Abstract
Toll-like receptors (TLRs) play a key role in the recognition of pathogen-associated molecular patterns, including immunostimulatory nucleic acids (INAs). INAs are recognized by TLRs in endosomes, leading to the activation of signalling pathways that activate the innate immune response. This feature makes INAs and their synthetic analogues useful as adjuvants in vaccines and in cancer treatment. We tested a delivery system for the improvement of the therapeutic effect of INAs which consists of a conjugate between transferrin (Tf) and poly-
Keywords
Introduction
Innate immunity is the first step of pathogen recognition and eradication, and is required for development and polarization of adaptive immune responses, 1 which finally eradicate pathogens and also include immunological memory. The innate immune system comprises cellular sensors, the so-called pathogen recognition receptors (PRRs), which recognize broadly conserved pathogen-associated molecular patterns (PAMPs). Perhaps the most investigated PRRs are TLRs. There are 10 known TLR members in the human immune system, which recognize various ligands, for example TLR4 recognizes LPS, TLR5 flagellin, TLR1/2 triacylated peptides, TLR3 dsRNA and TLR9 unmethylated CpG oligonucleotides. 2 TLRs are expressed on various immune cells, including macrophages, dendritic cells (DCs), B cells, specific types of T cells and even on non-immune cells, such as fibroblasts and epithelial cells. 3 When TLRs recognize their cognate ligand they dimerize, which activates a signalling cascade and induces genes involved in a host immune response. TLRs signal through the MyD88-dependent signalling pathway (all TLRs except for TLR3), which leads to the induction of inflammatory cytokines, and through the TRIF-dependent pathway (TLR3 and TLR4), which leads to the production of type I IFNs. TLRs are located at the cell surface (TLRs 1, 2, 4, 5 and 6) or in intracellular compartments, such as endosomes, where they recognize mainly nucleic acids (TLRs 3, 7, 8 and 9). 3 However, in several cell lines, including HMVEC-dLy Ad, 4 TLR3 was also shown to be present at the cell surface.5–7
TLR3 agonists, such as dsRNA or its synthetic analogue polyinosinic:polycytidylic acid [poly(I:C)], have many unique immunostimulatory properties which make them interesting candidates for adjuvants in modern vaccines, as well as for cancer treatment.8,9 TLR3 activation leads to the TRIF-dependent signalling resulting in type I IFN production, which enables effective anti-viral immune response without causing excessive inflammation. TLR3 is expressed in myeloid dendritic cells (mDC), which are the most effective APCs that are able to cross-present Ags.
8
It has been demonstrated that TLR3 signalling and type I IFN induction stimulate cross-presentation in APCs, which leads to the effective cytotoxic T lymphocyte (CTL) response.10,11 Moreover, activation of TLR3 has been shown to activate NK cells, which greatly contribute to cancer regression.12,13 Concerning classical vaccines, so far TLR3 ligands have been used as effective adjuvants in a mice virus infection model.14,15 In addition to preventing disease, vaccines could also be used in a therapeutic manner to enhance the host defence against cancer cells. One important obstacle in the cancer remediation by the immune system is the immunosuppressive environment that supports T-cell tolerance, which prevents the development of an effective immune response.
16
Ampligen, a synthetic analogue of dsRNA with reduced toxicity, was shown to be capable of breaking the tumor-related immunosuppressive mechanisms.
17
It has been demonstrated
18
that poly(I:C) induces apoptosis of human breast cancer cells in a TLR3-dependent manner. Moreover, poly(I:C) stabilized with poly-
However, the therapeutic potential of immunostimulatory nucleic acids (INAs) in general and poly(I:C) is limited owing to the inefficient cellular uptake and rapid degradation. It has been shown that adsorption of CpG oligodeoxynucleotides (CpG ODN) onto cationic polylactide-co-glycolide (PLG) microparticles accelerated and enhanced protective immunity elicited by Anthrax Vaccine Adsorbed (AVA, the licensed human anthrax vaccine).
20
Thus, we hypothesized that by stabilization and delivery of poly(I:C) into the endolysosomal pathway we could enhance its immunostimulatory effect. As a proof of a principle we prepared a conjugate between transferrin (Tf) and poly-
Materials and methods
Plasmids and reagents
pUNO-hTLR3-HA was purchased from InvivoGen (San Diego, CA, USA), phRL-TK from Promega (Madison, WI, USA), pIFNβ-FLuc was a kind gift from Dr John Hiscott (McGill University) and pELAM-Luc was a kind gift from Dr Terje Espevik (Institute of Cancer Research and Molecular Medicine, NTNU-Norwegian University of Science and Technology). For cell-transfection assays all plasmids were isolated using Purelink™ Quick Plasmid Miniprep Kit (Invitrogen, Carlsbad, CA, USA). Human holo-transferrin and PLL were purchased from Sigma (St Louis, MO, USA). N-succinimidyl 3-(2-pyridyldithio) propionate (SPDP) was purchased from Thermo Scientific (Waltham, MA, USA). GeneRuler™ DNA Ladder Mix and SDS-PAGE Page Ruler™ standards were purchased from Fermentas (Burlington, ON, Canada). Fluorescent dyes LysoTracker™ Green and HOECHST 33258 were obtained from Invitrogen. FITC-labelled anti-EEA1 Abs were purchased from BD Biosciences (San Jose, CA, USA). Synthetic ligand poly(I:C) with an average size of 1.5–8 kb, as well as rhodamine-labled poly(I:C) were purchased from InvivoGen. ODN 10104 labelled with Alexa fluor 633 dye was purchased from Sigma. All chemicals used in buffer preparation were also from Sigma. The RNase ONE was from Promega. Reagents used in dual-luciferase reporter assay were from Lux (Laurel, NJ, USA) and Promega. Human IL-6 ELISA kit was purchased from BenderMedsystems (Burlingame, CA, USA).
Cell culture and reagents
Cells were grown in a humidified atmosphere at 37°C and 5% CO2. The human embryonic kidney (HEK) 293 cell line was purchased from the American Type Culture Collection and the cervical cancer cell line HeLa was kindly provided by Irena Oven (University of Ljubljana). HEK 293 and HeLa cells were maintained in DMEM + GlutaMAX-I with 10% FBS (Invitrogen). HMVEC-dLy Ad cells (Lonza, Walkersville, MD, USA) were maintained in primary endothelial cell growth medium with endothelial cell supplements EGM-2-MV Bullet Kit (Lonza).
Preparation of TfPLL conjugate
Synthesis of TfPLL conjugate
Tf was coupled to PLL via a disulphide bond using a heterobifunctional reagent SPDP (Thermo Scientific) as described previously24,25 (Supplementary Figure S1). Firstly, Tf and PLL were dissolved in PBS-EDTA buffer (100 mM sodium phosphate, 150 mM sodium chloride, 1 mM EDTA, pH 7.5) and then separately modified with SPDP (Supplementary Figure S1, reaction A). Briefly, Tf was mixed with SPDP at a molar ratio of (1:10) and PLL, with an average molecular mass of 50 ku or 100 ku (approximate 400 or 800 lysine residues respectively), at a molar ratio of (1:20). After 1 h incubation with gentle shaking at room temperature (20–22°C), the reaction mixture was desalted using PD-10 Deslting columns (Amersham Biosciences, Piscataway, NJ, USA) to remove reaction by-products. The level of SPDP-modification was determined by Pyridine-2-Thione Assay according to the manufacturer’s instructions (Thermo Scientific). Then, SPDP-modified PLL was reduced by 50 mM DTT (final concentration) and 1-h incubation with gentle shaking which produced a free sulfhydryl group (Supplementary Figure S1, reaction B). Sulfhydryl-activated PLL was desalted again to remove DTT. Finally, pyridyldithiol-activated Tf and sulfhydryl-activated PLL were conjugated by mixing and 18 h incubation at room temperature with gentle shaking (Supplementary Figure S1, reaction C). By varying the amount of sulfhydyl-activated PLL while the amount of pyridyldithiol-activated Tf remained the same we prepared conjugates at three different molar ratios, namely: Tf: PLL = 1:0.1, 1:0.2 and 1:1.
Isolation of TfPLL conjugate
Conjugates were isolated by gel-filtration chromatography using BioSep-SEC-s3000 column (Phenomenex, Torrance, CA, USA) and PBS-EDTA buffer containing 1 M guanidine hydrochloride (20 mM sodium phosphate, 150 mM sodium chloride, 1 M guanidine hydrochloride, pH 7.5). Fractions containing TfPLL conjugates were combined and dialyzed against 20 mM Hepes buffer containing 100 mM sodium chloride (pH 7.5). Isolated conjugates were validated by SDS-PAGE.
Formation of complexes between TfPLL conjugate and poly(I:C)
Gel mobility shift assay
The poly(I:C) + TfPLL complexes were prepared as described before. 26 Various amounts of the TfPLL conjugate were added to a constant mass of poly(I:C) so that the mass ratio between poly(I:C) and TfPLL conjugate varied from 1:0.25 to 1:1.5. The TfPLL conjugate ranging from 62.5 to 375 ng (dissolved in 20 mM Hepes buffer containing 100 mM sodium chloride, pH 7.5) was slowly added to the 250 ng of poly(I:C) (dissolved in 20 mM Hepes buffer containing 100 mM sodium chloride, pH 7.5) and mixed gently. The samples were incubated for 1 h at room temperature and analyzed by 1% agarose gel electrophoresis in TAE buffer (40 mM Trisma-base, 20 mM glacial acetic acid, 1 mM EDTA, pH 8.0). The poly(I:C) + PLL complexes were prepared and analyzed in exactly the same manner as described above.
Stability of poly(I:C) in complexes
Spectrophotometric measurements of poly(I:C) degradation
Various amounts of the PLL solution (dissolved in 20 mM Hepes buffer containing 100 mM sodium chloride, pH 7.5) were added to a 20 µg/ml poly(I:C) solution (dissolved in 20 mM Hepes buffer containing 100 mM sodium chloride, pH 7.5) so that mass ratio between poly(I:C) and PLL varied from 1:0.025 to 1:1. Samples were mixed gently and incubated for 30 min at room temperature. Then, 10 U of the RNase ONE (Promega) were added to the 1 ml of poly(I:C) + PLL solution and samples were immediately analyzed spectrophotometrically (UV/VIS Lambda 25; Perkin Elmer, Waltham, MA, USA) at 260 nm for 20 min. We made use of the so-called hyperchrome effect 27 in which a monomeric solution of nucleotides released during degradation displays a higher optical density at 260 nm than the same molar concentration of polymerized nucleotides where bases are stacked and hence absorb less light.
Biological activity of poly(I:C) in complexes after RNase treatment
Dual luciferase assay
HEK 293 cells were harvested from an actively growing culture and plated into CoStar White 96-well plates (Corning) at 3 × 104 cells per well for transfection. After 24 h at 70% confluency, they were transfected with a mixture of jetPEI™ (Polyplus transfection) according to the manufacturer’s instructions (Invitrogen), and plasmids pIFN-β-FLuc (40 ng per well), pUNO-hTLR3-HA (20 ng) and phRL-TK (5 ng) coding for the IFN-β-induced firefly luciferase reporter, full-length human TLR3 and the Renilla luciferase transfection control, respectively. Twenty-four h post-transfection the culture-medium was replaced with 90 µl fresh DMEM with 10% FBS; cells were subsequently stimulated with 10 µl of samples (the final concentration of poly(I:C) was 10 µg/ml). Samples for stimulation were prepared as follows: to the solution of poly(I:C) (100 µg/ml) in complexes with PLL, prepared as previously described, or poly(I:C) alone, 10U of the RNase ONE (Promega) was added. After 20 min incubation at room temperature we also added the same amount of PLL to the sample containing poly(I:C) treated with the RNase alone and incubated for 30 min at room temperature so that the composition of the samples was the same. The so-prepared samples were added onto the cells and left to incubate for 18 h. After stimulation, cells were harvested and relative luciferase activity was determined using the Dual Glo Luciferase Assay System reagents (Promega). Luminescence was quantified using the plate reader Orion II (Berthold Detection Systems, Pforzheim, Germany).
Internalization of poly(I:C) in complexes with the TfPLL conjugate
Confocal fluorescence microscopy
Receptor-mediated internalization of complexes of INAs with TfPLL was tested with confocal fluorescence microscopy of HeLa cells. Twenty-four hours before the experiment, cells were seeded onto 8-well microscopic chambers (Ibidi) at a density of 5 × 104 per well. Complexes of INAs and TfPLL were prepared as follows. 1 µg of poly(I:C)-Rhodamine (InvivoGen) was diluted in OptiMEM (Invitrogen). 1 µg of TfPLL or 1.25 µg of PLL were added to the poly(I:C) mixture. After gentle mixing, samples were incubated for 30 min at room temperature. Then, DMEM with 10% FBS was added to a final volume of 200 µl and transferred onto the cells. The final concentration of poly(I:C)-Rhodamine in the growth medium was 5 µg/ml. After 4 h incubation at 37°C cells were washed 5 times with prewarmed DMEM and stained for microscopy. Lysosomes were stained with LysoTracker™ Green (Invitrogen) and nuclei with HOECHST 33289 (Invitrogen). Internalization of complexes was visualized with Leica TCS SP5 confocal scanning microscope, using laser lines 405, 488, 475 and 633.
Flow cytometry
HeLa cells were seeded onto a 6-well plate (3 × 105 cells per well) in 2 ml medium. The following day poly(I:C)-Rhodamine was complexed with PLL or TfPLL. Poly(I:C)-rhodamine (0.8 µg) was diluted in OptiMEM. An equal mass of TfPLL or 1.25 times the mass of PLL was added to the poly(I:C)-Rhodamine in OptiMEM. The mixture was incubated for 30 min at room temperature, diluted to 1 ml with fresh DMEM with 10% FBS and transferred onto the cells. After 4 h cells were washed in the wells 5 times with cold PBS, harvested and washed another 5 times with cold PBS, and resuspended in 500 µl of FACS buffer (0.5% BSA in PBS). Flow cytometry analysis was performed on EPICS ALTRA flow cytometer (Beckman Coulter, Brea, CA, USA). In each sample 10000 cells were analyzed. Collected data were analyzed by using WinMDI flow cytometry application.
Biological activity of complexes
Cytokine detection by ELISA
HMVEC-dLy cells were cultured at 4 × 104 cells per well in 100 µl EGM2-MV medium in 96-well plates. The cells were treated with poly(I:C) alone or in complexes with PLL and TfPLL. The complexes were prepared using different concentrations of poly(I:C) (InvivoGen) [diluted in 20 mM Hepes buffer containing 100 mM sodium chloride (pH 7.5)] to which we added an appropriate amount (as determinated by a gel mobility shift) of TfPLL or PLL. After gentle mixing and 30 min incubation at room temperature the complexes were added to the cells. The final concentration of poly(I:C) in the growth medium was 0.5 µg/ml, 1 µg/ml or 5 µg/ml. When testing the efficiency of cellular uptake, the growth medium was removed after 3 h and cells were washed thoroughly to remove residual poly(I:C) which was not internalized during this time period. Inhibition of the uptake of poly(I:C) complexes with TfPLL conjugates was tested with 100 × molar excess of Tf (Sigma). After 24 h the supernatants were collected and human IL-6 protein levels were measured by a sandwich ELISA, according to the manufacturer’s instructions (Bender MedSystems).
Results
Preparation of TfPLL conjugate
We conjugated Tf and PLL through disulphide bonds. Using the heterobifunctional reagent SPDP, 25 pyridyldithiol groups were introduced into both Tf and PLL (Supplementary Figure S1, reaction A). Addition of 10 × or 20 × molar excess of SPDP resulted in Tf and PLL with 7 and 16 incorporated pyridyldithiol moieties, respectively, as determinated by the Pyridne-2-Thione Assay. Pyridyldithiol groups in PLL were subsequently reduced to produce a free sulfhydryl group (Supplmentary Figure S1, reaction B), which, upon addition of pyridyldithiol-activated Tf, reacted to form the TfPLL conjugate (Supplementary Figure S1, reaction C). To determine the stoichiometry of the synthesis, we varied the amount of sulfhydryl-activated PLL at the constant amount of pyridyldithiol-activated Tf. In this way we prepared conjugates at three different molar ratios, namely: Tf:PLL = 1:1, 1:0.2 and 1:0.1. The disulfide bond between Tf and PLL is sensitive to reducing agents. We can observe a Tf band at 77 ku on an analytical SDS gel (Supplementary Figure S2, lanes 2, 4 and 6), while PLL cannot be separated by SDS-PAGE because of the highly positive charge that prevents entry into the gel. 24 The same holds true for the high molecular mass and positively-charged TfPLL conjugates which cannot be observed without the addition of a reducing agent. By comparison of the non-reduced (Supplementary Material Figure S2, lanes 1, 3 and 5) and reduced (Supplementary Material Figure S2, lanes 2, 4 and 6) conjugate samples, we can estimate the amount of conjugated Tf, which is proportional to the amount of added sulfhydryl-activated PLL.
The reaction mixture was purified by gel-filtration chromatography (Figure 1) to remove the reaction by-products and non-conjugated Tf which would interfere with further biological activity assays. Because the only component that absorbs at 280 nm is Tf, peaks eluting earlier than Tf represent TfPLL conjugates. The chromatogram in Figure 1 shows the stoichiometry of TfPLL conjugation as a function of the molar ratio between Tf and PLL. At the molar ratio of 1:1 almost all of the Tf is conjugated to PLL. Fractions containing the TfPLL conjugates were combined and dialyzed against the 20 mM Hepes buffer containing 100 mM sodium chloride (pH 7.5).
Purification of the TfPLL conjugate (conjugate between transferrin and PLL) by gel-filtration chromatography. By varying the amount of sulfhydryl-activated PLL at constant amount of pyridyldithiol-activated Tf, we prepared conjugates at three different molar ratios, namely: Tf: PLL = 1:1 (thick line), 1:0.2 (thin line) and 1:0.1 (broken line). The stoichiometry of TfPLL conjugation is dependent on the molar ratio between Tf and PLL. The fractions containing TfPLL conjugates were combined as indicated in the figure.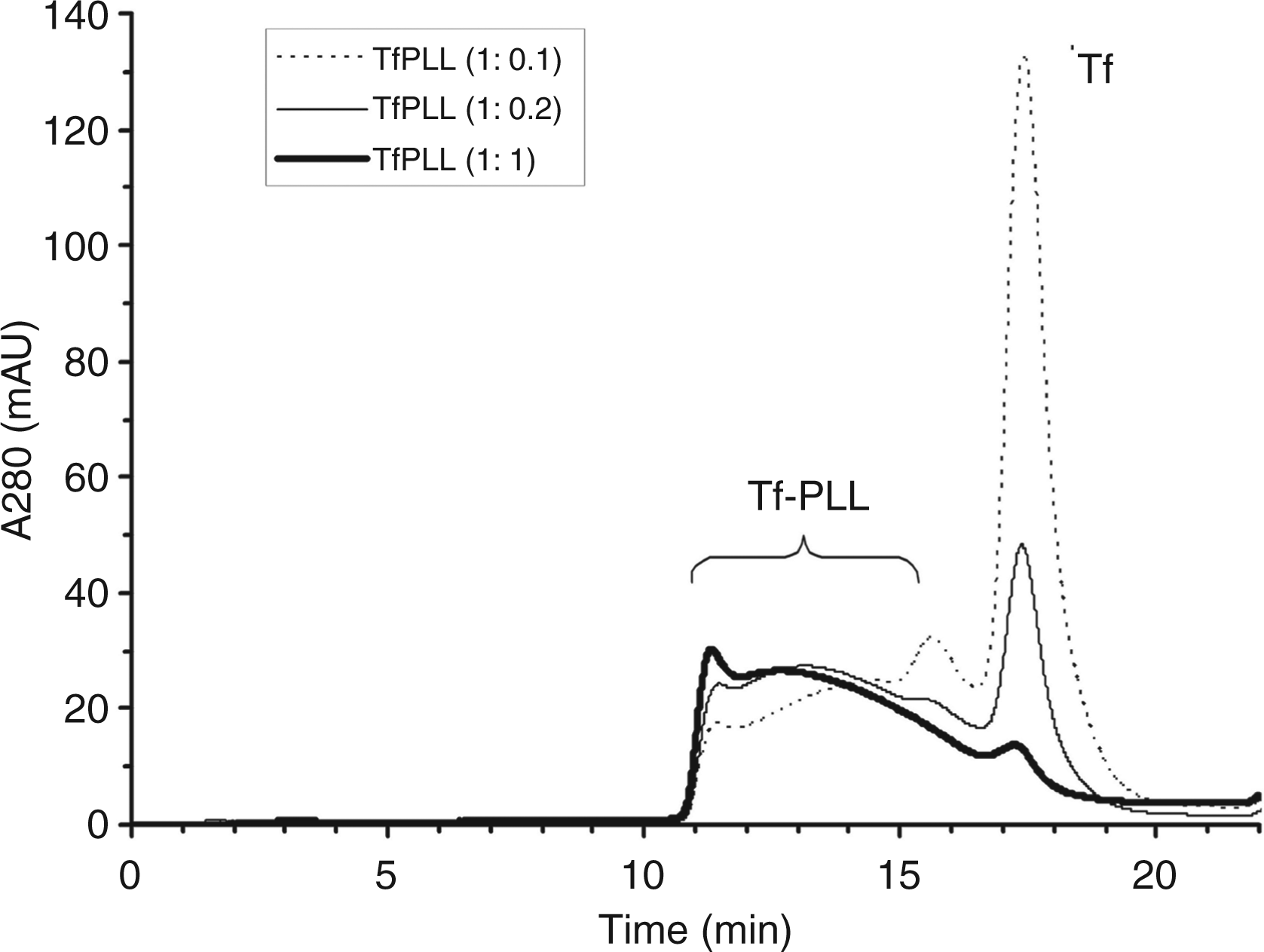
Formation of the complex between TfPLL conjugate and poly(I:C)
The synthetic polycation PLL of the TfPLL conjugate binds negatively-charged poly(I:C). To test if this is, indeed, the case we used gel-mobility shift method in which the poly(I:C) band retardation occurs because of the neutralization of negative charges by the positive charges of TfPLL.
26
As shown in Figure 2, electrophoretic mobility of poly(I:C) + TfPLL complexes decreased gradually with the increasing amount of added TfPLL conjugates. When the mass ratio between TfPLL and poly(I:C) was 1:1, complete retardation of poly(I:C) occurred as the negative charge was neutralized (Figure 2, lane 5, bold). Therefore, we used this mass ratio in all further biological activity tests. As complexes poly(I:C) + PLL are the proper control for evaluating the effect of Tf on endocytosis efficiency, the gel-mobility shift was done also for these (Supplementary Figure S3). The ratio between PLL and poly(I:C) used in further tests was 1.25:1.
Agarose gel mobility shift of poly(I:C) in complexes with TfPLL conjugate (conjugate between transferrin and PLL). Mobility of poly(I:C) gradually decreases as the amount of the TfPLL conjugate is increased. Lane 1 represents poly(I:C) (250 ng) alone, while lanes 2–7 represent poly(I:C) (250 ng) in complexes with the increasing amounts (62.5–375 ng) of the TfPLL conjugate. Complete retardation of poly(I:C) occurs at the 1:1 mass ratio between TfPLL and poly(I:C) (lane 5, bold). Lane M represents a DNA length standard (GeneRuler™ DNA Ladder Mix).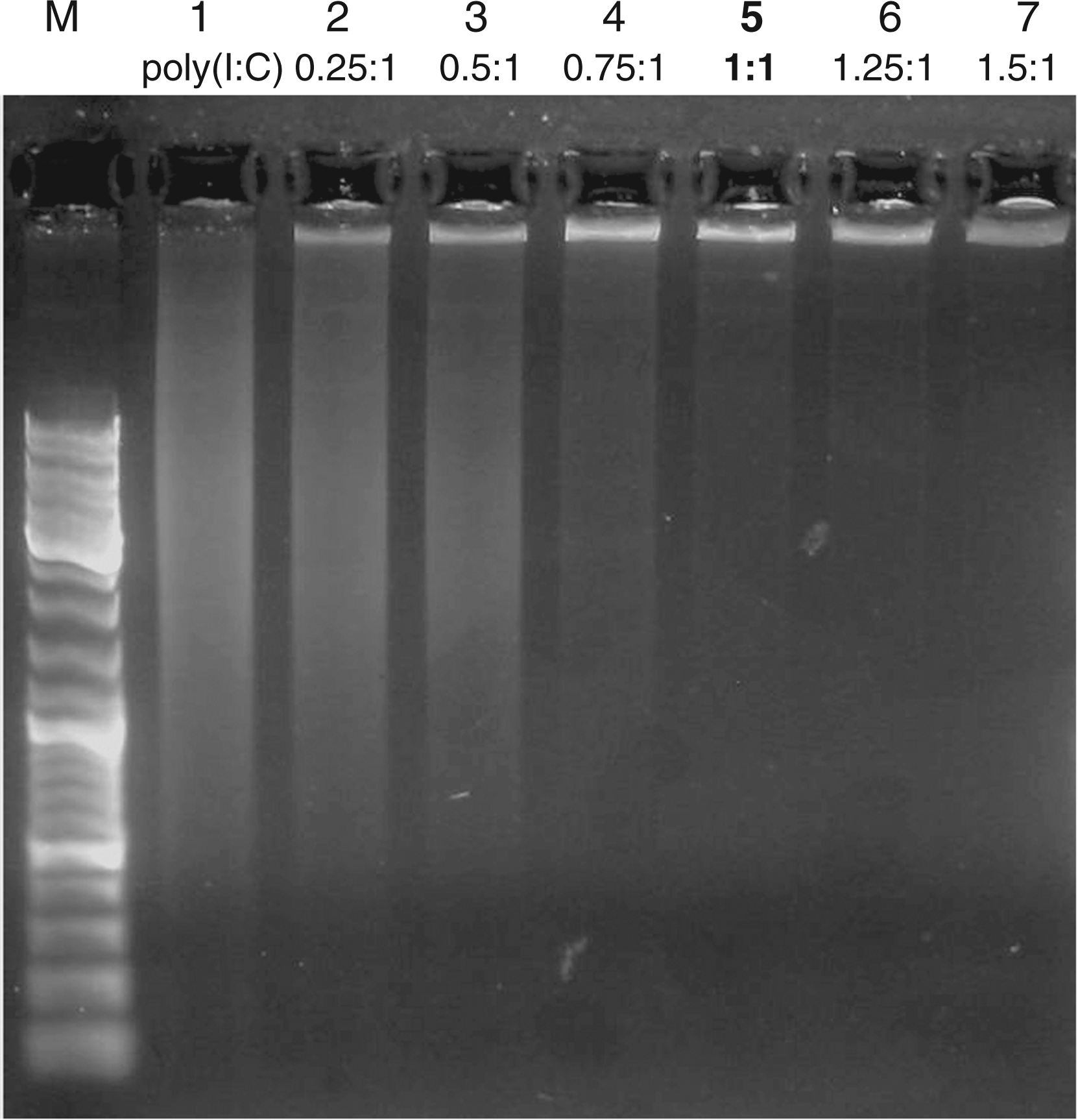
Nuclease protection of poly(I:C) in complexes
Poly(I:C) is sensitive to degradation by RNases. We wanted to investigate if the TfPLL conjugate prevents degradation of poly(I:C) and thus confers its protection against nucleases. We designed an experiment which enabled us to spectrophotometrically monitor the degradation of poly(I:C) in complexes by the RNase in the presence of PLL, as PLL is the only component of the conjugate which is supposed to bind and stabilize poly(I:C). By increasing the amount of PLL the amount of the non-degraded poly(I:C) increases. At the poly(I:C): PLL mass ratio of 1:0.5, approximately half of poly(I:C) remains non-degraded but at the ratio of 1:1, almost no degradation of poly(I:C) occurs (Figure 3A). This result indicates the efficient protection from the RNase degradation, which depends on the ratio between PLL and poly(I:C).
PLL protects poly(I:C) from degradation by the RNase. (A) Degradation of poly(I:C) is monitored spectrophotometrically where higher absorbance indicates more degraded poly(I:C). By increasing the amount of PLL the amount of the non-degraded poly(I:C) increases. The thin broken line represents poly(I:C) + PLL complexes with PLL being 2.5% of poly(I:C) mass. The degradation of this sample is very rapid. In contrast, the boldest lane represents sample where almost no poly(I:C) degradation occurs, as the amount of PLL is high enough to bind and protect total poly(I:C). (B) Activation of TLR3 receptor (measured by dual luciferase test) by poly(I:C) alone and in complexes with PLL after RNase treatment. The first column represents non-stimulated HEK 293 cells (transfected with pUNO-hTLR3-HA, phRL-TK and pIFNβ-FLUC). The second column represents cells stimulated with poly(I:C) (10 µg/ml), pre-incubated with RNase and subsequently supplemented with PLL. In this case no TLR3 activation occurs as the RNase degraded poly(I:C). In contrast, poly(I:C) + PLL complexes (the last column) are able to significantly [*P < 0.005 (Student’s t-test assuming unequal variances)] activate TLR3 receptor, which means PLL effectively protected poly(I:C) from degradation, as only non-degraded poly(I:C) is capable of receptor activation.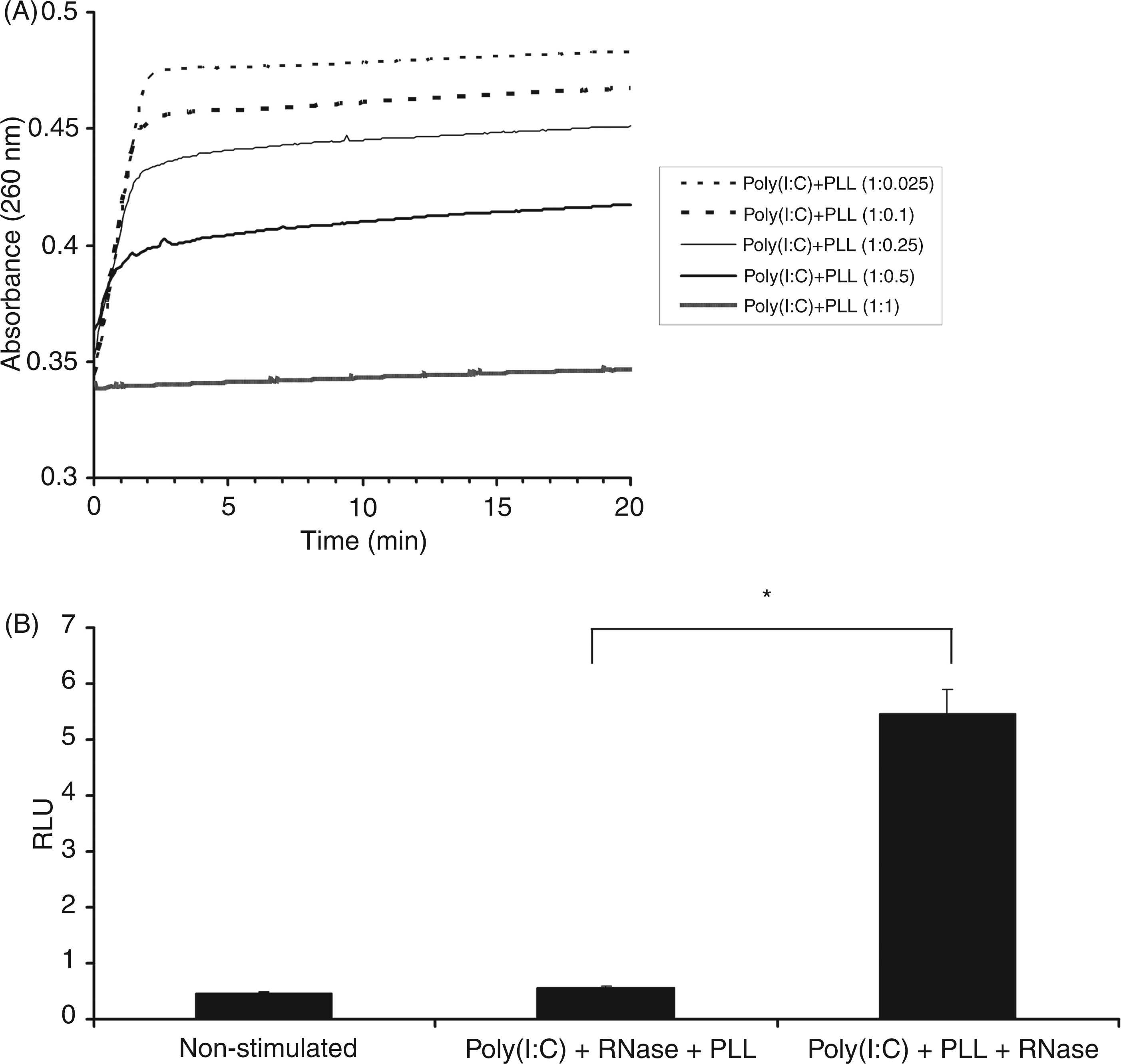
To test whether protection of poly(I:C) from the RNase degradation also has biological importance we tested activation of the TLR3 receptor by poly(I:C) alone and in complexes with PLL after 20 min RNase treatment. As demonstrated in Figure 3B, poly(I:C) alone was unable to activate the TLR3 receptor as it was degraded by the RNase. In contrast, poly(I:C) + PLL complexes showed significant (P < 0.005) biological activity, indicating that PLL protected poly(I:C) from degradation.
Enhanced uptake of poly(I:C) in complexes with TfPLL
We aimed to develop a targeted INAs delivery system to enhance the uptake of INAs into mammalian cells and boost their innate immune response. The Tf/TfR receptor-mediated endocytosis seemed to be the system of choice, as the TfR expression is highly up-regulated, especially on tumor cells. To demonstrate the improved INAs internalization in complexes with TfPLL we used fluorescently-labelled poly(I:C) and HeLa cells. Rhodamine-labelled poly(I:C) (poly(I:C)_R) was complexed with TfPLL or PLL and added to growing cells. After 4 h incubation live cells were assayed for poly(I:C)_R internalization with confocal fluorescence microscopy. Results show that poly(I:C)_R alone was barely internalized in 4 h (Figure 4, top two panels). It had aggregated appearance and did not enter the lysosomes. Poly(I:C)_R premixed with PLL formed markedly increased number of aggregates which could not be washed of the cells, but also did not enter the lysosomes (Figure 4, middle two panels). Poly(I:C)_R complexed with TfPLL was successfully internalized and localized in lysosomes (Figure 4, lower two panels). We also observed its colocalization with anti-early endosome antigen 1 Abs (Supplementary Figure S4).
Confocal fluorescence microscopy showing improved internalization of poly(I:C) in complexes with TfPLL. Poly(I:C)_rhodamine (poly(I:C)_R) was premixed with PLL or TfPLL at a mass ratio 1:1.25 and 1:1 respectively. Mixtures were left to form complexes for 30 min at room temperature and added onto HeLa cells. The final concentration of poly(I:C)_R in the growth medium was 5 µg/ml. After 4 h of incubation internalization was detected using confocal scanning laser microscopy. Cell nuclei were stained with Hoechst 33258 (blue) and acidic compartments with LysoTrackerTM Green (green). Poly(I:C)_R is shown in red. The top two panels show micrographs of cells, which were given poly(I:C)_R only; the middle two panels are cells with poly(I:C)_R + PLL; the lower two panels are cells with poly(I:C)_R + TfPLL. White arrows indicate spots of co-localization between LysoTracker Green and poly(I:C)_R.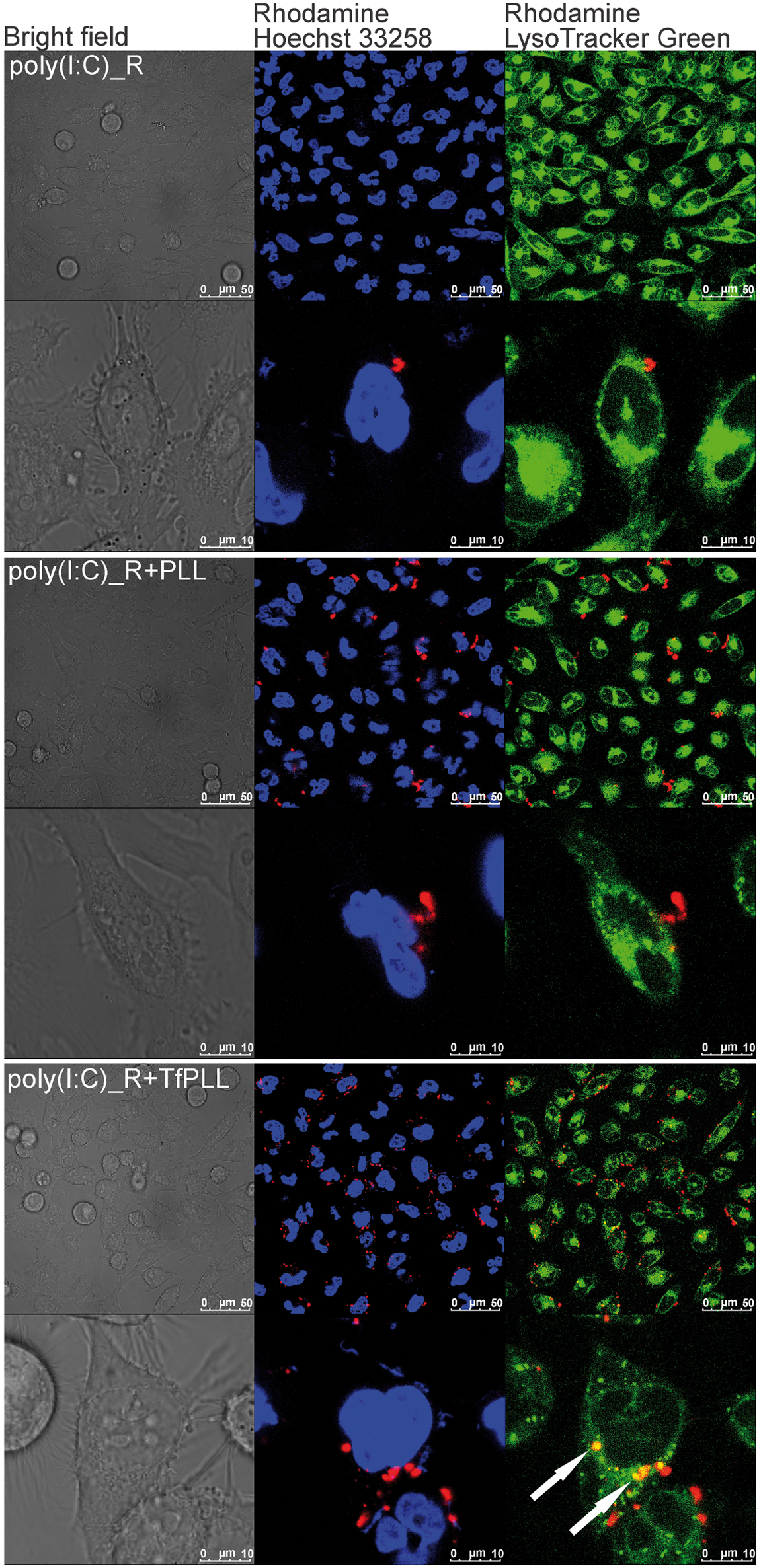
In addition to dsRNA, endosomal TLRs are also activated by ssDNA (TLR9). We wanted to test if the uptake of CpG ODN in HeLa cells was also more efficient when complexed with TfPLL. Internalization of fluorescently-labelled CpG ODN was considerably faster in complexes with TfPLL and was observed after only 4 h, whereas the CpG ODN control entered the cells only after overnight incubation (Supplementary Figure S5). In addition, the observed co-localization between CpG ODN and lysosomes indicated trafficking into the endosomal pathway. However, ODN 10104 + TfPLL complexes enhanced the activation of TLR9 transiently expressed in HEK 293T cells only by 20% in comparison with ODN 10104 alone or in complexes with PLL (data not shown). The reason for this may be the low level of TfR expression in HEK 293T cells or a significantly larger degree of polymerization state of poly(I:C) in comparison with ODNs, which can affect the size of complexes and internalization.
We also tested the internalization of poly(I:C) into HeLa cells with flow cytometry. Results show a considerably higher percentage of rhodamine-positive cells when poly(I:C) was complexed with TfPLL than with PLL or poly(I:C) alone (Figure 5A, B). Poly(I:C)_R complexed with PLL was internalized at a similar degree as poly(I:C)_R alone (Figure 5A, B).
Flow cytometry analysis of the uptake of poly(I:C)_rhodamine (poly(I:C)_R) alone, complexed with PLL and TfPLL. Poly(I:C)_R was premixed with PLL or TfPLL at a mass ratio 1:1.25 and 1:1 respectively. Mixtures were left to form complexes for 30 min at room temperature and added onto HeLa cells. The final concentration of poly(I:C)_R in the growth medium was 0.8 µg/ml. After 4 h of incubation cells were washed 5× with cold PBS in the growth vessel, detached with trypsinization, washed another 5× and resuspended in FACS buffer. Internalization of poly(I:C)_R was detected using flow cytometry. Unstained cells were used as negative control. (A) Flow chart showing percentage of rhodamine-positive cells. (B) Median fluorescence intensity of linearized data—the negative control was deducted from results.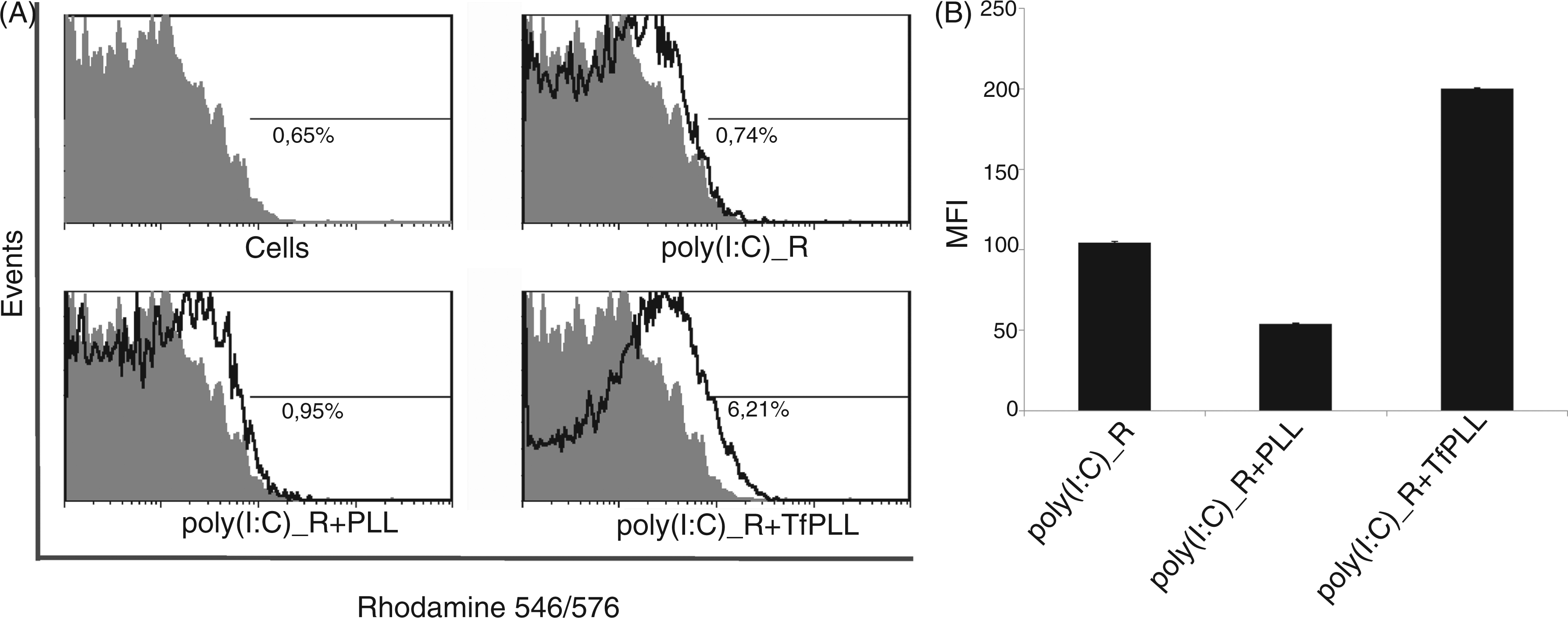
Enhanced uptake of poly(I:C)+TfPLL complexes enables rapid TLR3 activation
Results from confocal fluorescence microscopy demonstrated the increased uptake of INAs in complexes with TfPLL. In the following experiments we wanted to show the biological effect of the enhanced cellular uptake of poly(I:C) into primary HMVEC-dLy Ad cell line, which expresses TLR3.
4
We compared the stimulation with poly(I:C) or poly(I:C) in complexes with TfPLL or PLL. Stimulation for 3 h by poly(I:C) + TfPLL complexes and a subsequent washing step to remove non-internalized poly(I:C), elicited a strong immune response after 24 h, whereas poly(I:C) alone did not (Figure 6A). An increased cellular uptake of poly(I:C) + TfPLL complexes and, consequently, a higher IL-6 response was observed also at lower poly(I:C) concentrations (1 µg/ml) (Figure 6A). This result suggests the biological importance of an effective cellular uptake. The viability of HMVEC-dLy Ad cells was not affected by poly(I:C) + TfPLL stimulation (data not shown). We also excluded potential endotoxin contamination by the Limulus amebocyte lysate assay, as HMVEC-dLy Ad cells express TLR4 receptor and can state that endotoxin contamination was too low to overlap with TLR3 activation by poly(I:C) (data not shown). This was further demonstrated by stimulating HMVEC-dLy Ad cells with TfPLL and PLL at the highest concentrations used where no IL-6 production was observed (Figure 6B). Poly(I:C) + PLL complexes induced significantly lower IL-6 production than poly(I:C) + TfPLL which suggests that accelerated transport of poly(I:C) into the endosomal pathway was a result of Tf/TfR receptor-mediated endocytosis. This was further demonstrated by the competitive inhibition of the cellular uptake with free Tf (Figure 6C). Pre-incubation with free Tf for 15 min before stimulation with poly(I:C) + TfPLL significantly decreased IL-6 production.
The TfPLL conjugates enable effective cellular uptake of poly(I:C) resulting in a strong TLR3 activation. (A) HMVEC-dLy Ad cells were stimulated with different concentrations of poly(I:C) (0.5 µg/ml, 1 µg/ml and 5 µg/ml) and poly(I:C) in complexes with PLL or TfPLL. After 3 h cells were washed twice and fresh medium was added. The supernatants were collected 24 h after stimulation and human IL-6 protein levels were measured by ELISA. (B) To exclude potential endotoxin contamination, HMVEC-dLy Ad cells were also stimulated with TfPLL and PLL at the highest concentrations used. LPS at 50 ng/ml was used as a control. (C) Competitive inhibition of poly(I:C) + TfPLL complexes uptake with free Tf. HMVEC-dLy Ad cells were treated with 100 × molar excess of transferrin for 15 min and then stimulated with poly(I:C) (5 µg/ml) in complexes with TfPLL. After 24 h the supernatants were collected and human IL-6 protein levels were measured by ELISA. Inhibition with free Tf decreases the efficiency the Tf/TfR-mediated endocytosis [*P < 0.05, **P < 0.005 (Student’s t-test assuming unequal variances]).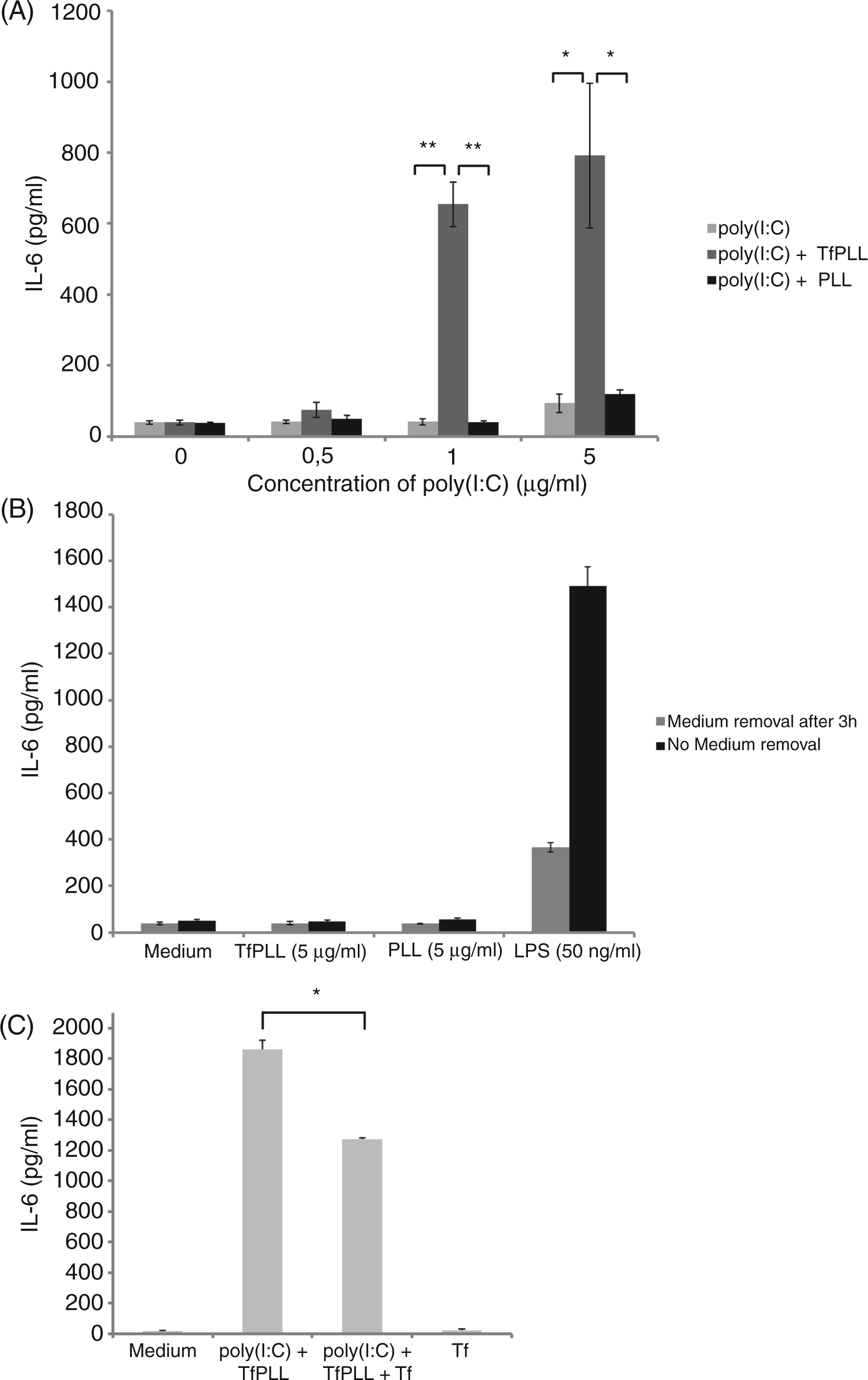
TfPLL conjugate facilitates a gradual release of poly(I:C) from complexes
The TfPLL conjugate is designed to bind, stabilize and effectively deliver INAs into the endosomal pathway. Poly(I:C) + TfPLL complexes have to dissociate or degrade proteolytically before gradually released poly(I:C) can activate TLR3.
We performed time monitoring of IL-6 production on primary HMVEC-dLy Ad cells (Figure 7). The results at the 12-h time point show the strongest activation by poly(I:C) alone (P < 0.05) whereas poly(I:C) + TfPLL or poly(I:C) + PLL are inefficient, probably because poly(I:C) is bound in complexes and, as such, unable to activate receptor TLR3. After 18 h we could already observe the enhanced effect of TfPLL in comparison with PLL. However, after 24 h or 36 h, poly(I:C) was already released from internalized poly(I:C) + TfPLL complexes and caused stronger activation of TLR3 receptor in comparison with poly(I:C) and poly(I:C) + PLL (P < 0.001), in particular (Figure 7). It is likely that a large fraction of complexes with PLL remained outside cells when TfPLL enabled an effective cellular uptake. These results suggest that TfPLL conjugate in comparison with PLL enabled faster internalization of poly(I:C) into cells; however, both of them caused delayed release of poly(I:C) from complexes in comparison with poly(I:C).
Gradual release of poly(I:C) from complexes with TfPLL and PLL. HMVEC-dLy cells were stimulated with poly(I:C) (5 µg/ml) and poly(I:C) in complexes with TfPLL or PLL. After 12 h, 18 h, 24 h and 36 h supernatants were collected and human IL-6 protein levels were measured by ELISA. Poly(I:C) is gradually released from the complexes which results in a delayed TLR3 activation in comparison with poly(I:C) alone [*P < 0.05, **P < 0.001 (Student’s t-test assuming unequal variances)].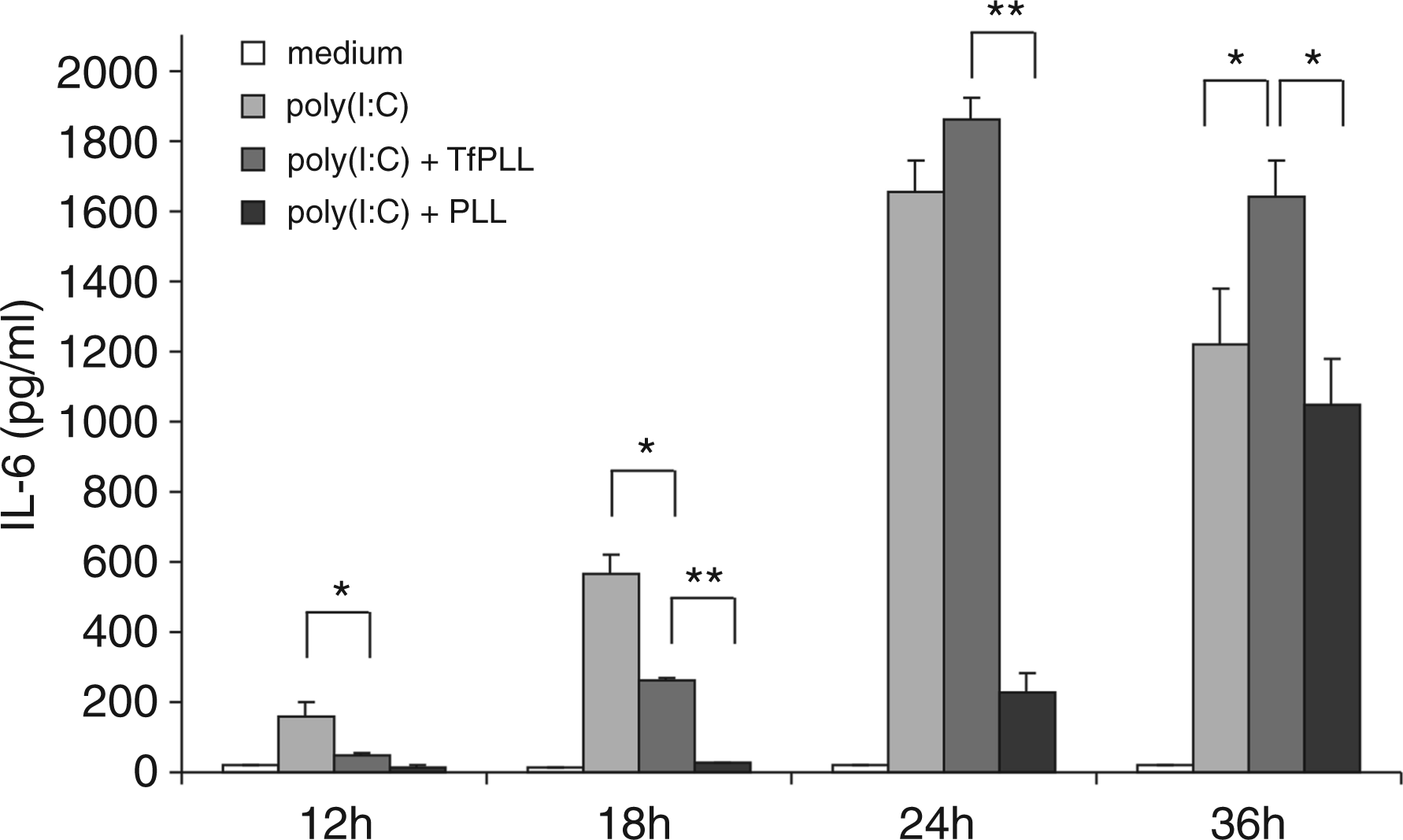
Discussion
In recent years, agonists of endosomal TLRs have shown great therapeutic potential as vaccine adjuvants and in the treatment of cancer, infectious diseases or allergies. 19 Several of these therapeutic approaches have already entered clinical trials and have shown promising results. 19 However, INAs and especially dsRNA and its synthetic analogues are sensitive to enzymatic degradation by serum nucleases. 28 As nucleic acids are hydrophilic polyanions, their entry into intracellular compartments across the cell membrane is rather inefficient, which limits their biological activity. 29 Whereas the structural basis of TLR3 activation by dsRNA has already been determined,30–32 the mechanism underlying the entry of ligand into the organelles containing TLR3 is not yet fully understood. Recently, clathrin-mediated endocytosis was proposed to be responsible for CpG DNA 33 and also dsRNA 34 uptake into cells. In order to trigger the activation of signalling pathways and subsequent immune response, INAs first have to be internalized into endosomes, where they bind their cognate TLR. 33 Therefore, it is important to develop a delivery system that provides protection to INAs and their targeted delivery into the endosomal compartments. 35
In the current study we focused on the development of a delivery system that would enable stabilization, targeted delivery, enhanced cellular uptake and gradual release of poly(I:C) which could then effectively activate innate and adaptive immune responses. We hypothesized that the conjugate between Tf and PLL could carry out these tasks. The physiological role of Tf is to mediate the cellular uptake of iron. A complex of serum Tf with bound iron is internalized by the well-characterized TfR-mediated endocytosis.
21
Tf as a ligand for this efficient receptor-mediated endocytotic pathway has been used for targeted drug delivery into malignant cells which overexpress TfR.
36
We conjugated Tf to PLL, a synthetic polycation of (A) Schematic representation of TfPLL conjugate (conjugate between transferrin and PLL) complexed with immunostimulatory nucleic acids (INAs). Such a complex enables targeted delivery of INAs (poly(I:C) or CpG ODN) into the desired cell type and their enhanced internalization for activation of innate immune response. (B) The proposed mechanism of INAs + TfPLL complex delivery and release. Transferrin/transferrin receptor-mediated endocytosis facilitates efficient uptake of INAs into endocytotic compartments where they activate endosomal TLRs.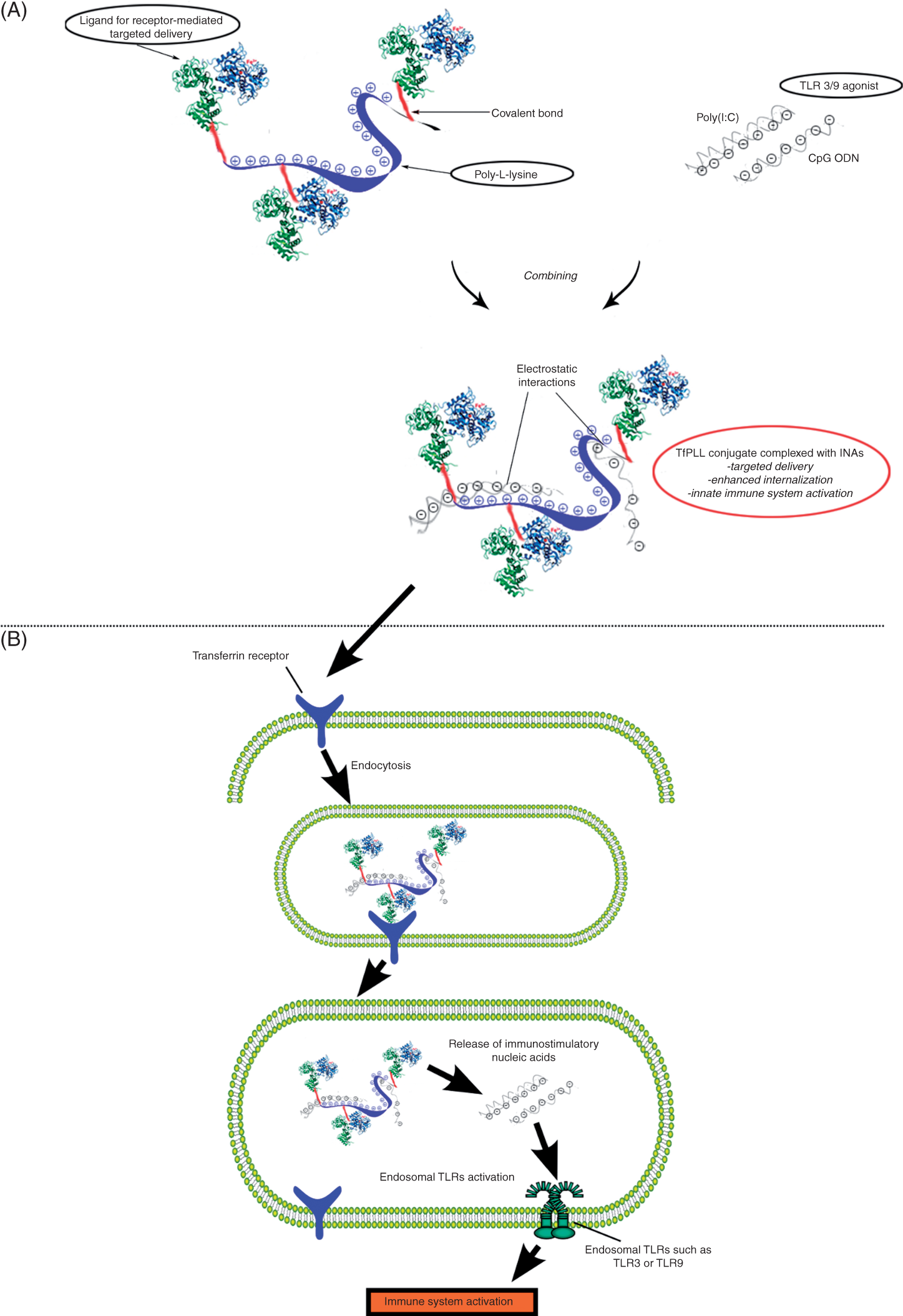
We demonstrated that the TfPLL conjugate successfully bound poly(I:C) as indicated by complete retardation of the complexes at a 1:1 mass ratio between poly(I:C) and TfPLL in a gel-mobility shift assay (Figure 2). Nucleic acids are rapidly degraded by nucleases and protection of ODNs against nucleases has been achieved by the introduction of phosphothioate bonds to CpG ODNs. Immunostimulatory dsRNA, such as poly(I:C), has, however, not been generally modified, despite greater sensitivity against nucleases. The stabilization of poly(I:C) against enzymatic degradation by the RNase was strongly improved by PLL. As shown in Figure 3A, at a mass ratio 1:1 between poly(I:C) and PLL, no degradation was observed. This is consistent with previous studies where an improved stability of plasmid DNA complexed with PLL during DNase I treatment was shown. 23 The improved stability of poly(I:C) was also demonstrated by the RNase resistance of TLR3 activation by poly(I:C) + PLL complexes (Figure 3B). We conclude that stabilization is based on the compaction of the structure of poly(I:C) in complexes which makes it unavailable for the RNase.
As Tf/TfR endocytosis is an effective and rapid process 39 we anticipated that poly(I:C) in complexes with TfPLL would enter HeLa cells which overexpress TfR much faster in comparison with poly(I:C) alone. To test these assumptions we compared the amount of internalized rhodamine-labelled poly(I:C) either alone or in complexes with Tf or PLL after a 4-h time period. Results shown in Figure 4 (lower 2 panels) demonstrate an accelerated uptake of poly(I:C)_R + TfPLL complexes. Similar enhancement of INAs uptake was also observed with CpG ODN + TfPLL conjugates (Supplementary Figure S5). Although PLL neutralizes negative charges of poly(I:C), it did not by itself provide an enhanced cellular uptake (Figure 4, middle 2 panels). PLL formed large aggregates with poly(I:C) which were not internalized and rather remained adhered to the cell surface even after washing steps.
Next, we wanted to investigate whether enhanced uptake of poly(I:C) results in an enhanced TLR3 activation after short time stimulation. Indeed, results shown in Figure 6A demonstrate that only 3 h of stimulation with poly(I:C) + TfPLL complexes, even at low concentrations, resulted in a strong TLR3 activation in contrast to poly(I:C) + PLL or poly(I:C) alone. Activation of HMVEC-dLy Ad cells with poly(I:C) + TfPLL was even higher than stimulation with 50 ng/ml of LPS in the same experiment with medium removal (Figure 6B).
TLR3 is also expressed at the cell surface of HMVEC-dLy Ad cells. 4 However, TLR3 needs to be internalized in order to trigger TRIF-dependent activation; therefore, in HMVEC-dLy Ad cells the activation was mainly the result of engaging endosomal TLR3.
We demonstrated that poly(I:C) should be released from TfPLL to activate TLR3 (Figure 7). In comparison with PLL, the TfPLL conjugate enabled faster internalization of a greater amount of poly(I:C). However, although TfPLL enabled rapid internalization of complexed poly(I:C), the latter was not able to activate TLR3, unless gradually desorbed from the complex with TfPLL. This could be the reason why an overall effect on TLR3 activation surpassed poly(I:C) only after 24 h and 36 h of stimulation. In comparison with TfPLL conjugate, PLL does not facilitate a rapid internalization of poly(I:C) but at the same time it sequesters poly(I:C), which results in an inhibitory effect of PLL on TLR3 activation by poly(I:C).
Lande et al. 40 reported that cationic antimicrobial peptide LL37 binds endogenous DNA resulting in a formation of aggregates which are delivered and retained within early endocytotic compartments of plasmacytoid dendritic cells where they trigger TLR9 signalling. This highlights the importance of efficient delivery and proper cellular localization of INAs. Additionally, delivery of INAs using lipofection also enhanced the immunostimulatory potency of INAs, 41 although this approach may not be feasible therapeutically. Our findings suggest that poly(I:C) + TfPLL complexes are rapidly internalized and effectively routed into the endosomal pathway where poly(I:C) is gradually released from the complexes, leading to endosomal TLR3 activation (schematically shown in Figure 8B). Poly(I:C) + TfPLL complexes could also activate cytoplasmic dsRNA receptors, such as MDA5, which could additionally contribute to the immunostimulation. It might be interesting to investigate the endosomal TLR activation by INAs complexed to a conjugate having weaker interactions with INAs [e.g. above-mentioned LL37, high-mobility group box protein 1 (HMGB-1) 42 or other weakly cationic molecule]. This modification may result in a decreased delay of receptor activation.
Activating both MyD88 and TRIF parallel branches of TLR signalling pathways has a beneficial effect on the quality and quantity of the elicited immune response. 43 The presented TfPLL conjugate could easily be implemented for such a purpose using a mixture of poly(I:C) (TLR3 ligand, activating TRIF pathway) and CpG ODN (TLR9 ligand, activating MyD88 pathway) (schematically shown in Figure 8A). Such complexes could be particularly useful for the treatment of cancer, as Tf provides cancer-targeting whereas TLR3/9 agonists confer immune remediation by breaking T-cell tolerance and eliciting a strong response against tumor-associated antigens. A similar principle combining cationic liposomes and TLR3/9 agonists has already been shown to elicit an effective T-cell immune response against antigens. 14 The receptor ligand-TLR ligand scaffold provides a platform for extension to the design of efficient vaccines by adding an antigen of interest to a TfPLL conjugate. A physical connection between the antigen and TLR ligand is also consistent with recent findings that rather than mixing TLR agonists and antigens, physical linking is favourable to ensure delivery into the same phagosome, which is important for proper antigen processing, MHC class II presentation and the subsequent immune response.44–46
In addition to the enhancement of the immunostimulatory effect of nucleic acids, we believe that the TfPLL conjugate could be therapeutically useful as a delivery system for antagonists of endosomal TLRs for the treatment of autoimmune and allergy diseases.
Conclusions
Receptor-mediated delivery of TLR ligands is a promising strategy to improve their therapeutic immunostimulatory properties. Here, we have shown that the TfPLL conjugate represents an attractive platform for the delivery of INAs, such as poly(I:C) or CpG ODNs. This provides the INAs protection against nuclease degradation, enhanced uptake, and strong and rapid activation of endosomal TLRs as receptors of innate immune response. This type of complexes have potential use in the development of advanced vaccine adjuvants and for immunotherapy, particularly in cells that express higher levels of TfR, such as cancer cells.
Footnotes
Funding
We thank the Slovenian Research Agency, Centre of excellence EN-FIST and ICGEB Grant for the financial support.
Acknowledgements
We thank Mateja Manček Keber for her help, Robert Bremšak for assistance with the TfPLL conjugate isolation, and Darija Oven and Irena Škraba for technical support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
