Abstract
Peripheral blood monocytes are recruited to inflammatory and tumor lesions where they undergo terminal differentiation into macrophages. Monocytes/macrophages integrate stimulatory and inhibitory signals present in the pathologic microenvironment through a defined repertoire of cell surface receptors, and deregulated expression of these molecules may result in amplification of inflammation or establishment of immune escape mechanisms. Characterization of the expression and function of these receptors is required for a better understanding of the regulation of monocyte/macrophage activity at pathologic sites. Hypoxia is a common feature of many pathological situations and an important regulator of monocyte/macrophage pro-inflammatory responses. In this study, we identify the leukocyte membrane antigen, CD300a, a member of the CD300 superfamily of immunoregulatory receptors, as a new hypoxia-inducible gene in primary human monocytes and monocyte-derived macrophages. CD300a mRNA up-regulation by hypoxia was rapid and reversible, paralleled by increased surface protein expression, and mediated by hypoxia-inducible factor-1α. CD300a induction was also triggered by the hypoxia-mimetic agent, desferrioxamine. CD300a exhibited both activating and inhibitory potential, differentially regulating CCL20 and vascular endothelial growth factor pro-inflammatory cytokine production by monocytes/macrophages upon triggering by an agonist Ab. These results suggest that CD300a induction by the hypoxic environment represents a mechanism of regulation of monocyte/macrophage pro-inflammatory responses at pathologic sites.
Introduction
Mononuclear phagocytes (MP) are important regulators of immunity and inflammation. They are recruited as primary monocytes (Mn) from the circulation to sites of infection, inflammation and tumor growth, where they terminally differentiate into macrophages (Mf).1–4 Mn/Mf reactivity in pathologic tissues is tightly regulated by a balance between stimulatory and inhibitory signals of various nature, such as cytokines, damage- and pathogen-associated molecular patterns, and cell metabolites, that, on the one hand, promote their responses and, on the other hand, prevent excessive activation.4–8 These signals are transduced by a defined repertoire of activating and inhibitory cell surface receptors, whose deregulated expression can lead to an aberrant response resulting in the amplification of inflammation or establishment of immune escape mechanisms.9,10 Among them, the immunoregulatory signaling (IRS) receptors play a major role in the regulation of MP responses in diseased tissues.11,12 Characterization of the expression and function of these receptors is thus required for a better understanding of the regulation of Mn/Mf activity at pathologic sites.
The leukocyte membrane Ag, CD300a (also known as CMRF-35 H and IRp60) is a member of the Ig-like CD300 family of paired activating/inhibitory IRS receptors clustered on human chromosome 17. 12 CD300a is broadly expressed on cells of both myeloid and lymphoid lineages, 12 and differently modulated upon stimulation with a variety of immunoregulators or pathogenic stimuli.13–21 CD300a plays a role in the regulation of immune responses, acting predominantly as an inhibitory receptor. Its engagement by an agonist mAb decreases NK cell cytotoxicity,14,22 inhibits IgE-induced mast cell and basophil degranulation,17,21 T-cell proliferation and IFNγ secretion, 19 and B-cell receptor-mediated signaling, 20 reduces FcgRIIa-triggered reactive oxygen species production and Ca2+ flux in neutrophils, 16 and suppresses eosinophil survival, migration and inflammatory mediator production triggered by eotaxin, IL-5 and granulocyte macrophage colony-stimulating factor. 18 Furthermore, in vivo studies in mice with bispecific Abs have shown that CD300a triggering can reverse tissue remodeling and airway inflammation in a model of asthma, 23 and abrogate IgE-mediated allergic reactions 17 and stem cell factor-induced cutaneous anaphylaxis. 24 However, CD300a is also endowed with an activating potential, as shown by its ability to induce IFNα/β secretion by CpG-stimulated human plasmacytoid dendritic cells (DCs). 15 CD300a is expressed on MP,13,25,26 but its regulation and function in these cells remain to be elucidated.
Low partial oxygen pressure (pO2, hypoxia) is a common feature of many pathological situations3,4,27–29 and an important regulator of MP recruitment, gene expression profile, differentiation and functions.1–3,30–34 Response to hypoxia is mediated primarily by a family of hypoxia-inducible transcription factors (HIFs), composed of the constitutive HIF-1β subunit and an O2-sensitive α subunit (HIF-1α,2α), which binds to and transactivates the hypoxia responsive element (HRE) present in the promoter of many hypoxia-inducible genes, activating transcription.2,29,35 HIF-1α/2α accumulation in response to low pO2 was demonstrated in vitro and in vivo in Mn/Mf infiltrating avascular and hypoxic/necrotic areas of diseased tissues,8,31,32,36 and HIF-α relevance for their inflammatory and immunoregulatory functions was reported.2–4,34,36,37
Recent gene expression profiling studies demonstrated that hypoxia can finely tune the expression of genes coding for IRS receptors in MP.30,32,38,39 The aim of this work was to characterize the regulation of CD300a expression by hypoxia in Mn and Mn-derived Mf (MDMs) and to establish its biological functions under conditions of low pO2. We provide evidence that CD300a is a hypoxia-inducible gene and a regulator of pro-inflammatory cytokine production by these cells. These results point to a role for this receptor in tuning MP pro-inflammatory responses at hypoxic sites.
Materials and methods
Cells and culture conditions
Peripheral blood was obtained by platelet apheresis of healthy donors according to the Gaslini’s Ethics Committee-approved protocol, and PBMCs were isolated by density gradient centrifugation over a Ficoll cushion (Ficoll-Paque PLUS, Milano, Italy). Mn were purified by counter-current centrifugal elutriation using a Beckman JE-6 elutriation chamber and Avanti J-20XP rotor system (Beckman, Milano, Italy), followed by magnetic activated cell sorting magnetic bead separation (Human Monocyte Isolation kit-II; Miltenyi Biotec, Bologna, Italy). MDMs were obtained by Mn adherence for 5 d to tissue culture plates (BD Falcon, Sacco, Milano, Italy) in RPMI 1640 (Euroclone, Celbio, Milano, Italy) supplemented with 2.5% human AB serum. Mn and MDM purity was confirmed by positivity for the pan-monocytic lineage markers, CD14 and CD68, which ranged between 92% and 98% among different donors in both cell populations. MDM differentiation was established by morphological criteria, such as cytoplasmic volume, shape, spreading, and adherence to plastic surface, assessed under a phase-contrast microscope (Olympus CKX41 equipped with an Ultra20 Olympus Photocamera for image capturing; Olympus, Milano, Italy) and by flow cytometric analysis of the cell forward side scatter (FSC) and side scatter (SSC) properties on a FACScan to measure size and granularity (Supplementary Figure S1). The human myelomonocytic cell lines THP-1 and U937 were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA). For experimental purposes, cells were cultured in RPMI 1640 supplemented with 10% heat-inactivated FCS (HyClone, Celbio, Milano, Italy), 2 mM
Synovial fluid (SF) samples were obtained at the time of therapeutic knee arthrocentesis from children affected by oligoarticular juvenile idiopathic arthritis (JIA) 4 and collected into sodium–heparin tubes under vacuum. Informed consent was obtained according to the procedure approved by the Gaslini’s Ethics Committee. pO2 levels in SF samples were monitored with the O2 analyzer to confirm hypoxic conditions, and specimens were handled in the anaerobic incubator to prevent cell re-oxygenation, as detailed. 31 Mononuclear cells (SFMCs) were isolated over a Ficoll cushion, and Mn cells were purified from SFMCs by adherence to tissue culture plates (Costar, Celbio, Milano, Italy) for 1 h at 37℃ (purity 93%) 31 and re-suspended in complete medium, as detailed. 31
Flow cytometry
Flow cytometry was performed as described. 38 For the detection of surface markers, cells were re-suspended with FACS buffer (PBS supplemented with 0.2% BSA, 0.01% NaN3) and incubated with the fluorochrome-conjugated mAbs, anti-CD14-PE and anti-CD300a (IRp60, clone P192 produced as described in Cantoni et al. 14 ), the isotype-matched control Abs, mouse IgG2a-PE (Biolegend, Campoverde, Milano, Italy) and mouse IgG2a (R&D Systems, Space Import Export, Milano, Italy), and the secondary FITC-conjugated goat anti-mouse Ab (Invitrogen Life Technologies, Monza, Italy) for 30 min at 4℃, after blocking nonspecific sites with rabbit IgG (Sigma, Milano, Italy). Intracellular staining with anti-CD68-FITC (Dako, Milano, Italy) or mouse IgG1-FITC (Dako, Milano, Italy) was performed after cell fixation in 4% paraformaldehyde and permeabilization in PBS containing 1% AB serum and 0.1% saponin (Sigma, Milano, Italy). Fluorescence was quantitated on a FACSCalibur flow cytometer equipped with CellQuest software (BD Biosciences, Milano, Italy). Cells were gated according to their light-scatter properties to exclude debris.
Western blot analysis
Whole protein extracts (100 µg) were electrophoresed on 10% SDS-PAGE and electroblotted to polyvinylidene membranes (Millipore, Milano, Italy). A prestained protein marker (175–6.5 ku) (New England Biolab, Ipswich, UK) was run as a molecular size standard. Immunoblotting was performed with rabbit anti-human HIF-1α (BD Biosciences, Milano, Italy) and anti-β-actin mAb (Santa Cruz Biotechnology, DBA Italia, Milano, Italy), as an internal loading control, as described previously. 40 Chemiluminescence detection was carried out with peroxidase-conjugated goat anti-rabbit Ab (Sigma, Milano, Italy), using an ECL kit (Pierce, Celbio, Milano, Italy), according to the manufacturer’s instructions.
RT-PCR
Total RNA was purified using the RNeasy MiniKit from Qiagen, and controlled for integrity with an Agilent Bioanalyzer 2100 (Agilent Technologies, Waldbroon, Germany). RNA was quantified by NanoDrop (NanoDrop Technologies, Wilmington, NC, USA) and reverse-transcribed into double-stranded cDNA on a GeneAmp PCR System 2700 thermal cycler (Applied Biosystems, Milano, Italy) using the SuperScript Double-Stranded cDNA synthesis kit (Invitrogen, Monza, Italy). qRT-PCR was performed on a 7500 Real Time PCR System (Applied Biosystems, Milano, Italy) in triplicate for each target transcript using SYBRGreen PCR Master Mix and sense/antisense oligonucleotide primers synthesized by TIBMolbiol (Genova) or from Qiagen (Milano, Italy) (RSP18), as detailed. 30 Expression data were normalized on the values obtained in parallel for three reference genes (actin-related protein 2/3 complex 1B, ARPC1B; lysosomal-associated multispanning membrane protein-5, LAPTM5; ribosomal protein S18, RPS18) using the Bestkeeper software, and relative expression values were calculated using Q-gene software. 30
CD300a cross-linking
Twelve-well flat-bottom tissue culture plates (Corning Life Sciences, Milano, Italy) were precoated with 10 µg/ml of goat anti-mouse IgG-F(ab’)2 (Sigma, Milano, Italy) overnight at 37℃. The following day, plates were washed three times with PBS and incubated with either agonist anti-CD300a mAb (clone P19214), or control IgG2a for 4 h at 37℃ before seeding 1 × 106 Mn or MDM/well/ml. Plates were spun at 126g to engage CD300a and then incubated for 24 h under normoxia or hypoxia. Culture supernatants were harvested by centrifugation and tested for CCL20 and VEGF content by ELISA. Total RNA was purified from triggered cells and analyzed by qRT-PCR.
ELISA
Secreted CCL20 and vascular endothelial growth factor (VEGF) were measured in supernatants from Mn and MDMs triggered with anti-CD300 mAb or control mAb by specific ELISA (R&D Systems Space, Milano, Italy). The OD of the plates was determined using a Spectrafluor Plus plate reader from TECAN (Milano, Italy). All assays were done in duplicate and repeated three times. Data were analyzed with Graph Pad Prism 5 software v. 5.00 (GraphPad Software, La Jolla, CA, USA).
Statistical analysis
Data are the mean ± SEM of three independent experiments, unless differently specified. The paired Student’s t-test was used to determine the significance of the result (P ≤ 0.05).
Results
Hypoxia is an inducer of CD300a in human Mn and MDMs
Experiments were carried out to investigate the effects of hypoxia on CD300a expression in primary human Mn. Peripheral blood Mn were purified from six different donors and cultured for 24 h in 1% O2, a condition that effectively modulates gene expression in these cells.
30
CD300a mRNA levels in hypoxic versus normoxic cells were then quantified by qRT-PCR. Expression of HIF-1α and the HIF-1-target gene, VEGF, was assessed in parallel as an index of response to hypoxia.
30
Mn exposure to 1% O2 resulted in HIF-1α protein accumulation (Figure 1A) and VEGF mRNA increase (Figure 1B). This was paralleled by a significant and consistent up-regulation of CD300a transcript levels (Figure 1B), with the extent of CD300a increase ranging from five- to 19-fold over normoxic levels among individual donors. mRNA up-regulation was an early event detectable within 3 h culture (Figure 1C) and was reversible because re-oxygenation by exposure to normal pO2 had already restored CD300a mRNA expression to normoxic levels after 6 h (Figure 1D). To determine whether CD300a transcript up-regulation by hypoxia was associated with increased surface protein expression, CD300a staining was measured by flow cytometry (Figure 1E). The left panel depicts CD300a staining pattern of cells from a representative donor, and the right panel shows the percentage of CD300a + Mn in the six donors examined by qRT-PCR (shown in Figure 1B). Seventy-nine to 88% Mn constitutively expressed moderate surface levels of CD300a under normoxic conditions (right panel) that were significantly increased upon exposure to hypoxia, both in terms of percentage of positive cells (P = 0.001) and mean fluorescence intensity.
CD300a up-regulation by hypoxia in primary human Mn. Human peripheral blood Mn purified from six different donors were cultured under hypoxic (Hypo, +) or normoxic (Hypo, –) conditions. (A) Whole protein extracts (100 µg) were prepared after 24 h culture, resolved on 10% SDS-PAGE and analyzed for HIF-1α protein expression by hybridization with a specific Ab. β-Actin was evaluated in parallel as a loading control. Immunoblots from three different donors are shown. (B) CD300a mRNA expression (solid bars) was assayed by qRT-PCR on total RNA from Mn exposed to hypoxia for 24 h. VEGF mRNA levels were assessed in parallel (open bars). Relative transcript expression was calculated in relation to the values obtained for three reference genes, as detailed in the ‘Materials and methods’. Data from one representative experiment/donor are expressed as fold increases (1% O2 relative to 20% O2) and represent the mean of triplicate determinations. (C) Mn cultured under hypoxia (Hypo) for the indicated time points or (D) exposed to hypoxia (Hypo, +) for 24 h and subsequently re-oxygenated by exposure to normoxia (Reox) for 6 h were analyzed for CD300a mRNA levels by qRT-PCR. Data from one representative experiment of three performed are expressed as mean normalized expression relative to the values obtained for three reference genes. Fold increase values in hypoxic versus normoxic (considered as equal to 1) cells are indicated by a number associated with each bar. (E) CD300a surface expression was evaluated by flow cytometry. Normoxic and hypoxic cells were stained with anti-CD300a mAb followed by FITC-conjugated goat anti-mouse Ab and analyzed on a FACScan, as specified in the ‘Materials and methods’. Left panels: data are plotted as fluorescence intensity on a log scale versus the number of positive cells. Open histograms represent the fluorescent profile of CD300a-expressing cells; solid histograms represent the fluorescent profile of cells stained with an isotype-matched control Ab. The percentage of CD300a+ cells and the mean fluorescence intensity (in parentheses) are indicated. Results are from one representative experiment of six performed. Right panel: data are shown as box plots and are expressed as percentage of CD300a+ cells in normoxic (Normo) and hypoxic (Hypo) cells from six different donors. Boxes contain the values falling between the 25th and 75th percentiles, horizontal lines represent median values and whiskers (lines that extend from the boxes) represent the highest and lowest value for each group. Values significantly different from normoxic cells according to the Student’s t-test: **P ≤ 0.01.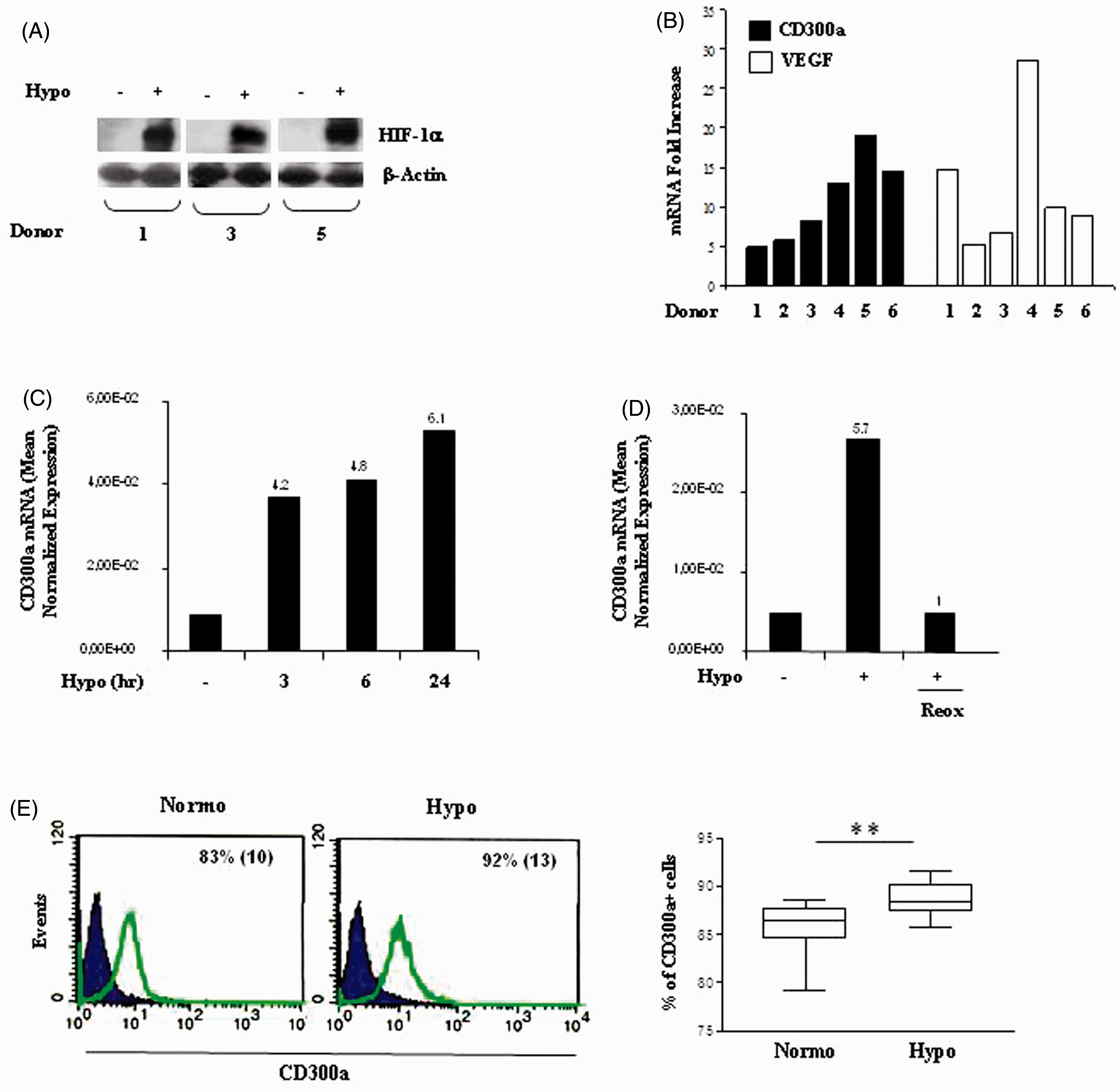
Mn differentiated into Mf in hypoxic areas of pathologic tissues.1,2 It was of interest to investigate CD300a expression in MDMs derived from primary Mn under long-term hypoxic conditions (hereafter referred to as ‘chronic hypoxia’). As depicted in Supplementary Figure S1, MDMs generated after 5 d culture under normal and reduced pO2 displayed morphologic features (panel A) and FSC/SSC profiles (panel B) typical of mature macrophages,41,42 and up-regulated the CD68 marker to a similar extent with respect to matched Mn (panel C), indicating that chronic hypoxia does not affect the differentiation process, which is consistent with previous observations.
42
MDM adaptation to chronic hypoxia resulted in HIF-1α protein stabilization (Figure 2A), VEGF mRNA increase (Figure 2B) and CD300a transcript up-regulation (Figure 2B), with CD300a induction ranging from two- to sixfold in different donors. The role of hypoxia as a stimulus for CD300a in MDMs was further supported by data showing that CD300a mRNA was also increased in MDMs exposed to 24 h hypoxia after differentiation under normoxic conditions, although at levels lower than those detectable in matched MDMs generated under chronic hypoxia (Figure 2C). As shown by immunophenotypic analysis of CD300a surface expression (Figure 2D), the percentage of MDMs constitutively expressing CD300a under normoxic conditions was lower than that of primary Mn and extremely variable, ranging from 1% to 56% in different donors (right panel). MDM generation under chronic hypoxia led to a significant increase (P = 0.003) in the percentage of CD300a-positive cells, with up to 90% of hypoxic MDMs expressing the surface protein similarly to what shown in hypoxic Mn.
Hypoxia-dependent increase of CD300a in MDMs. MDMs were generated under normoxic or hypoxic conditions by Mn adherence for 5 d to tissue culture plates, as detailed in the ‘Materials and methods’. (A) HIF-1α protein expression was assessed by Western blot analysis, as specified in the legend to Figure 1A. β-Actin expression was evaluated in parallel to control for protein loading. Immunoblots from three different donors are shown. (B) Total RNA was tested for CD300a expression by qRT-PCR, as detailed in the legend to Figure 1B. Data from one representative experiment/donor are expressed as fold increases (1% O2 relative to 20% O2) and represent the mean of triplicate determinations. (C) CD300a mRNA levels were determined in MDMs generated under normoxia (N 0–5) or hypoxia (H 0–5), or exposed to 24 h hypoxia after generation under normoxia for 4 d (H 4–5). Data from one representative experiment of three performed are expressed as in the legend to Figure 1C, D. Fold increase values in hypoxic versus normoxic (considered as equal to 1) cells are indicated by a number associated with each bar. (D) CD300a surface expression was evaluated by flow cytometry in the same MDMs preparations shown in panel B, and data are expressed as in the legend to Figure 1E. Values significantly different from normoxic cells according to the Student’s t-test: **P ≤ 0.01.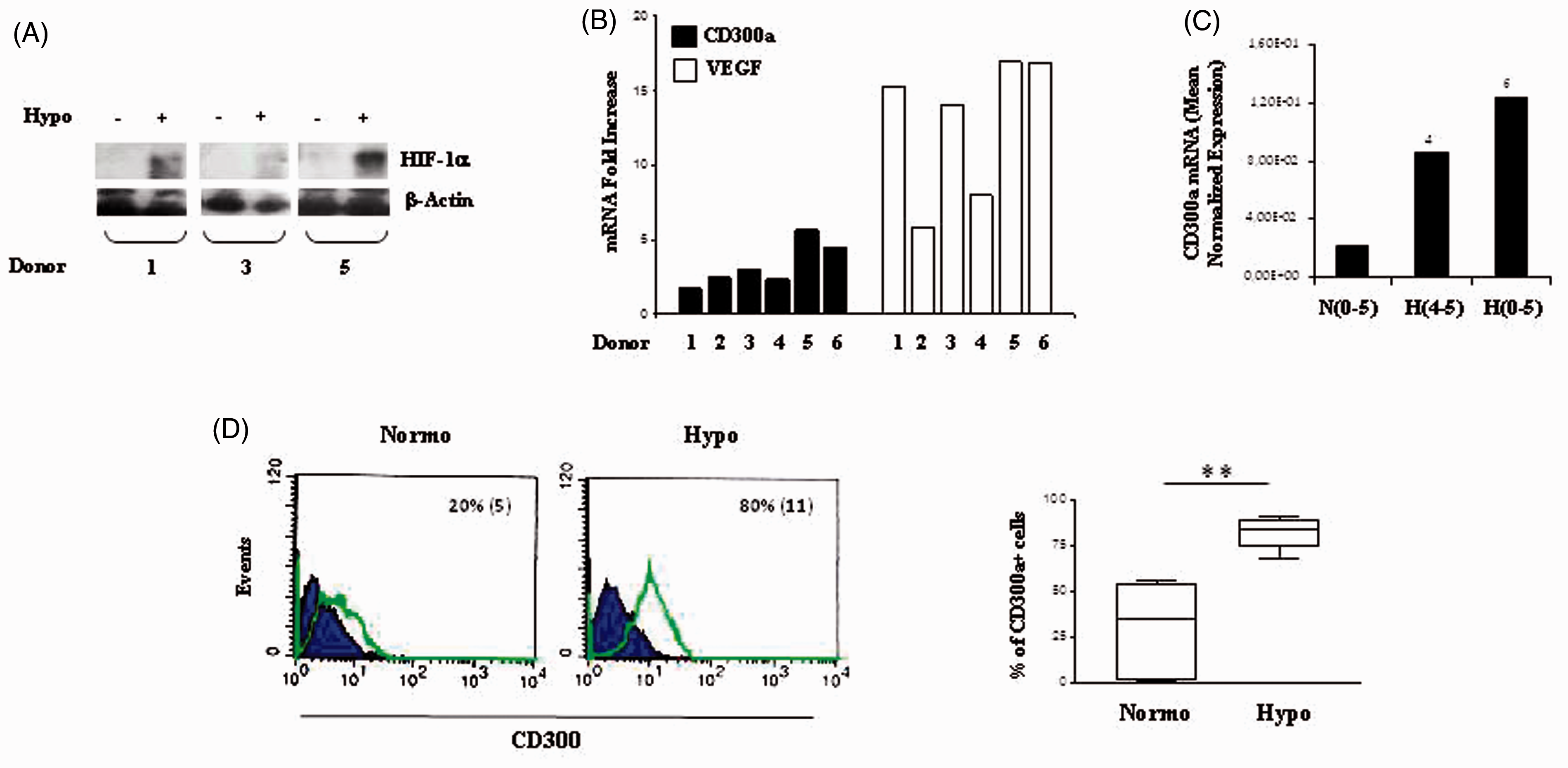
To investigate CD300a regulation by hypoxia in Mn cells at earlier stages of differentiation, CD300a mRNA expression was assessed in the human myelomonocytic cell lines, THP-1 and U937, cultured for different time points in 1% O2. As shown in Figure 3A, THP-1 exposure to hypoxia had resulted in a marked up-regulation of CD300a mRNA after 6 h culture, with a further increase at 24 h, and a strong CD300a induction occurred in U937 after 24 h treatment.
Hypoxic regulation of CD300a mRNA expression in promonocytic cell lines and Mn cells isolated from JIA-SF. (A) THP-1 and U937 cell lines were cultured under hypoxic conditions (Hypo) for the indicated time points, and CD300a mRNA expression was determined by qRT-PCR on total RNA. Data from one representative experiment of three performed are expressed as in the legend to Figure 1C, D. Fold increase values in hypoxic versus normoxic (considered as equal to 1) cells are indicated by a number associated with each bar. (B) Mn cells were purified from the SF of three JIA patients, as detailed in the ‘Materials and methods’, and left untreated (Fresh) or cultured for 16 h under normoxic (Reox) or hypoxic (Hypo) conditions. Left panel: CD300a transcript levels were quantified by qRT-PCR in total RNA from fresh (solid bars), re-oxygenated (grey bars), or hypoxic (open bars) cells. Data are expressed as in the legend to Figure 1C, D. mRNA fold-decrease values in re-oxygenated relative to fresh cells (considered as equal to 1) are indicated by a number associated with each bar. Right panel: HIF-1α expression was assessed by Western blot on whole protein extracts (100 µg) from fresh, re-oxygenated, or hypoxic SF-Mn as described in the legend to Figure 1A. β-Actin was evaluated in parallel as a loading control.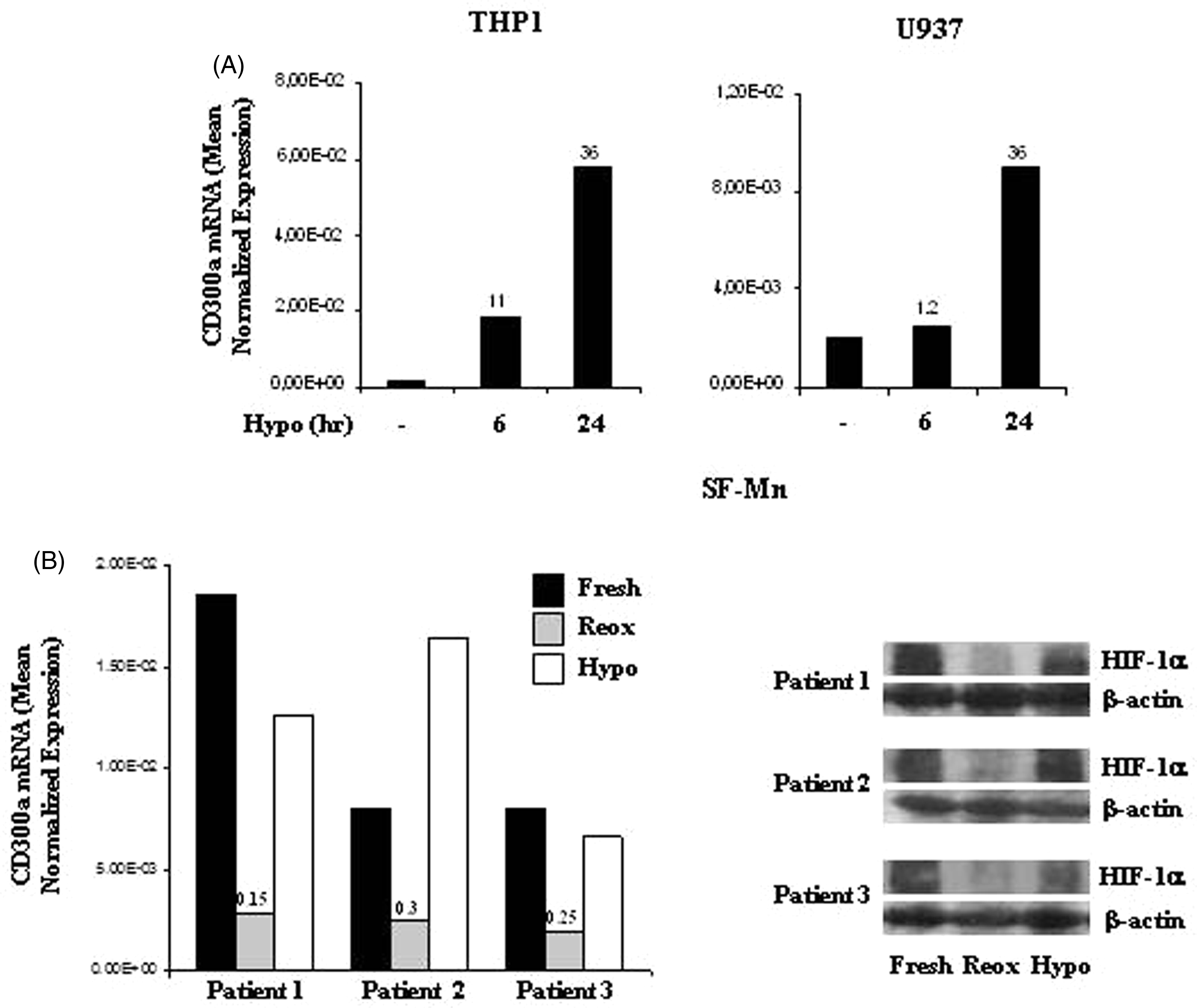
CD300a has been implicated in the development of chronic inflammatory diseases, including rheumatoid arthritis. 43 Hypoxia is a common feature of the inflamed rheumatoid synovium, 4 and Mn/Mf are enriched in arthritic joints where they adapt to the local hypoxic environment.4,31 To assess the effects of in vivo hypoxia on CD300a expression, Mn cells were isolated from the SF (SF-Mn) of patients affected by JIA, and CD300a mRNA levels were compared by qRT-PCR in SF-Mn freshly purified, allowed to re-oxygenate by exposure to 20% O2 for 16 h, or maintained under a controlled hypoxic environment (1% O2) for the same length of time (Figure 3B, left panel). HIF-1α protein accumulation was assessed in parallel to confirm adaptation to the hypoxic environment (Figure 3B, right panel). Elevated levels of HIF-1α were present in freshly purified SF-Mn and associated with CD300a mRNA expression. Re-oxygenation, which inhibited HIF-1α basal expression, caused a pronounced down-regulation of CD300a transcript levels relative to fresh cells (up to 85% reduction), whereas maintenance of the hypoxic environment sustained both HIF-1α and CD300a expression at levels comparable to those detected in fresh cells. These data support a role for intra-articular hypoxia as a stimulus for CD300a in SF-Mn and indicate a functional correlation between HIF-1α accumulation and CD300a expression.
Given the presence of a HRE sequence in the CD300a gene promoter,39,44 we next investigated the role of HIF-1α in CD300a inducibility by hypoxia by assessing the effects of echinomycin, a specific inhibitor of HIF-1α binding to HRE,39,45 on CD300a mRNA expression in Mn cultured under hypoxia for 6 and 24 h. As shown in Figure 4, treatment with echinomycin resulted in a dose-dependent decrease of CD300a mRNA levels, with a 70% reduction observed with 10 nmol/l of the drug by 6 h and almost complete inhibition achieved after 24 h.
Down-regulation of hypoxia-induced CD300a mRNA expression by echinomycin. Primary Mn were treated with the indicated concentrations of echinomycin for 6 (full triangles) and 24 h (open triangles) under hypoxic conditions. Total RNA was isolated and analyzed for CD300a mRNA expression by qRT-PCR, as described in the legend to Figure 1B. Results from one representative experiment of three performed are presented as percentage of mRNA remaining after echinomycin treatment, taking as 100% the expression levels of untreated hypoxic cells.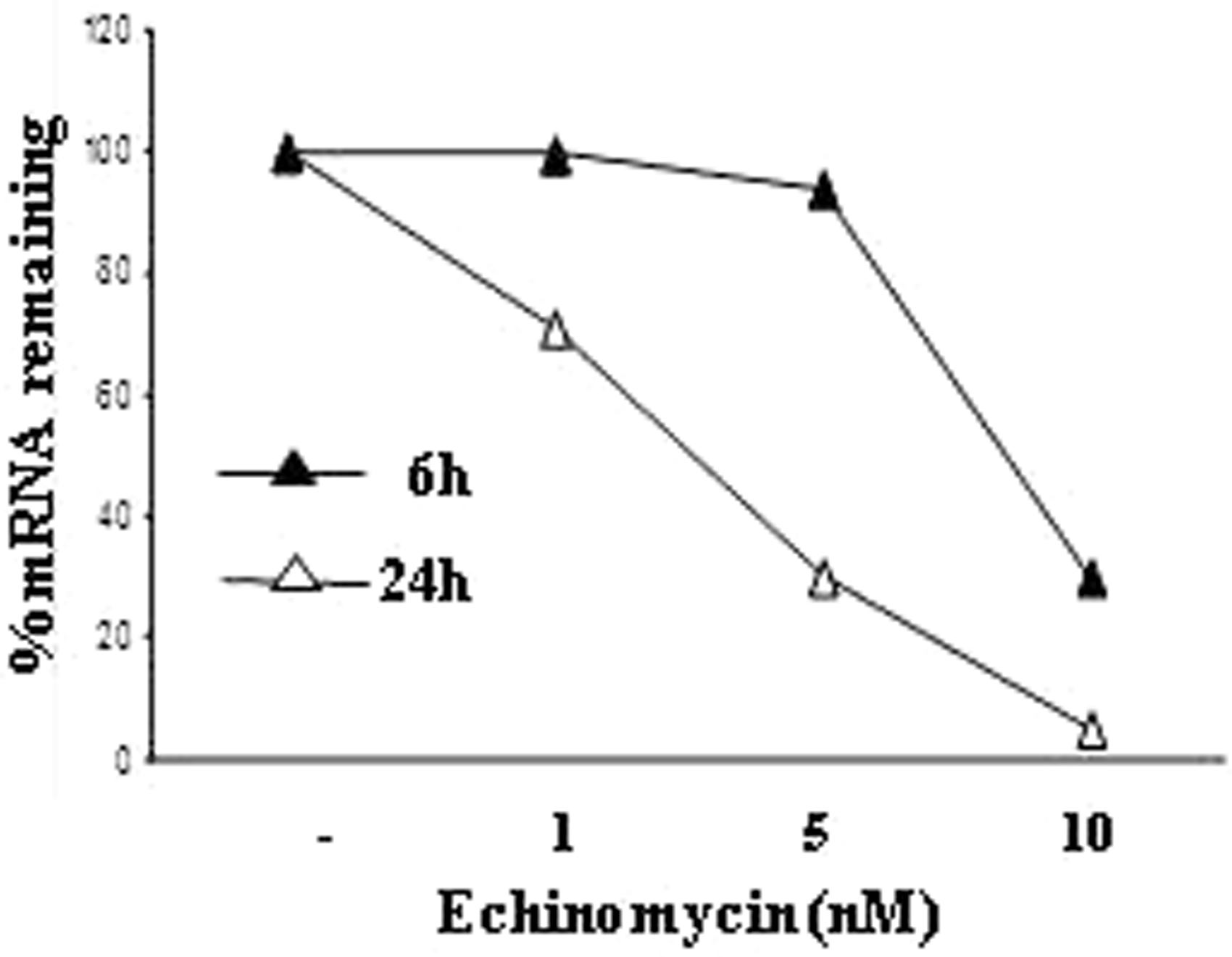
Taken together, these data indicate that CD300a gene inducibility by hypoxia is a common event in human Mn lineage cells at different stages of differentiation and that it is mediated by HIF-1α.
DFX increases CD300a expression in Mn and MDMs
The iron chelator DFX is a known hypoxia-mimetic agent that, as well as hypoxia, has the property of inducing gene expression under normal pO2 through the HIF-1α/HRE system in several cell types, including MP.40,46–49 Experiments were carried out to assess the effects of DFX on CD300a expression. Primary Mn and matched MDMs were stimulated for 24 h with 200 µM DFX, a concentration previously shown to modulate gene expression in these cells,7,47–49 and CD300a mRNA levels were quantified by qRT-PCR. HIF-1α protein expression was assessed in parallel. As shown in Figure 5, which depicts the results obtained in one representative donor, HIF-1α protein accumulation was triggered by DFX in both Mn and MDMs (Figure 5A) and was associated with CD300a mRNA up-regulation (Figure 5B). Similar results were obtained with other two donor-derived cell preparations (data not shown).
CD300a up-regulation by DFX in Mn and MDMs. Primary Mn and matched MDMs derived from 5 d in vitro culture under normoxia were stimulated with 200 µM DFX for 24 h. (A) HIF-1α expression was assessed by Western blot analysis as detailed in the legend to Figure 1A. β-Actin expression was evaluated in parallel to control for protein loading. A representative Western blot of three performed with cells from different donors is shown. (B) CD300a transcript levels were evaluated by qRT-PCR in cells treated with DFX in the presence (+) or absence (–) of 300 µM FeSO4. Data from one representative experiment of three performed are expressed as in the legend to Figure 1C, D. Fold increase values in DFX- versus medium- (considered as equal to 1) treated cells are indicated by a number associated with each bar. (C) CD300a surface expression was evaluated by flow cytometry, as described in the legend to the left panels of Figure 1E. Histograms depict the results obtained from one of three independently tested donors. The percentage of CD300a+ cells and the mean fluorescence intensity (in parenthesis) are indicated.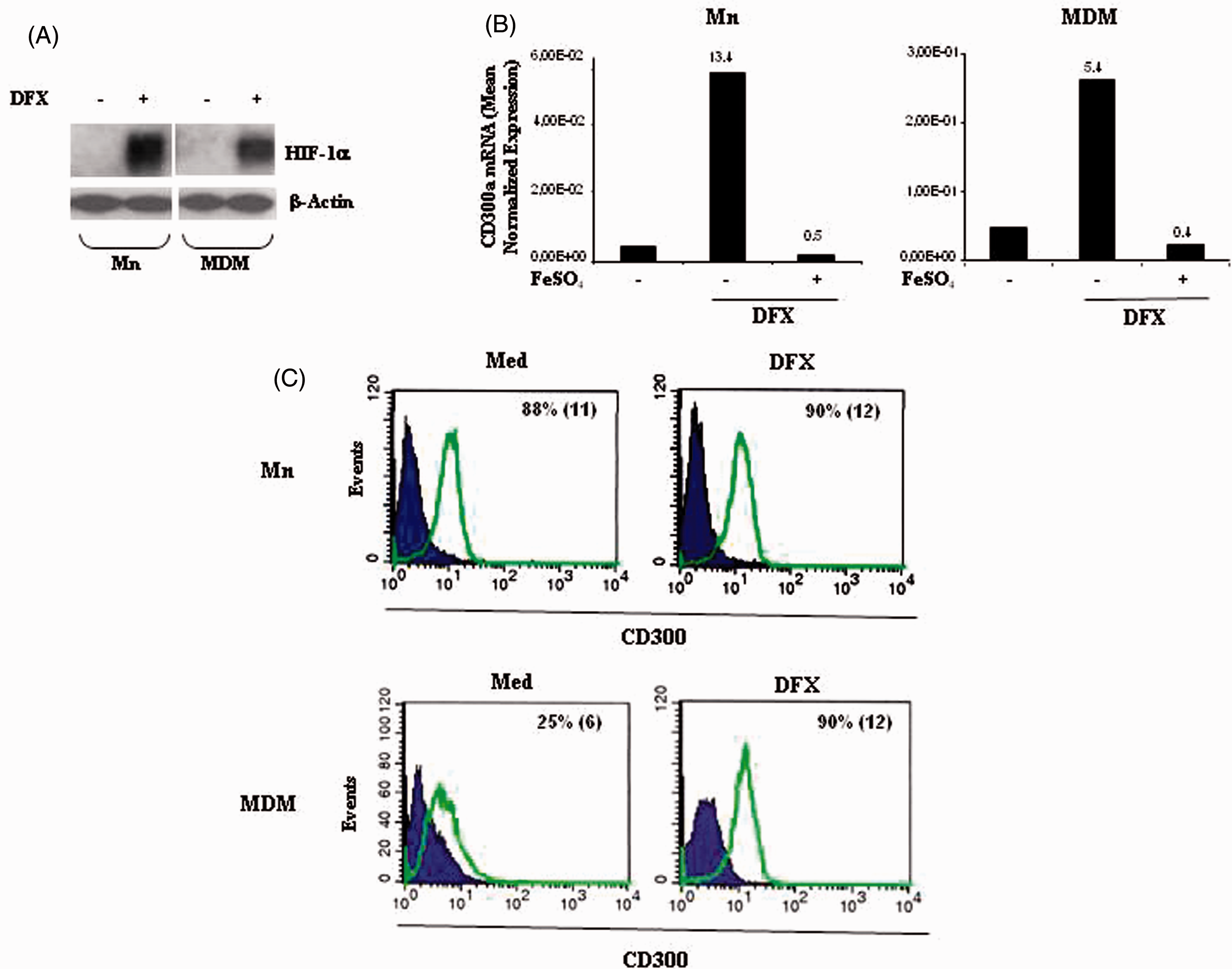
To determine the requirement for iron chelation in DFX-dependent CD300a mRNA induction, Mn and MDMs were treated with DFX in the presence or absence of 300 µM FeSO4, a concentration able to counteract DFX biological activities in MP,7,47–49 and CD300a mRNA levels were assessed by qRT-PCR. As shown in Figure 5B, CD300a transcript induction by DFX was completely reversed by the addition of FeSO4, confirming that DFX acts by targeting intracellular iron.
DFX treatment also led to an increase in CD300a surface expression that was particularly evident in MDMs with up to 90% of cells expressing the protein (Figure 5C), similarly to what shown for hypoxia.
These data indicate that DFX is active on Mn lineage cells as an inducer of CD300a gene expression
CD300a cross-linking on Mn and MDMs differentially modulates CCL20 and VEGF production
To investigate CD300a biological functions in MP, primary Mn and MDMs were cross-linked with a specific anti-CD300a agonist mAb for 24 h under normoxic and hypoxic conditions, and production of the two pro-inflammatory cytokines, CCL20 and VEGF, was assessed. Cross-linking experiments were carried out in parallel, using DFX as a stimulus. As shown in Figure 6A, Mn exposure to hypoxia or treatment with DFX resulted in a marked up-regulation of CCL20 mRNA expression compared with normoxic levels, confirming previous findings.30,47,50 CD300a engagement in the absence of other stimuli increased CCL20 transcripts with respect to an isotype-matched control Ab, although at levels lower (∼ threefold) than those induced by stimulation with hypoxia (∼ 10-fold) or DFX (∼ 19-fold). Interestingly, CD300a cross-linking concomitant to hypoxia stimulation resulted in a synergistic induction of CCL20 mRNA expression, which was approximately threefold higher than that exerted by hypoxia alone (Figure 6A). In contrast, CD300a cross-linking in conjunction with DFX treatment reduced by DFX-dependent CCL20 mRNA expression by ∼ 70% (Figure 6A). In keeping with mRNA data, hypoxia and DFX stimulated CCL20 secretion, and CD300a engagement increased CCL20 release triggered by hypoxia significantly (P = 0.0267), whereas it significantly reduced (P = 0.05) CCL20 induction by DFX (Figure 6B). A marked up-regulation of VEGF mRNA expression was observed in primary Mn in response to hypoxia (∼ 13-fold) and DFX (∼ 24-fold) (Figure 6C) and was associated with increased protein secretion (Figure 6D). Interestingly, CD300a cross-linking in the absence of other stimuli induced a slight increase of VEGF production, but significantly amplified VEGF induction by both hypoxia (P = 0.0067) and DFX (P = 0.038) (Figure 6D).
Differential modulation of CCL20 and VEGF production by CD300a cross-linking on primary Mn. Mn were seeded onto plates precoated with a specific anti-CD300a agonist mAb (+) or a control IgG1 (–) and goat anti-mouse IgG-F(ab′)2, and cultured for 24 h under hypoxic conditions (Hypo, +) or in the presence of DFX (+). Cells were harvested and analyzed for (A) CCL20 and (C) VEGF mRNA expression by qRT-PCR. Relative transcript expression was calculated as detailed in the legend to Figure 1B. Data shown are expressed as fold increase relative to control Ig-cross-linked cells (considered equal to 1) and are representative of one of three experiments performed. (B) CCL20 and (D) VEGF content in conditioned medium were assayed by ELISA. Results are expressed as pg/106 cells/ml and represent the mean ± SEM of three different experiments. Values significantly different from those of cells cross-linked with isotype-matched Ab according to the Student’s t-test: *P ≤ 0.05; **P ≤ 0.01.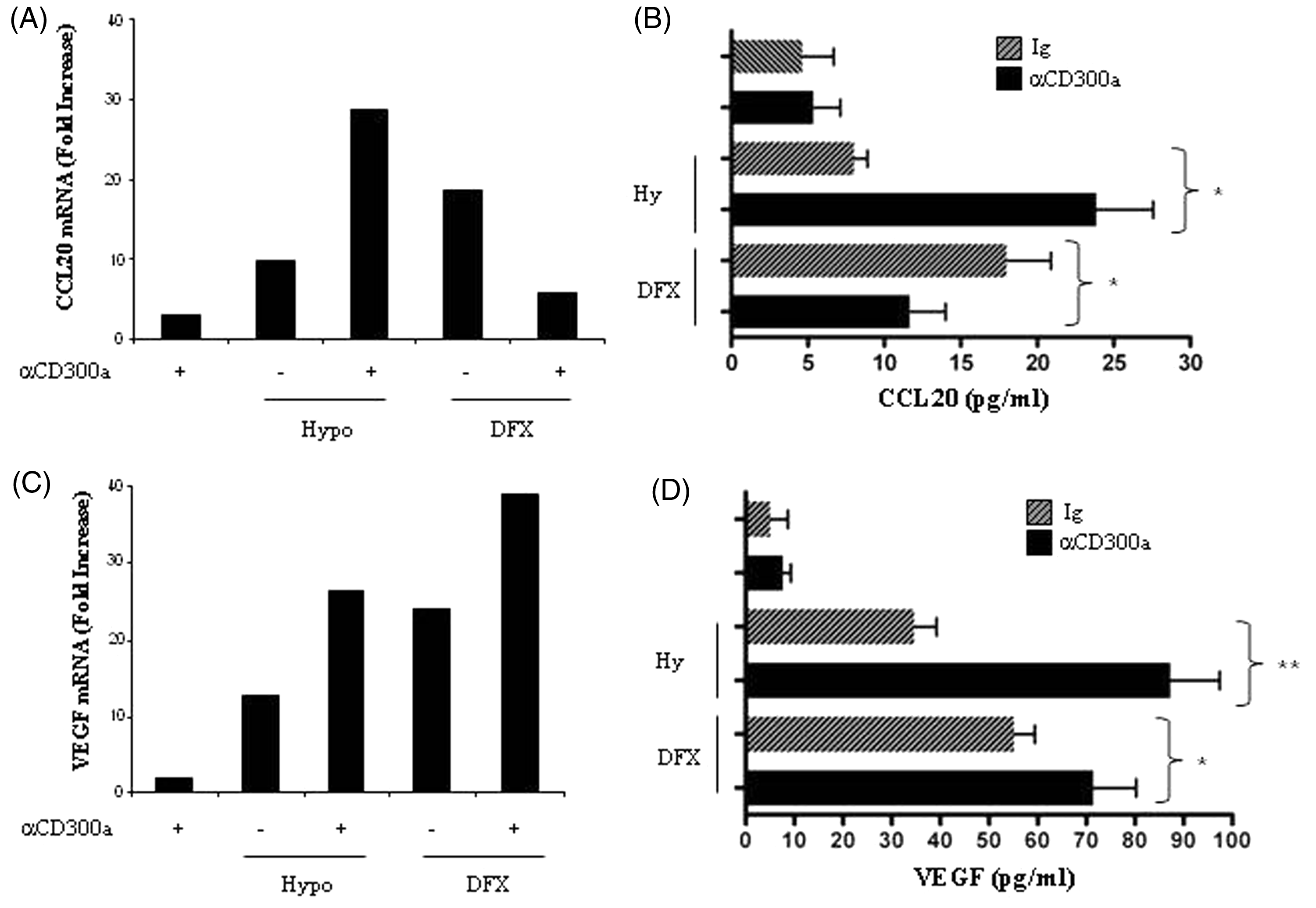
In contrast to what was observed in Mn, CD300a triggering in MDMs did not affect CCL20 and VEGF mRNA expression (Figure 7A, C) and protein secretion (Figure 7B, D), probably because of the low constitutive levels of the molecule on these cells. CCL20 mRNA expression was markedly increased by hypoxia (∼ sevenfold) and at a lower extent by DFX (∼ twofold) (Figure 7A) and associated with increased protein secretion (Figure 7B). MDM treatment with anti-CD300a mAb under hypoxia resulted in a synergistic induction of CCL20 mRNA expression, which was approximately 4.5-fold higher than that exerted by hypoxia alone, whereas no effect was observed on the response to DFX (Figure 7A). Accordingly, CD300a cross-linking enhanced significantly (P = 0.0144) hypoxia-induced CCL20 secretion (Figure 7B). A marked up-regulation of VEGF mRNA expression (Figure 7C) and a significant induction of VEGF secretion (Figure 7D) were detectable upon CD300a triggering in MDMs both during hypoxia exposure (P = 0.036) and DFX treatment (P = 0.0155).
CCL20 and VEGF production regulation by CD300a triggering on MDMs. MDMs were incubated with anti-CD300a Ab (+) or control isotype IgG1 (–) and goat anti-mouse IgG-F(ab′)2 for 24 h under hypoxic conditions (Hypo, +) or in the presence of DFX (+). (A) CCL20 mRNA expression, (C) VEGF mRNA expression and (B, D) cytokine release into the supernatant were analyzed 24 h after stimulation, as detailed in the legend to Figure 6. Values significantly different from those of cells cross-linked with isotype-matched Ab according to the Student’s t-test: * P ≤ 0.05.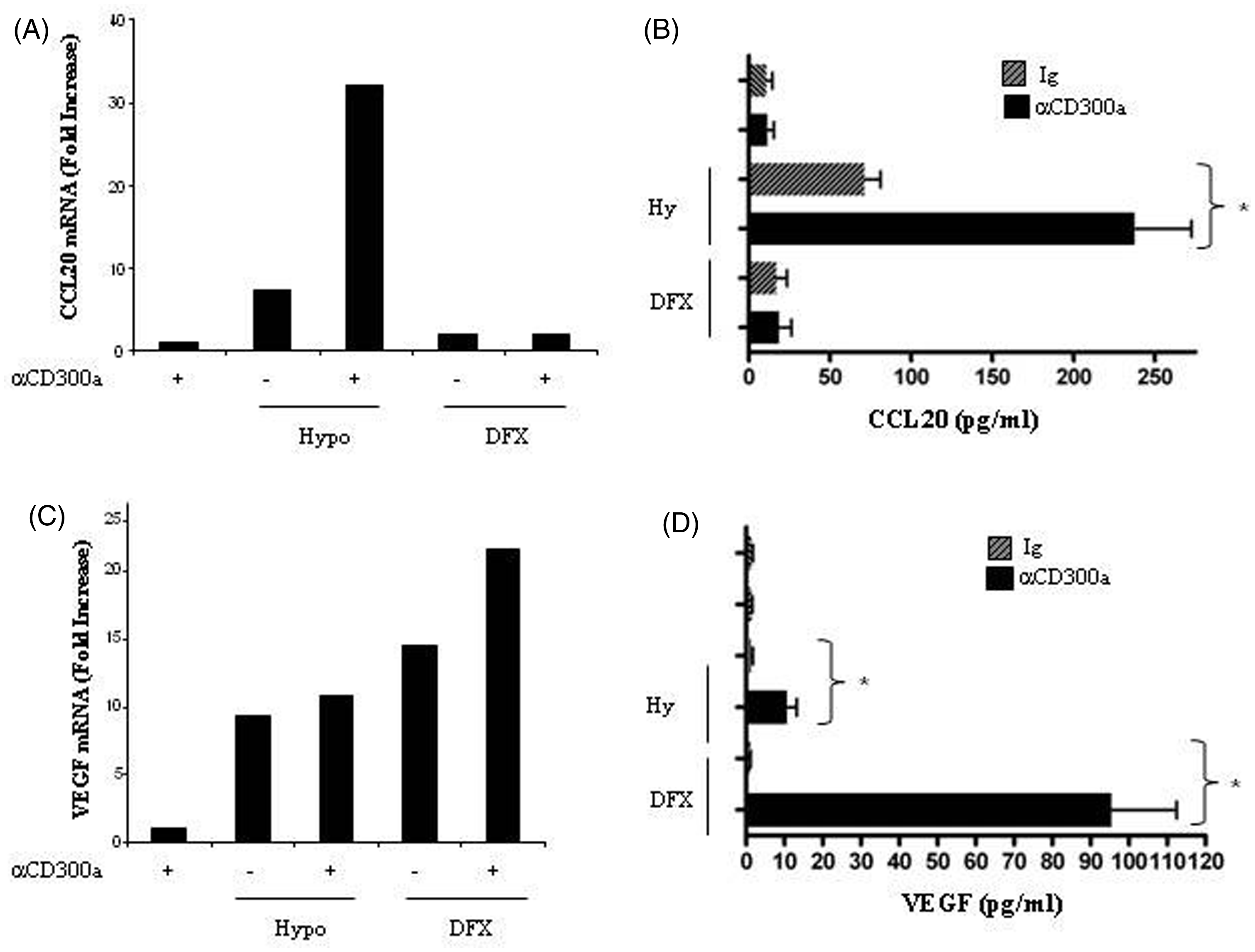
We can conclude that CD300a exerts both activating and inhibitory effects upon ligation on MPs, finely tuning cytokine production induced by hypoxia and DFX.
Discussion
IRS receptors play a major role in the regulation of MP responses in diseased tissues by continuously surveying the extracellular environment and integrating stimulatory and inhibitory signals.10–12 The relevance of hypoxia in strictly controlling the expression of various members of these receptor family in Mn lineage cells was recently demonstrated.30,32,38,39 In this study, we focused on CD300a, demonstrating its inducibility by hypoxia and the hypoxia-mimetic compound, DFX, in both primary human Mn and MDMs and characterizing a role for this molecule in the regulation of pro-inflammatory cytokine production by these cells.
CD300a is expressed in several leukocyte subsets, where it can be modulated by various stimuli, such as mitogens, bacterial products, and pro-inflammatory cytokines.13–21 Our results confirm previous evidence of CD300a expression in MP13,25,51 and identify new stimuli regulating the receptor in these cells. Mn exposure to hypoxia resulted in the increase of CD300a transcripts, which was detectable by 3 h and was counteracted by re-oxygenation, which restored expression levels to those of normoxic cells and was associated with HIF-1α accumulation. The critical role of HIF-1α in mediating CD300a inducibility by hypoxia was supported by the demonstration that Mn treatment with echinomycin, a known specific inhibitor of HIF-1α binding to HRE and transcriptional activity,39,45 prevented CD300a mRNA up-regulation by hypoxia and is consistent with the finding that the CD300a gene promoter contains a HRE sequence.39,44 CD300a transcript increase was also observed in myelomonocytic precursors and in MDMs in response to hypoxia, demonstrating the general inducibility of this gene by the hypoxic environment in Mn lineage cells.
Moderate surface levels of CD300a were constitutively expressed in both primary Mn and MDMs derived from different donors, but the percentage of positive cells was quite different among the two populations, ranging from approximately 80% to 90% in Mn and from 1% to 56% in matched MDMs. These data support previous findings, which suggest that CD300a expression is developmentally regulated in Mn lineage cells, decreasing during Mn differentiation into Mf. 25 mRNA up-regulation by hypoxia was paralleled by increased surface protein expression in both Mn and MDMs. Of note, MDMs generated under chronic hypoxic conditions were induced to express CD300a up to the levels of primary Mn (approximately 90% of cells), indicating that hypoxic stimulation can overcome CD300a developmental down-regulation and that Mf generated at hypoxic sites are characterized by high CD300a expression. Consistent with this conclusion is the observation that Mn cells freshly isolated from the hypoxic synovium of JIA patients expressed high levels of CD300a mRNA that were down-regulated upon re-oxygenation and sustained by maintenance of the hypoxic environment, correlating with HIF-1α protein expression. These findings raise the interesting possibility that CD300a expression in Mn lineage cells varies dynamically in vivo with the degree of local oxygenation, which is quite heterogeneous and rapidly fluctuating in diseased tissues, 5 giving rise to distinct cell subsets potentially endowed with different functional properties.
CD300a mRNA and protein up-regulation was also observed in response to treatment with DFX, a known hypoxia-mimetic stimulus that stabilizes HIF-1α by inhibiting prolyl hydroxylase domain (PHD) enzyme activity and the HIF-1α degradation pathway, 52 and was shown to correlate with HIF-1α accumulation in both Mn and MDMs. This effect seemed to be dependent on the iron-chelating property of this agent because addition of iron sulfate counteracted CD300a mRNA induction by DFX.
CD300a acts predominantly as an inhibitory receptor modulating a broad and diverse array of immune cell processes, including survival, differentiation, chemotaxis, phagocytosis, inflammatory mediator production, and cytotoxic activity.12,14–16,18–21,24 Its inhibitory activity can be ascribed to four putative immunoreceptor tyrosine-based inhibitory motifs (ITIM) present in the cytoplasmic tail, whose phosphorylation after engagement of the receptor starts a cascade of intracellular signaling events promoting cell inhibition.12,14,22 However, ITIM domains have also been found in receptors with no evident inhibitory function or even with activating function, and some ITIM-containing receptors have been shown to alternatively function as inhibitory or activating molecules.10,53 Interestingly, a few members of the CD300 family were reported to present a functional duality being endowed with the ability to deliver both inhibitory and stimulatory signals upon ligation, depending on the cell type and activity analyzed.26,54 Among them, CD300a triggering was shown to exert a reciprocal effect on cytokine production by CpG-stimulated human plasmacytoid DCs, specifically reducing TNF-α and IL-6 production, and increasing IFN-α/β secretion. 15 In this study, we demonstrate that CD300a activation can differentially regulate the production of CCL20 and VEGF in Mn and MDMs. CD300a cross-linking in the absence of other stimuli was able to up-regulate the expression of both cytokines in Mn, but not in MDMs, probably because of the low constitutive surface expression of the protein in these cells. Interestingly, CD300a triggering significantly increased CCL20 and VEGF production driven by hypoxia in both cell types. In contrast, it reduced CCL20 induction by DFX in Mn, without affecting MDM response, and enhanced VEGF production triggered by DFX both in Mn and MDMs. These data, together with recent findings showing that CD300a engagement differentially modulates IL-8 and metalloproteinase-9 induction by distinct TLR ligands in human Mn cell lines, 51 suggest that activation of this receptor in Mn lineage cells can concomitantly exert activating and inhibitory effects on cytokine production synergizing with or antagonizing the effects of different stimuli, potentially resulting in different immunological outcomes. Although the downstream mediators of CD300a inhibitory activity—specifically the SH2-containing tyrosine phosphatases, SHP-1 and SHP-2, have been identified in different cell types, including monocytes,12,14,18,51,55 the signaling events associated with CD300a stimulatory effects have not been elucidated yet. It is conceivable that CD300a interacts with activating signaling molecules through residues in the cytoplasmic domain distinct from ITIMs, similarly to what has been shown for CD300f, which can recruit PI3K in addition to SHP-1 and SHP-2. 25 A possible explanation for the dual activity of the CD300a receptor has been proposed by Martinez-Barriocanal et al., 54 who showed that the CD300 family members can interact with each other through their Ig domain and form homo- and heterosignaling complexes capable of modifying the properties of individual receptors, resulting in different signaling outcomes upon binding by their natural ligand. 26 We can hypothesize that the expression of distinct CD300 family members and/or their dimerization with CD300a is differentially affected by hypoxia and DFX in Mn/Mf, giving rise to heterocomplexes with unique signaling properties; further research is required to address this issue.
CD300a plays a pathogenic role in a number of diseases, including asthma, cutaneous anaphylaxis, pulmonary inflammation, acute lymphoblastic leukemia, ulcerative colitis, Alzheimer’s disease and AIDS,12,26,56 and the potential of CD300a as a therapeutic target for allergic and autoimmune diseases has been proposed.10,12,23,24 CD300a has also been implicated by genome-wide association studies in the development of chronic inflammatory diseases, such as psoriasis, atopic dermatitis and rheumatoid arthritis.25,26,43,56 It is reasonable to suggest that the concerted stimulatory activity of CD300a and hypoxia on CCL20 and VEGF production by Mn/Mf plays a role in the amplification and perpetuation of the inflammatory state in these pathological conditions.1,3,4,27 The CC chemokine, CCL20, is a selective chemoattractant for immature DCs, activated/memory T lymphocytes, and naive B-cells, and modulation of its production is a critical set point for the control of the kinetics and composition of the cellular infiltrate in diseased tissues. 30 However, VEGF not only can drive new blood vessel formation via stimulation of endothelial cell (EC) survival, proliferation and migration, but is also a potent chemoattractant and activating factor for Mn/Mf, both in vitro and in vivo, promoting their recruitment and stimulating pro-inflammatory cytokine production from these cells.1,4,57 We have previously documented the presence of reduced pO2 in the JIA synovium and the contribution of the hypoxic environment to the persistence of synovitis through the stimulation of VEGF and CCL20 production in Mn cells recruited to affected joints.4,31 Sustained CD300a expression in synovial Mn cells by intra-articular hypoxia is likely to represent a mechanism of amplification of their pro-inflammatory functions because its ligation by appropriate ligand(s) present in the inflamed arthritic synovium could trigger further VEGF and CCL20 production. Recent studies have suggested a role for CD300a in the recognition of the aminophospholipids, phosphatidylserine and phosphatidylethanolamine, expressed on the membrane of apoptotic cells at sites of inflammation and/or tissue damage,26,56,58,59 and have shown that hypoxia can induce their externalization on the cell surface. 60 The challenge of future studies will be to clarify the effective role played by these molecules in triggering CCL20 and VEGF production by hypoxic Mn/Mf via CD300a engagement. Furthermore, because aminophospholipids are also exposed on activated T- and B-cells, 58 future investigation of the role of CD300a in the regulation of MP and lymphoid cell cross-talk at hypoxic sites is warranted.
The demonstration that CD300a cross-linking can affect Mn and MDM response to DFX is likewise intriguing, adding new insights into our understanding of the mechanisms by which iron perturbations influence inflammatory processes. 61 Both iron deficiency and overload have been implicated in the regulation of Mn/Mf pro-inflammatory and immunoregulatory responses, and microbial effector functions by triggering the expression of a number of inflammation-related genes.48,61,63 We previously demonstrated that iron chelation by DFX can affect the MP chemotactic network by differentially modulating the production of CCL2, CCL3 and CCL4 in mouse Mf,7,40 and CCL20 and VEGF in human Mn and MDMs. 47 These findings, showing the ability of CD300a to reciprocally modulate CCL20 and VEGF release induced by DFX, define a fine mechanism of control of Mn/Mf pro-inflammatory activity, which may have not only biological, but also potential clinical, implications. DFX is, in fact, used as a therapeutic agent for the treatment of a number of diseases in which deregulated iron metabolism, MP and CD300a play a critical role in pathogenesis, such as Alzheimer’s disease, malignancies, infections, autoimmune disorders, chronic inflammatory cardiomyopathies and arthropathies.64–67
In conclusion, we have characterized a novel property of hypoxia as an inducer of CD300a in Mn/Mf and shed new light on CD300a involvement in the fine-tuning of pro-inflammatory cytokine production by these cells, adding novel mechanistic clues on the contribution of reduced O2 availability to the regulation of MP inflammatory activity at pathologic sites. Further understanding of the way this molecule regulates MP functions may provide an opportunity to manipulate the immune response to therapeutic effects.
Footnotes
Funding
This work was supported by grants from the Italian Association for Cancer Research (AIRC, Code: IG – 10565); the European Network for Cancer Research in Children and Adolescents (ENCCA); the Fondazione Umberto Veronesi. FR was supported by a fellowships from AIRC; F B was supported by a fellowship from the “Fondazione Italiana per la Lotta al Neuroblastoma”.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
