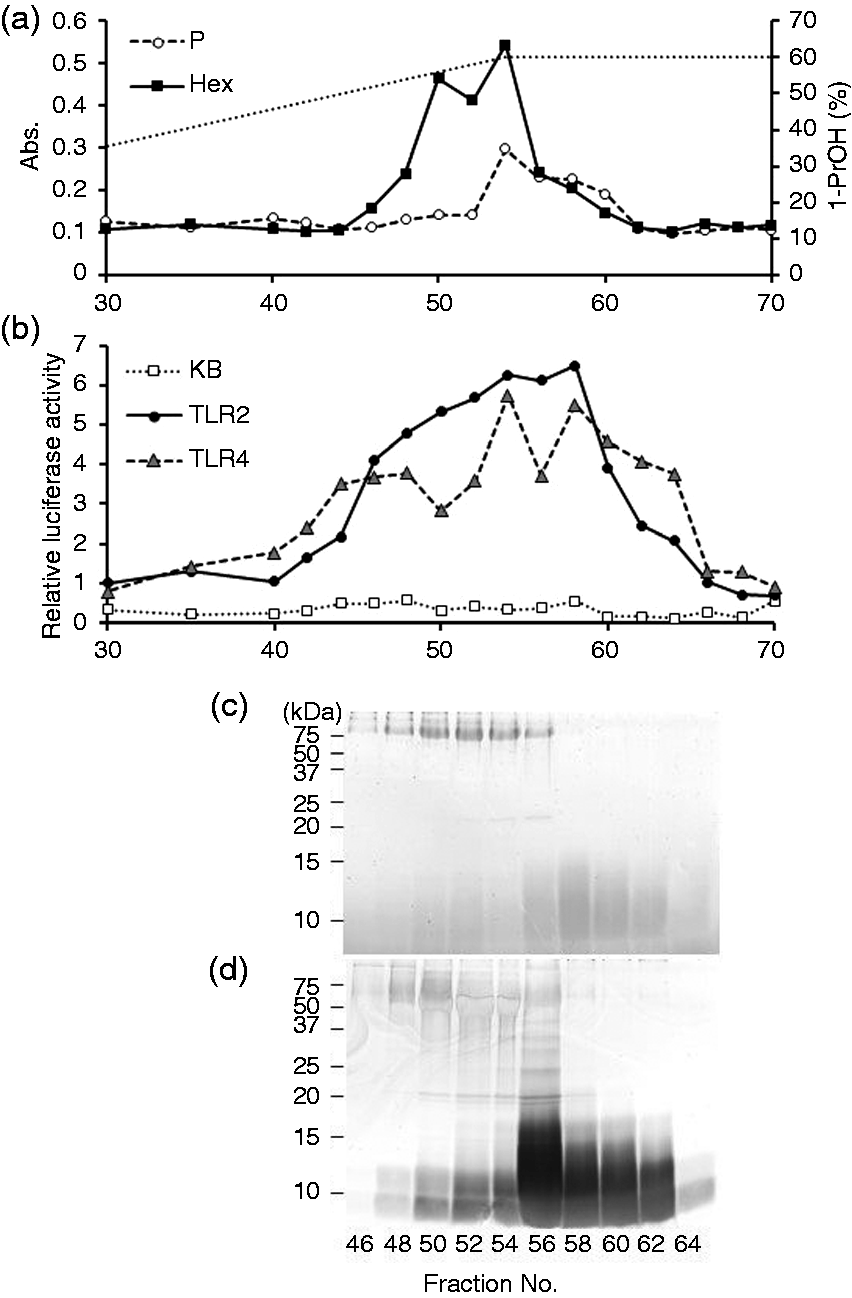
TLR-stimulating activity of the fractions
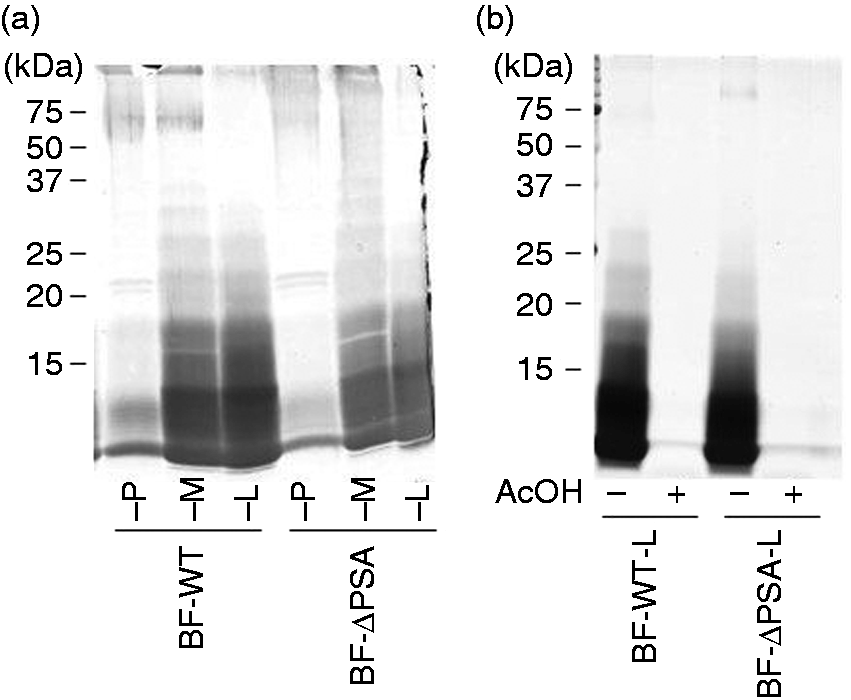
As shown in Figure 2(a), Bf-WT-P and Bf-ΔPSA-P were mainly composed of high-molecular-mass PS, and Bf-WT-L and Bf-ΔPSA-L were mainly composed of short-chain LPS. Thus, the TLR-stimulating activity of the fraction was assayed (Figure 4). Comparisons between equivalent fractions showed that the TLR2-stimulating activities of WT fractions (Figure 4a–c) were similar to those from ΔPSA (Figure 4d–f), suggesting that PSA may not be responsible for the majority of TLR2-stimulating activity. In contrast, TLR2-stimulating activities of LPS-containing fractions (Bf-WT-L and Bf-ΔPSA-L; Figure 4c, f) were lower than those of polysaccharide-containing fractions (Bf-WT-P and Bf-ΔPSA-P; Figure 4a, d). The results indicate that less hydrophobic fractions contain more TLR2-stimulating components.
TLR-stimulating activities of the glycoconjugate fractions (a) Bf-WT-P, (b) Bf-WT-M, (c) Bf-WT-L, (d) Bf-ΔPSA-P, (e) Bf-ΔPSA-M or (f) Bf-ΔPSA-L. The cells, Ba/κB, Ba/mTLR2 and Ba/mTLR4/mMD-2, were incubated with specimens for 4 h, and their NF-κB activation levels were measured in a luciferase assay.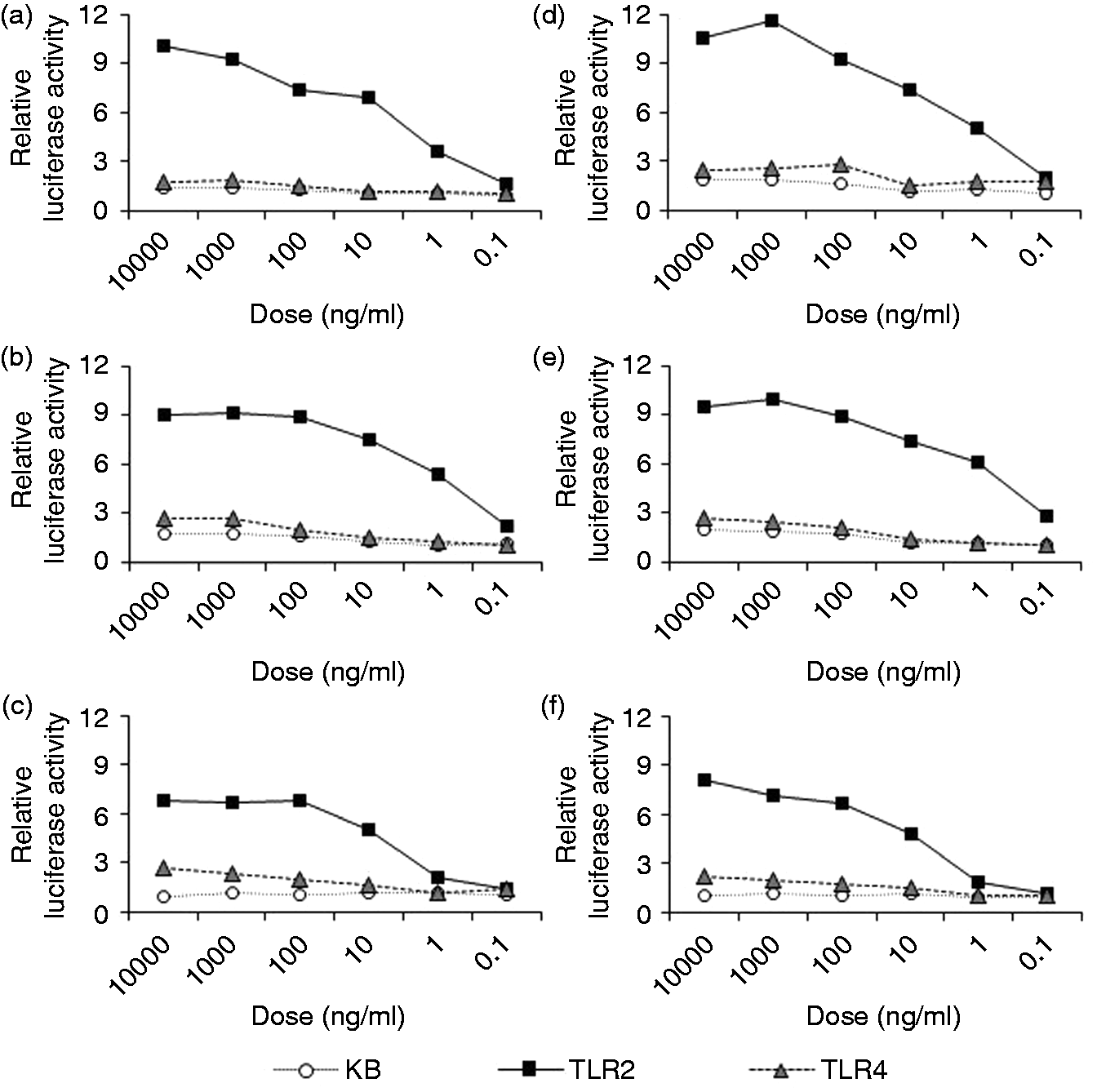
CPS and LPS are not the principal components responsible for TLR2 stimulation
The principal components responsible for TLR2-stimulation in these fractions were examined by in-gel TLR-stimulation assay. Bf-WT-P was separated by SDS-PAGE and the gel strips were subjected to TLR2-stimulation assay. As shown in Figure 5(a), high TLR2-stimulating activities were observed in a molecular-mass range of 15–40 kDa, while the activities were low, in the area above 50 kDa, where PSA was mainly migrated. After Bf-WT-P was digested with proteinase K, almost all TLR2-stimulating activities were found in the area below 15 kDa (Figure 5b). These results suggest that the predominant TLR2-stimulating components in Bf-WT-P are proteinous molecules but not PSA. Similarly, TLR2-stimulating activities in the CPS migrated area in Bf-ΔPSA-P were low (Figure 5c) and the most of its active components were digested into low-molecular-mass molecules by proteinase K (Figure 5d). The results also indicate that the predominant TLR2-stimulating components in Bf-ΔPSA-P are proteinous molecules but not CPS.
In-gel TLR-stimulation assay of the CPS fractions. (a) Bf-WT-P, (b) proteinase K-digested Bf-WT-P, (c) Bf-ΔPSA-P or (d) proteinase K-digested Bf-ΔPSA-P were separated by SDS-PAGE and visualized by CBB staining, and the stripped gels were subjected to NF-κB activation assay on Ba/mTLR2 cells.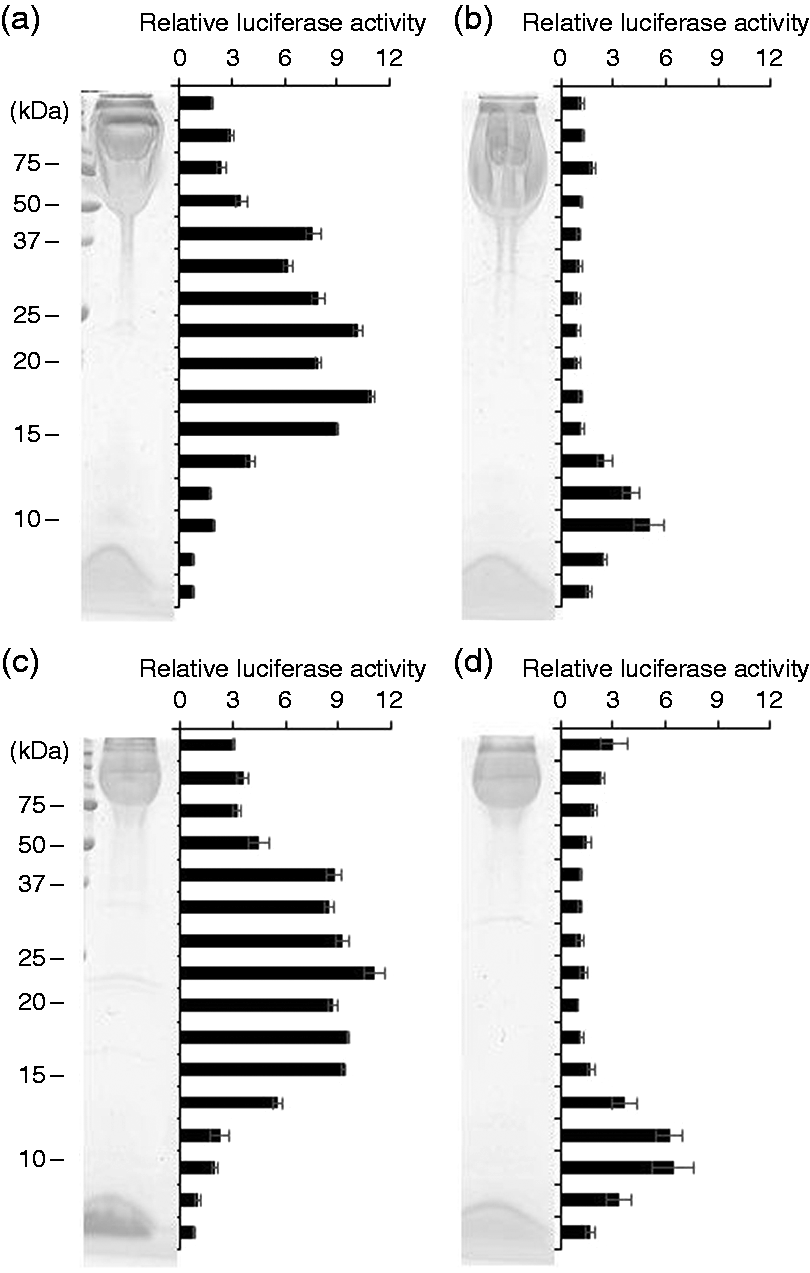
In the LPS-containing fractions, we observed similar results (Figure 6). TLR2-stimulating activities in both Bf-WT-L and Bf-ΔPSA-L were mainly observed in a molecular-mass range of 15–25 kDa (Figure 6a, c). After proteinase K digestion, the activities almost disappeared in this area (Figure 6b, d). The results support that the predominant TLR2-stimulating components in Bf-WT-L and Bf-ΔPSA-L are proteinous molecules but not LPS.
In-gel TLR-stimulation assay of the LPS fractions. (a) Bf-WT-L, (b) proteinase K-digested Bf-WT-L, (c) Bf-ΔPSA-L or (d) proteinase K-digested Bf-ΔPSA-L were separated by SDS-PAGE and visualized by CBB staining, and the stripped gels were subjected to NF-κB activation assay on Ba/mTLR2 cells.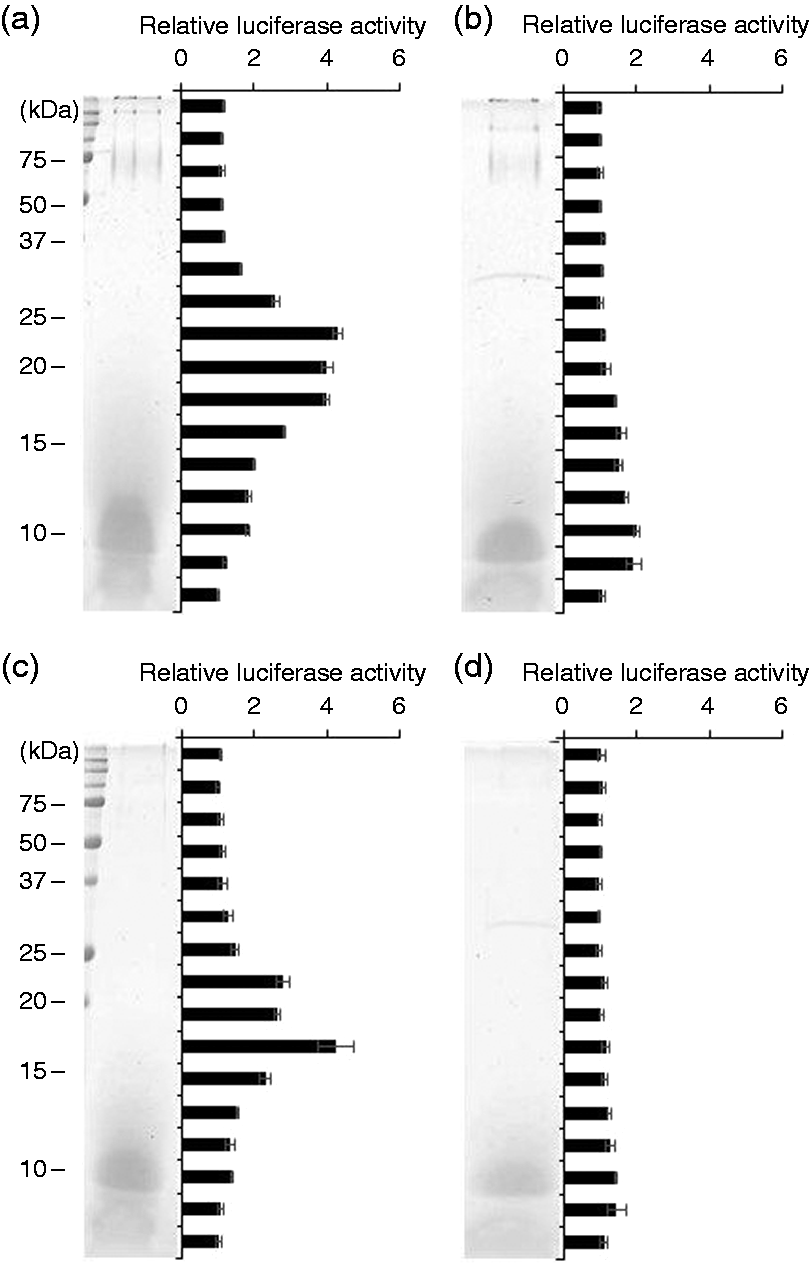
Lipoprotein expression in B. fragilis
We assumed that the TLR2-stimulating proteinous components are lipoproteins. Thus, Bf-WT-P was subjected to phenol re-purification to obtain re-purified Bf-WT-P recovered in an aqueous phase (Bf-WT-P-aq) and extracted components recovered in a phenolic phase (Bf-WT-P-ph). As expected, phenol re-purification of Bf-WT-P had nearly no effect on TLR2-stimulant removal (Figure 7). The SDS-PAGE profile of Bf-WT-P-aq (Figure 7b) was similar to that of intact Bf-WT-P (Figure 7a), while that of Bf-WT-P-ph showed that only a small amount of PSA was extracted (Figure 7c). Furthermore, the TLR2-stimulating profile of Bf-WT-P-aq (Figure 7b) was also analogous to that of intact Bf-WT-P (Figure 7a), but Bf-WT-P-ph possessed only slight activities (Figure 7c). The results concur with previous observations of P. gingivalis LPS.16,18
In-gel TLR-stimulation assay of the phenol repurificated fractions. (a) intact Bf-WT-P, (b) repurified fraction Bf-WT-P-aq, (c) extracted components Bf-WT-P-ph were separated by SDS-PAGE and visualized by CBB staining, and the stripped gels were subjected to NF-κB activation assay on Ba/mTLR2 cells.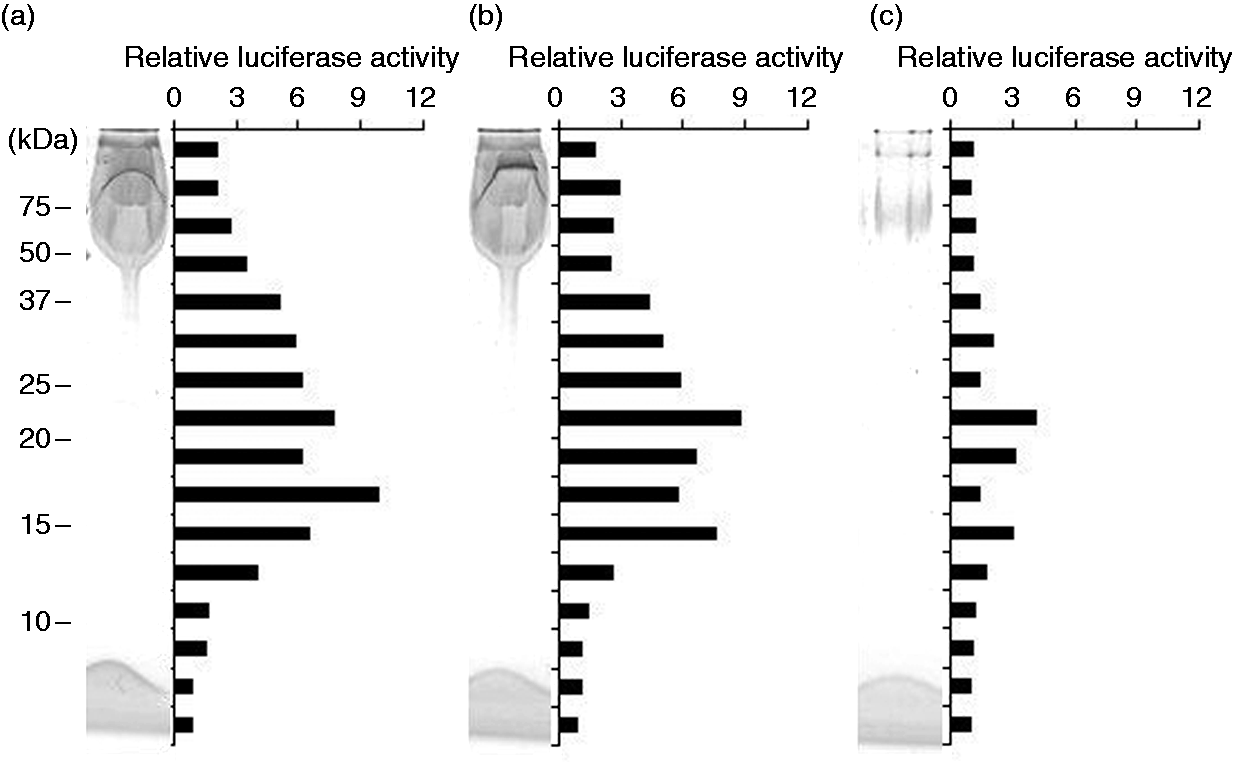
Thus, we attempted to identify lipoproteins in Bf-WT-P. Separation was performed by SDS-PAGE, and the gels were subjected to mass spectrometric analysis of in-gel or on-membrane tryptic digests. Unfortunately, however, we could not identify any protein, probably because the amount of protein was too small to detect.
To confirm whether lipoproteins expressed in B. fragilis, we prepared BF-WT-TX again and analyzed its proteins. We further identified additional lipoprotein candidates, BF0156 (BF9343_0155), BF0586 (BF9343_0559), BF2739 (BF9343_2654), BF3319 (BF9343_3233) and BF4309 (BF9343_4295) by mass spectra. The results suggest that B. fragilis expressed multiple lipoproteins in addition to BF1333 (BF9343_1268), which were previously identified. 19
Discussion
The cell-surface glycoconjugate fractions of B. fragilis, such as LPS and PSA fractions, are reported to modulate the immune system via TLR2.5,9–11 In this study, we re-investigated the principal molecules responsible for TLR2 stimulation in B. fragilis. We separated glycoconjugate fractions by hydrophobic interaction chromatography. We found that LPS was adsorbed on the column and eluted at a 60% 1-propanol concentration, as expected (Figures 1a and 3a). Furthermore, CPS also interacted with hydrophobic interaction chromatography, indicating CPS possess hydrophobic moieties. It is reported that CPS extracted from several Gram-negative bacteria are covalently linked with phospholipid moieties.30,31 Thus, we hypothesized that B. fragilis CPS also possess phospholipid moieties as a membrane anchor. Elution profiles of glycoconjugates on hydrophobic interaction chromatography indicated that both TLR2- and TLR4-stimulating activities were eluted at similar fractions, which contained CPS and LPS (Figures 1b and 3b). However, the results of an in-gel TLR-stimulation assay clearly showed that proteinous molecules, but not CPS nor LPS, are predominant TLR2 ligands (Figures 5 and 6). There are several studies showing that LPS fractions naturally separated from bacterial cells contain a trace amount of lipoproteins as a TLR2 ligand.14,16,32,33 Since both LPS and lipoprotein are amphiphiles, some lipoproteins are considered to possess similar chemical and physical properties to LPS and cannot be separated from LPS. In the case of B. fragilis LPS fractions, a similar explanation should also be applied. In addition, these studies also show that the LPS molecule itself is not a TLR2-stimulating molecule but a TLR4 stimulant. Lipid A, a glycolipid part of LPS, is proven to be responsible for the TLR4-stimulating activity of LPS. 34 The lipid A part of B. fragilis LPS is also characterized as a low TLR4-stimulating structure, which has a diglucosamine monophosphate backbone carrying five acyl groups. 35 Therefore, it is expected that B. fragilis LPS molecules are weak TLR4 stimulants, and that contaminants are responsible for TLR2 stimulation. This hypothesis is supported by our observation that most of the TLR2-stimulating components in Bf-WT-L and Bf-ΔPSA-L can be digested by proteinase K (Figure 6).
In contrast, the structure in PSA causing TLR2 stimulation has not been identified. One structural feature of PSA is a zwitterionic motif composed of FucN4N and a pyruvate group on Gal. It is shown that elimination or introduction of charges on polysaccharides altered the TLR2-stimulating activities.5,36 Thus, the zwitterionic motif may be the structure responsible for TLR2 stimulation. However, this structure is not a typical TLR2-stimulating molecular structure. 37 Furthermore, these polysaccharides were separated as natural and heterogeneous fractions from bacterial cells, like LPS fractions. In particular, molecules contained in Bf-WT-P and Bf-ΔPSA-P possessed a certain degree of hydrophobicity, as shown in Figures 1 and 3, that is, the molecules should be amphiphiles and be substituted by fatty acids or several lipophilic groups, like LPS. Thus, it is possible that the fractions contain several amphiphilic contaminants. Indeed, our results support this possibility. Most of the TLR2-stimulating components in Bf-WT-P and Bf-ΔPSA-P can be digested by proteinase K (Figure 5). Furthermore, the TLR2-stimulating components could not be extracted by phenol re-purification (Figure 7). The action of the components agrees with the behavior of the lipoproteins in P. gingivalis LPS fractions.16,18 These results indicate that complete purification of CPS is impossible at present.
Consequently, we concluded that almost all TLR2-stimulating components are proteinase K-labile molecules, at least in these glycoconjugate fractions. Unfortunately, we could not identify the proteins existing in the fractions. At present, however, we hypothesize that the proteins are lipoproteins. We previously identified TLR2-stimulating lipoprotein BF1333 (BF9343_1268) from B. fragilis. 19 Although we constructed B. fragilis BF1333-deficient mutants, TLR2-stimulating activities of the lipoprotein fractions and glycoconjugate fractions were not altered compared with those from WT bacteria (data not shown). In the B. fragilis NCTC 9343 genomes, 177 lipoprotein candidates have been identified. These data imply that more lipoproteins can be expressed in B. fragilis bacterial cells. In fact, we identified an additional five lipoprotein candidates from BF-WT-TX by mass spectra. Thus, we estimated that the fractions may contain multiple lipoproteins distributed in the molecular mass range of 15–40 kDa, and that the amount of each lipoprotein may be too little to detect.
To analyze the lipoproteins, we attempted to express a large amount of lipoproteins in B. fragilis by overexpression of the lipoprotein biosynthetic gene, phosphatidylglycerol:prolipoprotein diacylglycerol transferase (lgt). However, B. fragilis carrying the expression plasmid pVAL-Exp:BF4367FLAG did not increase the TLR2-stimulating activities of the fractions (data not shown), that is the amount of lipoprotein is estimated to be unchanged.
In Gram-positive bacteria, it has been shown that TLR2-stimulating lipoproteins are contaminated in cell surface components such as lipoteichoic acids (LTA). 38 Since LTA are also amphiphile, complete separation of lipoproteins and LTA is impossible in most bacteria. However, the lipoproteins in Gram-positive bacteria can be depleted by deletion of lgt in the genome. In Staphylococcus aureus, an lgt-deficient mutant has been constructed, 39 and it has been shown that the inflammatory response of the mutant is dramatically reduced. 40 Furthermore, lipoprotein-free LTA, which hardly activates TLR2, can be separated from the lgt-deficient mutant. 41 Thus, it is possible that biological activities of LTA and lipoproteins have been separately investigated to date.
Therefore, to investigate the roles of lipoproteins in the glycoconjugate fraction, we attempted to construct lipoprotein-deficient mutants of B. fragilis by deletion of lgt via homologous recombination. However, the construction of lgt single deletion mutants has not yet been successful. Briefly, we aimed to disrupt the lgt gene in B. fragilis NCTC 9343 using a previously described method. 42 The chromosomal lgt gene was successfully deleted only when B. fragilis NCTC 9343 possessed the plasmid pLYL05 harboring the lgt gene (pLYL05-lgt). This Δlgt strain harboring pLYL05-lgt was repeatedly subcultured in the media without selective antibiotics to cure the plasmid. However, the clone that lost the plasmid was not obtained despite pLYL05 being easily cured from the parent B. fragilis NCTC 9343. Deletion of lgt is known to be lethal to most Gram-negative bacteria,43,44 with a few exceptions. 45 The lgt deletion mutant should help to elucidate the functions of lipoproteins, as well as CPS in B. fragilis. Thus, this is a challenging issue for our next studies.
In conclusion, we demonstrated here that the predominant TLR2-stimulating components in glycoconjugate fractions prepared from B. fragilis are neither CPS nor LPS, but proteinous molecules. Although we could not identify them at present, they may be TLR2-stimulating lipoproteins that we previously found to be expressed in B. fragilis cells. However, we have not yet clarified the roles of the lipoproteins in the glycoconjugate fractions against the reported immune responses. In addition, CPS molecules in B. fragilis, especially zwitterionic PSA, are thought to have important immunomodulatory functions. Discrimination of the roles of both components is necessary for clarifying the function of B. fragilis in humans. Complete elimination of lipoproteins from the glycoconjugate fractions or genetically complete depletion of lipoproteins from bacteria is required in future studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Grant-in-Aid for Scientific Research (No. 26460530) from the Japan Society for the Promotion of Science.
