Abstract
Nicotinamide phosphoribosyltransferase (NAMPT), an enzyme involved in NAD biosynthesis, has recently been identified as a novel mediator of innate immunity.
In the present study, we report that treatment of LPS-primed monocytes with ATP greatly enhanced the secretion of NAMPT in a time- and concentration-dependent manner without displaying any cytotoxic effect. NAMPT release was suppressed by pretreatment with the P2X7 receptor (P2X7R) inhibitors oxidized ATP (oxATP) and KN-62, indicating the engagement of P2X7Rs. Furthermore, P2X7R was found to be involved in mediating cell permeability caused by the addition of ATP. To define a role of endogenous ATP in NAMPT secretion, LPS-primed monocytes were incubated in the presence of oxATP and KN-62, as well as the ATP-hydrolyzing enzymes apyrase and hexokinase. With the exception of oxATP, neither substance led to a decrease in NAMPT release, suggesting that autocrine/paracrine ATP is unlikely to be responsible for the LPS-induced release of NAMPT. In conclusion, the enhanced release of NAMPT by extracellular ATP described here indicates the requirement of a second stimulus for the efficient secretion of NAMPT. This mode of secretion, which also applies to IL-1β, might represent a general mechanism for the release of leaderless secretory proteins at locally restricted sites.
I ntroduction
NAD, initially characterized as a coenzyme for oxidoreductases, also serves as a substrate for NAD-consuming enzymes, such as mono-ADP-ribosyltransferases, 1 poly-ADP-polymerases 2 and sirtuins. 3 A common feature of these enzymatic reactions is the breakdown of NAD and the formation of free nicotinamide. 4
To avoid depletion of intracellular NAD eukaryotic cells have evolved a rescue pathway, leading to NAD re-synthesis from nicotinamide. 4 The rate-limiting enzyme in the rescue pathway is nicotinamide phosphoribosyltransferase (NAMPT). Its expression is widely induced by inflammatory stimuli in immune cells. 5 By inhibiting NAMPT activity in activated human monocytes we recently showed that the cells strongly depend on this enzyme to mount an appropriate inflammatory response. 6
In addition to its function as an intracellular enzyme there is some evidence that NAMPT also has a role as an extracellular mediator, showing cytokine-like effects. Recombinant NAMPT has been shown to induce cytokine production in monocytes and to increase cell surface expression of co-stimulatory molecules. 7
Moreover, it displays anti-apoptotic effects in macrophages 8 and neutrophils 9 . When NAMPT was first discovered as a visceral fat-derived adipokine 10 its role in glucose metabolism and obesity has been extensively studied, however, with contradictory results. 5
Its increased expression and release has also been associated with the pathogenesis of acute and chronic conditions, including acute lung injury, 11 atherosclerosis, 12 rheumatoid arthritis 13 and inflammatory bowel disease. 7
The extracellular appearance of NAMPT raises the question of how it can be transported out of the cell. Analysis of mRNA transcripts revealed that NAMPT does not contain cytokine-specific secretion sequences such as a consensus leader sequence or a canonical caspase-1 cleavage site. 14 Furthermore, NAMPT secretion was not reduced by brefeldin A and monensin, inhibitors of endoplasmic reticulum (ER)-Golgi-dependent protein secretion. Thus, NAMPT seems to be secreted through a non-classical secretory pathway. 15 One well characterized representative of proteins that do not follow the classical ER-Golgi pathway is IL-1β, a key initiator of an acute inflammatory response. For the efficient generation of active IL-1β two separate stimuli appear to be required: a priming stimulus and a secretion stimulus. 16
As ATP has been identified as a potent secretion stimulus, 16 we questioned whether this nucleotide is also capable of stimulating the release of NAMPT. We found that NAMPT secretion is greatly enhanced by extracellular ATP in LPS primed monocytes, whereas endogenous ATP does not seem to be involved in its release to the extracellular compartment.
Materials and methods
Reagents
Unless otherwise indicated, materials used in this study were from the following manufacturers: RPMI 1640 (with
Cell separation and cell culture
Human peripheral blood monocytes from healthy donors were obtained by counterflow elutriation using the JE-6B elutriation system and cultured as described previously. 17
Detection of NAMPT and IL-1β protein in culture supernatants
NAMPT and IL-1β release into cell culture supernatants was quantified by NAMPT-ELISA (Adipogen, San Diego, CA, USA) and IL-1β-ELISA (BD Bioscience, Franklin Lakes, NJ, USA), respectively, according to the manufacturer’s instructions.
MTT Assay
The viability of monocytes was determined by the MTT assay, a quantitative method based on the metabolic reduction of tetrazolium salt (MTT) into purple formazan-crystals. Monocytes (2 × 106/ml) were incubated in the presence of 0.3 mg/ml MTT. After 2 h the cells were lysed and incubated for another 6 h before absorbance was read at 570 nm. Every sample was measured in duplicate.
Lactate dehydrogenase assay
Lactate dehydrogenase (LDH) release into cell culture supernatants was quantified using a Cytotoxicity Detection Kit (Roche) following the manufacturer’s instructions. To determine the maximum release of LDH, monocytes were lysed with 1% Triton X-100. The spontaneous LDH release was calculated as percentage of the maximum LDH release.
Analysis of pore formation by flow cytometry
A 500 µl sample of human monocytes (2 × 106/ml) was re-suspended in culture medium and incubated for 15 h with LPS before the addition of oxATP or KN-62. After 15 min 2.5 µM ethidium bromide was added and the monocytes were incubated in the presence or absence of 2 mM ATP for another 15 min. Thereafter, cells were washed twice with PBS and measured by FACS analysis (Becton Dickinson, San Jose, CA, USA).
Statistical analyses
Statistical significance was calculated with the tests indicated and classified as follows: *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.
Each experiment (n) refers to a preparation of monocytes isolated from one healthy donor.
Results and discussion
ATP triggers NAMPT release in a dose- and time-dependent fashion
There is long standing evidence that ATP induces a variety of different responses in immune cells. 18 In monocytic cells, it leads to a drastic release of mature IL-1β when added after a primary stimulus.18–20
Considering that NAMPT, similarly to IL-1β, lacks a signal sequence, thus precluding its secretion via the classical endoplasmic reticulum to Golgi pathway, we reasoned that its efficient release from activated cells may require a second stimulus, such as extracellular ATP. When adding different concentrations of ATP for 1 hr to monocytes that had been incubated for 15 h with LPS, we observed a marked dose-dependent increase in NAMPT release over that induced by LPS (Figure 1A). As there was hardly any difference in NAMPT release at doses of 2 and 3 mM ATP, a concentration of 2 mM was used in the following experiments. According to the kinetics carried out with 2 mM ATP (Figure 1B), the release of NAMPT was found to be highest after an incubation time of 60 min. In the absence of ATP, extracellular NAMPT concentrations rose from 2.2 ± 1.1 ng/ml (mean ± SD; untreated controls; n = 12) to 10.4 ± 5.8 ng/ml (mean ± SD; P ≤ 0.001, student’s t-test; n = 12) in response to LPS. According to the large SD there was a substantial LPS-dependent donor-dependent variation in NAMPT release. Such variation was also observed when treating the activated cells with ATP (Figures 1A, B). Under the here-described experimental conditions we also found IL-1β to be secreted in response to ATP. While LPS-activated monocytes stimulated for 15 h with LPS secreted 8.7 ± 7.5 ng/ml (mean ± SD; n = 4) IL-1β, the subsequent incubation with ATP (2 mM) for 1 h led to a release of 73.3 ± 32.0 ng/ml (mean ± SD; P ≤ 0.01, student’s t-test; n = 4).
Effect of ATP on NAMPT and LDH release after LPS-priming. Monocytes (2 × 106/ml) were incubated in the presence of LPS (100 ng/ml) for 15 h before adding ATP in the given concentrations for 1 h (A) or 2 mM ATP for various times up to 1 h (B). NAMPT concentrations in the supernatants were determined by ELISA. Bars represent means ± SEM (n = 3); one-way ANOVA with Bonferroni post-hoc test calculated to the controls (black bar = 100%; values obtained in the absence of ATP). (C) Monocytes (2 × 106/ml) were incubated in the presence of LPS (100 ng/ml) for 15 h before ATP (2 mM) was added. After 15, 30 and 60 min, supernatants were assayed for LDH release. Bars represent means ± SEM (n = 3); calculated to total releasable LDH (100%). **P ≤ 0.01.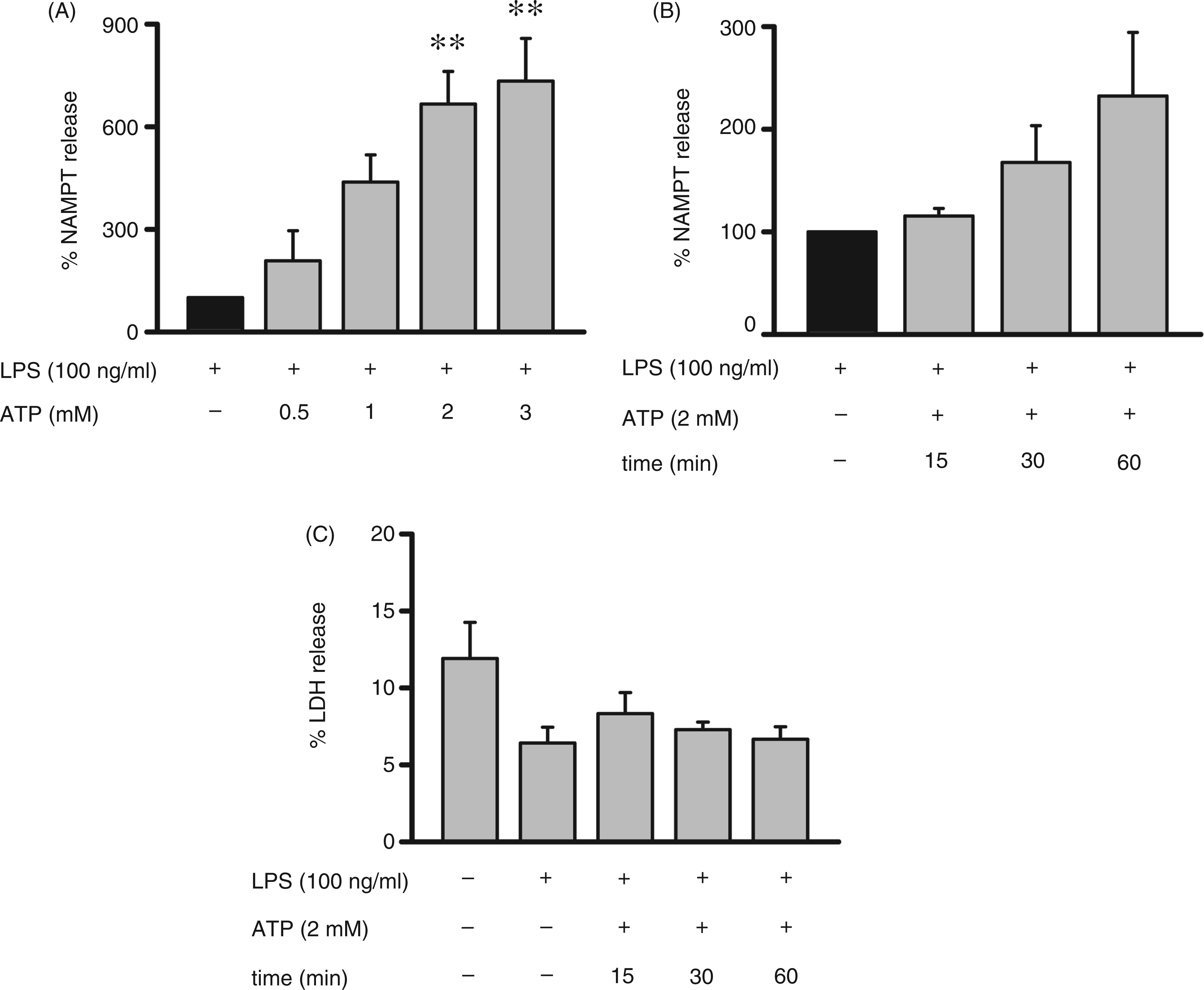
ATP does not display any cytotoxic effects
To assess whether the release of NAMPT was a result of cell death, we quantified LDH in the cell supernatants as a marker of cell damage. Figure 1C shows that slightly elevated and low levels of LDH are detectable in supernatants of unstimulated and stimulated cells, respectively, and that exposure of stimulated cells to ATP up to 60 min had no effect on LDH release. The increased release of LDH by unstimulated monocytes seems to correlate with a loss in cell viability which, as shown previously, 6 amounts to 15–20% after a 16 h incubation time.
Consistent with this finding cell viability, as measured by the use of the MTT test (a method suited to determine the metabolic activity of the cells), was not affected by ATP (data not shown). Although the mechanism of ATP-induced NAMPT release is unknown, the low, but similar, levels of LDH released from LPS-activated monocytes treated with or without ATP and the fact that the kinetics of NAMPT and LDH release did not match, argue against an unspecific release caused by ATP-induced cell lysis. These data are in line with those reported by Ferrari et al. and Sluyter et al.20,21 Measuring ATP-induced IL-1β release from human monocytes and macrophages both authors claim that this process is not necessarily associated with cell death. However, the susceptibility to ATP-induced cell lysis may very well be species- and cell type-dependent.
Engagement of P2X7 receptors in NAMPT release
To determine how far ATP receptors participate in the ATP-induced NAMPT release we pretreated the cells with PPADS, a selective P2 receptor antagonist,
22
before adding ATP. As shown in Figure 2A, PPADS led to a significant decrease in NAMPT release, indicating the engagement of P2 receptors. To define the receptor subtype mediating the ATP effect we treated the cells with the P2 receptor agonist BzATP, a potent activator at both the P2X1 and P2X7 receptor.23,24 Having found that BzATP strongly induced the release of NAMPT, we next incubated the cells with suramin prior to the addition of ATP. Suramin is highly active as a P2X1 and weak or nearly inactive as a P2X7 and P2X4 receptor antagonist.
25
The fact that suramin did not inhibit the ATP-induced NAMPT release (Figure 2A) argues against the participation of P2X1 receptors. A more likely candidate is the P2X7R. Indeed, we showed that ATP activity can be inhibited by either KN-62 (Figure 2A), an isoquinoline derivative widely used as a potent and selective antagonist at the P2X7R, and oxATP, an efficient P2X7R blocker.26,27 Furthermore, the concentration of ATP (∼1 mM) required to promote the release of NAMPT is in line with the requirements of the P2X7R.
28
We therefore conclude that the effect of ATP on NAMPT release involves engagement of the P2X7R at the binding site for ATP.
ATP induces P2X7R-dependent NAMPT release and pore formation. (A) Monocytes (2 × 106/ml) were incubated in the presence of LPS (100 ng/ml) for 15 h. Before addition of ATP (2 mM) or BzATP (300 µM) for 1 h, the cells were treated with P2X7R inhibitors for 15 min. NAMPT concentrations in the supernatants were determined by ELISA. Bars represent means ± SEM (n = 3); one-way ANOVA with Bonferroni post-hoc test calculated to the controls (black bars = 100%; values obtained in the presence of ATP alone or together with DMSO). (B) Monocytes (2 × 106/ml) were incubated in the presence of LPS (100 ng/ml) for 15 h. Before addition of ethidium bromide (2.5 µM) and ATP (2 mM) for 15 min the cells were treated with P2X7R inhibitors for 15 min. After washing the cells twice with PBS ethidium bromide uptake was measured by FACS-analysis. Bars represent means ± SEM (n = 3); one-way ANOVA with Bonferroni post-hoc test calculated to the controls (black bars = 100%; values obtained in the presence of ATP alone or together with DMSO). *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001.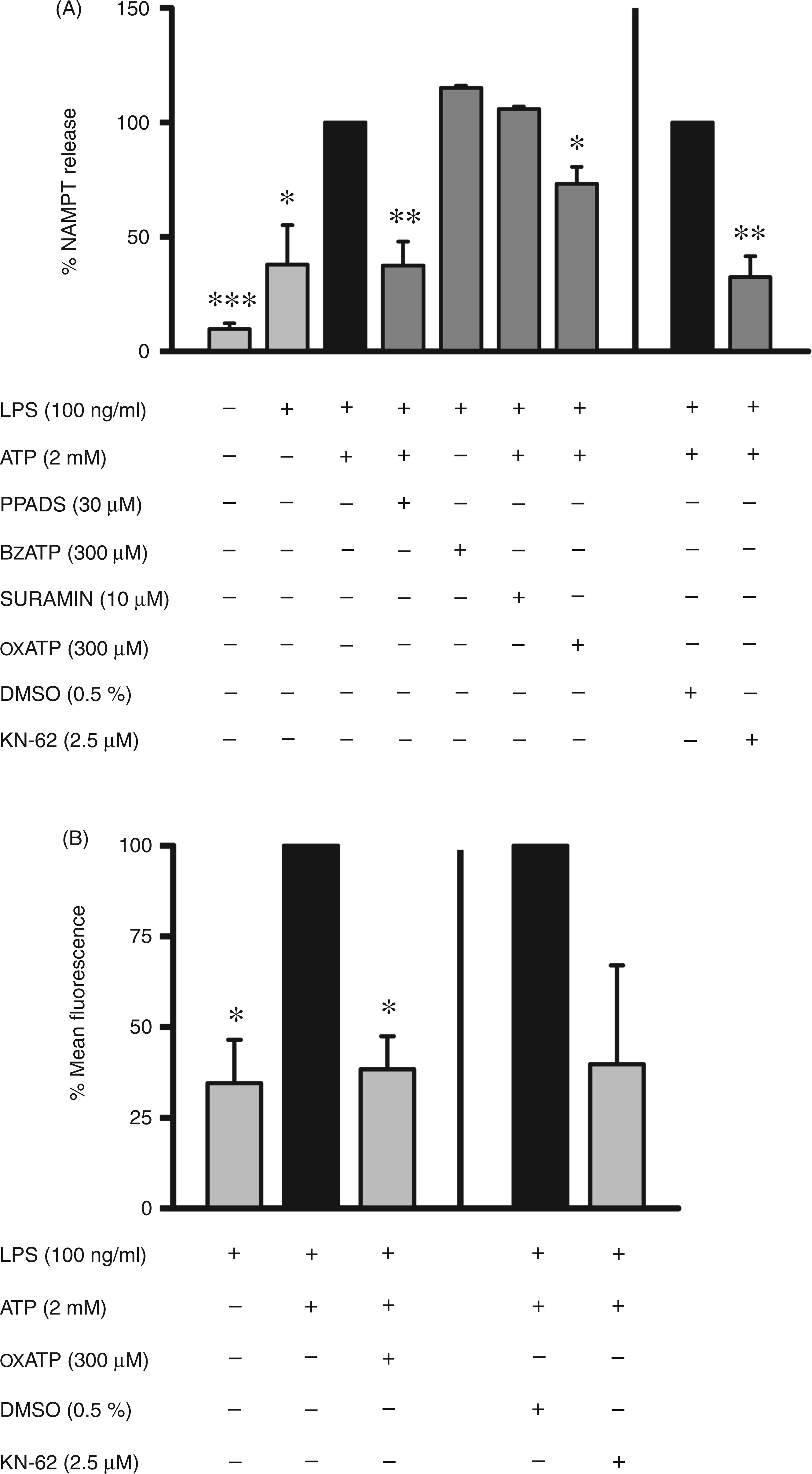
ATP-dependent pore formation
Considering that ATP, by activating the P2X7R, is able to trigger induction of a non-selective pore, 29 we measured pore formation after treatment of the activated cells with 2 mM ATP for 15 min. The pore is permeable to molecules with molecular masses up to 800 Da. 28 With its molecular mass of 340 Da, ethidium is normally excluded by intact plasma membranes. Upon entering cells ethidium fluorescence is induced by the intercalation of ethidium with RNA and DNA. Measuring ethidium bromide uptake by FACS-analysis we observed a fourfold increase in ethidium fluorescence in ATP-treated monocytes compared with cells incubated with LPS alone (Figure 2B). This finding was surprising as we have previously reported that resting monocytes lack this pore-forming response under physiological conditions. 30 Thus, the acquirement of pore formation during activation of monocytes seems to be of physiological relevance. In the presence of the P2X7R inhibitors oxATP and KN-62 the uptake of ethidium was diminished, indicating that the ATP-induced pore formation resulted from engagement of P2X7Rs (Figure 2B).
Activation of the P2X7 pore by ATP is well known to cause a massive K+-efflux leading to cytoplasmic K+ depletion. 31 For the secretion of mature IL-1β, this fall in cytoplasmic K+ concentration is thought to be essential, 32 as it leads to the activation of IL-1β-converting enzyme (ICE)/caspase-1, a protease that catalyzes the maturation of IL-1β.33,34
It is generally assumed that the pore itself is not directly involved in the transmembrane transfer of IL-1β, but that the pore-dependent depletion of K+ underlies IL-1β maturation and secretion. 35 It remains to be analyzed whether pore formation and K+-efflux have any impact on ATP-induced NAMPT release.
NAMPT release in the absence of extracellular ATP
Having shown that ATP is a secretory stimulus of NAMPT release, we next addressed the question of whether endogenous amounts of this nucleotide might be involved in the autocrine/paracrine stimulation of LPS-induced NAMPT secretion. ATP may be provided by activated monocytic cells, which, according to Ferrari et al., are capable of releasing ATP upon stimulation with LPS.
35
We incubated monocytes for 6 h with LPS in the presence and absence of oxATP and KN-62 (Figure 3A). During this time NAMPT concentrations in supernatants rose from 0.4 ± 0.4 ng/ml to 10.9 ± 4.7 ng/ml (means ± SD; n = 3). Although both agents are efficient P2X7R blockers only treatment with oxATP resulted in a pronounced decrease in NAMPT release, whereas KN-62 led to a minor reduction which failed to reach statistical significance.
Effect of P2X7R inhibitors, ATP-hydrolyzing enzymes and EGTA on NAMPT release by LPS-activated monocytes. Monocytes (2 × 106/ml) were incubated with LPS (100 ng/ml) in the presence or absence of (A) oxATP (300 µM), DMSO (0.5%), KN-62 (2.5 µM), EGTA (5 mM), or (B) hexokinase (10 U/ml), apyrase (2 U/ml). After 6 h NAMPT concentrations in the supernatants were determined by ELISA. Bars represent means ± SEM (n = 3); one-way ANOVA with Bonferroni post-hoc test calculated to the controls (black bars = 100%; values obtained in the presence of LPS alone or together with DMSO). *P ≤ 0.05, **P ≤ 0.01.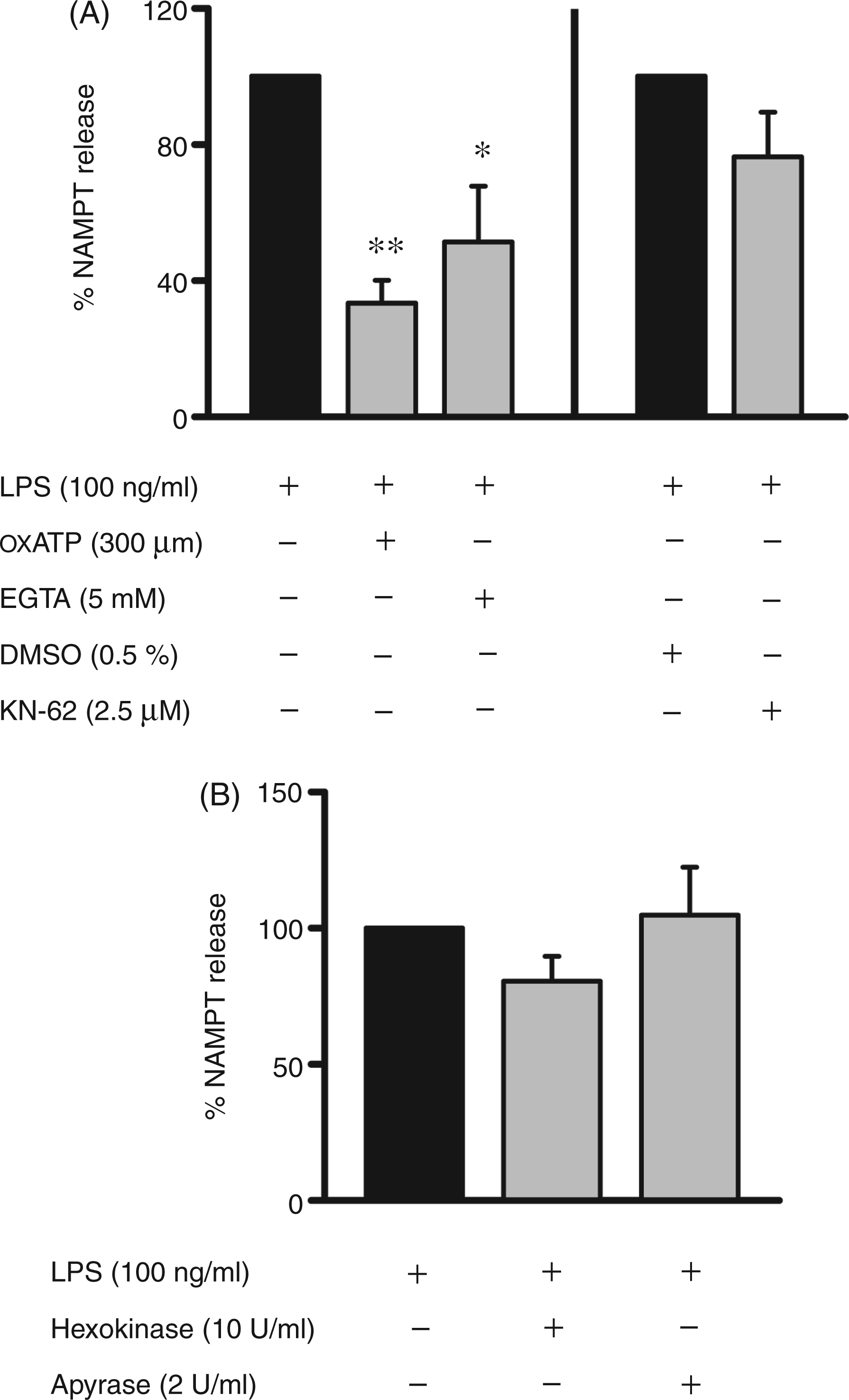
As discussed by Di Virgilio, oxATP may have other targets in addition to the P2X7R. It seems to be a valuable tool when used to antagonize ATP-dependent stimulation of this receptor, but less valuable when used to infer the participation of P2X7R in cellular responses caused by stimulation with non-nucleotide ligands. 36
Next, we incubated activated monocytes in the presence of ATP-consuming enzymes, such as apyrase and hexokinase. Apyrase hydrolyzes ATP and ADP to AMP, whereas hexokinase uses ATP to generate glucose-6-phosphate and ADP. Figure 3B illustrates that neither apyrase nor hexokinase were able to block NAMPT release to any significant extent. Considering that none of the pharmacologic modulators tested except for oxATP displayed any inhibitory effects, we conclude that LPS-induced NAMPT release is unlikely to be ATP-dependent. The ATP concentration required to stimulate NAMPT release may not be reached under the experimental conditions described here.
However, considering that intracellular ATP concentrations range from 5 to 10 mM, relatively high concentrations might be expected, especially at sites of inflammation where cell injury is a common event. Once released, ATP may then act as an extracellular mediator and enhance NAMPT secretion.
Interestingly, in the presence of EGTA (Figure 3A) NAMPT release was decreased, suggesting that the influx of extracellular Ca2+ is required for optimal NAMPT output.
Conclusion
Our studies, showing that activated monocytes require a second stimulus to efficiently secrete NAMPT, suggest that the process of stimulus-induced secretion may be of physiological relevance. On one hand this mode of regulation prevents NAMPT secretion in the absence of genuine need and, on the other hand, it ensures the rapid secretion of large amounts of NAMPT when required. With ATP being the second stimulus and NAMPT displaying cytokine like functions, ATP-induced secretion may be of importance at sites of inflammation where, as a result of cell damage or even cell death, high concentrations of this nucleotide can be achieved.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
We thank Nora Raulien and Dr Manuela Rossol (Department of Internal Medicine, Rheumatology, University of Leipzig) for performing the IL-1β assays.
