Abstract
Saliva interacts with blood after mucosal damage or leakage of gingival crevicular fluid. Surface-adsorbed salivary agglutinin (SAG) activates the lectin pathway (LP) of the complement system via mannose-binding lectin, while SAG in solution inhibits complement activation. In the present study we investigated if, next to SAG, whole and glandular saliva itself and other salivary glycoproteins activate or inhibit the LP. Complement activation was measured by detecting C4 deposition on microtiter plates coated with saliva or purified proteins. Complement inhibition was measured after incubating serum with saliva or proteins in microtiter plates coated with mannan, an LP activator. Adsorbed whole, sublingual and submandibular saliva showed LP-dependent complement activation. Blood group secretors, but not non-secretors, activated the LP. Saliva of both secretors and non-secretors inhibited C4 deposition on mannan. After depletion of SAG, saliva no longer inhibited the LP. Other salivary proteins, including amylase, MUC5B and histatin 2, did not activate or inhibit the LP. Surface-adsorbed whole saliva and glandular saliva samples activate the LP of complement, depending on the presence of SAG and the secretor status of the donor. In solution, saliva inhibits the LP, depending on the presence of SAG, but independent of the secretor status.
Introduction
Saliva is essential for the health of the oral cavity, where it is involved in processes such as lubrication, digestion of food, and protection of the teeth and oral mucosa. Most human saliva is produced by the larger pairs of parotid, sublingual (SL) and submandibular (SM) glands. In addition, saliva contains contributions from the minor buccal, labial, lingual and palatal salivary glands. 1 When there is a wound in the mucosa or when the gingiva become slightly damaged, for example after tooth brushing, blood comes into contact with saliva.2,3 Also in gingivitis and periodontal disease, gingival crevicular fluid, which originates from plasma, leaks into the oral cavity and allows the proteins in the different fluids to interact. 4
One group of proteins in plasma is the complement system, part of the innate immune system. Complement activation leads to a cascade of protein cleavages, ultimately leading to the formation of the anaphylatoxins C3a and C5a, which are chemotactic for leukocytes and have strong pro-inflammatory effects. Pathogens may be opsonized with C4b and C3b, which facilitate adhesion to phagocytes, leading to phagocytosis and killing of the pathogen. Additionally, the membrane attack complex is formed, which creates a pore in the cell membrane of a pathogen and causes cell lysis. Activation of the classical pathway (CP) or the lectin pathway (LP) leads to cleavage of complement components C2 and C4, resulting in deposition of C4b. IgG or IgM Abs within immune complexes are activators of the CP. Carbohydrates on pathogens, such as Man and fucose (Fuc), activate the LP via Man-binding lectin (MBL) or ficolins.5–8 Also salivary agglutinin (SAG) activates the LP of the complement system.
SAG, also known as gp-340 in the lungs and deleted in malignant brain tumors 1 (DMBT1), belongs to the scavenger receptor cysteine-rich (SRCR) superfamily. These proteins are characterized by the presence of SRCR domains, highly conserved protein fragments of 110 amino acids that play a role in bacterial binding. 9 In saliva, SAG causes agglutination of microorganisms, thereby promoting their clearance from the oral cavity. Surface-adsorbed SAG activates the complement system via the LP,10,11 while SAG in solution inhibits LP-mediated complement activation. 11 Activation of the complement system by SAG is mediated by Fuc-containing carbohydrate chains on SAG and is influenced by a donor’s secretor status. Secretors have the enzyme α-(1→2)-fucosyltransferase, and therefore express Lewis b, Lewis y and Ags from the ABO blood group system on SAG.12,13 It has been suggested that the Ags found on SAG of secretors are responsible for a higher complement activation, 14 although it is unknown whether these structures are also involved in complement inhibition by SAG. Saliva contains a number of other glycosylated proteins, such as amylase, the mucins MUC5B and MUC7 from the SM and SL glands, and proline-rich glycoproteins from parotid saliva, 15 which may potentially modulate the complement system via the LP as well. This could mean that in case of a wound or on infection, defense mechanisms of two different sources, saliva and blood, may collaborate to clear a potential pathogen via activation of the complement system or to avoid inflammation by inhibiting the complement system.
In the present study, we examined if, besides SAG, other salivary glycoproteins activate or inhibit the LP of the complement system. We found that SAG is the only glycoprotein in saliva that modulates the complement system through the LP.
Materials and methods
Collection of saliva
Human whole saliva was collected without stimulation from healthy donors, three secretors and three non-secretors (aged 25–63 yr). In addition, secretions of the SM and SL glands were separately collected from one donor (male, blood group B, secretor) using custom-fitted devices, as previously described. 16 From the same donor, parotid saliva was collected with a Lashley cup, under stimulating conditions (chewing on a sugar-free menthol chewing gum). All saliva samples were homogenized on a Vortex mixer (1 min) and centrifuged (10,000 g, 10 min) to remove cellular debris. The supernatant was stored at −20℃ until use. Whole saliva was subsequently mentioned clarified human whole saliva (CHWS).
Serum samples
Blood was withdrawn from MBL-sufficient (MBL+) and MBL-deficient (MBL−) healthy volunteers, allowed to clot for 1 h at room temperature (20–25℃) and centrifuged (2000 g, 10 min). Serum was stored at −80℃ until use. Informed consent was obtained from all donors.
Determination of saliva-mediated activation of complement
C4 deposition assays on saliva were conducted as previously described.10,14 Wells of microtiter plates (NuncMaxiSorp; Nunc, Roskilde, Denmark) were coated [overnight (16 h), 4℃] with saliva, twofold serially diluted in 0.1 M Na2CO3, pH 9.6. Serum (1%) was added in veronal buffer supplemented with 10 mM CaCl2, 2 mM MgCl2 and 0.02% Tween-20 (VB++) or in veronal buffer with 10 mM EDTA and 0.02% Tween-20 (VB/EDTA), and incubated for 1 h at room temperature, followed by incubation with biotinylated anti-C4-10 mouse mAb (Sanquin, Amsterdam, the Netherlands) 1:2000 diluted in PBS with 0.1% Tween-20 (PBST) supplemented with 1% BSA (blocking buffer). HRP-conjugated avidin (Abcam, Cambridge, UK) (0.5 µg/ml in blocking buffer) was added (30 min, room temperature), followed by 100 µl o-phenylenediamine (OPD) substrate (SIGMAFAST; Sigma-Aldrich, St. Louis, MO, USA). Reaction was stopped by the addition of 50 µl 1 M H2SO4 and the absorbance at 492 nm was measured using a Multiskan FC microplate photometer (Thermo Scientific, Rockford, IL, USA). After each incubation step, plates were washed three times with PBST.
ELISA of SAG in saliva
Saliva-coated microtiter plates (NuncMaxiSorp) were incubated in blocking buffer for 1 h, and then incubated with mouse mAb 213-06 HYB (Bioporto, Hellerup, Denmark) directed to a protein epitope of SAG (1:2000 diluted in blocking buffer). After washing, plates were incubated with HRP conjugated to rabbit anti-mouse IgG (DAKO, Glostrup, Denmark) (1:2000 in blocking buffer, 1 h, room temperature). After each incubation step, plates were washed three times with PBST. Visualization of the reaction was conducted as described above using SIGMAFAST OPD as substrate.
Inhibition of complement activation by saliva
Microtiter plate wells were coated with 10 µg/ml mannan (Sigma-Aldrich) (in 0.1 M Na2CO3, pH 9.6). 10 Wells were incubated with serial dilutions of serum in VB++ supplemented with 20% saliva or 20% saliva buffer (2 mM potassium phosphate, 50 mMKCl, 1 mM CaCl2, 0.1 mM MgCl2, pH 6.8) as control. Incubation steps and detection of C4 deposition were performed as described above. Binding of MBL was determined with biotinylated anti-MBL/1 (Sanquin), 1:1000 diluted in VB++, followed by incubation with streptavidin poly-conjugated with HRP (1:5000; Sanquin). For MBL-binding assays plates were washed with H2O between incubation steps.
Determination of secretor status
The secretor status of each donor was determined as earlier described, 17 with minor alterations. Briefly, microtiter plate wells were coated with saliva (1:800 diluted in 0.1 M Na2CO3, pH 9.6) and blocked with blocking buffer. After washing, biotinylated α-Fuc-(1→2)-Gal-binding Ulex europaeus agglutinin (UEA-1; Pierce, Rockford, Il, USA) (1:2000 diluted in blocking buffer) was added, followed by incubation for 1 h. Wells were washed and incubated with HRP-conjugated avidin (Abcam) (0.5 µg/ml in blocking buffer) for 30 min. Color was developed with SIGMAFAST OPD. Donors with a positive result for UEA-1 binding were defined as secretors.
Salivary proteins
Human amylase was from Sigma-Aldrich. Histatin 2 was synthesized by solid-phase peptide synthesis using Fmoc chemistry with a MilliGen 9050 peptide synthesizer (Milligen-Biosearch, Bedford, MA, USA) and purity was checked by mass spectrometry, as previously described. 18 SAG was purified from parotid saliva by applying a precipitate on a Sepharyl S-400 gel filtration column as described before.19,20 SAG-enriched samples were obtained as previously described and dissolved in 150 mM NaCl. 14 MUC5B was isolated from human saliva as described by Veerman et al. 21
Preparation of SAG-depleted saliva
Streptococcus mutans Ingbritt was cultured overnight in 10 ml tryptic soy broth at 37℃. Bacteria were washed and re-suspended in 2.5 ml PBS. Saliva samples were incubated with an equal volume of an S. mutans Ingbritt suspension or with PBS for 1 h at 37℃. Bacteria were pelleted by centrifugation (14,000 g, 5 min). The supernatant was centrifuged again (14,000 g, 20 min) and stored at −20℃ until use.
SDS-PAGE analysis of saliva
SDS-PAGE was performed on NuPAGE 4–12% BisTris gels (Thermo Scientific) under reducing conditions. Saliva samples were boiled for 10 min with LDS sample buffer and reducing agent (Thermo Scientific). Samples were loaded on the gel and run for 35 min at 120 V. Novex sharp pre-stained proteins standards (Thermo Scientific) were used as molecular mass markers. Subsequently, gels were incubated with Coomassie R 350 stain (GE Healthcare, Uppsala, Sweden) for 2 h at room temperature, followed by overnight de-staining with 10% acetic acid.
MALDI-TOF analysis
After electrophoresis, protein bands of interest were cut out of the gels with a razor blade, sliced into smaller pieces, washed with 25% acetonitrile and 0.1% trifluoroacetic acid (TFA) in water, and sonicated for 30 min in 25% acetonitrile and 0.1% TFA. The samples were extracted from the gel pieces with 500 µl 65% acetonitrile and 0.1% TFA in water overnight at room temperature while shaking. The samples were lyophilized in a Speed vac evaporator (RVC 2–25 CD plus; Martin Christ, Osterode am Harz, Germany), dissolved in 50 µl H2O, vortexed and lyophilized again. Then, the samples were analyzed by MALDI-TOF-MS as described previously. 19
Results
Adsorbed saliva activates complement in an MBL-dependent way
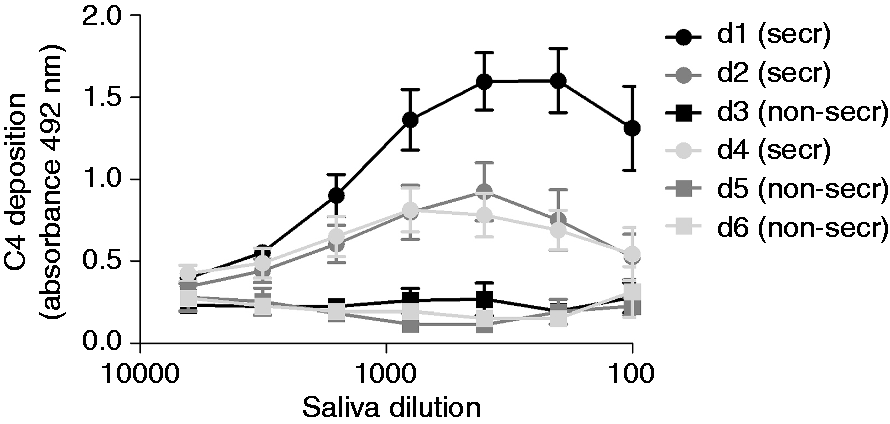
The complement-activating properties of whole and glandular human saliva were investigated by determining saliva-mediated C4 deposition. To this end, serum was incubated on saliva-coated microtiter plates (Figure 1). Serum diluted in a buffer containing 10 mM EDTA was included as a negative control. To explore whether the CP or the LP was involved, both MBL+ and MBL− serum were used. C4 deposited only with MBL+ serum on all types of saliva, indicating that saliva activates the complement system via the LP. Most C4 deposition was found for CHWS, followed by SM and SL saliva, while the least C4 deposition was found for parotid saliva. The amount of C4 deposition corresponded with the amount of SAG present in the different samples that were coated on the microtiter plates as determined with anti-SAG (Figure 1e). Equal amounts of SAG were detected on microtiter plates coated with CHWS, SM and SL saliva, whereas SAG was hardly detectable on microtiter plates coated with parotid saliva. C4 deposition was also tested in CHWS samples of three secretor and three non-secretor donors, as defined by UEA-1 binding (Supplementary Figure 1). C4 deposited on saliva of secretors, but not on saliva of non-secretors (Figure 2), in line with a previous study of SAG.
14
Complement activation by saliva secretions. Serial dilutions of (a) CHWS, (b) parotid saliva (c) SL saliva and (d) SM saliva from one donor (positive secretor status) were coated in microtiter plates, incubated with sera (1% in veronal buffer) and C4 deposition was measured. (e) SAG content of serially diluted saliva secretions, measured in an ELISA. Each point is the mean ± SE of three experiments performed in duplicate. VB++: veronal buffer supplemented with 10 mM CaCl2 and 2 mM MgCl2; VB/EDTA: veronal buffer supplemented with 10 mM EDTA; MBL+: serum containing MBL (two donors); MBL−: serum without MBL (two donors). The effect of the secretor status on saliva-mediated complement activation. CHWS of six donors (d1–d6) was coated in serial dilutions on microtiter plates. After incubation with MBL+ serum (1% in VB++) C4 deposition was measured. These results show the mean ± SE of three experiments performed in duplicate. secr: secretor; non-secr: non-secretor.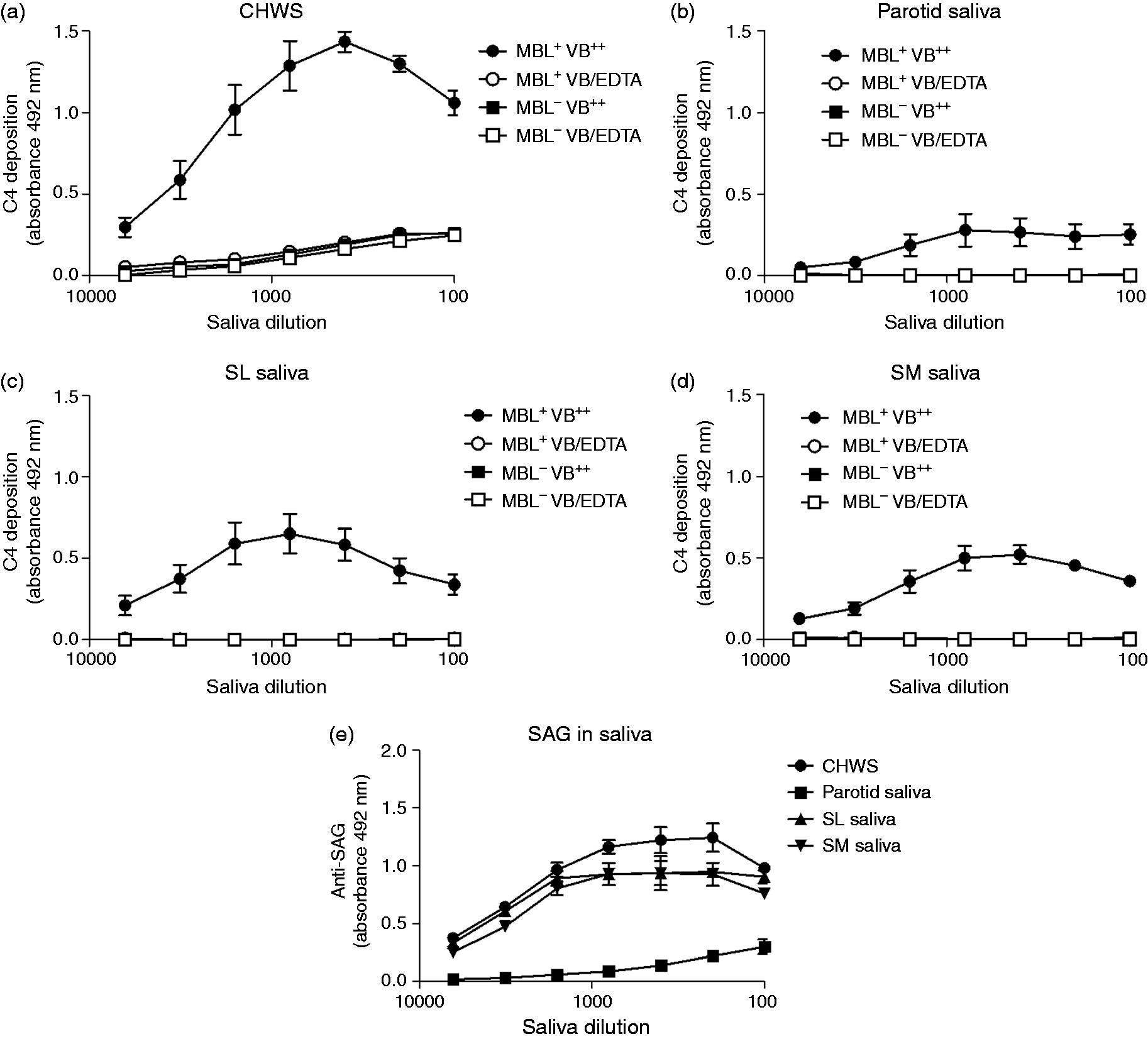
Saliva inhibits complement activation and MBL binding in solution
To investigate the complement-inhibiting properties of saliva, MBL+ serum was twofold serially diluted in VB++ in the presence of 20% saliva solution or saliva buffer, which simulates the ionic composition of saliva, and added to mannan-coated microtiter plate wells. After incubation, deposition of C4 was determined. CHWS and SM saliva of a secretor inhibited both the C4 deposition on mannan and the MBL binding to mannan for 85–90% (Figure 3a, b), while parotid saliva had little, if any, effect. In contrast to C4 activation by adsorbed saliva, which was only mediated by saliva from secretors, saliva of both secretors and non-secretors inhibited the MBL-mediated C4 deposition in solution (Figure 3c), as well as the binding of MBL to mannan (Figure 3d). These results suggest that different interactions are responsible for activation and inhibition of the LP, depending on whether saliva is in adsorbed phase or in solution.
Inhibition of the MBL pathway by saliva in solution. Serial dilutions of MBL+ serum in VB++ were mixed with 20% glandular saliva samples from (a, c) one donor or (b, d) CHWS from different secretor (n = 3) and non-secretor (n = 3) donors. (a, b) C4 deposition on mannan and (c, d) MBL binding to mannan were detected. VB++ with 20% saliva buffer was used as a control. Values are presented as means ± SE of three experiments performed in duplicate.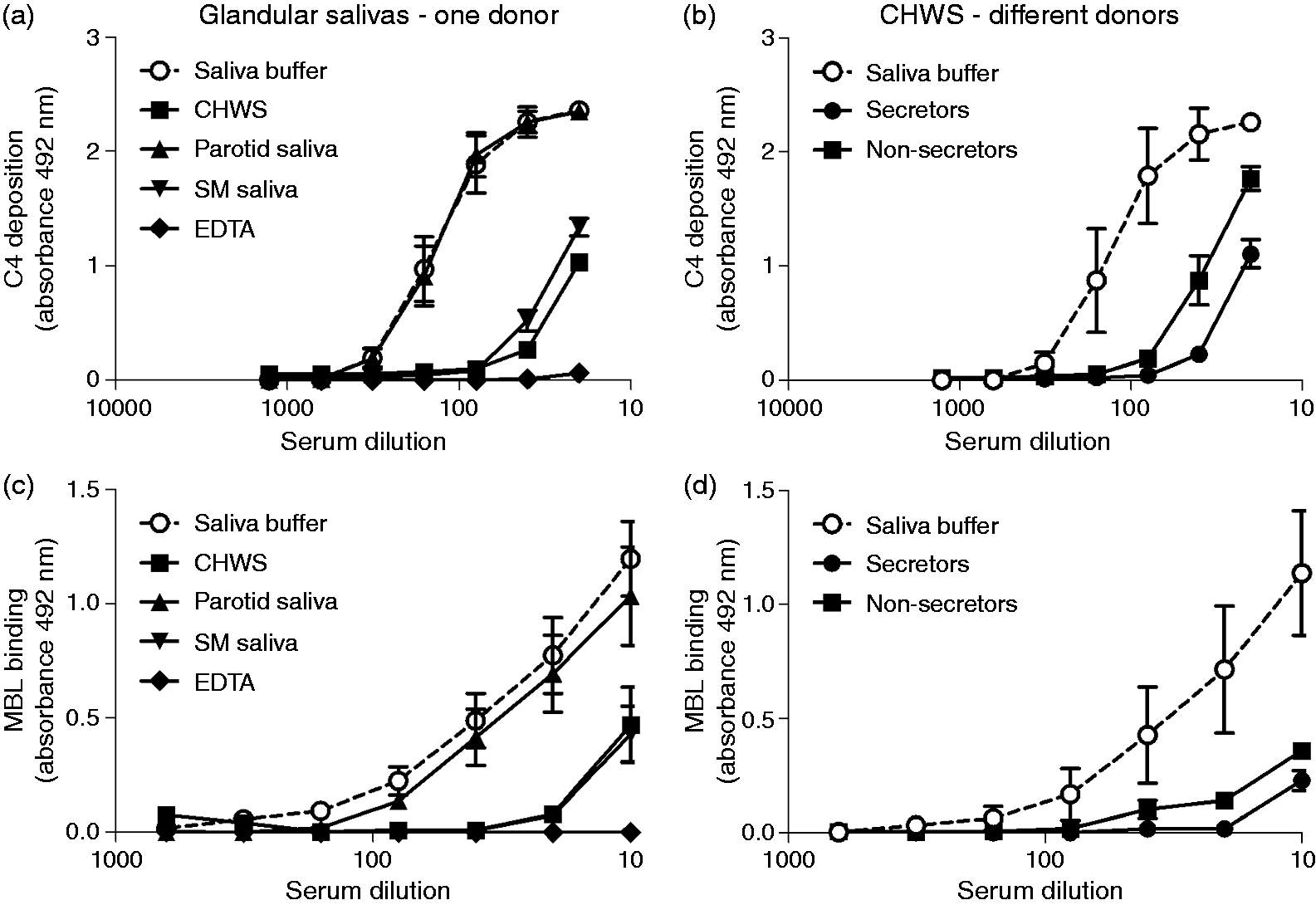
Effect of SAG depletion on the complement-activating and complement-inhibiting properties of saliva
To test if besides SAG, other salivary proteins modulate the LP, saliva samples of one secretor donor were depleted from SAG by incubation with S. mutans. ELISA revealed that by these treatments approximately 70% of SAG was depleted from CHWS, 90% from SL saliva and virtually 100% from SL saliva (Figure 4a). SDS-PAGE followed by Coomassie Blue staining, showed that other salivary proteins were not depleted, except for a peptide from parotid and SM saliva (Figure 4b). MALDI-TOF analysis of the band excised from the gel revealed that this was histatin 2 (Figure 4c).
SAG in saliva after depletion with S. mutans. (a) The presence of SAG was demonstrated with an ELISA on serially diluted immobilized saliva samples. (b) SDS-PAGE and Coomassie staining showed that SAG and histatin 2 were depleted from saliva after incubation with S. mutans, while other proteins remained present. An arrow indicates the position of SAG and the arrowhead indicates the position of putative histatin. (c) The putative histatin band was excised and analyzed by MS. This showed a peak of 3442.045 Da (lower panel), which was identical to synthesized histatin 2 (3442.872 Da, upper panel), and therefore confirmed the band on the gel was histatin 2.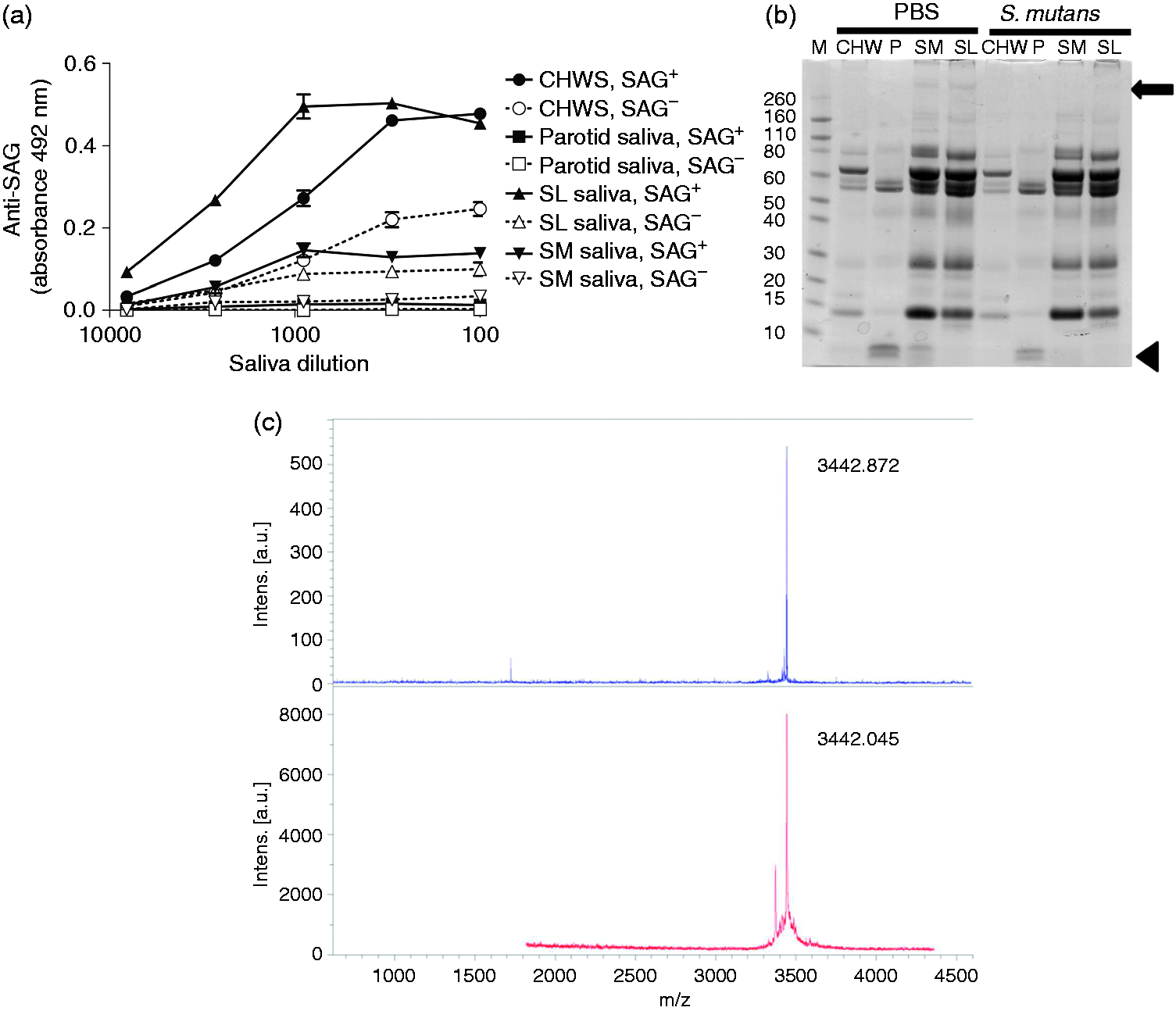
Next, the SAG-depleted samples were tested for C4 activation or inhibition in comparison with the SAG-containing samples. Less C4 deposited on SAG-depleted CHWS and SL saliva than on the SAG-containing samples (Figure 5a). This corresponded with the lower amount of SAG detected in the ELISA (Figure 4a). In line with this, little C4 deposited on SM saliva (Figure 5a). CHWS and SM saliva in solution, but not the SAG-depleted saliva samples, inhibited MBL-mediated deposition of C4 on mannan, partially (CHWS; Figure 5b) or completely (SM saliva; Figure 5c). The partial inhibition by SAG-depleted CHWS may be due to residual SAG in this sample (Figure 4a).
Complement activation and inhibition by SAG-depleted saliva. (a) To measure complement activation, sera (1% in VB++) were incubated in microtiter plates coated with serially diluted SAG-containing or SAG-depleted saliva and C4 deposition was detected. To measure complement inhibition, serial dilutions of MBL+ serum from three donors in VB++ were mixed with 20% SAG-containing or SAG-depleted (b) CHWS samples and (c) SM saliva samples and C4 deposition on mannan was measured. Values are means ± SE of three samples of an independent experiment, which was repeated at least twice. Statistically significant differences between SAG-containing and SAG-depleted saliva are shown, which were calculated using one-way ANOVA and Tukey’s post-test (*P < 0.05, **P < 0.01, ***P < 0.001). SAG−: SAG-depleted saliva; SAG+: SAG-containing saliva.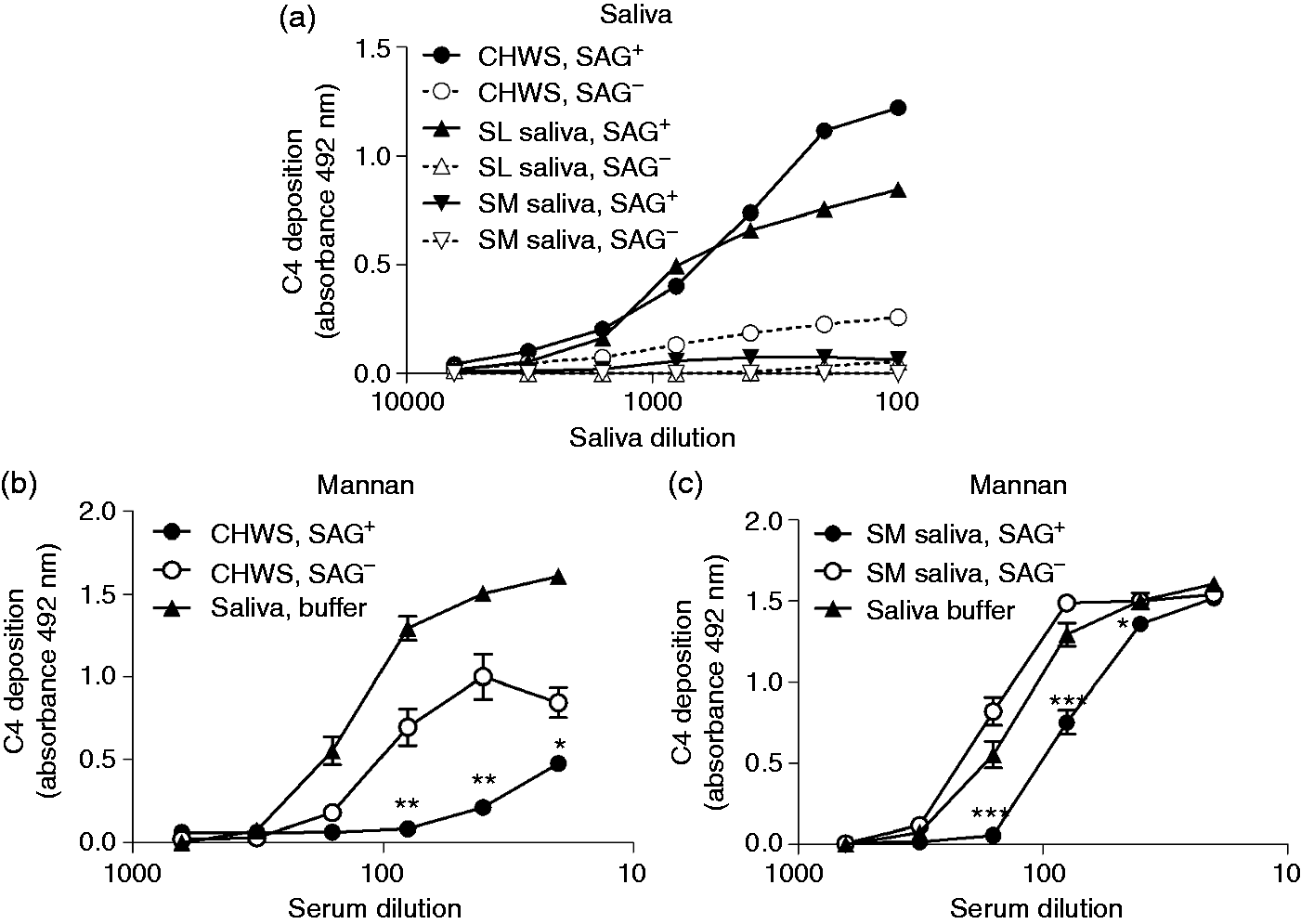
In addition to SAG, saliva contains a number of other glycosylated proteins, such as MUC5B, MUC7 and amylase, which could potentially activate the LP by binding carbohydrate recognition domains of MBL.
15
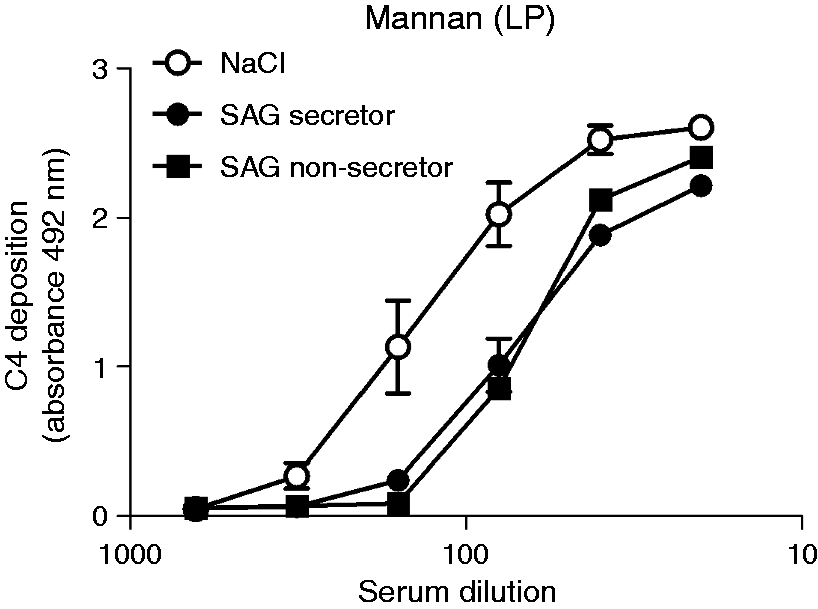
Thus, we tested the complement-modulating properties of a number of these proteins. Furthermore, we tested the un-glycosylated peptide histatin 2 because, besides SAG, it was the only compound that was removed from saliva by treatment with S. mutans (Figure 4b). C4 deposition was only found on adsorbed SAG, but not on amylase, MUC5B or histatin 2 (Figure 6a). In line with this, MBL bound only to adsorbed SAG but not to amylase, MUC5B or histatin 2 (Figure 6b). In solution, only purified SAG inhibited the C4 deposition on mannan, suggesting that when saliva is in solution, SAG is the major inhibitor of the MBL pathway. As complement activation by saliva is dependent on the secretor status, while complement inhibition by saliva in solution is independent of the secretor status, we assessed the complement-inhibiting properties of SAG-enriched parotid saliva samples from a secretor and a non-secretor. In solution, both SAG samples inhibited C4 deposition on mannan to the same extent (Figure 7). Taken together, these results show that SAG is the protein in saliva involved in LP modulation and that LP inhibition by saliva or SAG in solution is independent of a donor’s secretor status.
Effect of isolated salivary proteins on the MBL pathway. One µg/ml of SAG, amylase, MUC5B and histatin 2 were coated on microtiter plates, incubated with MBL+ serum (1%), and tested for complement activation by detecting (a) C4 deposition and (b) MBL binding. (c) Inhibition of the MBL pathway was measured. MBL+ serum from three donors was serially diluted and incubated in VB++ in the presence of 10 µg/ml (20% of total volume) SAG of a secretor donor, amylase, MUC5B, histatin 2 or TBS and C4 deposition on mannan was measured. Values are means ± SE of three samples of an independent experiment, which was repeated at least twice. In (a), statistically significant differences between means were calculated using one-way ANOVA and Tukey’s post-test. Statistical significance of differences compared with the blank (b) or TBS (c) were estimated using one-way ANOVA and a Dunett post-test (***P < 0.001). Inhibition of C4 deposition by SAG. MBL+ sera were serially diluted in VB++ supplemented with 10 µg/ml SAG (20%) of a secretor (filled circle), a non-secretor (filled square) or 30 mM NaCl (open circle) (NaCl), C4 deposition on mannan was measured. Results represent mean ± SE of three experiments performed in duplicate.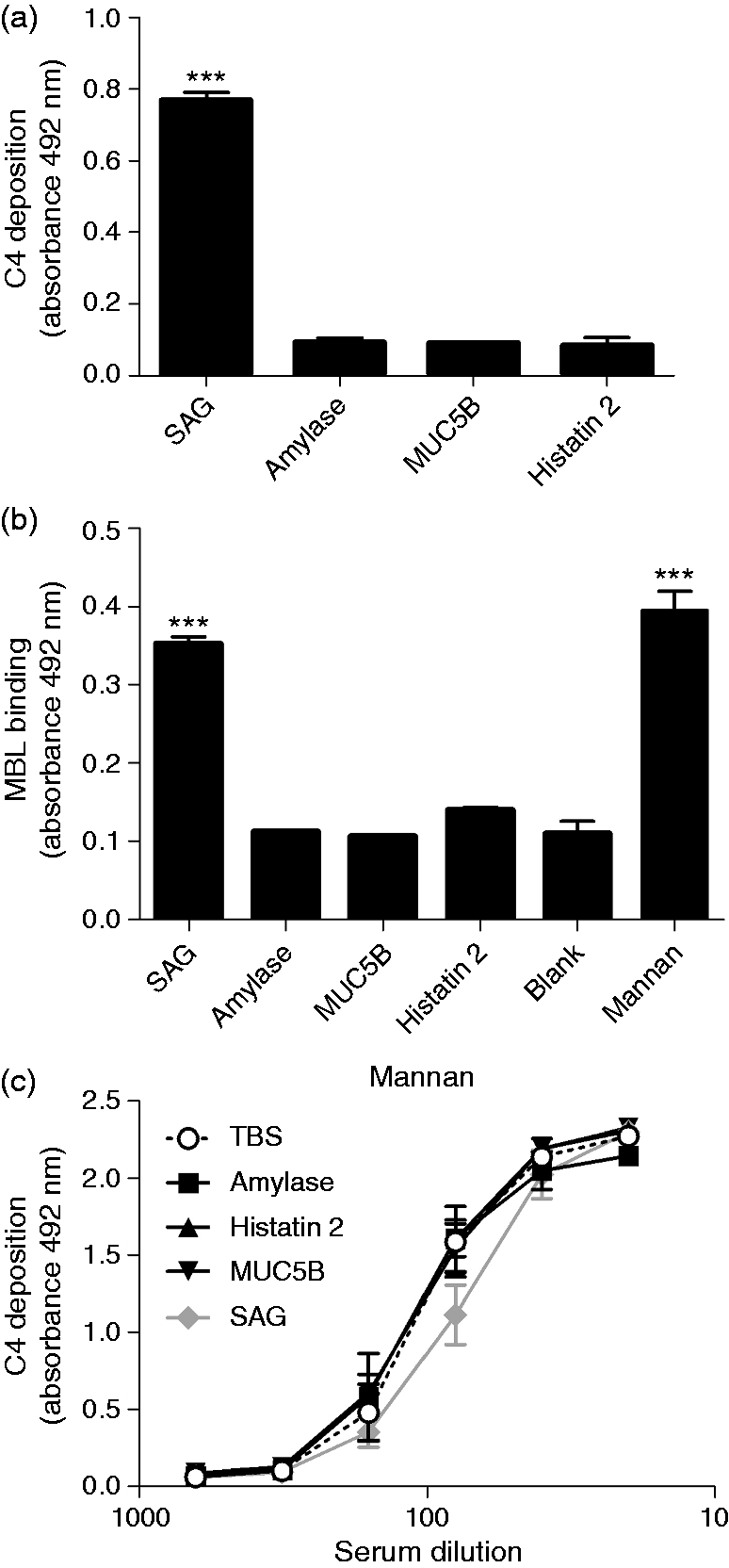
Discussion
In this study we obtained evidence that SAG is the only component in saliva that interacts with the MBL pathway of the complement system. In line with previous studies SAG or saliva, when adsorbed onto a surface, activates complement, resulting in deposition of C4 on SAG. In solution, however, SAG and SAG-containing secretions inhibited mannan-induced complement activation.10,11
CHWS of secretors activated the complement system, while CHWS of non-secretors did not. This is in accordance with previous research that showed that complement activation and MBL binding is higher on SAG of secretors than on SAG of non-secretors. 14 Other studies showed residual C4 deposition when complement activation was measured on SAG with MBL− serum,10,11 although we did not observe this for our CHWS samples (Figure 1). This implies that there is a difference between purified surface-adsorbed SAG and SAG in surface-adsorbed CHWS. Purified SAG might have more accessible regions for ficolins and C1q. The complement-inhibiting activity of saliva, however, was independent of the secretor status (Figure 3), suggesting that protein domains rather than carbohydrate epitopes are involved in this process. This is in line with a previous study showing that after heat treatment, which affects the protein conformation of SAG, SAG no longer inhibited the binding of MBL to Candida albicans. 11 An alternative explanation is that, in solution, monofucosylated Ags, such as Lewis a and Lewis x, which are present on SAG of non-secretors, may also bind to MBL. Indeed, we previously found that the Fuc monosaccharide inhibits MBL-mediated complement activation. 14
The existence of several inter-individual differences in glycosylation and isoforms has been reported for SAG.22–25 This may also have an effect on the complement-modulating properties of SAG. Future studies using well-characterized SAG isoforms should explore in more detail to what extent the various SAG isoforms differ in their complement-modulating activities. MUC7 and MUC5B, although both highly glycosylated, did not activate the complement system. The structures on SAG involved in the complement-activating properties are Fuc-containing Ags, in particular Lewis b and Lewis y.14,26 The majority of the Lewis Ags on MUC7, in both secretors and non-secretors, are sialylated Lewis a and sialyl-Lewis x, which may not bind to MBL. 27 MUC5B from secretors contain Lewis b Ags, but neither activated nor inhibited the MBL pathway, suggesting that the presence of Fuc-containing oligosaccharides by itself is not sufficient for complement activation. This may be owing to the fact that MUC5B carries an extremely heterogeneous repertoire of oligosaccharides. 28 It can be envisaged that the lack of clusters of identical oligosaccharides will hamper the formation of a stable multivalent binding between a polyvalent lectin such as MBL and its cognate carbohydrate ligands.
The complement system is activated in periodontal inflammation. The disease is induced by bacteria, but susceptibility and progression of the disease are determined by the host’s response.29,30 Continuous activation of the complement system may enhance local inflammation and tissue destruction. Despite activation of the complement system, periodontal pathogens have complement-evading proteins. 29 The bacteria resist immune-mediated killing and the subgingival biofilm remains intact, thus constantly triggering inflammation. 31 For future research, it would be interesting to determine the presence of salivary proteins, such as SAG, in the supra-gingival or subgingival area, and to investigate how this modulates the complement system. Diminished MBL binding, C4 deposition and C3 deposition was found on C. albicans when MBL or human serum was pre-incubated with recombinant SAG in solution. 11 When saliva is coated on a bacterial surface, complement proteins may bind to SAG and activate downstream pathways, while saliva in solution might prevent complement proteins from binding to other surfaces, thereby inhibiting further activation and subsequent inflammation. We should consider the dual role of SAG in both complement activation and inhibition, and keep in mind that LP activation by adsorbed SAG is individually different, determined by a donor’s secretor status, while LP inhibition by SAG or saliva in solution is equal among secretors and non-secretors.
Footnotes
Acknowledgements
This study was supported by the Academic Centre for Dentistry Amsterdam (ACTA) Graduate School.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
