Abstract
Modification of a lipid A moiety in Gram-negative bacterial LPS to a less acylated form is thought to facilitate bacterial evasion of host innate immunity, thereby enhancing pathogenicity. The contribution of less-acylated lipid A to interactions of whole bacterial cells with host cells (especially in humans) remains unclear. Mutant strains of Salmonella enterica serovar Typhimurium with fewer acylated groups were generated. The major lipid A form in wild-type (WT) and the mutant KCS237 strain is hexa-acylated; in mutant strains KCS311 and KCS324 it is penta-acylated; and in KCS369 it is tetra-acylated. WT and KCS237 formalin-killed and live bacteria, as well as their LPS, strongly stimulated production of pro-inflammatory cytokines in human U937 cells; this stimulation was suppressed by TLR4 suppressors. LPS of other mutants produced no agonistic activity, but strong antagonistic activity, while their formalin-killed and live bacteria preparations had weak agonistic and no antagonistic activity. Moreover, these less-acylated mutants had increased resistance to phagocytosis by U937 cells. Our results indicate that a decrease of one acyl group (from six to five) is enough to allow Salmonella to evade human innate immunity and that the antagonistic activity of less-acylated lipid A is not utilized for this evasion.
Introduction
Bacterial components called pathogen-associated molecular patterns (PAMPs) allow mammalian cells to recognize bacterial invasion and trigger innate immune responses. In Gram-negative bacteria, LPS (as a cell wall component) is known to be the principal PAMP and to be recognized by the TLR4/MD-2 receptor complex of mammalian cells.1–3 The lipid component of LPS, lipid A, plays a critical role in interactions with TLR4/MD-2 complexes,4–5 which discriminate structural variations in lipid A as either endotoxic or anti-endotoxic. Moreover, these discrimination patterns have been reported to differ depending on the mammalian species involved.6–7 The structure of lipid A is basically a β–1→6)-linked glucosamine disaccharide backbone phosphorylated at the 1 and 4’ positions and acylated at the 2, 3, 2’ and 3’ positions with R-3-hydroxymyristate. A hexa-acylated type, which is further acylated with a laurate and a myristate at the OH groups at positions 2’ and 3’, respectively, of the R-3-hydroxymyristate, was found to be the major component of Escherichia coli LPS and to elicit robust immunological activity.8–10 Although this particular type is relatively conserved among a wide variety of Gram-negative bacteria, major lipid A types sometimes differ among bacterial species, and some variants frequently coexist within a single species. 11
Our previous report provides details of the LPS of Yersinia pestis, which contains a heterogeneous mix of lipid A moieties, ranging from hexa-acylated to tri-acylated forms when the bacterium grows at 27°C (the temperature of the vector flea), and shifting to contain only the less acylated homologs (tetra- and tri-acylated) when grown at the mammalian body temperature of 37°C. 12 Such less-acylated types have been shown to have weaker stimulatory potency for mouse cells and to be inactive, but antagonistic, for human cells.1,6,7 The alteration of Y. pestis lipid A to less acylated types following transmission from flea to human is therefore thought to facilitate its evasion of human innate immune responses. The increased virulence associated with this alteration was shown to be greater than that provided by other virulence factors described for this bacterium. 13
A similar role for less-acylated lipid A forms has been indicated in some other bacterial species, such as Francisella tularensis, a causative agent of zoonotic tularemia,14,15 Helicobacter pylori in gastric ulcers 16 and Porphyromonas gingivalis in periodontal disease. 17 Such a virulence role for less-acylated lipid A may be fairly widespread among the broad spectrum of pathogenic Gram-negative bacteria, but, to the best of our knowledge, this aspect has been analyzed in only a few species. In addition, most previous studies have investigated the activity of isolated LPS having less acylated lipid A; however, the contribution of this activity to the interaction of live bacteria with host mammalian cells, especially with human cells, during infection has not been well studied.
Salmonella is a representative pathogenic Gram-negative bacterium; the importance of LPS in modulating the infected host response is emphasized by the fact that LPS-hyporesponsive mice (C3H/HeJ), which have a dysfunctional mutant TLR4, are more susceptible to infections with this bacterium.18,19 For the present study, mutant strains of Salmonella enterica serovar Typhimurium (S. Typhimurium) having fewer lipid A acyl groups were established; LPS, formalin-killed bacteria (FKB) and live bacteria preparations of these were used to stimulate human U937 macrophages for evaluation of the role of less-acylated lipid A during infection.
Materials and methods
Bacterial culture
Bacteria were grown at 37°C overnight (16–18 h) with shaking in LB broth (Nacalai Tesque, Inc., Kyoto, Japan), containing 10 g tryptone, 5 g yeast extract and 5 g NaCl per liter of distilled water. Optical densities of suspensions at 600 nm were used to estimate bacterial number. Viable bacterial counts were obtained from growth on LB agar (Nacalai Tesque); the results are expressed as CFU per ml.
Generation of mutant strains
Salmonella Typhimurium strain 14028s [American Type Culture Collection (ATCC), Manassas, VA, USA] 20 was used as the wild-type (WT) strain and is named KCS015 in this report. Non-polar deletion mutant strains were generated according to the method established by Datsenko and Wanner. 21 A DNA fragment containing the kanamycin resistance cassette pKD421 was amplified by PCR. Primers KK128 (CGTTTTGCCGGACGGCTGGGGAAGAGTTCT CGTCGCCGGGCGCTAATTAAGTGT AGGCTGGAGCTGCTTC) and KK129 (ATTATACACCGGGAAAAGCGGTATCACGCGT GCGCGGCACACTTTCATCACATAT GAATATCCTCCTTAG) were designed to delete 500 bp of the 969 bp lpxM-coding DNA region. Primers KK105 (TAAAGGGGGGTTTAACACGTTTACCGATAACGTC GCAGAAACGTGGCGACGTGTA GGCTGGAGCTGCTTC) and KK106 (AACGCATCCAGGCGAAATAGACGTTTCCGTTGTT ATACGAACCCGGAATGCATATG AATATCCTCCTTAG) were designed to delete 370 bp of the 570 bp pagP-coding DNA region. The PCR products were introduced into the WT strain carrying the plasmid pKD46,21,22 which encodes Red recombinase. Generation of the ΔlpxM::kan and ΔpagP::kan alleles in the resulting strains was confirmed by PCR. Kanamycin resistance cassettes were eliminated from the ΔlpxM::kan and ΔpagP::kan strains using the plasmid pCP20; 21 elimination in the resulting strains ΔlpxM (KCS311) and ΔpagP (KCS237) were confirmed by PCR. The double deletion mutant strain ΔlpxM ΔpagP (KCS324) was generated by introduction of the ΔpagP::kan allele into KCS311 by phage P22-mediated transduction 23 followed by elimination of the kanamycin resistance cassette. The S. Typhimurium pagL region was amplified by PCR using primers KK137 (CGCGGATCCTTCAG CCAGCAACTCGCTAA) and KK141 (CCGGAATTCGATTATT GTTTTCAGCGCGG), which are designed to amplify pagL, including 708 bp in the upstream region; the PCR products were cloned into the EcoRI/BamHI site of pBluescript II KS(+). The resulting plasmid construct pKK109 was introduced into KCS324 by electroporation to generate the mutant strain pagL ΔlpxM ΔpagP (KCS369).
Mass spectrometry analysis of lipid A
Bacterial lipid A was purified as previously described. 24 Briefly, pellets of bacterial cells from 10 ml of overnight cultures were re-suspended in 500 µl of Tri-reagent (Molecular Research Center Inc., Cincinnati, OH, USA). After incubation for 30 min at room temperature (20–25°C), 100 µl chloroform was added. After 15 min, the mixture was centrifuged, and the aqueous phase was recovered. LPS was extracted three times by addition of 500 µl water to the organic phase; the aqueous phase containing LPS was dried up with a vacuum concentrator; and 500 µl of 10 mM sodium acetate buffer (pH 4.5) containing 1% SDS was added. The LPS was then hydrolyzed to remove sugar chains from lipid A by incubation at 100°C for 1 h followed by drying. The dried lipid A was washed once with 0.02 N HCl in 95% ethanol and three times with 95% ethanol. The washed lipid A was dried up with a vacuum concentrator and then dissolved in 20 mg/ml 5-chloro-2-mercaptobenzothiazole matrices in chloroform/methanol (1:1, v/v). The mixtures were allowed to dry at room temperature on the sample plate prior to analysis. Spectra were obtained in the negative reflection mode using a matrix-assisted laser desorption/ionization time of flight Voyager-DE STR mass spectrometer (Applied Biosystems Japan, Tokyo, Japan). Each spectrum was the average of 200 shots.
Extraction of LPS and preparation of FKB
Bacterial cells obtained by overnight culture in LB broth were suspended in saline and killed by incubation at 100°C for 30 min. LPS was extracted from the heat-killed bacteria in the aqueous phase by the conventional hot phenol-water extraction method.25,26 To prepare FKB, bacterial cells from overnight cultures were washed once with PBS and suspended in 0.3% formalin at 4 × 109 CFU/ml. Suspensions were incubated at 37°C with shaking for 3 d to kill the bacteria completely and then washed with PBS to remove formalin. Finally, the FKB were re-suspended in PBS and kept at 4°C as stock solutions. Density of the killed bacteria was estimated from that of live bacteria used for formalin treatment.
Cell cultures
Complete medium (CM) used for cell culture was RPMI-1640 medium (Wako Pure Chemical Industry, Ltd, Osaka, Japan) with 2 mM
Cytokine assay
RAW264.7 and U937 cells were cultured in 48-well culture plates (400 µl/well) (Becton Dickinson, Franklin Lakes, NJ, USA) and stimulated with LPS, FKB or live bacteria. A synthetic TLR2 ligand, Pam3-Cys-Ser-(lys)4 hydrochloride (Pam3-Cys) (Calbiochem, EMD Bioscience, Inc., San Diego, CA, USA) was also used as a stimulant. Culture supernatants obtained 6 h after stimulation were assayed for mouse (m) and human (h) cytokines using a specific sandwich ELISA. ELISAs were performed according to instructions supplied by Pierce Biochemistry (Rockford, IL, USA) using matched Ab pairs. Each cytokine was quantified (ng/ml or pg/ml) based on the standard curve obtained for each assay. For suppression experiments, anti-human TLR4/CD284 (anti-TLR4) Ab (MBL Co., Ltd, Nagoya, Japan), 27 anti-human CD282 (anti-TLR2) Ab (BD Pharmingen, Franklin Lakes, NJ, USA) 28 and a human TLR4 antagonist, compound 406 (Daiichi Kagaku Co., Tokyo, Japan),6,7 were used as suppressors. Cells were stimulated in the presence of suppressors (added 1 h prior to stimulation) and culture supernatants were assayed for cytokines 6 h after stimulation.
Enumeration of phagocytosed bacteria
U937 cells pre-cultured in the presence of PMA for 3 d in a 24-well culture plate (5 × 105 cells/ml/well) were washed with antibiotic-free medium (CM containing neither penicillin nor streptomycin). Adherent cells were infected with the WT and mutant strains of Salmonella at a multiplicity of infection (MOI) of 10 at 37°C for 1 h in the antibiotic-free medium. To enumerate the internalized bacteria, cells were first washed and then cultured for 30 min in the presence of 100 µg/ml gentamicin (Wako Pure Chemical Industry). A preliminary experiment showed that extracellular bacteria were completely killed by this antibiotic treatment. Cells were washed and lysed in PBS containing 0.1% Triton X-100 (Wako Pure Chemical Industry). The cell lysate was diluted with PBS and inoculated onto LB agar plates. CFUs were counted after 24 h incubation.
Statistical analysis
Differences between experimental groups were analyzed by the Student’s t test. Significance was assigned to differences having P < 0.05.
Results
Generation and characterization of mutant strains
The assumed chemical structure of WT KCS015 lipid A is shown in Figure 1A; it has five myristic acids (14:0), one lauric acid (12:0) and a partially substituted palmitic acid (16:0). Acyl groups targeted for modification to generate mutant strains are also shown. Genes for the acyltransferases (pagP and lpxM) participating in the biosynthesis of lipid A were deleted from, or the gene for a deacylase (pagL) was introduced into, the WT strain, as described in the Materials and methods. Lipid A isolated from bacterial cells of each strain was analyzed by MALDI-TOF mass spectrometry (Figure 1B). The negative-ion spectrum of lipid A prepared from the WT strain included a major peak at 1798 u (unmodified hexa-acylated lipid A) and a smaller peak at 2036 u (palmitoylated form). This means that some of the hexa-acylated lipid A was palmitoylated by a PagP enzyme; the number of acyl groups is thus shown as 7 ∼ 6 in Figure 1A. This result is in accordance with previous reports.29–31 The spectrum of lipid A from KCS237 included a major peak at 1798 u and loss of the peak at 2036 u, indicating the existence of a single lipid A species having six acyl groups in this mutant strain. A major peak at 1588 u in the spectra of lipid A from KCS311 and KCS324 indicated presence of a penta-acylated type as a result of the lack of a myristic acid from loss of the LpxM enzyme. An additional smaller peak at 1826 u—the lipid A spectrum of KCS311, but not KCS324—indicated the presence of a palmitoylated species because of the presence of PagP. Therefore, the numbers of acyl groups in KCS311 and KCS324 are described as 6 ∼ 5 and 5 respectively. The spectrum of KCS369 lipid A included a major peak at 1362 u (tetra-acylated lipid A) and a smaller peak at 1588 u, indicating deacylation (although not complete) of 3-O-myristic acid by the introduction of PagL. The acyl group number for this mutant is therefore shown as 5 ∼ 4. Considered together, these results confirmed generation of the mutant strains planned for this study.
Analysis of acyl components of lipid A in S. Typhimurium strains. (A) Chemical structure of wild type (WT) lipid A and acyl groups targeted for deletion. Acyltransferases (PagP and LpxM) for biosynthetic introduction (↑) of acyl groups were deleted or a deacylase (PagL) for elimination (↓) of acyl group was introduced. Estimated number of acyl groups based on results presented in (B) also indicated. (B) Purified lipid A from WT and mutant strains analyzed by MALDI-TOF mass spectrometry; mass values are shown.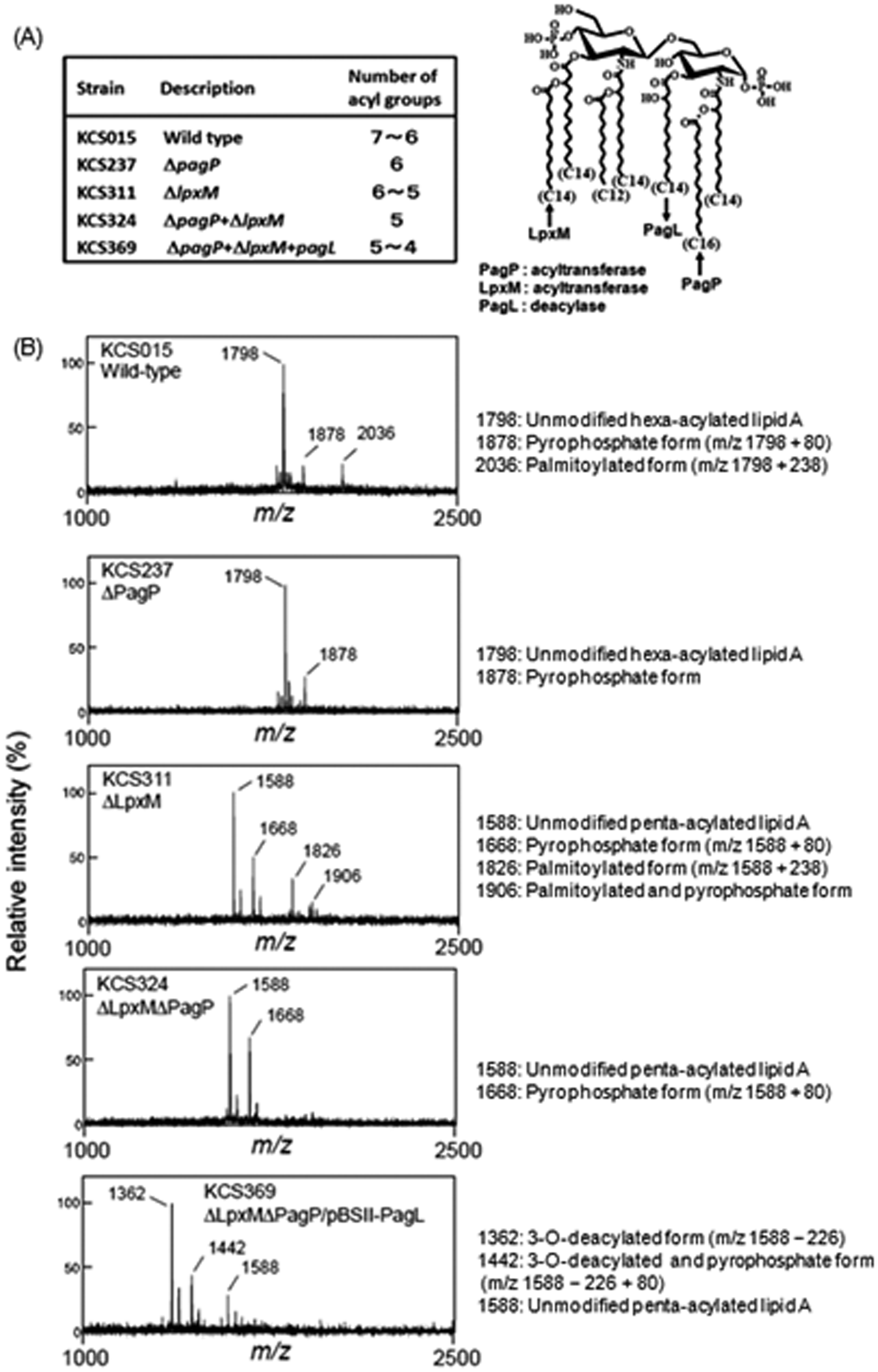
Stimulatory activity of WT and mutant LPS in mouse and human cells
RAW264.7 and U937 cells were used as mouse and human macrophage-like cells, respectively, and LPS obtained from each mutant bacterial strain was assessed for ability to stimulate these cells to produce pro-inflammatory cytokines such as TNF-α and IL-6. LPS from KCS015 (WT) exhibited the greatest stimulatory activity for production of both cytokines in mouse RAW264.7 cells (Figure 2); activity decreased gradually with decreasing LPS acyl group numbers, from KCS237 to KCS311, KCS324 and KCS369. Mutant KCS369 LPS activity was the weakest, although significantly positive. LPS from KCS015 and KCS237 showed similar strong stimulatory activity for human U937 cells (Figure 2), both inducing substantial amounts of TNF- α and IL-6 at doses as low as 1 ng/ml, with maximum levels of both cytokines occurring at 10 ng/ml and 100 ng/ml doses. However, none of the three LPS from the other mutant strains (KCS311, KCS324 and KCS369) exhibited any detectable activity up to the highest dose examined (100 ng/ml). These results indicate that activity in mouse cells gradually decreased with a decrease in acyl number, while activity in human cells disappeared suddenly when one acyl group was eliminated from the WT LPS, from a hexa- to a penta-acylated lipid A type.
Stimulation of mouse macrophage RAW264.7 cells or human macrophage U937 cells with LPS isolated from wild type and mutant strains. Cells stimulated with LPS at indicated doses. Cytokine levels in culture supernatants 6 h after stimulation determined by ELISA for mouse (m) or human (h) cytokines. Mean ± standard errors for triplicate samples; representative result of three independent experiments.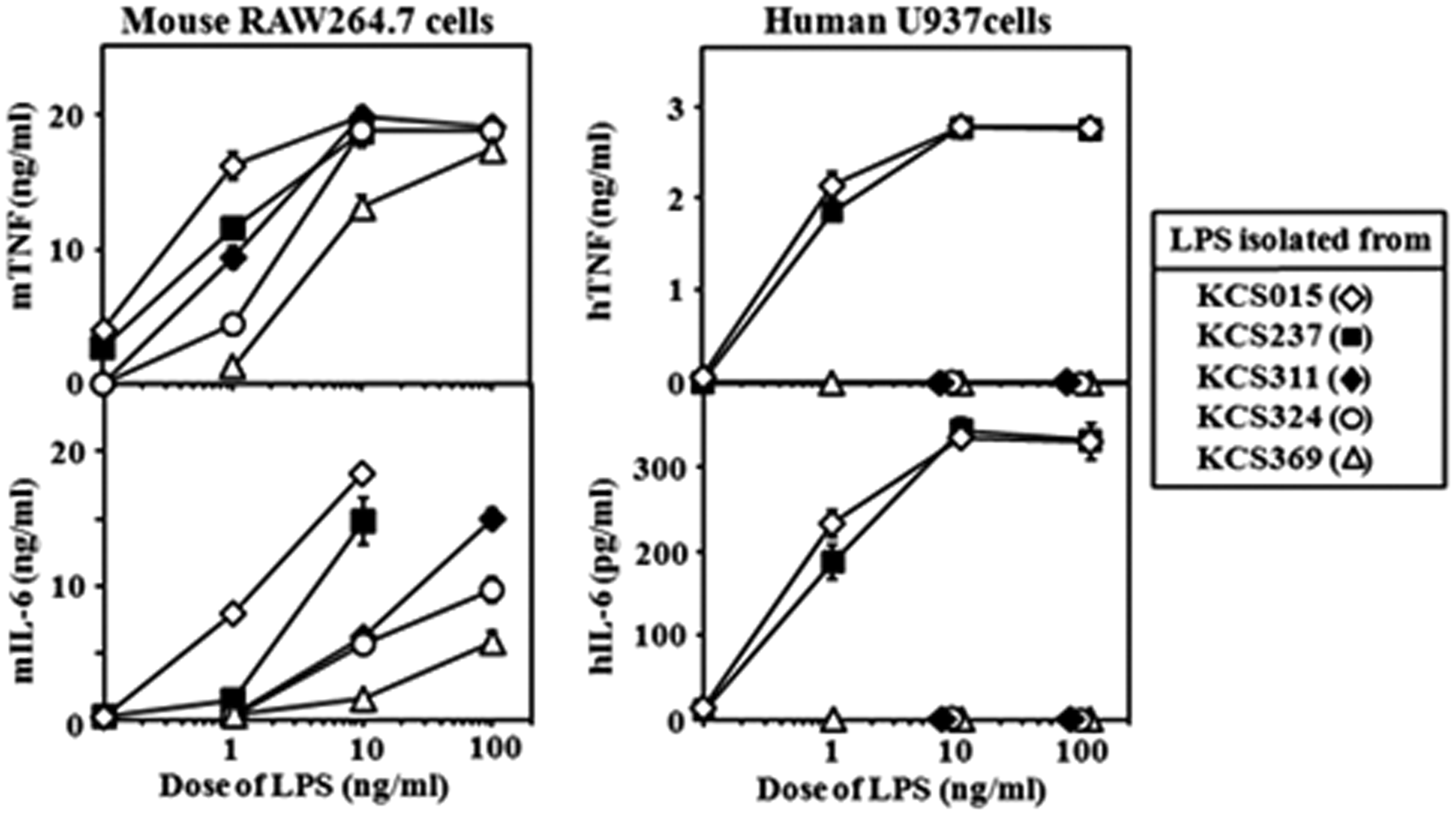
Stimulation of human U937 cells by mutant FKB and live bacteria strains
As shown in Figure 3, KCS015 and KCS237 FKB preparations elicited similar brisk induction of cytokine production in human cells. However, FKB preparations of the other three strains (KCS311, KCS324 and KCS369) had only weak activities at similar concentrations. There was a clear difference in stimulatory activity between the two groups. Similarly, as shown in Figure 3, live KCS015 and KCS237 bacteria had high activity, while the live KCS311, KCS324 and KCS369 strains had markedly reduced, weak activity in comparison. Effective doses of live bacteria for stimulation of U937 cells seemed much lower than those of FKB. In contrast to killed bacteria, live bacteria multiplied after addition to the cell cultures. Within 3 h of stimulation, doses of live bacteria increased more than 10-fold of the initial (indicated) doses and still increased vigorously thereafter. This may be the reason why about 100 × lower doses of live bacteria than those of killed bacteria were comparable for stimulation of U937 cells (e.g. 1 × 105 CFU/ml of live bacteria vs 1 × 107 killed bacteria/ml of FKB).
Effect of wild type and mutant strain FKB and live bacteria preparations on stimulation of U937 cells for cytokine production. U937 cells stimulated with indicated doses of FKB or live bacteria and cytokine levels measured as described in Figure 2. Mean ± standard errors for triplicate samples; representative result of three independent experiments.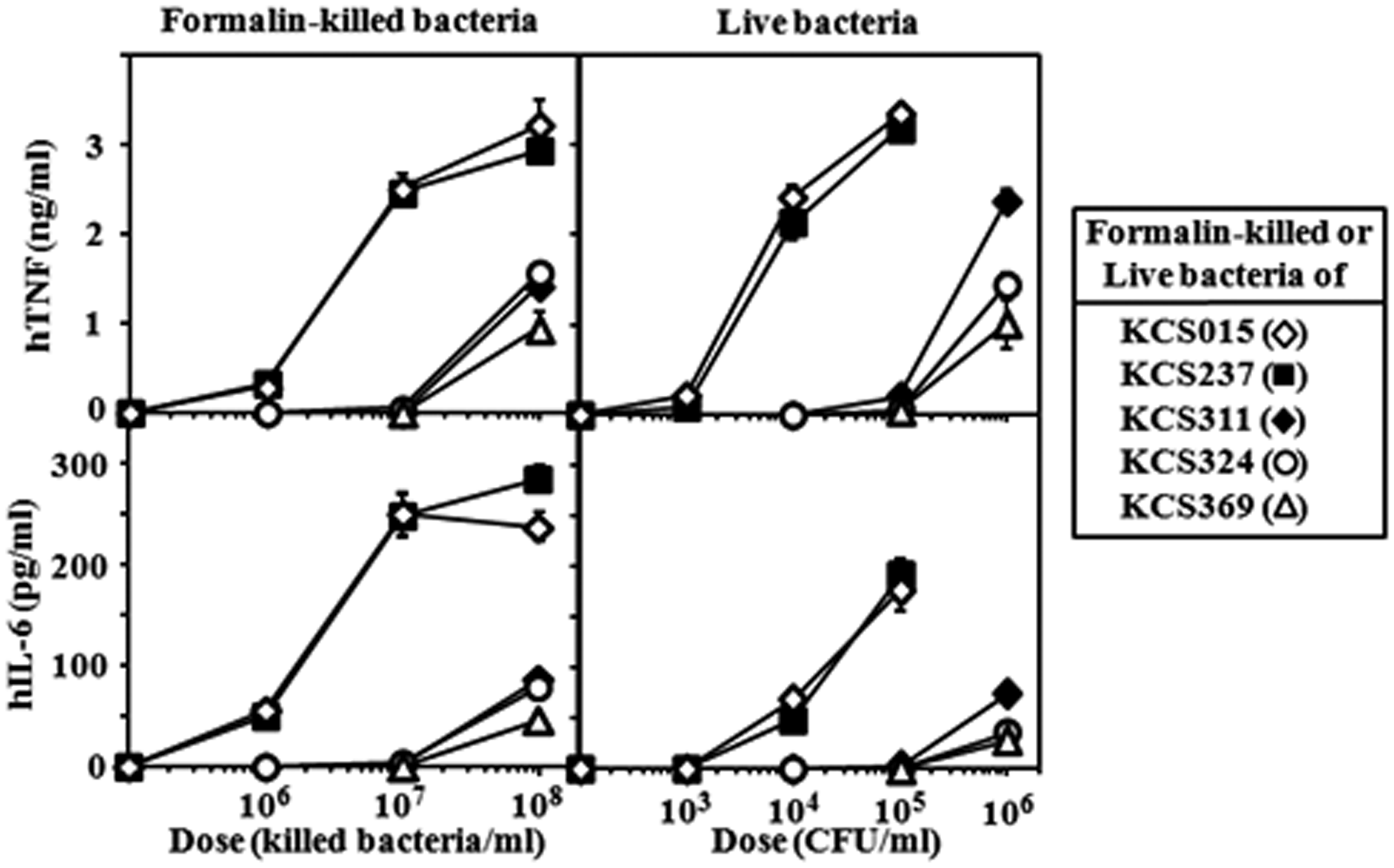
Antagonistic effect of inactive LPS on activation of human U937 cells by WT LPS contrasts with the effect of the corresponding FKB and live bacteria
Less acylated LPS that was inactive in human cell assays was assessed for antagonistic effects on the stimulatory potency of WT LPS. All three LPS exhibited strong antagonistic activity (Figure 4A, B) comparable to the activity of the synthetic human TLR4 antagonist, compound 406 (data not shown). Cytokine levels induced by WT LPS at a dose of 1 ng/ml were suppressed by the LPS starting at the 1 ng/ml level and reaching complete suppression at 100 ng/ml. However, neither FKB (Figure 4C, D) nor live bacteria (Figure 4E, F) preparations of the corresponding strains were antagonistic to WT LPS stimulation. These results indicate that antagonistic activity of LPS disappears in reactions with killed and live bacteria in contrast to the agonistic activity of LPS, which appears clearly in the bacterial reactions (Figure 3).
Antagonistic effect of mutant strain [having less acylated lipid A (KCS311, KCS324 and KCS369)] LPS, FKB and live bacteria preparations on stimulation of U937 cytokine production by wild type (WT) (KCS015) LPS. U937 cells stimulated with WT LPS (1 ng/ml) in the presence of indicated doses of LPS (A, B), FKB (C, D) or live bacteria (E, F) preparations of mutant strains. Culture supernatants obtained 6 h after stimulation assessed for cytokine level by ELISA. Gray bars: cytokine levels produced by stimulation with wild type LPS only. Mean ± standard errors for triplicate samples; representative result of three independent experiments.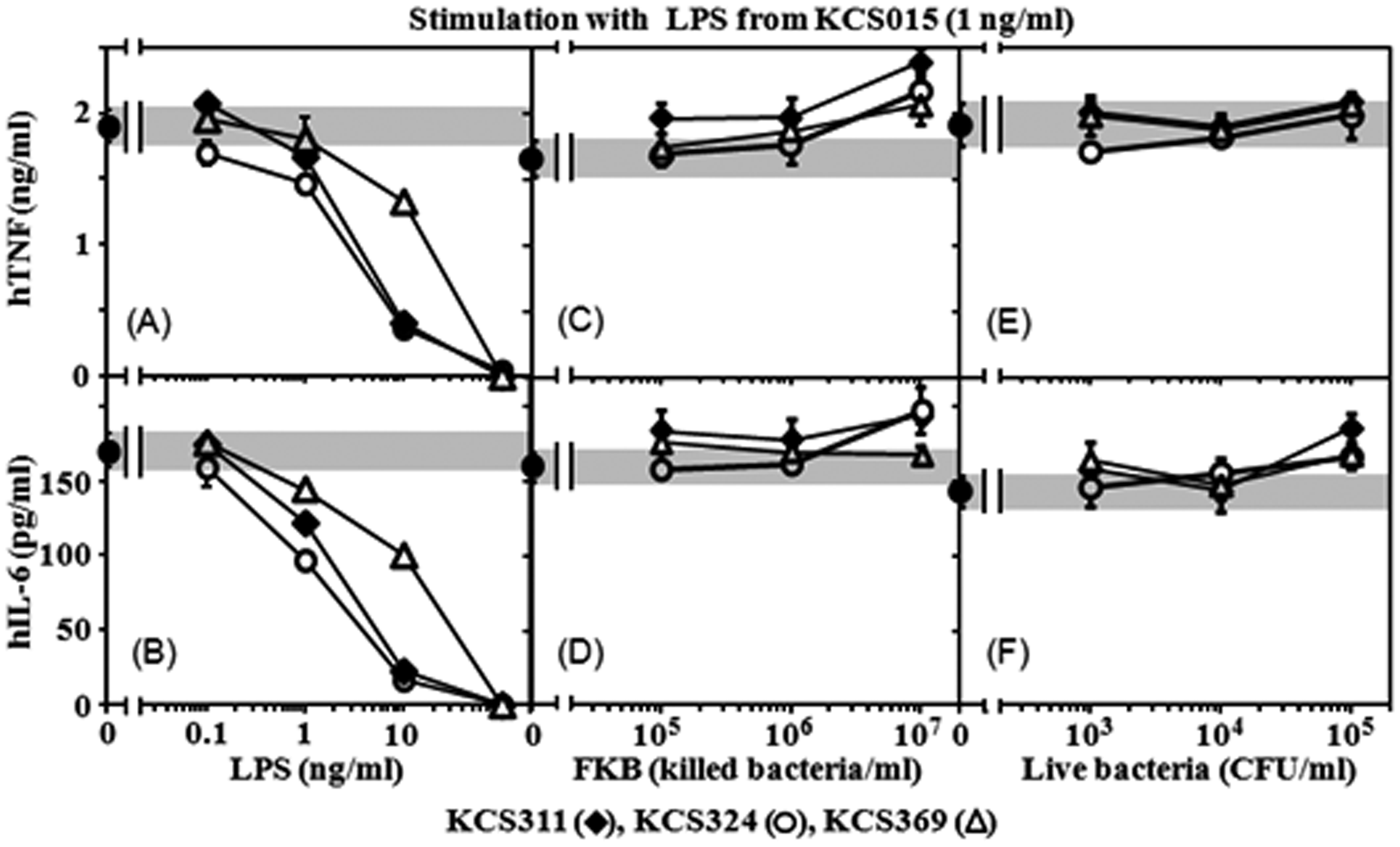
Effects of suppressors of TLR4 and TLR2 on cytokine production in U937 cells stimulated by FKB or live bacteria
Stimulation of U937 cells by KCS015 and KCS237 FKB and live bacteria was strongly suppressed by anti-TLR4 Ab and compound 406, but not at all by anti-TLR2 Ab; the same was true for activity of the respective LPS (Figure 5A, B). The same anti-TLR2 Ab suppressed cytokine production when U937 cells were stimulated with a synthetic TLR2 ligand, Pam3-Cys (Figure 5C). These results indicate that, among various bacterial components, it is predominantly the LPS of Salmonella that is recognized by human cells during bacterial infection for activation of innate immune responses.
Effects of a synthetic human TLR4 antagonist (compound 406), an anti-TLR4 Ab or an anti-TLR2 Ab on capacity of KCS015 or KCS237 LPS, FKB and live bacteria preparations to stimulate U937 cells. Each Ab added to cell culture at 10 µg/ml and compound 406 at 1 ng/ml. Stimulants added 1 h later at 1 ng/ml LPS, 5 × 106 FKB/ml or 1 × 105 CFU/ml. Cytokine levels of TNF (A) and IL-6 (B) in culture supernatants 6 h after addition of stimulants determined by ELISA. A synthetic TLR2 ligand, Pam3-Cys (10 nM), was also used as a stimulant, and cytokine levels determined (C) to check the capacity of the anti-TLR2 Ab. Mean ± standard errors for triplicate samples; representative result of three independent experiments. **P < 0.01, *P < 0.05, compared with control (none).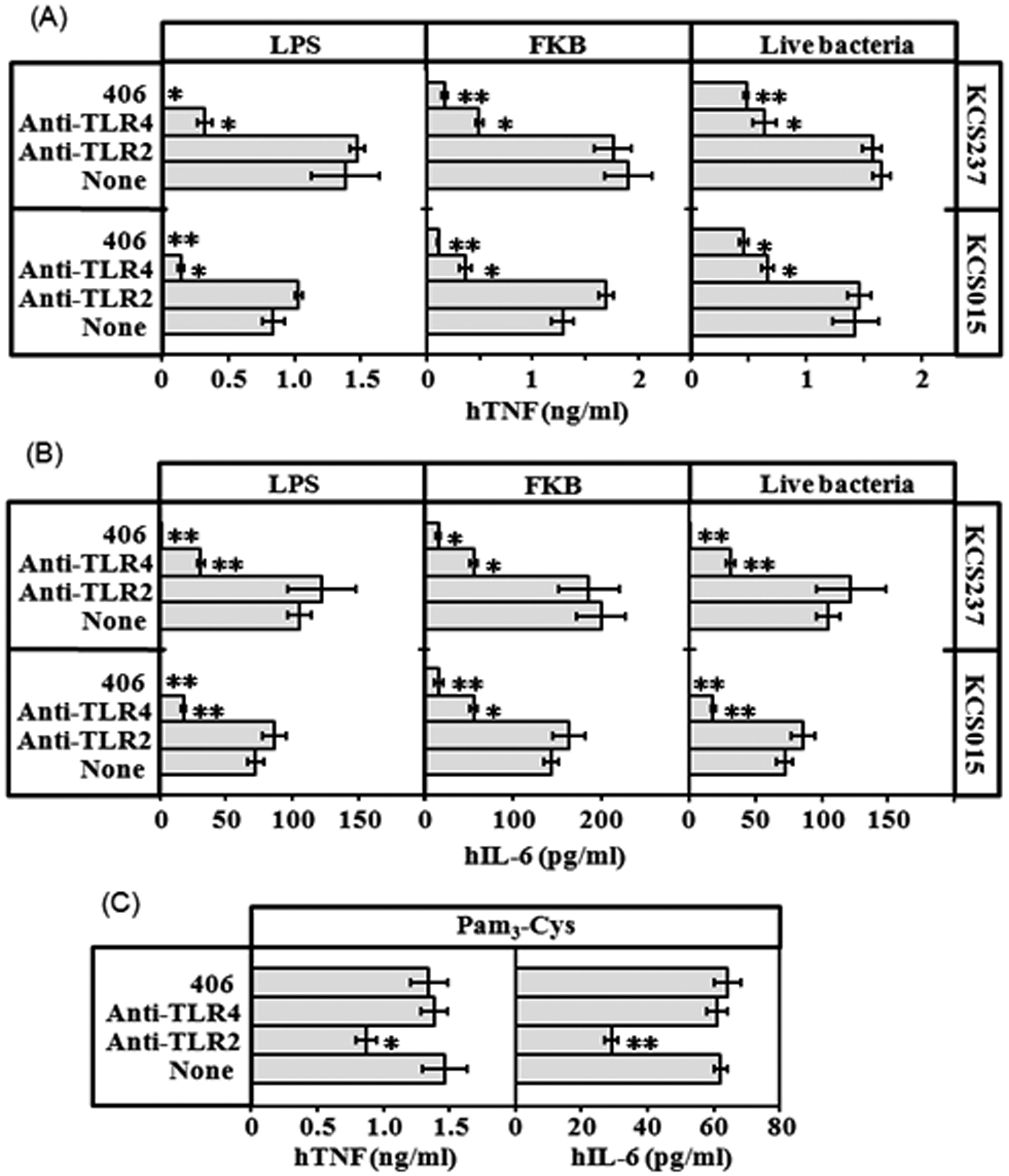
Time course for cytokine production by U937 cells stimulated with FKB and LPS
As shown in Figure 6, no differences in the time courses for cytokine induction by FKB and LPS preparations of the KCS015 and KCS237 strains were observed. This result indicates that FKB can induce cytokine production as quickly as free LPS and suggests that the bacterial-bound form of agonistic LPS can interact directly with U937 cells for cytokine induction without requiring degradation of bacteria to release free LPS.
Time course of cytokine production by U937 cells after stimulation with KCS015 or KCS237 LPS and FKB. LPS (10 ng/ml) or 1 × 107 FKB/ml added to cell cultures. Supernatants obtained at indicated times for cytokine determination. Mean ± standard errors for triplicate samples; representative result of three independent experiments.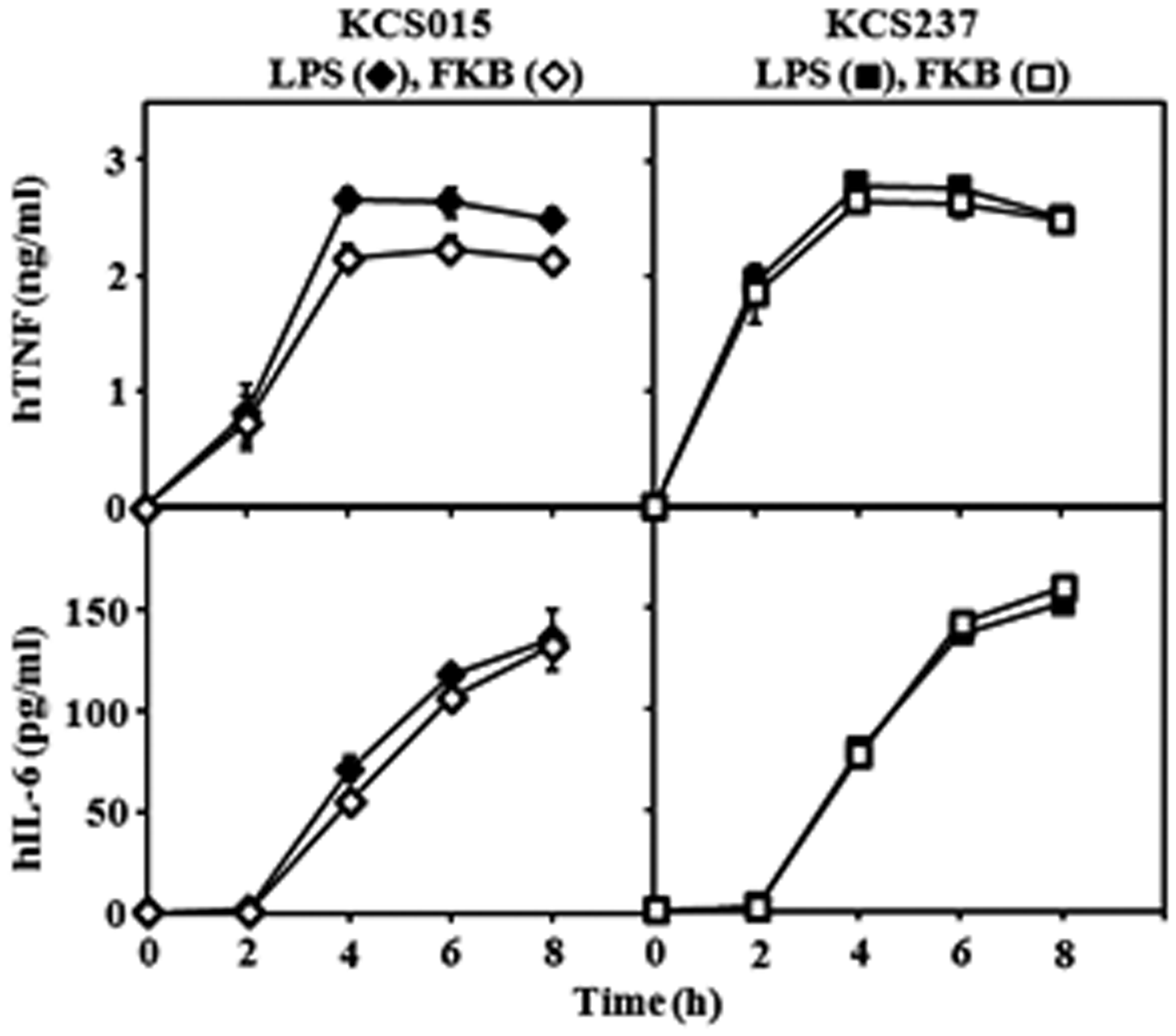
Phagocytosis of WT and mutant strains by U937 cells
To analyze the effect of decreased acylation of lipid A on phagocytosis of bacteria by human macrophage U937 cells we infected the cells with each strain and determined the number of phagocytosed bacteria using the gentamicin protection assay. As shown in Figure 7, WT bacteria were phagocytosed to about 10 × 104 CFU, which was comparable to results for KCS237—the strain possessing agonistic type LPS. However, strains KCS311, KCS324 and KCS369, all of which have antagonistic-type LPS, were phagocytosed at significantly lower numbers compared with the WT strain. These results suggest that those strains possessing less acylated lipid A became more resistant than the WT strain to phagocytosis by human macrophages. It means that, not only by cytokine-induction analysis but also by phagocytosis analysis, those mutants are judged to evade human defense mechanisms more effectively than the WT strain.
Phagocytosis of wild type (WT) and mutant strains of Salmonella by U937 cells. U937 cells were infected with bacteria at a MOI of 10 for 30 min and then treated with 100 µg/ml gentamicin for 1 h. Number of phagocytosed bacteria determined. Mean ± standard errors for triplicate samples; representative result of three independent experiments. **P < 0.01 compared to WT strain (KCS015).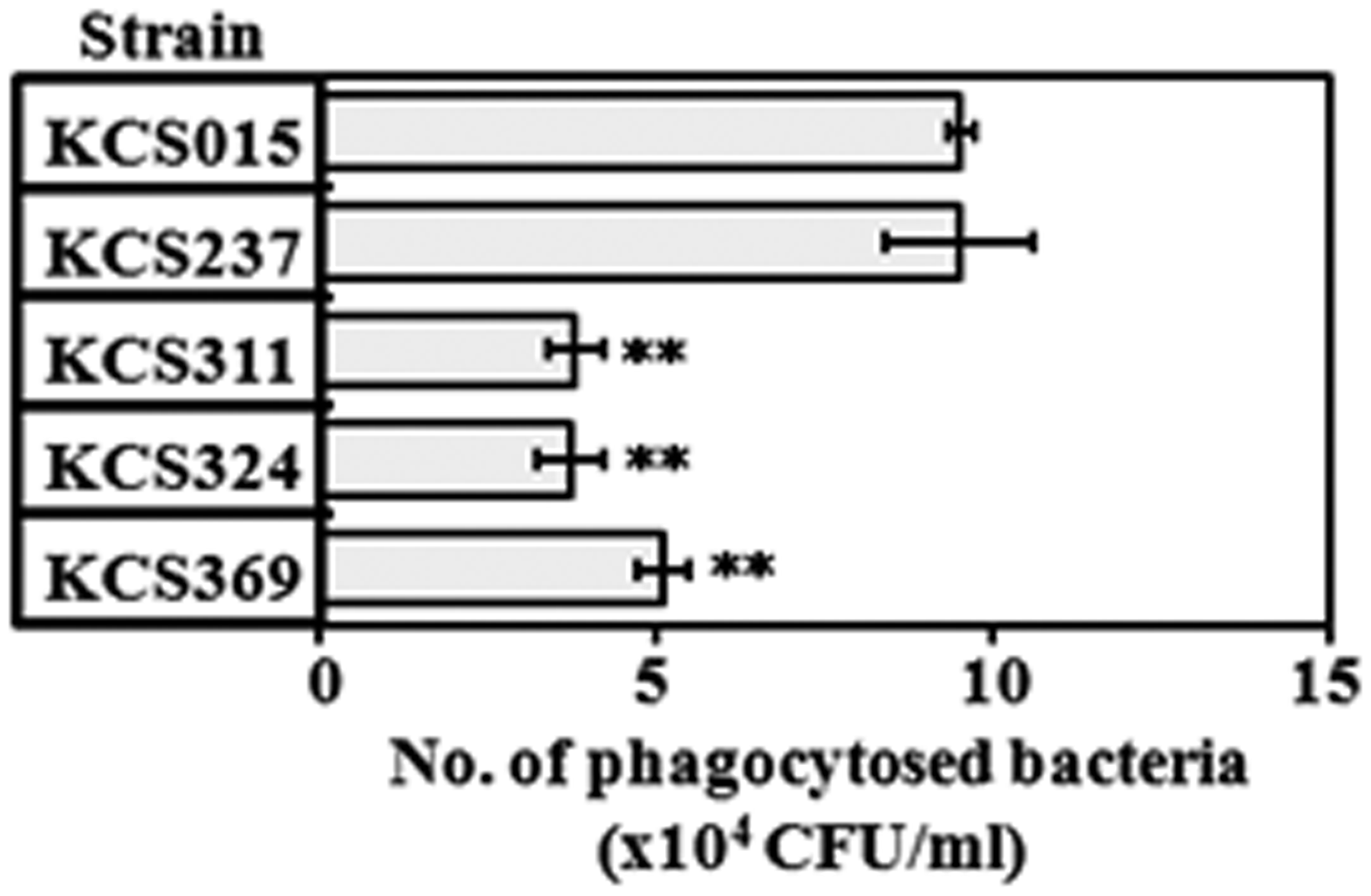
Discussion
It is very interesting that reactions of whole bacterial cells (live bacteria and FKB) were agonistic, even though weak (Figure 3), and not antagonistic (Figure 4C–F), while their LPS displayed no agonistic, but strong antagonistic, activity. Similar results were obtained in a previous study of Y. pestis LPS and FKB, which has less acylated lipid A. 32 Weak agonistic activities presented by whole bacterial reactions probably come from PAMPs other than LPS, as those activities were not suppressed by anti-TLR4 Ab (data not shown). These results indicate that mutants with less acylated lipid A successfully infect humans after markedly decreasing TLR4 agonistic activity, but not through increased antagonistic activity. This mechanism was sufficiently effective for the mutants studied to evade human innate immune responses and to establish infection. To the best of our knowledge, the results of the present study indicate, for the first time, that antagonistic activity of LPS scarcely contributes to successful live bacterial evasion of human innate immunity.
Agonistic LPS activity (WT and KCS237) was displayed, even in whole bacterial reactions with human cells, as opposed to the antagonistic effects of isolated LPS. However, participation of free LPS after release from bacterial cells in whole cell reactions is hard to determine, as the time required for cytokine induction by FKB was not longer than that for LPS (Figure 6). Bacterial-bound LPS is probably responsible for the activity observed. In general, an agonist interacts reversibly with its receptor, but an antagonist reacts irreversibly. LPS on the bacterial surface may exist in a state that permits reversible, but not irreversible, interaction with TLR4/MD-2.
The final steps for biosynthesis of lipid A are catalyzed by the acyltransferases LpxL and LpxM, which add 12:0 and 14:0 consecutively to the tetra-acylated intermediate. 31 In Salmonella species, deletion of the lpxM gene has been reported to allow bacterial growth at 37°C, unlike the lpxL gene deletion, which creates temperature-sensitive mutants. 33 The lpxM mutants, like KCS311 and KCS324, are useful for examining the role of penta-acylated lipid A when viable bacteria are presented to host defense cells at the normal body temperature of 37°C. It had been reported that the lpxM mutation of Salmonella did not appear to affect the stimulatory capacity of live bacteria for mouse cells, 34 in contrast to the results of infection of human cells presented in this study. LPS of the lpxM mutants exhibited substantial agonistic activity to mouse RAW264.7 cells (Figure 2), which was only somewhat weaker than that of the WT strain. Such a small difference in LPS activity may be submerged in the final outcome of live bacterial reactions. The same mutant LPS, however, had no agonistic activity in human cells, while WT LPS had strong activity. Such large differences in LPS activity may contribute to the outcomes of final live bacterial reactions. These results indicate that modification of the lipid A structure from the hexa- to the penta-acylated type is more effective for bacterial evasion of human, rather than mouse, innate immunity and that use of human cell experimental systems is a prerequisite for understanding the precise role played by LPS in human infections.
LPS containing tetra-acylated lipid A as a major component has been isolated from several species of bacteria and has been suggested to contribute to bacterial evasion of host innate immunity.12–17 Preparation of a mutant KCS369 containing only a tetra-acylated type of lipid A had been planned for this study, but the resulting strain preserved some of the penta-acylated form (Figure 1). A mutant strain of E. coli containing only a tetra-acylated type of lipid A has been prepared and was reported to lyse easily on centrifugation because of a weak cell wall structure; 35 it did not grow on nutrient broth above 32°C. Similarly, it may be hard for Salmonella to construct a cell wall strong enough to support stable growth with only tetra-acylated lipid A. Mutant strains used in this study grew well in LB broth at 37°C and, like the WT strain, hardly lysed on centrifugation. Therefore, bacterial cell surface remodeling upon alteration of the lipid A structure from a hexa- to a penta-acylated type occurs without substantial loss of bacterial cell wall stability.
Salmonella possess a range of cellular components that act as PAMPs and are recognized by host TLRs, including LPS (TLR4), lipoprotein (TLR2), flagellin (TLR5) and bacterial DNA (TLR9). 36 In the current study, strong suppression by TLR4 suppressors, but not by the TLR2 suppressor, suggests that among the various PAMPs, it is LPS that plays a predominant role in evading human innate immune responses during Salmonella infection. We suggest that modification of the lipid A structure is an effective bacterial strategy for evasion of human innate immune attack.
Recently, it was reported that a surprisingly large fraction of meningococcal clinical isolates have LPS with underacylated (penta-acylated) lipid A as a result of mutation of the lpxL gene, 37 which likely arises spontaneously in hosts. This finding revealed the important pathogenic role of lipid A modification in a practical infectious disease. Investigations standing on such a point of view are required to find out similar lipid A mutants among clinical isolates of the other infectious diseases and those findings will give great help to further understanding of the pathogenic role of lipid A modification.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
