Abstract
We hypothesized that development of cervical cancer is associated with alterations in the expression of innate immune receptors, i.e. integrins and TLRs, and that these alterations can be induced by infectious agents. We have studied the expression of these proteins in cervical biopsy tissues and cervical cancer-derived cell lines HeLa, CaSki, SiHa, C-33 A, and ME180. Immunohistochemistry analysis demonstrated an increase in integrin αv, β3, β4, and β6 expression in the epithelium during the development of cervical cancer. A clear trend towards higher expression of integrin β6 in cell lines harbouring human papillomavirus (HPV) genetic material, compared to HPV-negative C-33 A, was observed. To investigate whether bacterial infection can alter the expression of TLRs and integrins, we infected HeLa cells by two pathogens, Escherichia coli and Pseudomonas aeruginosa, using a common bacterium of the female genital tract, Lactobacillus reuteri, as a control. Infection with E. coli or P. aeruginosa, but not with L. reuteri, significantly altered the expression of TLR and integrins, particularly of TLR4 and integrin β6. Considering that both integrin β6 and TLR4 play important roles in tumorigenesis, our data suggest that bacterial infection may trigger cancer development in HPV-infected cervical epithelium.
Introduction
Cervical cancer is the second most common malignancy in women world-wide and the human papillomavirus (HPV) is recognized as its causative agent.1,2 However, as HPV-induced carcinogenesis is highly complex, its underlying molecular mechanisms are not completely understood. Although persistent HPV infection is a necessary element for the development of cervical cancer, it is insufficient. In contrast to other pathogens involved in carcinogenesis, HPV per se does not cause inflammation. However, other infectious agents, i.e. common bacterial, viral, or protozoan pathogens in the female genital tract cause mucosal inflammation and such infections are often associated with cervical cancer. 3 Some evidence suggests that bacterial vaginosis, characterized by a shift in normal vaginal flora toward the prevalence of anaerobes, such as Gardnerella vaginalis, may play an important role in cervical carcinogenesis. 3 Case-control studies have indicated that Chlamydia trachomatis infection increases the risk of development of cervical squamous cell carcinoma. 4 Although such observations implicate an association between an infection-induced inflammatory process in the female genital tract and the development of cervical cancer, the underlying biological mechanisms are poorly understood.
During the last decade, a causal relationship between some infectious agents, inflammation, and cancer development has been recognized. It has been established that inflammatory processes are directly responsible for tumour formation and progression in carcinomas induced by hepatitis B virus (liver cancer) and Helicobacter pylori (stomach cancer).5,6 Some of the mechanisms underlying the interrelations between infection, inflammation, and cancer may be mediated via the activation of innate immune receptors recognizing pathogen-associated molecules. In particular, the activation of TLRs, which is essential for the initiation of innate and adaptive immune responses to microbial pathogens, may provide signals for tumour cell proliferation and resistance to apoptosis. 7
Integrins are a large family of αβ heterodimeric transmembrane adhesion receptors that mediate cellular interactions with the extracellular matrix (ECM) and other cells in the micro-environment. Following ligand recognition, integrins undergo clustering and conformational changes followed by activation of several signalling cascades; the resulting intracellular signalling regulates vital cellular functions, such as proliferation, differentiation, migration, cytokine release, etc. 8 The involvement of integrin receptors in the development and progression of many cancers has been recognized. 9 For example, the overexpression of some integrin subunits is correlated with an increased tumour invasion and metastasis. It is also associated with poor prognosis because of an enhanced tumour cell adhesion to the ECM, anti-apoptotic effect, activation of TGF-β, enhanced signalling from the epidermal-growth-factor receptor and other receptor tyrosine kinases, and activation of metalloproteinases (MMP). 10 Based on the importance of integrins in tumorigenesis, these molecules have been extensively explored as potential therapeutic targets in some cancers, and several integrin-targeting agents are currently in clinical trials. 11
While the overexpression of αvβ6 and β3 integrins has been found to have a prognostic impact on cervical cancer outcomes, some studies have suggested that down-regulation, rather than up-regulation of integrins, i.e. α5β1 α2β1 is involved in cervical cancer pathogenesis. 12 – 14 The mechanisms behind the changes in integrin expression during cancer development remain unclear, as does the role of TLRs. Although a recent study suggested the involvement of TLR2 and TLR4 gene polymorphisms in cervical cancer susceptibility, 15 the published data on the expression of TLRs in cervical cancer remain controversial.16,17
We hypothesize that inflammation induced by an infectious process in the female genital tract can up-regulate the expression of integrins and TLRs, and the resulting alterations in intracellular signalling mechanisms may promote malignant transformation of HPV-infected cells and the progression of cervical carcinoma. To clarify the role of TLRs and integrins in cervical cancer, we have studied the expression of these molecules in cervical biopsy tissues, covering the full spectrum of cervical disease, as well as in several cervical cancer-derived cell lines. To address the question of whether inflammatory responses can trigger cervical cancer development via effect on TLR and integrin expression we have used an in vitro model of infection with two bacterial pathogens, Escherichia coli and Pseudomonas aeruginosa.
Materials and methods
Human tissue
Cervical biopsies were obtained with informed consent from 17 women attending the Colposcopy Clinic at the Thunder Bay Regional Health Science Centre (TBRHSC) in 2006–2007. A portion of each biopsy sample was examined and graded by a pathologist as normal, low-grade squamous intra-epithelial lesion (LSIL), high-grade squamous intra-epithelial lesion (HSIL), or carcinoma. Five of the samples were considered normal, five were LSIL, three were HSIL, and four samples were carcinomas. All biopsies contained both epithelium and stroma compartments. The study has been approved by the TBRHSC Research Ethics Board.
Cell lines
The cervical cancer-derived cell lines HeLa, CaSki, SiHa, C-33A, and ME180 were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA). Frozen stocks of cells were stored in liquid nitrogen until thawed for culturing. The cells were maintained at 37°C, 5% CO2 in 75-cm2 flasks containing Dulbecco’s Modified Eagle’s Medium (DMEM; Sigma-Aldrich, Oakville, ON, Canada) supplemented with 10% heat-inactivated fetal bovine serum (FBS, HyClone, Logan, UT, USA) and 100 U penicillin, 100 µg streptomycin, 0.25 µg amphotericin B per ml (antibiotic/antimycotic, AB/AM; Gibco, Grand Island, NY, USA). Cells were passaged every 2 d to sustain a 60–80% confluent monolayer. The cells used in experiments were passaged from 3 to a maximum of 7 times from originally purchased stocks.
Bacterial strains
Escherichia coli 25922 (ATCC catalogue #25922), P. aeruginosa PAK, 18 and Lactobacillius reuteri 23272 (ATCC catalogue #23272) were kindly provided by Dr Kam Leung (Lakehead University, Thunder Bay, ON, Canada). Dr R.J. Irvin (University of Alberta, Edmonton, AB, Canada) and Dr P. Forsythe (McMaster University, Hamilton, ON, Canada), respectively.
Preparation of bacteria for experiments
Escherichia coli and P. aeruginosa were grown in Luria–Burtani (LB) medium (Sigma-Aldrich, Steinheim, Switzerland) at 37°C for 16 h in a shaking incubator at 150 rpm. An aliquot of each culture (1 ml) was transferred to 20 ml of fresh LB medium and allowed to grow for an additional 1 h. Lactobacillus reuteri were grown in MRS medium (Sigma-Aldrich) in anaerobic conditions (GasPak100™ system; BD Diagnostic Systems, Sparks, MD, USA) at 37°C for 16 h. Indicator strips within the sealed anaerobic jar confirmed that the oxygen was displaced.
Each bacterial culture was centrifuged at 3500 g for 20 min at 4°C, and pellets were washed 3 times with 10 ml of cold, sterile PBS, pH 7.4. The final pellets were resuspended in PBS to achieve optical density at 600 nm (OD600) of 0.39–0.44. This corresponded to 2 × 108 CFU/ml for E. coli, 5 × 107 CFU/ml for L. reuteri, and 4 × 108 CFU/ml for P. aeruginosa as was determined through multiple drop plating assays.
Bacterial infection of HeLa cells
HeLa cells were trypsinized for a maximum of 5 min at 37°C (0.05% trypsin-EDTA; Invitrogen, Grand Island, NY, USA), washed with sterile PBS, counted using Vi-CELL™ XR viability analyzer (Beckman Coulter, Mississauga, ON, Canada), resuspended in culture medium and seeded at 2 × 106 cells per 75-cm2 flask 24 h prior to each experiment. Medium from the flasks was removed, and the cells were rinsed twice with 5 ml of sterile PBS. The appropriate volume of bacterial suspension calculated for the multiplicity of infection (MOI) of 50 : 1, or equal volume of PBS as a control, was added. After adding serum- and antibiotic/antimycotic-free DMEM to achieve a final volume of 15 ml, the flasks were incubated at 37°C, 5% CO2 for 4 h. Although CFU numbers corresponding to specific OD600 were determined during optimization of the assay, the MOI was confirmed in all the experiments with counting HeLa cells in control flasks and serial dilutions and drop plating of bacteria.
In some experiments, HeLa and ME180 cells were stimulated for 24 h with LPS from E. coli O111:B4 (catalogue #tlrl-pelps; InvivoGen, San Diego, CA, USA) at concentrations of 10 µg/ml and 100 µg/ml. Cells were seeded 24 h prior to treatment and washed twice with PBS. During the treatment period, medium was changed from DMEM supplemented with 10% FBS and 1 × AB/AM to DMEM supplemented with 5% FBS and 1 × AB/AM. Following stimulation, cells were prepared for flow cytometry.
Flow cytometry analysis
Following bacterial infection or LPS stimulation, the cells were washed three times with PBS, trypsinized, centrifuged at 500 g for 6 min at 4°C and resuspended in PBS to proceed with immunostaining and flow cytometry analysis. For analysis of unstimulated cells, all five cell lines were seeded 24 h prior to each experiment, then medium was removed from each flask, and cell monolayers were washed twice with PBS. Cells were trypsinized and washed as described above. Cell counting was performed with the Vi-CELL™ XR.
For immunostaining, the cell pellets were resuspended in PBS containing 0.1% BSA (Sigma); 100 μl of cell suspension containing 1 × 105 cells, was incubated with 10 µg/ml of PE-conjugated mAb against integrin αv (mouse IgG1, clone 13C2; Southern Biotech, Birmingham, AL, USA), α3 (mouse IgG1,κ, clone C3 II.1, BD Pharmingen), α5 (mouse IgG1,κ, clone IIA1, BD Pharmingen), α6 (rat IgG2a,κ, clone GoH3, BD Pharmingen), β1 (mouse IgG1,κ, clone 12G10, AbD Serotec, Raleigh, NC), β3 (mouse IgG1,κ, clone VI-PL2, BD Pharmingen), β4 (mouse IgG1, clone 450-9D, BD Pharmingen), TLR4 (mouse IgG2a,κ, clone HTA125, eBiosciences), or unconjugated mAb against β6 (mouse IgG, clone 442.5C4, Calbiochem), TLR3 (mouse IgG1,κ, clone TLR3.7; Santa Cruz Biotechnology, Santa Cruz, CA, USA), TLR9 (mouse IgG2a; clone 5G5, Santa Cruz) at 4°C for 1 h in the dark. Following immunostaining with unconjugated antibodies, cells were washed twice with PBS and incubated with either R-PE or FITC-conjugated anti-mouse IgG at 4°C for 1 h. Mouse IgG1,κ, mouse IgG (H + L), and rat IgG2a were used as isotype controls. For detection of intracellular TLR3, TLR4, and TLR9, prior to immunostaining, cells were treated with the permeabilization buffer (BD Biosciences, San Jose, CA, USA) for 10 min at 20–22°C according to the manufacturer’s protocol. After washing, the cells were resuspended in PBS and analyzed by flow cytometry using a FACSCalibur with CELLQuest Pro software (BD Biosciences). For each test, 10,000 gated events have been analyzed. The results were expressed as relative mean fluorescence intensity (MFI) normalized to the corresponding isotype control: Relative MFI = [ln(Antibody geo mean) – ln(Isotype control geo mean)].
Isolation of RNA and real-time PCR analysis
Isolation of RNA and quantitative real-time PCR (qRT-PCR) was conducted as previously described. 19 In brief, all cells were trypsinized, centrifuged at 150 g for 5 min at 4°C and washed twice with PBS. Cell pellets were then frozen and stored at −80°C (maximum 48 h) until RNA extraction. The RNA extraction was done using the RNAqueous®-4PCR kit (AM1914; Ambion Inc., Austin, TX, USA), following the recommended protocol for mammalian cells. The RNA quantity and integrity was determined by loading RNA into standard-sensitivity chips for the Bio-Rad Experion Automated Electrophoresis System (Bio-Rad, Hercules, CA, USA). In addition to calculated 28 s/18 s ratio, electrophoregram and digital gel images were considered to assess RNA integrity. RNA isolated from samples was reverse transcribed at 4 ng/µl to complementary DNA (cDNA) using the High Capacity cDNA Archive Kit (Applied Biosystems, Foster City, CA, USA) according to manufacturer’s directions, with random hexamer primers. Complimentary DNA (50 ng) was then amplified using the TaqMan® PreAmp Master Mix Kit (Applied Biosystems). Gene amplification was analyzed for uniformity within each sample by comparing unamplified versus amplified gene expression between two genes using the ΔΔCt method as per the manufacturer’s recommendations (Applied Biosystems TaqMan® PreAmp Master Mix Kit Protocol), as previously described. 19 Reactions were carried out according to the qRT-PCR protocol specified in the TaqMan® PreAmp Master Mix Kit. Triplicate reactions of 25 μl volume were added to a 96-optical well plate (Applied Biosystems) and incubated at standard qRT-PCR conditions (50°C for 5 min, 95°C for 10 min and then cycled at 95°C for 15 s and 60°C for 1 min for 40 cycles [detection limit]). Target genes were normalized to the housekeeping gene hypoxanthine phosphoribosyltransferase1 (HPRT1), as previous results indicate its expression is unaffected by HPV infection. 19 TaqMan® gene expression on demand assays for HPRT1, TLRs 1–9 and integrins α3, α5, α6, αv, β1, β3, β4, and β5 (Applied Biosystems) were run according to the manufacturer’s protocol. Negative controls where cDNA was omitted or the enzyme was missing in the RT reaction were run to monitor for contamination or non-specific primer binding. A positive control was included on every plate to control for variation between runs. Relative quantification of target genes was performed using auto Ct and baseline settings and a threshold of 0.20 (Applied Biosystems 7300/7500/7500 Fast Real-Time PCR System Software). As CT values designate the number of amplification cycles required for detection of the fluorescent signal above a threshold level, a higher CT values indicate lower amounts of starting template (cDNA).
Immunohistochemical analysis of cervical tissue biopsies
Tissue biopsy samples were immediately snap frozen in liquid nitrogen and embedded in Tissue Tek™ embedding medium (O.C.T. Compound; Sakura Finetek, Torrance, CA, USA). Tissues were kept for long-term storage at −80°C. Serial sections (7 µm thick) were cut in a Leica CM1850 cryostat and mounted onto Fisherbrand Colorfrost®/Plus precleaned microsocope slides (Fisher Scientific, Pittsburgh, PA, USA). Slides with cut sections were stored at −20°C prior to staining.
Tissues were fixed immediately before staining by immersing the slides into methanol at −20°C for 10 min. Immunostaining was performed according to DakoCytomation EnVision+ Dual Link System-HRP (DAB+) protocol (DakoCytomation, Carpinteria, CA, USA). In brief, tissue sections were incubated in Dual Endogenous Enzyme Block for 10 min followed by 3 washes for 5 min each in PBS. Concentrations of primary antibodies were optimized; dilutions were done in Dako ChemMate antibody diluent (Dako, Denmark), i.e. 1 : 50 for all TLRs, 1 : 100 for integrins αv, β3, and β4, and 1 : 1000 for β6. Sections were incubated with primary antibodies in the dark at 4°C in a humid chamber for 16 h and thereafter washed with PBS as described above. One to two drops of Labelled Polymer-HRP was added to each section and incubated at 20–22°C for 30 min, followed by three PBS washes. Sections were then covered with 50 μl of DAB+ Substrate-Chromagen mixture and incubated for 10 min at 20–22°C. DAB+ Substrate-Chromagen mixture was removed, and sections were rinsed for 15 min in gently running reverse osmosis water. The slides were immediately transferred to the pathology laboratory at the TBRHSC for haematoxylin staining. Sections were automatically mounted with an acetate slip for protection and preservation. Sections designated as negative controls were treated in parallel with the other sections, but no primary antibodies were added.
Tissue sections were examined using a Nikon DXM light microscope with a Nikon Pan fluor ×10 objective. Analysis of staining of both epithelium and stroma was done blindly by two independent investigators and scored semiquantitatively as undetectable (−), weak (+), moderate (++), strong (+++), or very strong (++++), compared to negative control sections.
Statistical analysis
Effect of bacterial infection on integrin and TLR expression was assessed using two-tailed Student’s t-test and SPSS v16.0; all comparisons among five different cell lines were done with ANOVA followed by Tukey’s test using Prism Graph software. P-values for all statistical comparisons are reported with at most two significant digits. Statistical significance is stated when P < 0.05.
Results
Expression of integrins in cervical tissue biopsies
To investigate the expression of integrins during the development of cervical cancer, we performed immunohistochemistry analysis of αv, β3, β4, and β6 integrin subunits in normal and abnormal ex vivo cervical tissue. Seventeen frozen cervical tissue biopsies including five with low-grade dysplasia (LSIL), three with high-grade dysplasia (HSIL), four carcinomas and five normal samples, were examined.
Immunochemisty analysis of integrin and Toll-like receptor expression in cervical biopsy samples

N, normal; L, low-grade squamous intra-epithelial lesion; H, high–grade squamous intra-epithelial lesion; C, carcinoma.
Immunostaining: – negative, + weak, ++ moderate, +++ strong, ++++ very strong, NA: data not available.
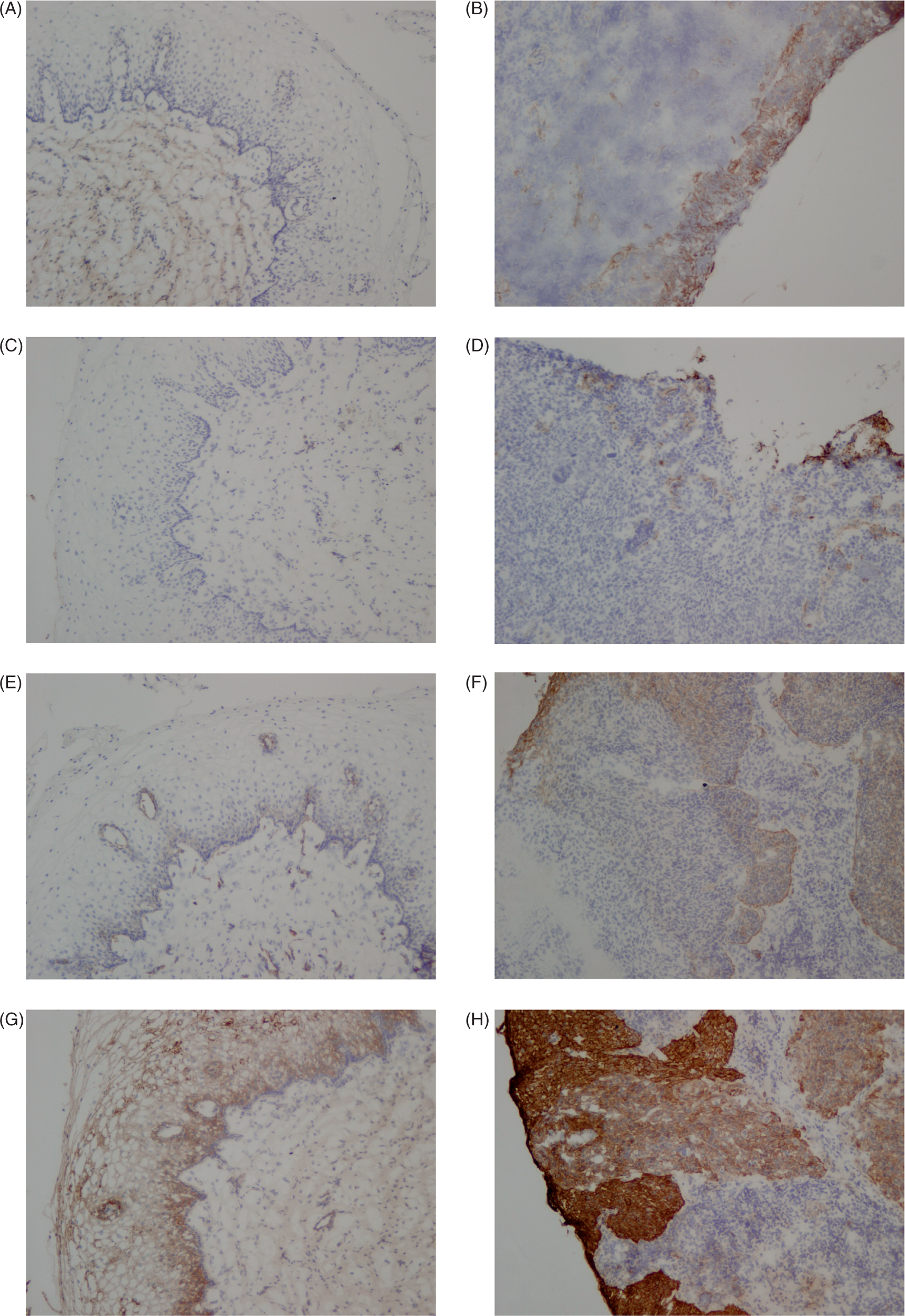
Immunohistochemistry analysis of integrin expression in cervical tissue biopsy samples. Frozen cervical tissue biopsy samples were processed and immunostained as described in Materials and methods. Representative sections of normal (A, C, E, G) and carcinoma (B, D, F, H) samples immunostained for integrin αv (A,B), β3 (C,D), β4 (E,F), and β6 (G,H) are shown (original magnification ×10).
Immunohistochemistry analysis revealed either undetectable or weak expression of TLR3 in both stroma and epithelium of the studied biopsy samples, without differences between normal and diseased tissues. Toll-like receptor 4 was undetectable in all, but one biopsy sample. As far as for TLR9, although this protein was detected in all samples, with an exception of the moderate expression in two carcinoma biopsies, its expression in other samples was weak (Table 1).
Hence, during the development of cervical cancer, there was a tendency towards an increased expression of some integrin subunits in the epithelium, i.e. of αv, β3, and β6, and appearance of β4 integrin in focal areas. However, no apparent changes in TLR3, TLR4, or TLR9 expression were detected.
Expression of integrins in cervical cancer-derived cell lines
As the HPV is a causative agent of cervical cancer, the observed differences in integrins αv, β3, β4, and β6 expression between carcinoma and normal tissue might depend on HPV infection. To address a question of possible relationship between HPV infection and the expression of integrins, we have examined four cervical cancer-derived cell lines, which are known to have different copy numbers and genotypes of HPV (HeLa, SiHa, CaSki, and ME180), and one cervical cancer-derived cell line negative for HPV genetic material (C-33 A, Product information sheet for HTB-31, ATCC). Among HPV-positive cell lines, SiHa contains 1–2 genome copies of HPV-16 per cell (Product information sheet for HTB-35, ATCC), CaSki contains 60–600 genome copies of HPV-16 and some HPV18 (Product information sheet for CRL-1550, ATCC), ME180 contains HPV-68 DNA, and HeLa contains 10–50 copies of HPV-18 genome equivalent. 20 – 22
Analysis of surface protein expression of various integrin subunits showed that all cell lines exhibited integrins α3, α5, α6, αv, β1, β4, and β6, although at different levels, but integrin β3 was present only in HeLa cells and undetectable or barely detectable in other cell lines (Figure 2). HeLa cells expressed higher levels of integrins β1, β3, αv, and α5 compared to the other cell lines (P < 0.05) and CaSki expressed the highest level of β6 integrin (P < 0.05). ME180 cells expressed the lowest levels of α5 and the highest levels of α6 and β4 integrins (P < 0.05). C-33 A cells showed a tendency to lower expression of most intergin subunits and had the lowest levels of α3, α6, and β1 subunits compared to all other cell lines (P < 0.05; Figure 2).
Flow cytomery analysis of surface expression of integrins α3, α5, α6, αv, β1, β3, β4, and β6, TLR3 and TLR4 in five cervical cancer-derived cell lines. Immunostaining followed by flow cytometry analyses of unstimulated, unpermeabilized cells was performed as described in Materials and methods; the results are shown as relative mean fluorescence intensity (MFI) of n = 3 independent experiments: Mean ± SEM; *P < 0.05 compared to other cell lines.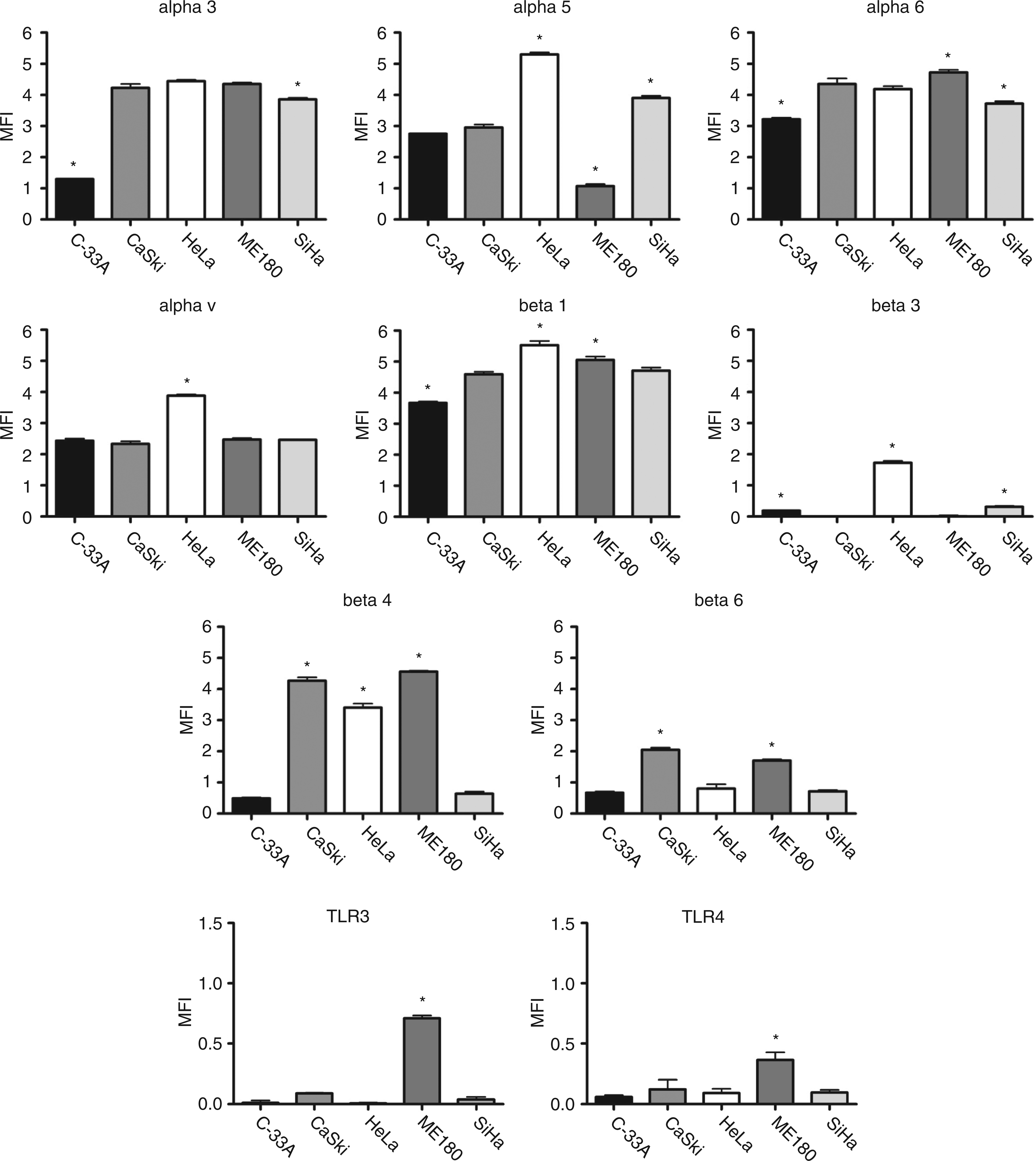
Hence, the expression of integrins in cervical cancer-derived cell lines containing different amounts of HPV genetic material significantly varies; this suggests a possibility that the surface expression of integrins can be up-regulated in cell lines containing HPV.
Expression of TLRs in cervical cancer-derived cell lines
Using qRT-PCR, we detected TLR1, 2, 5, 6, 7, and 9 mRNA in all studied cell lines and TLR3 mRNA in all cell lines except for C-33A (HPV-negative). Toll-like receptor 8 was absent from HeLa, SiHa, CaSki, and ME180 and present at low level in C-33 A (Figure 3). The largest variability in mRNA expression was detected for TLR4: this molecule was absent from ME180 and CaSki, but present at varying degrees in the remaining cell lines (Figure 3).
Gene expression of Toll-like receptors in five cervical cancer-derived cell lines. Isolation of RNA and real-time PCR was performed as described in Materials and methods; mRNA expression of TLR1–9 is shown as normalized CT values of n = 2 independent experiments; bars represent a mean ± range of two experiments; *not detected; **not detected in one out of two samples.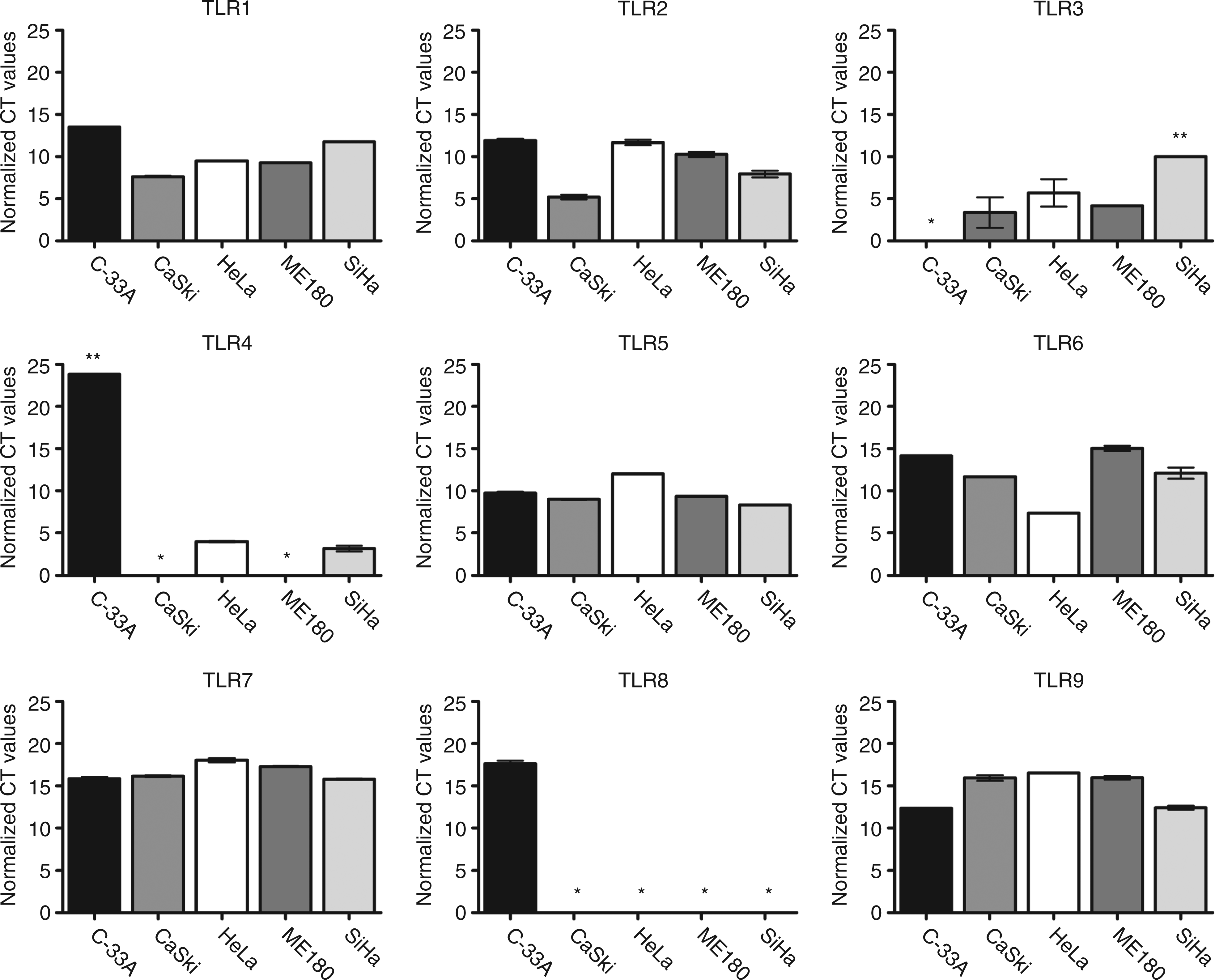
Despite the presence of TLR3 and TLR4 mRNA in most cell lines, the surface protein expression of these receptors was very low or undetectable in all studied cell lines (Figure 2). Although TLR9 mRNA was present in all cell lines, this molecule was not detected on the surface in any cell line (not shown). However, intracellular TLR3 and TLR9, but not TLR4 were detected in HeLa cells following the permeabilization of cell membranes (Figure 4) corroborating previous observations that the expression of TLR3 and TLR9 in cervical epithelial cells is almost exclusively intracellular.
23
Flow cytometry analysis of surface and intracellular expression of TLR3, TLR4, and TLR9 in HeLa cells. Unstimulated permeabilized or unpermeabilized HeLa cells were immunostained and analyzed by flow cytometry as described in Materials and methods; original histograms of one representative experiment are shown. Black filled histogram: isotype control, white line: unpermeabilized cells, grey line: permeabilized cells.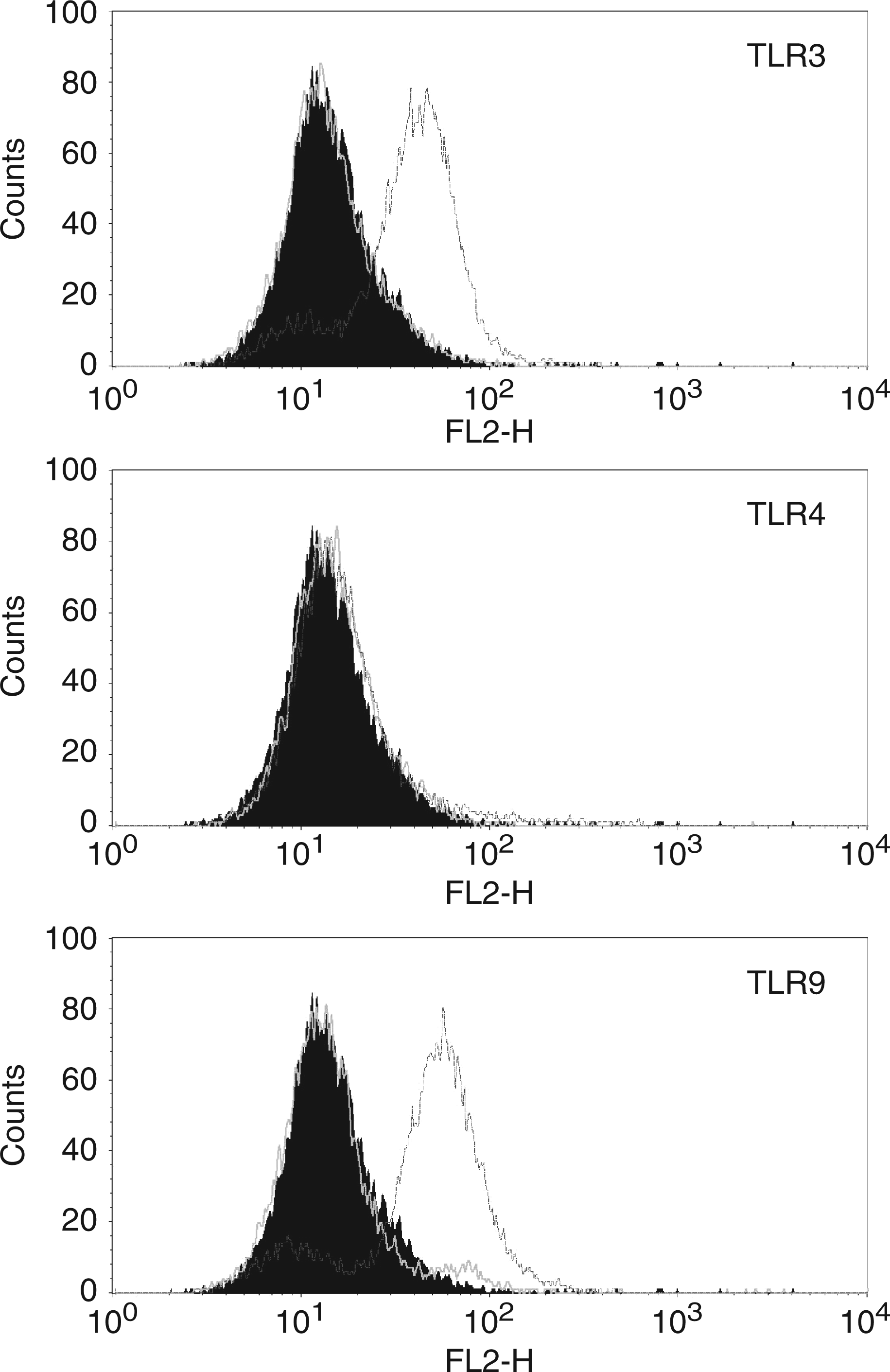
Effect of bacterial infection on the expression of integrins and TLRs
We hypothesized that the expression of integrins and TLRs in cervical epithelial cells can be altered as a result of bacterial infection. To test this hypothesis, we stimulated HeLa cells with either pathogenic Gram-negative bacteria E. coli and P. aeruginosa, or non-pathogenic Gram-positive L. reuteri. Expression of α3, α5, α6, αv, β1, β3, and β4 integrin subunits on HeLa cells significantly decreased after 4 h infection with E. coli or P. aeruginosa (Figs 5 and 6; P < 0.05). In contrast, expression of β6 integrin and TLR4 increased after infection with both pathogenic bacteria (Figs 5 and 6; P < 0.05). In addition, E. coli infection induced expression of TLR3 in HeLa cells (P < 0.05), although TLR3 was barely detectable on cell surface prior to the infection (Figure 5). However, no changes in any integrin, TLR3, or TLR4 expression were detected after 4 h infection with non-pathogenic L. reuteri, a commensal of the female genital tract (Figure 7).
Effect of infection with E. coli on the expression of integrins and TLRs in HeLa cells. HeLa cells were infected with E. coli during 4 h, then collected, immunostained and analyzed with flow cytometry to detect the surface expression of integrins α3, α5, α6, αv, β1, β3, β4, β6, TLR3 and TLR4 as described in Materials and methods; the results are shown as relative mean fluorescence intensity (MFI) of n = 3 independent experiments: Mean ± SEM; *P < 0.05.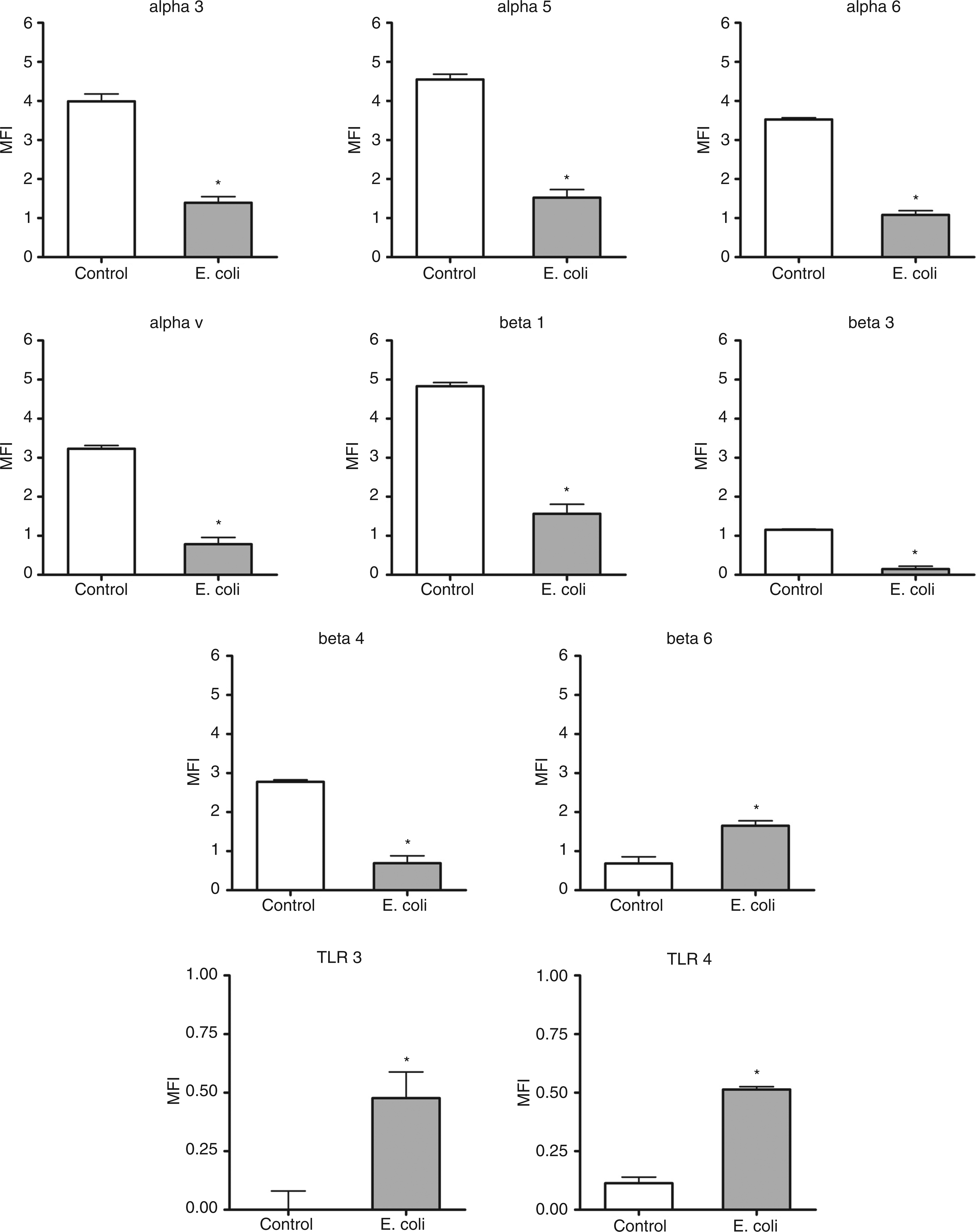
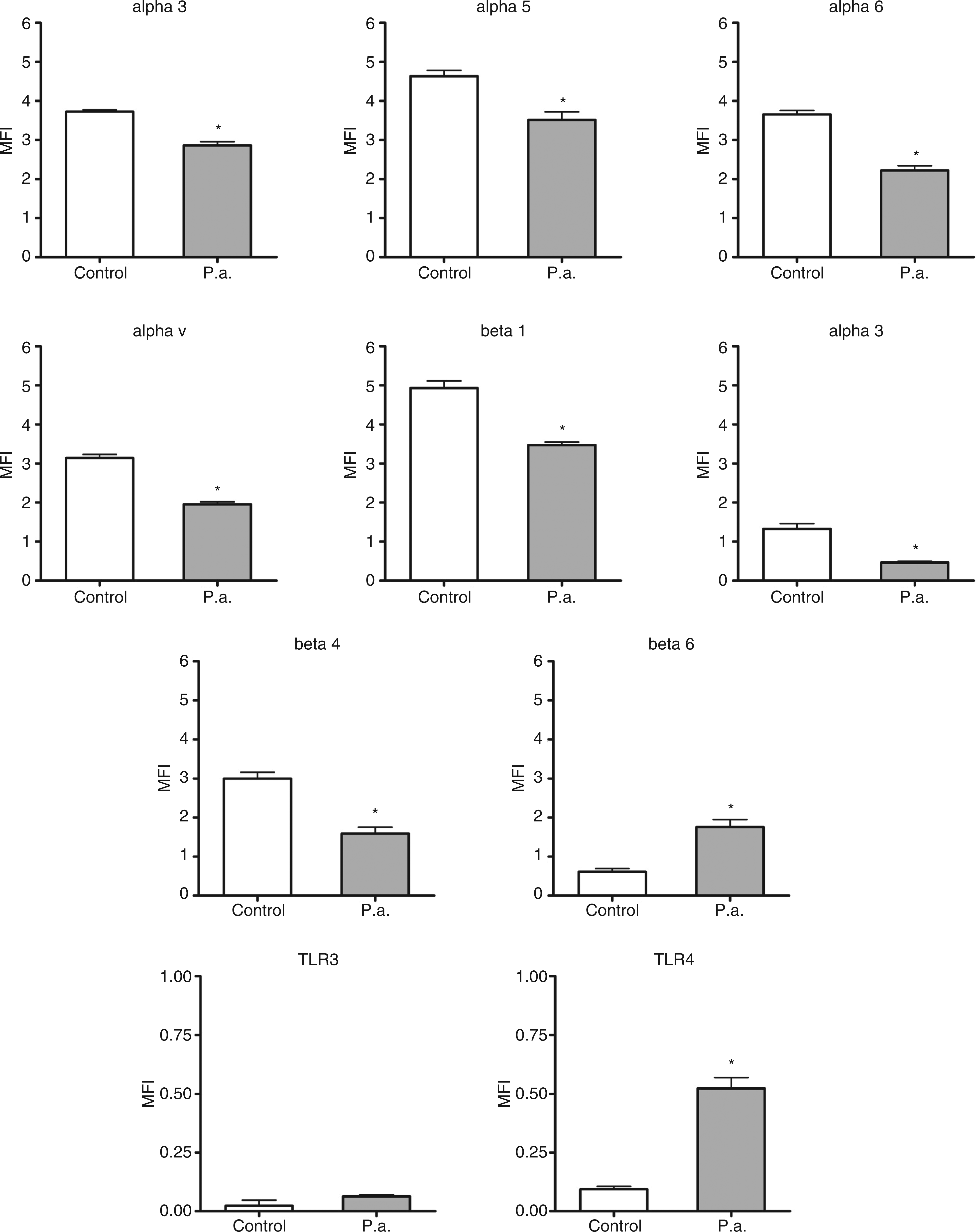
Effect of infection with P. aeruginosa on the expression of integrins and TLRs in HeLa cells. HeLa cells were infected with P. aeruginosa during 4 h, then collected, immunostained and analyzed with flow cytometry to detect the surface expression of integrins α3, α5, α6, αv, β1, β3, β4, β6, TLR3 and TLR4 as described in Materials and methods; the results are shown as relative mean fluorescence intensity (MFI) of n = 3 independent experiments: Mean ± SEM; *P < 0.05.
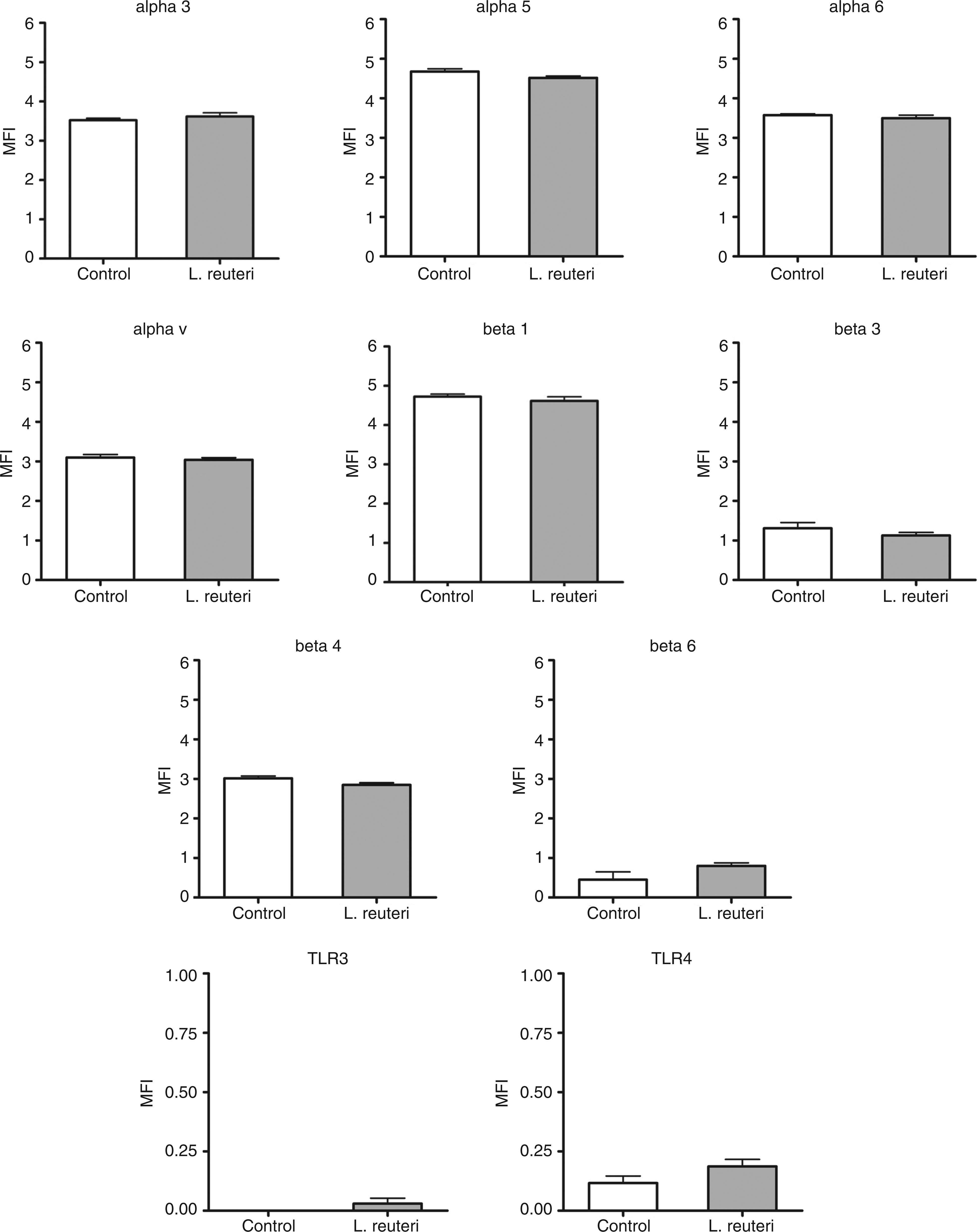
Effect of infection with L. reuteri on the expression of integrins and TLRs in HeLa cells. HeLa cells were infected with L. reuteri during 4 h, then collected, immunostained and analyzed with flow cytometry to detect the surface expression of integrins α3, α5, α6, αv, β1, β3, β4, β6, TLR3 and TLR4 as described in Materials and methods; the results are shown as relative mean fluorescence intensity (MFI) of n = 3 independent experiments: Mean ± SEM; *P < 0.05.
Since we observed the effect of Gram-negative, but not Gram-positive bacteria, on integrin and TLR expression, we tested if this effect was dependent on LPS, which is the major virulence factor of Gram-negative bacteria and a TLR4 ligand. However, stimulation of HeLa and ME180 cells with either 10 μ
Discussion
In this study, we hypothesized that the development of cervical cancer is associated with alterations in the expression of integrins and TLRs and that these alterations can be induced by pathogenic micro-organisms. Although HPV is recognized as a causative agent of cervical cancer, the mechanisms of malignant transformation of HPV-infected cells remain to be understood. Clinical and epidemiological data indicate that infection caused by Gram-negative bacteria, such as E. coli or C. trachomatis genital infection, can promote cervical cancer development.3,4 During the last decade, it became increasingly clear that the inflammatory micro-environment provides essential signals for the neoplastic development in various tissues. 24 However, the role of innate immune receptors, including TLRs and integrins, in the interface between inflammation and cancer, although intriguing, remains obscure. In this study, we have made several observations implicating the role of bacterial infection as a triggering factor in the development of cervical cancer.
To obtain insight into the clinical relevance of these observations, we have studied the expression of four integrin subunits previously shown to have prognostic significance in cervical cancer, i.e. αv, β3, β4, and β6. Among these integrins, β4 can combine only to α6 subunit, and overexpression of α6β4 integrin was detected in invasive and metastatic squamous-cell carcinomas. 10 Also, the expression of α6β4 integrin was recognized as an indicator of poor prognosis in head and neck cancer. 25 The αv subunit can combine with several β subunits, including β3 and β6, forming receptors for vitronectin (αvβ3, αvβ6), as well as fibronectin and tenascin (αvβ6). Overexpression of αvβ3, αvβ6, and of β3 subunit has been found in malignant cervical tumours and is associated with poor prognosis.12,13 An increased expression of αv, β3, β4, and β6 integrins in carcinomas compared to normal or dysplastic cervical biopsy tissues found in our study corroborates previous observations and suggests integrin involvement in the neoplastic progression.
Our data also suggest that the presence of HPV genetic material may up-regulate integrin expression in cervical cancer-derived cell lines. Although it is risky to generalize the data obtained on cell lines that exhibit a high genetic instability, we have observed a clear trend towards higher expression of integrins in cells harbouring greater amount of HPV genetic material, such as CaSki, compared to HPV-negative C-33A, e.g. of integrin β6 which is recognized as an indicator of cervical cancer progression and invasiveness. 13 In addition, HPV-positive cell lines exhibited higher expression of α6 and β4 subunits compared to HPV-negative cells. Considering that HPV can use α6β4 integrin bound to the ECM protein laminin 5, as a receptor, these data imply that HPV can potentially up-regulate its receptor on infected cells.26,27
Integrin involvement in tumorigenesis may be mediated by various pathways, e.g. by interactions between integrin-overexpressing cancer cells and the ECM proteins, such as fibronectin or vitronectin that facilitate cellular migration. 10 Overexpression of αvβ6 in cervical squamous carcinomas is an unfavourable prognostic factor. This might reflect an increased capacity of αvβ6-expressing tumour cells to migrate in a fibronectin-rich ECM and/or to activate TGF-β1 at the tumour/stroma interface, both of which processes may contribute to cervical cancer progression. 13 Likewise in colon and oral carcinomas, overexpression of αvβ6 may create an environment conducive to tumour progression.28,29 Moreover, integrin αvβ6 binds the latency-associated peptide of TGF-β1 and consequently activates this growth factor. 30 As a result, TGF-β signalling leads to the formation of Smad transcriptional complexes that regulate the expression of genes involved in cellular proliferation and differentiation. 31 Indeed, it has been established that TGF-β promotes progression of carcinomas, particularly in later stages of carcinogenesis. 31 Integrins like αvβ6 also regulate the activity of matrix metalloproteinases and via this mechanism may promote matrix degradation and hence alleviate spreading of cervical and other cancers. 32
Recent studies have described some specific integrin-mediated signalling pathways which are critically involved in tumorigenesis. One of these is the recruitment and phosphorylation of focal adhesion kinase (FAK), followed by activation of Src and c-Jun N-terminal kinase (JNK), as well as phosphorylation of phosphatidylinositol 3-kinase (PI3-K), leading to activation of protein kinase B (PKB)/Akt, Rac, and extracellular signal-regulated kinase (ERK). 33 As these signalling events control the cell cycle machinery, up-regulation of β6 integrin induced by bacterial infection can promote cancer cell proliferation and survival.
To investigate whether bacterial infection can contribute to cervical cancer development via altering the expression of innate immune receptors, we infected HeLa cells by two bacterial pathogens, E. coli and P. aeruginosa, using a common commensal bacterium of the female genital tract, L. reuteri, as a control. It is known that various bacteria use integrins as receptors during their adherence and invasion of susceptible cells. Moreover, pathogenic microbes can affect the expression of integrins via their transcriptional regulation or receptor binding followed by internalization. 34 However, to the best of our knowledge, the impact of E. coli, P. aeruginosa, and L. reuteri on the expression of integrins and TLRs in HeLa cells has not yet been investigated. Unexpectedly, both pathogenic bacteria greatly down-regulated the expression of all integrin subunits studied, with the exception of β6, which was up-regulated. Considering the role of the overexpression of β6 integrin in progression of cervical cancer as well as our findings of increased β6 integrin expression in ex vivo carcinoma samples, we suggest that bacterial infection may contribute to the neoplastic development of HPV-infected cervical keratinocytes, particularly via up-regulating integrin β6. 13
It is possible that the activation of pro-inflammatory signalling pathways initiated by the engagement of immune recognition receptors by microbial components can specifically induce transcriptional up-regulation of integrin β6. Indeed, we have found that the same infectious stimuli (E. coli and P. aeruginosa) caused up-regulation of TLR4, while E. coli infection also up-regulated TLR3. In contrast, stimulation with non-pathogenic and non-pro-inflammatory L. reuteri did not have any effect on integrins or TLRs. It has been demonstrated that LPS, or endotoxin of Gram-negative bacteria, which is the TLR4 ligand, can promote colon cancer development via the up-regulation of β1 integrin expression. 35 However, in our research, we did not find LPS to alter integrin expression either in cervical cancer-derived (this study) or in lung adenocarcinoma cell lines. 36 It is possible that, even if TLR4 is induced by bacterial infection, it may not be functional because of the lack of some essential components of the LPS receptor complex, such as CD14 or MD-2. Similar to our findings, some previous studies have demonstrated the lack of LPS-responsiveness in cervical keratinocytes, potentially related to low TLR expression and/or absence of CD14 or MD-2.37,38 Interestingly, the expression of TLR3, TLR4, and TLR9 at the protein level was barely detectable in cervical tissue biopsy samples and it did not differ between normal tissue and the samples obtained at different stages of cervical cancer development. Given that TLR protein expression in cervical cancer-derived cell lines was not detected in the absence of strong bacterial stimulation, these data suggest that the TLRs may be normally absent from uninfected cervical keratinocytes, similarly to some other epithelial cells. This may be biologically important because the absence of functional TLR signalling would prevent an excessive activation of pro-inflammatory signalling pathways in mucosal tissues, which are continuously exposed to various micro-organisms. As the expression of TLRs is strictly regulated, various pathogenic micro-organisms may have differential effects on TLRs. For example, the effect of HPV infection may depend on its specific type. Indeed, we have detected differential expression of various TLRs in cervical-cancer-derived cell lines harbouring different HPV genetic material. However, it is possible that cervical keratinocytes become responsive to LPS following bacterial infection as a result of the induction of TLR4 expression.
The regulation of integrin expression by infectious agents remains poorly understood. It is recognized that the transcriptional regulation of integrins is complex, cell-type specific, and controlled by several cytokines and growth factors, particularly TGF-β1. The TGF-β1-mediated signalling pathways have been found to be significantly involved in the regulation of several integrin subunits in lung fibroblasts and epithelial cells.39,40 Recent studies suggest that stimulation of TLR-mediated signalling by pathogen-associated molecules can activate TGF-β signalling pathways in the epithelium. These findings may explain how infection and inflammation affect integrin expression. 40
Up-regulation of TLRs induced by pathogenic micro-organisms, including those that can cause infections in female genital tract, has been previously demonstrated. For example, infection with Trichomonas vaginalis, a protozoan which infects the genito-urinary tract, was found to up-regulate TLR2, TLR4 and TLR9 gene expression in HeLa cells. 41 However, an increase in TLR4 expression caused by P. aeruginosa, or TLR3 and TLR4 caused by E. coli infection was not previously detected. Although the role of TLRs in tumorigenesis in general and in particular in cervical cancer development remains poorly understood, significant data support the idea that the up-regulation of some members of the TLR family, such as TLR4, can facilitate tumour evasion from immune surveillance. 42
Notably, in clear contrast to β6 integrin, the infection of HeLa cells with either E. coli or P. aeruginosa caused significant decrease in the surface expression of αv, β3, and β4 subunits. As endocytosis of α6β4 integrin was demonstrated in epithelial cells during wound healing, it is possible that a similar process can be induced in our model by cellular injury caused by the pathogenic bacteria. 43 In addition, integrin internalization was found in cells infected with adenoviruses which bind αvβ3 and αvβ5 integrins. 44 Whether infection with E. coli and P. aeruginosa may cause receptor internalization or steric hindrance is a puzzling issue and the subject of our on-going research.
Conclusions
The data suggest that the expression of integrins and TLRs in cervical epithelial cells can be affected by bacterial pathogens causing the inflammatory process in the female genital tract, and such mechanisms can potentially contribute to the neoplastic development of HPV-infected cells. In particular, we have found that bacterial infection can cause up-regulation of integrin β6, which has been found to be an important prognostic factor in cervical cancer. We have also observed up-regulation of β6 subunit expression in carcinoma biopsy tissues and in cervical cancer-derived cell lines harbouring HPV genetic material, i.e. CaSki and M180. We propose that up-regulation of integrin β6, TLR3 and TLR4 caused by bacterial infections, may contribute to cervical cancer development because these proteins can promote survival, proliferation, and immune evasion of cancer cells. Our findings emphasize the need to understand better the mechanisms underlying malignant transformation of epithelial cells under the influence of infections and inflammatory micro-environment. Since integrin αvβ6 is involved in migration, development and morphogenesis and typically absent from normal differentiated epithelia, unless associated with wound repair and inflammation, this protein may be considered as a potential therapeutic target in cervical neoplasia. 45
Footnotes
Acknowledgements
Ingeborg Zehbe and Marina Ulanova are joint senior authors. This work was supported by the Northern Cancer Research Foundation (Thunder Bay, ON) and a National Science and Engineering Research Council (NSERC) Discovery Grant. We are grateful to Dr Randal Irvin (University of Alberta, Edmonton, AB, Canada) for providing P. aeruginosa PAK strain, Dr Kam Leung (Lakehead University, Thunder Bay, ON) for providing E. coli 25922 and Dr P. Forsythe (McMaster University, Hamilton, ON) for providing L. reuteri 23272.
