Abstract
Members of the nucleotide binding and oligomerization domain-like receptors (NLRs) and the PYD and CARD domain containing adaptor protein (PYCARD) assemble into multi-protein platforms, termed inflammasomes, to mediate in the activation of caspase-1 and the subsequent secretion of IL-1β and IL-18, and the induction of pyroptotic cell death. While the recognition site for caspase-1 is well conserved in mammals, most of the non-mammalian IL-1β genes cloned so far lack this conserved site. We report here that stimulation or infection of seabream macrophages (MØ) led to the caspase-1-independent processing and release of IL-1β. In addition, several classical activators of the NLRP3 inflammasome failed to activate caspase-1 and to induce the processing and release of IL-1β. Furthermore, the processing of IL-1β in seabream MØ is not prevented by caspase-1 or pan-caspase inhibitors, and recombinant seabream caspase-1 failed to process IL-1β. However, the pharmacological inhibition of caspase-1 impaired Salmonella enterica sv. Typhimurium-induced cell death. These results suggest a role for the inflammasome and caspase-1 in the regulation of pyroptotic cell death in fish and support the idea that its use as a molecular platform for the processing of pro-inflammatory cytokines arose after the divergence of fish and tetrapods.
Introduction
The inflammasomes are cytosolic multiprotein platforms required for the activation of inflammatory caspases, namely caspase-1 1 and caspase-11 (also known as caspase 4). 2 Genetic studies in mice have revealed at least four inflammasomes of distinct composition, namely those containing the nucleotide-binding and oligomerization domain-like receptors (NLRs) NLRP1B, NLRP3 and NLRC4, and a recently characterized inflammasome complex assembled around the HIN-200 protein absent in melanoma 2 (AIM2). 1 An adaptor protein, PYD and CARD domain containing (PYCARD, also known as ASC), which recruits NLRs carrying the pyrin homologous domain (PYD) and procaspase-1 through PYD and CARD, respectively, is responsible for the formation of some inflammasomes and also for the activation of procaspase-1. 3 However, two recent, elegant studies have demonstrated a further complex step—at least in the NLRC4 inflammasome—where another family of intracellular receptors called NAIPs was directly involved in the binding and recognition of specific virulence-associated molecules.4,5
Once activated, caspase-1 modulates the inflammatory and host defense responses by processing the pro-inflammatory cytokines IL-1β and IL-18 into their biologically active forms: a prerequisite for their secretion.6–9 In addition to secreting IL-1β and IL-18, caspase-1 and –caspase-11 contribute to the host defense through an inflammatory cell death program known as pyroptosis, which occurs in myeloid cells infected with bacterial pathogens such as Salmonella enterica sv. Typhimurium (S. typhimurium), Francisella tularensis and Bacillus anthracis.1,10–12
Although mammalian IL-1β is relatively well characterized, little information is available on IL-1β from lower vertebrates. Identification of the first non-mammalian sequences has resulted in an even more puzzling scenario, as the fish, amphibian and bird IL-1β genes cloned so far lack a conserved caspase-1 recognition site. 13 We have determined the molecular identity and tissue localization of IL-1β in the teleost fish gilthead seabream (Sparus aurata L.). 14 Like its mammalian counterpart, we found that endotoxin challenge led to a significant increase in IL-1β expression in seabream leukocytes and that it accumulates intracellularly. 14 In contrast, the classical activator of the NLRP3 inflammasome, ATP, 15 fails to provoke IL-1β release from endotoxin-stimulated sea-bream leukocytes, 14 despite phosphatidylserine externalization and cell permeabilization in seabream leukocytes treated with ATP. 16 In addition, neither seabream nor zebrafish P2X7 receptors induced the secretion of mammalian or fish IL-1β when expressed in HEK293 cells, while a chimeric receptor harboring the ATP-binding domain of seabream P2X7 and the intracellular region of its rat counterpart did. 16 These findings indicate that P2X7 receptor-mediated activation of caspase-1 and the release of IL-1β result from different downstream signaling pathways, and suggest that, although the mechanisms involved in IL-1β secretion are conserved throughout evolution, distinct inflammatory signals have been selected for the secretion of this cytokine in different vertebrates.
To throw light on the evolutionary history of the inflammasome and its role in the regulation of the inflammatory response in primitive vertebrates, an immunologically tractable teleost fish species was studied, namely the gilthead seabream (S. aurata, Perciformes). We found that stimulation of seabream macrophages (MØ) with flagellin or bacterial DNA, or infection with either invasive or non-invasive bacteria, led to the processing and release of proIL-1β into an 18 kDa mature form. Interestingly, although pharmacological inhibition of caspase-1 had no effect on the processing of IL-1β, they impaired S. typhimurium-induced cell death in seabream.
Materials and methods
Animals
Healthy specimens (150 g mean mass) of the hermaphroditic protandrous marine fish gilthead seabream (S. aurata) were maintained at the Oceanographic Centre of Murcia (Spain) in 14 m3 running seawater aquaria (dissolved oxygen 6 ppm, flow rate 20% aquarium vol/h) with natural temperature and photoperiod, and fed twice daily with a commercial pellet diet (Trouvit, Burgos, Spain). Fish were fasted for 24 h before sampling. The experiments performed comply with the Guidelines of the European Union Council (86/609/EU) and the Bioethical Committee of the University of Murcia (approval no. #333/2008) for the use of laboratory animals.
Cell culture and treatments
Seabream head kidney (bone marrow equivalent in fish) leukocytes obtained as described elsewhere 17 were maintained in sRPMI [RPMI-1640 culture medium (Gibco, Madrid, Spain) adjusted to gilthead seabream serum osmolarity (353.33 mOs) with 0.35% NaCl] supplemented with 5% fetal calf serum (FCS) (Gibco), 100 I.U./ml penicillin and 100 µg/ml streptomycin (Biochrom, Cambridge, UK). Some experiments were conducted using purified cell fractions of MØ and acidophilic granulocytes (AG), the two professional phagocytic cell types of this species.17,18 Briefly, AG were isolated by magnetic-activated cell sorting (MACS) using a monoclonal Ab (mAb) specific to gilthead seabream AG (G7). 17 MØ monolayers were obtained after overnight culture of G7- fractions in FCS-free medium and their identity was confirmed by the expression of the macrophage-colony stimulating factor receptor (M-CSFR). 18
Seabream MØ, AG and total leukocytes from seabream head kidney were stimulated for 16 h at 23°C with 50 µg/ml phenol-extracted genomic DNA from Vibrio anguillarum ATCC19264 cells (VaDNA) or 100 ng/ml flagellin (Invivogen, San Diego, CA, USA) in sRPMI supplemented with 0.1% FCS and penicillin/streptomycin.
19
These pathogen-associated molecular patterns (PAMPs) were found to be most powerful in the activation of gilthead seabream professional phagocytes.
19
In some experiments, cells were then washed twice with sRPMI, and incubated for 1 h with 5 m
Cell supernatants from control and stimulated/infected leukocytes were collected after overnight incubation, unless otherwise indicated, clarified with a 0.45 µm filter and concentrated by precipitation with 20% trichloroacetic acid (Sigma-Aldrich).
Cell viability
Aliquots of cell suspensions were diluted in 200 µl PBS containing 40 µg/ml propidium iodide. The number of red fluorescent cells (dead cells) from triplicate samples was analyzed by using flow cytometry (BD Biosciences, Madrid, Spain).
Caspase-1 activity assay
Caspase-1 activity was determined with the fluorometric substrate Z-YVAD-AFC (caspase-1 substrate VI; Calbiochem-Merck), as described previously.
22
In brief, cells were lysed in hypotonic cell lysis buffer HEPES, 5 m
Western blot
Cells were lysed at 4°C in lysis buffer (10 m
Bactericidal assay
Leukocytes were lysed with 0.2% saponin (Sigma) and the number of surviving bacteria was determined by using a 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide inner salt (XTT; Sigma) colorimetric assay. 19 Bacteria were also enumerated by plating cell extracts on LB-agar plates containing the appropriate antibiotics.
Cell transfection
Seabream caspase-1 and pro-IL-1β were cloned into pcDNA3.1/V5-His-TOPO and pcDNA4His/Max expression vectors (Invitrogen, Madrid, Spain) for the expression of C-terminal V5/His6-tagged or N-terminal His/Xpress proteins respectively.16,22 Other expression constructs were rat P2X7ee,
24
human proIL-1β,
25
and human caspase-1.
26
Plasmid DNA was prepared using the Mini-Prep procedure (Qiagen, Madrid, Spain). DNA pellets were re-suspended in water and further diluted, when required, in PBS. Transfections were performed with a cationic lipid-based transfection reagent (LyoVec, Invivogen), according to the manufacturer’s instructions. Briefly, HEK293 cells were plated in six-well plates (400,000 cells/well) together with 100 µl of transfection reagent containing 0.15 µg of the rat P2X7-ee, 1.5 µg of either the human or seabream proIL-1β expression constructs and 1.5 µg of the human or seabream caspase-1 expression vector. Forty-eight hours after transfection, cells were washed twice with serum-free medium and incubated for 30 min with 1 m
Statistical analysis
Data were analyzed by analysis of variance (ANOVA) and a Tukey multiple range test to determine differences between groups. The differences between two samples were analyzed by Student’s t-test.
Results
Infection of seabream head-kidney leukocytes with invasive and non-virulent bacteria leads to the processing and release of mature IL-1β
To evaluate the production, processing and release of fish IL-1β, seabream head-kidney leukocytes pre-stimulated with bacterial DNA or flagellin—two powerful PAMPs for this species able to drastically induce the expression of the IL-1β gene and to promote the intracellular accumulation of IL-1β19,27,28—were infected with wild type S. typhimurium SL1344 and the non-invasive isogenic derivative strain SB169, which harbors a mutation in the sipB translocation machinery component (sipB::aphT) of the Salmonella pathogenicity island 1 (SPI1)-encoded type III secretion system (T3SS).
20
The SPI1 T3SS can be readily recognized by caspase-1 in mammalian cells by detecting inadvertently translocated flagellin or PrgJ rod protein to the cytosol mediated by NLRC4.4,5 Unexpectedly, although stimulation of leukocytes with PAMPs for 16 h led to the processing of proIL-1β to a ∼18 kDa IL-1β mature form (mIL-1β) that mainly remained cell-associated, infection of PAMP-stimulated leukocytes with both wild type and non-invasive S. typhimurium strains promoted the released of mature IL-1β (Figure 1A, B). Similarly, wild type E. coli and an isogenic derivative strain expressing recombinant LLO from L. monocytogenes
21
were similarly effective in the processing and release of IL-1β (Figure 1C). We next sought to determine whether PAMPs alone also induce the secretion of IL-1β and found that this was the case (Figure 1D), although at lower levels than bacteria (Figure 1A–C). As these results suggest the caspase-1-independent processing and release of IL-1β in this species, we stimulated seabream leukocytes with ATP (Figure 2A) and nigericin (Figure 2B), two classic activators of mammalian NLRP3 inflammasome,
15
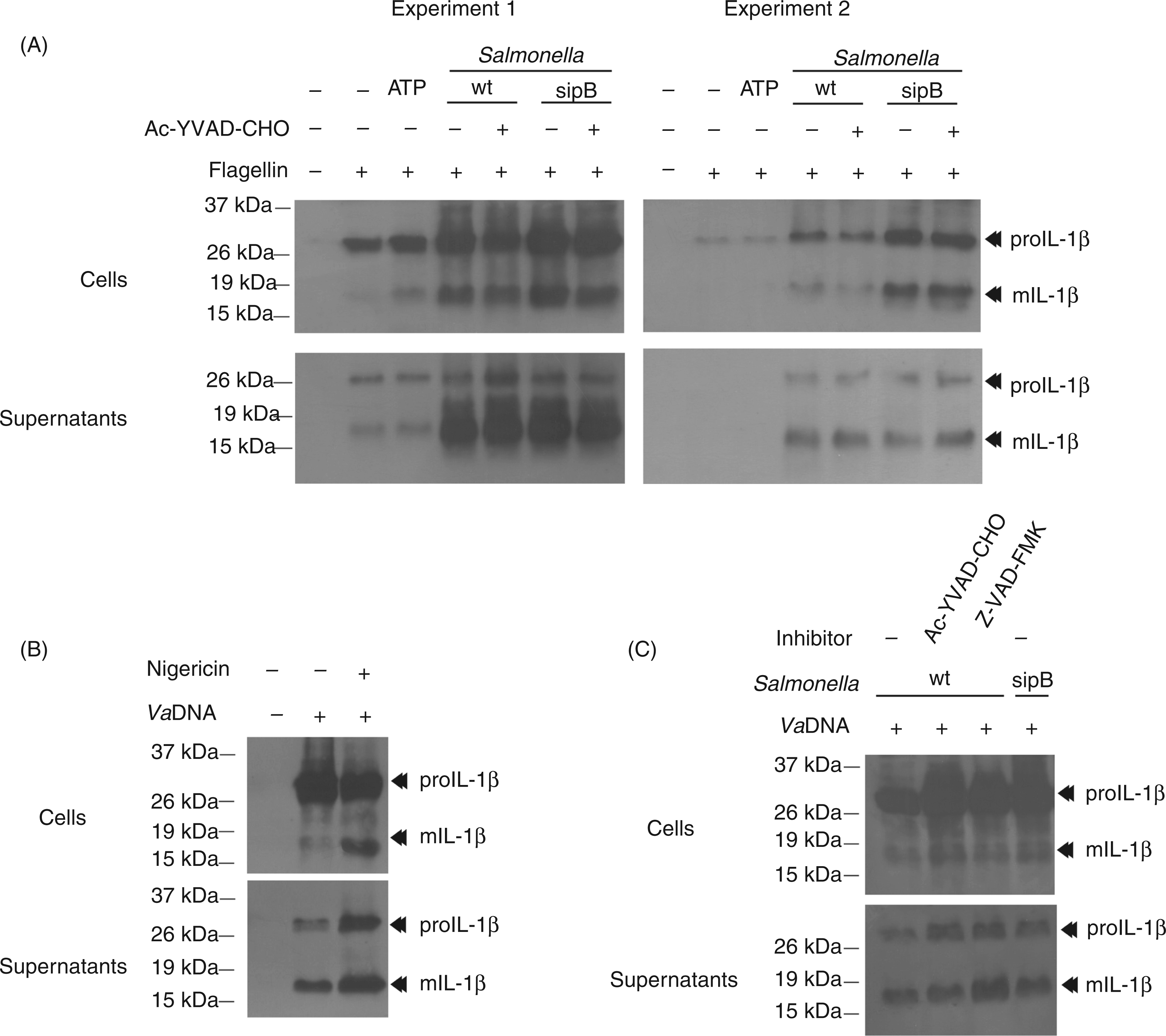
and found that both activators also failed to trigger IL-1β processing and release in seabream leukocytes. Interestingly, the pharmacologic inhibition of caspase-1 failed to inhibit the processing and secretion of IL-1β in seabream leukocytes in response to S. typhimurium infection (Figure 2B, C), even though this inhibitor is able to fully inhibit caspase-1 activity in seabream leukocytes.
22
These results were further confirmed with the pan-caspase inhibitor Z-VAD-FMK, which also failed to affect IL-1β processing and release (Figure 2C).
Infection of seabream leukocytes with invasive and non-virulent bacteria leads to the processing and release of mature IL-1β. Seabream head-kidney leukocytes were stimulated for 16 h with 50 µg/ml VaDNA (A, B, D) or 1 µg/ml flagellin (B, D) and then infected with a MOI of 1 (A) or 10 (B–D) of wild-type and sipB mutant strain of S. typhimurium (A, B) or wild-type E. coli and its isogenic derivative expressing recombinant LLO (C). Two hours after the infection, gentamycin (10 µg/ml) was added to limit the growth of extracellular bacteria and the infected leukocytes were incubated for 24 h. Cell lysates (5 × 106 cells) and concentrated supernatants obtained from 107 cells were probed with a monospecific polyclonal Ab to seabream IL-1β. Migration positions for the mature (m) and pro-cytokine forms are indicated. Results are representative of five independent experiments. The processing and release of IL-1β is caspase-1 independent in seabream leukocytes. Seabream head-kidney leukocytes pre-treated for 1 h with 100 µ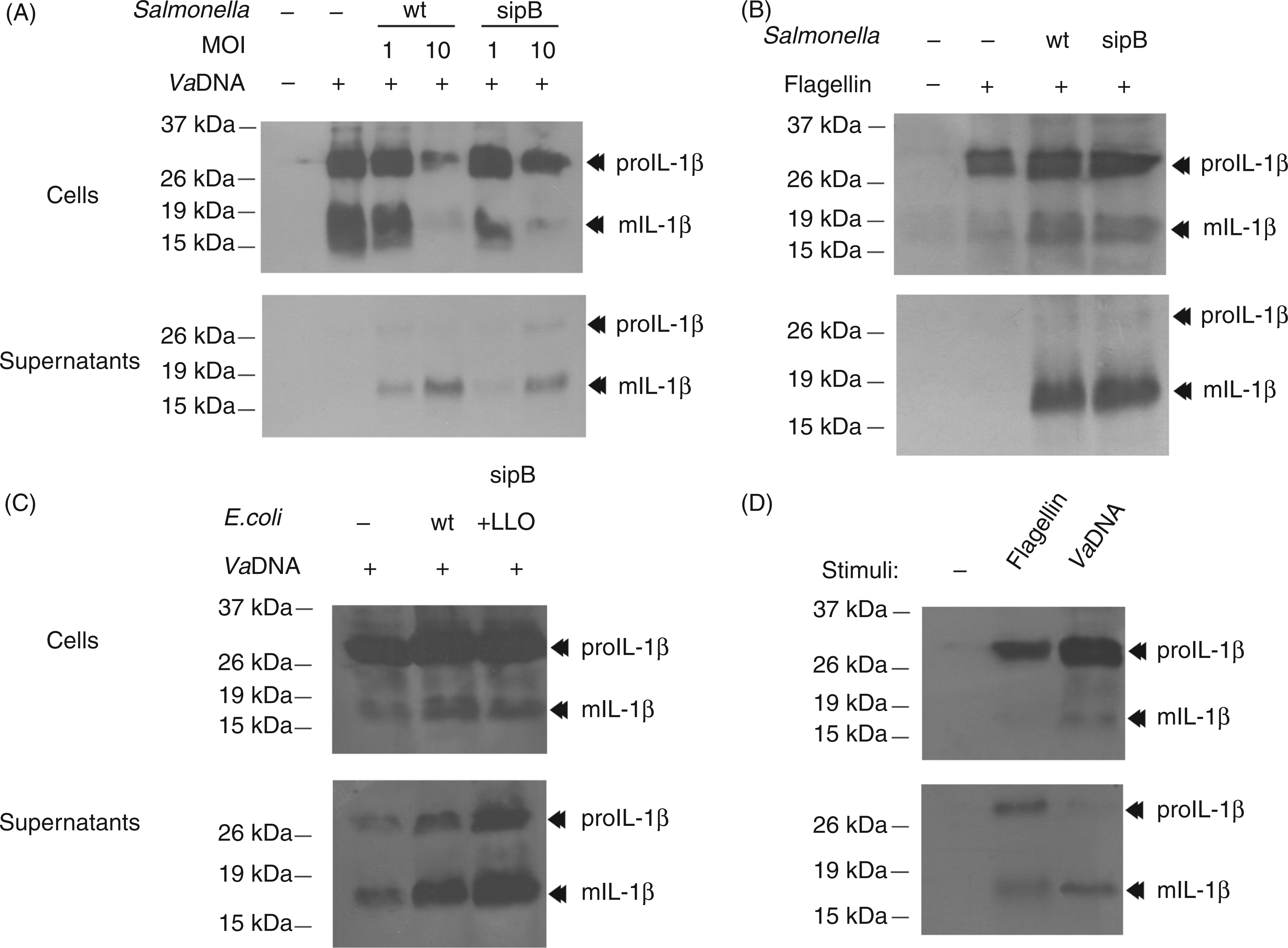
Seabream caspase-1 fails to process IL-1β
We next studied whether seabream caspase-1 ectopically expressed in HEK293 cells was able to process seabream IL-1β following activation. Seabream caspase-1 was unable to process seabream proIL-1β, and activation of the rat P2X7 receptor led to the secretion of proIL-1β. In sharp contrast, human caspase-1 processed human proIL-1β in both non-stimulated and ATP-stimulated cells, while activation of the P2X7 receptor with ATP led to the release of precursor and mature IL-1β forms (Figure 3). In accordance with our previous results,
22
activation of the rat P2X7 receptor with ATP resulted in the activation of caspase-1 at similar levels to that observed in its human counterpart (Figure 3).
Seabream caspase-1 fails to process seabream IL-1β. HEK293 cells were transiently transfected with the seabream (left panel) or human (right panel) caspase-1 and the rat (r) P2X7 expression vectors. After 2 d, cells were stimulated with 1 m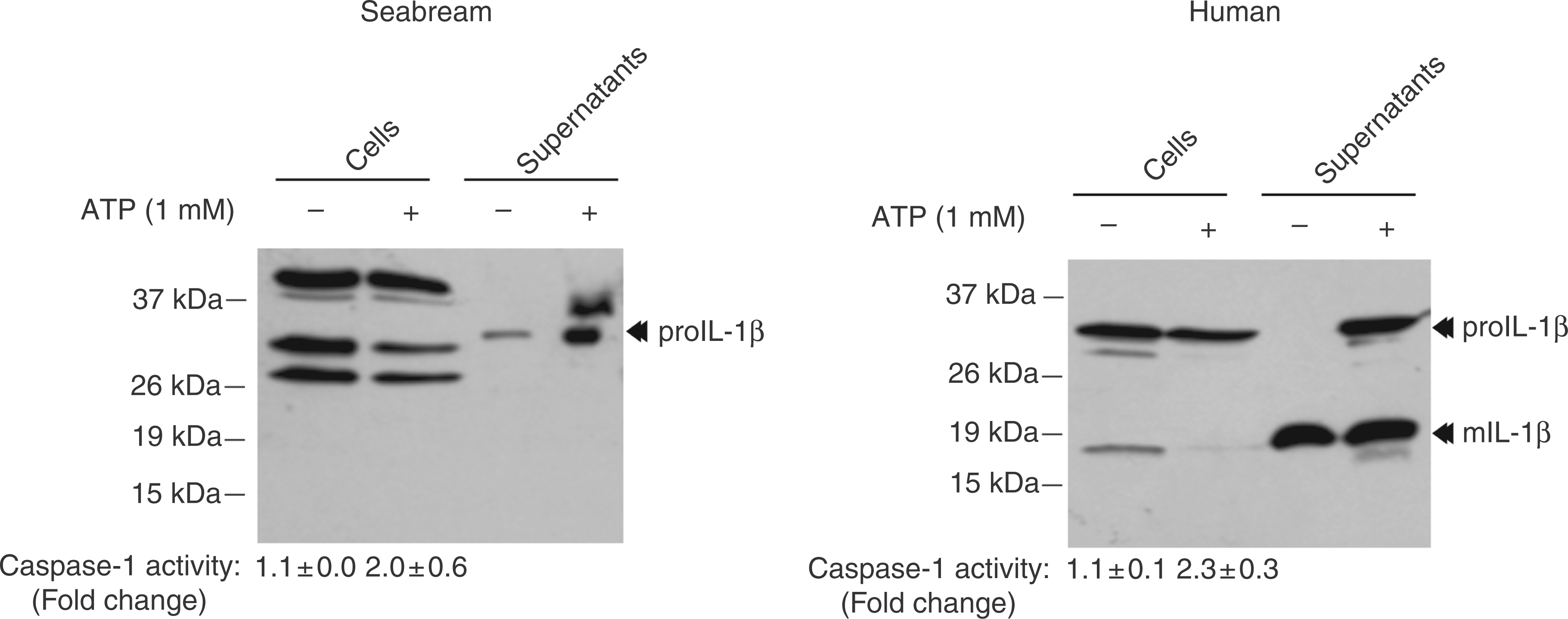
Classical NLRP3 inflammasome activators fail to activate caspase-1 and IL-1β processing in seabream MØ
As the relevance of the inflammasome and caspase-1 in the processing and release of IL-1β by mammalian neutrophils has been little studied and neutrophils may show inflammasome/caspase-1-independent mechanisms to process IL-1β,
29
we next examined the activation of caspase-1 and the processing/release of IL-1β in purified seabream MØ. The results show that ATP, K+ efflux (nigericin) and several crystals, including MSU and aluminum, all failed to activate caspase-1 in seabream MØ (Figure 4A). Similarly, none of these stimuli was able to promote the processing and release of IL-1β, although some of them decreased the release of mature IL-1β (Figure 4B). Interestingly, a hypotonic solution, which activated caspase-1 in seabream and mouse MØ through the activation of NLRP3 inflammasome (unpublished data) was also unable to trigger the processing of IL-1β (Figure 4A, B).
Classical NLRP3 inflammasome activators fail to activate caspase-1 and IL-1β processing in seabream MØ. Seabream head-kidney MØ were stimulated with 50 µg/ml VaDNA for 16 h, washed twice and then incubated for 1 h with a hypotonic buffer (90 mOs), 500 µg/ml MSU crystals, 5 m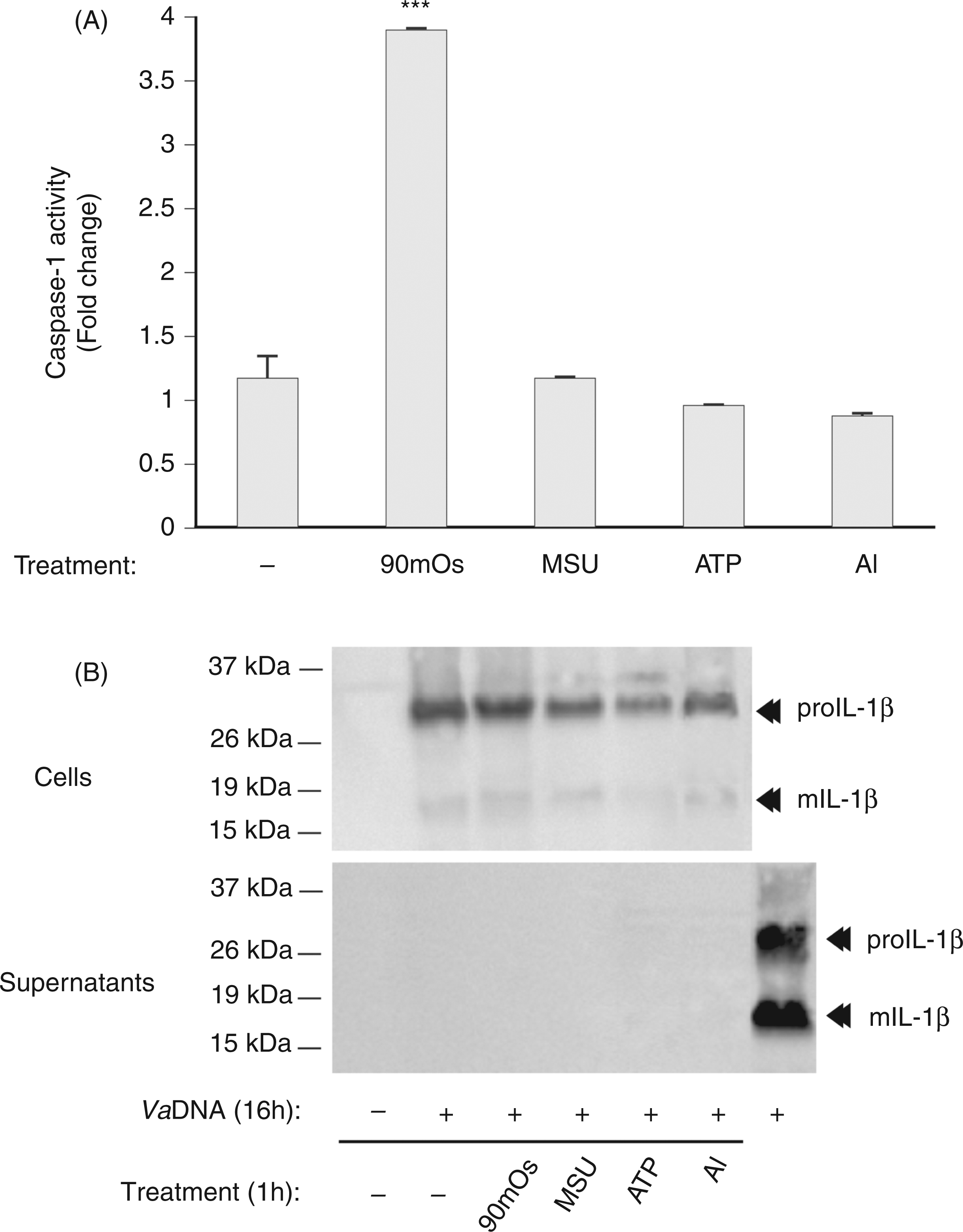
Invasive S. typhimurium promotes caspase-1 activation and cell death in seabream
We next analyzed the impact of wild-type and sipB mutant strains of S. typhimurium in seabream leukocytes and MØ. The results show that the wild type strain induced the rapid activation of caspase-1 in infected MØ, while the isogenic mutant strain failed to do so (Figure 5A, B). Notably, although both bacterial strains were able to trigger the processing and release of IL-1β at similar levels (Figure 5C), wild type S. typhimurium induced leukocyte death at higher levels (Figure 5D) and showed higher proliferative capacity in leukocytes (Figure 5E) than its sipB mutant isogenic derivative. In addition, pharmacologic inhibition of caspase-1 impaired S. typhimurium-induced cell death (Figure 5F).
Invasive S. typhimurium promotes caspase-1 activation and cell death in seabream. Seabream head-kidney MØ (A–C) and total leukocytes (D–F) pre-stimulated for 16 h with 50 µg/ml VaDNA in the presence or absence of 100 µ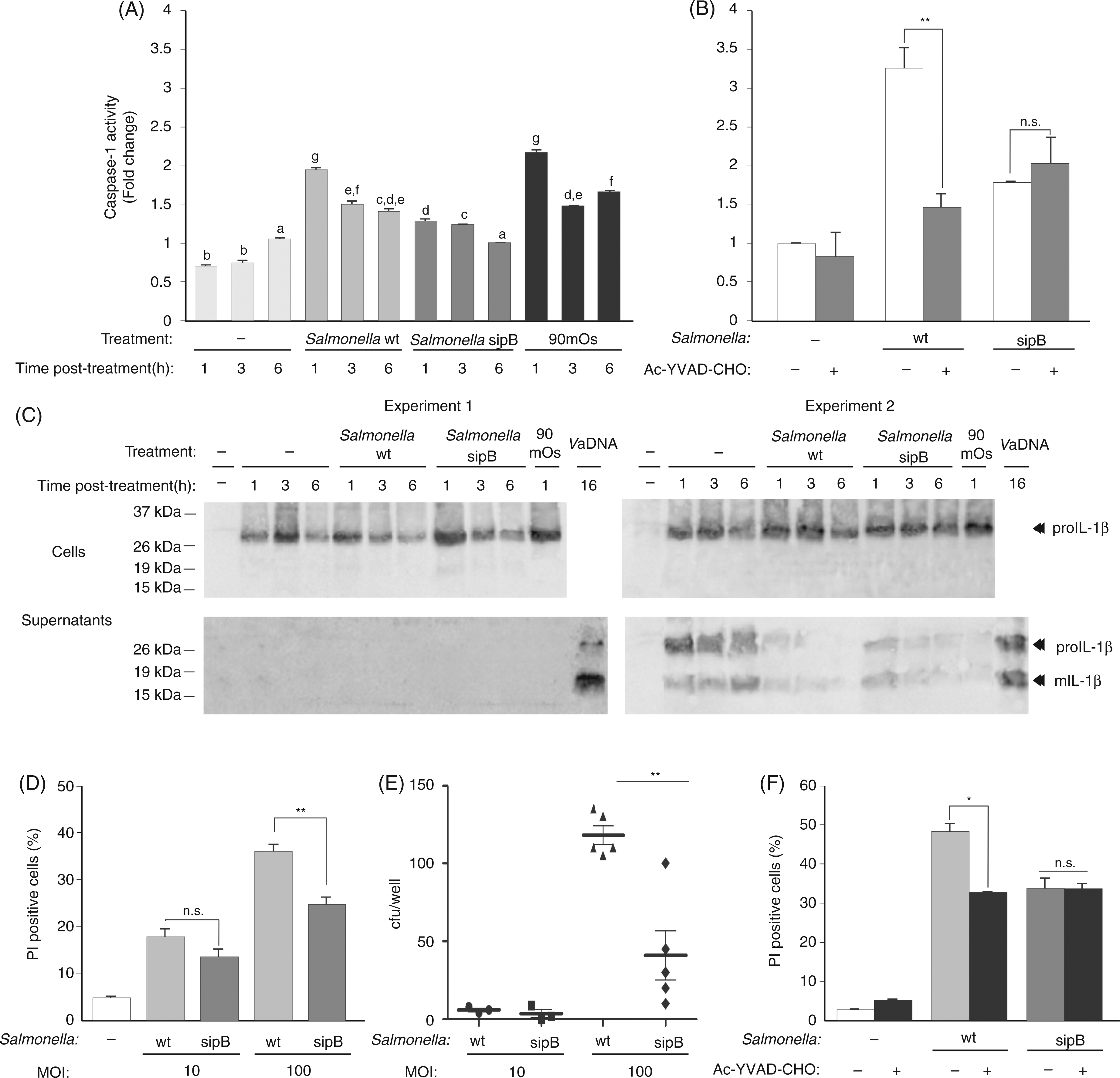
Discussion
In recent years, many studies have reported the crucial role of the inflammasome as a molecular platform involved in the sensing of microbial presence in the cytosol and the subsequent activation of caspase-1.1,2 Once activated, caspase-1 modulates inflammatory and host defense responses by processing the pro-inflammatory cytokines IL-1β and IL-18 into their biologically active forms—a prerequisite for their secretion.6–9 In sharp contrast, we found that although PAMP stimulation leads to the intracellular accumulation of IL-1β in seabream leukocytes,
14
the classical activator of the NLRP3 inflammasome ATP
15
fails to provoke IL-1β release from PAMP-stimulated seabream leukocytes,
14
despite phosphatidylserine externalization and cell permeabilization in these cells following ATP stimulation.
16
As these obvious differences in the processing and secretion of IL-1β between fish and mammals hinge on the presence of a functional P2X7 receptor in fish and as ATP was seen to be less potent in seabream P2X7 receptors than in mammalian P2X7 (EC50 ∼2 m
Despite the crucial importance of the inflammasome in sensing intracellular pathogens, little information exists concerning this gene family in non-mammalian vertebrates. A recent phylogenetic study has discovered three distinct NLR subfamilies in teleost fish by mining genome databases of various species: the first subfamily (NLR-A) resembles mammalian nucleotide-binding oligomerization domain (NOD) proteins, the second (NLR-B) resembles mammalian nacht, leucine rich repeat and pyrin domain (NALP) proteins, while the third (NLR-C) appears to be unique to teleost fish. 30 In zebrafish, while NLR-A and NLR-B subfamilies contain five and six genes, respectively, the third subfamily is strikingly large, containing several hundred NLR-C genes, many of which are predicted to encode a C-terminal B30.2 domain. 30 Although genetic depletion of zebrafish NOD1 and NOD2 orthologs belonging to subfamily NLR-A have been found to reduce the ability of embryos to control systemic infection, 31 the functional relevance of this extended array of NLR genes of zebrafish needs to be investigated.
The inability of caspase-1 to process IL-1β in sea-bream questions the relevance of caspase-1 and the inflammasome in the regulation of the immune response of early vertebrates. However, in addition to secreting IL-1β and IL-18, it has been found more recently that caspase-1 and caspase-11 contribute to host defense through the clearance of infected immune cells by inducing pyroptotic cell death.1,10–12 Indeed, an interesting study has very recently found that a S. typhimurium strain persistently expressing flagellin was cleared by the cytosolic flagellin-detection pathway through the activation of caspase-1 by the NLRC4 inflammasome; however, this clearance was independent of IL-1β and IL-18, but closely dependent on caspase-1-induced pyroptotic cell death of the infected MØ. 10 Strikingly, bacteria released from pyroptotic MØ exposed them to uptake and rendered them susceptible to killing by reactive oxygen species in neutrophils. 10 Similarly, the activation of caspase-1 cleared unmanipulated Legionella pneumophila and Burkholderia thailandensis through cytokine-independent mechanisms. 10 These results were later confirmed in L. monocytogenes and Francisella novicida, where mutations that caused bacteriolysis in the MØ cytosol promoted pyroptosis through activation of PYCARD-dependent AIM2 inflammasome, suggesting that pyroptosis is also triggered by bacterial DNA released during cytosolic lysis32,33 to clear these intracellular bacteria. 34 Consistent with these results, we found that invasive S. typhimurium, but not a non-invasive isogenic derivative strain harboring a mutation in the sipB translocation machinery component of the SPI1-encoded T3SS, 20 was able to promote the activation of caspase-1. This, in turn, resulted in leukocyte cell death. As the SPI1 T3SS can be recognized readily by caspase-1 in mammalian cells through the detection of flagellin or PrgJ rod protein inadvertently translocated to the cytosol mediated by NLRC4,4,5 our results suggest a role for the inflammasome and caspase-1 in the regulation of pyroptotic cell death and the clearance of intracellular bacteria in fish.
In conclusion, our results support the idea that the use of the inflammasome as a molecular platform for the processing of pro-inflammatory cytokines arose after the divergence of fish and tetrapods. Although our data argue against a role for caspase-1 in the processing of IL-1β in seabream, they do not rule out the possibility that caspase-1 might mediate the processing of other cytokines, such as IL-18, in fish. Furthermore, we hypothesize that, because of the relatively less elaborated and restrictive adaptive immune response of early vertebrates, 35 fish would display a more sophisticated intracellular sensing system than mammals, strengthening their ability to clear intracellular pathogens through the induction of pyroptotic cell death.
Footnotes
Funding
This work was supported by the Spanish Ministry of Science and Innovation (grant numbers BIO2008-01379, CSD2007-00002, and PhD fellowship to D.A.) and the Fundación Marcelino Botín (Spain).
Acknowledgements
We thank I. Fuentes and P. Martínez for excellent technical assistance, F. García del Portillo and Jorge Galán for the S. typhimurium strains, D. Higgins for E. coli strains, A. Surprenant for the rP2X7ee, S.B. Mizel for pRc/RSV-proIL-1β and J. Yuan for pBactM10Z.
