Abstract
Diphenyl diselenide (PhSe)2, a simple organoselenium compound, possesses interesting pharmacological properties that are under extensive research. As macrophages respond to microenvironmental stimuli and can display activities engaged in the initiation and the resolution of inflammation, in the present report we describe the ability of (PhSe)2 to modulate the macrophage activation. Our data indicate that (PhSe)2 could inhibit the NO production in a dose-dependent fashion in peritoneal macrophages activated by LPS or treated with vehicle alone. We could demonstrate that this effect correlated with a reduction in the expression of the inducible NO synthase in (PhSe)2-treated cells. Furthermore, (PhSe)2 suppressed the production of reactive oxygen species, diminished the activity of the arginase enzyme, and the accumulation of nitrotyrosine modified proteins in LPS-stimulated macrophages. This compound also diminished the antigen presentation capacity of classically activated macrophages, as it reduced MHCII and CD86 expression. In addition, (PhSe)2 modulated the alternative activation phenotype of macrophages. Dexamethasone-activated macrophages presented higher production of IL-10 and CD206, which were both down-regulated by the addition of (PhSe)2. These results suggest that (PhSe)2 possesses antioxidant and anti-inflammatory activities in classically-activated macrophages. We could demonstrate that (PhSe)2 can be also utilized to modulate the alternative activation phenotype of macrophages.
Introduction
Macrophages (Mph) represent a population of cells with marked phenotypic heterogeneity. They can be involved in both the initiation and the resolution of inflammation because of their ability to respond to signals they receive from the microenvironment. Mph have been categorized into subsets in analogy to the dichotomous Th1 and Th2 classification of T lymphocytes. Classically activated Mph (CaMph) or M1 Mph are generated following stimulation with microbial products and Th1 cytokines. Activation of macrophages to the M1 phenotype leads to enhanced microbicidal capacity and high secretion of pro-inflammatory cytokines (TNF-α, IL-1, IL-6, IL-12 and IL-23), thus polarizing a type I response. These cells are associated with augmented expression of MHCII and co-stimulatory molecules (CD86), and enhanced antigen-presenting capacity. 1 CaMph produce high levels of oxidative metabolites, NO and reactive oxygen species (ROS) to increase their killing activity. 2 CaMph have been implicated in several autoimmune pathologies, such as multiple sclerosis 3 , orchitis 4 and rheumatoid arthritis 5 , and in chronic inflammation-associated processes, such as obesity and atherosclerosis.6,7
Inflammatory processes triggered during infection are critical for pathogen removal. However, an excessive or prolonged inflammatory response is deleterious for the host, and must be stopped to allow healing. Mph also undergo alternative activation when stimulated with IL-4 or IL-13 (AaMph or M2 Mph), and represent a population involved in tolerance and the resolution of inflammation. These cells can be found in wound healing
8
, human term placenta
9
and in the lung.
10
AaMph are generated in pathologies that involve a Th2 profile, like parasitic infections, asthma and allergy.
11
These cells can function as modulators of the immune response given that IL-4 stimulates the production of anti-inflammatory cytokines (IL-10 and IL1-RA) while inhibiting the expression of pro-inflammatory cytokines. Several phenotypic markers have been identified for M2 Mph, such as Ym1 and Ym2, Found in Inflammatory Zone (FIZZ) 1 and Mph mannose receptor (CD206).
12
Th2-mediated induction of arginase I (Arg1) is a general feature of murine AaMph. This enzyme hydrolyzes
Despite the fact that the concept of AaMph was originally created to specifically distinguish the population of cells activated in the presence of IL-4 or IL-13, it was broadened to consider the phenotype of Mph activated by another set of stimuli that share properties involved in type II responses. To distinguish between the different, but overlapping, populations it has been proposed that the Mph activated by IL-4/IL-13 should be named M2a, M2b should be used to refer to Mph stimulated by immune complexes and TLR agonists and Mph treated with IL-10 or glucocorticoids should be named M2c. In particular, M2b Mph are characterized by high levels of production of IL-10 and low levels of IL-12. They better resemble the M1 population, as they produce pro-inflammatory cytokines, they do not induce Arg1 or FIZZ, and retain the ability to present antigens. 14 On the other hand, M2c Mph present a deactivated phenotype, they switch off the production of pro-inflammatory cytokines and reactive species (NO and ROS), down-regulate the expression of MHC class I and II expression and antigen processing. 15
Considering that the activation of pro-inflammatory situations is strictly related to ROS generation, and both circumstances are involved in several pathologies, increasing attention has focused on the development of new drugs with antioxidant/anti-inflammatory properties. In this way, several organoselenium compounds are proposed as promising antioxidant and anti-inflammatory agents. In this scenario, we have been studying the pharmacological properties of diphenyl diselenide (PhSe)2, a simple diaryl diselenide whose biological activities can be explained by the
Bearing in mind that ROS regulate redox-sensitive transcription factors which transcribe various inflammatory genes 22 , and that (PhSe)2 possesses antioxidant properties, it could be attractive to explore its anti-inflammatory properties. In this study, we aimed to explore the pharmacological properties of (PhSe)2 in cultured peritoneal Mph and how it can modulate the different activation pathways of these cells. Our results indicate that (PhSe)2 significantly inhibited the production of NO and ROS in activated Mph, as well as the immune content of iNOS and nitrotyrosine. Furthermore, (PhSe)2 treatment induced a reduction in the percentage of MHCII+ and CD86+ cells, and also diminished the activity of Arg1. Regarding the effects on M2c Mph, (PhSe)2 treatment inhibited the up-regulation of CD206 and the production of IL-10 induced by dexamethasone.
Materials and methods
Reagents
(PhSe)2 was synthesized according to published methods. Analysis of the 1H-NMR and
13
C-NMR spectra showed that the obtained compound presented analytical and spectroscopic data in full agreement with its assigned structure. The chemical purity of (PhSe)2 (99.9%) was determined by GLC/HPLC.
23
RPMI 1640 modified medium without phenol red, phorbol 12-myristate 13-acetate (PMA), LPS from
Animals
Albino six- to eight-week-old rats from a Wistar strain inbred in our laboratory for 40 years were used. All animal care and use was in accordance with the National Institutes of Health regulations and the Institutional Care and Use of Animals Committee (exp. No. 15-99-40426) approved animal handling and experimental procedures. Every effort was made to minimize both the number of animals used and their suffering.
Isolation of peritoneal Mph
Normal resident peritoneal cells were obtained by a peritoneal washing with 20 ml Dulbecco's PBS containing 2% FBS and 40 µg/ml gentamicin. After centrifugation at 350 ×
Quantification of nitrite in culture supernatants
Supernatants from Mph which had been cultured for 48 h with or without the indicated stimuli were analyzed. The concentration of nitrites was assayed in duplicate by a standard Griess reaction adapted to microplate as an indirect measurement of NO synthesis. The absorbance at 550 nm was obtained with a microplate reader model 680 (Bio-Rad Laboratories, Hercules, CA, USA). The data were referred to a standard curve of sodium nitrite. 25
Measurement of ROS production
Mph which had been cultured overnight (16–18 h) with or without different concentrations of (PhSe)2 were harvested and incubated simultaneously with 100 ng/ml PMA and the ROS probe DCF-DA (2 μM) for 1 h at 37 °C. Then, the cells were washed twice with PBS and the fluorescence intensity was analyzed by flow cytometry in a FACSCanto II (BD). 26 The fluorochrome DCF-DA was excited by the 488-nm laser and the fluorescent emission was obtained by the 530/30 filter of the flow cytometer.
iNOS and nitrotyrosine Western blotting
Peritoneal Mph which had been cultured for 24 h (for immunodetection of iNOS) or 48 h (for immunodetection of nitrotyrosine) with 5 μM (PhSe)2, 1 mM aminoguanidine and/or 0.5 µg/ml LPS were harvested and lyzed with 50 μl of 0.5% Triton X-100. The solution was then centrifuged at 4 °C at 10,000
Flow cytometry
Peritoneal Mph which had been cultured for 24 h with different stimuli were harvested and stained with the following reagents for surface activation markers: MHC-II, CD86 and CD206. Appropriate isotype controls were used. The cells were analyzed in a FACSCanto II.
Measurement of Arg1 activity
To analyze the Arg1 activity of cultured Mph, the cells were harvested and lyzed with 100 μl 0.2% Triton X-100. Then, 50 μl of the lysate was incubated with 50 μl of 10 nM MnCl2 and 50 μl 50 mM Tris-HCl (pH 7.5) for 10 min at 56 °C to induce the activation of the enzyme. Arginine hydrolysis was conducted by incubating the samples with 100 μl 0.5 M
Measurement of IL-10
Cytokine secretion was determined in supernatants collected from Mph which had been cultured for 48 h with the indicated stimuli. The concentration of IL-10 was measured using matching antibodies in a sandwich ELISA according to the indications of the manufacturer.
Statistical analyses
The results are presented as mean ± standard error of the mean (SEM). Comparisons between groups were performed by analysis of variance (ANOVA) followed by LSD Fisher test when appropriated.
Results
Effect of (PhSe)2 on cell viability and LPS toxicity
The effect of (PhSe)2 on the viability of Mph is presented in Figure 1. We evaluated the toxicity of this compound by means of the Trypan blue exclusion assay. A two-way ANOVA of the percentage of viable cells yielded a significant effect of the addition of both, LPS and (PhSe)2, and LPS × (PhSe)2 interaction. Effect of (PhSe)2 on the viability of macrophages. Peritoneal macrophages were cultured for 48 h and treated with increasing concentrations of (PhSe)2; concomitantly, the cells were activated or not with LPS. The cells were dyed with Trypan blue to determine their viability. Data indicate the percentage of viable cells and are presented as mean ± SEM of six independent experiments (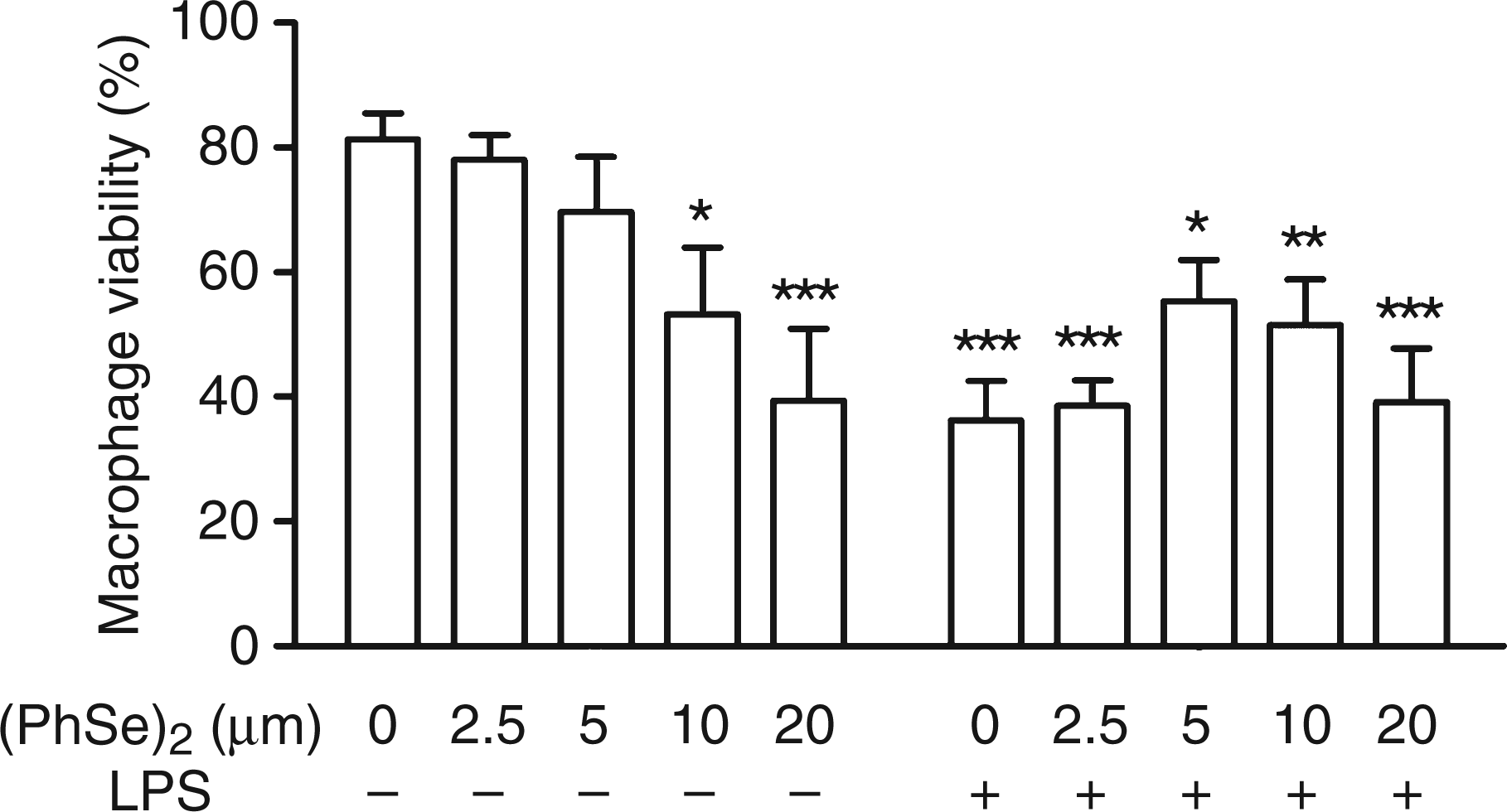
(PhSe)2 modulates NO production and iNOS expression
In order to analyze the anti-inflammatory properties of (PhSe)2, we assessed whether this compound can regulate the production of NO. Mph were cultured for 48 h and then the NO production in culture supernatants was determined by the Griess reaction. At basal state, the addition of (PhSe)2 induced a dose-dependent decrease in the nitrites production [F(1,16) = 5.22; (PhSe)2 modulation of NO production and iNOS expression. (A) Peritoneal macrophages were cultured for 48 h in the absence (A) or presence (B) of LPS and then treated with increasing concentrations of (PhSe)2 or 1 mM aminoguanidine (AG). The supernatants were collected and the nitrite concentration was determined by the Griess assay (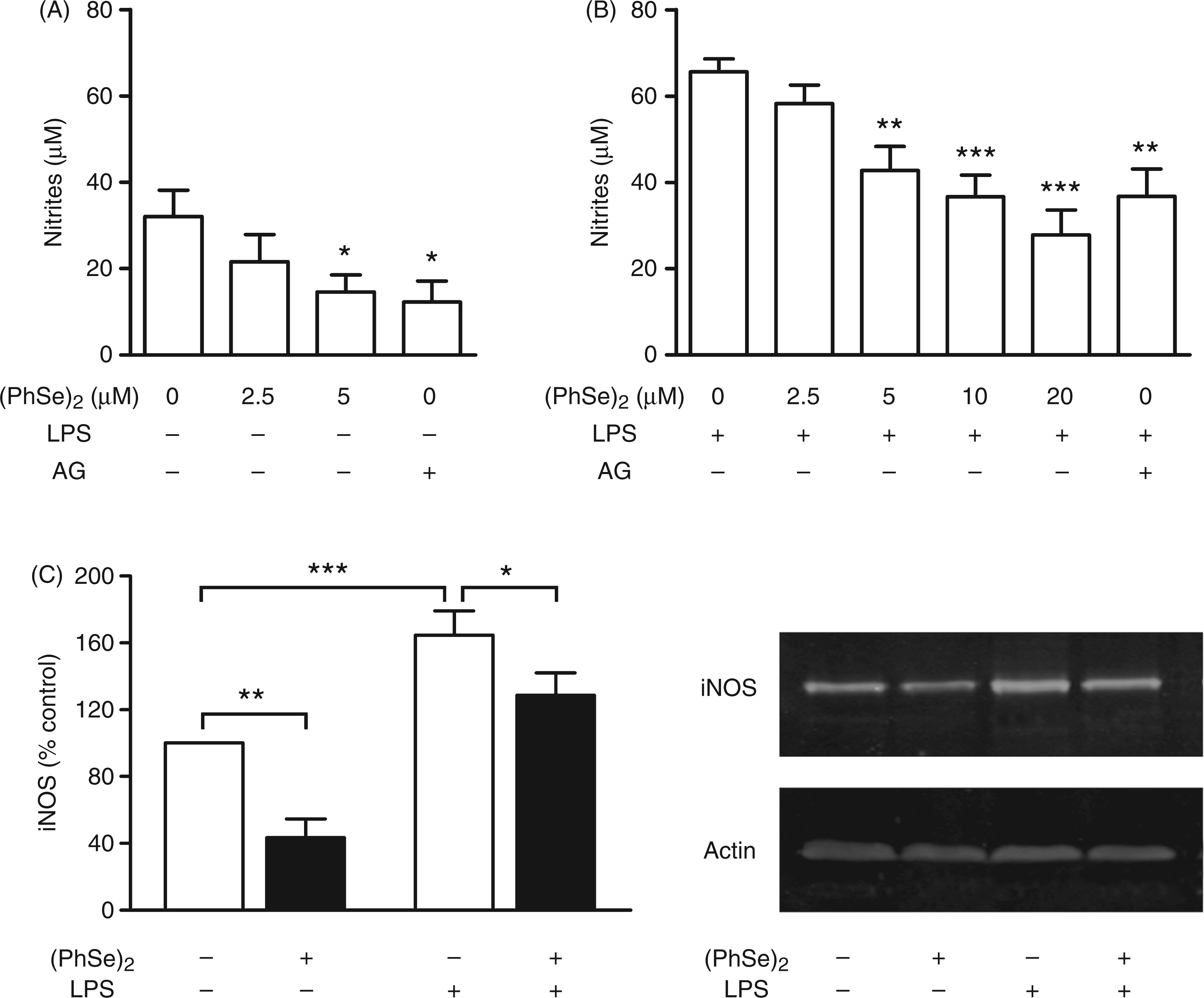
Western blot analyses were performed to determine whether the inhibitory effect of (PhSe)2 on NO production is related to its modulation of iNOS. A two way ANOVA of the iNOS/actin ratio revealed a significant effect of (PhSe)2 and LPS. In unstimulated Mph, we found that the addition of (PhSe)2 induced a statistically significant decrease in the amount of iNOS. Moreover, the expression of iNOS proteins was induced by LPS, and the treatment with (PhSe)2 inhibited this up-regulation (Figure 2C).
(PhSe)2 diminishes the ROS production
Next, we sought to further characterize the antioxidant properties of (PhSe)2 by evaluating the ROS production by means of the intracellular fluorescent probe DCF-DA. We stimulated the cells in the presence or absence of PMA to induce the production of ROS and increasing concentrations of (PhSe)2. Statistical analysis by two way ANOVA of the mean fluorescence intensity (MFI) revealed a significant effect of (PhSe)2, PMA and a PMA × (PhSe)2 interaction. We found that the addition of (PhSe)2 to Mph in the basal state diminished ROS production in a dose dependent manner [F(1,33) = 24.48; Effect of (PhSe)2 on the production of ROS in PMA-stimulated Mph. Peritoneal Mph were cultured for 24 h with different concentrations of (PhSe)2 and treated for 2 h with or without PMA. The production of ROS was assessed by incubating the cells with DCF-DA and analyzing them by flow cytometry. Data indicate the MFI and are presented as mean ± SEM of three independent experiments (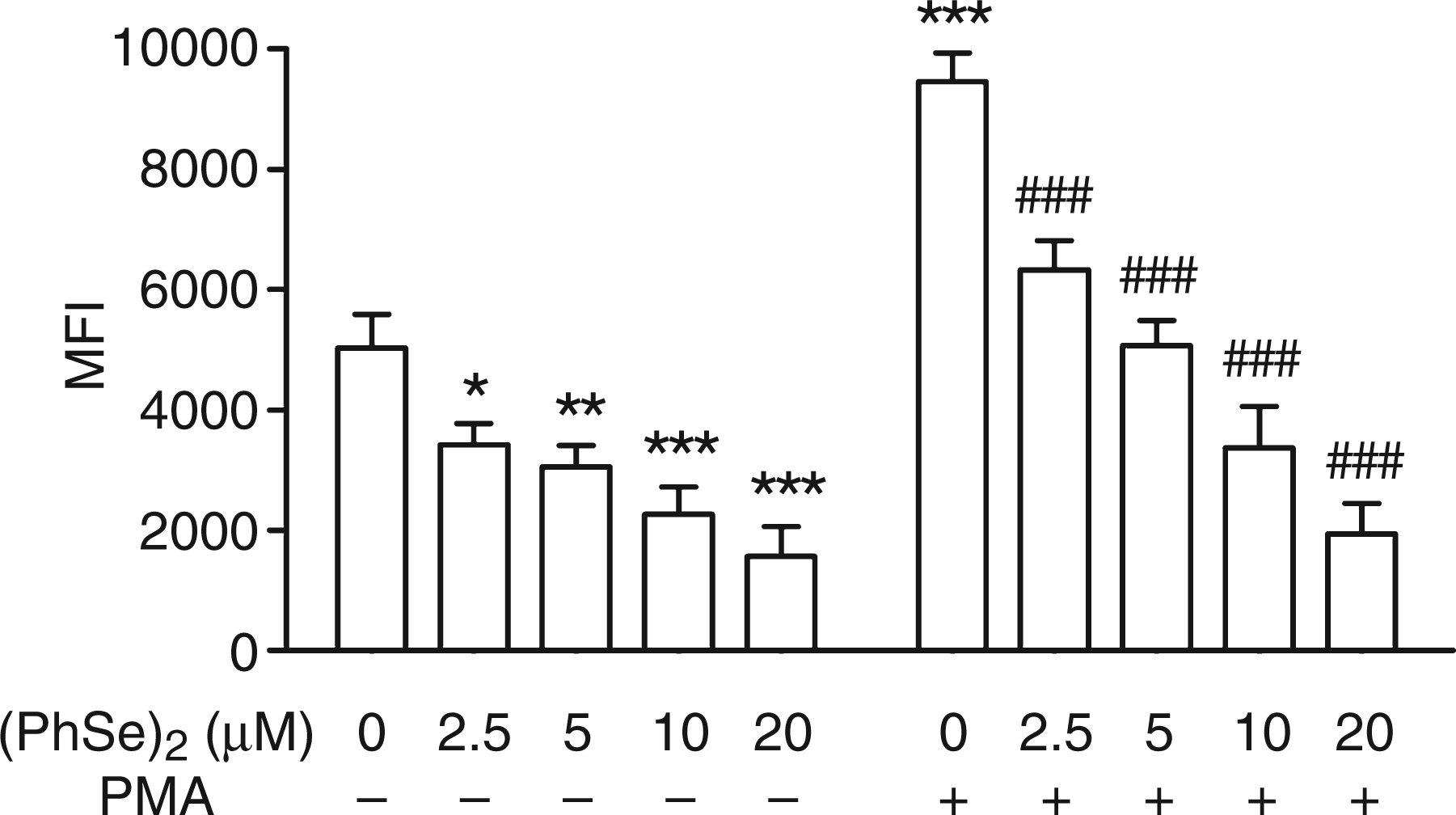
(PhSe)2 diminishes nitrotyrosine formation
Peroxynitrite (ONOO−.), the reaction product of NO and superoxide radicals, is a potent oxidant that has been implicated as a pathogenic mediator in a variety of disease conditions. Indeed, under appropriate stimulation conditions, immune cells, and, most notably Mph, can produce substantial amounts of peroxynitrite as a result of the iNOS− and NADPH oxidase-dependent formation of NO and O2–, respectively. Peroxynitrite promotes nitration (incorporation of a −NO2 group) of aromatic and aliphatic protein residues. Most notably, protein tyrosine residues constitute key targets for peroxynitrite-mediated nitration and the presence of 3-nitrotyrosine in proteins represents a usual modification introduced by the biological formation of peroxynitrite.
29
To study tyrosine nitration, peritoneal Mph were isolated and stimulated Effect of (PhSe)2 on the immunocontent of nitrotyrosine in peritoneal macrophages lysates. Mph were cultured for 48 h with or without 0.5 µg/ml LPS and 5 μM (PhSe)2. Homogenates of the cells were probed for nitrotyrosine by SDS-PAGE and immunoblotting using an Ab raised in rabbits against nitro-keyhole limpet hemocyanin (KLH). The intensity of all the bands that appeared on the nitrocellulose membrane were quantified to perform the statistical analysis. The cells were treated with vehicle (lane 1), (PhSe)2 (lane 2), LPS (lane 3), LPS and (PhSe)2 (lane 4), or LPS plus aminoguanidine (AG) (lane 5). The blot shown is representative of three independent experiments (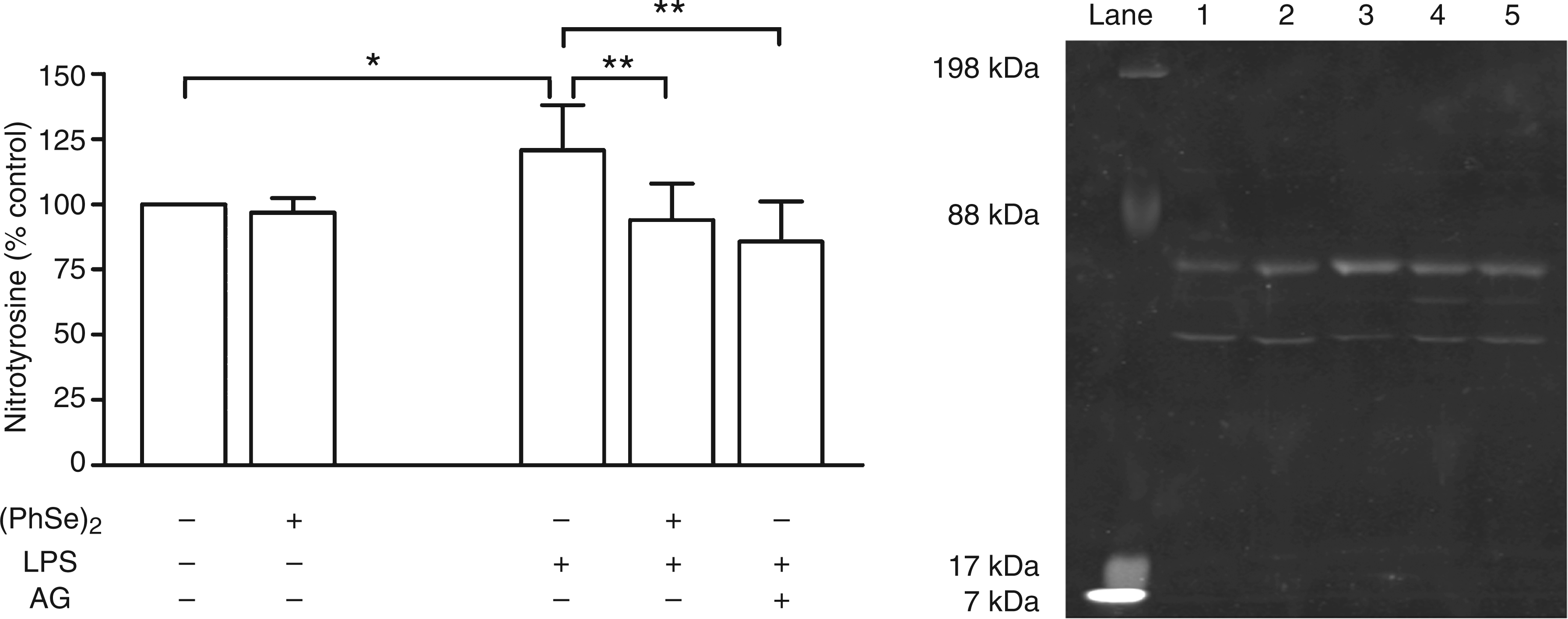
(PhSe)2 diminishes the expression of MHCII and costimulatory molecules
The effect of (PhSe)2 on the expression of co-stimulatory molecules and MHCII on the surface of Mph was examined by flow cytometry. To test whether expression of these molecules is modulated by (PhSe)2, the cells were treated with IFN-γ + LPS and the seleno-organic compound, and after about 18 h of culture, the cells were double labeled with anti-MHCII and -CD86 mAbs and analyzed by flow cytometry. According to a two way ANOVA, both (PhSe)2 and IFN-γ + LPS had a significant effect on the expression of CD86. Classical activation of Mph enhanced CD86 expression (130 ± 13% with respect to control,
A two-way ANOVA of the percentage of MHCII + cells revealed a significant effect of the addition of (PhSe)2 and IFN-γ + LPS. Treatment with IFN-γ + LPS augmented the percentage of MHCII+ Mph by 30% ( (PhSe)2 modulation of the expression of MHCII and CD86 in classically-activated Mph. Purified peritoneal Mph were treated 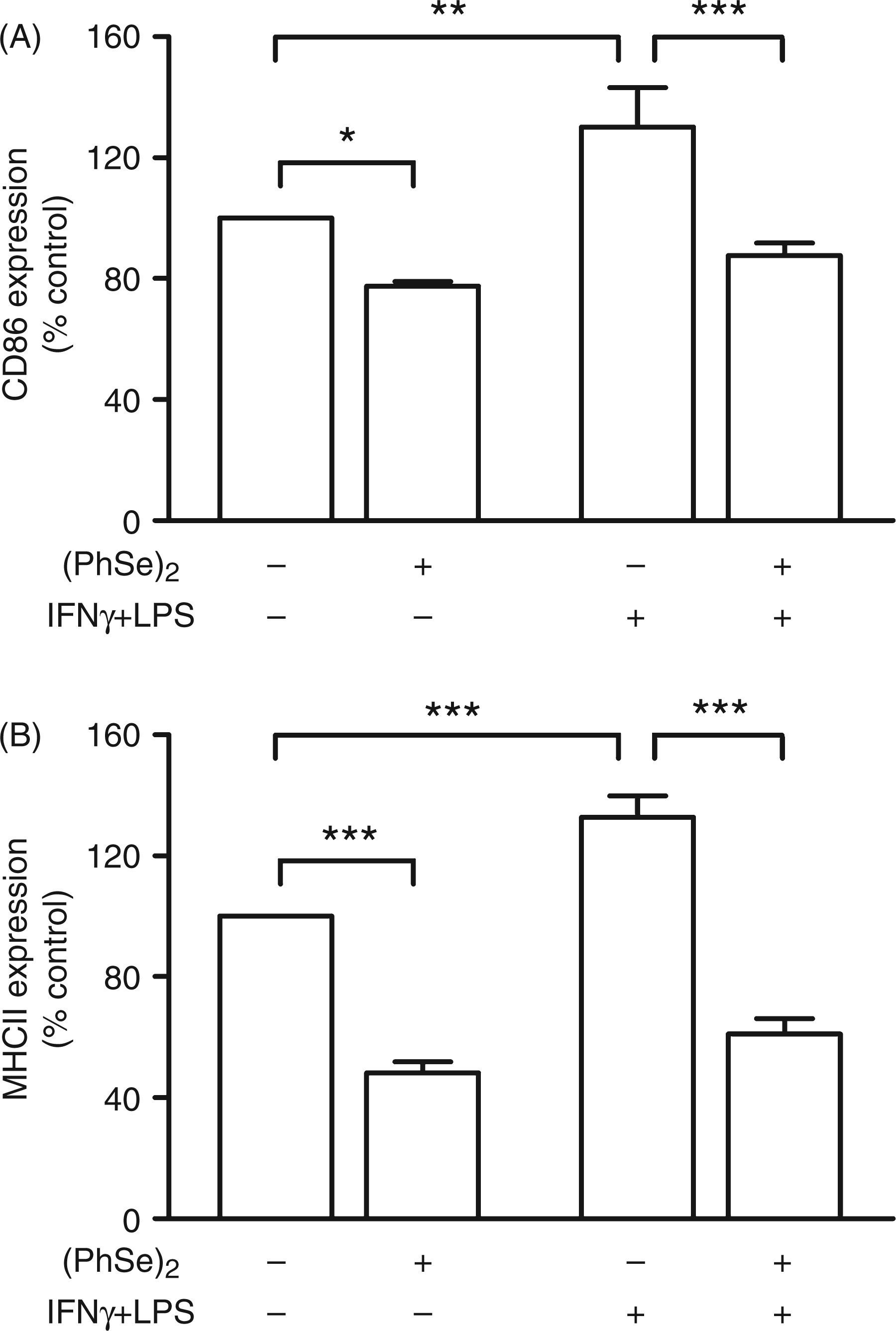
(PhSe)2 modulates the activity of Arg1
NO produced by Mph is toxic to host tissues and invading pathogens and its regulation is therefore essential to suppress host cytotoxicity. Mph Arg1 inhibits the production of NO by competing with NO synthases for arginine, the common substrate of NO synthases and arginases. Distinct mechanisms regulate Arg1 expression;
30
AaMph require the functions of both STAT6 and C/EBPβ, but are independent of MyD88. In contrast, expression of Arg1 induced by mycobacteria is independent of the STAT6 pathway but depends on C/EBPβ and MyD88. These data are consistent with studies documenting the induction of Arg1 expression by LPS. In order to analyze whether (PhSe)2 could modulate the activity of the Arg1 enzyme, Mph were cultured with this compound, then the cells were collected and Arg1 activity was determined in cell lysates by a colorimetric method. Statistical analysis by one way ANOVA of the Arg1 activity in cells cultured in the absence of LPS did not reveal a significant effect of the presence of (PhSe)2 (Figure 6A). On the other hand, in LPS-treated Mph, Arg1 activity revealed a significant concentration-dependent effect of the addition of (PhSe)2 (F(1,43) = 14.52; Effect of (PhSe)2 on the Arg1 activity in LPS treated Mph. Arg1 activity was measured in cell lysates from macrophages cultured in the absence (A) or presence (B) of LPS (0.5 µg/ml) for 48 h. Data indicate the concentration of urea normalized to protein content and are presented as mean ± SEM of six independent experiments (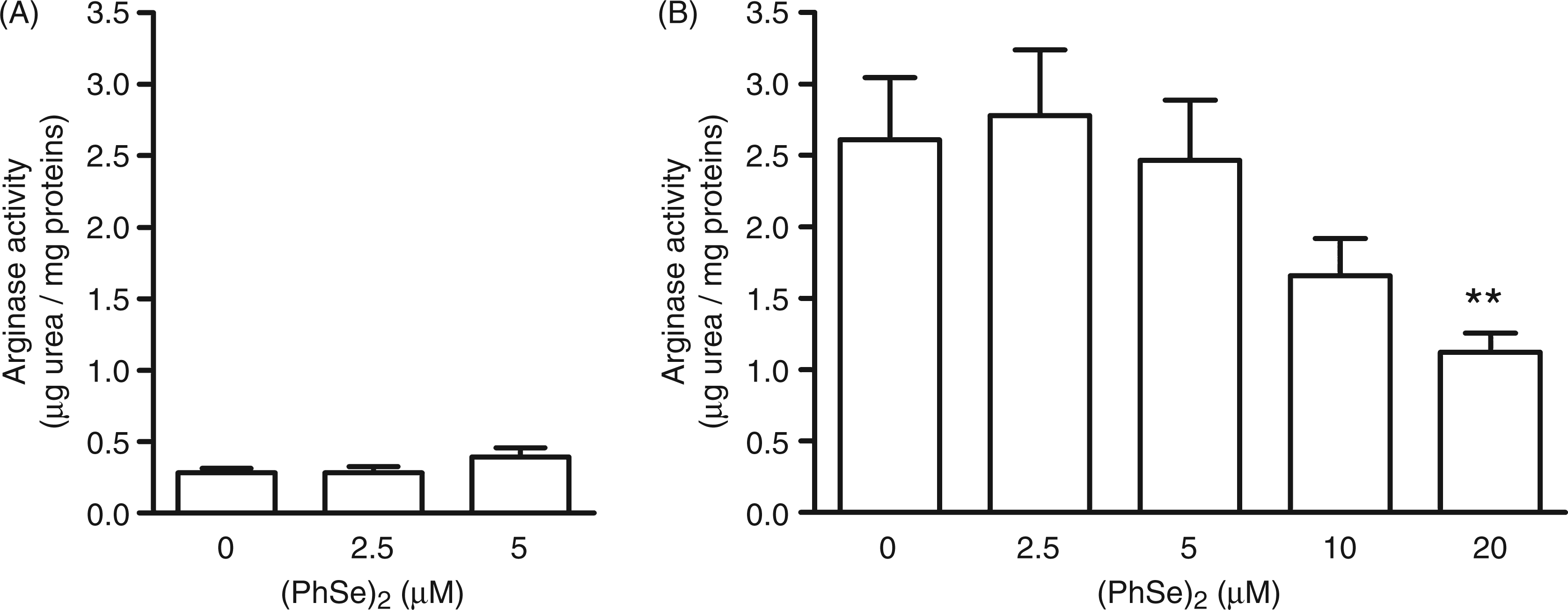
(PhSe)2 diminishes the expression of CD206 and the production of IL-10 in Mph activated by dexamethasone
Taking into account that (PhSe)2 presented several properties as an anti-inflammatory compound, we sought to evaluate if it could also modulate the alternative activation of Mph. For that purpose, peritoneal Mph were cultured with 1.5 μM dexamethasone concomitant to the addition of 5 μM (PhSe)2. The cells were collected and the surface expression of CD206, an alternative activation marker was assessed by flow cytometry. A two way ANOVA of the percentage of CD206+ Mph showed a significant effect of (PhSe)2 and dexamethasone. The expression of CD206 was significantly higher in dexamethasone-treated cells (136 ± 11% of the control, Effect of (PhSe)2 on the expression of alternative activation markers in dexamethasone-treated Mph. Peritoneal Mph were cultured with 1.5 μM dexamethasone to induce the alternative activation phenotype, and 5 μM (PhSe)2. (A) The cells were collected and the expression of CD206 was evaluated by flow cytometry. The expression of CD206 was determined by the percentage of CD206+ respect to control (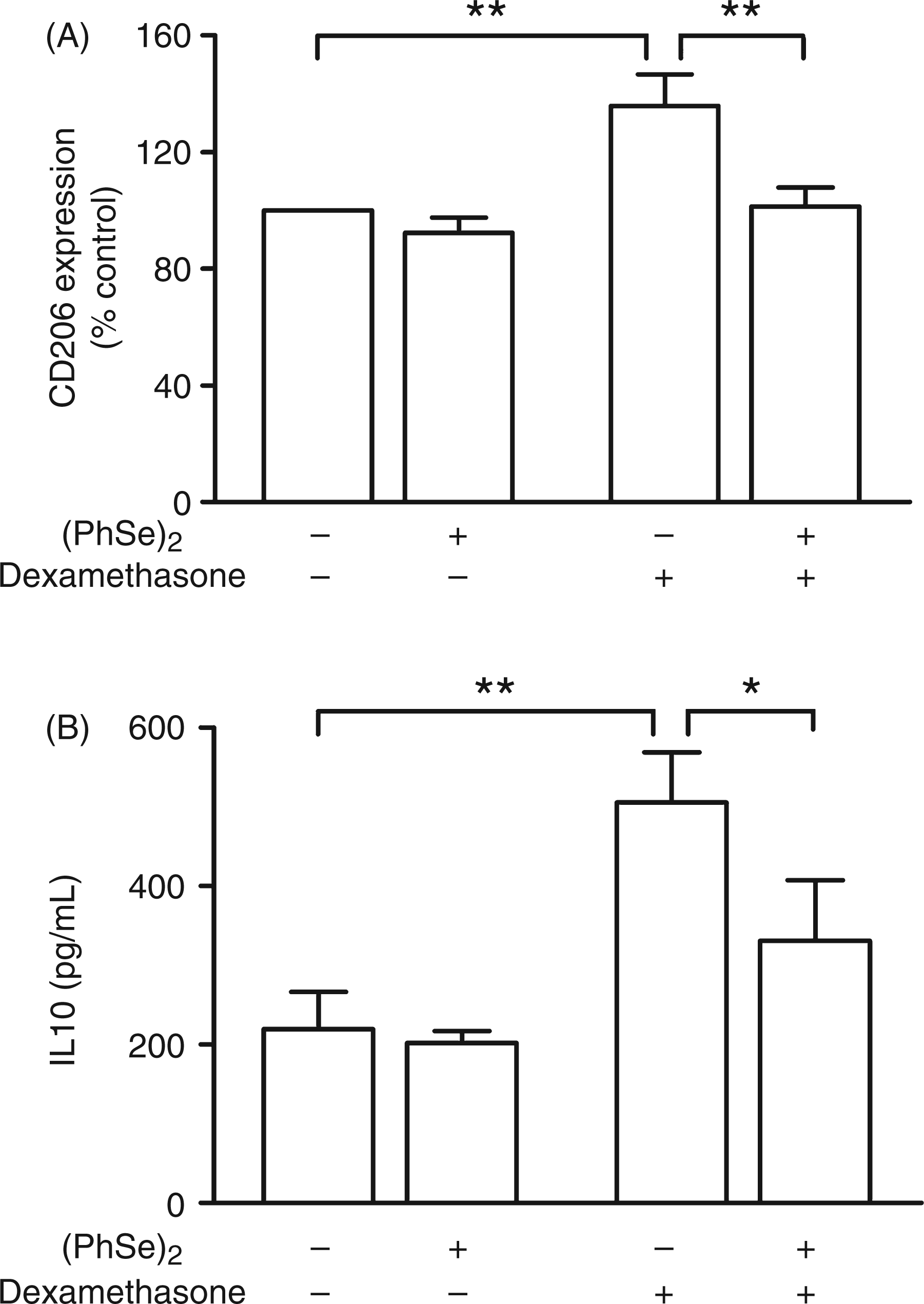
Discussion
The biological importance of selenium led to the development of pharmacologically active organoselenium compounds, among them (PhSe)2, whose biological activities have begun to be studied and it has become a good candidate for therapeutic purposes. 16 – 21 In the present study, we aimed at deepening the knowledge of the properties of (PhSe)2 in a model of Mph polarization. We found that (PhSe)2 was able to modulate Mph activation through the down-regulation of the release of pro-inflammatory mediators and the markers of alternative activation.
The role of ROS in inflammatory disorders has been the subject of intensive investigation. During the inflammatory process, activated immune cells increase the expression of cytokines, chemokines, ROS and NO, that amplify and perpetuate the inflammation. ROS and NO are known to directly affect macromolecules, producing a modification and possibly affecting their function. It means that ROS and NO can be responsible for the cellular damage that is present in inflammation. Nevertheless, ROS may play a role in enhancing inflammation through the activation of redox-sensitive transcription factors, such as the NF-κB and the activator protein-1. This resulted in an increased expression of a battery of distinct pro-inflammatory mediators.
22
Therefore, considerable effort has been deployed in the search for low-toxicity scavengers and inhibitors of ROS. To identify and characterize the properties of (PhSe)2 in cultured Mph, we analyzed the production of ROS in presence of an increasing concentration of this compound. We could demonstrate that (PhSe)2 efficiently inhibited ROS production, either via substrate for TrxR or because of the mimicking of the endogenous antioxidant enzyme, GPx.
16
–
18
In line with this, a recent study showed that
Antigen presentation is a crucial process during immunity for the generation of protective T-cell responses against pathogens or other foreign structures. The ‘professional’ antigen-presenting cells equipped to initiate a primary immune response by the presentation of antigen to naïve T cells are dendritic cells and Mph. After exposure to microbial products or inflammatory stimuli, these cells undergo an activation program that induces major phenotypic and functional modifications, especially affecting antigen capture, processing, and MHCII trafficking. Furthermore, maturation coincides with an increase in the expression of co-stimulatory signals required for efficient priming. In the present study, we found that the treatment with (PhSe)2 inhibited the surface expression of both, MHCII and CD86 molecules, and that this compound also prevented the augmentation induced by IFN-γ + LPS. These observations suggest that (PhSe)2 may serve as a compound used to prevent unwanted activation of T-cell responses. In accordance with our results, Matsue et al. 32 found that Ebselen at 10–20 µM efficiently inhibited LPS-induced CD86 up-regulation in bone marrow-derived dendritic cells, and this was accompanied by suppression in antigen-specific, dendritic cells-dependent proliferation by DO11.10 T cells in a dose-dependent manner. Furthermore, there are some reports that evaluate the role of selenium intake in antigen presentation. In this respect, it was found that low selenium intake could weaken the ability of recognizing and presenting OVA antigen by peritoneal macrophages of Wistar rats. 33
On the other hand, alternative activation of Mph encompasses a series of phenotypical changes, including low production of pro-inflammatory cytokines, ROS and NO, and abundant levels of non-opsonic receptors. Until recently, the discrimination between CaMph and AaMph in mice was mainly demonstrated at the biochemical level, in the metabolism of
Regarding the alternative activation, we evaluated the ability of (PhSe)2 to modulate the dexamethasone activation of these cells. We found that (PhSe)2 had no effect on the expression of CD206 and production of IL-10 of Mph. However, when the cells were treated with dexamethasone, (PhSe)2 was able to inhibit the up-regulation of both markers. In accordance with our results, it was described that selenomethionine and sodium selenite could inhibit UVB induction of IL-10 protein in murine keratinocytes. 37 These findings could be helpful in reinforcing the idea that selenium compounds could be used to prevent immunosupression triggered by UVB, thereby protecting against skin tumors. They also led us to speculate that there might be an unexplored redox-regulation of CD206 and IL-10 expression; however, further research needs to be carried out to clarify this matter.
Conclusions
These results indicate that (PhSe)2 possesses antioxidant activity and that it also functions as an anti-inflammatory compound in classically-activated macrophages and can also modulate the alternative activation phenotype of these cells.
Footnotes
Funding
This work was supported in part by grants from:
Conflict of interest
The authors declare that there is no conflict of interest.
