Abstract
Deficiency of mannose-binding lectin (MBL), a serum protein involved in killing and promoting phagocytosis of pathogens, is associated with respiratory infection and disease progression in a number of acute and chronic lung diseases, including cystic fibrosis (CF)- associated bronchiectasis. No such association has been studied in non-CF bronchiectasis (nCF-Br). One hundred and thirty-three adult patients with nCF-Br were studied. Serum MBL levels were measured and deficiency defined using two cut-off levels, i.e. MBL ≤100 ng/ml and ≤600 ng/ml. Parameters of severity included lung function impairment, annual exacerbation and hospital admission rates, breathlessness, and Pseudomonas aeruginosa and Haemophilus influenzae infection rates. The incidence of MBL deficiency using cut-off levels of 100 ng/ml and 600 ng/ml was 10% and 26% respectively, similar to rates seen in the general population. There was no significant difference in mean FEV1% predicted between MBL deficient and sufficient patients at both cut-off levels (≤100 ng/ml: 63.8% vs. 64.6%, P = 0.91; ≤ 600 ng/ml: 66.5% vs. 63.9%, P = 0.56). In addition, exacerbation/hospital admission rates, symptoms of breathlessness and isolation/colonisation rates with P. aeruginosa and H. influenzae were similar in both groups at both cut-off levels. In conclusion, MBL deficiency is not associated with markers of disease severity in patients with nCF-Br.
Introduction
Non-cystic fibrosis bronchiectasis (nCF-Br) is a syndrome of dysregulated immunity with evidence of impaired clearance of bacteria and elevated neutrophilic inflammation that leads to irreversibly damaged and dilated bronchi. These collectively result in disabling symptoms of chronic sputum production and repeated lower respiratory tract infections. Bronchiectasis is the final common pathway of a large range of known underlying disease processes. It can develop secondary to overt cellular and humoral immune defects; however, in over 50% of cases no cause is found. 1 In these so-called ‘idiopathic’ cases, it is plausible that there are undetected subtle defects in the innate immune responses to commonly encountered respiratory pathogens. Indeed, the recently published British Thoracic Guideline for nCF-Br 2 recommends the development of local respiratory/immunology protocols for thorough immunological profile testing in all patients, as the detection of an underlying cause impacts greatly on the prognosis and treatment available. 1
Mannose-binding lectin (MBL) is one of the major plasma pattern recognition molecules capable of killing organisms by activation of the lectin complement pathway or by aiding opsonophagocytosis. 3 Common polymorphisms in the structural and promoter region of its encoding gene MBL2 give rise to variable complement-activating ability and up to 1000-fold variation in serum MBL concentrations in humans. 4 In clinical studies, MBL deficiency is associated with a predisposition to developing infection 5 with high virulence and severity (as in the case of invasive pneumococcal disease),6–8 particularly within the respiratory tract.9–17
In CF-associated bronchiectasis, there is good evidence that MBL deficiency affects disease course and severity. 18 Although there is also evidence implicating MBL deficiency in the development of nCF-Br in the specific context of common variable immunodeficiency,19,20 no previous study has sought to investigate a potential link between low MBL levels and disease severity in nCF-Br. However it has recently been shown that MBL deficiency is equally prevalent in patients with nCF-Br and in healthy volunteers. 21
Patients and methods
Study design
A retrospective cohort analysis of consecutive adult patients attending a specialist nCF-Br clinic at Freeman Hospital, Newcastle-upon-Tyne, UK, from August 2008 to January 2011.
Patients
Causes and associations of bronchiectasis (n = 133).
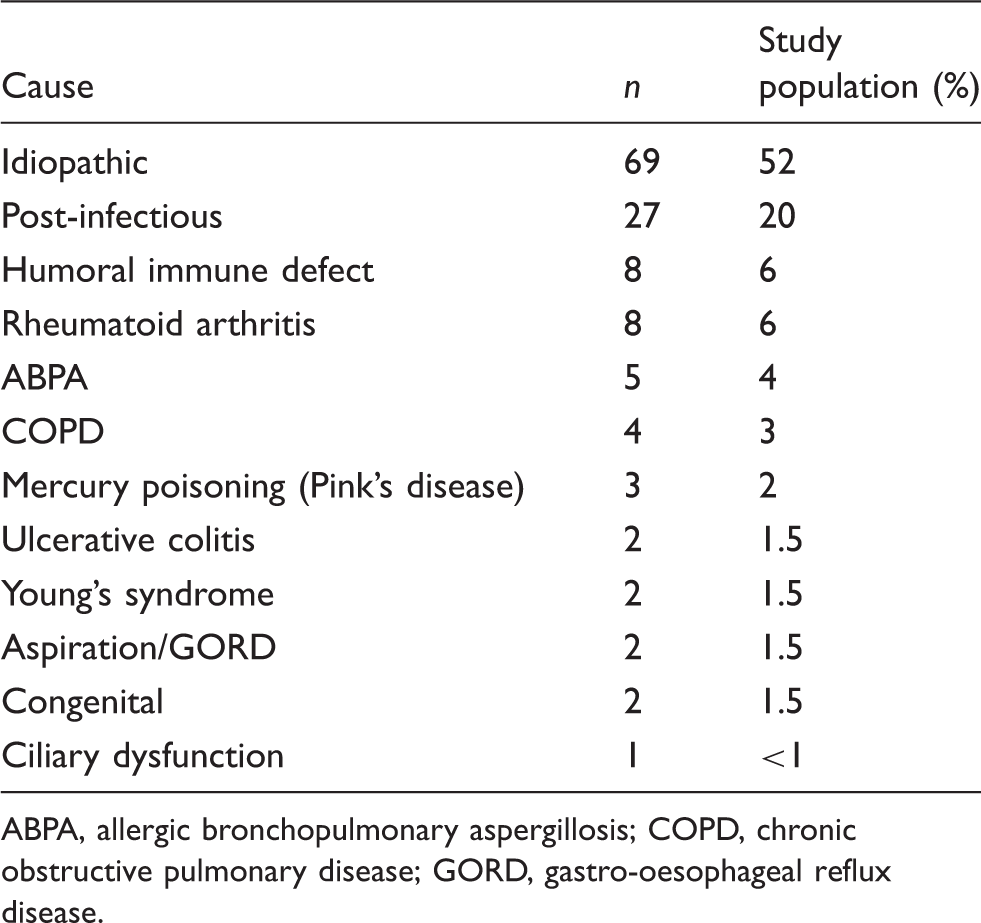
ABPA, allergic bronchopulmonary aspergillosis; COPD, chronic obstructive pulmonary disease; GORD, gastro-oesophageal reflux disease.
Serum MBL assay
According to our specialist clinic protocol, 2 a blood sample to measure a range of detailed immunological defects was taken from all patients in a period of stable disease defined as the absence of an infective exacerbation within the preceding two weeks. The routine immunological profile included a full blood count, total IgE level, Aspergillus fumigatus radioallergosorbent test (RAST), serum immunoglobulins (IgG, IgA and IgM) and serum electrophoresis, baseline specific antibody levels against tetanus toxoid and the capsular polysaccharides of Streptococcus pneumoniae. In addition, our local policy was to check MBL levels, which were measured using an MBL Oligomer ELISA kit (Alphalabs, Eastleigh, UK). There is no consensus on the precise serum MBL level below which an individual is defined as ‘MBL deficient’. We therefore divided our population into ‘MBL sufficient’ and ‘MBL deficient’ in two ways, using two different ‘cut-off ’ values: 23 a serum concentration ≤100 ng/ml (which corresponds to approximately 5–10% of healthy adults) and a value of ≤600 ng/ml (which corresponds to approximately 40% of the healthy population).
Spirometry and dyspnoea
Forced expiratory volume in 1 s (FEV1) was measured to American Thoracic Society/European Respiratory Society standards 24 and expressed as a percentage of predicted values for age, height, mass, sex and ethnicity. 25 We also assessed a multicomponent aspect of bronchiectasis, namely breathlessness, by recording the Medical Research Council (MRC) dyspnoea score 26 (range 1–5) of all patients.
Infective exacerbation and admission frequency
Patients’ self-reported 12-month infective exacerbation frequencies and the number of hospital admissions for bronchiectasis symptoms in the past 12 months are routinely recorded at the clinic and were extracted from the clinical records.
Sputum microbiology
All sputum microbiology results collected since the first sputum sample were retrospectively reviewed and all microorganisms recorded. Up to 12 years’ data were analysed for the growth of Pseudomonas aeruginosa and Haemophilus influenzae. Microbiology samples were processed in a Clinical Pathology Accreditation-accredited laboratory to routine UK National Health Service diagnostic standards. ‘Colonisation’ was defined as the organism being isolated on two or more occasions, more than three months apart within a one-year period. 1 ‘Isolation’ was defined as the growth of the organism on at least one occasion without colonisation.
Statistics
Statistical analysis was performed using Prism software v5 (Graphpad Software; GraphPad, La Jolla, CA, USA) to examine relationships between FEV1% predicted, MRC dyspnoea scores, exacerbation/admission frequency and P. aeruginosa/H. influenzae infection. Spearman’s rank correlation, Student’s t-test, Mann-Whitney U test and Fisher’s Exact test were used to identify differences in the distributions between groups. A probability (P) value of <0.05 was deemed to indicate statistical significance.
Results
MBL serum levels
The mean MBL level in the population was 1885 ng/ml (SD ± 1446 ng/ml) and the median level was 1574.5 ng/ml (interquartile range 584.4–3462.4 ng/ml). Using the higher cut-off MBL serum level of ≤600 ng/ml, we found that 34 patients (26%) were MBL deficient and 99 patients (74%) were MBL sufficient. With the lower cut-off value of ≤100 ng/ml, 13 patients (10% of total) were MBL deficient and 120 patients (90%) had normal levels. No patients in the MBL-deficient groups had other identified major immune function defects such as common variable immunodeficiency.
MBL, dyspnoea and airflow obstruction
Comparison of severity parameters in bronchiectasis patients with mannose-binding lectin (MBL) deficiency and those with normal serum concentrations using two different cut-off levels of ≤100 ng/ml and ≤600 ng/ml to define deficiency.
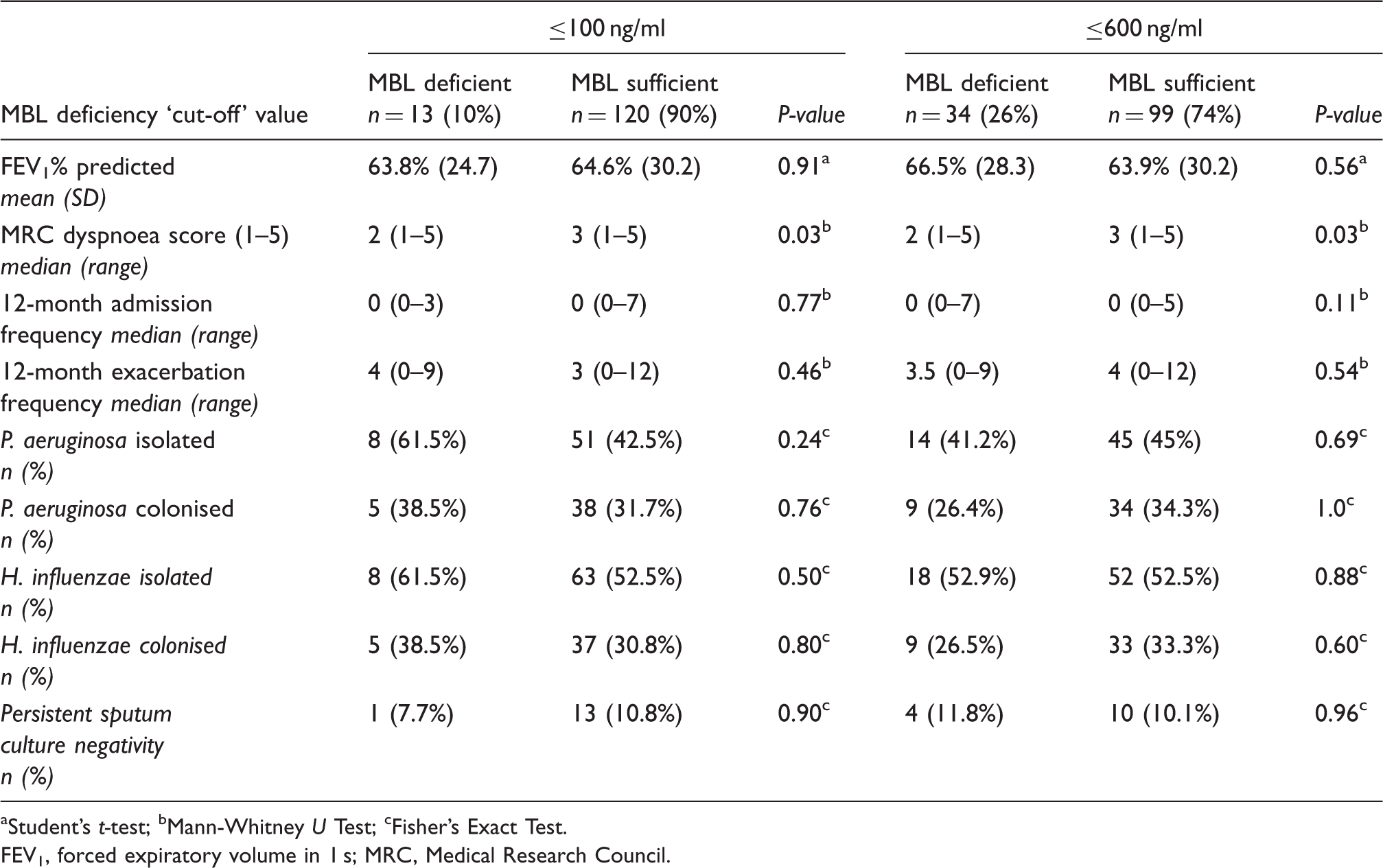
aStudent’s t-test; bMann-Whitney U Test; cFisher’s Exact Test.
FEV1, forced expiratory volume in 1 s; MRC, Medical Research Council.
The median MRC dyspnoea score was 2 (range 1–5) in patients with MBL deficiency and 3 (range 1–5) in patients with normal MBL levels using both cut-off levels. This difference did appear significant (P = 0.03, Mann Whitney); however, there was no correlation between absolute MBL values and MRC dyspnoea scores (P = 0.2, r = −0.02; Spearman’s rank correlation).
MBL and infective exacerbation/admission frequency
Median exacerbation and hospital admission frequency over 12 months amongst all patients was 4 (range 0–12) and 0 (range 0–7), respectively. There was no significant difference in median exacerbation or admission frequency between MBL deficient and MBL sufficient patients using both MBL cut-off levels (P >0.05, Mann Whitney).
MBL and sputum microbiology
Using the lower cut-off value of 100 ng/ml, 8 (61.5%) of the 13 patients with MBL deficiency had isolated P. aeruginosa and 5 of these patients (38.5% of total) had P. aeruginosa colonisation (either past or present). The trend of P. aeruginosa isolation was increased compared with MBL-sufficient patients (see Table 2), but did not reach clinical significance (61.5% vs. 42.5%, P = 0.24; Fisher’s Exact test). Similarly, using the higher cut-off value of 600 ng/ml, there was no statistically significant difference in P. aeruginosa isolation or colonisation between MBL-deficient and MBL-sufficient patients.
In addition, there was no significant difference between H. influenzae isolation/colonisation rates between MBL-deficient and MBL-sufficient patients at both cut-off levels or in the size of the small proportion of patients with persistently negative sputum cultures in the MBL-deficient and MBL-sufficient groups.
Discussion
Bronchiectasis is an increasingly recognised chronic pulmonary infection syndrome. The recent UK national audit of bronchiectasis captured 1601 patients seen in October 2010, suggesting a patient population of at least 18,000 in the UK. 27 Estimates from the USA suggest that more than 110,000 patients suffer from bronchiectasis. 28 In a prior series, almost 10% of patients with non-CF bronchiectasis have been found to have an identifiable deficit of humoral immune function. 1 There is also good evidence of a maladaptive cellular response in the bronchial mucosa, with a marked influx of CD4+ T lymphocytes, neutrophilia and increased expression of IL-8. 29 Whilst L-ficolin insufficiency is over-represented in patients with bronchiectasis, 21 relatively little is known about the influence of other soluble pattern recognition molecules of the innate immune system, such as MBL, on the development of the disease, and, in particular, its clinical severity. This is despite increasing evidence of the importance of MBL deficiency in respiratory infection in other acute and chronic lung diseases, such as acute fungal infections 30 and disease progression in CF respectively.
Our data in a larger cohort extend the previous findings that MBL deficiency at two different threshold levels (≤100 ng/ml and ≤600 ng/ml) is not more prevalent in patients with nCF-Br compared with healthy controls, 21 with an incidence in our population of 10% and 26% respectively.
Our key finding is the novel observation that there was no significant influence of MBL deficiency on a range of clinical features normally associated with more severe cases of bronchiectasis, such as the severity of airflow obstruction, 12-month infective exacerbation or hospital admission frequency. Furthermore, there was no increased rate of being culture positive for any pathogen in the MBL deficient groups. Specifically, we were also unable to demonstrate any increased risk of infection with either of the key pathogens P. aeruginosa and H. influenzae in this population. 1 In contrast, a previous large Canadian series of over 1000 children with CF demonstrated earlier acquisition of P. aeruginosa and a greater rate of decline in lung function in patients with MBL-deficient genotypes. 31 Surprisingly, our data suggested that patients with sufficient MBL levels were subjectively more breathless. This finding remains unexplained.
A recent review and meta-analysis of almost 6000 patients from 9 countries confirmed an association between MBL deficiency and reduced FEV1% predicted and forced vital capacity (FVC) in CF adults, earlier acquisition of P. aeruginosa and increased progression to end-stage CF lung disease requiring transplantation. 18 Although P. aeruginosa isolation and colonisation rates are much higher in CF patients than in nCF-Br patients, the lack of an association between P. aeruginosa infection and MBL deficiency in the current study compared with the correlations seen in CF remains difficult to explain. Plausible explanations may include the variable time between disease onset and diagnosis, differing aetiological precipitants and the more variable disease progression rates seen in adult bronchiectasis compared to CF. In the context of a coexistent common variable immunodeficiency (CVID), characterised by defective immunoglobulin production, low MBL levels are associated with low capacity of complement activation through the lectin pathway and an increased frequency of lower respiratory tract infections and bronchiectasis.19,20 This ‘two-hit hypothesis’ of MBL deficiency in association with another immunodeficiency translating to clinical effect 4 is further supported by studies of susceptibility to infection in HIV-positive patients 32 and systemic lupus erythematosus patients. 33 In our cohort, none of the MBL-deficient or MBL-insufficient patients had a pre-identified immune defect and could, in part, explain our lack of association with severity indices.
Limitations of the current study include our sample size as it does not permit accurate comparison of MBL deficiency among different nCF-Br aetiologies or other microbiological patterns, such as S. pneumoniae or Aspergillus sp. Additionally, we did not perform MBL genotyping, often measured in other clinical studies in place of, or in addition to, MBL serum concentrations. Although less than 5% of individuals with a homozygous ‘wild-type’ genotype (A/A) have deficient serum MBL levels, 34 selected normal MBL genotypes can have low MBL level and function. 35 Therefore, serum levels and functional assays of complement activation may be more practical tests. There has been more research interest recently into the influence of MBL-associated serine protease (MASP) levels on complement activation, 36 deficiency of which is regarded as a separate primary immunodeficiency. 37 Our study measured lung function on a cross-sectional basis with variation in time periods after disease onset. We are therefore unable to conclude from our data whether low MBL levels are associated with an accelerated decline in lung function, as was the case in CF patients. 31
Our study may have also been limited by the definition we used for ‘clinical stability’ (no infective episode in the preceding 2 wk). Some patients only report full recovery up to 4 wk, or even longer after an exacerbation. As MBL is an acute phase protein, its serum levels rise in response to infection, but in the absence of infection its serum levels are relatively stable. This may explain our relatively higher mean MBL levels (1885 ng/ml) than those previous published (1466 ng/ml). 21 A further limitation may be the use of lung function tests as a marker of disease severity without specific assessment of CT scores. The available CT scoring systems are, however, not universally accepted or widely implemented, so have little routine clinical applicability in contrast to FEV1%. We chose to measure only FEV1% as a marker of respiratory function. Bronchiectasis is commonly associated with an obstructive ventilatory deficit, but patients may also have restrictive spirometry or entirely normal lung function. The inclusion of forced vital capacity (FVC) and inspiratory capacity (IC) measurements may have provided us with additional markers of functional status.
In summary, our data show that low MBL levels are not associated with impaired lung function, exacerbation frequency or rates of P. aeruginosa or H. influenzae infection in adult patients with nCF-Br. These findings are in contrast to the findings of similar studies in patients with bronchiectasis caused by CF. To further investigate any potential association, studies assessing other aspects of alternative pathway complement activation with MBL genotype, MBL function and MASP level/function and L-ficolin would be most appropriate. 38 Our current data would support national clinical guidelines that advise restricting detailed immunological investigation to patients with abnormal screening tests or clinical presentation. Further multicentre studies will be required to validate our findings but our data do not support the widespread use of MBL level testing in the diagnostic work-up of nCF-Br.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
The authors are grateful to all the patients and staff in the specialist bronchiectasis service, Freeman Hospital, Newcastle-upon-Tyne, UK.
Conflict of interest
The authors have no conflicts of interest to declare.
