Abstract
The functional phenotype of resident macrophages significantly determines the character of an inflammatory response. In this study we identified two phenotypes of tissue macrophages in bovine teat tissue based on expression of Calprotectin and CD163. To investigate a possible link between the dichotomy in phenotype and functional properties of cells in association with different host mediators we set up an in vitro model with bovine monocyte-derived macrophages (MdM). In vitro differentiated MdM invariably and uniformly expressed both antigens. Classically activated MdM (IFN-γ priming and LPS stimulation) showed a decreased CD163 expression while alternative activation (IL-4/IL-13 priming) did not change expression of CD163 and Calprotectin. Differently activated MdM showed a clearly distinct expression of genes related to classical (IL-12, inducible NO synthase) or alternative activation (IL-10, arginase I). The presence of the inflammatory host mediator prostaglandin E2 (PGE2) neither influenced expression of Calprotectin and CD163 nor gene expression profiles in MdM generated in the presence of PGE2 (PGE2-MdM). Supernatants of PGE2-MdM, however, significantly dampened the migration of neutrophilic granulocytes. The results of this study highlight the discrepancy between in vivo and in vitro obtained macrophages and point to the necessity to analyze the functional capacities of bovine tissue macrophages in situ.
Introduction
Functional polarization of tissue macrophages is a well-known phenomenon, which decisively influences the immune response towards invading pathogens, as well as the resolution of inflammation and tissue remodelling.1–3 Macrophage diversity is reflected by a continuous transition displaying a whole spectrum of different phenotypes ranging from classically activated, pro-inflammatory subtypes to alternatively activated macrophages.4,5 The presence of these different macrophage subtypes is the result of a distinct combination of host mediators and pathogen-associated molecular patterns (PAMPs) in their microenvironment.6,7 Priming with IFN-γ and stimulation with bacterial endotoxin leads to classical activation of cells resulting in the up-regulation of mainly pro-inflammatory chemokines and cytokines (CCL2, IL-1β, TNF-α, IL-12) and an increased production of reactive oxygen species (ROS), as well as inducible nitrogen synthase (iNOS). 6 Alternative activation by IL-4 and IL-13 induces a phenotype which is characterized by the production of arginase I (Arg I). The alternatively activated macrophage is not anti-inflammatory per se but comprises a diverse phenotype contributing to the immune response towards parasites, as well as tissue repair, but also with the ability to mount a pro-inflammatory reaction after TLR4 stimulation.3,8
There are only few studies describing the distribution and importance of different macrophage phenotypes for cattle. Flynn et al. 9 showed the development of an alternatively activated macrophage phenotype after exposure to IL-4 and excretory/secretory products of Fasciola hepatica displaying reduced iNOS and high Arg I production, as well as low IFN-γ levels, after stimulation with TLR2 and TLR4 ligands. Stijlemans et al. 10 describe the kinetics of classically and alternatively activated macrophages in the context of bovine African trypanosomiasis. They show that the persistence of classically activated macrophages induced by parasite-derived glycolipids contributes to anaemia while a switch to alternatively activated macrophages mediated by host-derived IL-10 reduced pathological signs, including anaemia. The influence of local host mediators apart from cytokines during differentiation of monocytes into macrophages is scarcely investigated. We recently showed a modulation of the in vitro LPS responsiveness of bovine monocyte-derived macrophages (MdM) differentiated in the presence of the neuromediator Substance P. 11
Mastitis is a major problem in dairy cows. The performance of the host immune system seems to play a key role during the infection, particularly with Gram- negative pathogens. 12 Differentially activated macrophages in the bovine udder could play a central role in governing the immune responses in the onset and the resolution of mastitis. The aim of the present study was, therefore, to characterize functional properties of macrophages in the context of classical and alternative activation and a host-derived inflammatory mediator in an in vitro differentiation model after identifying different macrophage phenotypes in teat tissue. We focused on the teat compartment as a defined site within the organ where an early contact between the host and the pathogen takes place. The calcium binding protein Calprotectin and the scavenger receptor CD163 have been associated with classically activated (Calprotectin) or alternatively activated macrophages (CD163) in tissue of different species, including cattle,13–15 although they have not, so far, been described in udder tissue. Whether such macrophage populations represent the result of a biased monocyte-to-macrophage differentiation or whether they represent the contact of macrophages with distinct host/pathogen molecules is not known either. To obtain a more detailed view on monocyte differentiation and polarization, the functional phenotype of differentially activated MdM, was studied in an in vitro set up combining polarization of macrophages with the presence of inflammatory mediators during their differentiation. We used prostaglandin E2 (PGE2) to exemplify a host-derived mediator known to increase the expression of IL-1β, TNF-α, IL-10, IL-23 and IL-6, and, therefore, favoring the development of Th17 cells by human dendritic cells in vivo. 16
We postulate that there is an impact of local host mediators on the monocyte-to-macrophage differentiation resulting in a specific functional macrophage phenotype encountering a specific combination of cytokines and PAMPs during the inflammatory response.
Material and methods
Bovine teat tissue
Teats from seven lactating Holstein Friesian cows between the age of four and five years old were obtained from the local abattoir during the routine slaughtering process. A general examination was performed and animals showing signs of clinical mastitis (heat, pain, redness and swelling of the udder) were not used for tissue sampling.
Tissue sample preparation
Udder tissue samples were collected within 5 to 10 min after slaughtering. Udder and teat tissue were macroscopically checked for prominent alterations. Mammary glands and teats showing signs of an inflammatory process or other morphological abnormalities were discarded. Teats were separated from the udder using a scalpel, rinsed three times in sterile PBS, pinned on a board and cut longitudinally through the teat canal to the upper limit of the teat cistern. For precise cutting, a cannula was inserted into the teat canal without perforating it. Afterwards, teats were cut into four quarters. Tissue of two quarters was fixed in 4% neutral buffered formalin according to Lillie and Fullmer 17 for 24 h. Tissue samples were subsequently dehydrated in a graded ethanol series and finally embedded in paraffin. Every tissue block included the proximal end of the teat canal, the Fürstenberg’s Rosette and the teat cistern for tissue analysis of macrophages. Two quarters of each teat were analyzed.
Immunohistochemistry
Markers for macrophage phenotypes were located in formalin-fixed and paraffin-embedded teat tissue sections (4 µm). Preparation of the sections included inactivation of endogenous peroxidase with H2O2 (30%, 30 min), pre-treatment with Pronase (0.1%, 30 or 15 min, respectively; Biologo, Kronshagen, Germany) and blocking with normal goat serum (20% in PBS, 20 min). Abs used for detection of macrophage phenotypes were mouse anti-human CD163 (10 µg/ml; TransGenic Inc., Kobe, Japan) and mouse anti-human Calprotectin (0.2 µg/ml; Biologo, Kronshagen, Germany). 18 Primary Abs were incubated overnight (18 h) at 4°C. The immunoreaction was visualized using EnVision+™ anti-mouse immunoglobulin conjugated to peroxidase-labeled dextran polymer (DAKO, Glostrup, Denmark) and DAB (Sigma-Aldrich, Steinheim, Germany). To check for unspecific binding, isotype-specific negative controls were performed by replacing the primary Abs by non-immune anti-mouse IgG (Sigma-Aldrich). Sections were counterstained with Delafield’s Hematoxylin and analyzed with a Zeiss Axiovert 200 M microscope (Zeiss, Oberkochen, Germany). Samples were histopathologically examined. Tissue showing leukocyte influx and damaged epithelia was discarded. Frequency of positively stained macrophages was graded by use of a semiquantitative scale between 0 and 3 [0 = no positive cells, 3 = great number of positive cells (in increments of 0.5)] according to the amount of stained cells. Five vision fields per slide (including subepithelial and underlying connective tissue, 200 × magnification) following the epithelial line on the slide were analyzed.
Cell separation
Blood from four healthy cows was obtained by puncture of the vena jugularis externa into heparinized vacutainer tubes (Becton Dickinson, Heidelberg, Germany). Blood was layered on Ficoll-Isopaque (PAA, Pasching, Austria) and centrifuged at 4°C for 30 min at 1000 g. The interphase containing mononuclear cells was washed three times in PBS (200 g, 100 g and 80 g, 4°C). Polymorphonuclear granulocytes (PMN) were obtained for migration assays from one healthy cow from the packed cells below the Ficoll-Isopaque. Erythrocytes were lysed with distilled water. This was repeated twice until complete erythrolysis. Subsequently PMN were washed three times in PBS (200 g, 100 g, 60 g) and finally re-suspended in RPMI 1640 (5 × 106 cells/ml). Mononuclear cells were re-suspended in 5 ml PBS containing 2 mmol/l EDTA and 0.5% BSA and filtered through a pre-separation filter (mesh size: 30 µm) (MiltenyiBiotec, BergischGladbach, Germany) to remove cell clumps. Cells were centrifuged at 70 g (10 min, 4°C) and labeled with a monoclonal Ab specific for CD14 coupled with paramagnetic beads (MiltenyiBiotec) (15 min, 6°C). After labeling, the cells were washed with PBS-EDTA (300 g, 10 min, 4°C). Subsequent magnetic separation with the Magnetic-activated cell sorting (MidiMACS) separation unit (MiltenyiBiotec) was conducted according to the manufacturer’s instructions. Positively selected cells (monocytes) were suspended in 5 ml PBS-EDTA and checked flow cytometrically for their viability (≥ 98%) and purity (≥ 95%).
In vitro culture of monocytes and MdM
The procedure to generate MdM was basically adapted from Jungi et al. 19 except that monocytes were used instead of blood mononuclear cells. MACS-separated monocytes were suspended in Iscové medium (PAA, Pasching, Austria) supplemented with 10% (v/v) FCS and 1% penicillin/streptomycin at 4 × 105 cells/ml. The cells were plated in 24-well plates (4 × 105/well) and incubated for 4 or 7 d (37°C, 5% CO2) in the absence (MdM) or presence of PGE2 (10-9 mol/l, PGE2-MdM; Sigma-Aldrich). All experiments were carried out in duplicates. Viability during differentiation was controlled flow-cytometrically using LIVE/DEAD® fixable red stain (Invitrogen, Karlsuhe, Germany) according to the manufacturer’s instructions and was found to range between 85% and 95%. There was no significant difference in the viability between MDM cultured in the presence or absence of PGE2.
Days 4–7 MdM/PGE2-MdM were detached from the 24-well plates by 5 min incubation with Accutase (PAA). Accutase reaction was stopped by adding media containing 10% FCS. After detaching, cells were centrifuged (100 × g, 5 min, 4°C), and CD163 and Calprotectin expression were detected by membrane and intracellular immune fluorescence (see below).
On d 6, MdM were primed with either IFN-γ (20 ng/ml, Kingfisher Biotech, St. Paul, MN, USA) or IL-4 and IL-13 (20 ng/ml each, Kingfisher Biotech). On d 7 MdM were stimulated with LPS (from Escherichia coli O55:B5, 1 µg/ml; Sigma-Aldrich) for 3 h or left as untreated controls. Subsequently, culture supernatants were collected for in vitro migration assays with neutrophilic granulocytes and total RNA was extracted for RT-qPCR analysis (see below).
Membrane and intracellular immune fluorescence
List of monoclonal Abs cross-reactive for bovine monocytes/macrophages used for flow cytometric analyses.

Cells were stained with LIVE/DEAD® fixable red stain (Invitrogen) according to the manufacturer’s instructions and subsequently incubated with anti-CD163PE for 20 min at 4°C and washed twice (200 g, 4 min, 4°C) with PBS-buffer containing 0.5% BSA and 0.01% sodium fluoride (Magnetic-activated cell sorting (MIF) buffer). Afterwards, cells were fixed with paraformaldehyde (4%) for 45 min at room temperature (21°C, RT) and washed twice with PBS (200 g, 10 min, RT). The fixed cells were incubated with anti-Calprotectin in MIF buffer containing 0.5% saponin for 30 min at 4°C. Washing steps were performed twice (200 g, 4 min, 4°C) in MIF saponin buffer with an incubation of 20 min at 4°C in between to allow unbound antibody to diffuse out of the fixed cells.
After labeling, cells were analyzed by flow cytometer. Viable monocytes and MdM were identified in forward/side scatter dot plots based on their characteristic scatter properties. Compared with MdM, monocytes were positioned lower in the side scatter and forward scatter as a result of their less complex surface and smaller size. Also, monocytes represent a more homogenous population. Viable cells were determined as LIVE/DEAD® fixable red negative events in the red fluorescence channel.
Migration assay
The chemotactic properties of the MdM supernatants from four cows were tested in a migration assay using PMN obtained from one healthy cow. Migration assays were performed in a 96-well transmigration chamber (Neuro Probe, Gaithersburg, MD, USA). As MdM supernatants contained 10% FCS, they were diluted 1:3 in Iscové medium without FCS to minimize the intrinsic chemotactic properties of FCS. Lower chamber wells were filled with 200 µl culture medium (Iscové, 3% FCS) or the diluted culture supernatants of in vitro generated MdM. To prevent adhesion of migrated PMN to the walls of the wells, 100 µl Percoll® was layered underneath and 200 µl of granulocyte suspension (1 × 106 cells) were placed into the upper well. Experiments were performed in duplicate. The upper and lower parts of the transmigration chamber were separated by a membrane with a pore size of 3 µm and 2 × 106 pores/cm2. Transmigration chambers were incubated in a humidified 5% CO2 atmosphere for 2 h at 37°C. After incubation, the cell suspensions were completely retrieved from the lower and upper compartment, transferred to separate flow cytometry tubes, and quantified flow cytometrically (C6 Flow Cytometer®, Accuri Cytometers, Cambs, UK) by the standard cell dilution assay. 20 Neutrophils were identified in forward/side scatter and green fluorescence/side scatter dot plots based on their characteristic scatter and autofluorescence profiles. The procedure was adapted for the bovine system with slight modifications as described previously. 21 For each determination at least 104 events were acquired.
RNA preparation and RT-qPCR
Primer sequences and concentrations used for real-time PCR analysis.

bp, length of amplicons in base pairs; mod., modified; Acc.no., Gene Bank accession number of nucleotide sequence used for primer generation.
Statistical analyses
Statistical analysis was performed with the statistical software package SAS (version 9.1, SAS Institute Inc., Cary, NC, USA). Data were checked for normal distribution with the Shapiro Wilk test. Differences between means were tested with one-factorial analysis of variance (ANOVA) for repeated measurements and Bonferroni’s correction for normally distributed data. P values of ≤ 0.05 were considered significant.
Results
Identification of morphologically distinct Calprotectin+ and CD163+ macrophages in the teat with biased distribution pattern
In all tested animals CD163, as well as Calprotectin, could be detected in tissue of the teat by immunohistochemistry. Single positive cells showed a distinct morphology. Macrophages expressing Calprotectin were round to spindle-shaped with a reniform nucleus (Figure 1A). In contrast, the CD163+ macrophages showed a much larger and more complex cell body with a round, centric nucleus and elongated cytoplasma extensions (Figure 1B). Most macrophages expressed the antigens selectively. Serial slides revealed the presence of few double positive cells (Figure 1C, D; arrow). The distribution pattern of macrophages selectively positive for CD163 or Calprotectin was biased (Figure 1C–F). In sub-epithelial locations of the teat more Calprotectin+ cells were detected (Figure C, D) while CD163+ cells were more abundant in perivascular locations (Figure 1E, F).
Immunolocalization of cells positive for Calprotectin (A, C, E) or CD163 (B, D, F) in bovine teat tissue. A, B: Detailed view (peripheral connective tissue). C, D: Serial sections (subepithelial location). E, F: Serial slides (perivascular location). Brown: immune reaction; blue: counterstain with Delafield's Hematoxylin. Arrows: cells detected by both Abs. Pictures are representative for teat tissue slides from 7 animals.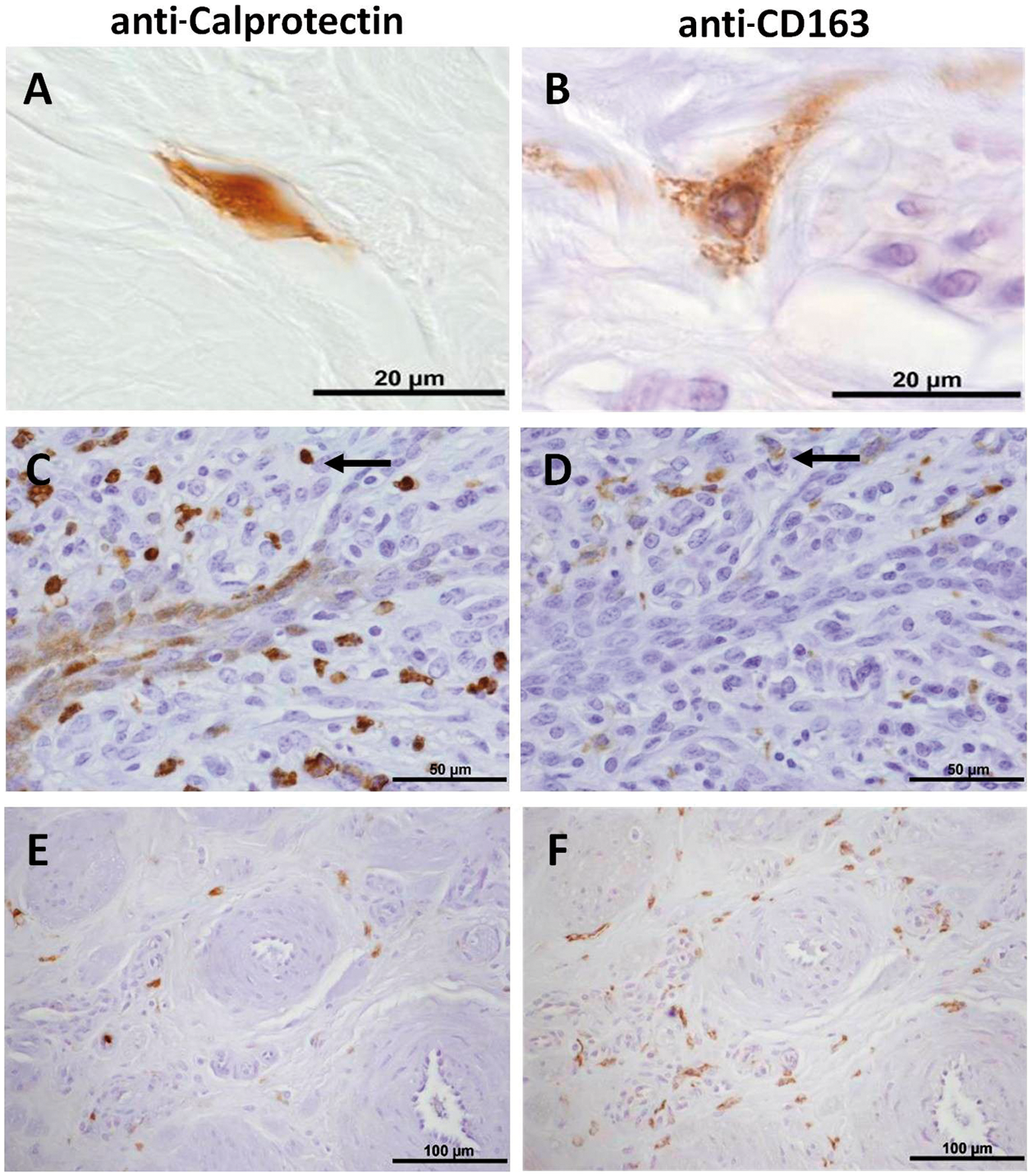
Expression of CD163 and Calprotectin during the in vitro monocyte–macrophage transition
To investigate a possible development and functional polarization of the two different macrophage phenotypes, further studies were made with in vitro-differentiated MdM. We also analyzed whether PGE2, a host factor expressed after pathogen contact, has an impact on the generation of different macrophage populations or modulates macrophage responses towards classical and/or alternative stimuli.
Monocytes expressed both antigens. The expression pattern for Calprotectin was broad, while CD163 displayed a defined, homogenous expression (Figure 2A). To analyze when and how selective positive MdM develop, the expression of CD163 and Calprotectin was followed during the monocyte to macrophage transition up to d 7 in vitro.
Two-colour immunofluorescence showing the kinetic of Calprotectin and CD163 expression during differentiation of MdM and PGE2-MdM (differentiated in the presence of PGE2, 1 nmol/l). (A) Monocytes (B) day 4 and day 7 MdM/PGE2-MdM were labelled with Calprotectin and CD163-specific monoclonal Abs. Data are representative for 3 independent experiments.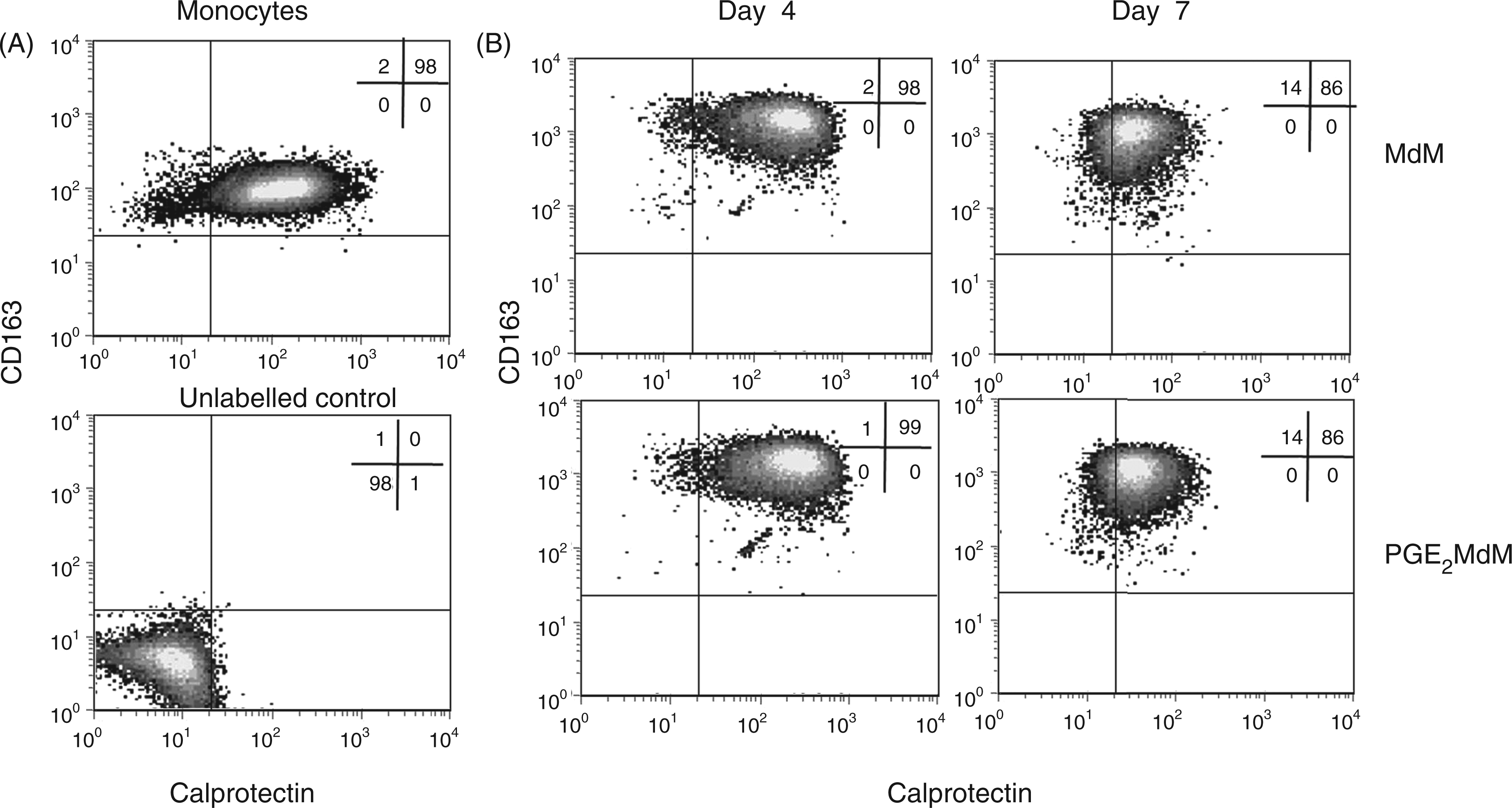
On d 4, MdM still expressed both antigens, with a markedly increased expression of CD163 and a homogeneous lower expression of Calprotectin (Figure 2B). Fully differentiated MdM on d 7 displayed an even decreased Calprotectin expression with a consistently high CD163 expression (Figure 2B). Thus, in vitro-generated macrophages displayed a phenotype different from those seen in vivo.
The presence of PGE2 during monocyte–macrophage differentiation did not influence the expression pattern of CD163 and Calprotectin (Figure 2B).
Classical, but not alternative, activation of MdM or PGE2-MdM alters CD163 expression pattern
In order to test whether different MdM-activation alters the expression pattern of CD163 and Calprotectin, MdM and PGE2-MdM were stimulated with IFN-γ and LPS (classical activation) or with IL-4 and IL-13 (alternative activation). Classical activation decreased expression of CD163 while Calprotectin expression was not altered (Figure 3A). In alternatively activated MdM no effect on the expression of both antigens could be observed (Figure 3B). Both MdM and PGE2-MdM displayed the same reduced CD163 expression after classical activation (Figure 3A, B).
Analysis of Calprotectin and CD163 expression in classically and alternatively activated MdM and PGE2-MdM. Monocytes were differentiated in the presence or absence of PGE2 (1 nmol/l). (A) Classically activated cells were primed with IFN-γ (20ng/ml, 12 h) and subsequently stimulated with LPS (1μg/ml, 3 h). (B) Alternatively activated cells were primed with IL-4 and IL-13 (each 20 ng/ml, 12 h). Cells were stained with CD163- or Calprotectin-specific monoclonal Abs. Filled histograms show unpolarized MdM. Data are representative for 3 independent experiments.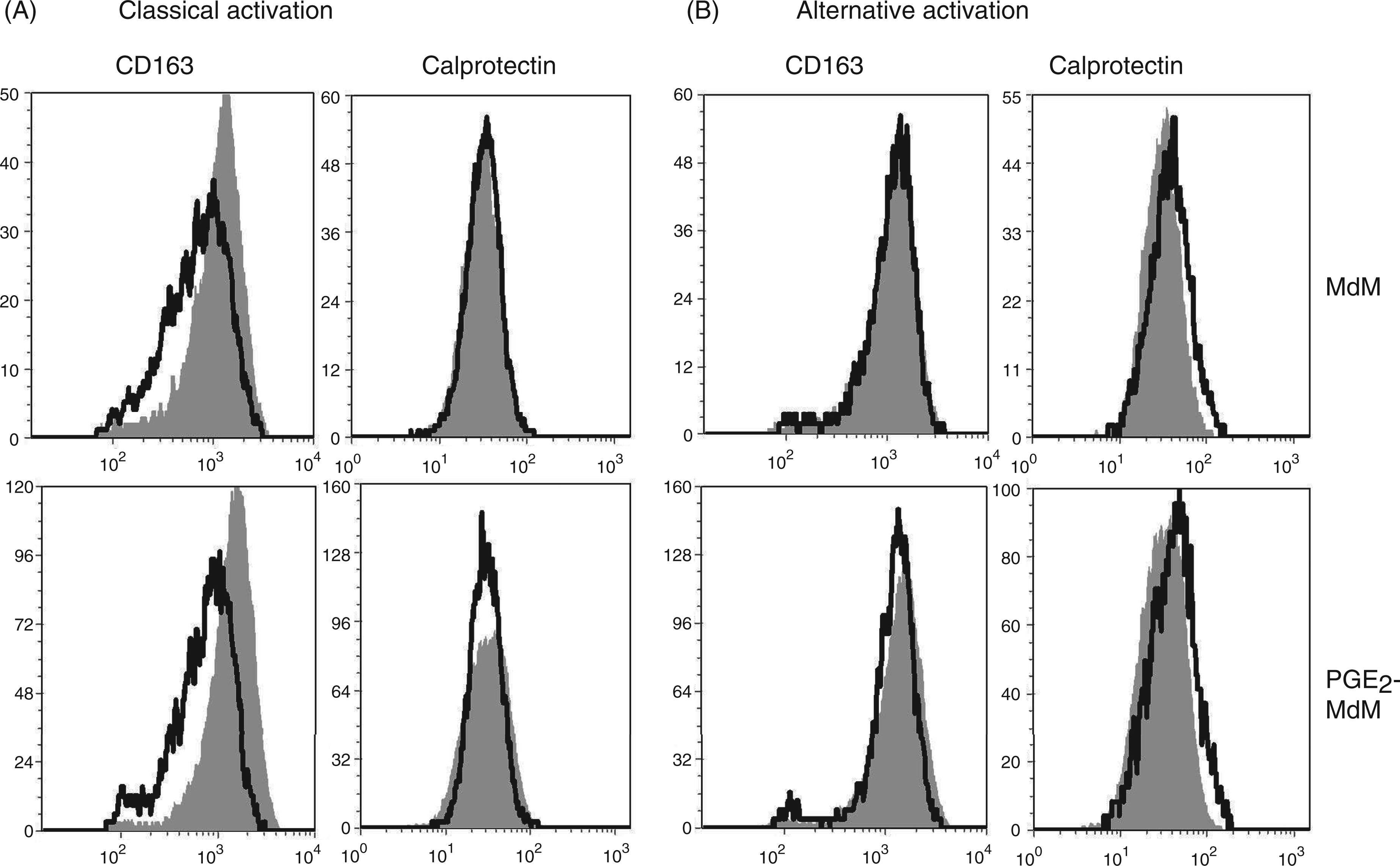
Gene expression profiles of classically and alternatively activated MdM and PGE2-MdM
To characterize the functional phenotype of the differently activated MdM and PGE2-MdM after contact with LPS, we analyzed the expression of genes related to classical activation (IL-12, iNOS) or to alternative activation (IL-10, Arg I), as well as the gene expression of CXCL8 as a classical chemoattractant for neutrophil granulocytes (Figure 4). All activation regimes induced a significantly increased CXCL8 gene expression (Figure 4A). Surprisingly, the expression of IL-10, a cytokine related to alternative activation, was only significantly up-regulated in LPS-stimulated MdM/PGE2-MdM without any polarization and not in alternatively activated cells (Figure 4B). Alternatively activated MdM/PGE2-MdM, however, showed up-regulated Arginase I expression—although it was not statistically significant—and significantly lower expression of IL-12 and iNOS compared with classically activated and LPS stimulated cells without polarization (Figure 4C–E). The expression of IL-12 and iNOS in classically activated MdM/PGE2-MdM did not significantly differ from those of LPS stimulated MdM/PGE2-MdM without polarization (Figure 4D, E). The presence of PGE2 during differentiation of MdM had no impact on gene expression profiles (Figure 4A–E).
Gene expression of classically and alternatively activated MdM and PGE2-MdM. Monocytes (n = 4) were differentiated in the presence or absence of PGE2 (1nmol/l) and for classical activation primed with IFN-γ (20ng/ml, 12h) and subsequently stimulated with LPS (1μg/ml, 3h) or for alternative activation primed with IL-4 and IL-13 (20ng/ml each, 12h) and subsequently stimulated with LPS (1μg/ml, 3h) or left untreated. Messenger RNA copy numbers (100ng total RNA) were determined by RT-qPCR. Distinct letters indicate significant differences (p < 0.05). n.d. = not detectable.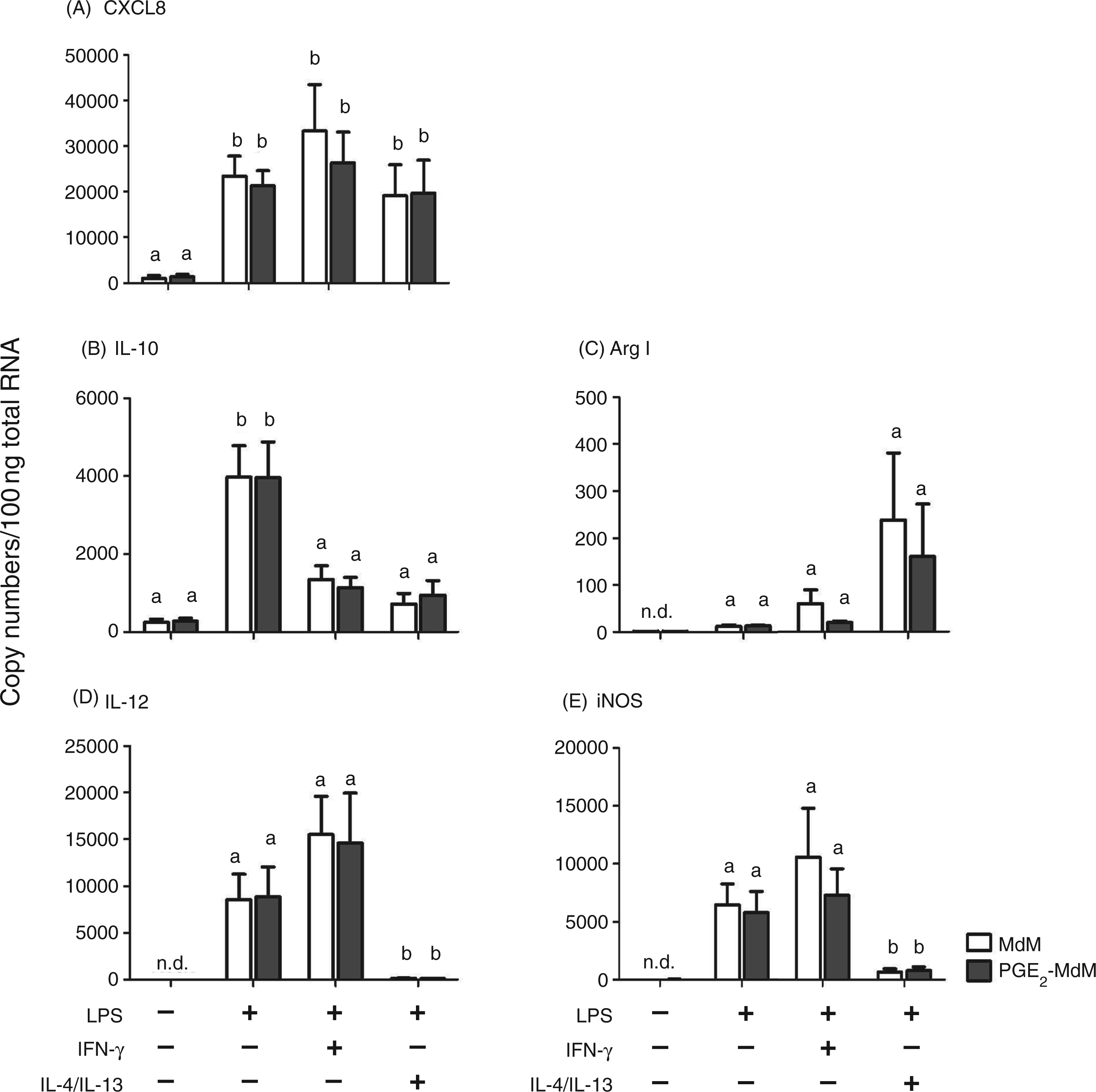
Supernatants of classically and alternatively activated MdM induce migration of neutrophil granulocytes which is dampened in PGE2-MdM supernatants
The chemotactic properties of differentially activated MdM/PGE2-MdM were analyzed by a quantitative in vitro migration assay using cell culture supernatants. Neither PGE2 nor LPS and the cytokines used for MdM-polarization themselves induced neutrophil migration (data not shown). Supernatants from LPS-stimulated MdM/PGE2-MdM did not induce a significantly higher neutrophil migration rate than supernatants of untreated MdM/PGE2-MdM (Figure 5). The migration rate was significantly increased with supernatants from classically activated MdM (31 ± 2%) compared with untreated (13 ± 2%) or only LPS-stimulated MdM supernatants (16 ± 3%) (Figure 5). Supernatants from classically activated PGE2-MdM induced a lesser increase of neutrophil migration rate reaching statistically significant levels compared to supernatants of untreated MdM (Figure 5). The strongest chemotactic properties were seen in supernatants of alternatively activated MdM. They significantly increased neutrophil migration rate up to 43 ± 6%. Compared with that, supernatants from alternatively activated PGE2-MdM significantly dampened neutrophil migration (31 ± 5%) (Figure 5).
Migration rates of neutrophilic granulocytes after quantitative in vitro migration. Lower wells of the transmigration chamber contained cell culture supernatants (n = 4) obtained from MdM differentiated in the presence or absence of PGE2 (1 nmol/l). MdM and PGE2-MdM were either primed with IFN-γ (20 ng/ml, 12 h) and subsequently stimulated with LPS (1 μg/ml, 3 h) to induce classical activation or primed with IL-4 and IL-13 (20 ng/ml each, 12 h) and subsequently stimulated with (LPS1μg/ml, 3 h) to induce alternative activation or left untreated. Distinct letters indicate significant differences (p < 0.05).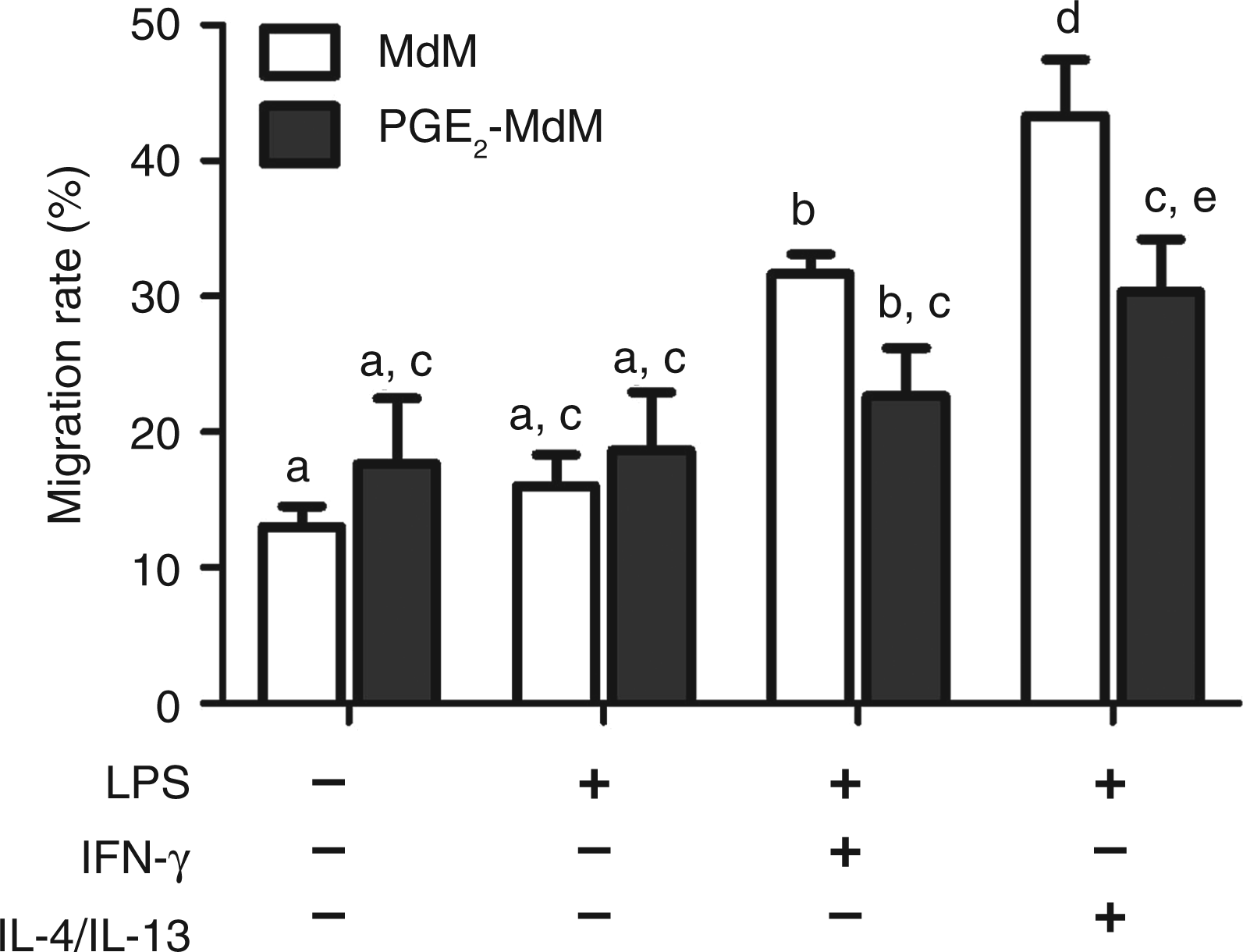
Discussion
Although macrophage plasticity is well studied in humans and mice, little is known about the situation in bovine tissue. The immunohistochemistry of teat tissue performed in this study provides a snap-shot of myeloid cells during their differentiation in the context of distinct mediators in their microenvironment. It reveals the presence of two clearly distinct phenotypes of macrophages in bovine udder tissue, expressing either CD163 or Calprotectin (Figure 1C–F). The expression of the hemoglobin scavenger receptor CD163 has been shown to be restricted to macrophages and is often related to the late inflammatory response because of its ability to bind hemoglobin haptoglobin complexes.22–24 Human macrophages stimulated with IL-10 showed a high expression of CD163 whereas classically activating stimuli, such as IFN-γ, induced only weak expression of the scavenger receptor, suggesting a relationship of CD163 expression with an anti-inflammatory macrophage subtype. 15 A recently published study, however, linked the iron uptake of macrophages promoted by CD163 to a pro-inflammatory macrophage phenotype. 25 The intracellular expression of the calcium binding protein Calprotectin can be induced in human monocytes by IL-1β, TNF-α and LPS, and is related to monocytes and freshly recruited macrophages under pro-inflammatory conditions.14,26,27 However, the expression of Calprotectin in later stages of inflammation is induced by IL-10 and a number of anti-inflammatory properties such as binding of ROS and chelating of zinc, and also suggests a role in resolving inflammation. 28 Therefore, the mere expression of either CD163 or Calprotectin cannot be used to characterize the functionality of the identified macrophages in teat tissue. The comparatively larger nucleus and cell body, as well as the elongated cytoplasma extensions of the CD163 positive cells, however, suggest that there could be a difference in cell metabolism or maturation stage between the two phenotypes (Figure 1A, B).
The in vitro model set up to investigate the functional properties of the different macrophage phenotypes showed that bovine MdM per se do not display the dichotomy concerning CD163 and Calprotectin expression which could be observed in vivo (Figure 2A, B). The increase of CD163 expression and the decrease of Calprotectin over time could reflect the proceeding maturation of the macrophages in culture (Figure 2B) as it is described by others. 24 Recent studies of our group showed a decreased CD163 expression and an increased Calprotectin expression in macrophages differentiated in the presence of monocyte attracting chemokines (unpublished data), suggesting that the biased phenotype in teat tissue is a matter of the local host mediator pattern. The two extremes of macrophage polarization, classically activated, pro-inflammatory MdM and alternatively activated, wound-healing MdM were induced by priming with IFN-γ and subsequent LPS stimulation for classical activation, or by priming with IL-4 and IL-13 for alternatively activated macrophages. In this set up, priming refers to the contact of the cells with host-derived mediators prior to stimulation with a PAMP. We adopted this procedure from general protocols for human and murine macrophages,29,30 but extended it by adding PGE2 during the differentiation of the cells exemplifying a local host mediator. Recent studies showed that induction of mastitis either by LPS or experimental infection with E. coli caused a transiently increased expression of prostaglandin endoperoxide synthase (PTGS2) in mammary gland tissue,31,32 demonstrating the relevance of PGE2 in the udder during mastitis. The rationale was to test whether the presence of PGE2 shapes MdM during their differentiation regarding the functions and phenotype of differentially primed MdM. As classical activation leads to a reduction in CD163 expression after 7 d of culture, the cells in the teat positive for Calprotectin could only be the result of an ongoing classical activation for more than 7 d (Figure 3). The gene expression profile of classically activated cells showed the anticipated pattern of significantly up-regulated IL-12 and iNOS expression (Figure 4D, E). The characteristics for alternatively activated macrophages were an almost complete shutoff in IL-12 expression compared with untreated MdM and a measurable ArgI expression (Figure 4C, D). Surprisingly, the expression of IL-10 was significantly up-regulated in unpolarized MdM stimulated with LPS and not in alternatively activated cells (Figure 4B). To our knowledge, this is the first study that has used IL-4/IL-13 priming for bovine MdM. Flynn and Mulcahy 9 induced IL-10 production in bovine MdM by using IL-4 and secretory products of F. hepatica. Therefore, another combination of priming agents might have induced an up-regulation in IL-10 gene expression above the level of merely LPS-stimulated MdM. As the ability to attract effector cells to the site of inflammation is an important step in the early phase of the inflammatory response, we also measured gene expression of CXCL8 as a classical neutrophil chemoattractant which was significantly up-regulated in all polarization regimes, as well as in unpolarized LPS stimulated MdM (Figure 4A). The presence of PGE2 during differentiation did not impact CD163 and Calprotectin expression or on the expression of the selected genes. The migration assay with MdM culture supernatants showed a significantly higher production of chemotactic mediators by classically activated MdM compared with only LPS-stimulated MdM. Alternatively activated MdM, however, showed the strongest production of chemotactic mediators (Figure 5). Alternatively activated macrophages play a key role in attraction of other cells to inflammatory foci. 3 The presence of PGE2 during macrophage differentiation significantly dampened the chemotactic activity of alternatively activated MdM (Figure 5). This effect is neither due to PGE2 itself, as controls showed no chemotactic activity of PGE2 (data not shown), nor induced by a reduction in CXCL8 expression as the gene expression of CXCL8 is not influenced by PGE2 (Figure 4A). Therefore, despite the lack of modulating selected genes typical for alternative or classical activation, and any impact on CD163 and Calprotectin expression, there must be other mechanisms influenced by PGE2 that shape the mediator pattern of alternatively activated macrophages. This stresses the potential of PGE2 in inducing the switch from classically to alternatively activated macrophages in an in vitro context, which could be exploited for therapeutic approaches aiming to modulate the inflammatory response to obtain a sufficient clearance of the pathogen but avoid an overshoot of pro-inflammatory cytokines that have a harmful effect on the host. Particularly in the case of bovine mastitis, the modulation of the host immune response might be promising strategy for future treatment designs.
Overall, the study gives a more detailed view on the phenotypic and functional properties of bovine macrophages in an in situ and in vitro context, highlighting the importance of host derived mediators in the process of macrophages differentiation in vitro. It also stressed that the classical way of linking the expression of certain surface markers on macrophages to their function is not appropriate.
Footnotes
Funding
This work was funded by Pfizer Animal Health.
Acknowledgements
It is a special pleasure to acknowledge the kind and skilful technical assistance of Silke Schoeneberg and Udo Rabe.
